Submitted:
09 March 2026
Posted:
10 March 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Experimental Part
Results and Discussion
Conclusions
References
- A. Sodiq et al., “A review on progress made in direct air capture of CO2,” Environ. Technol. Innov., vol. 29, p. 102991, 2023, . [CrossRef]
- M. Fasihi, O. Efimova, and C. Breyer, “Techno-economic assessment of CO2 direct air capture plants,” J. Clean. Prod., vol. 224, pp. 957–980, 2019, . [CrossRef]
- C. Breyer, M. Fasihi, C. Bajamundi, and F. Creutzig, “Direct Air Capture of CO2: A Key Technology for Ambitious Climate Change Mitigation,” Joule, vol. 3, no. 9, pp. 2053–2057, 2019, . [CrossRef]
- D. W. Keith, G. Holmes, D. St. Angelo, and K. Heidel, “A Process for Capturing CO2 from the Atmosphere,” Joule, vol. 2, no. 8, pp. 1573–1594, 2018, . [CrossRef]
- X. Zhu et al., “Recent advances in direct air capture by adsorption,” Chem. Soc. Rev., vol. 51, no. 15, pp. 6574–6651, 2022, doi: 10.1039/D1CS00970B.
- L. Jiang et al., “Sorption direct air capture with CO2 utilization,” Prog. Energy Combust. Sci., vol. 95, p. 101069, 2023, . [CrossRef]
- Y.-M. Wei et al., “Unlocking the economic potential of Direct Air Capture technology: Insights from a component-based learning curve,” Technol. Forecast. Soc. Change, vol. 215, p. 124109, 2025, . [CrossRef]
- L. A. Darunte, K. S. Walton, D. S. Sholl, and C. W. Jones, “CO2 capture via adsorption in amine-functionalized sorbents,” Curr. Opin. Chem. Eng., vol. 12, pp. 82–90, 2016, . [CrossRef]
- R. Navik, E. Wang, X. Ding, H. Yunyi, Y. Liu, and J. Li, “Enhanced post-combustion CO2 capture and direct air capture by plasma surface functionalization of graphene adsorbent,” J. Energy Chem., vol. 100, pp. 653–664, 2025, . [CrossRef]
- Y. Zhao, B. Zheng, J. Zhang, and H. Xu, “Research on Direct Air Capture: A Review,” 2025. doi: 10.3390/en18246632.
- N. McQueen, K. Gomes, C. McCormick, K. Blumanthal, M. Pisciotta, and J. Wilcox, “A review of direct air capture (DAC): scaling up commercial technologies and innovating for the future,” Prog. Energy, vol. 3, Apr. 2021, . [CrossRef]
- C. Dhoke, A. Zaabout, S. Cloete, and S. Amini, “Review on Reactor Configurations for Adsorption-Based CO2 Capture,” Ind. Eng. Chem. Res., vol. 60, no. 10, pp. 3779–3798, Mar. 2021, . [CrossRef]
- L. Liu, F. Gong, and R. Xiao, “Direct Air Carbon Capture and Recovery Utilizing Alkaline Solution Circulation,” Energy & Fuels, vol. 37, no. 13, pp. 9339–9346, Jul. 2023, . [CrossRef]
- F. Ghadyanlou, A. Azari, and A. Vatani, “Experimental Investigation of Mass Transfer Intensification for CO2 Capture by Environment-Friendly Water Based Nanofluid Solvents in a Rotating Packed Bed,” 2022. [CrossRef]
- J. F. Wiegner, A. Grimm, L. Weimann, and M. Gazzani, “Optimal Design and Operation of Solid Sorbent Direct Air Capture Processes at Varying Ambient Conditions,” Ind. Eng. Chem. Res., vol. 61, no. 34, pp. 12649–12667, Aug. 2022, . [CrossRef]
- R. Mennitto, R. Blom, M. Dörr, M. Rosental, and N. Rettenmaier, “Solid sorbents for direct air capture: a technological and environmental perspective,” Curr. Opin. Chem. Eng., vol. 50, p. 101195, 2025, . [CrossRef]
- M. C. Davis, W. J. M. Kort-Kamp, I. Matanovic, P. Zelenay, and E. F. Holby, “Design of active sites for amine-functionalized direct air capture materials using integrated high-throughput calculations and machine learning,” Commun. Chem., vol. 9, no. 1, p. 12, 2025, . [CrossRef]
- X. Zhang, H. Zhao, Q. Yang, M. Yao, Y. Wu, and Y. Gu, “Direct air capture of CO2 in designed metal-organic frameworks at lab and pilot scale,” Carbon Capture Sci. Technol., vol. 9, p. 100145, 2023, . [CrossRef]
- C. Zhang, S. Sun, S. He, and C. Wu, “Direct air capture of CO2 by KOH-activated bamboo biochar,” J. Energy Inst., vol. 105, pp. 399–405, 2022, . [CrossRef]
- S. Hashmi, M. Rasool, W. H., and H. Moiz, “Activated Carbons for Direct Air Capture: Adsorption Mechanisms, Material Design and Performance Optimization,” Feb. 2025.
