Submitted:
08 March 2026
Posted:
10 March 2026
You are already at the latest version
Abstract
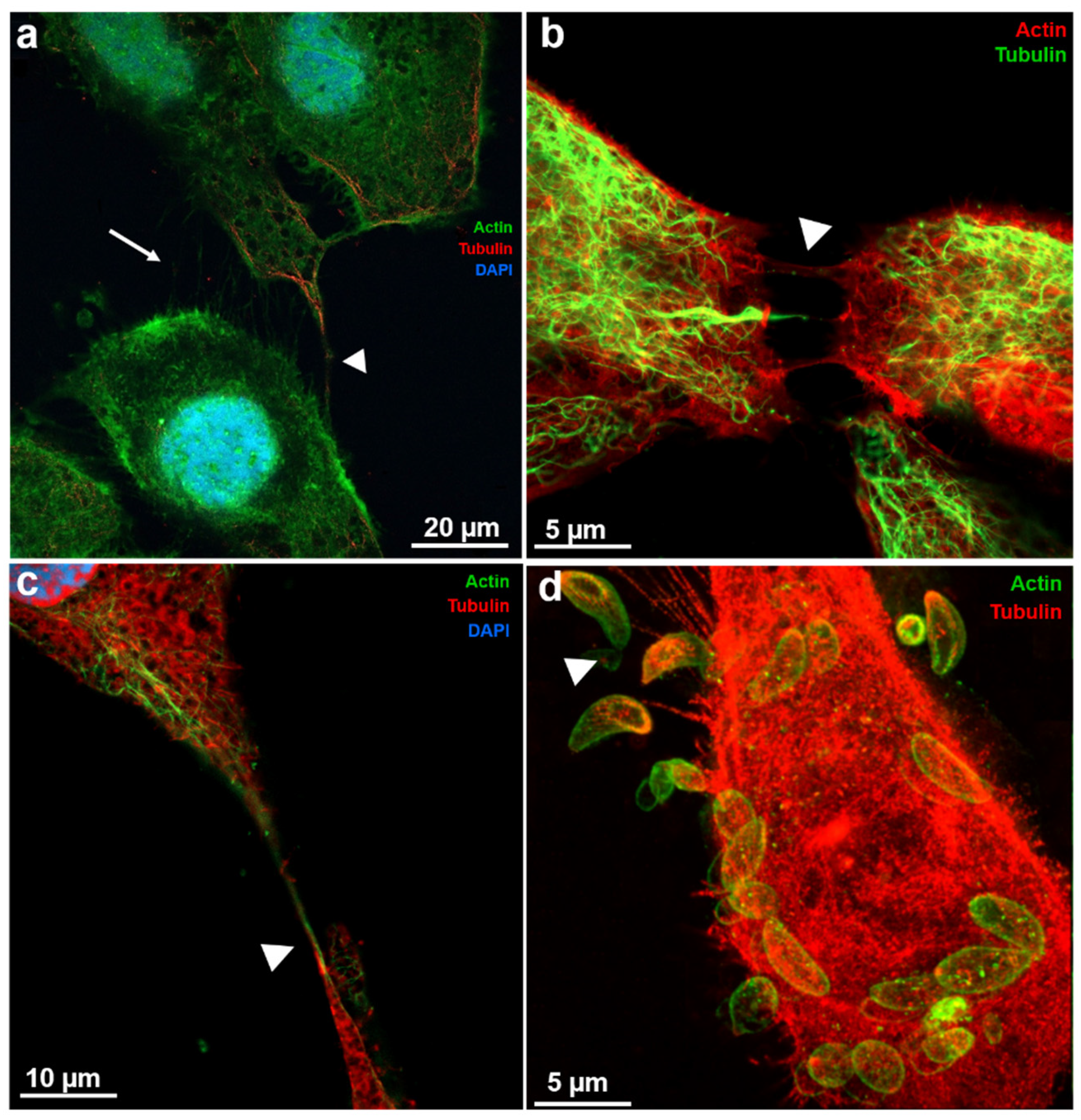
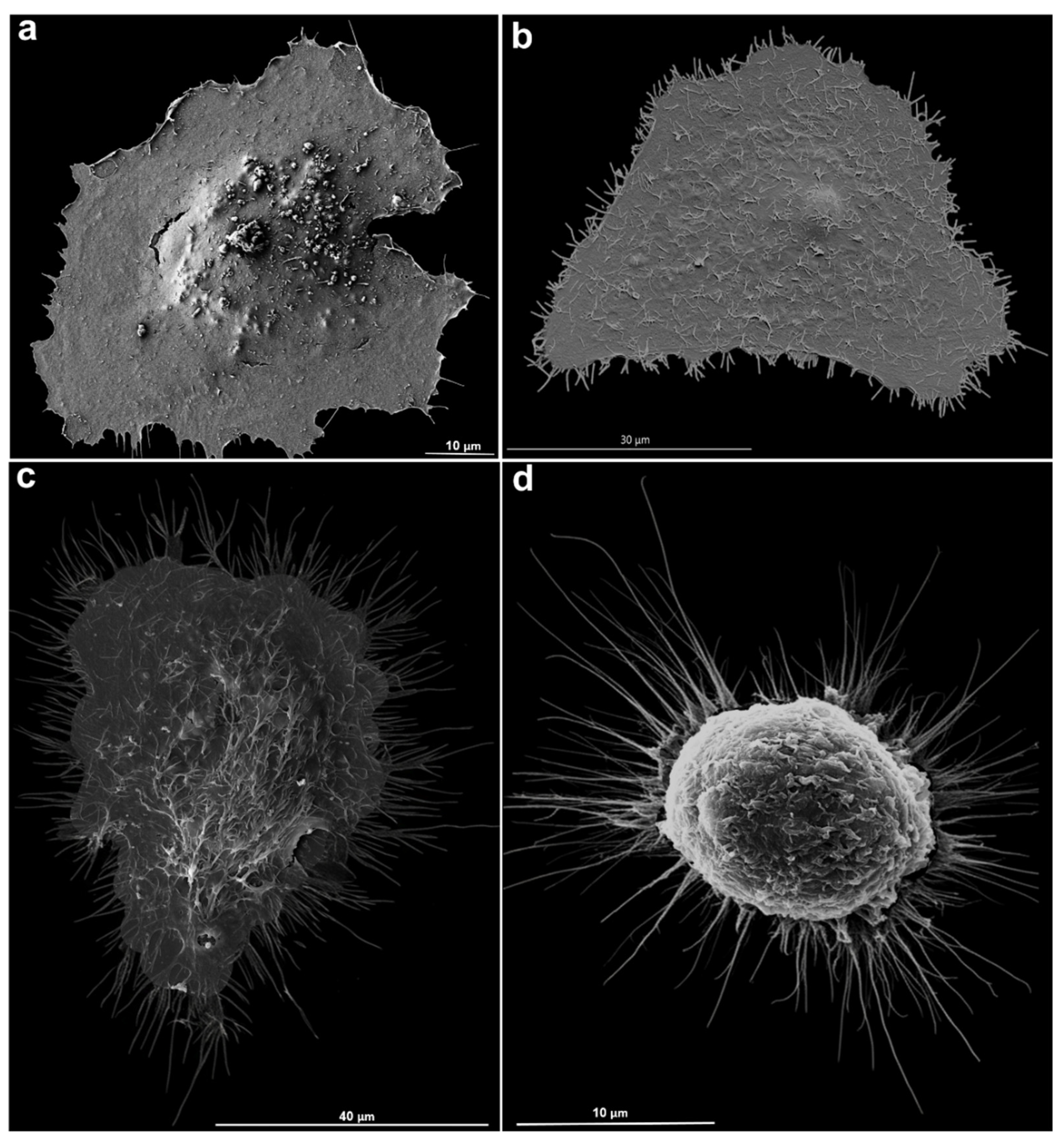
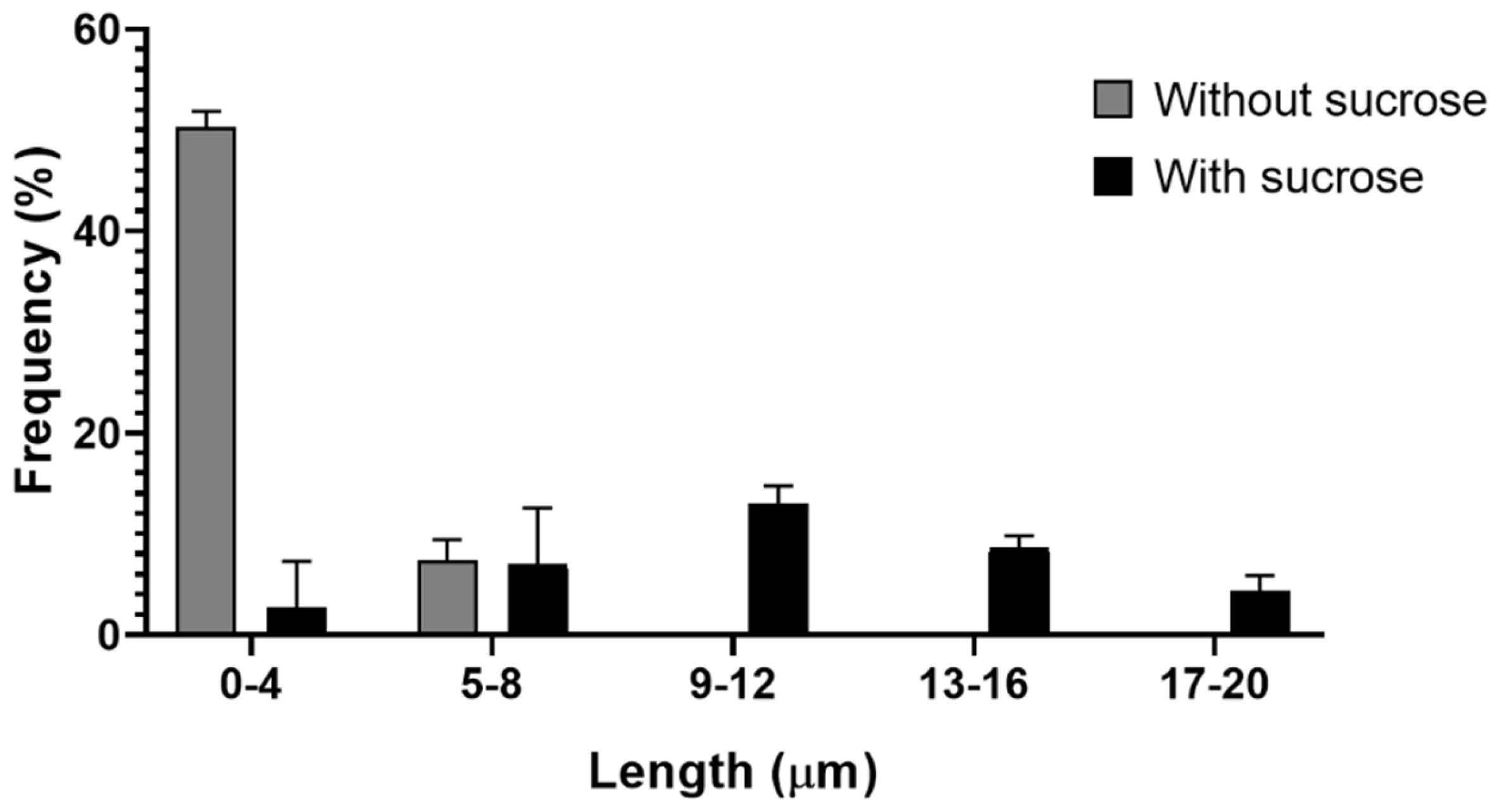
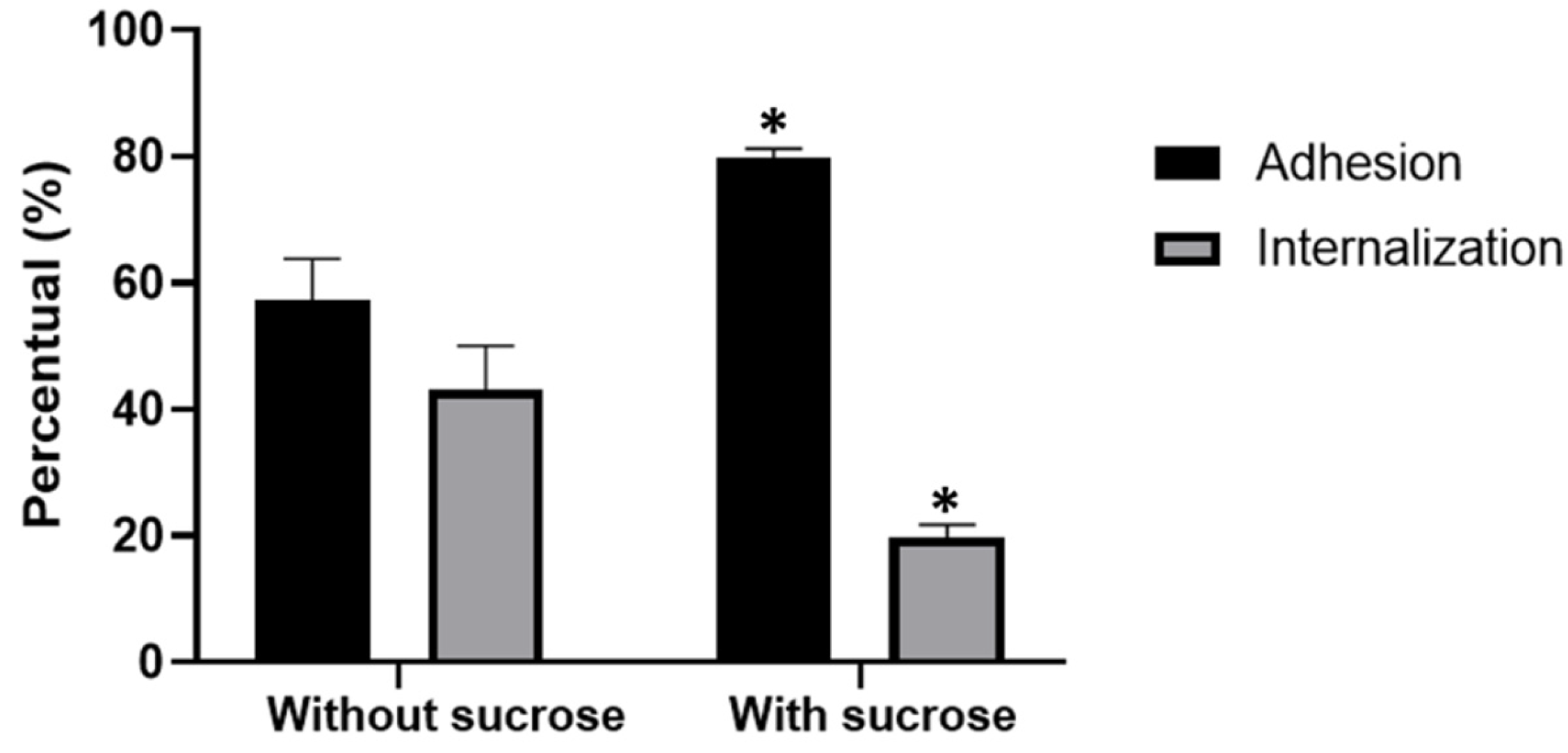
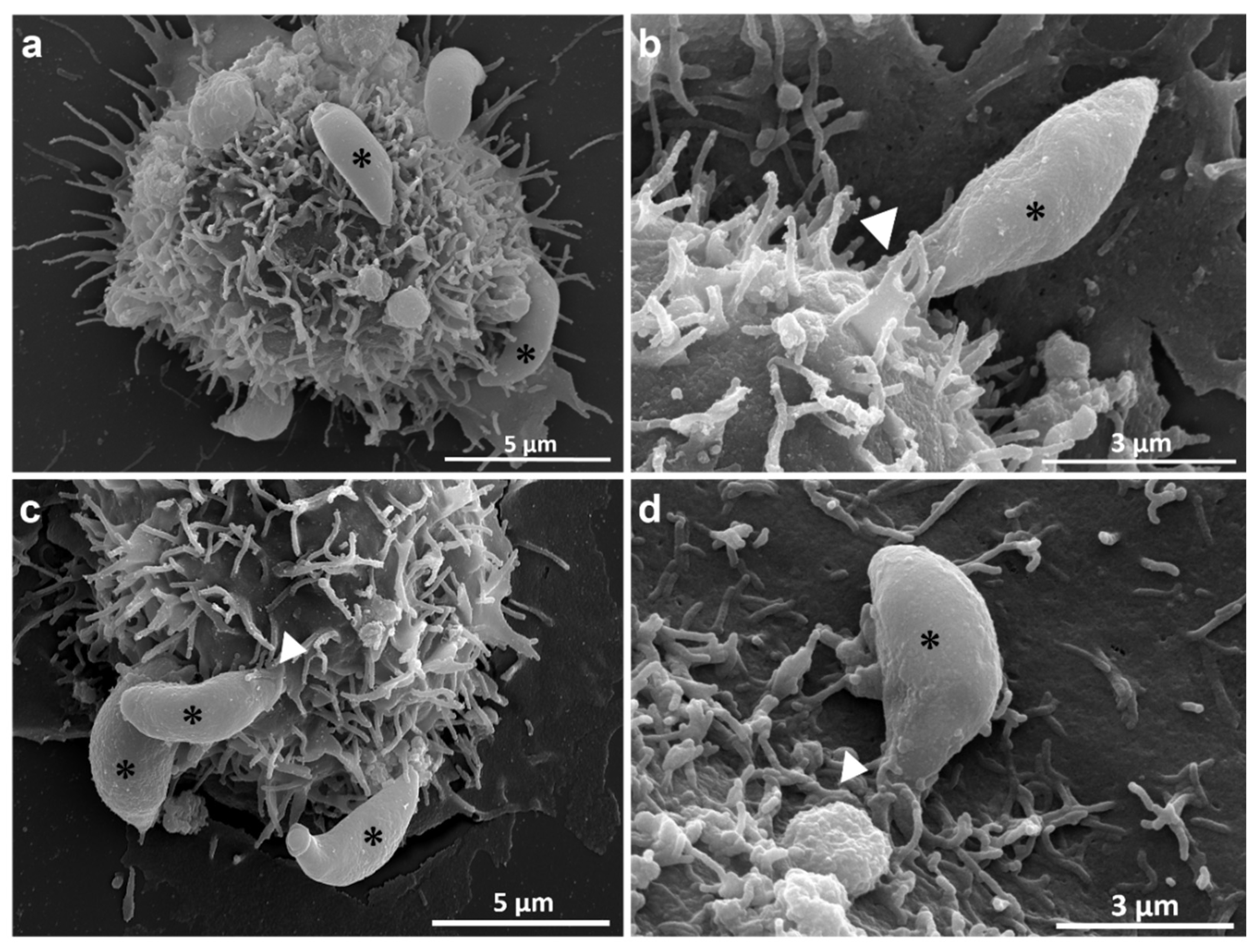
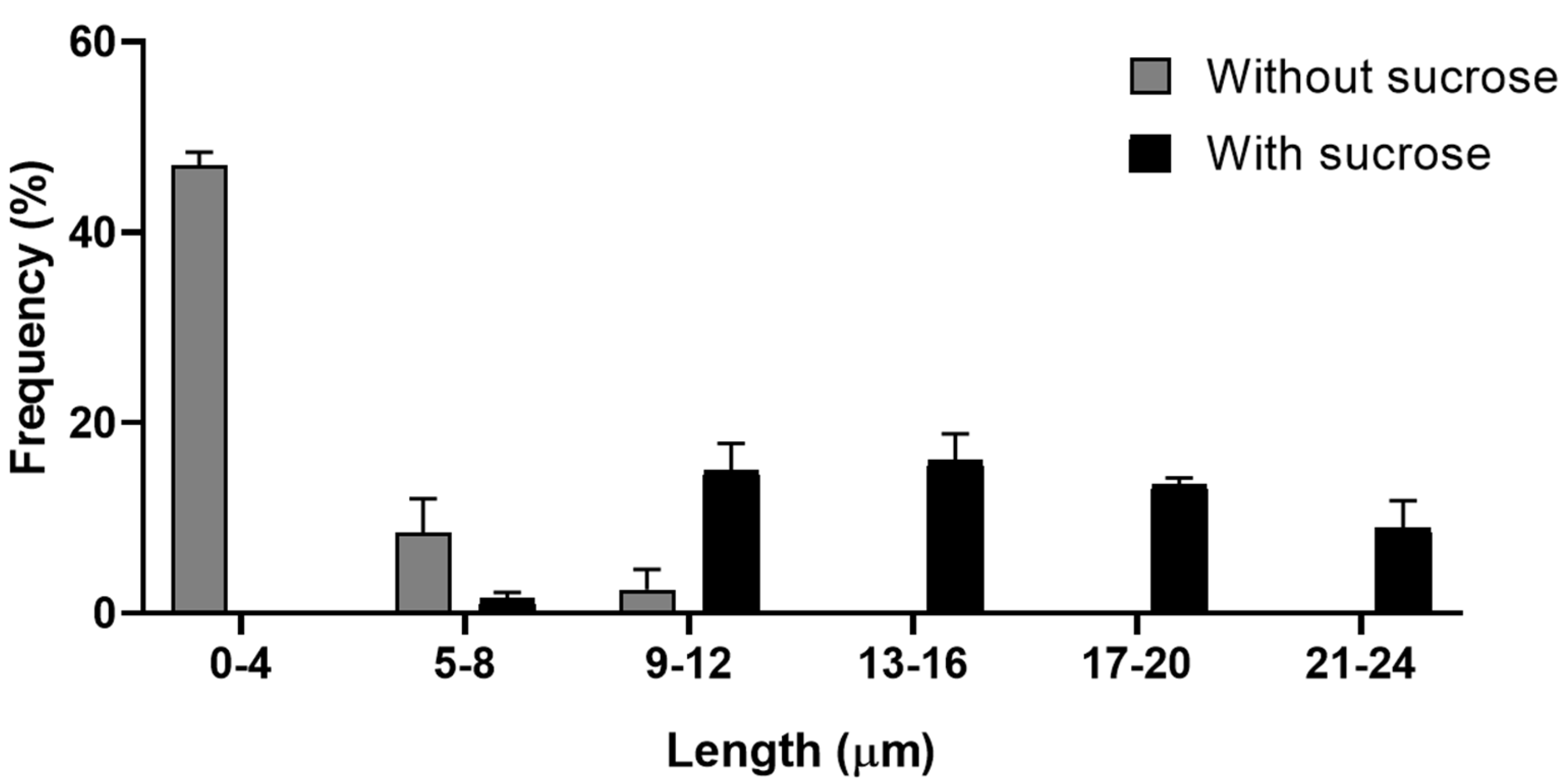
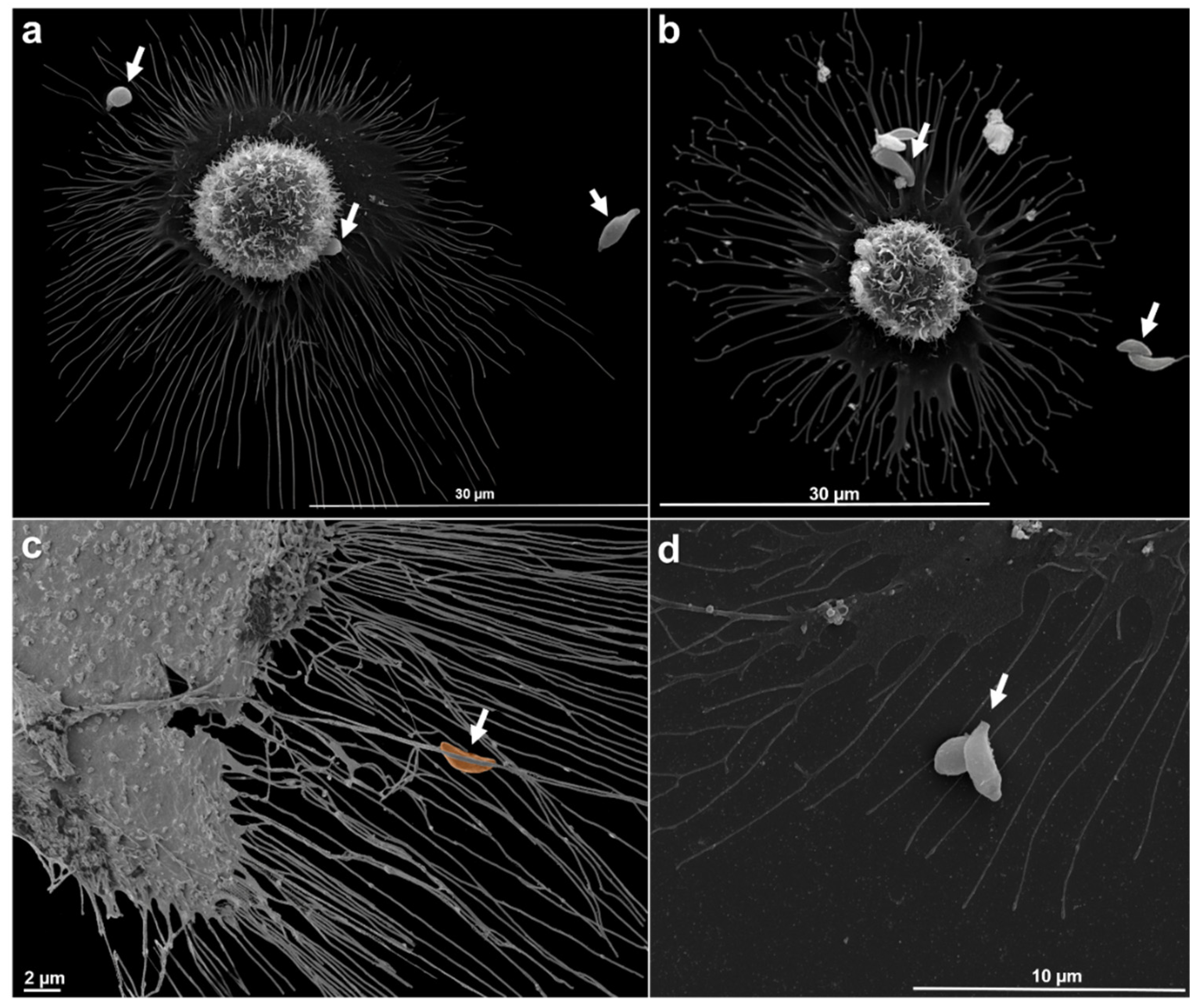
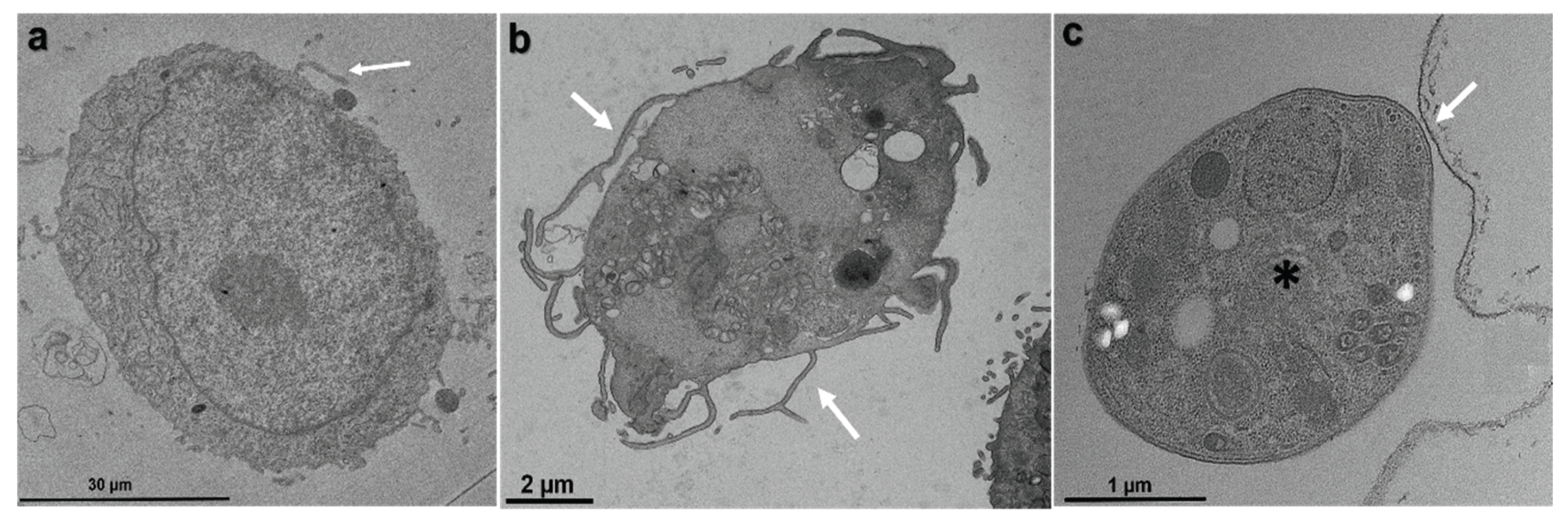
Toxoplasma gondii, the causative agent of toxoplamosis, a disease widely distributed, is an intracellular parasite that invades host cells of different tissues using specialized endocytic activity. Recent studies suggest that tunneling nanotubes (TNTs), thin cell surface projections, may participate in the parasite-host cell interaction process. We report results on the involvement of host cells TNTs in the adhesion and internalization of T. gondii tachyzoites to