Submitted:
06 March 2026
Posted:
09 March 2026
You are already at the latest version
Abstract
Keywords:
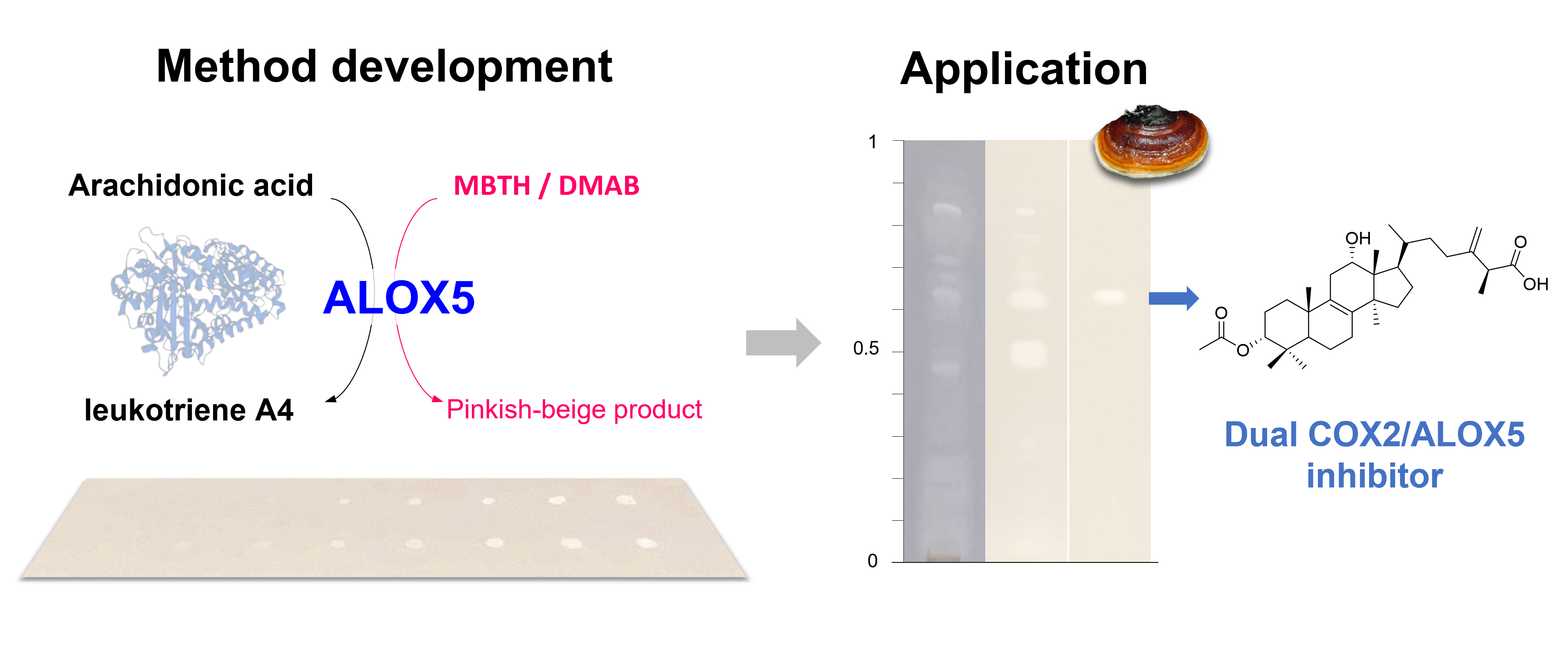
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Mushroom Material
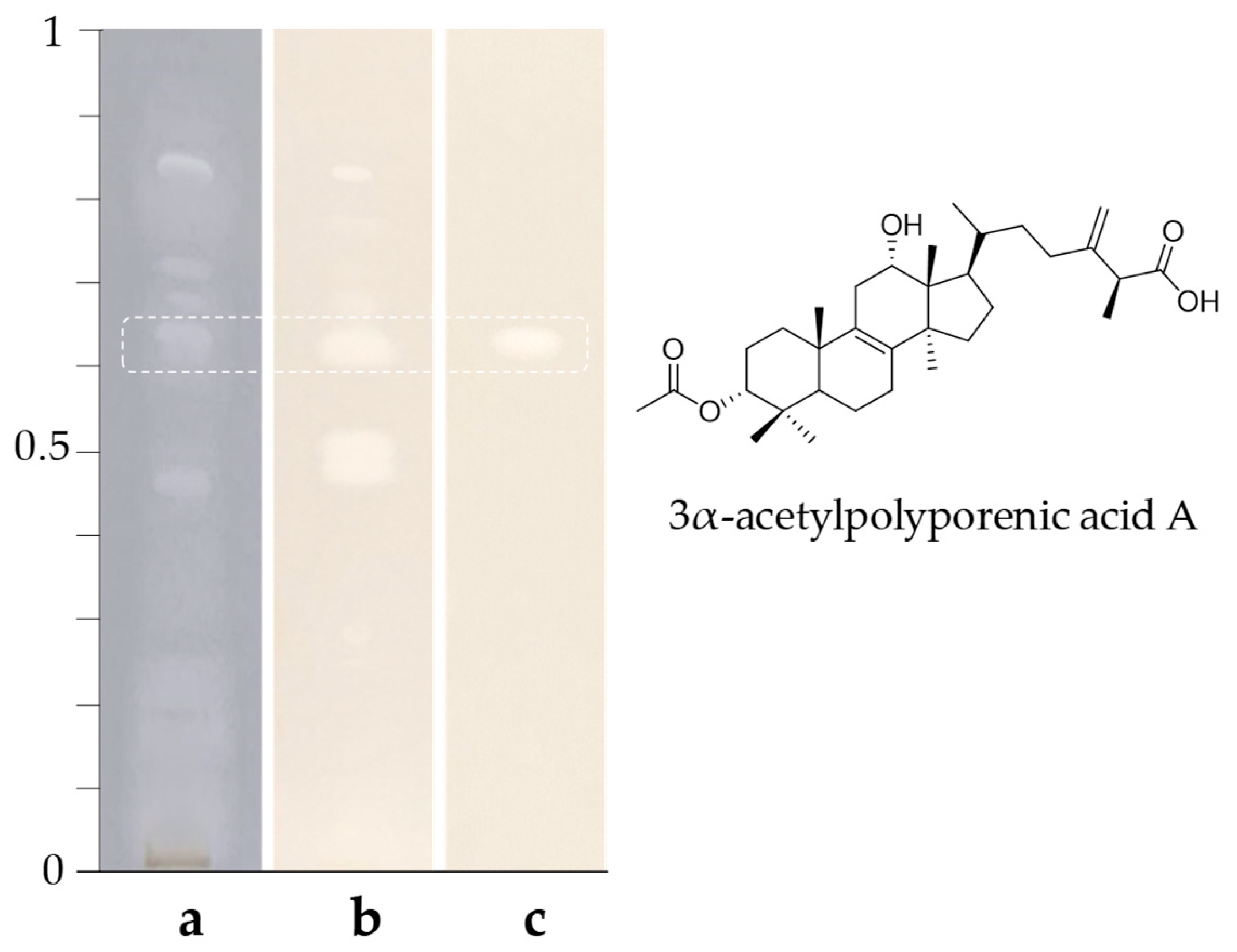
2.3. Purification and Identification of 3α-Acetylpolyporenic Acid A
2.4. Preparation of Solutions
2.5. Thin-Layer Chromatography
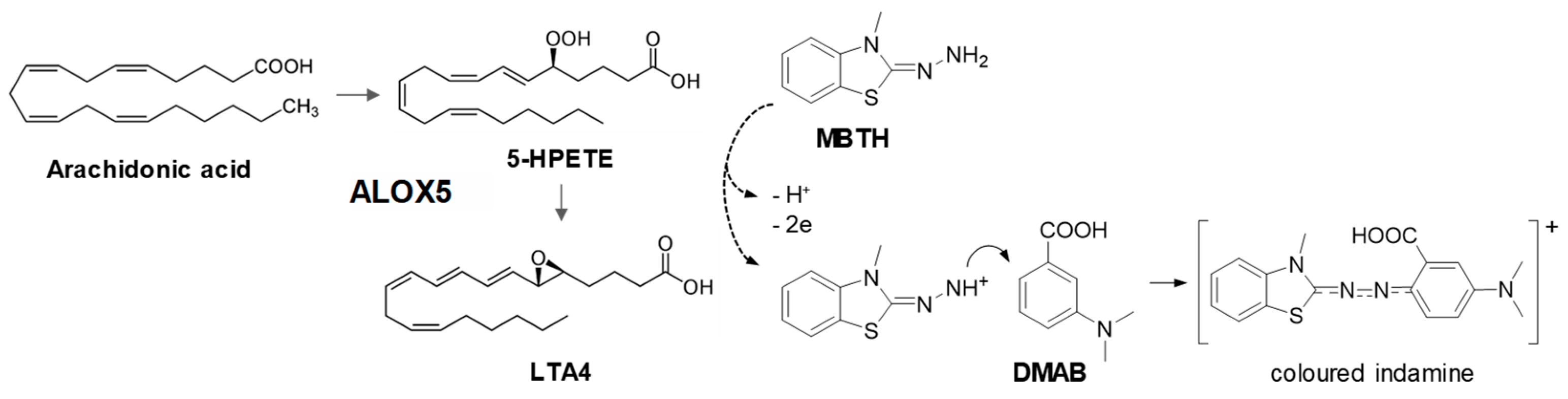
2.6. Arachidonate 5-Lipoxygenase Inhibition Assay
2.7. Data Processing
2.8. Optimization of Reagents Concentrations and Statistical Analyses
2.9. Sensitivity and Linearity
3. Results and Discussion
3.1. Development and Validation of the TLC-based Enzyme Assay
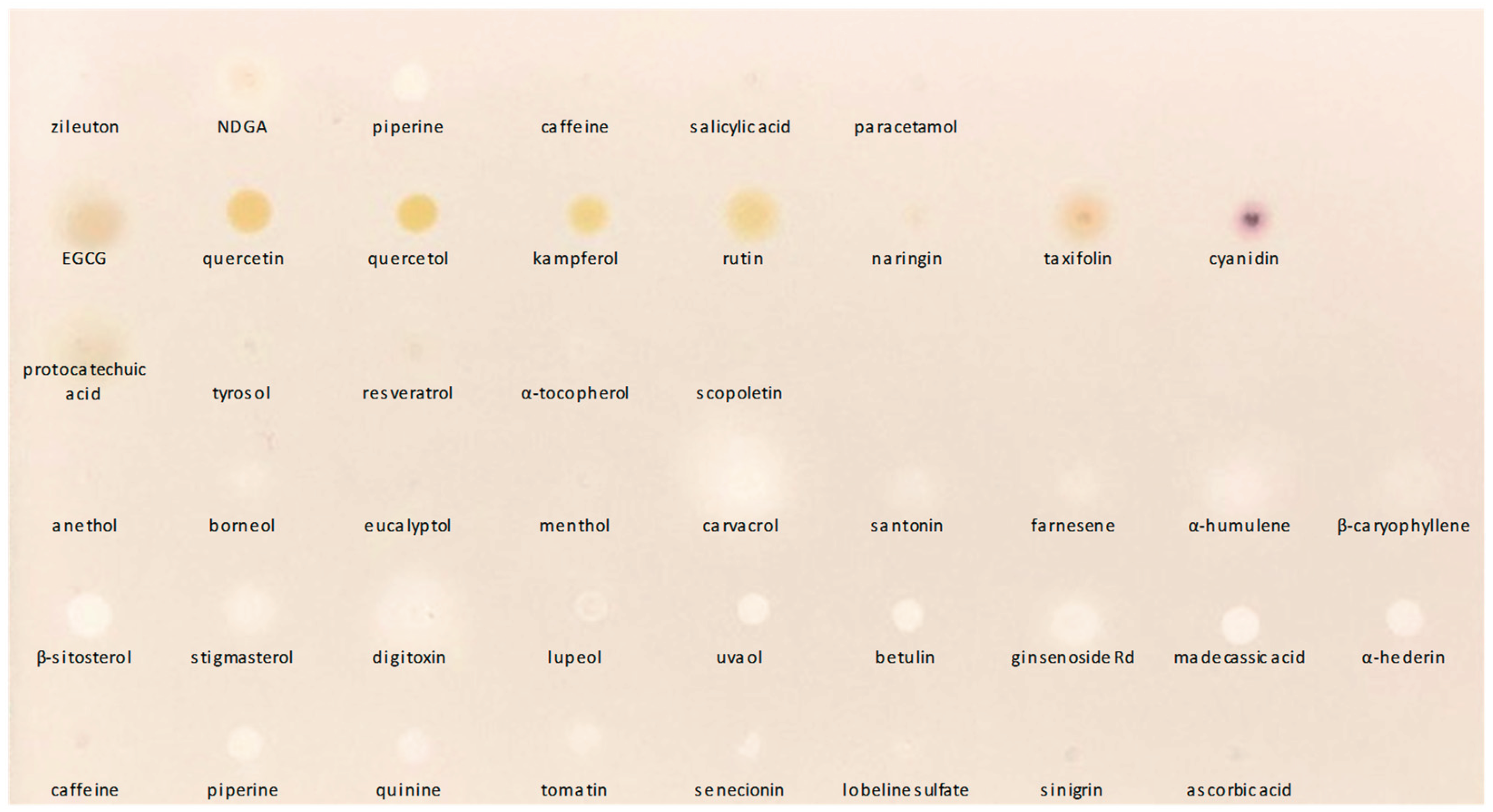
3.1.1. Specificity and Selectivity
3.1.2. Optimization of Experimental Parameters
- Heme cofactors
- Incubation conditions
- ATP and calcium
- Reagent concentrations
- Final Protocol
3.1.3. Limit of Detection and Linear Regression
3.2. Application to Natural Products
3.2.1. Application to Pure Compounds
3.2.2. Application to Mushroom Extracts
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ALOX5 | Arachidonate 5-lipoxygenase |
| COX | Cyclooxygenase |
| TLC | Thin-layer chromatography |
| HPLC | High-performance liquid chromatography |
| ELISA | enzyme-linked immunosorbent assay |
| DMAB | 3-(dimethylamino)benzoic acid |
| MBTH | 3-methyl-2-benzothiazolinone hydrazone |
| NDGA | Nordihydroguaiaretic acid |
| FOX | Ferrous-oxidized xylenol orange assay |