- S. Acevedo, L. Giraldo, and J. C. Moreno-Piraján, “Adsorption of CO2 on Activated Carbons Prepared by Chemical Activation with Cupric Nitrate,” ACS Omega, vol. 5, no. 18, pp. 10423–10432, May 2020, . [CrossRef]
- N. S. Mohamed Hatta, M. K. Aroua, F. Hussin, and L. T. Gew, “A Systematic Review of Amino Acid-Based Adsorbents for CO2 Capture,” 2022. [CrossRef]
- D. Barker-Rothschild et al., “Lignin-based porous carbon adsorbents for CO2 capture,” Chem. Soc. Rev., vol. 54, no. 2, pp. 623–652, 2025, . [CrossRef]
- H. E. Holmes, S. Banerjee, A. Vallace, R. P. Lively, C. W. Jones, and M. J. Realff, “Tuning sorbent properties to reduce the cost of direct air capture,” Energy Environ. Sci., vol. 17, no. 13, pp. 4544–4559, 2024, . [CrossRef]
- S. A. Khan, S. Ahmed, S. Ali, and F. Altaf, “Advances in CO2 Capture Materials: From Strategies to CO2-Based Fertilizers for a Sustainable Future,” Carbon Neutralization, vol. 4, no. 6, p. e70063, Nov. 2025, . [CrossRef]
- A. I. Hadi et al., “A comprehensive review of carbon capture: From conventional to emerging electrochemical technologies,” Next Energy, vol. 9, p. 100415, 2025, . [CrossRef]
- M. Zanatta, “Materials for Direct Air Capture and Integrated CO2 Conversion: Advancement, Challenges, and Prospects,” ACS Mater. Au, vol. 3, no. 6, pp. 576–583, Nov. 2023, . [CrossRef]
- M. B. Ahmed et al., “Activated carbon preparation from biomass feedstock: Clean production and carbon dioxide adsorption,” J. Clean. Prod., vol. 225, pp. 405–413, 2019, . [CrossRef]
- H. Jedli, M. Almonnef, R. Rabhi, M. Mbarek, J. Abdessalem, and K. Slimi, “Activated Carbon as an Adsorbent for CO2 Capture: Adsorption, Kinetics, and RSM Modeling,” ACS Omega, vol. 9, no. 2, pp. 2080–2087, Jan. 2024, . [CrossRef]
- N. A. Rashidi and S. Yusup, “An overview of activated carbons utilization for the post-combustion carbon dioxide capture,” J. CO2 Util., vol. 13, pp. 1–16, 2016, . [CrossRef]
- W. Jiao, S. Luo, Z. He, and Y. Liu, “Applications of high gravity technologies for wastewater treatment: A review,” Chem. Eng. J., vol. 313, pp. 912–927, 2017, . [CrossRef]
- K. Neumann et al., “A guide on the industrial application of rotating packed beds,” Chem. Eng. Res. Des., vol. 134, pp. 443–462, 2018, . [CrossRef]
- M. P. Singh, A. M. Alatyar, A. S. Berrouk, and M. Saeed, “Numerical modelling of rotating packed beds used for CO2 capture processes: A review,” Can. J. Chem. Eng., vol. 101, no. 11, pp. 6170–6202, Nov. 2023, . [CrossRef]
- D. P. Rao, A. Bhowal, and P. S. Goswami, “Process Intensification in Rotating Packed Beds (HIGEE): An Appraisal,” Ind. Eng. Chem. Res., vol. 43, no. 4, pp. 1150–1162, Feb. 2004, . [CrossRef]