epithelial LLC-MK2 cells. Microscopy analysis showed that incubating cells in 0.45 M sucrose induces reversible assembly of TNTs without affecting cell viability. The presence of extended TNTs correlated with increase on parasite adhesion and reduction of parasite entry, suggesting a structural or signaling role in mediating adhesion. TNTs assembled following sucrose incubation contain both actin and tubulin components. These results highlight the functional relevance of TNTs in T. gondii host cell interaction, especially in parasite adhesion, opening new perspectives for understanding T. gondii-host cell interaction.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Host Cells
2.2. Parasite Maintenance
2.3. Interaction Assay
2.4. Induction of Tunneling Nanotube Assembly
2.5. Cell Viability
2.6. Fluorescence Microscopy
2.7. Scanning Electron Microscopy
2.8. Transmission Electron Microscopy
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Abbreviation | Meaning |
| TNT | Tunneling Nanotube(s) |
| T. gondii | Toxoplasma gondii |
| SEM | Scanning Electron Microscopy |
| TEM | Transmission Electron Microscopy |
| LLC-MK2 | Rhesus monkey kidney epithelial cell line |
| RPMI | Roswell Park Memorial Institute medium |
| PBS | Phosphate-Buffered Saline |
| pH | Hydrogen Ion Concentration |
| CO₂ | Carbon Dioxide |
| FCS | Fetal Calf Serum |
| MTS/PMS | MTS: 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium / PMS: Phenazine Methosulfate |
| BSA | Bovine Serum Albumin |
| DAPI | 4′,6-diamidino-2-phenylindole |
| OsO₄ | Osmium Tetroxide |
| nm | Nanometer |
References
- Besteiro, S.; Dubremetz, J.; Lebrun, M. The moving junction of apicomplexan parasites: a key structure for invasion. Cell. Microbiol. 2011, 13, 797–805. [Google Scholar] [CrossRef] [PubMed]
