Submitted:
13 April 2026
Posted:
15 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Method
1.2. Production and Consumption of Animal Proteins
1.3. Nutritional, Functional, Sensory and Religious Reasons of Meat Consumption
1.4. Meat Production Environmental Impact
2. Single-Cell Protein
2.1. Fungal SCP as Alternative Protein
2.2. Evolution of Mycoprotein as Alternative Protein
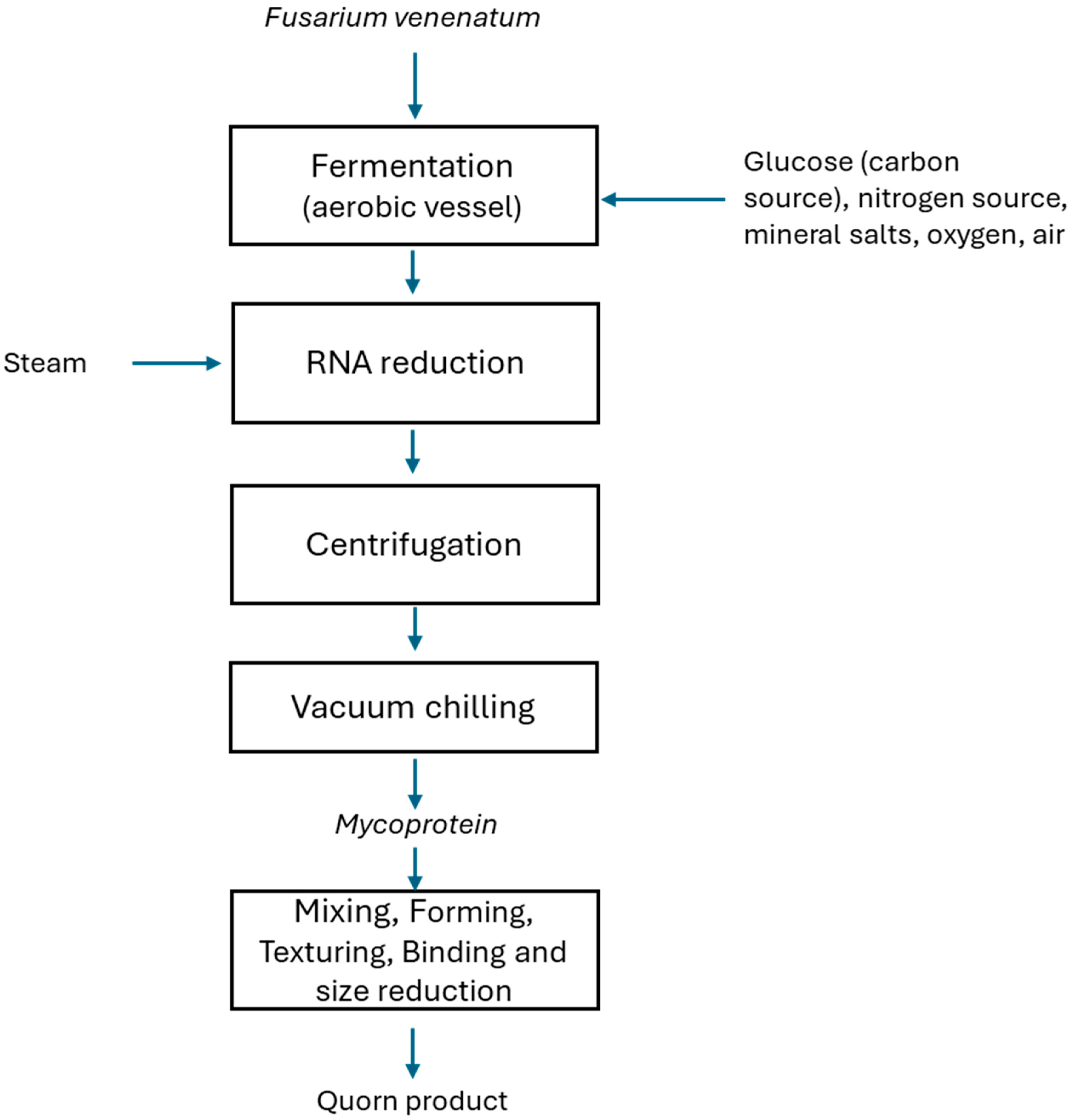
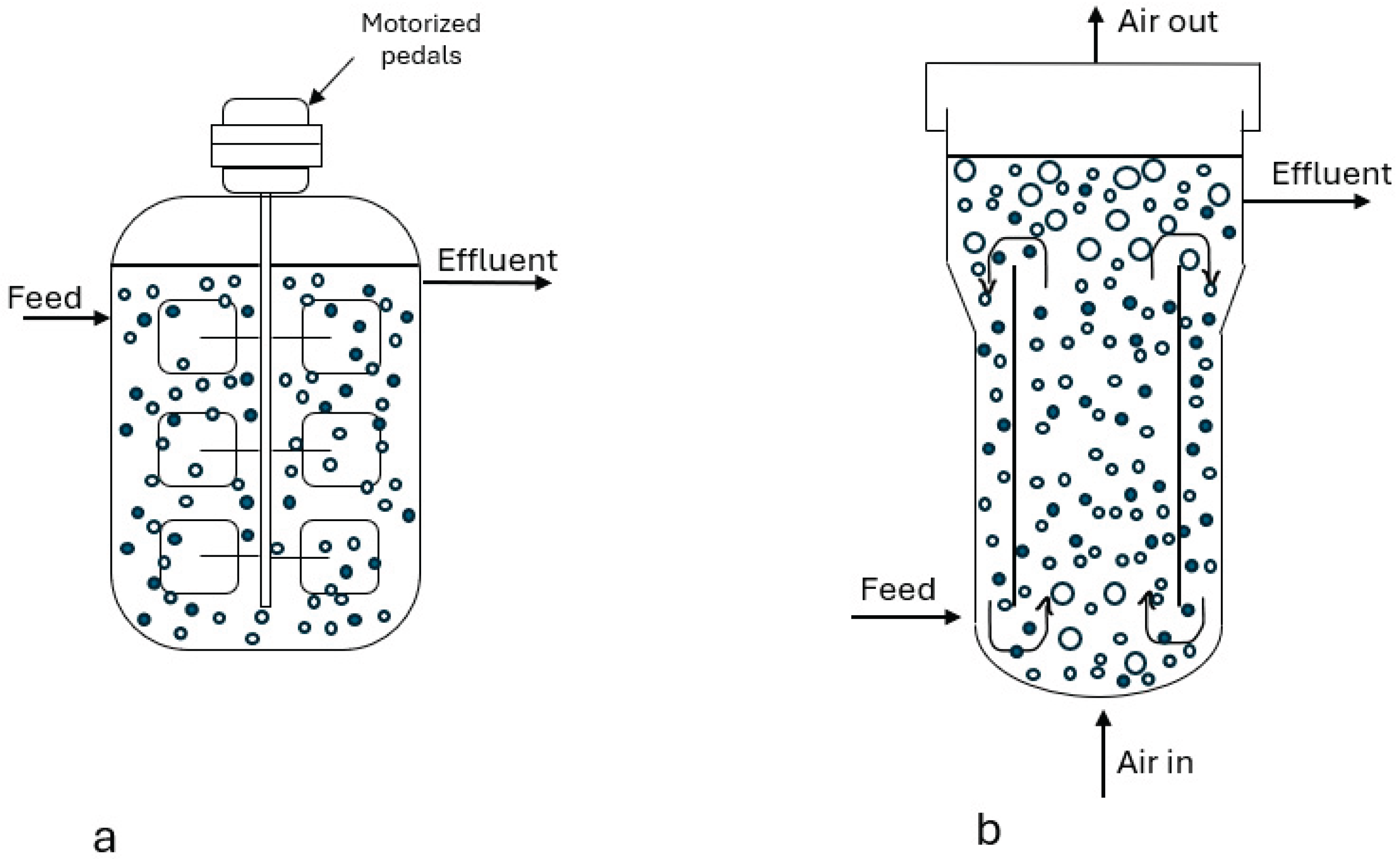
3. Fermentation
3.1. Types of Fermentation and Its Uses in the Food Industry
3.2. Limitations and Drawbacks of Fermentation
4. Acid and Enzymatic Hydrolysis of Substrates for Fermentation
5. Agricultural and Agro-Industrial Byproducts
5.1. Starch
5.1.1. Brewers’ Spent Grain
5.1.2. Rice Byproducts
5.1.3. Cassava Bagasse
5.1.4. Potato Peel Waste
6. Life Cycle Assessment
6.1. LCA on Mycoprotein
7. Conclusions
References
- United Nations. World population to reach 8 billion on 15 November 2022 | United Nations. https://www.un.org/en/desa/world-population-reach-8-billion-15-november-2022 (accessed on 1 Nov 2024).