- A. C. Mitropoulos, R. I. Kosheleva, M. Kostoglou, and T. D. Karapantsios, “The Effect of Rotation on Gas Storage in Nanoporous Materials,” Separations, vol. 11, no. 3, 2024, . [CrossRef]
- R. I. Kosheleva, T. D. Karapantsios, M. Kostoglou, and A. C. Mitropoulos, “A novel device for in situ study of gas adsorption under rotation,” Rev. Sci. Instrum., vol. 92, no. 4, p. 45106, Apr. 2021, . [CrossRef]
- S. Bulbul and M. Ergün, “Production and characterization of activated carbon from Black Poplar (Populus Nigra) wood waste with different chemical activation methods,” Int. Adv. Res. Eng. J., vol. 6, Nov. 2022, . [CrossRef]
- C. P. Bonaventura, S. Bartucci, G. Conte, G. Desiderio, R. G. Agostino, and A. Policicchio, “Optimizing CO2 and CH4 storage: The role of high skeletal density activated carbon in adsorption processes,” Colloids Surfaces A Physicochem. Eng. Asp., vol. 721, p. 137209, 2025, . [CrossRef]
- R. I. Kosheleva, T. D. Karapantsios, M. Kostoglou, and A. C. Mitropoulos, “Thermodynamic analysis of the effect of rotation on gas adsorption,” J. Non-Equilibrium Thermodyn., vol. 48, no. 4, 2023, doi: doi:10.1515/jnet-2022-0086.
- V. R. Choudhary and S. Mayadevi, “Adsorption of methane, ethane, ethylene, and carbon dioxide on silicalite-l,” Zeolites, vol. 17, no. 5–6, pp. 501–507, 1996.
- V. K. Singh and E. A. Kumar, “Experimental investigation and thermodynamic analysis of CO2 adsorption on activated carbons for cooling system,” J. CO2 Util., vol. 17, pp. 290–304, 2017, . [CrossRef]
- K. L. Tan and B. H. Hameed, “Insight into the adsorption kinetics models for the removal of contaminants from aqueous solutions,” J. Taiwan Inst. Chem. Eng., vol. 74, pp. 25–48, 2017, . [CrossRef]
- V. I. Geyko and N. J. Fisch, “Reduced Compressibility and an Inverse Problem for a Spinning Gas,” Phys. Rev. Lett., vol. 110, no. 15, p. 150604, Apr. 2013, . [CrossRef]
- S. Shi, “Diffusion model based on Fick’s second law for the moisture absorption process in wood fiber-based composites: Is it suitable or not?,” Wood Sci. Technol., vol. 41, pp. 645–658, Dec. 2007, . [CrossRef]
- Y. Kim and J. T. Gostick, “Measuring effective diffusivity in porous media with a gasket-free, radial arrangement,” Int. J. Heat Mass Transf., vol. 129, pp. 1023–1030, 2019, . [CrossRef]
- H. Naidu and A. P. Mathews, “Linear driving force analysis of adsorption dynamics in stratified fixed-bed adsorbers,” Sep. Purif. Technol., vol. 257, p. 117955, 2021, . [CrossRef]
- E. D. Revellame, D. L. Fortela, W. Sharp, R. Hernandez, and M. E. Zappi, “Adsorption kinetic modeling using pseudo-first order and pseudo-second order rate laws: A review,” Clean. Eng. Technol., vol. 1, p. 100032, 2020.
