Submitted:
09 March 2026
Posted:
12 March 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Methods
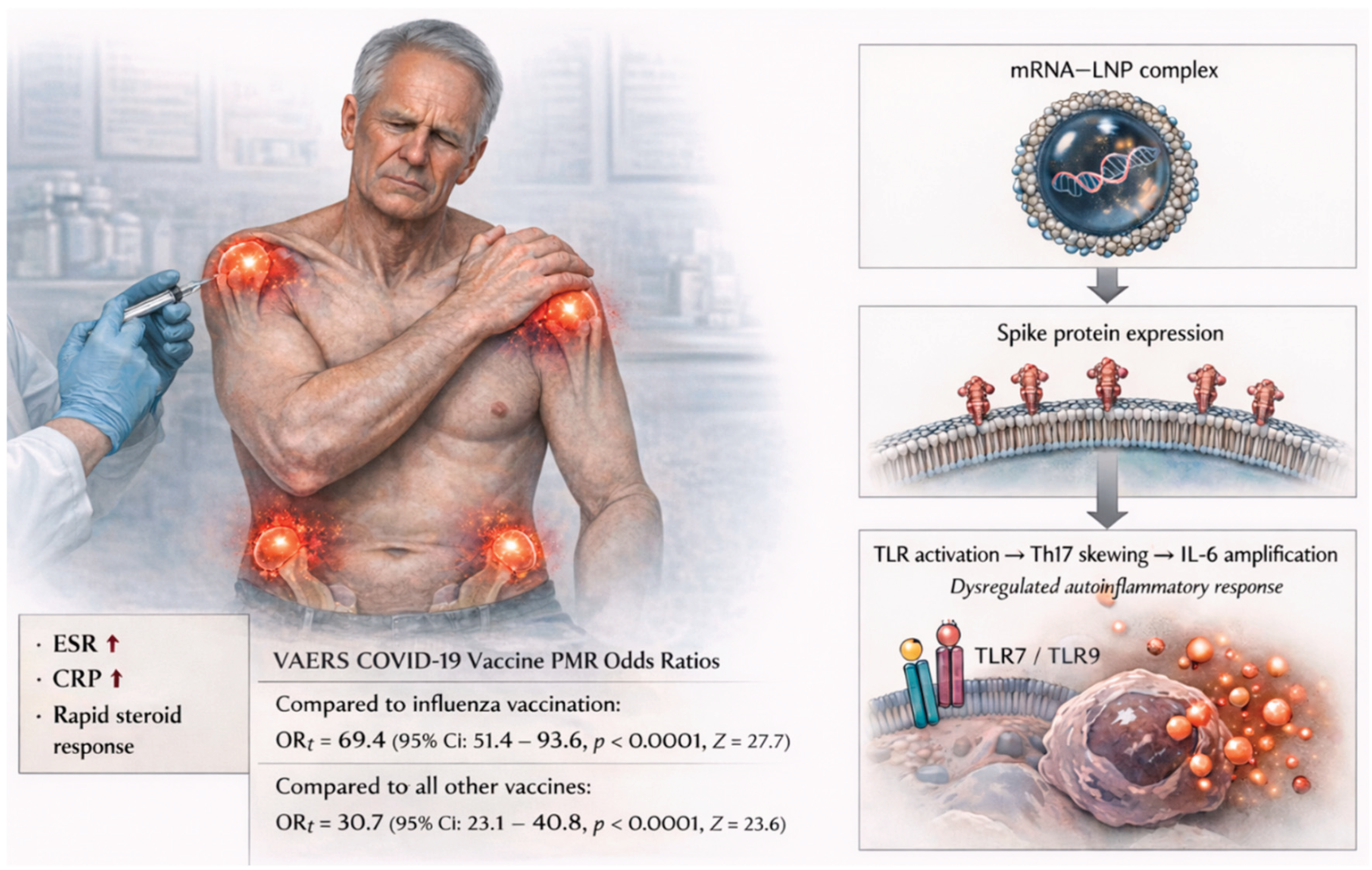
Results
- COVID-19 vaccination versus influenza vaccination: 69.4, 51.4–93.6, p < 0.0001, 27.7;
- COVID-19 vaccination versus all other vaccines combined: 30.7, 23.1–40.8, p < 0.0001, 23.6
Discussion
- Preexisting factors that predispose individuals to PMR following vaccination, such as the HLA-DRB1*13:01 allele, potentially triggered by vaccine adjuvants;
- Autoimmune syndromes induced by vaccine adjuvants; and
- Activation of Toll-like receptors, specifically TLR7 and TLR9, which may contribute to the development of PMR following immunization.
