1. Introduction
Hidradenitis suppurativa (HS) is a chronic inflammatory dermatosis characterized by deep-seated nodules and abscesses [
1,
2]. Among its clinical manifestations, pain is consistently identified as the most frequent and debilitating symptom, reported by over 95% of patients [
2]. Consequently, the disease imposes a profound impairment on patient quality of life, yielding dermatology life quality index (DLQI) scores significantly higher than those reported for other severe skin diseases, such as psoriasis and acne [
3].
The unique morbidity of HS stems directly from the anatomical localization of these lesions. The disease predominantly affects intertriginous zones, specifically the axillary, inguinal, gluteal, and perineal regions [
1]. Crucially, these areas function as the primary anatomical hinges essential for basic human locomotion. Because these hinges must continuously stretch and compress, ordinary daily movement generates friction and shearing forces across the inflamed tissue.
Furthermore, the pain generated by this mechanical stress is uniquely severe. HS pain is not exclusively nociceptive; chronic inflammation induces peripheral neuroplastic changes and central sensitization, resulting in a profound neuropathic pain profile [
2]. Clinical literature confirms that patients experience mechanical allodynia and hyperalgesia, frequently describing the pain as a burning, cutting, or pressing soreness [
3]. In this neuropathic state, even minor, non-threatening skin tension from walking or reaching registers to the central nervous system as severe pain stimuli.
This biomechanical reality fundamentally alters patient behavior. To prevent mechanically triggered allodynia, the initial, logical biological reflex is to cease movement. However, as the disease progresses, this protective reflex conditions the central nervous system to associate normal locomotion with agonizing pain, establishing the foundation for a profound psychological barrier.
2. Translating the Fear-Avoidance Model to Hidradenitis Suppurativa
In musculoskeletal rehabilitation, the transition from acute protective reflexes to chronic disability is explained by the fear-avoidance model [
4]. This model posits that a maladaptive cognitive appraisal of pain triggers kinesiophobia, defined as an excessive and irrational fear of movement due to a perceived vulnerability to re-injury [
4]. Kinesiophobia is a highly specific and measurable clinical entity, robustly quantified in chronic pain populations using the validated Tampa Scale for Kinesiophobia (TSK) [
5].
This psychological framework translates seamlessly to the biomechanical reality of HS. Qualitative studies demonstrate that HS patients spend extensive cognitive and physical energy anticipating and compensating for pain [
6]. Because ordinary movements such as reaching, walking, turning a steering wheel, or typing directly activate nerves in the lesions, patients actively avoid even minor postural changes [
6]. While initially an acute biological necessity during unpredictable disease flares, this anticipatory fear of movement may evolve into a chronic psychological barrier contributing to a sedentary lifestyle and driving a pro-inflammatory state. Recent epidemiological literature explicitly recognizes this phenomenon, hypothesizing a clinical cycle of pain-induced immobility, kinesiophobia, and severe physical deconditioning in patients with HS [
7].
Adjacent medical specialties increasingly recognize kinesiophobia as a critical amplifier of pain and disability in systemic inflammatory conditions like spondyloarthritis [
8] and psoriatic arthritis, where kinesiophobia afflicts nearly half of patients, directly driving central sensitization and functional impairment independent of inflammatory disease activity [
9]. While HS is among the dermatological conditions most strongly associated with mobility-related pain, standard tools like the Dermatology Life Quality Index (DLQI) and Patient Health Questionnaire-9 (PHQ-9) do not currently account for kinesiophobia [
6]. Kinesiophobia remains a critically unmeasured metric in the HS patient population. If actively identified, kinesiophobia could be mitigated using established musculoskeletal interventions, such as graded exposure therapy and cognitive-behavioral rehabilitation [
4]. However, left unchecked, this psychological avoidance and fear of movement may force the patient into sustained immobility, initiating a secondary cascade of physical deconditioning and metabolic dysfunction [
4,
6].