| EtOAc | Ethyl acetate |
Appendix A
References
- Haeggström, J.Z.; Funk, C.D. Lipoxygenase and Leukotriene Pathways: Biochemistry, Biology, and Roles in Disease. Chem. Rev. 2011, 111, 5866–5898. [Google Scholar] [CrossRef]
- Paljarvi, T.; Forton, J.; Luciano, S.; Herttua, K.; Fazel, S. Analysis of Neuropsychiatric Diagnoses after Montelukast Initiation. JAMA Netw. Open 2022, 5, e2213643. [Google Scholar] [CrossRef]
- Vishnupriya, P.; Aparna, A.; Viswanadha, V.P. Lipoxygenase (LOX) Pathway: A Promising Target to Combat Cancer. Curr. Pharm. Design 2021, 27, 3349–3369. [Google Scholar] [CrossRef]
- Yan, M.; Zhang, S.; Li, C.; Liu, Y.; Zhao, J.; Wang, Y.; Yang, Y.; Zhang, L. 5-Lipoxygenase as an Emerging Target against Age-Related Brain Disorders. Ageing Res. Rev. 2021, 69, 101359. [Google Scholar] [CrossRef] [PubMed]
- Pufahl, R.A.; Kasten, T.P.; Hills, R.; Gierse, J.K.; Reitz, B.A.; Weinberg, R.A.; Masferrer, J.L. Development of a Fluorescence-Based Enzyme Assay of Human 5-Lipoxygenase. Anal. Biochem. 2007, 364, 204–212. [Google Scholar] [CrossRef]
- Nair, D.G.; Funk, C.D. A Cell-Based Assay for Screening Lipoxygenase Inhibitors. Prostaglandins Other Lipid Mediat. 2009, 90, 98–104. [Google Scholar] [CrossRef]
- Lu, W.; Zhao, X.; Xu, Z.; Dong, N.; Zou, S.; Shen, X.; Huang, J. Development of a New Colorimetric Assay for Lipoxygenase Activity. Anal. Biochem. 2013, 441, 162–168. [Google Scholar] [CrossRef]