Conclusions
Funding
Conflicts of Interest
References
- Nunez-Castilla J, Stebliankin V, Baral P, et al. Potential Autoimmunity Resulting from Molecular Mimicry between SARS-CoV-2 Spike and Human Proteins. Viruses. 2022; 14(7):1415. [CrossRef]
- Mezache, L., Soltisz, A., Tili, E. et al. SARS-CoV-2 spike protein-induced inflammation underlies proarrhythmia in COVID-19. Sci Rep 15, 33991 (2025). [CrossRef]
- Syrmou, V., Liaskos, C., Ntavari, N. et al. COVID-19 vaccine-associated myositis: a comprehensive review of the literature driven by a case report. Immunol Res 71, 537–546 (2023). [CrossRef]
- https://pmc.ncbi.nlm.nih.gov/articles/PMC10018601/.
- Huynh TV, Rethi L, Lee T-W, Higa S, Kao Y-H, Chen Y-J. Spike Protein Impairs Mitochondrial Function in Human Cardiomyocytes: Mechanisms Underlying Cardiac Injury in COVID-19. Cells. 2023; 12(6):877. [CrossRef]
- Rurek M. Mitochondria in COVID-19: from cellular and molecular perspective. Front Physiol. 2024 Jun 21;15:1406635. doi: 10.3389/fphys.2024.1406635. PMID: 38974521; PMCID: PMC11224649. https://pmc.ncbi.nlm.nih.gov/articles/PMC11224649/. [CrossRef]
- Thorp KE, Thorp JA, Thorp EM. COVID-19 and the Unraveling of Experimental Medicine—Part III. G Med Sci. 2022; 3(1):118-158. https://www.doi.org/10.46766/thegms.pubheal.22042302.
- Acharya S, Musa R. Polymyalgia Rheumatica. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan. PMID: 30725959. https://pubmed.ncbi.nlm.nih.gov/30725959.
- Mahmood SB, Chohan S, Dasgupta B. Polymyalgia rheumatica: An updated review. Cleve Clin J Med. 2020;87(9):549-556. doi:10.3949/ccjm.87a.20008. PMID: 32868305. https://pubmed.ncbi.nlm.nih.gov/32868305. [CrossRef]
- Espígol-Frigolé G, Danda D, McAdoo SP, Luckie SL, Luqmani SM, Camellino D. Polymyalgia rheumatica. Lancet. 2023;402(10411):1459-1472. doi:10.1016/S0140-6736(23)01310-7. PMID: 37832573. https://pubmed.ncbi.nlm.nih.gov/37832573. [CrossRef]
- Choy EH, Unsworth DJ, Walsh DA, Dasgupta B, Filer A. Understanding the immunopathophysiology of polymyalgia rheumatica: implications for treatment. Ann Rheum Dis. 2026;85(1):6-21. doi:10.1016/j.ard.2025.09.005. PMID: 41107118. https://pubmed.ncbi.nlm.nih.gov/41107118. [CrossRef]
- Hysa E, Sobrero A, Camellino D, Rumi F, Cimmino MA, Cutolo M, Melsens K. Immune system activation in polymyalgia rheumatica: Which balance between autoinflammation and autoimmunity? A systematic review. Autoimmun Rev. 2022;21(2):102995. doi:10.1016/j.autrev.2021.102995. PMID: 34798314. https://pubmed.ncbi.nlm.nih.gov/34798314. [CrossRef]
- Florescu MM, Cinteză D, Sandru F, Gheorghe AG, Pana M, Mușetescu AE. Polymyalgia rheumatica: An update (Review). Exp Ther Med. 2023;26(6):543. doi:10.3892/etm.2023.12242. PMID: 37928511. https://pubmed.ncbi.nlm.nih.gov/37928511. [CrossRef]