3. Behavior and Lifestyle Positive Feedback Loops: A Macroscopic Framework
HS pathophysiology operates through a complex network of positive feedback loops that continuously converge to amplify systemic inflammation, the primary biological driver of the disease [
10,
11]. To date, dermatological literature has predominantly characterized these feedback loops at the microscopic levels. For instance, existing models define a localized vicious cycle of chronic inflammation driven by innate immune hyperactivation, aberrant antimicrobial peptide expression, and local microbiome dysbiosis [
12,
13,
14]. Specifically, molecular models have mapped self-amplifying cycles where TNF-α induces the production of lipocalin-2 in neutrophils, which in turn stimulates further TNF-α release and neutrophil chemotaxis, locking the local tissue in a state of chronic suppuration [
13].
We propose that this established pathophysiological paradigm can be expanded macroscopically. While the profound psychosocial and socioeconomic burdens of HS are individually well-documented across the literature, these downstream consequences have not been integrated in a single model as active mechanisms of disease pathogenesis. By combining known behavioral maladaptations with kinesiophobia, we construct a comprehensive macroscopic feedback network reflecting the burdens on the HS patient population. Rather than replacing localized microbiological models, this network approach complements them by illustrating how the behavioral, psychological, and socioeconomic adaptations to the disease structurally feed back into the systemic inflammatory core, functioning as interconnected positive feedback loops.
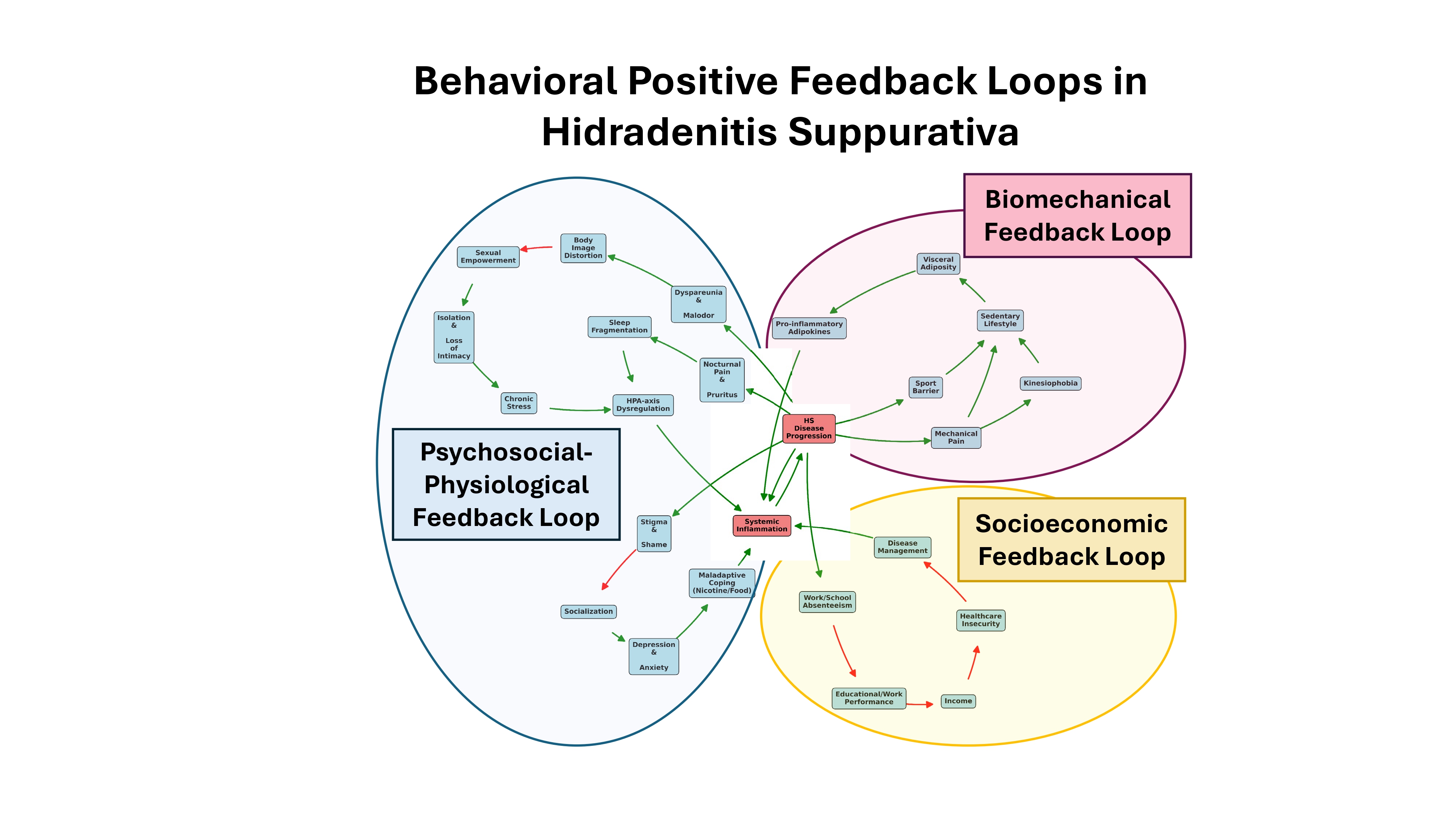
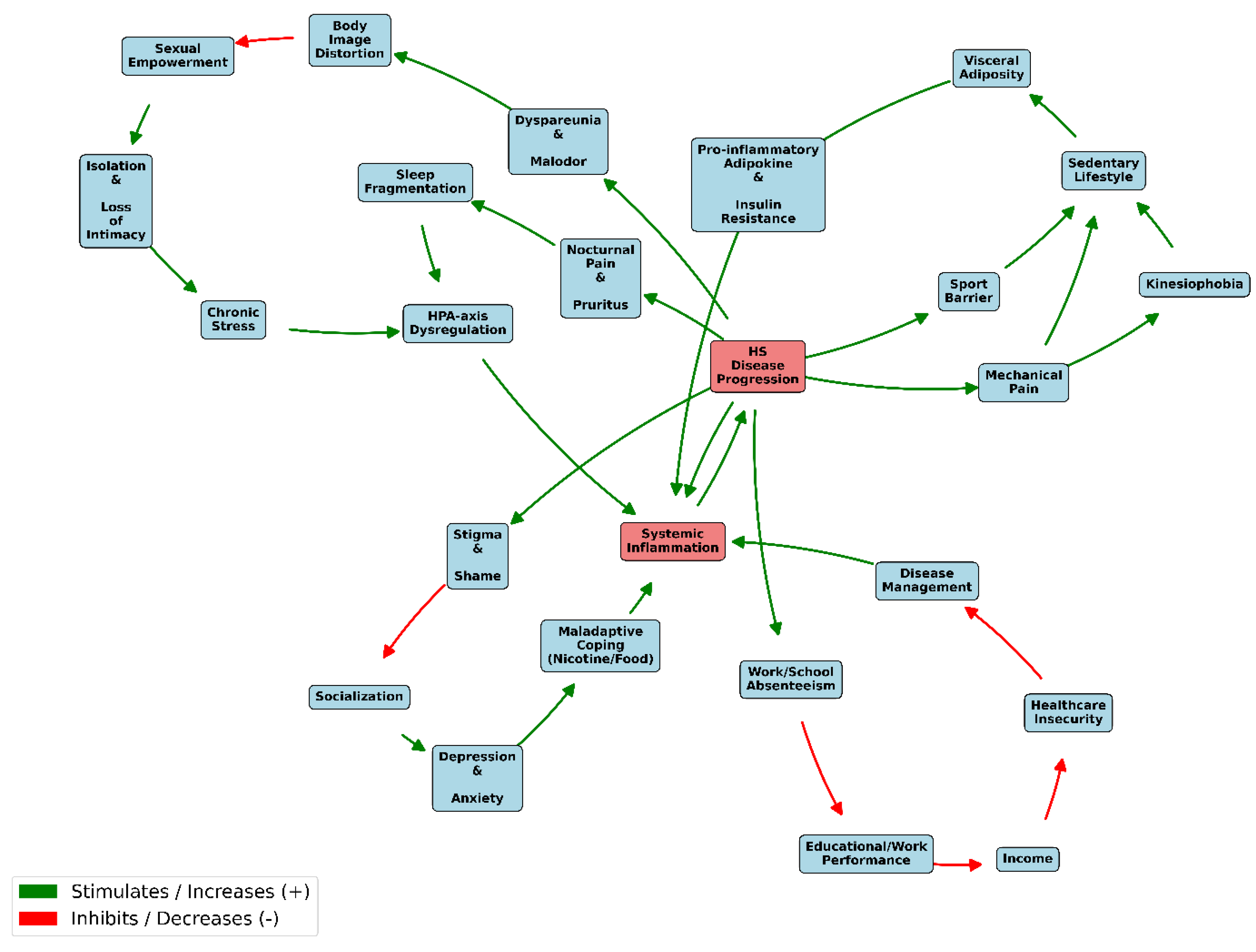
As illustrated in
Figure 1, we hypothesize that this self-sustaining cycle operates through three broad, interacting domains: (1) a biomechanical-metabolic loop, where pain-driven inactivity accelerates visceral adiposity and systemic metabolic dysfunction, including insulin resistance; (2) a psychosocial and physiological loop, where severe stigma, loss of intimacy, and sleep fragmentation collectively drive chronic stress, hypothalamic-pituitary-adrenal (HPA)-axis dysregulation, and maladaptive coping; and (3) a socioeconomic loop, where disease-related absenteeism leads to sustained healthcare insecurity.
