Submitted:
06 March 2026
Posted:
09 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
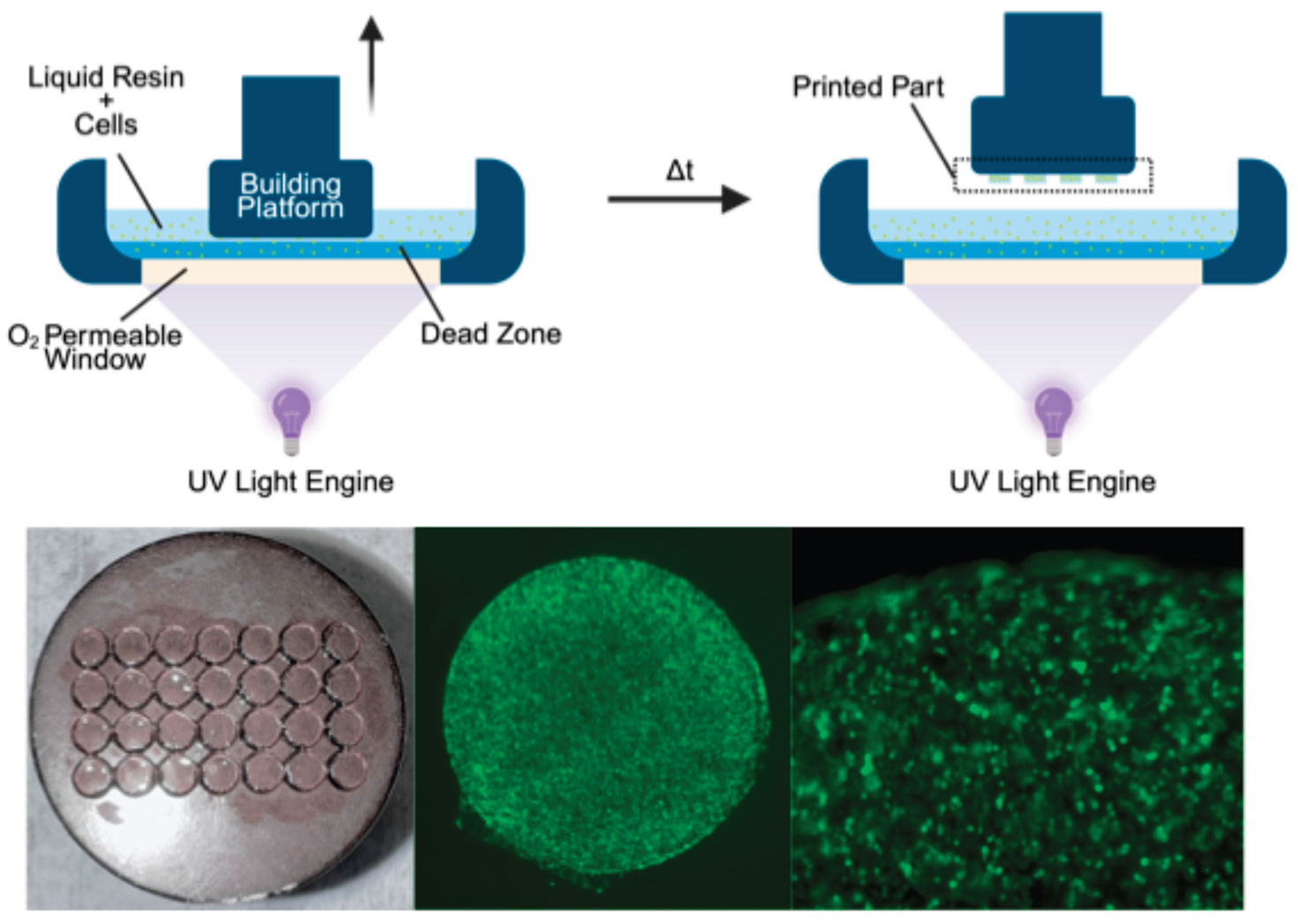
2. Results
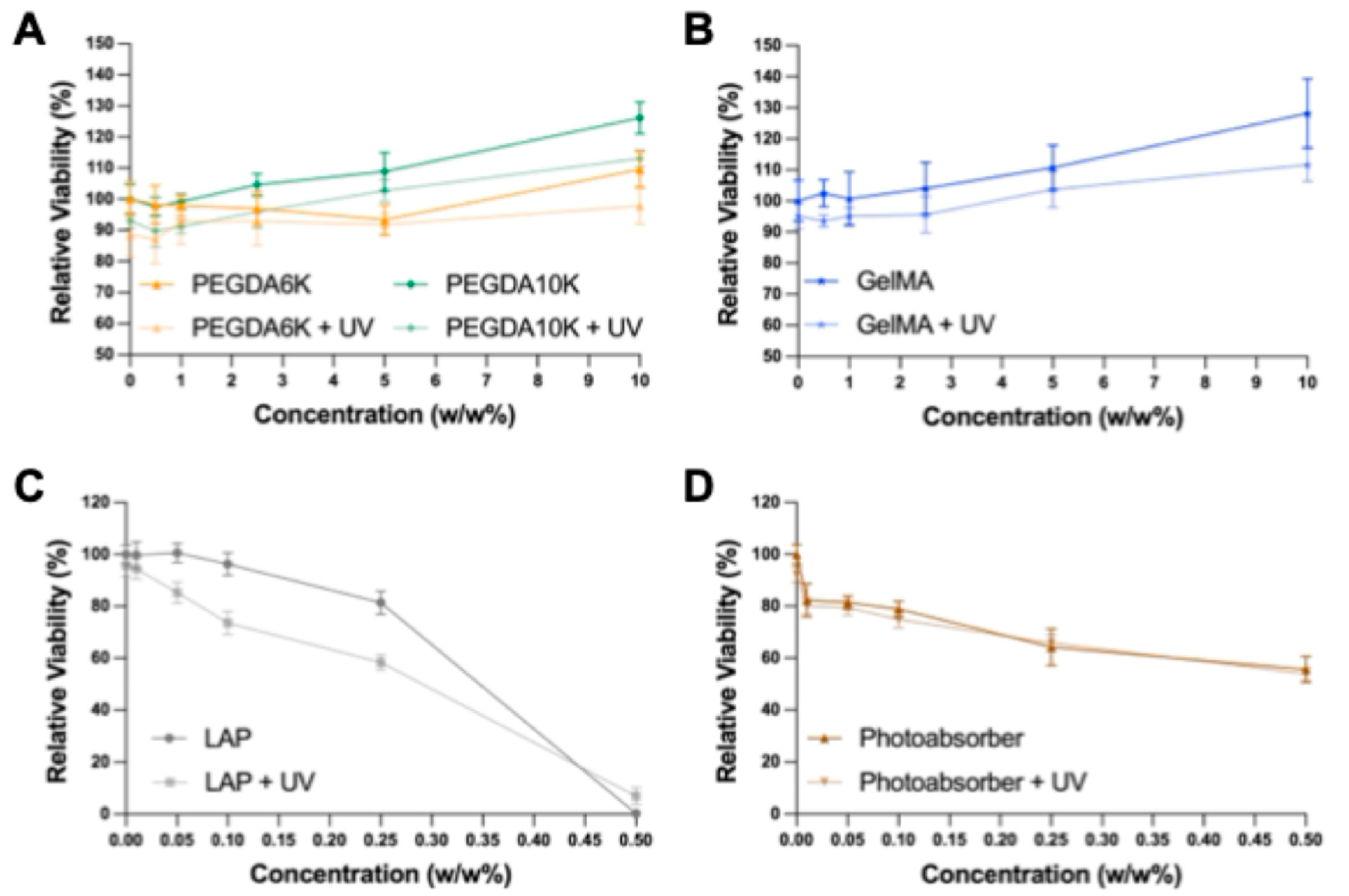
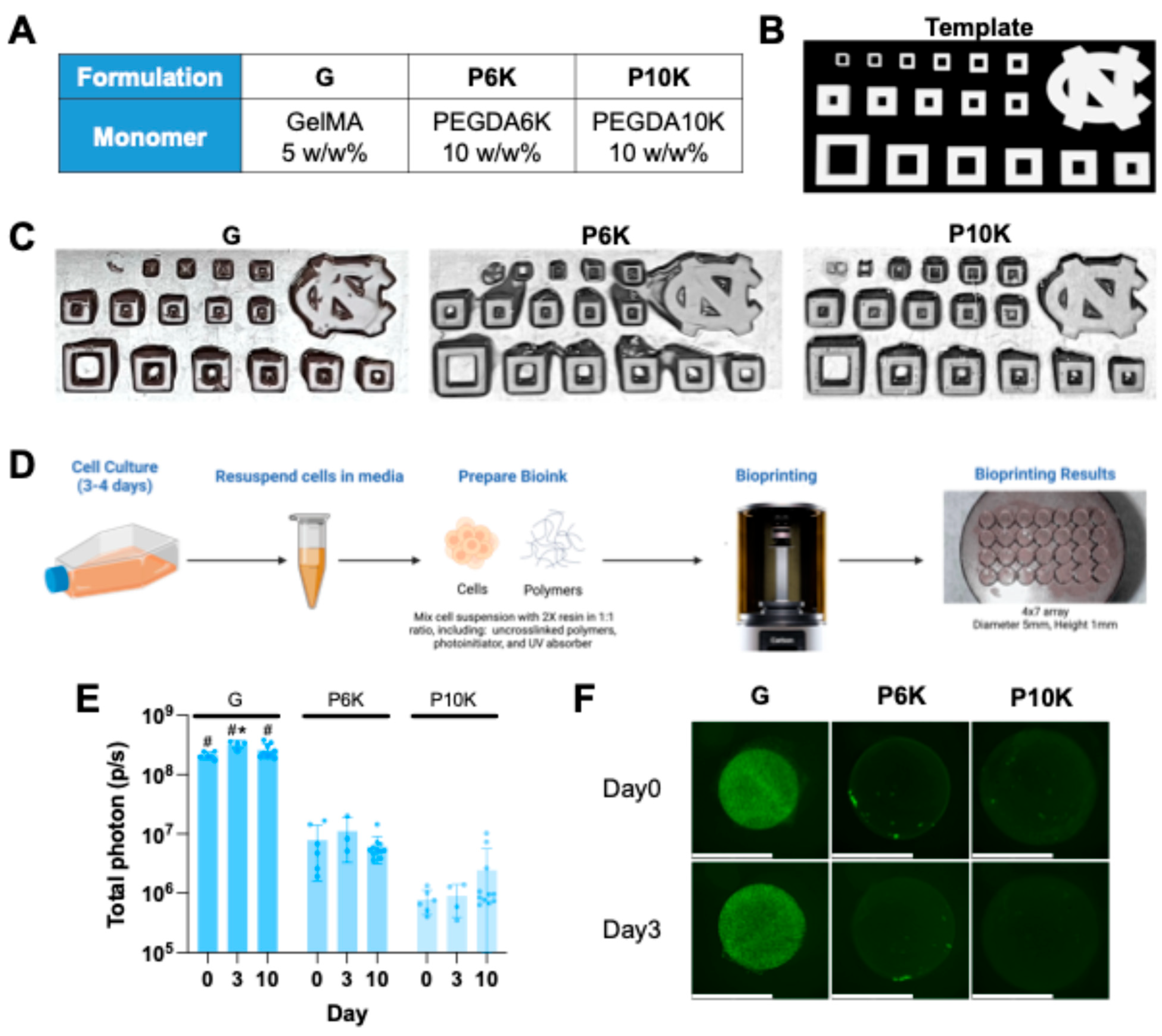
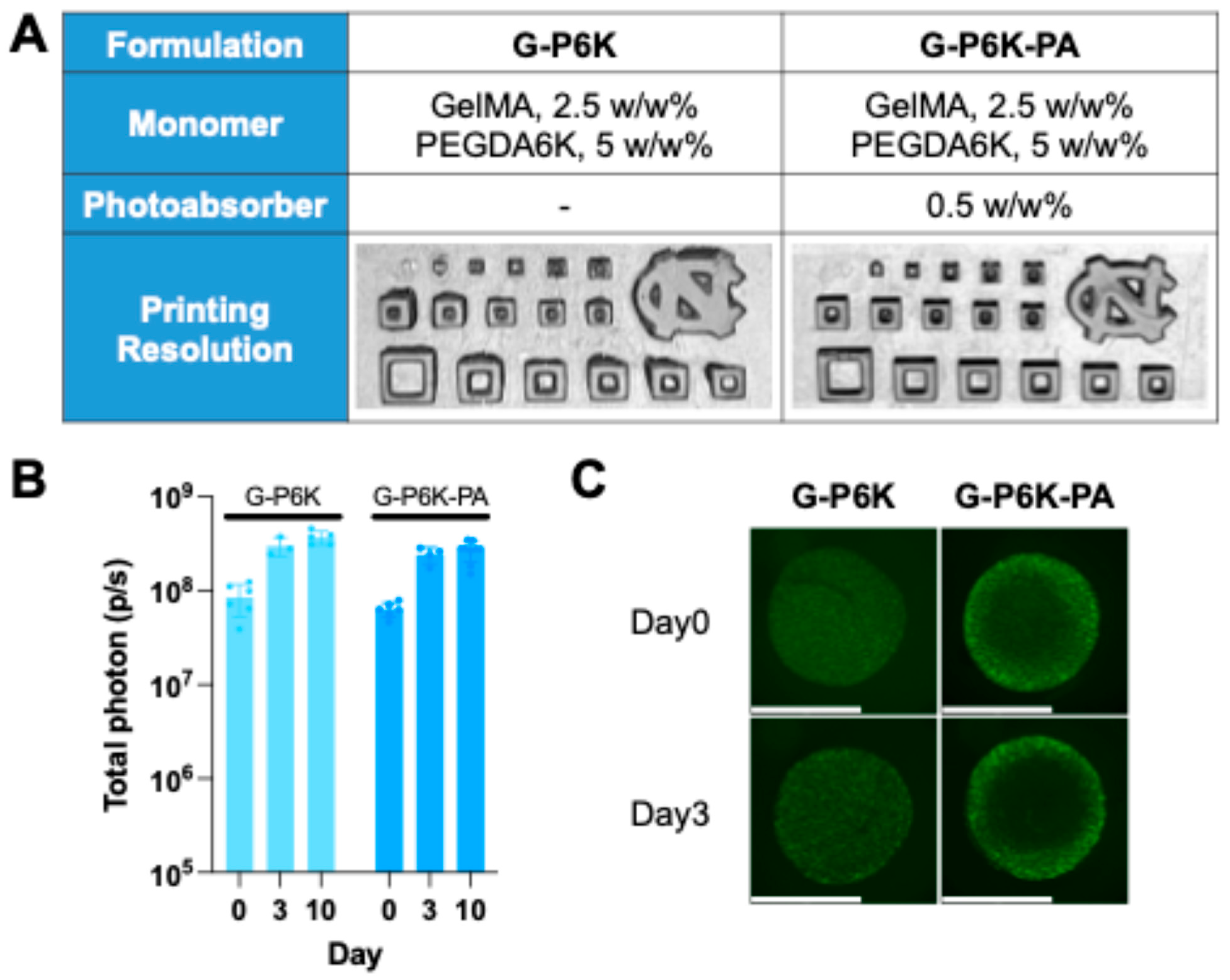
2.1. Development of a Biocompatible Bioprinting Resin Formulation
2.2. Evaluation of Printability and Cell Viability with Various Resin Formulations
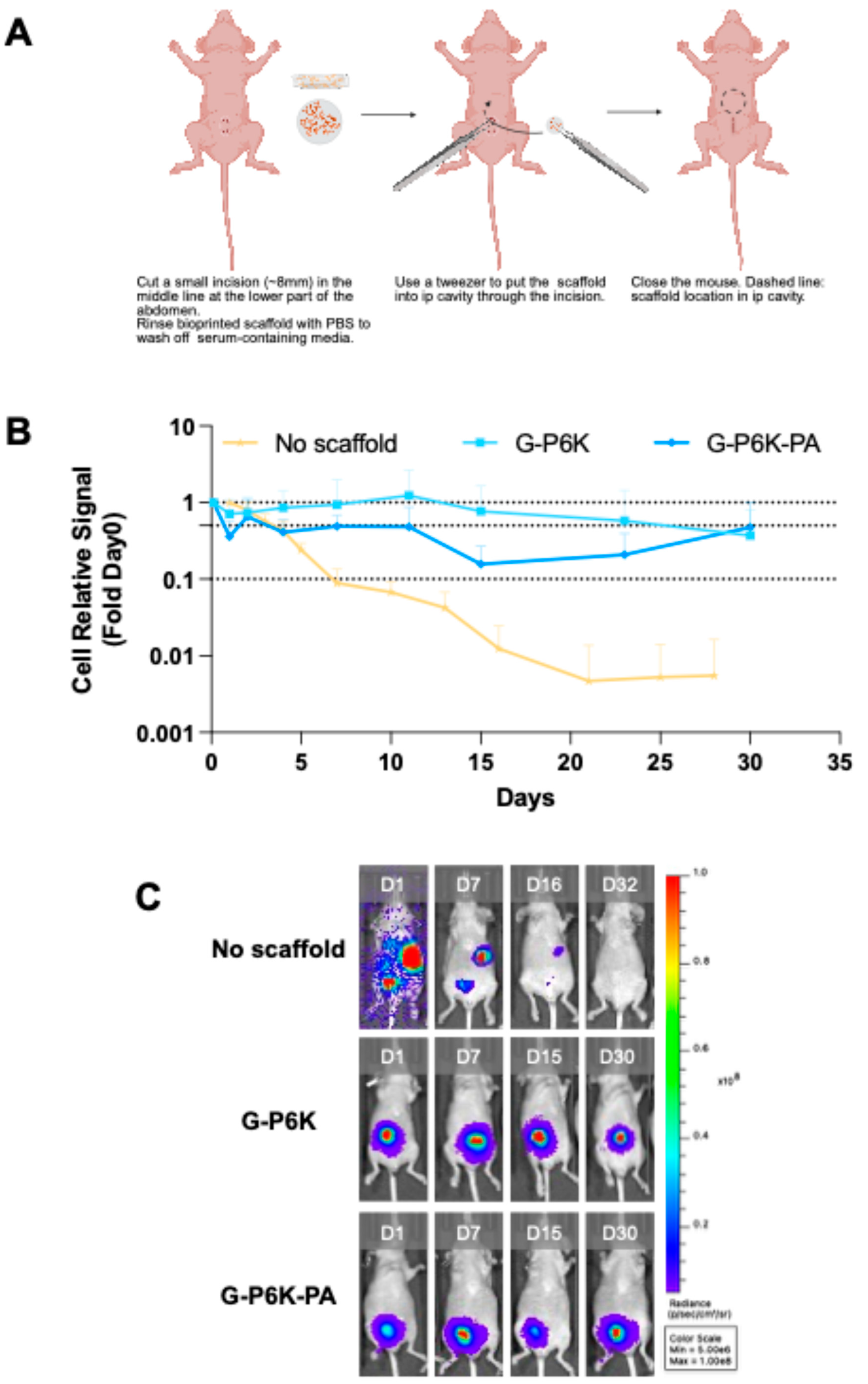
2.3. Impact of Material Composition on Cell In Vivo Persistence
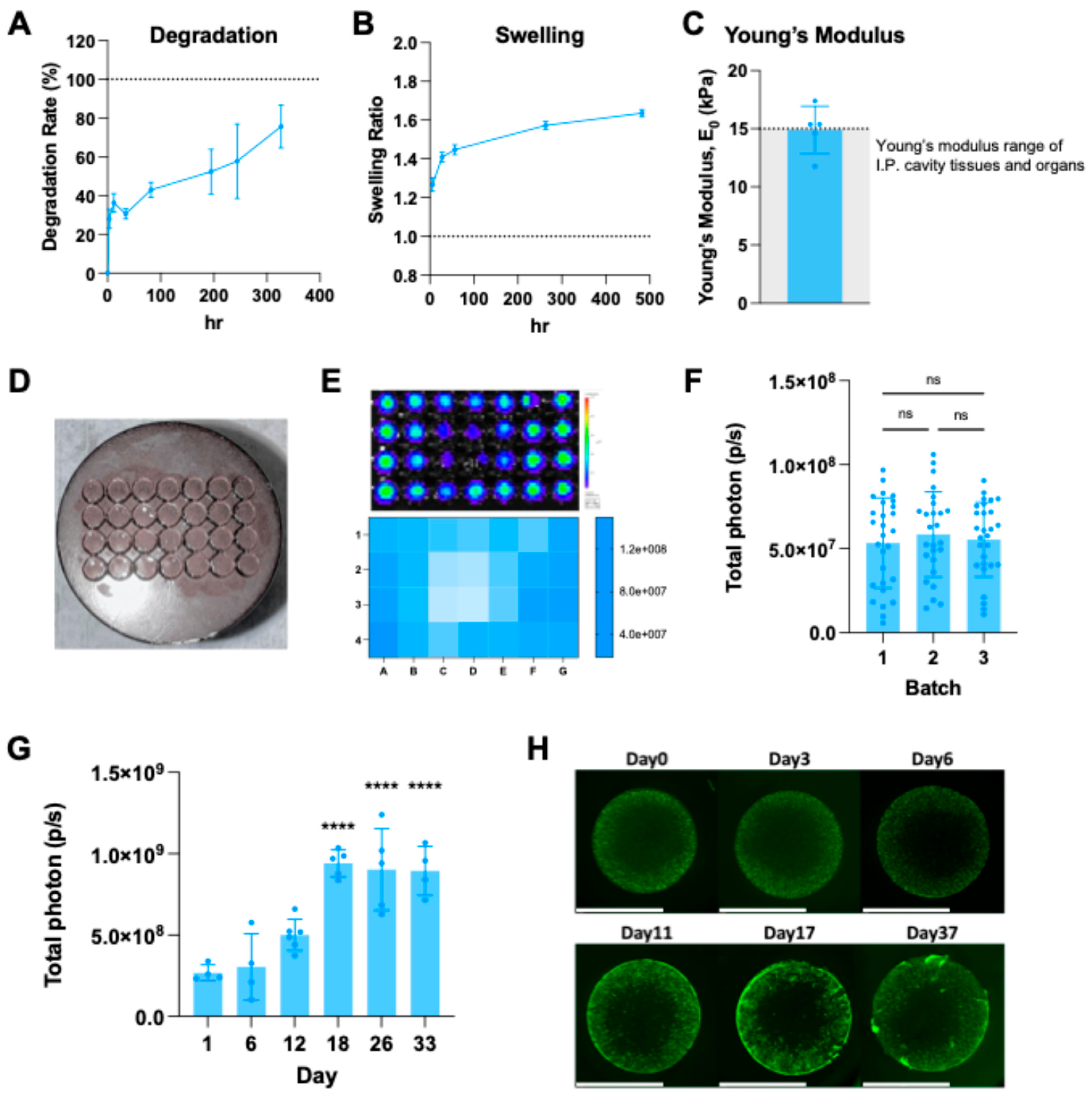
2.4. Characterization of Bioprinted NHF1GFP-FL G-P6K-PA Scaffolds
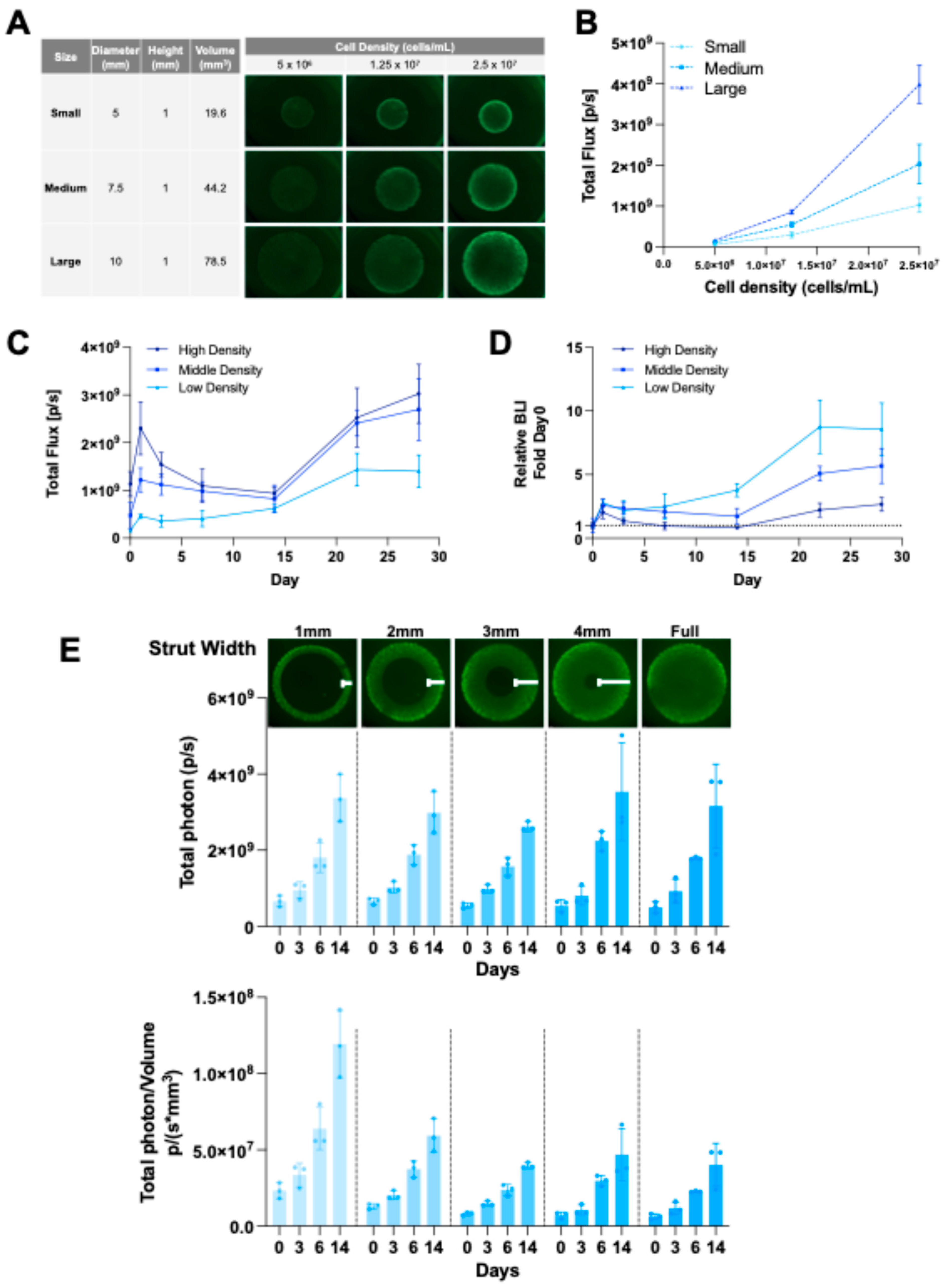
2.5. Tuning Cell Loading in CLIP 3D-Bioprinted Scaffolds
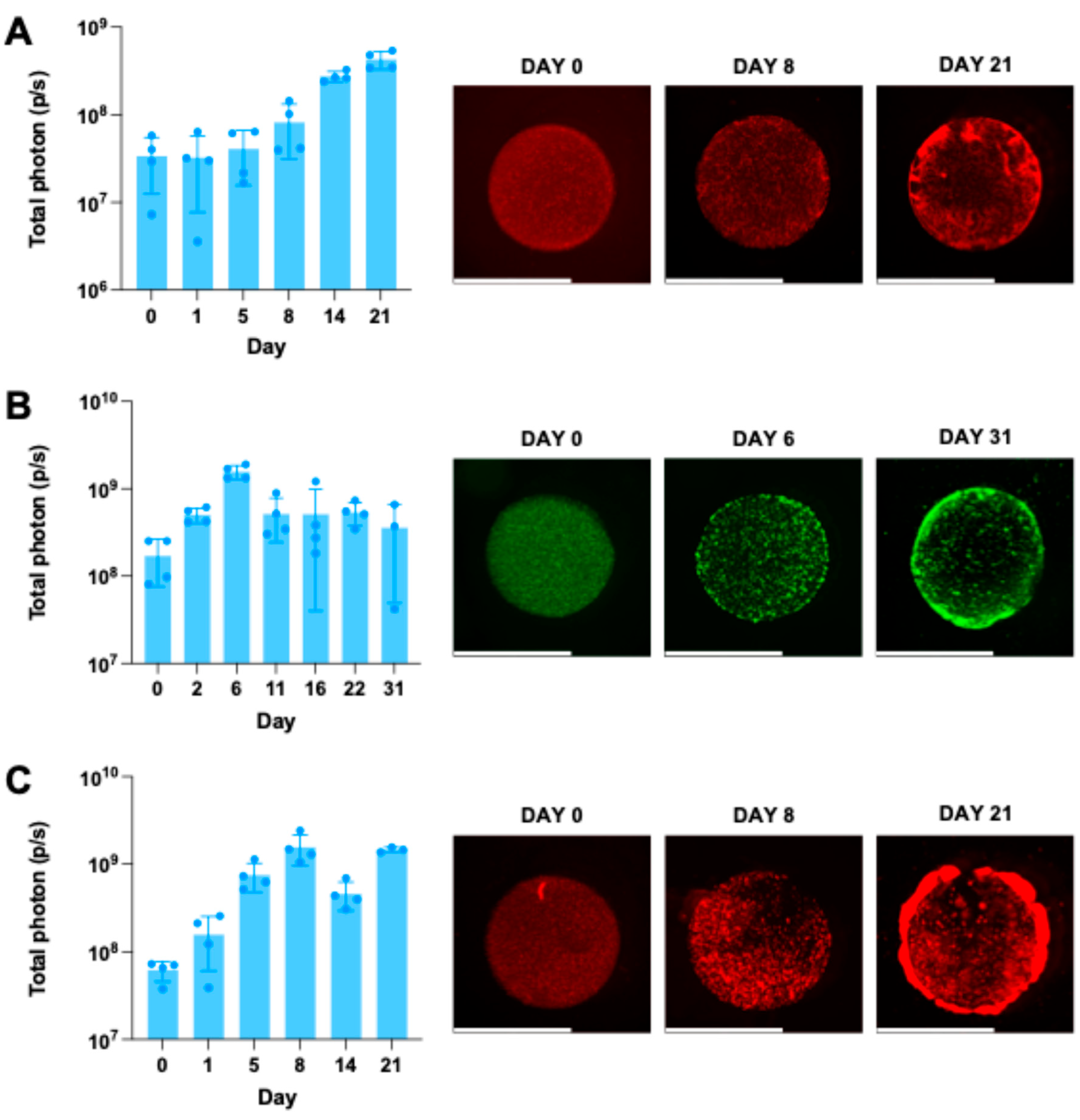
2.6. Extending Bioprinting to Stem Cells