- FAO. Meat Market Review: Overview of global market developments in 2023.
- Röös, E.; Sundberg, C.; Tidåker, P.; Strid, I.; Hansson, P. A. Can Carbon Footprint Serve as an Indicator of the Environmental Impact of Meat Production? Ecol. Indic. 2013, 24, 573–581. [Google Scholar] [CrossRef]
- GFI. State of Alternative Protein: A Global Glimpse at the State of the Industry. 2024. [Google Scholar]
- Poore, J.; Nemecek, T. Reducing Food’s Environmental Impacts through Producers and Consumers. Science (1979). 2018, 360(6392), 987–992. [Google Scholar] [CrossRef]
- Finnigan, T. J. A.; Wall, B. T.; Wilde, P. J.; Stephens, F. B.; Taylor, S. L.; Freedman, M. R. Mycoprotein: The Future of Nutritious Nonmeat Protein, a Symposium Review. In Current Developments in Nutrition; Oxford University Press, 2019; Vol. 3. [Google Scholar] [CrossRef]
- United Nations. Transforming our world: The 2030 agenda for sustainable development. https://sdgs.un.org/2030agenda (accessed on 6 Apr 2026).
- Caporgno, M. P.; Mathys, A. Trends in Microalgae Incorporation Into Innovative Food Products With Potential Health Benefits. Frontiers in Nutrition. Frontiers Media S.A. July 31, 2018. [CrossRef]
- van Huis, A. Nutrition and Health of Edible Insects. Current Opinion in Clinical Nutrition and Metabolic Car 2020, 22(3), 228–231. [Google Scholar] [CrossRef]
- Tuomisto, H. L.; Teixeira De Mattos, M. J. Environmental Impacts of Cultured Meat Production. Environ. Sci. Technol. 2011, 45(14), 6117–6123. [Google Scholar] [CrossRef] [PubMed]
- Santo, R. E.; Kim, B. F.; Goldman, S. E.; Dutkiewicz, J.; Biehl, E. M. B.; Bloem, M. W.; Neff, R. A.; Nachman, K. E. Considering Plant-Based Meat Substitutes and Cell-Based Meats: A Public Health and Food Systems Perspective. Frontiers in Sustainable Food Systems. Frontiers Media S.A. August 31, 2020. [CrossRef]
- Ayoo, C.; Bonti-Ankomah, S.; Aryee, A. N. A. Byproducts from Agriculture and Fisheries: Adding Value for Food, Feed, Pharma, and Fuels, 2020.
- Lynch, K. M.; Steffen, E. J.; Arendt, E. K. Brewers’ Spent Grain: A Review with an Emphasis on Food and Health. Journal of the Institute of Brewing 2016, 122(4), 553–568. [Google Scholar] [CrossRef]
- Ravindran, R.; Jaiswal, A. K. Exploitation of Food Industry Waste for High-Value Products. Trends in Biotechnology. Elsevier Ltd January 1, 2016, pp 58–69. [CrossRef]
- Betoret, N.; Betoret, E.; Glicerina, V. T. Valorization and Utilization of Food Wastes and By-Products: Recent Trends, Innovative Technologies and Sustainability Challenges. Foods. Multidisciplinary Digital Publishing Institute (MDPI) January 1, 2024. [CrossRef]
- Adejumo, I. O.; Adebiyi, O. a. Risky Benefits and Beneficial Risks of Animal Protein. 2024. [Google Scholar] [CrossRef]
- FAO. Meat and Dairy Production. https://ourworldindata.org/meat-production (accessed on 28 Jan 2026).
- Statista. Global Meat Production Forecast. https://www.statista.com/statistics/237632/production-of-meat-worldwide-since-1990/?srsltid=AfmBOoopwekf182lxfHttTaZFvvxv0EB04KBXbr1IkUlvR1E1BRyVO2L (accessed on 28 Jan 2026).
- Leroy, F.; Smith, N. W.; Adesogan, A. T.; Beal, T.; Iannotti, L.; Moughan, P. J.; Mann, N. The Role of Meat in the Human Diet: Evolutionary Aspects and Nutritional Value. Anim. Front. 2023, 13(2), 11. [Google Scholar] [CrossRef] [PubMed]
- Mottram, D. S. Flavour Formation in Meat and Meat a Review Products; 1998; Vol. 62. [Google Scholar]
- Li, L.; Belloch, C.; Flores, M. The Maillard Reaction as Source of Meat Flavor Compounds in Dry Cured Meat Model Systems under Mild Temperature Conditions. Molecules 2021, 26(1), 223. [Google Scholar] [CrossRef]
- Wang, L.; Huang, Y.; Wang, Y.; Shan, T. Effects of Polyunsaturated Fatty Acids Supplementation on the Meat Quality of Pigs: A Meta-Analysis. Front. Nutr. 2021, 8, 746765. [Google Scholar] [CrossRef]
- Shahidi, F.; Pegg, R. B. Hexanal as an Indicator of the Flavor Deterioration of Meat and Meat Products, 1994.
- Elmore, J. S.; Cooper, S. L.; Enser, M.; Mottram, D. S.; Sinclair, L. A.; Wilkinson, R. G.; Wood, J. D. Dietary Manipulation of Fatty Acid Composition in Lamb Meat and Its Effect on the Volatile Aroma Compounds of Grilled Lamb. Meat Sci. 2005, 69(2), 233–242. [Google Scholar] [CrossRef]
- Jones, D. T.; Woods, D. R. Acetone-Butanol Fermentation Revisited; 1986; Vol. 50. [Google Scholar]
- Chang, H.; Wang, Q.; Xu, X.; Li, C.; Huang, M.; Zhou, G.; Dai, Y. Effect of Heat-Induced Changes of Connective Tissue and Collagen on Meat Texture Properties of Beef Semitendinosus Muscle. Int. J. Food Prop. 2011, 14(2), 381–396. [Google Scholar] [CrossRef]
- Huff-Lonergan, E.; Lonergan, S. M. Mechanisms of Water-Holding Capacity of Meat: The Role of Postmortem Biochemical and Structural Changes. Meat Sci. 2005, 71(1), 194–204. [Google Scholar] [CrossRef]
- Jimenez-Colmenero, F.; Carballo, J.; Cofrades, S. Healthier Meat and Meat Products: Their Role as Functional Foods.
