1. Introduction
Natural products remain a major source of lead compounds in drug discovery due to their structural diversity and biological activities [
1,
2,
3]. Quassinoids, a class of highly oxygenated triterpenoids predominantly found in the Simaroubaceae family, are known for their potent anticancer and antimalarial properties [
4,
5,
6,
7]. Among them, Bruceine A, isolated from the seeds of
Brucea javanica, has attracted considerable attention because of its cytotoxic and antiplasmodial activities [
8,
9].
Semisynthesis has been widely applied as a strategic approach to overcome the limitations associated with natural product drug discovery, particularly when the target compounds are available only in small quantities from natural sources. By introducing specific functional groups into an existing bioactive scaffold, semisynthesis allows modulation of physicochemical properties, biological activity, and selectivity without the need for total synthesis [
10]. In the case of quassinoids, selective modification of hydroxyl functionalities has been reported to significantly influence cytotoxic and antimalarial activities, highlighting the importance of controlled acylation reactions in structure–activity relationship (SAR) studies [
11,
12,
13]. Although numerous quassinoids have been isolated from Brucea javanica, systematic semisynthesis modification of Bruceine-A remains limited. Most previous studies have focused on isolation and biological screening, while mechanistic aspects of selective acylation reactions have received less attention. In particular, comparative evaluation of protection–deprotection versus direct acylation strategies has rarely been discussed in detail [
14].
Bruceine-A possesses multiple oxygenated functional groups, including hydroxyl moieties at C-3 and C-15, which provide suitable reactive sites for chemical modification. Previous studies have demonstrated that the hydroxyl group at C-3 is particularly reactive due to electronic effects arising from the adjacent enone system, making it a preferred site for selective acylation [
12,
15]. Therefore, exploration of direct acylation reactions at this position is not only chemically rational but also relevant for the development of new Bruceine A derivatives with potential therapeutic value. In this context, the present study emphasizes mechanistic understanding and feasibility of semisynthesis rather than reaction optimization, which is appropriate considering the limited availability of the natural starting material.
Despite its promising biological activity, the development of Bruceine A as a therapeutic agent is limited by issues related to potency, selectivity, and availability. Semisynthesis provides a practical approach to structurally modify natural compounds while preserving their complex core skeleton. Previous studies have demonstrated that minor modifications of quassinoid structures can lead to significant changes in biological activity [
12]. Therefore, this study aims to provide a mechanistic and practical evaluation of semisynthesis approaches for Bruceine A derivatives, emphasizing reaction feasibility under limited material conditions. Understanding the regioselectivity and reaction pathway of acylation at the C-3 hydroxyl group is essential for future structure–activity relationship studies and rational design of quassinoid-based bioactive compounds.
2. Materials and Methods
Equipment
The equipment utilized were glassware (Pyrex) that is commonly employed in the field of natural product chemistry research, including glass funnels, stirring rods, dropper pipettes, column chromatography with a diameter of 10 mm and a length of 30 cm, desiccators, and a JEOL ECA 300 NMR spectrophotometer.
2.1. Material
The starting material Bruceine A used in the present study was obtained from our own previous isolation work on the seeds of Brucea javanica. The compound was isolated using standard natural product procedures involving solvent extraction, liquid–liquid partitioning, and silica gel chromatography. The purity of the isolated Bruceine A was confirmed prior to use through TLC and spectroscopic analysis (UV, IR, and NMR) prior to use in semisynthetic reactions.
The chemicals utilized in this study are ethyl acetate, chloroform , imidazole, Hydrochloric acid, Chlorobenzoyl chloride, DMF methanol solvents procured from E. Merck. The solvents acetonitrile high grade for employed HPLC, The solvent for TLC and column chromatography are 100% chloroform, a chloroform: methanol (9: 1) solution, and a chloroform: methanol (8: 2) solution, silica Gel 60 N (spherical, neutral, 40–50 μm, Sigma Aldrich Chemical Co., Inc.), TLC plate (silica gel 60 F254, Merck KGaA Darmstadt, Germany) with ultraviolet lamp 254 nm and DMSO-deuterium-ethanol solvent (DMSO-D6) and pyridine .
