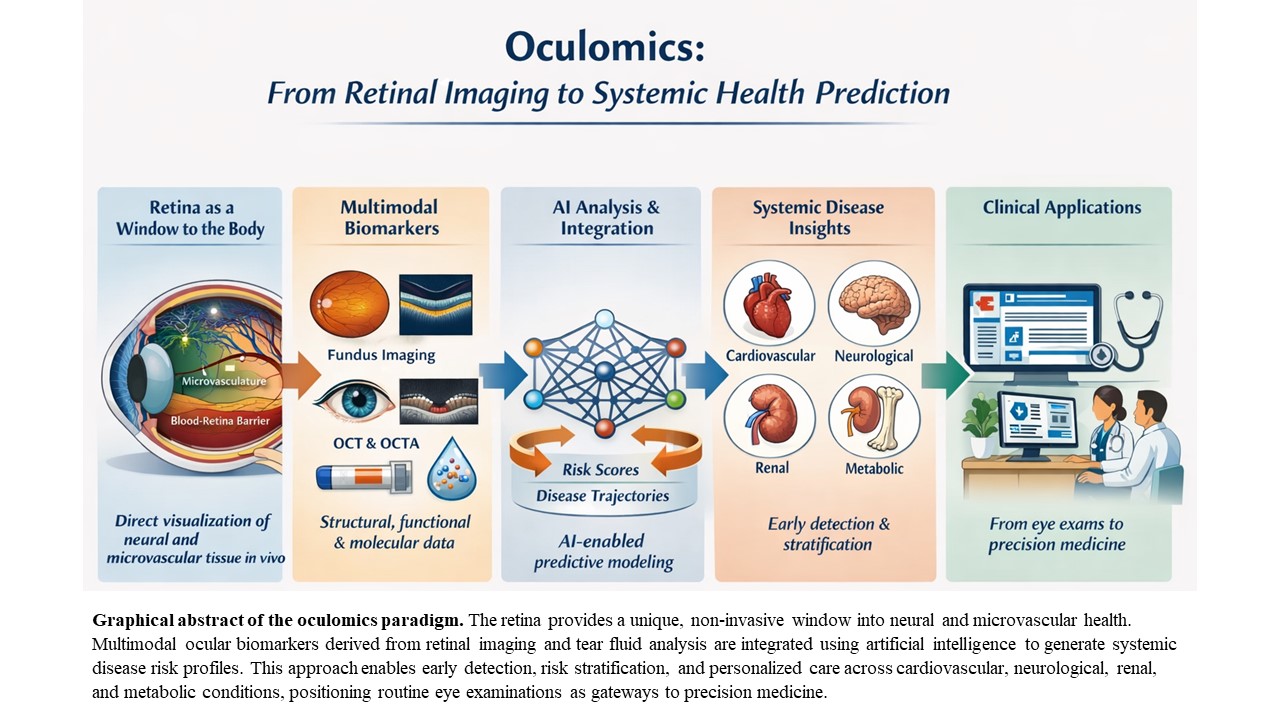
1. Introduction: What Is Oculomics?
For decades, ophthalmology has largely conceptualized ocular involvement as a downstream consequence of systemic disease. Neurological, metabolic, cardiovascular, and inflammatory disorders were known to increase the risk of ocular pathology, with retinal, optic nerve, and microvascular alterations interpreted primarily as secondary manifestations of broader systemic dysfunction [
1]. Within this traditional framework, the eye was regarded mainly as a
target organ—valuable for diagnosing local disease and monitoring complications, but rarely considered a source of primary, predictive information about systemic health.
This view is now being fundamentally challenged. Advances in ocular imaging, data science, and systems medicine have revealed that many systemic diseases leave quantifiable signatures in ocular tissues well before the onset of overt clinical symptoms elsewhere in the body. Oculomics emerges from this realization and inverts the classical question: rather than asking how systemic disease affects the eye, oculomics asks how the eye can be used to detect, stratify, and even predict systemic disease risk. This inversion is possible because the eye, and the retina in particular, shares fundamental embryological, anatomical, and physiological properties with the brain and systemic vasculature, making it a sensitive biosensor for early pathological change. The biological basis for this connection is detailed in the following section.
Oculomics capitalizes on this unique biological positioning and on the exceptional accessibility of the eye. Unlike other organs, ocular tissues can be examined non-invasively, repeatedly, and at high spatial resolution using widely available technologies such as color fundus photography, optical coherence tomography (OCT), and OCT angiography (OCTA). These modalities enable direct visualization and quantification of neural and microvascular structures in vivo, offering a practical and scalable window into systemic pathophysiology [
2,
3].
Formally, oculomics can be defined as an emerging interdisciplinary field that leverages ocular imaging and ocular-derived biological data to identify biomarkers of systemic health and disease. It integrates ophthalmology with omics sciences—including genomics, proteomics, metabolomics, and exposomics—and with advanced artificial intelligence (AI) analytics to transform the eye into a non-invasive biosensor of whole-body health [
2,
4,
5]. In this framework, ocular features are no longer interpreted in isolation but are analyzed as part of interconnected biological networks reflecting systemic function and dysfunction.
The rapid maturation of oculomics has been enabled by three converging developments. First, high-resolution multimodal ocular imaging has become routine in clinical practice, generating large volumes of standardized quantitative data. Second, population-scale datasets—such as biobanks and longitudinal health record linkages—have made it possible to associate ocular features with long-term systemic outcomes. Third, AI-driven analytical frameworks, particularly deep learning models, have demonstrated the capacity to extract complex, disease-relevant patterns from ocular images that are imperceptible to human observers [
6,
7,
8].
Together, these advances have enabled the identification of retinal, choroidal, corneal, and tear-film features associated with cardiovascular disease, diabetes, neurodegenerative disorders, chronic kidney disease, inflammatory conditions, and even systemic aging trajectories—often years before clinical diagnosis in non-ocular organs [
3,
5,
9]. Importantly, these associations are not merely correlative; growing evidence suggests that ocular biomarkers capture fundamental microvascular and neurodegenerative processes that are central to systemic disease pathogenesis [
10,
11].
In this context, oculomics redefines the boundaries of ophthalmology. Routine eye examinations are no longer viewed solely as tools for preserving vision, but as strategic entry points for preventive, predictive, and personalized systemic medicine. This paradigm—recently conceptualized as “Healthcare from the Eye”—positions ophthalmic data as an integral component of modern healthcare ecosystems, enabling earlier risk stratification, targeted referral, and more efficient allocation of preventive interventions across medical specialties [
12].
Oculomics enters a growing ecosystem of non-invasive approaches for systemic health assessment, which includes voice analysis, skin imaging, wearables-derived digital phenotyping, and gut microbiome profiling [
13,
14,
15,
16]. While these modalities offer valuable insights—such as autonomic nervous system tone from voice [
13] or cardiovascular dynamics [
15]—oculomics possesses distinctive advantages. The retina provides the only direct, in vivo visualization of central nervous system neurons and the microvasculature at micrometer-scale resolution, offering anatomically anchored and biologically interpretable structural and functional readouts. Unlike wearables, which capture transient physiological states (e.g., heart-rate variability), retinal imaging captures cumulative signatures of microvascular remodeling and neurodegeneration [
17,
18,
19]. Furthermore, ocular imaging is already embedded in routine clinical workflows, is highly standardized, and generates rich, objective data inherently suited for quantitative analysis and AI integration [
20]. This combination of biological depth, clinical accessibility, and analytical scalability positions oculomics as a complementary yet uniquely powerful pillar within the future of multimodal, data-driven preventive medicine.
While recent publications [
3,
12] emphasize frameworks for integrating oculomics into coordinated healthcare delivery and address operational challenges, the present work advances the field by offering a mechanistic, evidence-based synthesis of quantitative retinal biomarkers derived from imaging and fluidomics, their underlying biological plausibility grounded in embryology and neurovascular continuity, and their specific associations with systemic pathophysiology. Unlike implementation-oriented perspectives, our manuscript maps phenotypes to mechanisms and provides a granular disease-centric blueprint for future translational and analytical research.
2. Why the Eye? The Biological Rationale
At its core, oculomics is based on the premise that ocular structures---particularly the retina---share deep embryological, neurovascular, and metabolic commonalities with the brain, heart, kidneys, and peripheral vasculature. The retina is a direct extension of the central nervous system, characterized by highly organized neural layers, dense microvascular networks, and tightly regulated blood--tissue barriers. As a result, pathological processes such as microvascular rarefaction, neurodegeneration, inflammation, and metabolic stress often manifest in the eye in ways that closely mirror systemic pathology [
2,
21,
22]. The fundamental premise of oculomics—that the eye holds unparalleled insights into systemic and neurological health—is not merely observational but deeply rooted in embryology, anatomy, and physiology. Against this backdrop, it becomes essential to explore the strong biological rationale supporting the field and to consider how modern, non-invasive technologies have repositioned the eye from a mere organ of vision to a powerful portal for systemic diagnosis.
2.1. The Eye as an Extension of the Brain: Shared Origins and Architecture
The intimate connection between the eye and the central nervous system (CNS) begins at the earliest stages of development. During embryogenesis, the optic vesicles evaginate directly from the diencephalon of the neural tube, meaning the retina is ontologically a part of the brain itself. This shared embryological origin establishes a permanent biological kinship [
23]. Consequently, the retina consists of specialized neurons—photoreceptors, bipolar cells, and ganglion cells—and supporting glial cells (Müller cells and astrocytes) that are direct analogues to those found within the cerebral cortex.