- Huang, X.; Li, Y.; Sabier, M.; Si, J.; Wang, P.; Shen, Y.; Zhang, X.; Liu, J. Guidelines for the in Vitro Determination of Anti-Inflammatory Activity. eFood 2024, 5, e160. [Google Scholar] [CrossRef]
- Schneider, I.; Bucar, F. Lipoxygenase Inhibitors from Natural Plant Sources. Part 1: Medicinal Plants with Inhibitory Activity on Arachidonate 5-Lipoxygenase and 5-Lipoxygenase/Cyclooxygenase. Phytother Res 2005, 19, 81–102. [Google Scholar] [CrossRef]
- Werz, O. Inhibition of 5-Lipoxygenase Product Synthesis by Natural Compounds of Plant Origin. Planta Med 2007, 73, 1331–1357. [Google Scholar] [CrossRef] [PubMed]
- Urbain, A.; Trabelssi, N.; Bardot, V. Development of an Enzyme-Based Thin-Layer Chromatographic Assay for the Detection of Cyclooxygenase-2 Inhibitors. Separations 2022, 9, 238. [Google Scholar] [CrossRef]
- Cho, Y.S.; Kim, H.S.; Kim, C.H.; Cheon, H.G. Application of the Ferrous Oxidation-Xylenol Orange Assay for the Screening of 5-Lipoxygenase Inhibitors. Anal. Biochem. 2006, 351, 62–68. [Google Scholar] [CrossRef]
- Auerbach, B.; Kiely, J.; Cornicelli, J. A Spectrophotometric Microtiter-Based Assay for the Detection of Hydroperoxy Derivatives of Linoleic-Acid. Anal. Biochem. 1992, 201, 375–380. [Google Scholar] [CrossRef]
- Ngo, T.T.; Lenhoff, H.M. A Sensitive and Versatile Chromogenic Assay for Peroxidase and Peroxidase-Coupled Reactions. Anal. Biochem. 1980, 105, 389–397. [Google Scholar] [CrossRef] [PubMed]
- Anthon, G.E.; Barrett, D.M. Colorimetric Method for the Determination of Lipoxygenase Activity. J Agric Food Chem 2001, 49, 32–37. [Google Scholar] [CrossRef]