2.2. Design of Semisynthesis Pathways
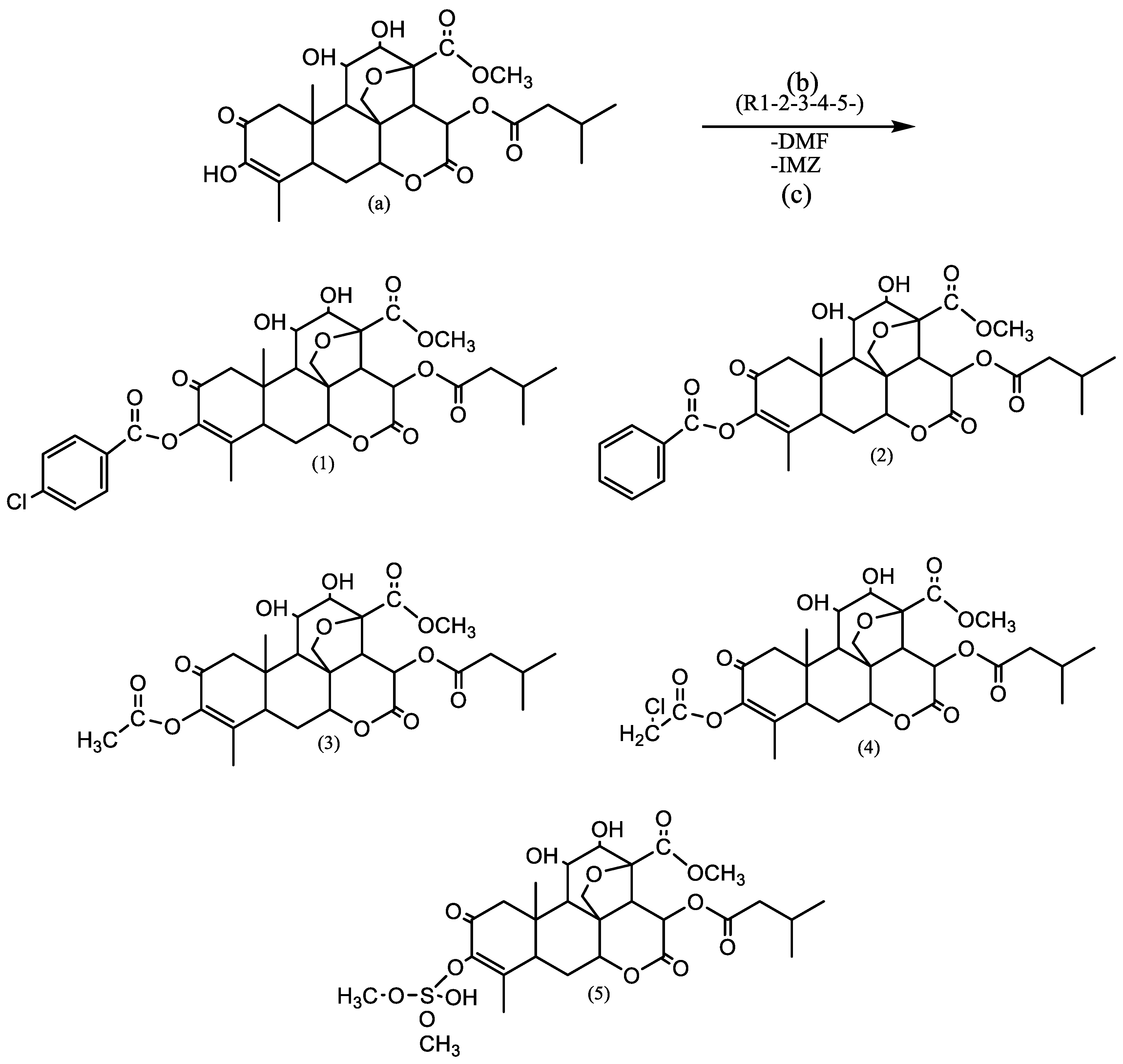
Two semisynthesis strategies for Bruceine-A were designed. The first pathway involved protection of the hydroxyl group at C-3 using tert-butyldimethylsilyl chloride (TBDMS-Cl) to prevent undesired reactions at this position. After protection, ester cleavage at C-15 was planned via alkaline hydrolysis using sodium methoxide (MeONa), followed by acylation at C-15. However, this pathway was not pursued further due to limited availability of Bruceine A, difficulties in solvent selection, and increased reaction complexity.
The second, alternative pathway involved direct acylation of Bruceine-A. This approach was based on the higher reactivity of the hydroxyl group at C-3 compared to other hydroxyl groups, attributed to the presence of an enone moiety in the Bruceine-A skeleton [
12]. This pathway was selected as the most practical and efficient route.
2.3. General Procedure for Direct Acylation
Bruceine-A was directly reacted with the acylating agent in N,N-dimethylformamide (DMF) ( E. Merck) as the solvent and imidazole as a base catalyst. The reaction mixture was heated at 40–50 °C for 24–36 h. Imidazole (E. Merck) facilitated the reaction by activating the hydroxyl group through proton abstraction, enabling nucleophilic substitution at the acyl carbon.