3. Discussion
4. Materials and Methods
4.1. Materials, Reagents, and Cell Lines
4.2. hiNeuroS Generation
4.3. Lentiviral Transduction
4.4. Resin Toxicity Screening
4.5. CLIP 3D Bioprinting
4.6. Printability
4.7. Degradation Study
4.8. Swelling Test
4.9. Modulus Test
4.10. In Vitro Bioluminescence Viability Assay
4.11. Free NHFGFP-FL Cell Injection into Mouse Intraperitoneal Cavity and Persistence Imaging
4.12. In Vivo NHFGFP-FL Bioprinted Scaffolds Implantation into Mouse Intraperitoneal Cavity
4.13. In Vivo NHFGFP-FL Persistence in Bioprinted Scaffolds
4.14. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BLI | Bioluminescence |
| CAD | Computer-Aided Design |
| CAR-M: | Chimeric Antigen Receptor - Macrophages |
| CAR-NK | Chimeric Antigen Receptor - Natural Killer Cells |
| CAR-T | Chimeric Antigen Receptor - T Cells |
| CLIP | Continuous Liquid Interface Production |
| DDS | Drug Delivery Systems |
| ECM | Extracellular Matrix |
| EudraCT | European Union Drug Regulating Authorities Clinical Trials Database |
| FBR | Foreign Body Response |
| GelMA | Gelatin Methacrylate |
| hiNeuroS | Spheroidal Human Induced Neural Stem Cell Line |
| I.P. | f |
| iPSC | Induced Pluripotent Stem Cell |
| LAP | Lithium Phenyl-2,4,6-Trimethylbenzoylphosphinate |
| MSC | Mesenchymal Stem Cells |
| MSC-INFβ | MSC-Infβ:Mesenchymal Stem Cells Secreting Infβ |
| NK | Natural Killer Cells |
| NSC | Neural Stem Cells |
| PA | Photoabsorber |
| PEGDA | Polyethylene Glycol Diacrylate |
| RPE-IL2 | Human Retinal Pigmented Epithelial Cells Producing Interleukin-2 |