3.1. The Biomechanical-Metabolic Feedback Loop
The most direct physical consequence of kinesiophobia is a profound reduction in mobility. While factors such as pain, active suppuration, malodor, and generalized physiological burden inherently inhibit regular exercise [
15,
16], we postulate that kinesiophobia may also act as the psychological stimulus contributing to infrequent exercise and a chronically sedentary lifestyle in HS patients. This sustained physical inactivity accelerates the development of visceral adiposity and drives a systemic decline in insulin sensitivity [
17]. Adipose tissue increases the systemic secretion of pro-inflammatory adipokines while simultaneously downregulating anti-inflammatory mediators [
18]. Furthermore, the resulting insulin resistance induces compensatory hyperinsulinemia, a state known to stimulate androgen-mediated epidermal hyperplasia and follicular hyperkeratosis [
19]. Because these adipokine and metabolic disruptions upregulate the exact pathways responsible for HS lesion formation, the psychological fear of movement perpetuates the biological chronicity of the disease by sustaining a pro-inflammatory metabolic state.
3.2. The Psychosocial-Physiological Feedback Loop
This metabolic cascade is accompanied by a concurrent neuroendocrine feedback loop. The physical realities of active HS include pain, suppuration, dyspareunia, and malodor which together precipitate anxiety, body image distortion, social stigma, and the loss of socialization and sexual intimacy [
16,
20,
21,
22,
23,
24]. These factors promote chronic psychological stress, which biologically dysregulates the HPA axis, elevating baseline systemic inflammatory markers [
25].
Moreover, the disease disrupts the foundational pillars of physiological recovery. HS profoundly impairs sleep architecture, primarily driven by disease-specific pain and severe pruritus [
26,
27,
28]. Clinical HS literature establishes pain and pruritus as a primary driver of prolonged sleep latency and frequent nocturnal awakenings [
27]. The specific biomechanical mechanisms underlying this disruption are likely that deep-seated nodules predominantly afflict intertriginous and weight-bearing regions, and mechanical allodynia [
3] severely restricts comfortable resting postures. This underexplored positional pain may also act as a direct, physical barrier to sleep onset and continuity. Crucially, this chronic sleep deprivation functions as an active pathophysiological stressor rather than a mere symptom. Insufficient sleep disrupts circadian homeostasis and upregulates the systemic production of pro-inflammatory cytokines, tumor necrosis factor-alpha (TNF-alpha) and interleukin-6 (IL-6) [
25]. This establishes a positive feedback loop where HS-driven pain and pruritus impair sleep, and the resulting sleep loss fuels the systemic inflammation promoting further disease progression [
25].
Furthermore, the physiological exhaustion resulting from chronic sleep loss critically depletes cognitive reserves, which directly exacerbates baseline emotional distress, anxiety, and depressive symptoms [
26]. To mitigate this compounding psychological and physiological fatigue, patients frequently resort to maladaptive coping behaviors, most notably nicotine consumption and high-glycemic emotional eating [
11]. These behaviors act as active vectors for disease progression. Nicotine stimulates specific receptors in the follicular infundibulum, promoting epidermal thickening as well as chemotaxis and neutrophil extravasation [
11]. Concurrently, a reliance on high-glycemic diets induces rapid spikes in blood glucose and compensatory hyperinsulinemia [
19]. This diet-induced loss of insulin sensitivity is deeply pathogenic. Chronic hyperinsulinemia directly amplifies the systemic inflammatory pool and stimulates androgen-driven sebaceous gland overactivity [
11,
19]. Additionally, insulin resistance and obesity disrupt metabolic homeostasis by promoting the continuous release of pro-inflammatory cytokines from adipose tissue, reinforcing a hostile systemic environment [
18].