- Wangun, H.V.K.; Berg, A.; Hertel, W.; Nkengfack, A.E.; Hertweck, C. Anti-Inflammatory and Anti-Hyaluronate Lyase Activities of Lanostanoids from Piptoporus betulinus. J Antibiot (Tokyo) 2004, 57, 755–758. [Google Scholar] [CrossRef]
- Prasad, N.S.; Raghavendra, R.; Lokesh, B.R.; Naidu, K.A. Spice Phenolics Inhibit Human PMNL 5-Lipoxygenase. Prostaglandins Leukot Essent Fatty Acids 2004, 70, 521–528. [Google Scholar] [CrossRef] [PubMed]
- Lapenna, D.; Ciofani, G.; Pierdomenico, S.D.; Neri, M.; Cuccurullo, C.; Giamberardino, M.A.; Cuccurullo, F. Inhibitory Activity of Salicylic Acid on Lipoxygenase-Dependent Lipid Peroxidation. Biochim. Biophys. Acta 2009, 1790, 25–30. [Google Scholar] [CrossRef]
- Häfner, A.-K.; Kahnt, A.S.; Steinhilber, D. Beyond Leukotriene Formation—The Noncanonical Functions of 5-Lipoxygenase. Prostaglandins Other Lipid Mediat. 2019, 142, 24–32. [Google Scholar] [CrossRef]
- Vo, N.N.Q.; Nomura, Y.; Muranaka, T.; Fukushima, E.O. Structure–Activity Relationships of Pentacyclic Triterpenoids as Inhibitors of Cyclooxygenase and Lipoxygenase Enzymes. J. Nat. Prod. 2019, 82, 3311–3320. [Google Scholar] [CrossRef]
- Gilbert, N.C.; Gerstmeier, J.; Schexnaydre, E.E.; Börner, F.; Garscha, U.; Neau, D.B.; Werz, O.; Newcomer, M.E. Structural and Mechanistic Insights into 5-Lipoxygenase Inhibition by Natural Products. Nat. Chem. Biol. 2020, 16, 783–790. [Google Scholar] [CrossRef]