This neural continuity is mirrored in the microvascular architecture. The retinal vasculature shares characteristics with the cerebral vasculature, including similar endothelial cell structures, tight junction proteins, and regulatory mechanisms. Both systems possess specialized blood-tissue barriers—the blood-retina barrier (BRB) and the blood-brain barrier (BBB)—that tightly control molecular exchange to maintain a precise microenvironment essential for neural function. These barriers, while protective, often respond similarly to systemic insults such as hypertension, hyperglycemia, and inflammation [
24].
Critically, the retina exhibits analogous neurodegenerative processes. The retinal ganglion cell axons, which bundle together to form the optic nerve, are true CNS tracts, unmyelinated until they pass the lamina cribrosa. This makes them vulnerable to the same pathophysiological mechanisms that affect neurons in the brain [
25]. Research indicates that biomarkers for neuroinflammatory processes, amyloid-beta accumulation (associated with Alzheimer's disease), and neuronal apoptosis can be concurrently observed in the brain and the retina, offering a unique opportunity to study CNS disease in vivo [
21].
2.2. A Unique Diagnostic Window: Clarity and Accessibility
This unique anatomy is rendered accessible by the eye's transparent optical media—the cornea, aqueous humor, lens, and vitreous body. These structures function as a natural "body window," allowing light to pass unimpeded to the retina and back. This clarity enables the direct, in vivo visualization of living neural tissue and microcirculation at a cellular-level resolution that is unmatched in any other part of the human body without invasive procedures [
19]. This optical advantage is harnessed through a suite of standardized, rapid, and non-invasive imaging modalities that form the technological backbone of oculomics.
Color Fundus Photography (CFP) is a widely available, cost-effective technique that provides a two-dimensional
en face view of the retinal surface, capturing the optic disc, macula, and vascular tree. It is the cornerstone for detecting classic signs of systemic diseases, such as hypertensive retinopathy (arteriovenous nicking, hemorrhages) and diabetic retinopathy (microaneurysms, exudates) [
2].
Optical Coherence Tomography (OCT) is analogous to in vivo optical biopsy. OCT generates high-resolution, cross-sectional tomographic images of the retina. It allows for the precise quantification of individual retinal layer thicknesses. Critically, the thinning of the ganglion cell-inner plexiform layer (GC-IPL) and the retinal nerve fiber layer (RNFL) serves as a direct measure of neurodegeneration, relevant to conditions like Alzheimer's disease, Parkinson's disease, and multiple sclerosis [
26].
OCT Angiography (OCTA) is a functional extension of OCT. OCTA visualizes retinal and choroidal vasculature without the need for invasive dye injection. It can map the delicate capillary networks, including the foveal avascular zone (FAZ), and quantify metrics like vessel density and perfusion. This makes it exquisitely sensitive to early microvascular changes in diabetes, hypertension, and vascular cognitive impairment [
27].
2.3. The Oculomics Proposition: From Correlation to Causation
It is at the intersection of strong biological rationale and cutting-edge imaging technologies that the concept of oculomics truly emerges. Rather than serving as a passive mirror of systemic pathology, the eye functions as an active biological interface in which disease-related processes appear early and can be quantitatively captured. In many systemic disorders, retinal alterations precede overt clinical manifestations, positioning the eye as a uniquely valuable site for early detection and timely intervention [
22].
Furthermore, the quantitative, digital nature of these imaging biomarkers is ideal for the application of artificial intelligence (AI). Machine learning algorithms can detect subtle, subclinical patterns across vast image datasets, uncovering novel biomarkers beyond human perception. This enables large-scale screening, precise longitudinal monitoring of disease progression or therapeutic response, and the development of predictive models for personalized medicine [
2].
In summary, the biological rationale for oculomics rests on the eye's status as CNS tissue, while its technological rationale is built on non-invasive, high-resolution imaging. Together, they establish the retina as a powerful, accessible biosensor for systemic health and neurological integrity, paving the way for a transformative shift in preventive diagnostics and disease management [
21].
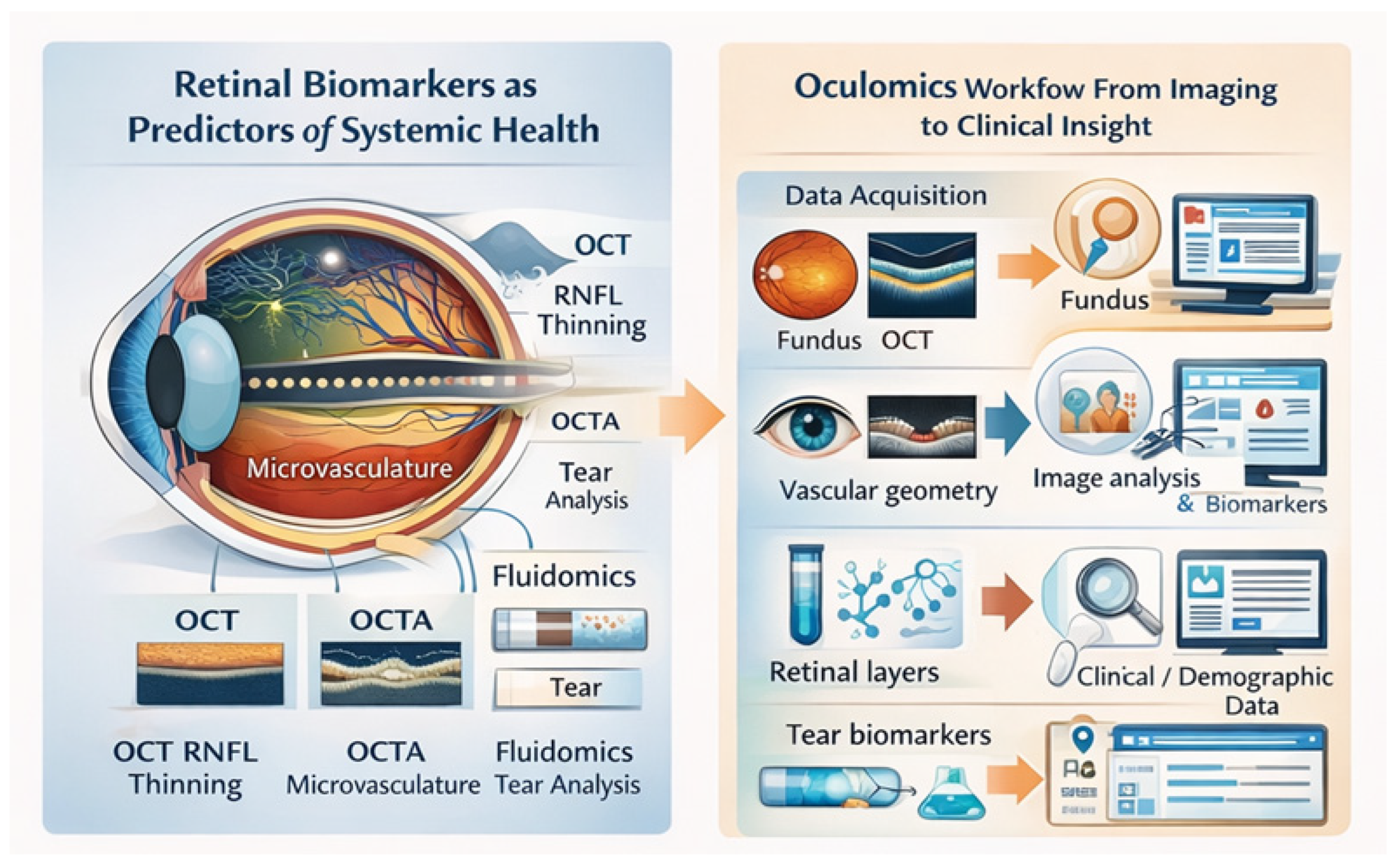
3. Key Technologies and Biomarkers
The practical realization of oculomics rests on the ability to capture, quantify, and interpret ocular features that faithfully reflect systemic biological processes. Unlike traditional ophthalmic assessment, which is often qualitative and disease-specific, oculomics relies on standardized, quantitative biomarkers derived from imaging and ocular biofluids. These biomarkers are inherently suited for longitudinal monitoring, population-level analysis, and integration with artificial intelligence (AI)–driven analytical frameworks. This chapter focuses on the core technologies underpinning oculomics and the principal classes of biomarkers they generate, while avoiding redundancy with the biological rationale discussed in previous sections (
Figure 1).
3.1. Retinal Imaging as a Quantitative Platform
Color fundus photography (CFP) remains one of the most widely deployed ocular imaging modalities worldwide, forming the historical and practical foundation of retinal phenotyping. Beyond its conventional diagnostic role, CFP enables the extraction of quantitative vascular features, including arteriolar and venular caliber, vessel tortuosity, branching angles, and fractal dimension. These parameters provide a structural fingerprint of systemic microvascular health and have been robustly associated with hypertension, diabetes, cardiovascular disease, and stroke risk in large population-based cohorts [
2,
22]. In the context of oculomics, the value of CFP lies not in isolated lesion detection, but in its capacity to capture global vascular architecture. Subtle changes in vascular geometry—often imperceptible to the human observer—can be quantified algorithmically and linked to systemic endothelial dysfunction, chronic inflammation, and altered hemodynamics [
28]. The ubiquity, low cost, and scalability of CFP make it particularly attractive for large-scale screening and epidemiological studies, especially when combined with automated image analysis.