3.3. The Socioeconomic Feedback Loop
Simultaneously, the severe physical and psychological disabilities imposed by HS drive a profound and measurable socioeconomic deterioration [
29]. The compounded burden of chronic pain, immobility, and generalized physiological exhaustion fundamentally compromises educational and professional capability. Clinical evaluations utilizing validated productivity metrics demonstrate that HS patients experience substantial rates of both absenteeism (missed work) and presenteeism (reduced productivity while at work) [
22,
30]. As inflammatory disease severity increases, overall work productivity and the capacity to perform essential daily activities decline markedly [
30].
This chronic professional disruption translates directly into long-term economic impairment. Compared to healthy demographics, patients with HS experience significantly hindered income trajectories, incur substantial indirect disease-related costs, and suffer disproportionately high rates of both unemployment and reliance on long-term disability support [
22,
31].
Crucially, we suggest that this socioeconomic decline establishes a barrier to physiological recovery. The progressive reduction in personal income directly restricts a patient's capacity to afford optimal and supplementary healthcare and consequently hinders effective disease management. Lacking the financial agency to access the clinical interventions required to disrupt these interconnected behavioral, neuroendocrine, and socioeconomic feedback loops, the patient is progressively drawn into a self-sustaining cycle of uncontrolled systemic inflammation.
4. Future Directions and Conclusions
The conceptualization of hidradenitis suppurativa must evolve from a focus on localized and systemic inflammation to include a complex, systemic network of behavioral, physiological, and socioeconomic feedback loops. While advanced biologic therapies and surgical interventions remain the foundational cornerstones of HS management, these pharmaceuticals are inherently limited if a patient remains structurally bound to a cycle of psychological, physical and socioeconomic decline. By recognizing maladaptive behavioral responses as active continuous drivers of systemic inflammation, clinicians can better understand the persistent chronicity of the disease and implement the appropriate treatments or referrals to address these key aspects of the disease. Consequently, optimal disease management requires a fundamental shift toward multidisciplinary care.
For kinesiophobia in HS, routine dermatological assessments must expand beyond standard quality-of-life indices to incorporate validated musculoskeletal and psychological screening tools, most notably the Tampa Scale for Kinesiophobia (TSK) [
5]. Characterizing this anticipatory fear of movement opens an interesting new research frontier and introduces a potential novel therapeutic target for HS management. Patients exhibiting high kinesiophobia scores should be proactively integrated into structured musculoskeletal rehabilitation programs. Utilizing established interventions such as cognitive-behavioral therapy and graded exposure therapy [
4] can effectively dismantle the psychological barrier to movement. Reversing this immobility is biologically imperative to prevent the secondary cascade of visceral adiposity, hyperinsulinemia, and subsequent inflammatory amplification.
To clinically validate this kinesiophobia, future research must prioritize prospective epidemiological and interventional trials. Initial observational studies should focus on quantifying the baseline prevalence and severity of kinesiophobia across diverse HS cohorts, explicitly correlating these behavioral metrics with systemic inflammatory markers and established disease severity staging. Furthermore, interventional studies are required to determine whether targeted physical rehabilitation and psychological desensitization to movement can biologically reduce the baseline systemic pro-inflammatory pool and disease severity.