- Benlidayi IC. Why is polymyalgia rheumatica a disease of older adults? Explanations through etiology and pathogenesis: a narrative review. Clin Rheumatol. 2024;43(3):851-861. doi:10.1007/s10067-023-06708-3. PMID: 37470883. https://pubmed.ncbi.nlm.nih.gov/37470883. [CrossRef]
- Shah S, Perera S, Deane KD, Dasgupta B. A history of polymyalgia rheumatica: a narrative review. Rheumatology (Oxford). 2025;64(2):470-476. doi:10.1093/rheumatology/keae569. PMID: 39412500. https://pubmed.ncbi.nlm.nih.gov/39412500. [CrossRef]
- Raleigh MF, Wilson JM, Hall RR, Nguyen G, Jump RL. Polymyalgia Rheumatica and Giant Cell Arteritis: Rapid Evidence Review. Am Fam Physician. 2022;106(4):420-426. PMID: 36260899. https://pubmed.ncbi.nlm.nih.gov/36260899.
- Nageswaran P, Unwin S, Adebajo A, Owen CE, Mackie SL, Deane KD, Dasgupta B. Review of phase 2/3 trials in polymyalgia rheumatica and giant cell arteritis. Expert Opin Emerg Drugs. 2024;29(1):5-17. doi:10.1080/14728214.2024.2303093. PMID: 38180809. https://pubmed.ncbi.nlm.nih.gov/38180809. [CrossRef]
- Buttgereit F, Dejaco C, Matteson EL, Dasgupta B. Polymyalgia Rheumatica and Giant Cell Arteritis: A Systematic Review. JAMA. 2016;315(22):2442-2458. doi:10.1001/jama.2016.5444. PMID: 27299619. https://pubmed.ncbi.nlm.nih.gov/27299619. [CrossRef]
- Castañeda S, García-Castañeda N, Prieto-Peña D, Martínez-López D, Gómez-Caverzaschi V, Atienza-Mateo B, Blanco R. Treatment of polymyalgia rheumatica. Biochem Pharmacol. 2019;165:221-229. doi:10.1016/j.bcp.2019.03.027. PMID: 30904473. https://pubmed.ncbi.nlm.nih.gov/30904473. [CrossRef]
- Hernández-Rodríguez J, Cid MC, López-Soto A, Espigol-Frigole G, Bosch X. Treatment of polymyalgia rheumatica: a systematic review. Arch Intern Med. 2009;169(20):1839-1850. doi:10.1001/archinternmed.2009.352. PMID: 19901135. https://pubmed.ncbi.nlm.nih.gov/19901135. [CrossRef]
- Harkins P, Cowley S, Conway R, Kane D, Burke E, Harrington R, Molloy D. Modern Management of Isolated Polymyalgia Rheumatica. Rheumatol Ther. 2025;12(6):1017-1041. doi:10.1007/s40744-025-00797-z. PMID: 41085890. https://pubmed.ncbi.nlm.nih.gov/41085890. [CrossRef]
- Boyadzhieva Z, Ruffer N, Bosch P, Buttgereit F, Dejaco C, Iagnocco A, Schäfer VS, Schmidt WA, Sattler L, Thiel J, Kleyer A, Schett G. Treatment and prognostic factors in PMR: a systematic literature review informing German, Austrian and Swiss guidelines. Rheumatology (Oxford). 2025;64(12):6036-6047. doi:10.1093/rheumatology/keaf418. PMID: 40795053. https://pubmed.ncbi.nlm.nih.gov/40795053. [CrossRef]
- MedAlerts. National Vaccine Information Center. Your Health. Your Family. Your Choice. https://medalerts.org/index.php. Accessed April 5, 2025.