Optical coherence tomography (OCT) has fundamentally transformed retinal imaging by providing micrometer-resolution, cross-sectional visualization of retinal microstructure in vivo. In oculomics, OCT functions as a form of non-invasive optical biopsy, enabling precise and reproducible measurement of individual retinal layer thicknesses. Of particular relevance are the retinal nerve fiber layer (RNFL) and the ganglion cell–inner plexiform layer (GC-IPL), which serve as quantitative surrogates for neuroaxonal integrity [
26]. Thinning of these layers has been consistently linked to neurodegenerative processes affecting the central nervous system. Importantly, OCT-derived metrics allow detection of subclinical neurodegeneration, often preceding overt neurological symptoms. This positions OCT as a powerful tool for risk stratification, disease monitoring, and therapeutic response assessment in conditions such as Alzheimer’s disease, Parkinson’s disease, and multiple sclerosis [
11]. From an oculomics perspective, the strength of OCT lies in its ability to translate complex neural pathology into simple, standardized numerical biomarkers suitable for longitudinal analysis.
OCT angiography (OCTA) extends the structural insights of OCT by providing depth-resolved visualization of retinal and choroidal blood flow without the need for exogenous contrast agents. By detecting motion contrast from erythrocytes, OCTA generates detailed maps of capillary networks, including the superficial and deep vascular plexuses and the foveal avascular zone (FAZ). Quantitative metrics such as vessel density, perfusion density, and flow voids can be extracted with high reproducibility [
27]. These parameters are exquisitely sensitive to early microvascular dysfunction, making OCTA particularly valuable for oculomics applications in systemic diseases characterized by vascular compromise. Alterations in capillary density and FAZ morphology have been linked to diabetes, hypertension, chronic kidney disease, and vascular cognitive impairment, often before the appearance of clinically evident retinopathy [
9]. As a functional complement to structural OCT, OCTA enables a more comprehensive assessment of neurovascular coupling and systemic microcirculatory health.
3.2. Emerging Imaging-Derived Biomarkers
Beyond conventional layer thickness and vessel density metrics, advanced image analysis has given rise to novel biomarkers with systemic relevance. Retinal ischemic perivascular lesions (RIPLs), identified on
en face OCT, represent permanent anatomical footprints of prior subclinical ischemic injury and may serve as cumulative markers of systemic vascular burden [
10]. Similarly, texture analysis, reflectivity profiling, and spatial pattern recognition across retinal layers are increasingly explored as higher-order biomarkers of metabolic stress and neurodegeneration.
These emerging features underscore a key principle of oculomics: the eye encodes systemic pathology not only through overt lesions, but also through subtle, spatially distributed alterations that require computational approaches for detection. Such biomarkers are inherently suited for AI-based discovery pipelines and may ultimately outperform traditional single-parameter measures in predictive accuracy.
3.3. Ocular Fluidomics: Molecular Readouts of Systemic Health
The term fluidomics refers to the comprehensive molecular analysis of biofluids—in this context, tears, aqueous humor, and vitreous—to identify protein, lipid, and metabolic signatures associated with health and disease. While imaging forms the backbone of oculomics, ocular biofluids provide a complementary molecular dimension. The tear film, in particular, has attracted growing interest due to its non-invasive accessibility and rich molecular composition. Proteomic, lipidomic, and metabolomic analyses of tears have revealed signatures associated with inflammation, metabolic dysregulation, neurodegeneration, and malignancy-related pathways [
5,
29].
While the tear film offers a rich molecular profile through non-invasive collection , the more invasive sampling of internal fluids—such as aqueous and vitreous humor—provides higher molecular specificity due to their immediate proximity to retinal tissue. Consequently, these surgical sampling procedures allow internal ocular fluids to function as localized liquid biopsies that reflect the precise metabolic and inflammatory state of the central nervous system [
30]. In the oculomics framework, fluid-derived biomarkers serve as adjuncts to imaging, enriching phenotypic resolution and complementing structural and functional metrics.
4. Cardiovascular and Metabolic Health
Cardiovascular and metabolic diseases represent the most mature and clinically substantiated application domain of oculomics. This prominence is biologically grounded. The retina is exquisitely sensitive to disturbances in microvascular integrity, endothelial function, and metabolic homeostasis—processes that are central to hypertension, diabetes mellitus, atherosclerosis, and cardiometabolic syndromes. Importantly, retinal alterations typically emerge along a continuum, often preceding clinically evident damage in the heart, brain, or kidneys. Within this framework, retinal biomarkers provide a unique means of capturing both early dysfunction and the cumulative burden of systemic vascular injury.
4.1. Retinal Microvasculature and Hypertensive Vascular Remodeling
The retinal microvascular network offers a direct and quantifiable representation of systemic small-vessel health. Unlike conventional imaging modalities that primarily assess large arteries, retinal imaging captures microvessels that are particularly vulnerable to chronic hemodynamic stress and endothelial dysfunction. Population-based studies have consistently shown that changes in retinal arteriolar and venular caliber reflect systemic blood pressure regulation, arterial stiffness, and inflammatory status [
2,
22].
Generalized arteriolar narrowing is strongly associated with hypertension and elevated cardiovascular risk, whereas venular dilation correlates with metabolic dysregulation, systemic inflammation, and incident cardiovascular events. Beyond simple caliber measurements, higher-order geometric descriptors—including tortuosity, branching asymmetry, and fractal dimension—encode information about long-term vascular remodeling and impaired autoregulation [
28]. Within the oculomics paradigm, these features are interpreted as integrative biomarkers that reflect cumulative vascular stress rather than isolated ophthalmic abnormalities.
Hypertension exemplifies the clinical value of these retinal biomarkers. Subtle microvascular changes can be detected years before the appearance of overt hypertensive retinopathy or clinically apparent end-organ damage. AI-driven analyses of fundus images have further enhanced sensitivity by capturing complex vascular signatures that reflect lifetime blood pressure exposure, outperforming risk stratification based solely on single-point clinical measurements [
2]. As such, retinal biomarkers may offer a biologically meaningful assessment of vascular risk, particularly in individuals with masked, nocturnal, or labile hypertension.
4.2. Diabetes and Neurovascular Metabolic Dysfunction
Diabetes mellitus provides a paradigmatic example of how oculomics extends beyond traditional disease classification. While diabetic retinopathy has long been recognized as a hallmark microvascular complication, oculomics shifts attention toward preclinical retinal alterations that precede clinically detectable retinopathy. Subtle impairments in capillary perfusion, photoreceptor integrity, and neuroretinal thickness have been documented in individuals with impaired glucose metabolism and early type 2 diabetes [
31,
32].
OCT and OCTA studies further demonstrate that neurodegenerative changes—such as thinning of inner retinal layers—can occur independently of overt microvascular lesions, underscoring the neurovascular nature of diabetic retinal pathology [
9]. These findings reinforce the concept that diabetes is not solely a metabolic disorder but also a disease characterized by early neural and microvascular dysfunction. From a systemic perspective, retinal biomarkers provide a sensitive readout of disease burden and therapeutic response, even in the absence of classical diabetic retinopathy.
4.3. Retinal Ischemic Signatures and Cumulative Vascular Injury
Beyond dynamic measures of vascular function, recent work has identified retinal ischemic perivascular lesions (RIPLs) as permanent structural markers of prior microvascular ischemic events. Visualized on en face OCT, RIPLs are thought to result from focal capillary occlusion followed by neuronal remodeling. Notably, these lesions may persist even after apparent normalization of systemic risk factors, effectively serving as a cumulative record of past ischemic injury [
10].
In the context of cardiovascular risk assessment, RIPLs represent a conceptual shift from transient biomarkers to structural indicators of vascular memory. Their presence has been associated with systemic vascular disease and may complement conventional risk scores by capturing historical exposure to ischemic stress. This notion aligns with the broader oculomics perspective, in which the retina functions as a biological archive of systemic pathology rather than a snapshot of current physiological state.
4.4. Cardiovascular Risk Prediction and Event Stratification
One of the most compelling promises of oculomics lies in its capacity to predict future cardiovascular events. Large-scale studies have demonstrated that retinal features derived from fundus photography and OCT can predict myocardial infarction, stroke, and cardiovascular mortality, often independently of established clinical risk factors [
2,
22].
AI-based models trained on retinal images have shown the ability to infer systemic parameters such as age, sex, smoking status, and glycemic control, highlighting the depth of physiological information encoded within ocular tissues. When combined with traditional clinical variables, these models improve risk discrimination and may enable personalized prevention strategies [
7]. Nevertheless, model interpretability remains a key challenge, and ongoing efforts aim to link algorithmic predictions to biologically meaningful retinal features.
4.5. Metabolic Syndrome, Systemic Inflammation, and Clinical Translation
Beyond overt diabetes, retinal biomarkers have been associated with broader cardiometabolic syndromes characterized by obesity, dyslipidemia, insulin resistance, and chronic low-grade inflammation. Venular dilation, altered fractal dimension, and capillary rarefaction reflect inflammatory and oxidative stress pathways that are central to cardiometabolic disease pathogenesis [
22]. These findings emphasize the role of the retina as an integrative sensor of systemic metabolic imbalance rather than a disease-specific endpoint.