- Gutierrez-Lugo, M.-T.; Deschamps, J.D.; Holman, T.R.; Suarez, E.; Timmermann, B.N. Lipoxygenase Inhibition by Anadanthoflavone, a New Flavonoid from the Aerial Parts of Anadenanthera colubrina. Planta Med. 2004, 70, 263–265. [Google Scholar] [CrossRef] [PubMed]
- Mikutis, S.; Lawrinowitz, S.; Kretzer, C.; Dunsmore, L.; Sketeris, L.; Rodrigues, T.; Werz, O.; Bernardes, G.J.L. Machine Learning Uncovers Natural Product Modulators of the 5-Lipoxygenase Pathway and Facilitates the Elucidation of Their Biological Mechanisms. ACS Chem Biol 2023, 19, 217–229. [Google Scholar] [CrossRef] [PubMed]
- Tomy, M.J.; Sharanya, C.S.; Dileep, K.V.; Prasanth, S.; Sabu, A.; Sadasivan, C.; Haridas, M. Derivatives Form Better Lipoxygenase Inhibitors than Piperine: In Vitro and in Silico Study. Chem Biol Drug Des 2015, 85, 715–721. [Google Scholar] [CrossRef] [PubMed]
- Stöhr, J.R.; Xiao, P.-G.; Bauer, R. Constituents of Chinese Piper Species and Their Inhibitory Activity on Prostaglandin and Leukotriene Biosynthesis in Vitro. J Ethnopharmacol 2001, 75, 133–139. [Google Scholar] [CrossRef]
- Fatima, K.; Luqman, S.; Meena, A. Carvacrol Arrests the Proliferation of Hypopharyngeal Carcinoma Cells by Suppressing Ornithine Decarboxylase and Hyaluronidase Activities. Front. Nutr. 2022, 9. [Google Scholar] [CrossRef]
- Chen, S. Natural Products Triggering Biological Targets- A Review of the Anti-Inflammatory Phytochemicals Targeting the Arachidonic Acid Pathway in Allergy Asthma and Rheumatoid Arthritis. Curr. Drug Targets 2010, 12, 288–301. [Google Scholar] [CrossRef]