- Thorp JA, Rogers C, Deskevich M, Tankersley S, Benavides A, Redshaw MD, McCullough PA. Covid-19 vaccines: the impact on pregnancy outcomes and menstrual function. J. Am. Phys.Surg. 2023;28(1):28-34. https://www.jpands.org/vol28no1/thorp.pdf.
- Our World in Data. Coronavirus (COVID-19) Vaccinations. https://ourworldindata.org/covid-vaccinations Published in 2020, Revised in 2024. Accessed February 22 2026.
- Norton EC, Dowd BE, Maciejewski ML. Odds Ratios-Cureent Best Practice and Use. JAMA Guide to Statistics and Methods. JAMA July 3, 2018:320(1).
- https://www.feinberg.northwestern.edu/sites/firstdailylife/docs/resources-docs/jama.2018.norton.guidetostatisticsandmedicine.odds-ratioscurrent-best-practice-and-use.pdf?utm_source=chatgpt.com.
- [26] Cash E, Boktor SW. Understanding Biostatistics Interpretation. National Library of Medicine StatPearls. March 13, 2023.
- https://www.ncbi.nlm.nih.gov/sites/books/NBK431056/?utm_source=chatgpt.com.
- CDC VAERS Team. Vaccine Adverse Event Reporting System (VAERS) Standard Operating Procedures for Covid-19. See Page 15. February 2, 2022. https://www.cdc.gov/vaccine-safety-systems/media/pdfs/vaers-covid19-sop-2-feb-2022-508.pdf?CDC_AAref_Val=https://www.cdc.gov/vaccinesafety/pdf/VAERS-COVID19-SOP-02-02-2022-508.pdf This has been accessed and previously referenced multiple times by the second author (JAT) over the past 3 years and published as such. However, the CDC removed this URL from their website in late 2024. Senator Ron Johnson, in a January 10, 2023, letter to CDC Director Rochelle Walensky, specifically points out his concerns for breach in safety signals in VAERS as measured by the disproportionality testing. In this letter Senator Ron Johnson also references this exact same URL in his letter (reference #2) https://www.ronjohnson.senate.gov/services/files/AB68101B-CDA4-49F1-8174-4274DDEB0120 The Wayback Machine archive to the removed URL is available here on November 15, 2024—click on that date. https://web.archive.org/web/20241115170847/https://www.cdc.gov/vaccine-safety-systems/media/pdfs/vaers-covid19-sop-2-feb-2022-508.pdf?CDC_AAref_Val=https://www.cdc.gov/vaccinesafety/pdf/VAERS-COVID19-SOP-02-02-2022-508.pdf.
- [28] Fleiss JS, Levin B, Myunghee CP. Statistical Methods for Rates and Proportions. Third Edition. New Your, NY:. John Wiley & Sons; 2003.
- Irani L, Linthicum M, Koleilat I. Polymyalgia rheumatica following COVID-19 vaccination: Case series of 3 patients and literature review on polymyalgia rheumatica induced by various vaccines. Medicine (Baltimore). 2024;103(44):e40218. doi:10.1097/MD.0000000000040218. https://journals.lww.com/md-journal/fulltext/2024/10250/polymyalgia_rheumatica_following_covid_19.42.aspx. [CrossRef]
- Shahid F, Shahid M, Ahmad M, et al. The Association of Polymyalgia Rheumatica and Giant Cell Arteritis With COVID-19 Vaccination: A Systematic Review. Clin Med Insights Arthritis Musculoskelet Disord. 2026;19:11795441251414673. doi:10.1177/11795441251414673. https://journals.sagepub.com/doi/10.1177/11795441251414673. [CrossRef]