From a translational perspective, the application of oculomics to cardiovascular and metabolic health illustrates a broader shift from reactive to proactive medicine. Retinal biomarkers offer a non-invasive, scalable means of assessing vascular and metabolic integrity across the lifespan. Importantly, they should be viewed not as replacements for established diagnostic tools, but as complementary sources of information that capture dimensions of disease biology inaccessible through conventional testing. As validation studies expand and AI-driven analytics mature, oculomics is poised to play an increasingly central role in cardiometabolic risk assessment, setting the stage for disease-specific applications discussed in subsequent chapters.
5. Neurodegenerative and Neurological Disease
Oculomics has shown particular promise in the study of neurodegenerative and neurological disorders, where direct access to central nervous system (CNS) tissue is limited and existing biomarkers are often invasive, costly, or insufficiently sensitive to early disease stages. The retina, as an accessible extension of the CNS, offers a unique opportunity to interrogate neuroaxonal integrity, neurovascular coupling, and neuroinflammatory processes in vivo. Retinal imaging thus provides a biologically grounded and clinically scalable window into brain pathology, positioning oculomics as a complementary approach to established neurological biomarkers.
5.1. Alzheimer’s Disease and Related Dementias
Alzheimer’s disease (AD) represents one of the most extensively studied neurological applications of oculomics. Converging evidence indicates that AD is associated with progressive thinning of the retinal nerve fiber layer (RNFL) and the ganglion cell–inner plexiform layer (GC-IPL), reflecting widespread neuroaxonal degeneration analogous to that observed in the cerebral cortex [
21]. These structural changes are detectable using optical coherence tomography (OCT) and may occur at preclinical or prodromal stages, preceding overt cognitive decline.
Beyond neurodegeneration, retinal microvascular alterations provide additional insight into AD pathophysiology. OCT angiography studies have demonstrated reduced capillary density, enlargement of the foveal avascular zone, and impaired perfusion in patients with mild cognitive impairment and AD, mirroring cerebral microvascular dysfunction and impaired neurovascular coupling [
22]. Together, neural and vascular retinal biomarkers support the concept of AD as a combined neurodegenerative and vasculopathic disorder.
Importantly, retinal biomarkers correlate with established central measures of disease burden, including hippocampal atrophy, cerebral amyloid load, and cognitive performance, underscoring their biological relevance [
21]. Within the oculomics framework, these findings suggest that the retina may function as an early indicator of cerebral pathology, offering opportunities for risk stratification, longitudinal monitoring, and potentially for evaluating therapeutic response in disease-modifying trials.
5.2. Parkinson’s Disease and Disorders of Dopaminergic Signaling
Parkinson’s disease (PD) further illustrates the relevance of retinal biomarkers in neurodegenerative conditions. Dopaminergic neurons play a critical role not only in basal ganglia circuits but also in retinal signal processing. In PD, alterations in retinal structure—particularly thinning of the inner retinal layers—have been consistently reported and are thought to reflect both neurodegeneration and disrupted dopaminergic modulation [
21].
Functional changes complement these structural findings. Abnormalities in contrast sensitivity, color vision, and pupillary light reflex dynamics have been documented in PD, indicating impaired retinal neurotransmission and autonomic dysfunction. Pupillometry, in particular, has emerged as a potential oculomics biomarker of central autonomic and dopaminergic impairment, capturing dynamic neural responses that are difficult to assess through static imaging alone [
21].
These retinal manifestations are increasingly recognized as part of the non-motor spectrum of PD and may precede classical motor symptoms. From a translational standpoint, retinal biomarkers could contribute to earlier diagnosis, phenotypic stratification, and monitoring of disease progression, complementing clinical assessment and neuroimaging.
5.3. Multiple Sclerosis and Inflammatory Demyelinating Disease
Among neurological disorders, multiple sclerosis (MS) represents one of the most clinically mature applications of retinal imaging. OCT-derived retinal layer analysis provides a robust, non-invasive measure of neuroaxonal loss, particularly within the RNFL and GC-IPL, which closely reflects cumulative damage to the optic nerve and broader CNS pathways [
11].
Importantly, retinal thinning in MS is observed not only following episodes of optic neuritis but also in eyes without a history of overt optic nerve inflammation, indicating diffuse neurodegeneration. These changes correlate with brain atrophy, lesion burden on magnetic resonance imaging, and long-term disability, positioning retinal biomarkers as valuable adjuncts to conventional imaging and cerebrospinal fluid markers [
11].
Oculomics further extends the utility of retinal assessment in MS by enabling high-frequency, longitudinal monitoring of disease activity and treatment response. Compared with MRI, OCT is less costly, more accessible, and more readily repeatable, making it particularly attractive for routine clinical follow-up and large-scale clinical studies.
5.4. Psychiatric and Neurodevelopmental Disorders
Emerging evidence suggests that retinal biomarkers may also index pathological processes in psychiatric and neurodevelopmental conditions. Thinning of retinal neural layers, particularly within the RNFL and GC-IPL, has been reported in schizophrenia and has been associated with cognitive impairment, disease severity, and functional outcomes [
33]. These findings support the hypothesis that schizophrenia involves progressive neurobiological changes extending beyond classical neurotransmitter dysregulation.
From an oculomics perspective, retinal alterations in psychiatric disorders may reflect shared neurodevelopmental vulnerabilities, neuroinflammatory pathways, or accelerated neurodegenerative processes. While the current evidence base remains limited and largely cross-sectional, these observations open new avenues for investigating brain–retina relationships in disorders traditionally diagnosed and monitored using behavioral and clinical criteria alone.
5.5. Integrative Perspective and Translational Implications
Collectively, findings from neurodegenerative and neurological disorders underscore the retina as a uniquely accessible surrogate of central nervous system pathology, capable of capturing neuroaxonal loss, neurovascular dysfunction, and inflammatory processes in vivo. Unlike conventional neurological biomarkers that often reflect static or late-stage disease, retinal measures offer a dynamic and repeatable readout of ongoing tissue injury and remodeling.
As multimodal analytics evolve, retinal structural and functional metrics may increasingly complement neuroimaging and cerebrospinal fluid markers by enabling earlier detection, finer phenotypic stratification, and high-frequency longitudinal monitoring. In this sense, oculomics contributes not merely an additional biomarker modality, but a fundamentally different temporal and biological perspective on neurological disease progression.
6. Emerging Frontiers: Bone, Kidney, and Environmental Health
While cardiovascular, metabolic, and neurological disorders represent the most mature applications of oculomics, recent evidence suggests that ocular biomarkers may also capture systemic processes traditionally considered remote from ophthalmology. Disorders of skeletal integrity, renal function, and environmental exposure share a common pathophysiological substrate characterized by chronic inflammation, microvascular dysfunction, and cumulative biological stress. In this context, the eye may function as a distal yet sensitive reporter of systemic vulnerability, extending the scope of oculomics beyond classical disease boundaries.
6.1. Retinal Biomarkers and Skeletal Health
Bone health has historically been assessed using localized measures such as dual-energy X-ray absorptiometry (DEXA), which provide static estimates of bone mineral density but limited insight into systemic biological aging. Recent AI-driven studies have challenged this compartmentalized view by demonstrating associations between ophthalmic features and osteoporosis risk. Retinal vascular parameters, lens status, and early age-related macular degeneration phenotypes have been linked to reduced bone mineral density and increased fracture risk [
34].
Several mechanisms may underlie this association. Bone and retinal tissues are both highly sensitive to microvascular perfusion, oxidative stress, and age-related alterations in extracellular matrix remodeling. Chronic low-grade inflammation and endothelial dysfunction—central drivers of osteoporosis—may therefore manifest in parallel within the retinal microcirculation. In addition, shared metabolic pathways involving calcium homeostasis, vitamin D signaling, and mitochondrial function may contribute to coupled degeneration of skeletal and ocular tissues [
35].
From an oculomics perspective, these findings suggest that retinal imaging could serve as an opportunistic marker of skeletal fragility, particularly in populations undergoing routine ophthalmic screening. While retinal biomarkers are unlikely to replace established diagnostic tools for osteoporosis, they may contribute to early risk stratification and identify individuals who would benefit from targeted bone health assessment.
6.2. Oculomics in Chronic Kidney Disease
Chronic kidney disease (CKD) represents another emerging domain in which oculomics offers biologically plausible insights. CKD is fundamentally a microvascular disease, characterized by endothelial dysfunction, capillary rarefaction, and impaired autoregulation across multiple organ systems [
36]. The retina and choroid, with their dense vascular networks and high metabolic demand, are particularly vulnerable to these systemic alterations.
Clinical and imaging studies have demonstrated that retinal arteriolar narrowing, venular dilation, reduced capillary density, and choroidal thinning correlate with renal dysfunction and disease severity [
9,
37]. OCT and OCTA metrics have been shown to reflect declining glomerular filtration rate and increased albuminuria, even in the absence of overt ocular disease. These associations support the concept that retinal microvascular pathology mirrors renal microangiopathy rather than simply reflecting shared cardiovascular risk factors.
Importantly, ocular biomarkers may capture aspects of cumulative microvascular injury that are not readily accessible through conventional renal biomarkers. In this sense, oculomics complements laboratory measures by providing a structural and functional readout of systemic vascular health. As CKD often progresses silently until advanced stages, retinal imaging may offer a non-invasive window into early disease progression and vascular burden.