- Fiddes, N. Meat: A Natural Symbol, 1st ed.; Routledge, 2004. [Google Scholar] [CrossRef]
- Gerber, P. J. Tackling Climate Change through Livestock: A Global Assessment of Emissions and Mitigation Opportunities; Food and Agriculture Organization of the United Nations, 2013. [Google Scholar]
- Shindell, D.; Bréon, F.; Collins, W.; Fuglestvedt, J.; Huang, J.; Koch, D.; Lamarque, J.; Lee, D.; Mendoza, B.; Nakajima, T.; et al. Anthropogenic and Natural Radiative Forc-Ing. In Climate Change 2013: The Physical Science Basis. Contribution of Working Group I; 2013. [Google Scholar]
- Strassburg, B. B. N.; Latawiec, A. E.; Barioni, L. G.; Nobre, C. A.; da Silva, V. P.; Valentim, J. F.; Vianna, M.; Assad, E. D. When Enough Should Be Enough: Improving the Use of Current Agricultural Lands Could Meet Production Demands and Spare Natural Habitats in Brazil. Global Environmental Change 2014, 28(1), 84–97. [Google Scholar] [CrossRef]
- Leip, A. Evaluation of the Livestock Sector’s Contribution to the EU Greenhouse Gas Emissions (GGELS)-Final Report. 2010. [Google Scholar]
- de Vries, M.; de Boer, I. J. M. Comparing Environmental Impacts for Livestock Products: A Review of Life Cycle Assessments. Livest. Sci. 2010, 128(1–3), 1–11. [Google Scholar] [CrossRef]
- Walker, P.; Rhubart-Berg, P.; McKenzie, S.; Kelling, K.; Lawrence, R. S. Public Health Implications of Meat Production and Consumption. Public Health Nutr. 2005, 8(4), 348–356. [Google Scholar] [CrossRef]
- Ugalde, U. O.; Castrillo, J. I. Single Cell Proteins from Fungi and Yeasts. Applied Mycology and Biotechnology 2002, 2 (C), 123–149. [Google Scholar] [CrossRef]
- Galán, B.; Santos-Merino, M.; Nogales, J.; de la Cruz, F.; García, J. Microbial Oils as Nutraceuticals and Animal Feeds. https://scholar.google.com/scholar?hl=en&as_sdt=0%2C4&q=Gal%C3%A1n%2C+Beatriz%2C+et+al.+%22Microbial+oils+as+nutraceuticals+and+animal+feeds.%22+Health+consequences+of+microbial+interactions+with+hydrocarbons%2C+oils%2C+and+lipids.+Cham%3A+Springer+International+Publishing%2C+2020.+401-445.&btnG= (accessed on 9 Dec 2025).
- Chamodi, K. K. D.; Vu, N. T.; Domingos, J. A.; Loh, J. Y. Cellular Solutions: Evaluating Single-Cell Proteins as Sustainable Feed Alternatives in Aquaculture. Biology (Basel). 2025, 14(7), 764. [Google Scholar] [CrossRef] [PubMed]
- Trinci, A. P. J. Evolution of the Quorn® Myco-Protein Fungus, Fusarium Graminearum A3/5. Microbiology (N. Y). 1994, 140(9), 2181–2188. [Google Scholar] [CrossRef]
- Ye, L.; Bogicevic, B.; Bolten, C. J.; Wittmann, C. Single-Cell Protein: Overcoming Technological and Biological Challenges towards Improved Industrialization. Curr. Opin. Biotechnol. 2024, 88, 103171. [Google Scholar] [CrossRef]
- Sharif, M.; Zafar, M. H.; Aqib, A. I.; Saeed, M.; Farag, M. R.; Alagawany, M. Single Cell Protein: Sources, Mechanism of Production, Nutritional Value and Its Uses in Aquaculture Nutrition. Aquaculture 2021, 531, 735885. [Google Scholar] [CrossRef]
- Al-Mudhafr, A. Microbiological Sources and Nutritional Value of Single Cell Protein (SCP). Nutrition and Food Processing 2019, 2(2), 01–02. [Google Scholar] [CrossRef] [PubMed]
- Braho, V.; Sar, T.; Taherzadeh, M. J. Cultivation of Edible Filamentous Fungi on Pomegranate By-Products as Feedstocks to Produce Mycoprotein. Systems Microbiology and Biomanufacturing 2024, 4(2), 675–686. [Google Scholar] [CrossRef]
- Abdul Manan, M.; Webb, C. Design Aspects of Solid State Fermentation as Applied to Microbial Bioprocessing. 2017. [Google Scholar] [CrossRef]
- Post, M. J. Cultured Meat from Stem Cells: Challenges and Prospects. Meat Science. November 2012, pp 297–301. [CrossRef]
- Arsov, A.; Tsigoriyna, L.; Batovska, D.; Armenova, N.; Mu, W.; Zhang, W.; Petrov, K.; Petrova, P. Bacterial Degradation of Antinutrients in Foods: The Genomic Insight. Foods 2024, Vol. 13 2024, 13(15). [Google Scholar] [CrossRef]
- Wei, Z.; Wang, H.; Zhong, R.; Chen, L.; Vigors, S.; Zhang, H. Biodegradation of α-Solanine and α-Chaconine: Insights into Microbial Detoxification and Enzymatic Deglycosylation Pathways. Food Chem. X 2025, 31, 102968. [Google Scholar] [CrossRef]
- United Nations. Goals 12: Ensure sustainable consumption and production patterns.
- Khumchai, J.; Wongchai, A.; On-uma, R.; Sabour, A.; Alshiekheid, M.; Narayanan, M.; Karuppusamy, I.; Pugazhendhi, A.; Brindhadevi, K.; Lan Chi, N. T. A Viable Bioremediation Strategy for Treating Paper and Pulp Industry Effluents and Assessing the Prospect of Resulted Bacterial Biomass as Single Cell Protein (SCP) Using Indigenous Bacterial Species. Chemosphere 2022, 304. [Google Scholar] [CrossRef]
- Rodero, M. del R.; Magdalena, J. A.; Steyer, J. P.; Escudié, R.; Capson-Tojo, G. Potential of Enriched Phototrophic Purple Bacteria for H2 Bioconversion into Single Cell Protein. Science of The Total Environment 2024, 908, 168471. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Yang, X.; Zeng, D.; Su, Y.; Zhang, Y. Microbial Conversion of Syngas to Single Cell Protein: The Role of Carbon Monoxide. Chemical Engineering Journal 2022, 450, 138041. [Google Scholar] [CrossRef]
- Persistence Market Research. Single Cell Protein Market Thrives with a Robust 6.8% CAGR, Set to Reach US$ 6,963.0 Million by 2033 | According to Persistence Market Research. https://finance.yahoo.com/news/single-cell-protein-market-thrives-180100370.html?guccounter=1&guce_referrer=aHR0cHM6Ly93d3cuZ29vZ2xlLmNvbS8&guce_referrer_sig=AQAAAK4KqKwKC04eyWUYpllc5McrKwMOH0rXTIlYOycINiqSUQjOk1STSIhnh3pAJ7o8NQHB94IgXURrozgAsD9NDQ7Sr9LDBlggYJ04_sYlys_Ko7krMLLqlpf_k7PkRDvgqWnFJ9OtcwUoi8lKljUAP0qj_ZqwkaRKUwcw3fcTIDhU (accessed on 7 Apr 2026).