- Giménez-Bastida, J.A.; González-Sarrías, A.; Laparra-Llopis, J.M.; Schneider, C.; Espín, J.C. Targeting Mammalian 5-Lipoxygenase by Dietary Phenolics as an Anti-Inflammatory Mechanism: A Systematic Review. Int. J. Mol. Sci. 2021, 22, 7937. [Google Scholar] [CrossRef]
- Laughton, M.J.; Evans, P.J.; Moroney, M.A.; Hoult, J.R.S.; Halliwell, B. Inhibition of Mammalian 5-Lipoxygenase and Cyclo-Oxygenase by Flavonoids and Phenolic Dietary Additives: Relationship to Antioxidant Activity and to Iron Ion-Reducing Ability. Biochem. Pharmacol. 1991, 42, 1673–1681. [Google Scholar] [CrossRef]
- Mukhopadhyay, N.; Shukla, A.; Makhal, P.N.; Kaki, V.R. Natural Product-Driven Dual COX-LOX Inhibitors: Overview of Recent Studies on the Development of Novel Anti-Inflammatory Agents. Heliyon 2023, 9, e14569. [Google Scholar] [CrossRef]
- Agrawal, N. A Comprehensive Review on the Advancements of Dual COX-2/5-LOX Inhibitors as Anti-Inflammatory Drugs. Chem. Biol. Drug Des. 2025, 105, e70114. [Google Scholar] [CrossRef]
- Chatzipieris, F.P.; Petsas, E.; Lambrinidis, G.; Vassiliou, S.; Chasapis, C.T. Recent Advances in Dual COX/LOX Inhibitor Design (2020-2024): Establishing “The Rule of Four for Inflammation.”. Life-Basel 2026, 16, 163. [Google Scholar] [CrossRef]
- Taofiq, O.; Martins, A.; Barreiro, M.F.; Ferreira, I.C.F.R. Anti-Inflammatory Potential of Mushroom Extracts and Isolated Metabolites. Trends Food Sci. Technol. 2016, 50, 193–210. [Google Scholar] [CrossRef]