6.3. Oculomics and Exposomics: Linking Environment to Systemic Biology
The integration of oculomics with exposomics— the study of the totality of environmental exposures (e.g., to pollutants, chemicals, diet, stress) over a lifetime and their associated biological responses—represents a novel and rapidly evolving frontier. Environmental stressors such as air pollution, heavy metals, ultraviolet radiation, and occupational toxins exert systemic effects through oxidative stress, inflammation, and vascular injury. However, quantifying their cumulative biological impact remains a major challenge in population health.
Ocular tissues are uniquely positioned to act as sensors of environmental exposure. The ocular surface is directly exposed to the external environment, while the retina reflects downstream systemic consequences of chronic exposure. Recent studies have identified associations between air pollution exposure and alterations in retinal vascular caliber, capillary density, and neuroretinal thickness, suggesting that environmental insults leave measurable signatures in ocular tissues [
30].
Beyond imaging, tear fluid analysis adds a molecular dimension to environmental oculomics, enabling the detection of biomarkers that reflect both external exposures and systemic biological responses. Human tears contain a complex repertoire of proteins, lipids, and small molecules that interface directly with the external environment at the air–tear surface and simultaneously integrate signals from the internal milieu by crossing the blood–tear barrier. Tear fluid has been proposed as a promising biomonitoring matrix for environmental and chemical exposures because it can non-invasively capture exposure markers from both the external and internal environments [
38] Amini 2024]. Real-world studies demonstrate that air pollution exposures are associated with changes in tear inflammatory mediators such as interleukin-6, interleukin-8, and VEGF, as well as alterations in tear film stability and ocular surface function, supporting the concept that external environmental stressors leave measurable molecular signatures in tears [
39,
40].
Proteomic and metabolomic profiling of tears reveals hundreds to thousands of detectable proteins and metabolites, including inflammatory-related proteins and metabolic intermediates that participate in key biological pathways relevant to oxidative stress and immune activation [
41]. Complementary metabolomics studies indicate that tear fluid contains metabolic signatures associated with systemic metabolic states, further underscoring its potential to serve as a window into internal biological processes influenced by external exposures [
42].
Collectively, these findings suggest that ocular biomarkers derived from tear fluid could bridge external environmental exposures and internal biological responses, providing an integrative readout of environmental risk and systemic physiology.
6.4. Implications for Precision Environmental and Systemic Medicine
The convergence of oculomics and exposomics supports a broader reconceptualization of the eye as an interface between environment and systemic biology. Rather than serving solely as a target organ for exposure-related disease, the eye may function as a biological dosimeter, recording the cumulative impact of environmental stress over time [
43]. This perspective aligns with emerging models of precision environmental health, which seek to move beyond exposure metrics toward individualized assessments of biological effect.
In clinical and public health contexts, retinal imaging and ocular fluid biomarkers could enable large-scale, non-invasive monitoring of environmental risk, particularly in vulnerable populations. While this field remains in its early stages, the integration of ocular biomarkers with environmental, genetic, and clinical data holds promise for advancing risk prediction, prevention strategies, and policy-relevant research.
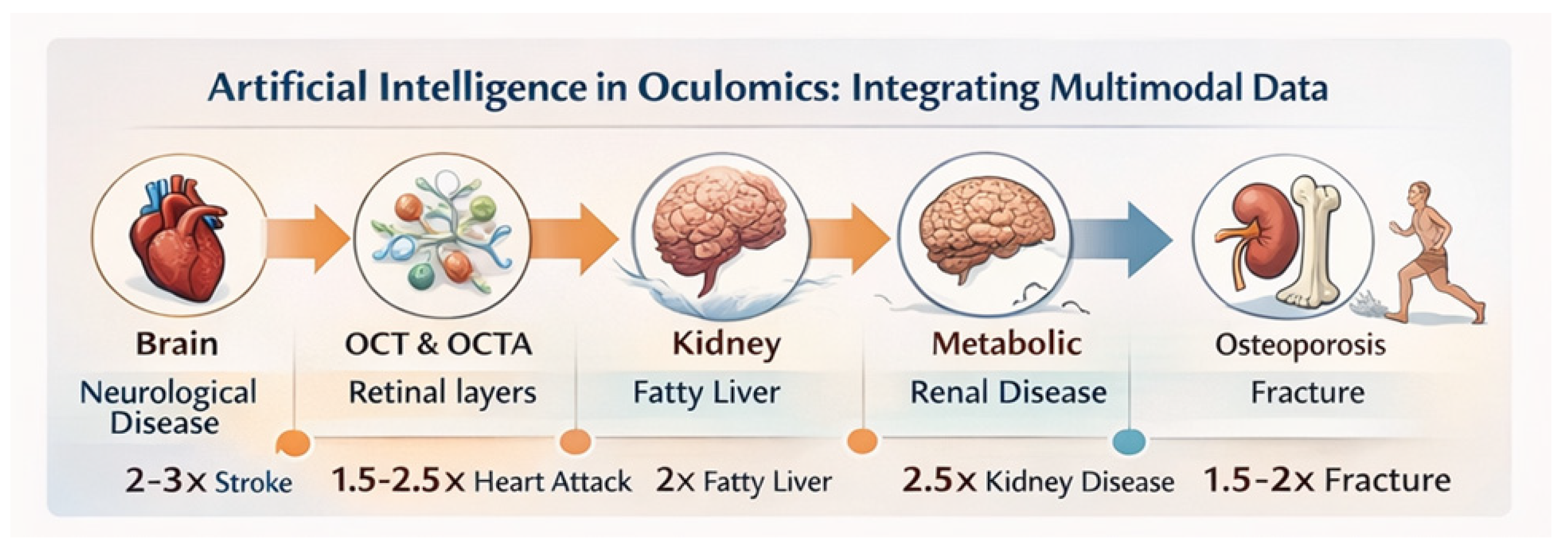
7. The Role of Artificial Intelligence
Artificial intelligence (AI) constitutes the methodological backbone that transforms oculomics from a descriptive framework into a scalable, predictive, and clinically actionable discipline. A defining feature of oculomics is the integration of heterogeneous data—retinal images, OCT/OCTA metrics, tear fluidomics, and clinical variables—into unified analytical models. The digital nature of this data makes it ideally suited for machine learning. AI, particularly deep learning, enables the extraction, integration, and interpretation of complex, high-dimensional patterns that are inaccessible to human interpretation, thereby unlocking the full informational content of ophthalmic data and shifting the field from descriptive correlation toward predictive modeling (
Figure 2).
7.1. From Human-Defined Features to Data-Driven Representation Learning
Traditional ophthalmic image analysis relies on predefined features such as vessel caliber, retinal layer thickness, or lesion counts. While biologically meaningful, these handcrafted metrics capture only a fraction of the information embedded within retinal images. Deep learning models, by contrast, operate through representation learning, identifying hierarchical patterns directly from raw data without prior assumptions about which features are most informative [
44,
45].
Seminal studies demonstrated that convolutional neural networks trained on fundus photographs could predict demographic and systemic variables—including biological age, sex, smoking status, blood pressure, and glycemic control—with remarkable accuracy [
6]. These findings provided early proof that retinal images encode systemic physiological signatures far beyond conventional ophthalmic phenotypes. Importantly, such predictions are not driven by single anatomical features but by distributed patterns spanning vascular geometry, texture, reflectivity, and spatial relationships across the retina.
7.2. Predictive Modeling and Disease Risk Stratification
Beyond cross-sectional inference, AI has enabled the use of ocular data for prospective disease prediction, a defining goal of oculomics. Deep learning models trained on large retinal datasets have been shown to predict incident cardiovascular events, including myocardial infarction and heart failure, independently of established clinical risk factors [
7]. These models capture cumulative biological exposure—often referred to as "biological memory," meaning the lasting physiological imprint of past risk factor exposure—that is not fully reflected by contemporaneous laboratory or clinical measurements.
More recently, foundation models trained on millions of retinal images have demonstrated strong generalizability across tasks and populations. Retina-specific architectures pretrained on large, heterogeneous datasets can be fine-tuned to predict multiple systemic outcomes with limited additional data, reflecting a shift from task-specific models toward reusable, general-purpose retinal representations [
8]. This paradigm substantially enhances scalability and reduces the data requirements for deploying oculomics across diverse healthcare settings.
7.3. Multimodal Integration and Systems-Level Inference
A key strength of AI in oculomics lies in its ability to integrate heterogeneous data modalities. Retinal images, OCT and OCTA metrics, tear fluidomics, demographic variables, and genetic information can be combined within unified analytical frameworks. Such multimodal models capture non-linear interactions between structural, functional, and molecular biomarkers, enabling systems-level inference of disease risk and progression [
46].
This integrative capacity is particularly relevant for complex, multifactorial conditions in which no single biomarker is sufficient. By learning joint representations across modalities, AI-based oculomics models can identify latent disease subtypes, stratify patients according to biological rather than purely clinical criteria, and support personalized risk profiling [
3]. Importantly, these approaches align with broader trends in precision medicine, where the emphasis shifts from isolated predictors to network-level biological signatures.
7.4. Explainability, Robustness, and Clinical Trust
Despite their predictive power, AI systems in oculomics face significant challenges related to interpretability and clinical trust. Early deep learning models were often criticized as “black boxes,” limiting their acceptance in high-stakes medical decision-making. In response, increasing emphasis has been placed on explainable AI techniques that link model outputs to biologically interpretable retinal features [
47].