- Grasso, A. C.; Hung, Y.; Olthof, M. R.; Verbeke, W.; Brouwer, I. A. Older Consumers’ Readiness to Accept Alternative, More Sustainable Protein Sources in the European Union. 2019. [CrossRef]
- Pojić, M.; Mišan, A.; Tiwari, B. Eco-Innovative Technologies for Extraction of Proteins for Human Consumption from Renewable Protein Sources of Plant Origin. Trends in Food Science and Technology. Elsevier Ltd May 1, 2018, pp 93–104. [CrossRef]
- Jeske, S.; Zannini, E.; Arendt, E. K. Past, Present and Future: The Strength of Plant-Based Dairy Substitutes Based on Gluten-Free Raw Materials. Food Research International 2018, 110, 42–51. [Google Scholar] [CrossRef]
- Boye, J. I.; Aksay, S.; Roufik, S.; Ribéreau, S.; Mondor, M.; Farnworth, E.; Rajamohamed, S. H. Comparison of the Functional Properties of Pea, Chickpea and Lentil Protein Concentrates Processed Using Ultrafiltration and Isoelectric Precipitation Techniques. Food Research International 2010, 43(2), 537–546. [Google Scholar] [CrossRef]
- Hertzler, S. R.; Lieblein-Boff, J. C.; Weiler, M.; Allgeier, C. Plant Proteins: Assessing Their Nutritional Quality and Effects on Health and Physical Function. Nutrients. MDPI AG December 1, 2020, pp 1–27. [CrossRef]
- Zhou, H.; Hu, Y.; Tan, Y.; Zhang, Z.; McClements, D. J. Digestibility and Gastrointestinal Fate of Meat versus Plant-Based Meat Analogs: An in Vitro Comparison. Food Chem. 2021, 364. [Google Scholar] [CrossRef] [PubMed]
- Reynaud, Y.; Buffière, C.; Cohade, B.; Vauris, M.; Liebermann, K.; Hafnaoui, N.; Lopez, M.; Souchon, I.; Dupont, D.; Rémond, D. True Ileal Amino Acid Digestibility and Digestible Indispensable Amino Acid Scores (DIAASs) of Plant-Based Protein Foods. Food Chem. 2021, 338. [Google Scholar] [CrossRef] [PubMed]
- You, X. Y.; Ding, Y.; Bu, Q. Y.; Wang, Q. H.; Zhao, G. P. Nutritional, Textural, and Sensory Attributes of Protein Bars Formulated with Mycoproteins. Foods 2024, 13(5). [Google Scholar] [CrossRef]
- Zahra Ahangi; Seyed Shojaosadati; Houshang Nikoopour. Study of Mycoprotein Production Using Fusarium Oxysporum and Reduction of RNA Content. Pakistan Journal of Nutrition 2008, 7(2), 240–243. [CrossRef]
- Perkins, D. D.; Davis, R. H. GUEST COMMENTARY Evidence for Safety of Neurospora Species for Academic and Commercial Uses; 2000; Vol. 66.
- FDA. GRAS Notice 1117, Mycelial Biomass from Neurospora Crassa. 2025. [Google Scholar]
- Wang, Z.; Lopez-Giraldez, F.; Slot, J.; Yarden, O.; Trail, F.; Townsend, J. P.; Hug, L. A. Secondary Metabolism Gene Clusters Exhibit Increasingly Dynamic and Differential Expression during Asexual Growth, Conidiation, and Sexual Development in Neurospora Crassa. mSystems 2022, 7(3). [Google Scholar] [CrossRef]
- Perkins, D. D. Neurospora: The Organism behind the Molecular Revolution. Genetics 1992, 130(4), 687–701. [Google Scholar] [CrossRef]
- Singh Nee Nigam, P.; Pandey, A. Biotechnology for Agro-Industrial Residues Utilisation: Utilisation of Agro-Residues; Springer Netherlands, 2009. [Google Scholar] [CrossRef]
- Wiebe, M. Myco-Protein from Fusarium Venenatum: A Well-Established Product for Human Consumption. Applied Microbiology and Biotechnology 2002, 421–427. [Google Scholar] [CrossRef]
- Dean, D.; Rombach, M.; de Koning, W.; Vriesekoop, F.; Satyajaya, W.; Yuliandari, P.; Anderson, M.; Mongondry, P.; Urbano, B.; Luciano, C. A. G.; et al. Understanding Key Factors Influencing Consumers’ Willingness to Try, Buy, and Pay a Price Premium for Mycoproteins. Nutrients 2022, 14(16). [Google Scholar] [CrossRef] [PubMed]
- PATRICK E. McGOVERN. THE NEAR EASTERN CHALLENGE. Uncorking the Past, 2009, 60.
- Steinkraus, K. H. Use of Lactobacillus from Pulque in Sourdough. Handbook of Indigenous Fermented Foods. 1996, 52–72. [Google Scholar]
- Pasteur (1866) Études Sur Le Vi.
- Chain, E.; Florey, H. W.; Gardener, A. D.; Heatley, N.; Jennings, M. A. THE CLASSIC: Penicillin as a Chemotherapeutic Agent. Clin Orthop Relat Res. 1940, 439, 23–26. [Google Scholar] [CrossRef]
- Edwards, G. Myco-Protein - the Development of a New Food [Fusarium Graminearum]. Food Laboratory Newsletter 1986, No. 6, 21–24. [Google Scholar]
- Denny, A.; Aisbitt, B.; Lunn, J. Mycoprotein and Health. Nutrition Bulletin. December 2008, pp 298–310. [CrossRef]
- Cedeno, F. R. P.; Olubiyo, O. J.; Ferreira, S. From Microbial Proteins to Cultivated Meat for Alternative Meat-like Products: A Review on Sustainable Fermentation Approaches. Journal of Biological Engineering 2025 19:1 2025, 19(1), 44. [Google Scholar] [CrossRef] [PubMed]
- Fox, P. F.; Guineee, T. P.; Cogann, T. M.; Mcsweeney, P. L. H. Fundamentals of Cheese Science Second Edition.
- Stanbury, P. F.; Whitaker, Allan.; Hall, S. J. Principles of Fermentation Technology; Pergamon, 2008. [Google Scholar]
- Lee, S. Y.; Ra, C. H. Comparison of Liquid and Solid-State Fermentation Processes for the Production of Enzymes and Beta-Glucan from Hulled Barley. J. Microbiol. Biotechnol. 2021, 32(3), 317. [Google Scholar] [CrossRef]
- Hutkins, R. W. Microbiology and Technology of Fermented Foods. Microbiology and Technology of Fermented Foods 2007, 1–473. [Google Scholar] [CrossRef]
- Boulton, R. B.; Singleton, V. L.; Bisson, L. F.; Kunkee, R. E. Principles and Practices of Winemaking. Principles and Practices of Winemaking 1999. [Google Scholar] [CrossRef]
- Soibam, H.; Ayam, V. S.; Chakraborty, I.; Chandra, B.; Viswavidyalaya, K. Natural Resource Utilization by Acetic Acid Fermentation: A Method for Post Harvest Management. Article in Advances in Bioresearch 2014. [Google Scholar] [CrossRef]
- Paddon, C. J.; Westfall, P. J.; Pitera, D. J.; Benjamin, K.; Fisher, K.; McPhee, D.; Leavell, M. D.; Tai, A.; Main, A.; Eng, D.; et al. High-Level Semi-Synthetic Production of the Potent Antimalarial Artemisinin. Nature 2013, 496(7446), 528–532. [Google Scholar] [CrossRef]
- Chai, K. F.; Ng, K. R.; Samarasiri, M.; Chen, W. N. Precision Fermentation to Advance Fungal Food Fermentations. Current Opinion in Food Science. Elsevier Ltd October 1, 2022. [CrossRef]
- Augustin, M. A.; Hartley, C. J.; Maloney, G.; Tyndall, S. Innovation in Precision Fermentation for Food Ingredients. Critical Reviews in Food Science and Nutrition. Taylor and Francis Ltd. 2024, pp 6218–6238. [CrossRef]