- Urbain, A.; Simões-Pires, C.A. Thin-Layer Chromatography for the Detection and Analysis of Bioactive Natural Products. In Encyclopedia of Analytical Chemistry; Meyers, R.A., Ed.; Wiley, 2020; pp. 1–29. ISBN 978-0-471-97670-7. [Google Scholar]


| Compound | LOD (µg) | LOD (nmol) | Regression equation | R2 |
|---|---|---|---|---|
| zileuton | 1.00 | 4.23 | y = 2.4175x + 232.75 | 0.98 |
| NDGA | 0.01 | 3.31 | y = - 15.64ln(x) + 140.72 | 0.97 |
| piperine | 0.01 | 1.65 | y = - 10.01ln(x) + 176.94 | 0.98 |
| uvaol | 0.50 | 1.13 | y = 0.3882x + 241.03 | 0.91 |
| betulin | 0.05 | 0.226 | y = 0.0194x + 241.53 | 0.98 |
| Compound | ALOX5 inhibition a |
Compound | ALOX5 inhibition a |
|
|---|---|---|---|---|
| NDGA | + | lobeline | ± | |
| betulin | + | caffeine | - | |
| digitoxin | + | anethol | - | |
| ginsenoside Rd | + | eucalyptol | - | |
| α-hederin | + | menthol | - | |
| lupeol | + | α-tocopherol | - | |
| madecassic acid | + | ascorbic acid | - | |
| piperine | + | epigallocatechin | - | |
| quinine | + | kampferol | - | |
| senecionin | + | naringin | - | |
| β-sitosterol | + | protocatechuic acid | - | |
| stigmasterol | + | quercetin | - | |
| tomatine | + | quercetol | - | |
| uvaol | + | resveratrol | - | |
| borneol | ± | rutin | - | |
| carvacrol | ± | salicylic acid | - | |
| caryophyllene | ± | scopoletin | - | |
| farnesene | ± | taxifolin | - | |
| humulene | ± | tyrosol | - | |
| santonin | ± | sinigrin | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