Attention mapping, saliency analysis, and concept-based explanations have revealed that AI models frequently focus on anatomically plausible regions, including the retinal vasculature, optic disc, and macular architecture, when predicting systemic outcomes [
48]. These findings provide reassurance that predictions are grounded in meaningful biological substrates rather than spurious correlations. Parallel efforts to improve robustness, fairness, and external validation aim to ensure that AI-driven oculomics models perform consistently across populations, imaging devices, and clinical environments.
7.5. Translation, Ethics, and Future Directions
The integration of AI into oculomics raises important translational and ethical considerations. Model deployment requires standardized imaging protocols, rigorous external validation, and continuous performance monitoring to avoid bias and model drift. Moreover, the ability of AI systems to infer sensitive health information from ocular data necessitates careful governance around data privacy, informed consent, and clinical responsibility [
8].
Looking forward, AI is expected to play an increasingly central role in transforming routine ophthalmic imaging into a platform for systemic health assessment. As foundation models mature and multimodal integration advances, AI-driven oculomics may enable opportunistic screening, early disease detection, and longitudinal monitoring at population scale. In this context, AI should be viewed not as an isolated technological layer, but as the connective tissue that unifies biological insight, clinical relevance, and scalable implementation within the oculomics paradigm.
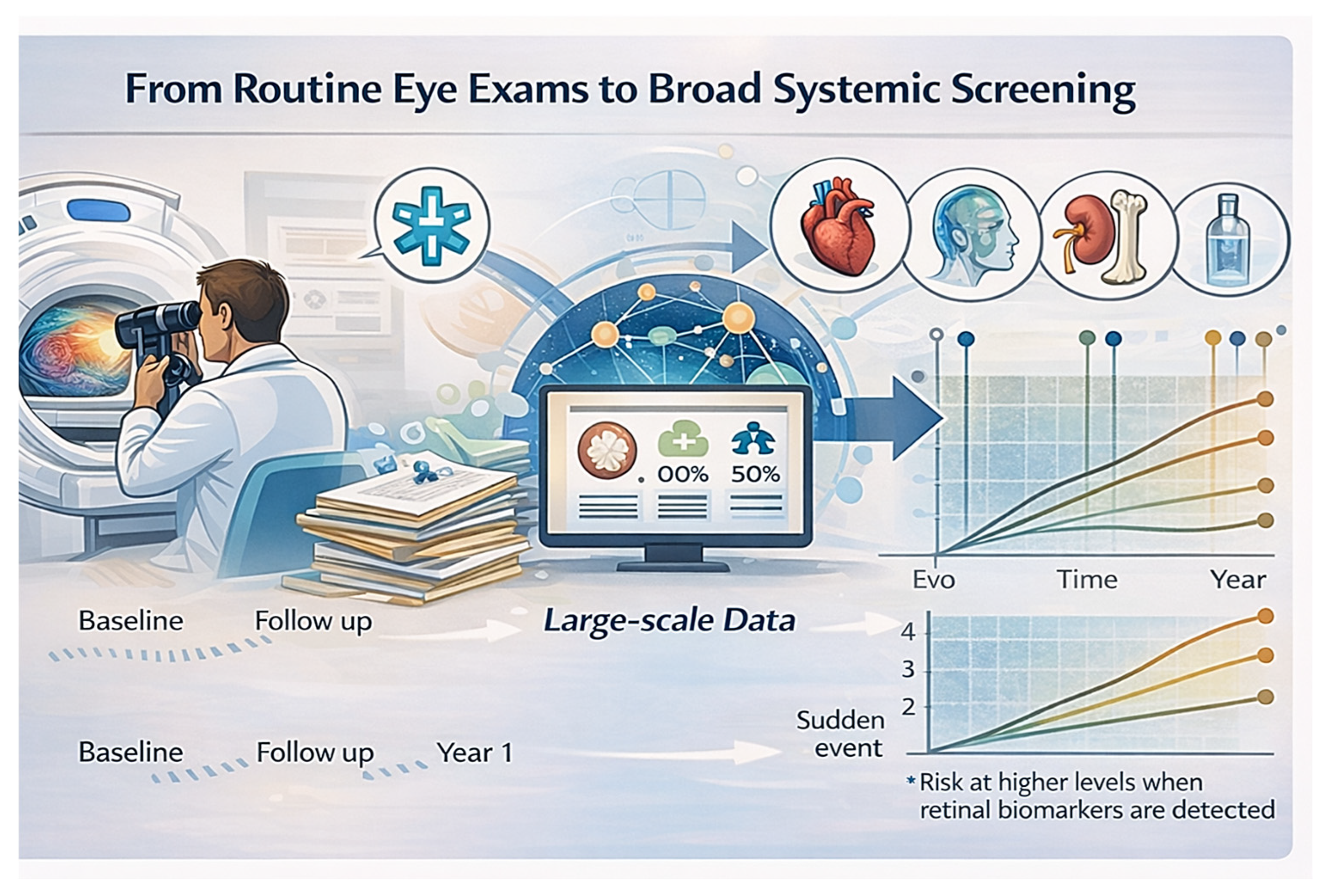
8. The “Healthcare from the Eye” Framework
The concept of
Healthcare from the Eye reframes routine ophthalmic examinations as structured entry points for proactive systemic health assessment. Rather than positioning ocular imaging as a specialty-isolated diagnostic activity, this framework recognizes ophthalmic data as a scalable source of systemic risk information that can be integrated into longitudinal, coordinated care pathways. Importantly, oculomics within this model is not intended to deliver definitive systemic diagnoses, but to support early risk stratification and triage in individuals who are frequently asymptomatic at the time of evaluation (
Figure 3).
The clinical rationale for this approach builds on accumulating evidence that retinal imaging—particularly when combined with artificial intelligence—encodes biologically meaningful information related to systemic health status. Deep learning analyses of fundus photographs have demonstrated the ability to infer a wide range of demographic and physiological variables, including age, sex, blood pressure, glycemic control, and medication use [
49]. These findings underscore the potent latent systemic signals embedded in routinely acquired ocular data [
6]. Large-scale studies and reviews further support the capacity of retinal biomarkers to predict systemic disease risk across cardiovascular, metabolic, and neurological domains, providing the scientific basis for broader clinical integration [
50].
A defining strength of the
Healthcare from the Eye framework lies in its alignment with existing care workflows. Eye examinations are already widely performed, repeatable across the lifespan, and frequently accessed even by individuals with limited engagement in primary care. Embedding oculomics-derived outputs into these encounters enables opportunistic identification of systemic risk without additional testing burden. When integrated into electronic health records, such outputs can support automated referral logic and decision support, facilitating targeted follow-up within appropriate clinical domains rather than creating parallel diagnostic pathways [
12].
Successful implementation depends less on algorithmic sophistication than on governance, interoperability, and clinical accountability. Recent systematic reviews of retinal fundus photograph–based artificial intelligence algorithms highlight that while predictive performance across medical domains is increasingly well established, clinical translation is now limited primarily by issues of standardization, external validation, interoperability, and workflow integration rather than algorithmic capability alone [
51]. Oculomics outputs must be standardized, interpretable, and explicitly framed as probabilistic risk indicators rather than diagnostic conclusions. Clear thresholds for action and predefined referral pathways are essential to ensure that generated insights translate into meaningful clinical responses. In this model, ophthalmologists and optometrists function as contributors to systemic risk detection, while responsibility for diagnosis and management remains with primary care physicians and relevant specialists, preserving established scopes of practice.
Data governance represents a central policy challenge. Retinal images and derived biomarkers may reveal latent information about systemic health that exceeds patient expectations at the time of imaging. Transparent consent processes, responsible data stewardship, and safeguards against unintended secondary use are therefore critical. In parallel, the risk of algorithmic bias necessitates continuous external validation across diverse populations, a priority echoed in recent strategic roadmaps that emphasize retinal biomarkers as promising but carefully governed tools for systemic disease research and clinical translation [
52].
Early real-world deployments suggest that oculomics-enabled workflows may improve diagnostic yield and health system efficiency by shifting elements of risk identification upstream and reducing reliance on late-stage, symptom-driven detection [
12]. These benefits arise not from replacing established diagnostic standards, but from augmenting them with non-invasive, biologically grounded signals derived from routine care. Accordingly,
Healthcare from the Eye should be viewed as an enabling infrastructure for preventive and precision medicine rather than as a standalone screening program.
In summary, the Healthcare from the Eye framework operationalizes oculomics as a health-system function rather than a technological novelty. By embedding ocular biomarkers into interoperable clinical pathways with appropriate governance and clinical oversight, this approach offers a pragmatic route toward earlier intervention, improved risk stratification, and more efficient use of healthcare resources.
9. Challenges and Limitations
Despite its strong biological rationale and rapidly expanding evidence base, oculomics has not yet transitioned into routine clinical practice. This gap between conceptual promise and real-world implementation reflects a convergence of technical, methodological, regulatory, and ethical challenges that must be addressed before oculomics can be safely, equitably, and effectively integrated into healthcare systems.
A primary limitation concerns standardization and reproducibility. Oculomics relies on subtle quantitative features extracted from ocular imaging, yet substantial variability exists across imaging devices, acquisition protocols, segmentation strategies, and analytic pipelines. Differences in camera hardware, illumination conditions, image resolution, and software updates can materially influence derived biomarkers and downstream model performance. This issue is particularly acute for metrics intended for individual risk prediction, where even modest variability can undermine clinical confidence.