- Trotto N, Tannenbaum C, Glicksman J. Polymyalgia Rheumatica After mRNA COVID-19 Vaccination. Ann Allergy Asthma Immunol. 2022;129(5):S12. doi:10.1016/j.anai.2022.08.546. https://www.annallergy.org/article/S1081-1206(22)01469-7/fulltext. [CrossRef]
- Yokote A, Fujioka S, Takahashi N, Mishima T, Tsuboi Y. Polymyalgia Rheumatica Following COVID-19 Vaccination. Intern Med. 2022;61(11):1777-1779. doi:10.2169/internalmedicine.8803-21. https://pmc.ncbi.nlm.nih.gov/articles/PMC9259323. [CrossRef]
- Mamootil D. New-Onset Polymyalgia Rheumatica Complicated by Giant Cell Arteritis Following COVID-19 Infection. Cureus. 2023 Jul 16;15(7):e41951. doi: 10.7759/cureus.41951. PMID: 37588300; PMCID: PMC10426384. [CrossRef]
- Bandinelli F. Post-COVID-19 and Post-COVID-19 Vaccine Arthritis, Polymyalgia Rheumatica and Horton’s Arteritis: A Single-Center Assessment of Clinical, Serological, Genetic, and Ultrasonographic Biomarkers. J Clin Med. 2023;12(24):7563. doi:10.3390/jcm12247563. PMID: 38137641. https://pubmed.ncbi.nlm.nih.gov/38137641. [CrossRef]
- Manzo C. Polymyalgia rheumatica and polymyalgia-like syndromes as adverse events following COVID-19 vaccines: working notes from a narrative review of published literature. Reumatologia. 2022;60(1):60-67. doi:10.5114/reum.2022.114244. PMID: 35645422. https://pubmed.ncbi.nlm.nih.gov/35645422. [CrossRef]
- Pinto Oliveira C, Ferreira Azevedo S, Vilafanha C, Prata AR, Barcelos A. Polymyalgia Rheumatica After COVID-19 Vaccination: Data from the EudraVigilance Database. Acta Med Port. 2024 May 2;37(5):396-397. doi: 10.20344/amp.20952. Epub 2024 Apr 12. PMID: 38607657. [CrossRef]
- Jarrot PA, Mirouse A, Ottaviani S, et al. Polymyalgia rheumatica and giant cell arteritis following COVID-19 vaccination: Results from a nationwide survey. Hum Vaccin Immunother. 2024 Dec 31;20(1):2334084. doi: 10.1080/21645515.2024.2334084. Epub 2024 Apr 2. PMID: 38563792; PMCID: PMC10989707. [CrossRef]
- Ottaviani S, Juge PA, Forien M, Ebstein E, Palazzo E, Dieudé P. Polymyalgia rheumatica following COVID-19 vaccination: A case-series of ten patients. Joint Bone Spine. 2022 Mar;89(2):105334. doi: 10.1016/j.jbspin.2021.105334. Epub 2021 Dec 22. PMID: 34954076; PMCID: PMC8694785. [CrossRef]
- Irani L, Bou Karroum M, Chehab Y, et al. Polymyalgia rheumatica following COVID-19 vaccination: Case series of 3 patients and literature review on polymyalgia rheumatica induced by various vaccines. Medicine (Baltimore). 2024 Oct 25;103(43):e40204. doi: 10.1097/MD.0000000000040204. PMID: 39470514; PMCID: PMC11521047. [CrossRef]
- Furr T, Garg M. Rare Cases of Polymyalgia Rheumatica After Receiving COVID-19 Vaccinations. Cureus. 2023 Apr 18;15(4):e37782. doi: 10.7759/cureus.37782. PMID: 37091488; PMCID: PMC10115741. [CrossRef]