After completion, the reaction mixture was extracted with chloroform (E. Merck) and ethyl acetate (E. Merck), and the crude product was purified by thin-layer chromatography (TLC). Structural elucidation was conducted using spectroscopic techniques.
Due to the limited availability of Bruceine-A and the exploratory nature of the semisynthesis work, reaction yields were not quantitatively determined. Product formation was monitored by TLC and confirmed by spectroscopic analysis after purification by preparative TLC.
3. Results
3.1. Reaction Mechanism of Acylation
Acylation, also known as alkanoylation, is defined as the introduction of an acyl group into a target molecule.[
16,
17] In this study, acyl chlorides acted as strong electrophilic acylating agents. The reaction proceeded via nucleophilic attack of the oxygen atom of the C-3 hydroxyl group of Bruceine A on the carbonyl carbon of the acyl chloride. This process was accompanied by the release of a chloride ion (Cl⁻) and a proton (H⁺), resulting in the formation of hydrogen chloride (HCl) as a by-product.
Imidazole enhanced the ionic nature of the reaction medium, facilitating the departure of the chloride leaving group and increasing the rate of acylation.[
18] The reaction occurred selectively at the C-3 hydroxyl position, as illustrated in the reaction schemes. Starting point Bruceine A (
Figure 1) and mechanistic reaction with acylation showed in (
Figure 2).
3.2. Semisynthesis of 3-O-Chlorobenzoylbruceine A (P1)
Bruceine-A (C₂₆H₃₄O₁₁, 30 mg) was dissolved in DMF (800–1000 µL) and stirred for 15 min until complete dissolution. Imidazole (25 mg) was then added, and the mixture was stirred for an additional 30 min to ensure effective activation of the C-3 hydroxyl group. Subsequently, chlorobenzoyl chloride (C₆H₄ClCOCl; molecular weight 175.17 g/mol) was added (25 µL) in DMF.