Recent empirical work has illustrated this challenge in concrete terms. For example, studies assessing retinal fractal dimension—a feature frequently proposed as a marker of systemic microvascular health—have demonstrated that its repeatability and robustness vary significantly depending on the analytic pipeline used. Using two widely adopted segmentation frameworks (DART and AutoMorph), Engelmann and colleagues showed that algorithmic choices alone can introduce variability sufficient to affect risk stratification at the individual level [
28]. These findings underscore that, without harmonized methods and cross-platform benchmarking, oculomic biomarkers risk being context-dependent rather than clinically generalizable.
Closely related is the limitation imposed by data scale, diversity, and longitudinal depth. Many oculomics studies draw on large retrospective datasets that are nevertheless demographically constrained, often enriched for specific ethnicities, age ranges, or disease prevalences. Models trained under these conditions may perform well internally but fail to generalize across populations, exacerbating the risk of biased predictions. Moreover, cross-sectional associations—while useful for hypothesis generation—are insufficient to support preventive or therapeutic decision-making. Longitudinal datasets linking ocular biomarkers to incident disease, progression, and outcomes are essential to establish temporal validity and clinical utility, yet remain relatively scarce.
Another major barrier lies in clinical interpretability and responsibility. AI-driven oculomics models typically generate probabilistic risk estimates rather than categorical diagnoses, raising questions about how such outputs should be interpreted, communicated, and acted upon in clinical practice. Many high-performing models remain functionally opaque, offering limited insight into which retinal features drive predictions. This lack of transparency complicates clinician trust, patient counseling, and medico-legal accountability. As emphasized in broader analyses of artificial intelligence in ophthalmology, interpretability is not merely a technical aspiration but a prerequisite for responsible clinical deployment [
53].
Ethical and governance considerations further constrain translation. Oculomics enables the inference of systemic health information from data acquired for another purpose, creating tension around informed consent, incidental findings, and secondary data use. Patients undergoing routine eye examinations may not anticipate that their images could be used to infer cardiovascular, neurological, or metabolic risk. Clear consent frameworks, transparent communication, and robust data governance structures are therefore essential. In parallel, algorithmic bias—arising from unbalanced training data or structural inequities in healthcare access—requires continuous monitoring and mitigation to ensure equitable performance across populations.
Regulatory and economic barriers also remain substantial. For oculomics to achieve widespread adoption, models must demonstrate not only predictive accuracy but also clinical benefit, safety, and cost-effectiveness. Regulatory pathways for AI-based risk stratification tools are still evolving, with limited consensus on appropriate validation endpoints, acceptable performance thresholds, and post-market surveillance requirements. At the same time, the absence of clear reimbursement mechanisms reduces incentives for healthcare providers to integrate oculomics into routine workflows, particularly when benefits accrue downstream or across specialties.
Finally, oculomics faces a challenge of clinical ownership and integration. Its inherently interdisciplinary nature—spanning ophthalmology, primary care, cardiology, neurology, data science, and public health—complicates responsibility for implementation and oversight. Without clearly defined roles and care pathways, oculomics risks remaining a technically sophisticated but operationally orphaned innovation. As highlighted in policy-oriented discussions of
Healthcare from the Eye, successful translation will require not only technological maturity but also organizational alignment and cultural acceptance within healthcare systems [
12].
In summary, the current limitations of oculomics do not reflect a lack of scientific plausibility, but rather the complexity of translating high-dimensional ocular biomarkers into accountable, reproducible, and equitable clinical tools. Addressing these challenges will require coordinated advances in methodological standardization, data infrastructure, explainable AI, ethical governance, and health system integration. Only by confronting these constraints explicitly can the field move beyond proof-of-concept toward responsible and sustainable clinical adoption.
10. Take-Home Message
This review highlights oculomics as a conceptual and technological shift in how ocular data are interpreted within medicine. Rather than viewing the eye solely as an end-organ affected by systemic disease, oculomics reframes the retina as an accessible extension of neural and vascular biology, offering a unique opportunity to interrogate systemic health non-invasively and at scale.
The retina represents the only anatomical site where microcirculation and neural tissue can be directly visualized in vivo with high spatial resolution. This distinctive property enables the detection of structural, vascular, and neurodegenerative alterations that reflect fundamental pathophysiological processes shared across cardiovascular, metabolic, neurological, renal, skeletal, and environmental disease domains. Importantly, many of these retinal changes emerge before the onset of overt clinical symptoms or irreversible organ damage, positioning ocular biomarkers as early indicators rather than late complications of systemic disease.
By integrating advanced retinal imaging, molecular ocular biomarkers, and artificial intelligence–driven analytics, oculomics creates a functional bridge between ophthalmology, internal medicine, and neurology. This interdisciplinary framework supports a transition from reactive disease management toward preventive, risk-stratified, and personalized care, in which routine eye examinations may contribute actionable information beyond visual health alone. In this context, ocular data complement—rather than replace—established diagnostic pathways, offering biologically grounded insights that are difficult to obtain through conventional testing.
From a practical perspective, the scalability and non-invasive nature of ocular imaging make oculomics particularly well suited for population-level screening and longitudinal monitoring. As AI-enabled analytic pipelines mature and methodological standardization improves, retinal assessments could be seamlessly integrated into routine clinical workflows, electronic health records, and care coordination pathways. Such integration has the potential to enhance early disease detection, improve risk stratification, and optimize the allocation of healthcare resources without imposing substantial additional burden on patients or clinicians (
Figure 4).
10.1. Future Directions
The successful integration of oculomics into mainstream healthcare hinges on progress along several interconnected fronts:
Technical Harmonization: Developing universally accepted standards for image acquisition, segmentation, and biomarker quantification is paramount. Initiatives like the INSIGHT Health Data Hub and roadmaps from bodies like the NHLBI are critical first steps [
52,
53].
Longitudinal & Diverse Data: Moving beyond cross-sectional associations requires large-scale, prospective cohorts with repeated ocular measurements linked to hard systemic outcomes. Prioritizing diversity in these datasets is essential to ensure equitable model performance and generalizability.
Explainable & Actionable AI: The next generation of AI models must prioritize interpretability, clearly linking predictions to biologically plausible retinal features to build clinical trust. Furthermore, AI outputs need to be integrated into electronic health records as actionable risk scores with clear referral pathways.
Expanded Biomarker Discovery: Research must move beyond the retina to fully characterize the systemic signal in anterior segment imaging (e.g., corneal confocal microscopy) and in the molecular profiles of tears and aqueous humor.
Clinical Trial Integration: Ocular biomarkers should be incorporated as secondary or exploratory endpoints in systemic disease trials to validate their utility for monitoring therapeutic response and disease progression.
Ethical and Policy Frameworks: Establishing robust governance for data privacy, consent for secondary use, and guidelines for managing incidental findings is non-negotiable for responsible deployment.
Addressing these challenges will transform oculomics from a promising research paradigm into a foundational component of preventive, precision medicine.
11. Conclusions
As outlined above, the clinical translation of oculomics is a multidisciplinary endeavor. Success will depend on coordinated advances not just in technology, but in data infrastructure, clinical workflow, and ethical policy. By systematically addressing these priorities, the field can fully realize its potential to redefine medicine—transforming the routine eye examination into a powerful, accessible, and strategic pillar of proactive systemic health assessment and precision prevention.
In fact, beyond its technological implications, the implementation of oculomics has the potential to reshape the professional role of the ophthalmologist. Traditionally focused on the diagnosis and treatment of ocular disease, the ophthalmologist may increasingly act as a first-line observer of systemic health, interpreting retinal biomarkers that reflect cardiovascular, neurological, metabolic, and inflammatory status. In this evolving framework, the ophthalmic examination extends beyond visual function, contributing clinically relevant information to broader health assessment and preventive care.
Rather than replacing the role of other medical specialties, oculomics positions ophthalmologists as key partners within multidisciplinary care pathways. Routine eye examinations may thus evolve into opportunistic, non-invasive health evaluations, prompting timely referral, risk stratification, and early intervention. This shift underscores the expanding clinical value of ophthalmology within precision and preventive medicine, while reinforcing the need for appropriate training, data governance, and integration with systemic care infrastructures.
Author Contributions
Conceptualization, D.R.; methodology, D.R., C.G., J.F.M.-V.; software, J.F.M.-V.; data curation, D.R., C.G., J.F.M.-V.; writing—original draft preparation, D.R.; writing—review and editing, D.R., C.G., J.F.M.-V.; supervision, D.R. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created.