Ultimately, investigating whether actively disrupting these behavioral feedback loops enhances the clinical efficacy and longevity of existing biologic therapies will be paramount in establishing comprehensive, disease-modifying treatment protocols for hidradenitis suppurativa. HS patients frequently experience delayed diagnosis, ineffective treatments, undertreatment of psychological and psychiatric comorbidities, as well as a lack of education regarding effective lifestyle modifications, ultimately leading to medical alienation. To restore patient trust in the healthcare system, greater emphasis must be placed on describing, quantifying and treating the neglected dimensions of the disease. From a broader philosophical and clinical perspective, it would be highly valuable to map the additional positive feedback loops in HS and develop an expanded network that combines both microscopic (molecular) and macroscopic (behavioral) feedback loops. While the macroscopic framework presented in this study compartmentalizes these burdens into three distinct modules, they are likely far more intricately interconnected in clinical reality. Moreover, it is essential that future research aim to expand upon the macroscopic network proposed in this study. The three distinct behavioral feedback loops identified within this review could be mathematically translated and integrated into existing ordinary differential equation frameworks such as the inflammatory flux model for lifestyle and pharmacotherapy in hidradenitis suppurativa [
10]. By undertaking this integration, we will be able to more comprehensively and accurately simulate the profound dynamic complexity of the disease.
Supplementary Materials
The following supporting information can be downloaded at the website of this paper posted on
Preprints.org.
Conflicts of Interest
The author declares no conflicts of interest.
Funding
This research received no external funding.
Abbreviations
DLQI, Dermatology Life Quality Index
HPA, hypothalamic-pituitary-adrenal
HS, Hidradenitis suppurativa
IL-6, interleukin-6
PHQ-9, Patient Health Questionnaire-9
TNF-alpha, tumor necrosis factor-alpha
TSK, Tampa Scale for Kinesiophobia
References
- Szepietowska, M.; Krajewski, P.K.; Pacan, P.; Wojas-Pelc, A.; Matusiak, L.; Jaworek, A.K. A Cross-Sectional Study on Relationships Between Depression and Anxiety in Hidradenitis Suppurativa Patients and Disease Severity, Subjective Symptoms and Quality of Life. J. Clin. Med. 2026, 15, 700. [Google Scholar] [CrossRef] [PubMed]
- Montero-Vilchez, T.; Diaz-Calvillo, P.; Rodriguez-Pozo, J.-A.; Cuenca-Barrales, C.; Martinez-Lopez, A.; Arias-Santiago, S.; Molina-Leyva, A. The Burden of Hidradenitis Suppurativa Signs and Symptoms in Quality of Life: Systematic Review and Meta-Analysis. Int. J. Environ. Res. Public. Health 2021, 18, 6709. [Google Scholar] [CrossRef] [PubMed]
- Ring, H.C.; Sørensen, H.; Miller, I.M.; List, E.K.; Saunte, D.M.; Jemec, G.B. Pain in Hidradenitis Suppurativa: A Pilot Study. Acta Derm. Venereol. 2016, 96, 554–556. [Google Scholar] [CrossRef] [PubMed]
- Vlaeyen, J.W.S.; Linton, S.J. Fear-avoidance and its consequences in chronic musculoskeletal pain: A state of the art. Pain 2000, 85, 317–332. [Google Scholar] [CrossRef]
- French, D.J.; France, C.R.; Vigneau, F.; French, J.A.; Evans, R.T. Fear of movement/(re)injury in chronic pain: A psychometric assessment of the original English version of the Tampa scale for kinesiophobia (TSK). Pain 2007, 127, 42–51. [Google Scholar] [CrossRef]