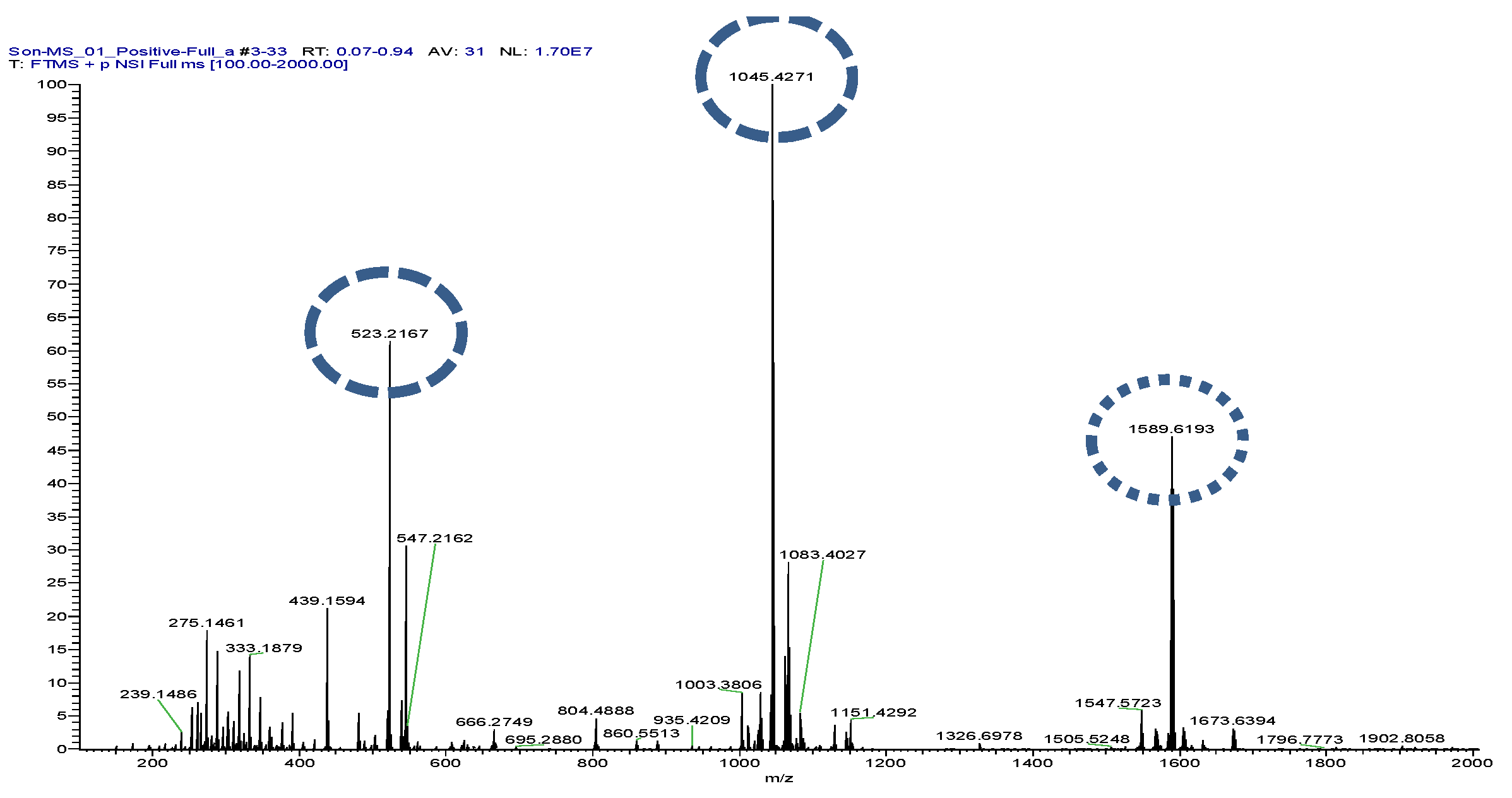
The reaction mixture was heated at 40–50 °C for 36–48 h. Reaction progress was monitored by TLC using chloroform:ethyl acetate (3:5 and 4:6) as the mobile phase. Completion of the reaction was indicated by the disappearance of the starting material spot and the appearance of a new product spot. After cooling, cold water (1 mL) was added to induce precipitation. The product was extracted with chloroform or ethyl acetate and purified by TLC. A total of 15.5 mg of the semisynthesis product was obtained. Spectroscopic analysis (UV, IR, ^1H-NMR, ^13C-NMR, and LC–MS) confirmed the structure of the product as 3-O-chlorobenzoylbruceine (P1). Full spectral data were HRESI/MS showed in
Figure 2.
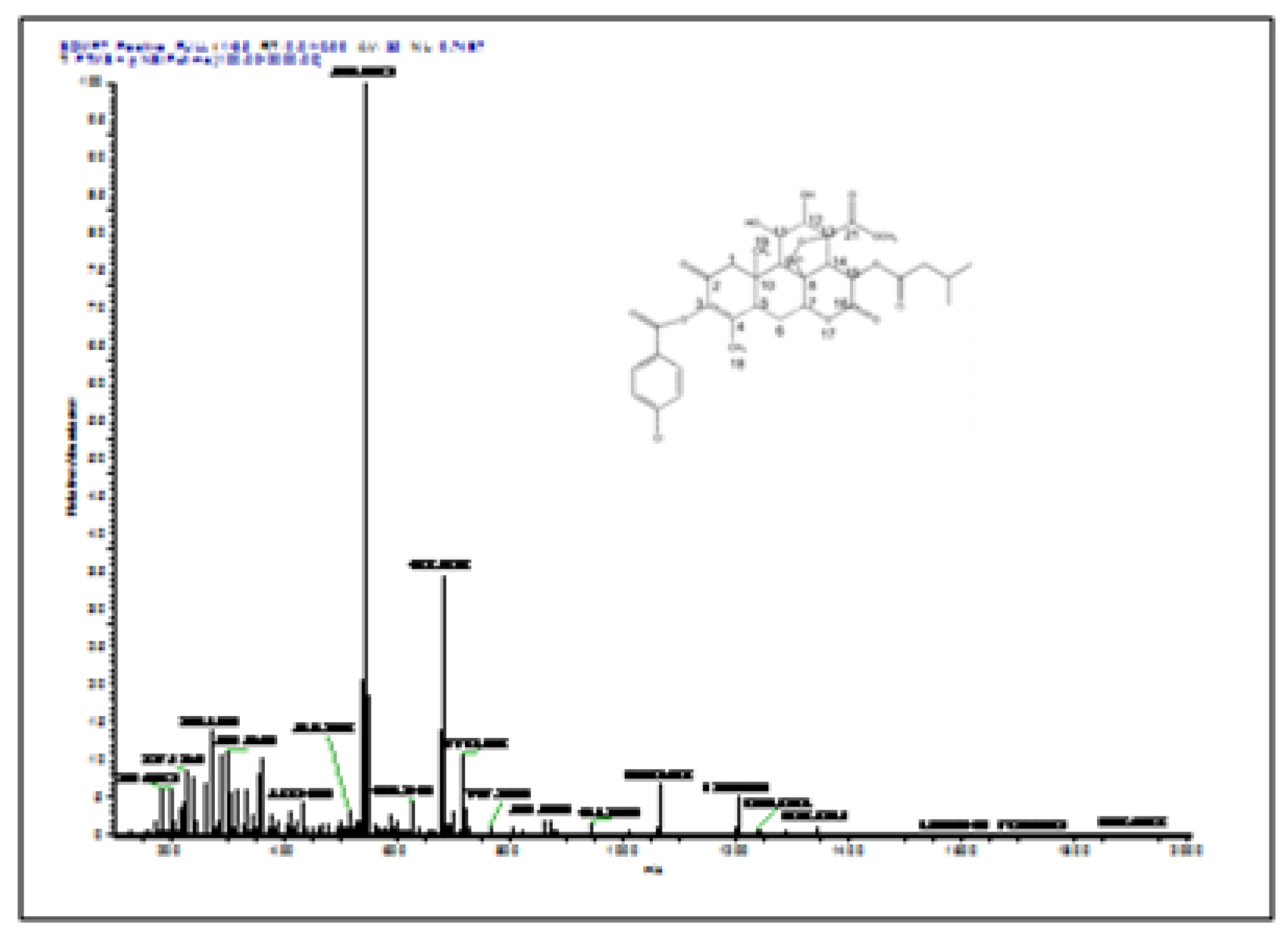
The semisynthesis route described herein is based on our laboratory work. The focus is on the synthetic strategy and reaction mechanism. Structural elucidation of the product was confirmed using standard spectroscopic techniques, including ¹H-NMR, ¹³C-NMR, and LC–MS, as presented in