- Wang, W. Y.; Wu, S. H.; Xie, Y. H.; Zhong, M.; Wei, M. L.; Li, Z. Y.; Long, X. F.; Niu, F. X. A High-Throughput Screening Procedure (Py-Fe3+) for Enhancing Ethanol Production by Saccharomyces Cerevisiae Using ARTP Random Mutagenesis. Processes 2022, 10(11). [Google Scholar] [CrossRef]
- Nielsen, J.; Larsson, C.; van Maris, A.; Pronk, J. Metabolic Engineering of Yeast for Production of Fuels and Chemicals. Current Opinion in Biotechnology. June 2013, pp 398–404. [CrossRef]
- Fink, M.; Cserjan-Puschmann, M.; Reinisch, D.; Striedner, G. High-Throughput Microbioreactor Provides a Capable Tool for Early Stage Bioprocess Development. Scientific Reports 2021 11:1 2021, 11(1), 2056. [Google Scholar] [CrossRef]
- Wang, S.; Copeland, L. Effect of Acid Hydrolysis on Starch Structure and Functionality: A Review. Crit. Rev. Food Sci. Nutr. 2015, 55(8), 1081–1097, REQUESTEDJOURNAL:JOURNAL:BFSN20. [Google Scholar] [CrossRef] [PubMed]
- Feldman, D.; Kowbel, D. J.; Cohen, A.; Glass, N. L.; Hadar, Y.; Yarden, O. Identification and Manipulation of Neurospora Crassa Genes Involved in Sensitivity to Furfural. Biotechnol. Biofuels 2019, 12(1). [Google Scholar] [CrossRef] [PubMed]
- Cabeza, C.; Ahmed, A. E. G.; Minauf, M.; Wieland, K.; Harasek, M. Starch Hydrolysates, Their Impurities and the Role of Membrane-Based Technologies as a Promising Sustainable Purification Method at Industrial Scale. Food Research International 2025, 209, 116300. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Ma, L.; Duan, W.; Gao, W.; Fang, Y.; Guo, L.; Yuan, C.; Wu, Z.; Cui, B. Maltogenic Amylase: Its Structure, Molecular Modification, and Effects on Starch and Starch-Based Products. Carbohydr. Polym. 2023, 319, 121183. [Google Scholar] [CrossRef]
- Hii, S. L.; Tan, J. S.; Ling, T. C.; Ariff, A. Bin. Pullulanase: Role in Starch Hydrolysis and Potential Industrial Applications. Enzyme Res. 2012, 2012, 921362. [Google Scholar] [CrossRef] [PubMed]
- Collares, R. M.; Miklasevicius, L. V. S.; Bassaco, M. M.; Salau, N. P. G.; Mazutti, M. A.; Bisognin, D. A.; Terra, L. M. Optimization of Enzymatic Hydrolysis of Cassava to Obtain Fermentable Sugars. J. Zhejiang Univ. Sci. B 2012, 13(7), 579. [Google Scholar] [CrossRef]
- Ghinea, C.; Ungureanu-Comăniță, E. D.; Țâbuleac, R. M.; Oprea, P. S.; Coșbuc, E. D.; Gavrilescu, M. Cost-Benefit Analysis of Enzymatic Hydrolysis Alternatives for Food Waste Management. Foods 2025, 14(3), 488. [Google Scholar] [CrossRef]
- Vieira, B. F.; Bueno, L. C. G.; Ferrari, L. D.; Masarin, F.; de Paula, A. V.; Andrade, G. S. S. A Novel Pre-Treatment of Brewery Spent Grains Using Mycelium-Bound Cellulase from Aspergillus Oryzae for Biogas Production. Biomass Bioenergy 2025, 194, 107691. [Google Scholar] [CrossRef]
- Rojas-Chamorro, J. A.; Romero, I.; López-Linares, J. C.; Castro, E. Brewer’s Spent Grain as a Source of Renewable Fuel through Optimized Dilute Acid Pretreatment. Renew. Energy 2020, 148, 81–90. [Google Scholar] [CrossRef]
- Ozer Uyar, G. E.; Uyar, B. Potato Peel Waste Fermentation by Rhizopus Oryzae to Produce Lactic Acid and Ethanol. Food Sci. Nutr. 2023, 11(10), 5908. [Google Scholar] [CrossRef]
- Tadesse, H. M.; Atnafu, T.; Kassahun, E.; Tessema, I.; Abewaa, M.; Tibebu, S. Optimization of Bioethanol Production from a Brewers’ Spent Grain and Sugarcane Molasses Mixture Utilizing Saccharomyces Cerevisiae. Biomass Conversion and Biorefinery 2025 15:14 2025, 15(14), 20765–20788. [Google Scholar] [CrossRef]
- Punia, S.; Sandhu, K. S.; Grasso, S.; Purewal, S. S.; Kaur, M.; Siroha, A. K.; Kumar, K.; Kumar, V.; Kumar, M. Aspergillus Oryzae Fermented Rice Bran: A Byproduct with Enhanced Bioactive Compounds and Antioxidant Potential. Foods 2021, Vol. 10 2020, 10(1). [Google Scholar] [CrossRef]
- Zhang, B.; Zhan, B.; Bao, J. Reframing Biorefinery Processing Chain of Corn Fiber for Cellulosic Ethanol Production. Ind. Crops Prod. 2021, 170, 113791. [Google Scholar] [CrossRef]
- Solanki, A.; Kumar, D.; Sharma, P. A Mini-Review on Agro Waste Mediated Technologies Used for Landfill Leachate Treatment. Journal of Water Process Engineering 2024, 57(1), 104685. [Google Scholar] [CrossRef]
- Megavitry, R.; Silamat, E. Utilisation of Agricultural Wastes as Alternative Raw Materials in Fermented Food Production. West Science Agro 2024, 2(03), 109–122. [Google Scholar] [CrossRef]
- Bamigbade, G. B.; Oyelami, O. I.; Babalola, O. O.; Adewolu, A.; Omemu, A. M.; Ogunsanya, T. F.; Sanusi, J. O. F.; Daniel, O. M. An Updated Comprehensive Review on Waste Valorization: Informetric Analysis, Current Insights and Future Perspectives on Cereal Waste and Byproduct Utilization for Sustainable Industrial Applications. Bioresour. Technol. 2025, 418, 131868. [Google Scholar] [CrossRef]
- Rasool, K.; Hussain, S.; Shahzad, A.; Miran, W.; Mahmoud, K. A.; Ali, N.; Almomani, F.; Rasool, K.; Mahmoud, K. A.; Hussain, S.; et al. Comprehensive Insights into Sustainable Conversion of Agricultural and Food Waste into Microbial Protein for Animal Feed Production. Reviews in Environmental Science and Bio/Technology 2023 22:2 2023, 22(2), 527–562. [Google Scholar] [CrossRef]
- Koukoumaki, D. I.; Tsouko, E.; Papanikolaou, S.; Ioannou, Z.; Diamantopoulou, P.; Sarris, D. Recent Advances in the Production of Single Cell Protein from Renewable Resources and Applications. Carbon Resources Conversion 2024, 7(2), 100195. [Google Scholar] [CrossRef]
- Rashwan, A. K.; Younis, H. A.; Abdelshafy, A. M.; Osman, A. I.; Eletmany, M. R.; Hafouda, M. A.; Chen, W. Plant Starch Extraction, Modification, and Green Applications: A Review. Environmental Chemistry Letters 2024 22:5 2024, 22(5), 2483–2530. [Google Scholar] [CrossRef]
- Junejo, S. A.; Flanagan, B. M.; Zhang, B.; Dhital, S. Starch Structure and Nutritional Functionality – Past Revelations and Future Prospects. Carbohydr. Polym. 2022, 277. [Google Scholar] [CrossRef]
- Wang, Y.; Ou, X.; Al-Maqtari, Q. A.; He, H. J.; Othman, N. Evaluation of Amylose Content: Structural and Functional Properties, Analytical Techniques, and Future Prospects. Food Chem. X 2024, 24, 101830. [Google Scholar] [CrossRef]