Acknowledgments
During the preparation of this manuscript, the author(s) used ChatGPT-4 for the purposes of critical proofreading of the manuscript, improvement of idiomatic English and to generate the figures. The authors have reviewed and edited the output and take full responsibility for the content of this publication.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Jafari N, Golnik K, Shahriari M, Karimzadeh P, Jabbehdari S. Ophthalmologic findings in patients with neuro-metabolic disorders. J Ophthalmic Vis Res. 2018;13(1):34-38. [CrossRef]
- Wagner SK, Fu DJ, Faes L, et al. Insights into systemic disease through retinal imaging-based oculomics. Transl Vis Sci Technol. 2020;9(2):6. [CrossRef]
- Zhu Z, Wang Y, Qi Z, et al. Oculomics: Current concepts and evidence. Prog Retin Eye Res. 2025;106:101350. [CrossRef]
- Honavar SG. Oculomics—the eyes talk a great deal. Indian J Ophthalmol. 2022;70(3):713. [CrossRef]
- Patterson EJ, Bounds AD, Wagner SK, et al. Oculomics: A crusade against the four horsemen of chronic disease. Ophthalmol Ther. 2024;13(6):1427-1451. [CrossRef]
- Wu JH, Liu TYA. Application of deep learning to retinal-image-based oculomics for evaluation of systemic health: A review. J Clin Med. 2022;12(1):152. [CrossRef]
- Wang J, Wang YX, Zeng D, et al. Artificial intelligence-enhanced retinal imaging as a biomarker for systemic diseases. Theranostics. 2025;15(8):3223-3233. [CrossRef]
- Yao J, Hong ASY, Fukutsu K, Ting DSW. Artificial intelligence oculomics for systemic health and longevity medicine: 2025 and beyond. Curr Opin Ophthalmol. 2025. [CrossRef]
- Merriott DJ, Parikh D, Najac MJ, et al. Optical coherence tomography and optical coherence tomography angiography in systemic disease. Taiwan J Ophthalmol. 2025;15(3):364-377. [CrossRef]
- Bair H. From risk markers to treatable traits in retinal oculomics. Exp Eye Res. 2025. [CrossRef]
- Suh A, Hampel G, Vinjamuri A, et al. Oculomics analysis in multiple sclerosis: Current ophthalmic clinical and imaging biomarkers. Eye (Lond). 2024;38(14):2701-2710. [CrossRef]
- Weinreb RN, Keane PA, Cooley A, et al. A framework for healthcare from the eye: Oculomics as a powerful window to systemic health. Ophthalmology. 2026. [CrossRef]
- Morales-Luque C, Carrillo-Franco L, López-González MV, et al. Mapping the neurophysiological link between voice and autonomic function: A scoping review. Biology (Basel). 2025;14(10):1382. [CrossRef]
- Lepri G, Hughes M, Allanore Y, et al. The role of skin ultrasound in systemic sclerosis: Looking below the surface to understand disease evolution. Lancet Rheumatol. 2023;5(7):e422-e425. [CrossRef]
- Khera R, Oikonomou EK, Nadkarni GN, et al. Transforming cardiovascular care with artificial intelligence: From discovery to practice. J Am Coll Cardiol. 2024;84(1):97-114. [CrossRef]
- Salvadori M, Rosso G. Update on the gut microbiome in health and diseases. World J Methodol. 2024;14(1):89196. [CrossRef]
- Hadoux X, Hui F, Lim JKH, et al. Non-invasive in vivo hyperspectral imaging of the retina for potential biomarker use in Alzheimer disease. Nat Commun. 2019;10:4227. [CrossRef]
- Cheung CY, Chan VTT, Mok VC, Chen C, Wong TY. Potential retinal biomarkers for dementia: What is new? Curr Opin Neurol. 2019;32(1):82-91. [CrossRef]
- London A, Benhar I, Schwartz M. The retina as a window to the brain—from eye research to CNS disorders. Nat Rev Neurol. 2013;9(1):44-53. [CrossRef]
- Schmidt-Erfurth U, Sadeghipour A, Gerendas BS, Waldstein SM, Bogunović H. Artificial intelligence in retina. Prog Retin Eye Res. 2018;67:1-29. [CrossRef]
- Suh A, Ong J, Kamran SA, et al. Retina oculomics in neurodegenerative disease. Ann Biomed Eng. 2023;51(12):2708-2721. [CrossRef]
- Arnould L, Meriaudeau F, Guenancia C, et al. Using artificial intelligence to analyse the retinal vascular network: The future of cardiovascular risk assessment based on oculomics? Ophthalmol Ther. 2023;12(2):657-674. [CrossRef]
- Heavner W, Pevny L. Eye development and retinogenesis. Cold Spring Harb Perspect Biol. 2012;4(12):a008391. [CrossRef]
- Maurissen TL, Pavlou G, Bichsel C, et al. Microphysiological neurovascular barriers to model the inner retinal microvasculature. J Pers Med. 2022;12(2):148. [CrossRef]
- Carelli V, La Morgia C, Ross-Cisneros FN, Sadun AA. Optic neuropathies: The tip of the neurodegeneration iceberg. Hum Mol Genet. 2017;26(R2):R139-R150. [CrossRef]
- Bouma BE, de Boer JF, Huang D, et al. Optical coherence tomography. Nat Rev Methods Primers. 2022;2:79. [CrossRef]
- Ong CJT, Wong MYZ, Cheong KX, Zhao J, Teo KYC, Tan TE. Optical coherence tomography angiography in retinal vascular disorders. Diagnostics (Basel). 2023;13(9):1620. [CrossRef]
- Engelmann J, Moukaddem D, Gago L, et al. Applicability of oculomics for individual risk prediction: Repeatability and robustness of retinal fractal dimension using DART and AutoMorph. Invest Ophthalmol Vis Sci. 2024;65(6):10. [CrossRef]
- Fotovat-Ahmadi N, Siddiqui O, Ong J, et al. The ocular surface tear film as a biomarker for systemic health. Ocul Surf. 2025;37:283-300. [CrossRef]
- Cheng H, Sarnat JA, Walker DI, et al. Oculomics meets exposomics: A roadmap for applying multi-modal ocular biomarkers in precision environmental health research. Exposome. 2025;5(1):osaf013. [CrossRef]
- Ma Y, Wu Y, Hu L, et al. Photoreceptor layer thinning is an early biomarker for type 2 diabetes. Eye (Lond). 2025. [CrossRef]
- Vaughan M, Tay N, Kalitzeos A, et al. Changes in waveguiding cone photoreceptors and color vision in diabetes mellitus. Invest Ophthalmol Vis Sci. 2024;65(14):28. [CrossRef]
- Silverstein SM, Keane BP, Corlett PR. Oculomics in schizophrenia research. Schizophr Bull. 2021;47(3):577-579. [CrossRef]
- Choi JY, Han E, Yoo TK. Application of ChatGPT-4 to oculomics: A cost-effective osteoporosis risk assessment model. EPMA J. 2024;15(4):659-676. [CrossRef]
- Liu S, Zhang O, Wang H, et al. Association between age-related macular degeneration and osteoporosis in the US. Sci Rep. 2025;15:29045. [CrossRef]
- Yeung L, Wu IW, Sun CC, et al. Early retinal microvascular abnormalities in chronic kidney disease. Microcirculation. 2019;26(7):e12555. [CrossRef]
- Drakopoulos M, Nadel A, Bains HK, et al. Quantitative OCT angiography and systemic conditions. Surv Ophthalmol. 2026;71(2):423-455. [CrossRef]
- Amini P, Okeme JO. Tear fluid as a matrix for biomonitoring environmental exposures. Curr Environ Health Rep. 2024;11(3):340-355. [CrossRef]
- Hao R, Zhang M, Zhao L, et al. Impact of air pollution on the ocular surface and tear cytokine levels. Front Med (Lausanne). 2022;9:909330. [CrossRef]
- Jing D, Jiang X, Zhou P, et al. Air pollution-related ocular signs and inflammatory cytokines. Sci Rep. 2022;12:18359. [CrossRef]
- Chen X, Rao J, Zheng Z, et al. Integrated tear proteome and metabolome reveal inflammatory pathways in dry eye syndrome. J Proteome Res. 2019;18(5):2321-2330. [CrossRef]
- Brunmair J, Bileck A, Schmidl D, et al. Metabolic phenotyping of tear fluid in type 2 diabetes. EPMA J. 2022;13(1):107-123. [CrossRef]
- Battistini R, Di Geronimo N, Porru E, et al. Multi-pollutant exposure and ocular surface health: The Bike-Eye study. Int J Environ Res Public Health. 2025;22(12):1818. [CrossRef]
- Lai Y. A comparison of traditional machine learning and deep learning in image recognition. J Phys Conf Ser. 2019;1314:012148. [CrossRef]
- Beyeler MJ, Trofimova O, Bontempi D, et al. Comparing retinal image characteristics with deep learning features for disease prediction. medRxiv. 2024. [CrossRef]
- Cleland C, Taylor E. Artificial intelligence and oculomics: Improving global health. Eye News. 2025.
- An S, Teo K, McConnell MV, et al. AI explainability in oculomics. Prog Retin Eye Res. 2025;106:101352. [CrossRef]
- An S, Squirrell D. Validation of neuron activation patterns for artificial intelligence models in oculomics. Sci Rep. 2024;14:20940. [CrossRef]
- Khan NC, Perera C, Dow ER, et al. Predicting systemic health features from retinal fundus images using transfer learning AI models. Diagnostics (Basel). 2022;12(7):1714. [CrossRef]
- Ranchod TM. Systemic retinal biomarkers. Curr Opin Ophthalmol. 2021;32(5):439-444. [CrossRef]
- Grzybowski A, Jin K, Zhou J, et al. Retina fundus photograph-based AI algorithms in medicine: A systematic review. Ophthalmol Ther. 2024;13(8):2125-2149. [CrossRef]
- Chew EY, Burns SA, Abraham AG, et al. Standardization and clinical applications of retinal imaging biomarkers for cardiovascular disease. Nat Rev Cardiol. 2025;22(1):47-63. [CrossRef]
- Denniston AK, Kale AU, Lee WH, Mollan SP, Keane PA. Building trust in real-world data: Lessons from INSIGHT, the UK’s health data research hub for eye health and oculomics. Curr Opin Ophthalmol. 2022;33(5):399-406. [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).