- Chaabene, R. Ben; Lentini, G.; Soldati-Favre, D. Biogenesis and discharge of the rhoptries: Key organelles for entry and hijack of host cells by the Apicomplexa. Mol. Microbiol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Dubois, D. J.; Soldati-Favre, D. Biogenesis and secretion of micronemes in Toxoplasma gondii. Cell. Microbiol. 2019, 21(5), e13018. [Google Scholar] [CrossRef] [PubMed]
- Aquilini, E.; Mendonça Cova, M.; Mageswaran, S. K.; Dos Santos Pacheco, N.; Sparvoli, D.; Penarete Vargas, D. M.; Najm, R.; Graindorge, A.; Suarez, C.; Maynadier, M.; Berry-Sterkers, L.; Urbach, S.; Ruga Fahy, P.; Guérin, A. N.; Striepen, B.; Dubremetz, J.-F.; Chang, Y.-W.; Turkewitz, A. P.; Lebrun, M. An Alveolata secretory machinery adapted to parasite host-cell invasion. Nat. Microbiol. 2021, 6, 425–434. [Google Scholar] [CrossRef]
- Teles, E.; Portes, J.; de Souza, W. New morphological observations on the initial events of Toxoplasma gondii entry into host cells. Vet. Parasitol. 2023, 322, 110006. [Google Scholar] [CrossRef] [PubMed]
- Kornberg, T. B. Cytonemes and the dispersion of morphogens. Wiley Interdiscip. Rev. Dev. Biol. 2014, 3, 445–463. [Google Scholar] [CrossRef] [PubMed]
- Rustom, A.; Saffrich, R.; Markovic, I.; Walther, P.; Gerdes, H.H. Nanotubular highways for intercellular organelle transport. Science 2004, 303, 1007–1010. [Google Scholar] [CrossRef] [PubMed]
- Onfelt, B.; Nedvetzki, S.; Benninger, R. K.; Purbhoo, M. A.; Sowinski, S.; Hume, A. N.; Seabra, M. C.; Neil, M. A.; French, P. M.; Davis, D. M. Structurally distinct membrane nanotubes between human macrophages support long-distance vesicular traffic or surfing of bacteria. J. Immunol. 2006, 177, 8476–8483. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Zhang, J.; Wu, Z.; Zhou, L.; Yu, B.; Jing, Y.; Lin, D.; Qu, J. Revealing the structure and organization of intercellular tunneling nanotubes (TNTs) by STORM imaging. Nanoscale Adv. 2022, 4, 4258–4262. [Google Scholar] [CrossRef] [PubMed]
- Seabra, S. H.; Damatta, R. A.; de Mello, F. G.; de Souza, W. Endogenous polyamine levels in macrophages is sufficient to support growth of Toxoplasma gondii. J. Parasitol. 2004, 90, 455–460. [Google Scholar] [CrossRef] [PubMed]
- Bárrias, E.; Reignault, L.; de Carvalho, T. M. U.; de Souza, W. Clathrin coated pit dependent pathway for Trypanosoma cruzi internalization into host cells. Acta Trop. 2019, 199, 105057. [Google Scholar] [CrossRef] [PubMed]
- Lou, E.; Fujisawa, S.; Morozov, A.; Barlas, A.; Romin, Y.; Dogan, Y.; Gholami, S.; Moreira, A. L.; Manova-Todorova, K.; Moore, M. A. Tunneling nanotubes provide a unique conduit for intercellular transfer of cellular contents in human malignant pleural mesothelioma. PLoS One 2012, 7(3), e33093. [Google Scholar] [CrossRef] [PubMed]