- Mamootil D. New-Onset Polymyalgia Rheumatica Complicated by Giant Cell Arteritis Following COVID-19 Infection. Cureus. 2023 Jul 16;15(7):e41951. doi: 10.7759/cureus.41951. PMID: 37588300; PMCID: PMC10426384. [CrossRef]
- Arias M, Cacabelos P, Arias-Rivas S. Polymyalgia rheumatica and giant cell arteritis with intracranial involvement postvaccination anti-COVID-19. Med Clin (Engl Ed). 2022 Sep 23;159(6):e43-e44. doi: 10.1016/j.medcle.2022.04.011. Epub 2022 Aug 30. PMID: 36060099; PMCID: PMC9425118. [CrossRef]
- Bandinelli F, Pagano M, Vallecoccia MS. Post-COVID-19 and Post-COVID-19 Vaccine Arthritis, Polymyalgia Rheumatica and Horton’s Arteritis: A Single-Center Assessment of Clinical, Serological, Genetic, and Ultrasonographic Biomarkers. J Clin Med. 2023 Dec 8;12(24):7563. doi: 10.3390/jcm12247563. PMID: 38137631; PMCID: PMC10743539. [CrossRef]
- Osada A, Sakuragi C, Toya C, Mitsuo A. New-onset Polymyalgia Rheumatica Following the Administration of the Pfizer-BioNTech COVID-19 Vaccine. Intern Med. 2022 Mar 1;61(5):749-753. doi: 10.2169/internalmedicine.8651-21. Epub 2021 Dec 11. PMID: 34897152; PMCID: PMC8943385. [CrossRef]
- Manzo C, Castagna A, Nune A, Isetta M. Polymyalgia rheumatica and polymyalgia-like syndromes as adverse events following immunisation with COVID-19 vaccines: a 15 months update. Reumatologia. 2023;61(5):408-409. doi: 10.5114/reum/172508. Epub 2023 Oct 31. PMID: 37970116; PMCID: PMC10634411. [CrossRef]
- Shahid F, Farooq H, Abeer H, et al. The Association of Polymyalgia Rheumatica and Giant Cell Arteritis With COVID-19 Vaccination: A Systematic Review. Clin Med Insights Arthritis Musculoskelet Disord. 2026 Jan 19;19:11795441251414673. doi: 10.1177/11795441251414673. PMID: 41567360; PMCID: PMC12816559. [CrossRef]
- Haruna K, Shirota S, Nishioka H. Polymyalgia Rheumatica (PMR) Lacking Shoulder Pain Following COVID-19 Vaccination. Cureus. 2023 Feb 7;15(2):e34714. doi: 10.7759/cureus.34714. PMID: 36909027; PMCID: PMC9996542. [CrossRef]
- Izuka S, Komai T, Natsumoto B, Shoda H, Fujio K. Self-limited Polymyalgia Rheumatica-like Syndrome Following mRNA-1273 SARS-CoV-2 Vaccination. Intern Med. 2022 Mar 15;61(6):903-906. doi: 10.2169/internalmedicine.8829-21. Epub 2021 Dec 28. PMID: 34980802; PMCID: PMC8987264. [CrossRef]
- Nielsen AW, Hansen IT, Gormsen LC, Hauge EM, Keller KK. Self-limiting reactive disease mimicking polymyalgia rheumatica following Moderna COVID-19 vaccine. Scand J Rheumatol. 2022 Sep;51(5):411-413. doi: 10.1080/03009742.2022.2070959. Epub 2022 Jun 6. PMID: 35658808. [CrossRef]
- Ahmad A, Baker DL. An Unusual Side Effect of the COVID-19 Vaccine: A Possible Trigger of Polymyalgia Rheumatica. Cureus. 2022 Jul 6;14(7):e26617. doi: 10.7759/cureus.26617. PMID: 35949778; PMCID: PMC9356652. [CrossRef]
- Matsuzono K, Fujimoto S. Case report of transient polymyalgia rheumatica after receiving the first dose of the BNT162b2 mRNA COVID-19 vaccine. SAGE Open Med Case Rep. 2024 Mar 4;12:2050313 × 241235830. doi: 10.1177/2050313 × 241235830. PMID: 38444691; PMCID: PMC10913505.