- Orenstein, L.A.V.; Salame, N.; Siira, M.R.; Urbanski, M.; Flowers, N.I.; Echuri, H.; Garg, A.; McKenzie-Brown, A.M.; Curseen, K.A.; Patzer, R.E.; Kavalieratos, D.; Chen, S.C. Pain experiences among those living with hidradenitis suppurativa: A qualitative study. Br. J. Dermatol. 2023, 188, 41–51. [Google Scholar] [CrossRef]
- Lu, H.-Y.; Su, Y.-J.; Chang, H.-C.; Gau, S.-Y. Risk of fracture in patients with hidradenitis suppurativa: A multi-center retrospective cohort study. ResearchSquare 2025. [Google Scholar] [CrossRef]
- Capparelli, E.; Iacovantuono, M.; Vescovo, S.D.; Monosi, B.; Bonini, C.; Fiannacca, L.; Greco, E.; Cela, E.; Conigliaro, P.; Dipietrangelo, G.G.; Iannone, F.; Lopalco, G.; Chimenti, M.S. Kinesiophobia as part of the psychological burden in Spondyloarthritis: A case–control study. Rheumatol. Adv. Pract 2025. [Google Scholar] [CrossRef]
- Llamas-Ramos, R.; Llamas-Ramos, I.; Alvarado-Omenat, J.J.; Toledano, E.; Queiro, R.; Martin, J.; Chacón, C.C.; Díaz-Peña, R.; Martín, D.; Calleja, C.H.; Sánchez, M.D.; Montilla, C. Prevalence and Predictors of Kinesiophobia in Psoriatic Arthritis: The Role of Central Sensitization and comorbidities. Front. Med. 2026, 13. [Google Scholar]
- Roukens, J.-J. Inflammatory Flux and Disease Progression in Hidradenitis Suppurativa: A Multi-Compartment Deterministic Model Simulating Lifestyle and Pharmaceutical Interventions in In Silico Cohorts. Preprints.Org 2026. [Google Scholar] [CrossRef]
- Sabat, R.; Alavi, A.; Wolk, K.; Wortsman, X.; McGrath, B.; Garg, A.; Szepietowski, J.C. Hidradenitis suppurativa. The Lancet 2025, 405, 420–438. [Google Scholar] [CrossRef] [PubMed]
- Chopra, D.; Arens, R.A.; Amornpairoj, W.; Lowes, M.A.; Tomic-Canic, M.; Strbo, N.; Lev-Tov, H.; Pastar, I. Innate immunity and microbial dysbiosis in hidradenitis suppurativa–vicious cycle of chronic inflammation. Front. Immunol. 2022, 13. [Google Scholar] [CrossRef] [PubMed]
- Mössner, R. New insights into hidradenitis suppurativa: Is lipocalin-2 acting as an amplifier? Br. J. Dermatol. 2017, 177, 1162–1164. [Google Scholar] [CrossRef] [PubMed]
- Yu, W.-W.; Barrett, J.N.P.; Tong, J.; Lin, M.-J.; Marohn, M.; Devlin, J.C.; Herrera, A.; Remark, J.; Levine, J.; Liu, P.-K.; Fang, V.; Zellmer, A.M.; Oldridge, D.A.; Wherry, E.J.; Lin, J.-R.; Chen, J.-Y.; Sorger, P.; Santagata, S.; Krueger, J.G.; Ruggles, K.V.; Wang, F.; Su, C.; Koralov, S.B.; Wang, J.; Chiu, E.S.; Lu, C.P. Skin immune-mesenchymal interplay within tertiary lymphoid structures promotes autoimmune pathogenesis in hidradenitis suppurativa. Immunity 2024, 57, 2827–2842.e5. [Google Scholar] [CrossRef]
- Zinko, O.; Buczyńska-Dymicka, K.; Bajek, M.; Puchalski, K.; Polakowska, I.; Urban-Michalak, K.; Bigdoń, A.; Pietrzak-Chmiel, M.M.; Malinowski, K.; Gosztyła, D. Quality of Life and Exercise Adherence in Individuals with Chronic Inflammatory Skin Diseases: A Narrative Review. Qual. Sport 2026, 50, 67932. [Google Scholar] [CrossRef]
- Ureña-Paniego, C.; Soto-Moreno, A.; Díaz-Calvillo, P.; Cuenca-Barrales, C.; Molina-Leyva, A.; Arias-Santiago, S. Impact of Hidradenitis Suppurativa on Major Life-Changing Decisions: A Cross-Sectional Study. Indian J. Dermatol. 2025, 70, 283–286. [Google Scholar] [CrossRef]