Figure 1,
Figure 2, Figure 3,
Figure 4 and
Figure 5.
4. Discussion
The compound was synthesized via direct acylation of Bruceine A. Characterization data (LC-MS) were generated (
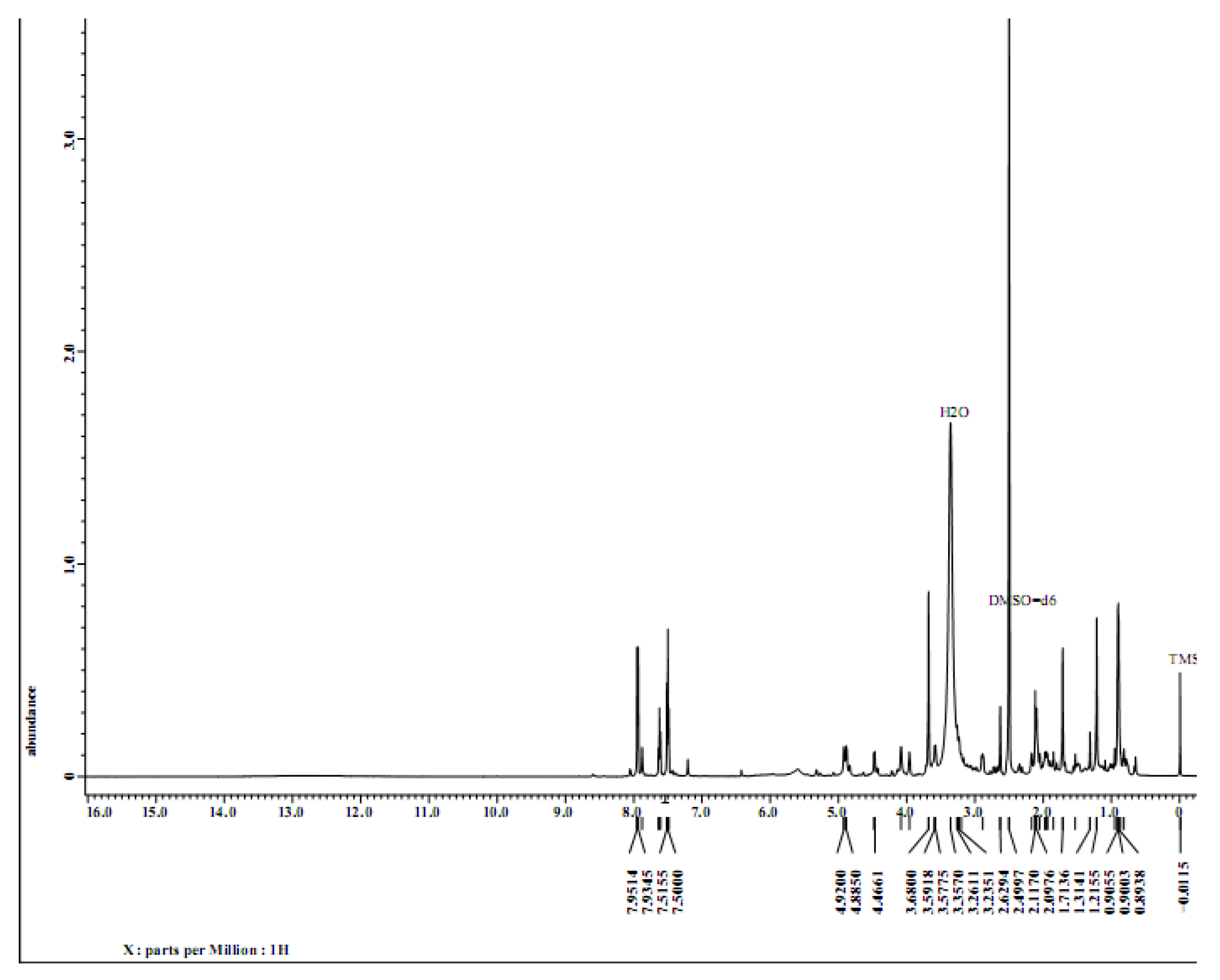
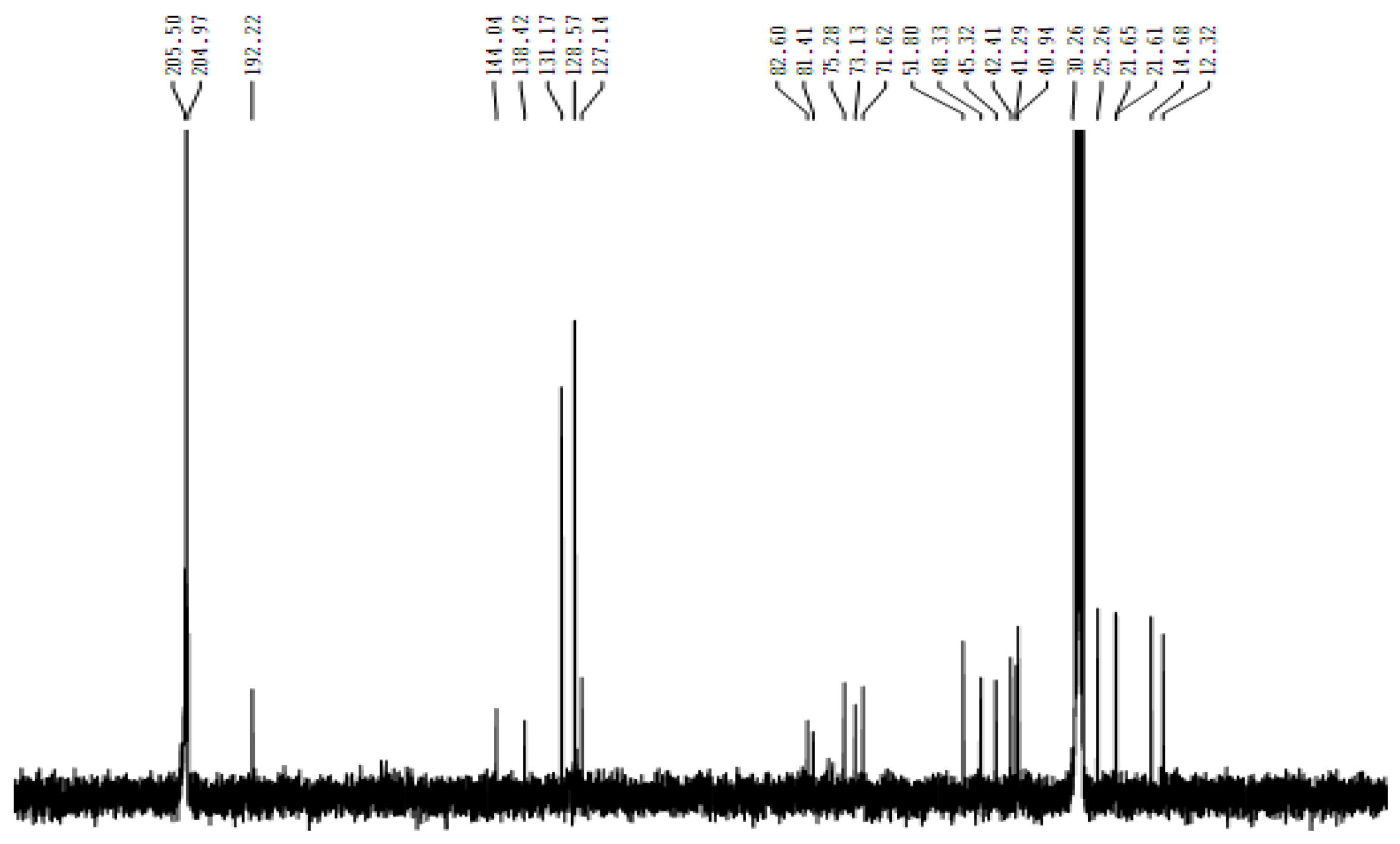
Figure 1). The chemical structure of pure compounds or chemical components that have been isolated is then elucidated. This is achieved through the use of nuclear magnetic resonance (NMR) spectroscopy, specifically proton (1H-NMR) and carbon (13C-NMR) spectroscopy, using a deuterium-methanol solvent (C2D5OD). The NMR spectra display the chemical shifts of the protons and carbons of the acylation compound
Figure 4 (specra HNMR) and
Figure 5 (spectra CNMR), indicating the presence of several protons and carbons in the compound acylation of Bruceine A to have results.