- Prückler, M.; Siebenhandl-Ehn, S.; Apprich, S.; Höltinger, S.; Haas, C.; Schmid, E.; Kneifel, W. Wheat Bran-Based Biorefinery 1: Composition of Wheat Bran and Strategies of Functionalization. LWT - Food Science and Technology 2014, 56(2), 211–221. [Google Scholar] [CrossRef]
- Zhang, C.; Xue, W.; Li, T.; Wang, L. Understanding the Relationship between the Molecular Structure and Physicochemical Properties of Soft Rice Starch. Foods 2023, 12(19), 3611. [Google Scholar] [CrossRef]
- Waterschoot, J.; Gomand, S. V.; Fierens, E.; Delcour, J. A. Production, Structure, Physicochemical and Functional Properties of Maize, Cassava, Wheat, Potato and Rice Starches. Starch - Stärke 2015, 67(1–2), 14–29. [Google Scholar] [CrossRef]
- Ranum, P.; Peña-Rosas, J. P.; Garcia-Casal, M. N. Global Maize Production, Utilization, and Consumption. Ann. N. Y. Acad. Sci. 2014, 1312(1), 105–112. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Jiang, H.; Campbell, M.; Blanco, M.; Jane, J. lin. Characterization of Maize Amylose-Extender (Ae) Mutant Starches. Part I: Relationship between Resistant Starch Contents and Molecular Structures. Carbohydr. Polym. 2008, 74(3), 396–404. [Google Scholar] [CrossRef]
- Singh, N.; Singh, J.; Kaur, L.; Sodhi, N. S.; Gill, B. S. Morphological, Thermal and Rheological Properties of Starches from Different Botanical Sources. Food Chem. 2003, 81(2), 219–231. [Google Scholar] [CrossRef]
- Pulido Diaz, A.; Lourdin, D.; Della Valle, G.; Fernandez Quintero, A.; Ceballos, H.; Tran, T.; Dufour, D. Thermomechanical Characterization of an Amylose-Free Starch Extracted from Cassava (Manihot Esculenta, Crantz). 2016. [Google Scholar] [CrossRef] [PubMed]
- Riley, C. K.; Wheatley, A. O.; Asemota, H. N. Isolation and Characterization of Starches from Eight Dioscorea Alata Cultivars Grown in Jamaica. Afr. J. Biotechnol. 2006, 5(17), 1528–1536. [Google Scholar]
- Buléon, A.; Colonna, P.; Planchot, V.; Ball, S. Starch Granules: Structure and Biosynthesis. Int. J. Biol. Macromol. 1998, 23(2), 85–112. [Google Scholar] [CrossRef]
- Dar, M. Z.; Deepika, K.; Jan, K.; Swer, T. L.; Kumar, P.; Verma, R.; Verma, K.; Prakash, K. S.; Jan, S.; Bashir, K. Modification of Structure and Physicochemical Properties of Buckwheat and Oat Starch by γ-Irradiation. Int J Biol Macromol 2018, 108, 1348–1356. [Google Scholar] [CrossRef]
- Bashir, K.; Aggarwal, M. Physicochemical, Structural and Functional Properties of Native and Irradiated Starch: A Review. Journal of Food Science and Technology 2019 56:2 2019, 56(2), 513–523. [Google Scholar] [CrossRef]
- El Farkhani, M.; Dadou, S.; El Miz, Y.; Elyoussfi, A.; El Miz, M.; Salhi, A.; Koudad, M.; Benchat, N. A Review of the Chemical Modification and Applications of Starch. In BIO Web of Conferences; EDP Sciences, 2024; Vol. 109. [CrossRef]
- Mussatto, S. I. Brewer’s Spent Grain: A Valuable Feedstock for Industrial Applications. J. Sci. Food Agric. 2014, 94(7), 1264–1275. [Google Scholar] [CrossRef]
- Wilkinson, S.; Smart, K. A.; James, S.; Cook, D. J. Bioethanol Production from Brewers Spent Grains Using a Fungal Consolidated Bioprocessing (CBP) Approach. BioEnergy Research 2016 10:1 2016, 10(1), 146–157. [Google Scholar] [CrossRef]
- Mussatto, S. I.; Fernandes, M.; Dragone, G.; Mancilha, I. M.; Roberto, I. C. Brewer’s Spent Grain as Raw Material for Lactic Acid Production by Lactobacillus Delbrueckii. Biotechnology Letters 2007 29:12 2007, 29(12), 1973–1976. [Google Scholar] [CrossRef]
- Waters, D. M.; Jacob, F.; Titze, J.; Arendt, E. K.; Zannini, E. Fibre, Protein and Mineral Fortification of Wheat Bread through Milled and Fermented Brewer’s Spent Grain Enrichment. European Food Research and Technology 2012 235:5 2012, 235(5), 767–778. [Google Scholar] [CrossRef]
- Mitri, S.; Salameh, S.-J.; Khelfa, A.; Léonard, E.; Maroun, R.; Louka, N.; Koubaa, M.; Veeravalli, S.; Leonard, E.; Maroun, R. G. Valorization of Brewers’ Spent Grains: Pretreatments and Fermentation, a Review Fermentation Valorization of Brewers’ Spent Grains: Pretreatments and Fermentation, a Review. 2022. [Google Scholar] [CrossRef]
- Nyhan, L.; Sahin, A. W.; Schmitz, H. H.; Siegel, J. B.; Arendt, E. K. Brewers’ Spent Grain: An Unprecedented Opportunity to Develop Sustainable Plant-Based Nutrition Ingredients Addressing Global Malnutrition Challenges. J. Agric. Food Chem. 2023, 71(28), 10543–10564. [Google Scholar] [CrossRef]
- Kavalopoulos, M.; Stoumpou, V.; Christofi, A.; Mai, S.; Barampouti, E. M.; Moustakas, K.; Malamis, D.; Loizidou, M. Sustainable Valorisation Pathways Mitigating Environmental Pollution from Brewers’ Spent Grains. Environmental Pollution 2021, 270, 116069. [Google Scholar] [CrossRef]
- Celus, I.; Brijs, K.; Delcour, J. A. Enzymatic Hydrolysis of Brewers’ Spent Grain Proteins and Technofunctional Properties of the Resulting Hydrolysates. J. Agric. Food Chem. 2007, 55(21), 8703–8710. [Google Scholar] [CrossRef] [PubMed]
- Jaeger, A.; Sahin, A. W.; Nyhan, L.; Zannini, E.; Arendt, E. K.; Casanova, F.; Jaeger, A.; Sahin, A. W.; Nyhan, L.; Zannini, E.; et al. Functional Properties of Brewer’s Spent Grain Protein Isolate: The Missing Piece in the Plant Protein Portfolio. Foods 2023, Vol. 12, Page 798 2023, 12(4), 798. [Google Scholar] [CrossRef] [PubMed]
- Vogelsang-O’Dwyer, M.; Bez, J.; Petersen, I. L.; Joehnke, M. S.; Detzel, A.; Busch, M.; Krueger, M.; Ispiryan, L.; O’Mahony, J. A.; Arendt, E. K.; et al. Techno-Functional, Nutritional and Environmental Performance of Protein Isolates from Blue Lupin and White Lupin. Foods 2020, Vol. 9, Page 230 2020, 9(2), 230. [Google Scholar] [CrossRef] [PubMed]
- Herreman, L.; Nommensen, P.; Pennings, B.; Laus, M. C. Comprehensive Overview of the Quality of Plant- And Animal-Sourced Proteins Based on the Digestible Indispensable Amino Acid Score. Food Sci. Nutr. 2020, 8(10), 5379–5391. [Google Scholar] [CrossRef]
- Mathai, J. K.; Liu, Y.; Stein, H. H. Values for Digestible Indispensable Amino Acid Scores (DIAAS) for Some Dairy and Plant Proteins May Better Describe Protein Quality than Values Calculated Using the Concept for Protein Digestibility-Corrected Amino Acid Scores (PDCAAS). British Journal of Nutrition 2017, 117(4), 490–499. [Google Scholar] [CrossRef]