- Lourenço C, Pascoal A, Paiva A, Campos I, Pagaimo J. Polymyalgia Rheumatica After ChAdOx1 nCov-19 Vaccine: A Case Report. Cureus. 2022 May 26;14(5):e25346. doi: 10.7759/cureus.25346. PMID: 35774673; PMCID: PMC9236670. [CrossRef]
- amada S, Yamada K, Nishida H. A Case of Sequential Development of Polymyalgia Rheumatica and Guillain-Barré Syndrome Following Administration of the Pfizer-BioNTech COVID-19 Vaccine. Intern Med. 2022 Oct 1;61(19):2995. doi: 10.2169/internalmedicine.0319-22. Epub 2022 Jul 22. PMID: 35871588; PMCID: PMC9593167. [CrossRef]
- Nahra V, Makandura M, Anthony DD, Mattar M. A Case Series on the COVID-19 Vaccines and Possible Immune-Related Adverse Events: A New Challenge for the Rheumatologists. Cureus. 2022 Sep 27;14(9):e29660. doi: 10.7759/cureus.29660. PMID: 36321010; PMCID: PMC9612893. [CrossRef]
- Hill AB. The environment and disease: association or causation? Proc R Soc Med. 196;58(5):295-300. PMID: 14283879.
- Hulscher N, Hodkinson B, Makris M, et al. Clinical Approach to Post-acute Sequelae After COVID-19 Infection and Vaccination. Cureus. 2023;15(11):e49204. doi:10.7759/cureus.49204. https://pmc.ncbi.nlm.nih.gov/articles/PMC10663976. [CrossRef]
- Lesgards JF, Cerdan D, Perronne C, et al. Toxicity of SARS-CoV-2 Spike Protein from the Virus and Produced from COVID-19 mRNA or Adenoviral DNA Vaccines. J Vaccines Vaccin. 2023;14(4):1000517. https://www.fortunejournals.com/articles/toxicity-of-sarscov2-spike-protein-from-the-virus-and-produced-from-covid19-mrna-or-adenoviral-dna-vaccines.html.
- Peters SE, Newman J, Ray H, Thorp JA, Parotto T, Hooker B, McDyer D, Murphy L, Stricker RB, McDonnell M, Mills PJ, Geick W, Northrup C. Menstrual Abnormalities Strongly Associated with Proximity to COVID-19 Vaccinated Individuals. IJVTPR. International Journal of Vaccine Theory, Practice, and Research. December 7, 2024. 3(2)1435-1461. https://www.ijvtpr.com/index.php/IJVTPR/article/view/113 https://www.ijvtpr.com/index.php/IJVTPR/article/view/113/365.
- Mihalcea AM. Further proof of shedding: detection of Pfizer BioNTech messenger RNA COVID-19 Vaccine in Human Blood, Placenta and semen: detects mRNA in half of unvaccinated women>. December 6, 2025. https://anamihalceamdphd.substack.com/p/further-proof-of-shedding-detection?r=45fey1&triedRedirect=true.
- Mordechay, L., Baum, G., Gabbay-Benziv, R., Weinberger, H., & Frenkel-Morgenstern, M. F. (2025). Detection of Pfizer-BioNTech Messenger RNA COVID-19 Vaccine in Human Blood, Placenta and Semen. Annals of Case Reports, 10, Article 2428. DOI: 10.29011/2574-7754.102428. [CrossRef]
- Wolf N. “Shedding” is real, DNA Harms are real. April 4, 2024. https://naomiwolf.substack.com/p/shedding-is-real-dna-harms-are-real?r=45fey1&triedRedirect=true.
- Kedl RM, Hsieh EWY, Morrison TE, et al. Evidence for Aerosol Transfer of SARS-CoV-2-Specific Humoral Immunity. Immunohorizons. 2023 May 1;7(5):307-309. doi: 10.4049/immunohorizons.2300027. PMID: 37159005; PMCID: PMC10579981. [CrossRef]
- Kory P. Shedding Part 6—Clinical case notes describing shedding phenomena at the leading edge clinic. Nov 1, 2023. https://pierrekorymedicalmusings.com/p/shedding-part-6-clinical-case-notes.