The results demonstrate that the direct acylation pathway is the most feasible semisynthesis route for Bruceine-A derivatives. The higher reactivity of the C-3 hydroxyl group, influenced by the enone system, enables selective acylation without the need for protecting groups [
12,
19]. The use of DMF as an aprotic polar solvent provided good solubility for Bruceine A, while imidazole effectively catalyzed the reaction by facilitating proton transfer and stabilizing reaction intermediates.
The successful synthesis
of 3-O-chlorobenzoylbruceine A confirms that structural modification of Bruceine-A can be achieved efficiently without disrupting the quassinoid core. These findings are consistent with previous reports on quassinoid semisynthesis and structure–activity relationships [
11,
15,
20].
Structural modification of quassinoids through acylation may significantly influence their physicochemical properties, particularly lipophilicity and membrane permeability. Introduction of an aromatic acyl moiety such as chlorobenzoyl can increase hydrophobic character, which may enhance passive diffusion across biological membranes and potentially improve intracellular access to
Plasmodium parasites. Alterations in lipophilicity are known to play a crucial role in optimizing antiplasmodial activity, as membrane penetration and interaction with parasite-specific molecular targets are key determinants of efficacy [
8,
21]. Therefore, acylation at the C-3 hydroxyl position of Bruceine A may not only serve as a synthetic modification but also represent a strategic approach for modulating biological activity. Further biological evaluation is required to confirm the impact of this structural modification on antiplasmodial potency [
22].
Reaction scheme of Bruceine A with acylating reagents showed in
Figure 6. Compound the reagents employed include chlorobenzoyl chloride, benzoyl chloride, dimethyl sulfate, acetyl chloride, chloroacetyl chloride, and tert-butyldimethylsilyl chloride (TBDMS-Cl).
The findings of the present study are aligned with previous reports indicating that chemical modification at the C-3 hydroxyl group of Bruceine A plays a crucial role in modulating antiplasmodial efficacy. The successful synthesis of 3-O-chlorobenzoylbruceine and its observed activity support earlier evidence that acylated derivatives of Bruceine A retain inhibitory effects against
P. falciparum and may display altered potencies depending on the introduced substituent. These observations correspond with the results of Tenda et al. [
23], who demonstrated that different acyl and alkyl substituents at the C-3 position produce measurable differences in antiplasmodial activity, further emphasizing the relevance of targeted semisynthetic modification for structure–activity relationship development.
✍️ Catatan kecil (opsional, untuk jurnal kimia)
Jika Bapak ingin lebih “journal-style”, saya bisa menambahkan satu kalimat mekanistik, misalnya:
The reactions proceed via selective acylation of the hydroxyl group at C-3 of the Bruceine A skeleton.
Silakan beri tahu:
apakah teks ini akan ditempatkan sebagai caption Figure (Reaction Scheme), atau
sebagai paragraf Methods / Results.
Saya siap menyesuaikan persis dengan target jurnal Bapak.
Memory full
5. Conclusion
This study successfully achieved the semisynthesis of 3-O-chlorobenzoylbruceine (P1) through direct acylation of Bruceine-A at the C-3 hydroxyl group using chlorobenzoyl chloride, DMF, and imidazole. Compared to protection–deprotection strategies, the direct acylation route proved to be more efficient, practical, and suitable under conditions of limited starting material. The results highlight the potential of semisynthesis as a valuable strategy for the development of Bruceine A derivatives with enhanced biological relevance.
Supplementary Materials
The following supporting information can be downloaded at the website of this paper posted on Preprints.org.