- Amati, F.; Dubé, J.J.; Coen, P.M.; Stefanovic-Racic, M.; Toledo, F.G.S.; Goodpaster, B.H. Physical Inactivity and Obesity Underlie the Insulin Resistance of Aging. Diabetes Care 2009, 32, 1547–1549. [Google Scholar] [CrossRef]
- Kirichenko, T.V.; Markina, Y.V.; Bogatyreva, A.I.; Tolstik, T.V.; Varaeva, Y.R.; Starodubova, A.V. The Role of Adipokines in Inflammatory Mechanisms of Obesity. Int. J. Mol. Sci. 2022, 23, 14982. [Google Scholar] [CrossRef]
- González-Saldivar, G.; Rodríguez-Gutiérrez, R.; Ocampo-Candiani, J.; González-González, J.G.; Gómez-Flores, M. Skin Manifestations of Insulin Resistance: From a Biochemical Stance to a Clinical Diagnosis and Management. Dermatol. Ther. 2017, 7, 37–51. [Google Scholar] [CrossRef]
- Cohn, E.; Palma, G.; Mastacouris, N.; Strunk, A.; Garg, A. Incidence of anxiety disorder in adults with hidradenitis suppurativa. Br. J. Dermatol. 2024, 191, 351–356. [Google Scholar] [CrossRef]
- Perche, P.O.; Singh, R.; Senthilnathan, A.; Feldman, S.R.; Pichardo, R.O. Hidradenitis Suppurativa’s Impact on Social Activities: An Observational Study. Cureus 2022, 14, e25292. [Google Scholar] [CrossRef] [PubMed]
- Schneider-Burrus, S.; Jost, A.; Peters, E.M.J.; Witte-Haendel, E.; Sterry, W.; Sabat, R. Association of Hidradenitis Suppurativa With Body Image. JAMA Dermatol 2018, 154, 447–451. [Google Scholar] [CrossRef]
- Caccavale, S.; Tancredi, V.; Boccellino, M.P.; Babino, G.; Fulgione, E.; Argenziano, G. Hidradenitis Suppurativa Burdens on Mental Health: A Literature Review of Associated Psychiatric Disorders and Their Pathogenesis. Life 2023, 13, 189. [Google Scholar] [CrossRef] [PubMed]
- Patel, K.R.; Lee, H.H.; Rastogi, S.; Vakharia, P.P.; Hua, T.; Chhiba, K.; Singam, V.; Silverberg, J.I. Association between hidradenitis suppurativa, depression, anxiety, and suicidality: A systematic review and meta-analysis. J. Am. Acad. Dermatol. 2020, 83, 737–744. [Google Scholar] [CrossRef]
- Nunez, S.G.; Rabelo, S.P.; Subotic, N.; Caruso, J.W.; Knezevic, N.N. Chronic Stress and Autoimmunity: The Role of HPA Axis and Cortisol Dysregulation. Int. J. Mol. Sci. 2025, 26, 9994. [Google Scholar] [CrossRef] [PubMed]
- Yeroushalmi, S.; Ildardashty, A.; Elhage, K.G.; Chung, M.; Bartholomew, E.; Hakimi, M.; Tahir, P.; Naik, H.B.; Bhutani, T.; Liao, W. Hidradenitis suppurativa and sleep: A systematic review. Arch. Dermatol. Res. 2023, 315, 1409–1415. [Google Scholar] [CrossRef]
- Kaaz, K.; Szepietowski, J.C.; Matusiak, Ł. Influence of Itch and Pain on Sleep Quality in Patients with Hidradenitis Suppurativa. Acta Derm. Venereol. 2018, 98, 757–761. [Google Scholar] [CrossRef]
- Chung, C.S.; Kherallah, K.; Fragoso, N.M.; Daveluy, S.; Hsiao, J.L.; Lee, K.H. Exploring the Pathophysiology, Burden, and Management of Itch in Hidradenitis Suppurativa: A Narrative Review. Curr. Dermatol. Rep. 2025, 14, 35. [Google Scholar] [CrossRef]
- Regensberger, F.; ANDRÉ, F.; MAIER, S.; POSCH, C.; SCHMUTH, M. The Hidden Socioeconomic Toll of Severe Hidradenitis Suppurativa. Acta Derm. Venereol. 2025, 105, 44495. [Google Scholar] [CrossRef]
- Yao, Y.; Jørgensen, A.-H.R.; Thomsen, S.F. Work productivity and activity impairment in patients with hidradenitis suppurativa: A cross-sectional study. Int. J. Dermatol. 2020, 59, 333–340. [Google Scholar] [CrossRef]
- Tzellos, T.; Yang, H.; Mu, F.; Calimlim, B.; Signorovitch, J. Impact of hidradenitis suppurativa on work loss, indirect costs and income. Br. J. Dermatol. 2019, 181, 147–154. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).