- Bartmann C, Schmidt V, Mörz M, et al. Detection of spike protein in term placentas of COVID-19 vaccinated and/or SARS-CoV-2 infected women. PLoS One. 2026 Mar 5;21(3):e0344185. doi: 10.1371/journal.pone.0344185. PMID: 41785286; PMCID: PMC12962466. https://pmc.ncbi.nlm.nih.gov/articles/PMC12962466/?utm_source=chatgpt.com. [CrossRef]
- Espígol-Frigolé G, Danda D, McAdoo SP, et al. Polymyalgia rheumatica. Lancet. 2023;402(10411):1459-1472. doi:10.1016/S0140-6736(23)01310-7. https://pubmed.ncbi.nlm.nih.gov/37832573. [CrossRef]
- Parry PI, Lefringhausen A, Turni C, et al. ‘Spikeopathy’: COVID-19 Spike Protein Is Pathogenic, from Both Virus and Vaccine mRNA. Biomedicines. 2023;11(8):2287. doi:10.3390/biomedicines11082287. https://pmc.ncbi.nlm.nih.gov/articles/PMC10452662. [CrossRef]
- McCullough PA. Painful Osteoporotic Microfractures in Younger Persons After COVID-19 Vaccination” by Peter A. McCullough, MD, MPH, on Focal Points/Substack, December 31, 2025. https://www.thefocalpoints.com/p/painful-osteoporotic-microfractures?utm_source=chatgpt.com.
- Ursini F, Ruscitti P, Raimondo V, et al. Spectrum of short-term inflammatory musculoskeletal manifestations after COVID-19 vaccine administration: a report of 66 cases. Ann Rheum Dis. 2022 Mar;81(3):440-441. doi: 10.1136/annrheumdis-2021-221587. Epub 2021 Nov 26. PMID: 34836886. [CrossRef]
- Sharma A, Parekh SG. Musculoskeletal sequelae following COVID-19 mRNA vaccination: a case report. J Orthop Rep. 2022;1(3):100044. doi:10.1016/j.jorep.2022.100044. https://doaj.org/article/927b9189708d46d8a995c613adfc4ff9?utm_. [CrossRef]
- Park YH, Kim MH, Choi MG, Chun EM. Correlation between COVID-19 vaccination and inflammatory musculoskeletal disorders. medRxiv preprint November 22, 2023. doi:https://doi.org/10.11012023.11.14.23298544. https://www.medrxiv.org/content/10.1101/2023.11.14.23298544v1.full.pdf?utm_. [CrossRef]
- Kawakami E, Blank A, Smith B. Sport and the coronavirus crisis special issue: An introduction. Int J Sport Commun. 2020;13(3):1-4. doi:10.1123/ijsc.2020-0234. [CrossRef]
- Peter Cowan @living_energy on X formerly Twitter: “David White, interim director of the NFL Player Association, stated: “The NFLPA considers EMFs to be a potential WORKPLACE SAFETY issue”. This was reported on The Mercury News. https://x.com/living_energy/status/2028546275772448889?s=46&t=E3lBa0k6ScUvJ7YwdVqK9w.
- Knoblauch E. For some 49ers, EMF injuries theory no joke: ‘I might pull up with a meter one day’. San Francisco Chronicle. 2026 Jan 23. https://www.sfchronicle.com/sports/49ers/article/49ers-emf-injury-theory-no-joke-might-pull-21311524.php.
- Wang F. NFL leaders monitoring viral EMF conspiracy theory about 49ers injuries that doctors have debunked. ABC7 News. 2026 Feb 4. https://abc7news.com/post/nfl-leaders-monitoring-viral-emf-conspiracy-theory-49ers-injuries-doctors-have-debunked/18541925.
- McCullough PA, Wynn C, Procter BC. Clinical Rationale for SARS-CoV-2 Base Spike Protein Detoxification in Post COVID-19 and Vaccine Injury Syndromes. J Am Phys Surg. 2023;28(3):80-84. https://jpands.org/vol28no3/mccullough.pdf.
- Lazarus R. Eletronic Support for Public Health—Vaccine Adverse Event Reporting System (ES::VAERS)—Final Report. Prepared by Harvard Pilgrim Health Care, Inc. under Grant No. R18 HS017045. Rockville, MD: Agency for Healthcare research and Quality (AHRQ); 2010. https://digital.ahrq.gov/ahrq-funded-projects/electronic-support-public-health-vaccine-adverse-event-reporting-system?utm_.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