Acknowledgments
The author declares that the compound and preliminary characterization data were generated in our laboratory. The authors acknowledge the laboratory facilities of Universitas Gadjah Mada for supporting this research.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Cragg, G.M.; Grothaus, P.G.; Newman, D.J. ‘Impact of natural products on developing new anti-cancer agents’. Chemical Reviews 2009, 109(7), 3012–3043. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M. ‘Natural products as sources of new drugs over the nearly four decades’. Journal of Natural Products 2020, 83(3), 770–803. [Google Scholar] [CrossRef] [PubMed]
- Atanasov, A.G.; Zotchev, S.B.; Dirsch, V.M.; Supuran, C.T. ‘Natural products in drug discovery: advances and opportunities’. Nature Reviews Drug Discovery 2021, 20, 200–216. [Google Scholar] [CrossRef] [PubMed]
- Polonsky, J. ‘Quassinoid bitter principles’. Progress in the Chemistry of Organic Natural Products 1985, 47, 221–264. [Google Scholar] [PubMed]
- Kuo, P.C.; Damu, A.G.; Lee, K.H.; Wu, T.S. ‘Cytotoxic and antimalarial quassinoids from the fruits of Brucea javanica’. Journal of Natural Products 2004, 67(3), 442–446. [Google Scholar]
- Rahman, S.; Fukamiya, N.; Tokuda, H.; Nishino, H. ‘Quassinoids from Brucea javanica and their biological activities’. Phytochemistry Reviews 2014, 13(4), 755–768. [Google Scholar]
- Zhao, Y.; Li, Y.; Wang, J.; Chen, X. ‘Recent advances in the chemistry and pharmacology of quassinoids’. Bioorganic & Medicinal Chemistry Letters 2016, 26(8), 2010–2014. [Google Scholar]
- Batista, R.; Silva, A.J.; Oliveira, A.B.; Braga, F.C. ‘In vitro and in vivo antimalarial activity of plant extracts and quassinoids’. Phytomedicine 2009, 16(11), 1031–1037. [Google Scholar]
- Bero, J.; Frédérich, M.; Quetin-Leclercq, J. ‘Antimalarial compounds isolated from plants’. Journal of Pharmacy and Pharmacology 2009, 61, 1401–1433. [Google Scholar] [CrossRef] [PubMed]
- Cragg, G.M.; Newman, D.J. ‘Natural products: a continuing source of novel drug leads’. Biochimica et Biophysica Acta – General Subjects 2013, 1830(6), 3670–3695. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.H.; Imakura, Y.; Sumida, Y. ‘Structure and chemical reactions of quassinoids’. Journal of Organic Chemistry 1979, 44(12), 2180–2185. [Google Scholar] [CrossRef]
- Ohno, N.; Fukamiya, N.; Lee, K.H. ‘Structure–activity relationships of quassinoids and related compounds’. Bioorganic & Medicinal Chemistry 1997, 5(8), 1489–1494. [Google Scholar]
- Zhang, H.; Tan, G.T.; Santarsiero, B.D.; Mesecar, A.D.; Van Hung, N.; Cuong, N.M.; Soejarto, D.D.; Pezzuto, J.M. ‘New quassinoids from Brucea javanica’. Journal of Natural Products 2003, 66(7), 903–907. [Google Scholar]
- Greene, T.W.; Wuts, P.G.M. Protective Groups in Organic Synthesis, 4th edn; Hoboken: Wiley, 2007. [Google Scholar]
- Hitotsuyanagi, Y.; Fukaya, H.; Takeya, K. ‘Quassinoid derivatives and their biological activities’. Journal of Natural Products 2006, 69(3), 451–456. [Google Scholar]
- Carey, F.A.; Sundberg, R.J. Advanced Organic Chemistry Part B: Reactions and Synthesis, 5th edn; Springer: New York, 2007. [Google Scholar]
- Otera, J.; Nishikido, J. Esterification: Methods, Reactions, and Applications; Hoboken: Wiley, 2010. [Google Scholar]
- Sundberg, R.J. ‘Imidazole-mediated acylation reactions’. Tetrahedron 1996, 52(26), 8619–8652. [Google Scholar]
- Lee, K.H.; Lin, Y.M.; Wu, T.S.; Hall, I.H. ‘Structure–activity relationships of quassinoids and their semisynthetic derivatives’. Journal of Medicinal Chemistry 1987, 30(6), 1099–1104. [Google Scholar]
- Wang, Y.; Chen, X.; Lee, K.H. ‘Anticancer quassinoids: chemistry and biology’. Current Medicinal Chemistry 2010, 17(6), 529–541. [Google Scholar]
- O’Neill, P.M.; Barton, V.E.; Ward, S.A. ‘The molecular mechanism of action of artemisinin—the debate continues’. Molecules 2010, 15, 1705–1721. [Google Scholar] [CrossRef] [PubMed]
- Cragg, G.M.; Newman, D.J. ‘Natural products as sources of new drugs from 1981 to 2014’. Journal of Natural Products 2016, 79(3), 629–661. [Google Scholar] [CrossRef] [PubMed]
- Tenda, P. E.; Solikhah, E. N.; Wijayanti, M. A. In vitro antiplasmodial activity of Brucein A semisynthetic compounds. TMJ 2014, 2(1), 34–38. [Google Scholar]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).