- Portes, J.; Barrias, E.; Travassos, R.; Attias, M.; de Souza, W. Toxoplasma gondii Mechanisms of Entry Into Host Cells. Front. Cell Infect. Microbiol. 2020, 10, 294. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.-L.; Zhao, H.-C.; Feng, X.-Q. Hypertonic pressure affects the pluripotency and self-renewal of mouse embryonic stem cells. Stem Cell Res. 2021, 56, 102537. [Google Scholar] [CrossRef] [PubMed]
- Salas, N.; Pedreros, M. B.; Melo, T. D. D.; Maguire, V. G.; Sha, J.; Wohlschlegel, J. A.; Pereira-Neves, A.; de Miguel, N. Role of cytoneme structures and extracellular vesicles in Trichomonas vaginalis parasite-parasite communication. eLife 2023, 12, e86067. [Google Scholar] [CrossRef] [PubMed]
- Stögerer, T.; Silva-Barrios, S.; Carmona-Pérez, L.; Swaminathan, S.; Mai, L. T.; Leroux, L. P.; Jaramillo, M.; Descoteaux, A.; Stäger, S. Leishmania donovani Exploits Tunneling Nanotubes for Dissemination and Propagation of B Cell Activation. Microbiol. Spectr. 2023, 11(4), e05096-22. [Google Scholar] [CrossRef] [PubMed]
- Midlej, V.; Tenaglia, A. H.; Luján, H. D.; de Souza, W. Tunneling Nanotube-like Structures in Giardia duodenalis. Cells 2024, 13(18), 1538. [Google Scholar] [CrossRef] [PubMed]
- Benchimol, M.; Miranda-Magalhães, A.; Pereira-Neves, A.; de Souza, W. Tritrichomonas foetus: new structures by high-resolution scanning Helium ion microscopy. Biocell 2021, 45, 259–266. [Google Scholar] [CrossRef]
- Gerdes, H.H.; Bukoreshtliev, N.V.; Barroso, J.F. Tunneling Nanotubes: A New Route for the Exchange of Components between Animal Cells. FEBS Lett. 2007, 581(11), 2194–2201. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