- Su, Y.; Du, R.; Guo, H.; Cao, M.; Wu, Q.; Su, R.; Qi, W.; He, Z. Fractional Pretreatment of Lignocellulose by Alkaline Hydrogen Peroxide: Characterization of Its Major Components. Food and Bioproducts Processing 2015, 94, 322–330. [Google Scholar] [CrossRef]
- Liguori, R.; Soccol, C. R.; de Souza Vandenberghe, L. P.; Woiciechowski, A. L.; Faraco, V. Second Generation Ethanol Production from Brewers’ Spent Grain. Energies 2015, Vol. 8, Pages 2575-2586 2015, 8(4), 2575–2586. [Google Scholar] [CrossRef]
- Nunes, L. F.; Ugalde, G. A.; Anschau, K. F.; Müller, E. I.; Tres, M. V.; Zabot, G. L.; Kuhn, R. C. Clean Production of Sugars from Brewer’s Spent Grains Using Subcritical Water Hydrolysis and Steam Explosion. Sustainable Chemistry 2024, 5(4), 308–323. [Google Scholar] [CrossRef]
- Paz, A.; Outeiriño, D.; Pérez Guerra, N.; Domínguez, J. M. Enzymatic Hydrolysis of Brewer’s Spent Grain to Obtain Fermentable Sugars. Bioresour. Technol. 2019, 275, 402–409. [Google Scholar] [CrossRef] [PubMed]
- Xiros, C.; Christakopoulos, P. Enhanced Ethanol Production from Brewer’s Spent Grain by a Fusarium Oxysporum Consolidated System. Biotechnol. Biofuels 2009, 2. [Google Scholar] [CrossRef]
- Cooray, S. T.; Chen, W. N. Valorization of Brewer’s Spent Grain Using Fungi Solid-State Fermentation to Enhance Nutritional Value. J. Funct. Foods 2018, 42, 85–94. [Google Scholar] [CrossRef]
- Rusbjerg-Weberskov, C. E.; Foley, J. D.; Yang, L.; Terp, M.; Gregersen Echers, S.; Orlien, V.; Lübeck, M. Combined Rhizopus Oryzae Fermentation and Protein Extraction of Brewer’s Spent Grain Improves Protein Functionality. Food and Bioprocess Technology 2025 18:11 2025, 18(11), 9574–9593. [Google Scholar] [CrossRef]
- Sganzerla, W. G.; Costa, J. M.; Tena-Villares, M.; Buller, L. S.; Mussatto, S. I.; Forster-Carneiro, T. Dry Anaerobic Digestion of Brewer’s Spent Grains toward a More Sustainable Brewery: Operational Performance, Kinetic Analysis, and Bioenergy Potential. Fermentation 2023, 9(1). [Google Scholar] [CrossRef]
- Drakopoulos, D.; Sulyok, M.; Krska, R.; Logrieco, A. F.; Vogelgsang, S. Raised Concerns about the Safety of Barley Grains and Straw: A Swiss Survey Reveals a High Diversity of Mycotoxins and Other Fungal Metabolites. Food Control 2021, 125, 107919. [Google Scholar] [CrossRef]
- Sharif, M. K.; Butt, M. S.; Anjum, F. M.; Khan, S. H. Rice Bran: A Novel Functional Ingredient. Crit. Rev. Food Sci. Nutr., 2014, 54 (6), 807–816; JOURNAL:JOURNAL:BFSN18;WGROUP:STRING:PUBLICATION. [CrossRef]
- Muthayya, S.; Sugimoto, J. D.; Montgomery, S.; Maberly, G. F. An Overview of Global Rice Production, Supply, Trade, and Consumption. Ann. N. Y. Acad. Sci. 2014, 1324(1), 7–14. [Google Scholar] [CrossRef]
- Research Nester Rice Husk Ash Market Outlook. 2025. https://www.researchnester.com/reports/rice-husk-ash-market/3376 (accessed on 11 Dec 2025).
- USDA. Rice - Rice Sector at a Glance. https://www.ers.usda.gov/topics/crops/rice/rice-sector-at-a-glance (accessed on 11 Dec 2025).
- FAO. FAO Rice Price Update. https://www.fao.org/markets-and-trade/commodities/rice/fao-rice-price-update/en (accessed on 11 Dec 2025).
- Jenkins, B. M.; Baxter, L. L.; Miles, T. R.; Miles, T. R. Combustion Properties of Biomass. Fuel Processing Technology 1998, 54(1–3), 17–46. [Google Scholar] [CrossRef]
- Yoswathana, N.; Phuriphipat, P.; Treyawutthiwat, P.; Eshtiaghi, M. N. Bioethanol Production from Rice Straw. Energy Research Journal 2010, 1(1), 26–31. [Google Scholar] [CrossRef]
- Manzoor, A.; Pandey, V. K.; Dar, A. H.; Fayaz, U.; Dash, K. K.; Shams, R.; Ahmad, S.; Bashir, I.; Fayaz, J.; Singh, P.; et al. Rice Bran: Nutritional, Phytochemical, and Pharmacological Profile and Its Contribution to Human Health Promotion. Food Chemistry Advances 2023, 2, 100296. [Google Scholar] [CrossRef]
- Faria, S. A. dos S. C.; Bassinello, P. Z.; Penteado, M. de V. C. Nutritional Composition of Rice Bran Submitted to Different Stabilization Procedures. Brazilian Journal of Pharmaceutical Sciences 2012, 48(4), 651–657. [Google Scholar] [CrossRef]
- Siepmann, F. B.; Kalschne, D. L.; Zabotti, C.; de Moraes Flores, E. L.; Canan, C.; Colla, E. Feasibility of Bioethanol Production from Rice Bran. Semina:Ciencias Agrarias 2020, 41(6), 2951–2966. [Google Scholar] [CrossRef]
- Oliveira, M. dos S.; Cipolatti, E. P.; Furlong, E. B.; Soares, L. de S. Compostos Fenólicos e Atividade Antioxidante Em Farelo de Arroz (Oryza Sativa) Fermentado. Ciencia e Tecnologia de Alimentos 2012, 32(3), 531–537. [Google Scholar] [CrossRef]
- Ghosh, S.; Chowdhury, R.; Bhattacharya, P. Sustainability of Cereal Straws for the Fermentative Production of Second Generation Biofuels: A Review of the Efficiency and Economics of Biochemical Pretreatment Processes. Appl. Energy 2017, 198, 284–298. [Google Scholar] [CrossRef]
- Morya, R.; Andrianantenaina, F. H.; Singh, S.; Pandey, A. K.; Kim, G. B.; Verma, J. P.; Kumar, G.; Raj, T.; Kim, S. H. Exploring Rice Straw as Substrate for Hydrogen Production: Critical Challenges and Opportunities. Environ. Technol. Innov. 2023, 31, 103153. [Google Scholar] [CrossRef]
- Zhang, W.; Xu, X.; Yuan, Y.; Wang, Z. Sustainable Application of Rice-Waste for Fuels and Valuable Chemicals-a Mini Review. Front. Chem. 2023, 11, 1225073. [Google Scholar] [CrossRef]
- Gohel, V.; Duan, G. No-Cook Process for Ethanol Production Using Indian Broken Rice and Pearl Millet. Int. J. Microbiol. 2012, 2012(1), 680232. [Google Scholar] [CrossRef]
- Babini, E.; Taneyo-Saa, D. L.; Tassoni, A.; Ferri, M.; Kraft, A.; Grän-Heedfeld, J.; Bretz, K.; Roda, A.; Michelini, E.; Calabretta, M. M.; et al. Microbial Fermentation of Industrial Rice-Starch Byproduct as Valuable Source of Peptide Fractions with Health-Related Activity. Microorganisms 2020, 8(7), 986. [Google Scholar] [CrossRef]
- Pandey, A.; Soccol, C. R.; Nigam, P.; Soccol, V. T. Biotechnological Potential of Agro-Industrial Residues. I: Sugarcane Bagasse. Bioresour. Technol. 2000, 74(1), 69–80. [Google Scholar] [CrossRef]
- FAOSTAT. Data on yield of crops and area harvested. https://www.fao.org/faostat/en/#data/QCL/visualize (accessed on 28 Jan 2026).
