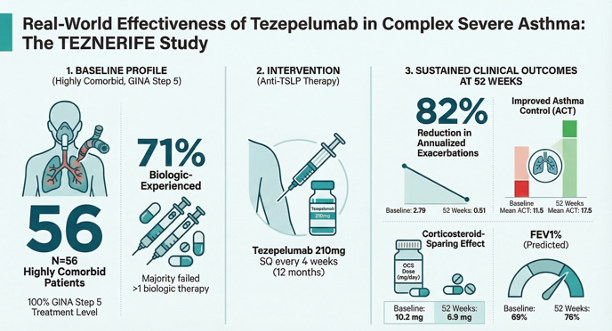
Background/Objectives: Severe asthma in routine practice often involves long-standing disease, multimorbidity, and prior biologic failure, settings underrepresented in pivotal tezepelumab trials. This study evaluated 52 week real world effectiveness and safety of tezepelumab in a highly comorbid, predominantly T2 high, biologic experienced severe asthma cohort from the Canary Islands. Methods: TEZNERIFE is a multicenter, retrospective phase IV study including consecutive adolescents and adults with GINA Step 5 severe uncontrolled asthma treated with tezepelumab 210 mg every 4 weeks for 12 months. Clinical outcomes, lung function, type 2 biomarkers, upper airway symptoms, and Biologics Asthma Response Score (BARS) were assessed at baseline, 26 weeks, and 52 weeks. Results: Fifty six patients (mean age 53.5 years, 71% female, mean asthma duration 30 years, 84% T2 high; 71% with ≥1 prior biologic) were analyzed. ACT improved from 11.5±3.7 to 15.9±4.7 at 26 weeks and 17.5±4.7 at 52 weeks (both p<0.0001), while annualized exacerbations declined from 2.79±2.0 to 0.50±0.72 and 0.51±0.89 (both p<0.0001). Maintenance oral corticosteroid dose fell from 10.2±8.3 to 6.9±2.4 mg/day at 52 weeks (p=0.014). FEV1% predicted increased from 69.3±19.2% to 75.3±17.7% and 76.2±20.6% (p=0.004 and p=0.001), and blood eosinophils decreased from 234±231 to 146±120 and 147±110 cells/µL (p=0.001 and p=0.013). At one year, 18.9% and 67.9% were classified as good and intermediate responders by BARS; 13.2% were insufficient responders. Two patients discontinued due to non serious adverse events while no treatment related serious events occurred. Conclusions: In this difficult to treat, multimorbid, biologic experienced population, tezepelumab achieved sustained improvements in asthma control, exacerbations, lung function, eosinophilic inflammation, and corticosteroid exposure over 52 weeks, supporting upstream alarmin inhibition as a versatile strategy in complex severe asthma.