- Y. S. Ho and G. McKay, “Pseudo-second order model for sorption processes,” Process Biochem., vol. 34, no. 5, pp. 451–465, 1999, . [CrossRef]
|
Section |
Transition (mbar) |
ΔG (J/mol) |
ΔS (J/mol.K) |
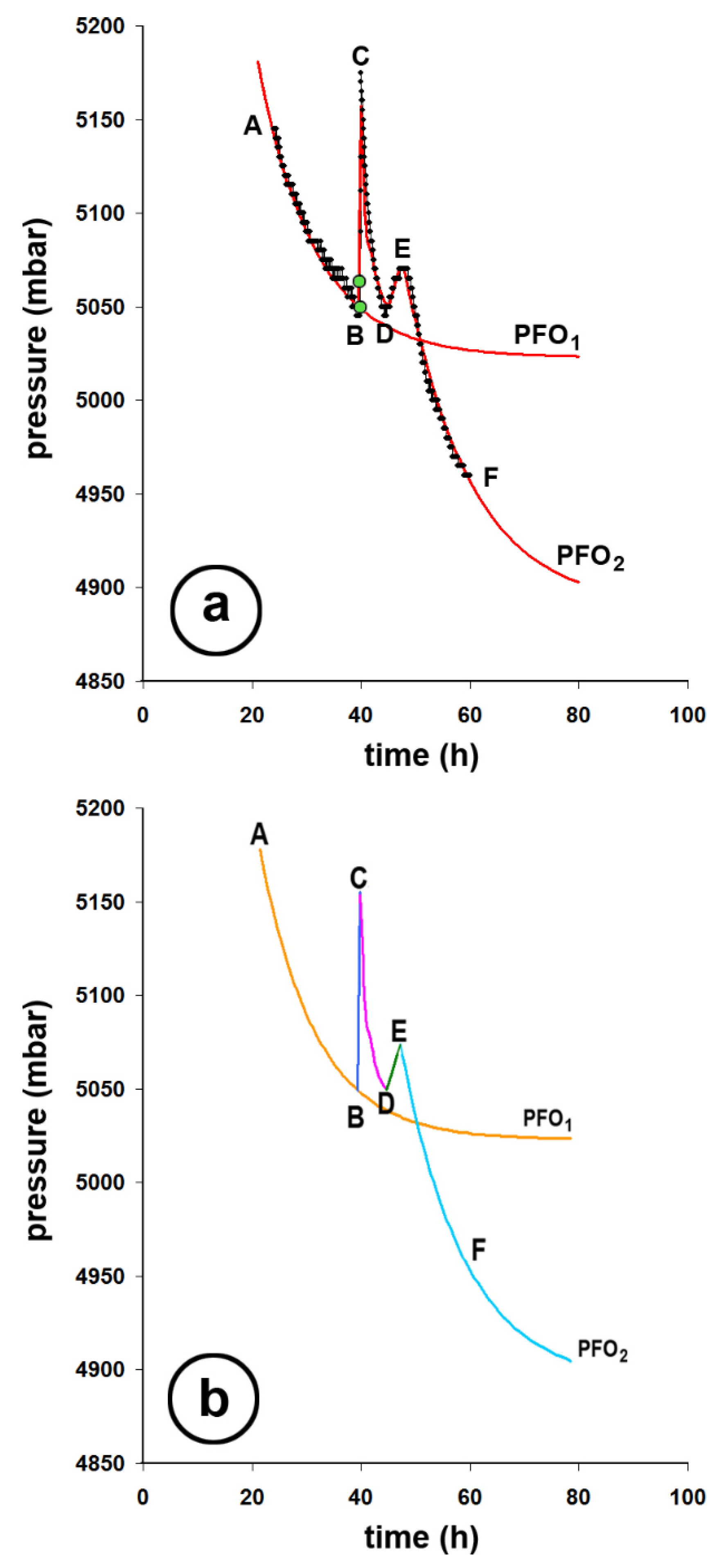
| AB | 6.190 to 5,045 | -507.01 | -79.80 |
| BC | 5,045 to 5,175 | +63,07 | +81.29 |
| CD | 5,175 to 5,045 | -63,07 | -81.29 |
| DE | 5,045 to 5,070 | +12.25 | +81.46 |
| EF | 5,070 to 4,960 | -54.37 | -81.32 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