- Mukhtar, A.; Latif, S.; Barati, Z.; Müller, J. Valorization of Cassava By-Products: Cyanide Content and Quality Characteristics of Leaves and Peel. Applied Sciences 2023, Vol. 13 2023, 13(10). [Google Scholar] [CrossRef]
- Ezekiel, O. O.; Aworh, O. C.; Du Preez, J. C.; Steyn, L. Cultivation of Candida Utilis on Cassava Peel Hydrolysates for Single-Cell Protein Production. J. Food Sci. Eng. 2012, 2, 452–461. [Google Scholar] [CrossRef]
- Bayitse, R.; Hou, X.; Laryea, G.; Bjerre, A. B. Protein Enrichment of Cassava Residue Using Trichoderma Pseudokoningii (ATCC 26801). AMB Express 2015 5:1 2015, 5(1), 80. [Google Scholar] [CrossRef]
- Pandey, A.; Soccol, C. R.; Nigam, P.; Soccol, V. T.; Vandenberghe, L. P. S.; Mohan, R. Biotechnological Potential of Agro-Industrial Residues. II: Cassava Bagasse. Bioresour. Technol. 2000, 74(1), 81–87. [Google Scholar] [CrossRef]
- Arapoglou, D.; Varzakas, T.; Vlyssides, A.; Israilides, C. Ethanol Production from Potato Peel Waste (PPW). Waste Management 2010, 30(10), 1898–1902. [Google Scholar] [CrossRef]
- Gebrechristos, H. Y.; Ma, X.; Xiao, F.; He, Y.; Zheng, S.; Oyungerel, G.; Chen, W. Potato Peel Extracts as an Antimicrobial and Potential Antioxidant in Active Edible Film. Food Sci. Nutr. 2020, 8(12), 6338. [Google Scholar] [CrossRef]
- Arapoglou, D.; Varzakas, T.; Vlyssides, A.; Israilides, C. Ethanol Production from Potato Peel Waste (PPW). Waste Management 2010, 30(10), 1898–1902. [Google Scholar] [CrossRef]
- Maxwell, O. I.; Chinwuba, U. B.; Onyebuchukwu, M. G.; Maxwell, O. I.; Chinwuba, U. B.; Onyebuchukwu, M. G. Protein Enrichment of Potato Peels Using Saccharomyces Cerevisiae via Solid-State Fermentation Process. Advances in Chemical Engineering and Science 2019, 9(1), 99–108. [Google Scholar] [CrossRef]
- Khan, A. R.; Ahmad, B.; Khan, S. A.; Sultana, K.; Hassan, S. Utilization of Potato and Cucumber Peels in Producing Single-Cell Protein by Aspergillus Parasiticus Nrrl 21369. BIOSCIENCE RESEARCH 2021, 18(4), 2845–2850. [Google Scholar]
- Tropea, A.; Ferracane, A.; Albergamo, A.; Potortì, A. G.; Turco, V. Lo; Di Bella, G. Single Cell Protein Production through Multi Food-Waste Substrate Fermentation. Fermentation 2022, Vol. 8 2022, 8(3). [Google Scholar] [CrossRef]
- ISO. ISO 14040:2006 - Environmental management — Life cycle assessment — Principles and framework. https://www.iso.org/standard/37456.html (accessed on 7 Feb 2026).
- Hauschild, M. Z.; Rosenbaum, R. K.; Olsen, S. I. Life Cycle Assessment: Theory and Practice. Life Cycle Assessment: Theory and Practice 2017, 1–1216. [Google Scholar] [CrossRef]
- Jolliet, O.; Margni, M.; Charles, R.; Humbert, S.; Payet, J.; Rebitzer, G.; Rosenbaum, R. IMPACT 2002+: A New Life Cycle Impact Assessment Methodology; 2003. [Google Scholar]
- Bare, J. C.; Norris, G. A.; Pennington, D. W.; McKone, T. TRACI: The Tool for the Reduction and Assessment of Chemical and Other Environmental Impacts. Journal of Industrial Ecology. MIT Press Journals 2003, pp 49–78. [CrossRef]
- Smetana, S.; Mathys, A.; Knoch, A.; Heinz, V. Meat Alternatives: Life Cycle Assessment of Most Known Meat Substitutes. International Journal of Life Cycle Assessment 2015, 20(9), 1254–1267. [Google Scholar] [CrossRef]
- Fernández-López, L.; González-García, P.; Fernández-Ríos, A.; Aldaco, R.; Laso, J.; Martínez-Ibáñez, E.; Gutiérrez-Fernández, D.; Pérez-Martínez, M. M.; Marchisio, V.; Figueroa, M.; et al. Life Cycle Assessment of Single Cell Protein Production–A Review of Current Technologies and Emerging Challenges. Cleaner and Circular Bioeconomy 2024, 8, 100079. [Google Scholar] [CrossRef]
- Upcraft, T.; Tu, W. C.; Johnson, R.; Finnigan, T.; Van Hung, N.; Hallett, J.; Guo, M. Protein from Renewable Resources: Mycoprotein Production from Agricultural Residues. Green Chemistry 2021, 23(14), 5150–5165. [Google Scholar] [CrossRef]
- Risner, D.; McDonald, K. A.; Jones, C.; Spang, E. S. A Techno-Economic Model of Mycoprotein Production: Achieving Price Parity with Beef Protein. Front. Sustain. Food Syst. 2023, 7. [Google Scholar] [CrossRef]
| Region | Production per year | ||
| 1961 (million tons) | 2022 (million tons) | Relative change | |
| Africa | 3.681 | 22.742 | +518% |
| Asia | 9.034 | 155.082 | +1617% |
| Europe | 29.454 | 59.082 | +101% |
| North America | 19.582 | 63.992 | +227% |
| Oceania | 2.296 | 6.537 | +185% |
| South America | 6.516 | 48.023 | +637% |
| Protein | Fat | Ash | Nucleic acid | |
| Fungi | 50-55 | 2-8 | 4-19 | 7-10 |
| Macroalgae | 40-60 | 7-20 | 8-10 | 3-8 |
| Yeast | 55-60 | 2-6 | 5-10 | 6-12 |
| Bacteria | 72-85 | 1-3 | 3-7 | 8-12 |
| Waste | Fungi | Fermentation type | Results | References |
| BSG | Aspergillus oryzae | SmF + enzymatic hydrolysis | Increased methane yield and COD removal of 85.2% | [94] |
| BSG | Saccharomyces cerevisiae | SmF +Dilute phosphoric acid | 92% total sugar recovery | [95] |
| Potato peel waste | Rhizopus oryzae | SmF + enzymatic hydrolysis | Increase ethanol and lactic acid yield | [96] |
| BSG and sugarcane molasses | Saccharomyces cerevisae | Dilute acid hydrolysis +SmF | Increased reducing sugar, ethanol yield and 67.1% lignin removal | [97] |
| RB | Aspergillus oryzae | SSF | Increased Total phenolic content, condensed tannin content | [98] |
| Starch Source |
Amylose (%) | Amylopectin (%) | Amylose:Amylopectin | Relative crystallinity | References |
| Wheat | 20-30 | 70-80 | 20:80 to 30:70 | 27-30 | [108] |
| Rice | 15-35 | 65-85 | 15:85 to 35:65 | 34 | [109] |
| Rice (waxy) | <1 | >99 | <1:99 | 30-35 | [110] |
| Maize (corn) | 25-30 | 70-75 | 25:75 to 30:70 | 32-33 | [111] |
| Maize (high) | >50 | <50 | >50:<50 | 20-25 | [112] |
| Potato | 20-30 | 70-80 | 20:80 to 30:70 | 37-38 | [110] |
| Sweet potato | 15-25 | 75-85 | 15:85 to 25:75 | 33 | [113] |
| Cassava | 15-25 | 75-85 | 15:85 to 25:75 | 38-40 | [114] |
| Yam | 20-30 | 70-80 | 20:80 to 30:70 | 25-35 | [115] |
| Maize (waxy) | <1 | >99 | <1:99 | 35-40 | [116] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