| UMIN-CTR | University Hospital Medical Information Network Clinical Trials Registry, Japan |
References
- Ding, Y.; Wang, Y.; Hu, Q. Recent advances in overcoming barriers to cell-based delivery systems for cancer immunotherapy. In Exploration; Wiley Online Library, 2022. [Google Scholar]
- Cao, P.; Mooney, R.; Tirughana, R.; Abidi, W.; Aramburo, S.; Flores, L.; Gilchrist, M.; Nwokafor, U.; Haber, T.; Tiet, P. Intraperitoneal administration of neural stem cell–nanoparticle conjugates targets chemotherapy to ovarian tumors. Bioconjugate chemistry 2017, 28, 1767–1776. [Google Scholar] [CrossRef]
- Perelló-Trias, M.T.; Serrano-Munoz, A.J.; Rodríguez-Fernández, A.; Segura-Sampedro, J.J.; Ramis, J.M.; Monjo, M. Intraperitoneal drug delivery systems for peritoneal carcinomatosis: Bridging the gap between research and clinical implementation. Journal of Controlled Release 2024, 373, 70–92. [Google Scholar] [CrossRef]
- Ceelen, W.P.; Flessner, M.F. Intraperitoneal therapy for peritoneal tumors: biophysics and clinical evidence. Nature reviews Clinical oncology 2010, 7, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Abe, Y.; Kobayashi, H.; Kanno, T.; Akizawa, Y.; Ishitani, K.; Hashimoto, K. Intraperitoneal Injection of Expanded Gamma Delta T Cells with Zoledronate: A Pilot Analysis of Seven Patients with Ovarian Cancer. Annals of Cancer Research and Therapy 2024, 32, 31–38. [Google Scholar] [CrossRef]
- Nash, A.M.; Jarvis, M.I.; Aghlara-Fotovat, S.; Mukherjee, S.; Hernandez, A.; Hecht, A.D.; Rios, P.D.; Ghani, S.; Joshi, I.; Isa, D. Clinically translatable cytokine delivery platform for eradication of intraperitoneal tumors. Science advances 2022, 8, eabm1032. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.K.; Hamilton, C.A.; Cheung, M.K.; Karimi, M.; Baker, J.; Gall, J.M.; Schulz, S.; Thorne, S.H.; Teng, N.N.; Contag, C.H. Enhanced killing of primary ovarian cancer by retargeting autologous cytokine-induced killer cells with bispecific antibodies: a preclinical study. Clinical Cancer Research 2006, 12, 1859–1867. [Google Scholar] [CrossRef]
- Hermanson, D.L.; Bendzick, L.; Pribyl, L.; McCullar, V.; Vogel, R.I.; Miller, J.S.; Geller, M.A.; Kaufman, D.S. Induced pluripotent stem cell-derived natural killer cells for treatment of ovarian cancer. Stem cells 2016, 34, 93–101. [Google Scholar] [CrossRef]
- Xiao, L.; Cen, D.; Gan, H.; Sun, Y.; Huang, N.; Xiong, H.; Jin, Q.; Su, L.; Liu, X.; Wang, K. Adoptive transfer of NKG2D CAR mRNA-engineered natural killer cells in colorectal cancer patients. Molecular Therapy 2019, 27, 1114–1125. [Google Scholar] [CrossRef]
- Wada, I.; Matsushita, H.; Noji, S.; Mori, K.; Yamashita, H.; Nomura, S.; Shimizu, N.; Seto, Y.; Kakimi, K. Intraperitoneal injection of in vitro expanded Vγ9Vδ2 T cells together with zoledronate for the treatment of malignant ascites due to gastric cancer. Cancer medicine 2014, 3, 362–375. [Google Scholar] [CrossRef]
- Kono, K.; Takahashi, A.; Ichihara, F.; Amemiya, H.; Iizuka, H.; Fujii, H.; Sekikawa, T.; Matsumoto, Y. Prognostic significance of adoptive immunotherapy with tumor-associated lymphocytes in patients with advanced gastric cancer: a randomized trial. Clinical Cancer Research 2002, 8, 1767–1771. [Google Scholar]
- Du, X.; Jin, R.; Ning, N.; Li, L.; Wang, Q.; Liang, W.; Liu, J.; Xu, Y. In vivo distribution and antitumor effect of infused immune cells in a gastric cancer model. Oncology reports 2012, 28, 1743–1749. [Google Scholar] [CrossRef]
- Cao, B.; Liu, M.; Huang, J.; Zhou, J.; Li, J.; Lian, H.; Huang, W.; Guo, Y.; Yang, S.; Lin, L. Development of mesothelin-specific CAR NK-92 cells for the treatment of gastric cancer. International journal of biological sciences 2021, 17, 3850. [Google Scholar] [CrossRef]
- Fitzpatrick, E.; Filippi, C.; Jagadisan, B.; Shivapatham, D.; Anand, H.; Lyne, M.; Stroud, K.-D.; Newton, R.; DeLord, M.; Douiri, A. Intraperitoneal transplant of Hepatocytes co-Encapsulated with mesenchymal stromal cells in modified alginate microbeads for the treatment of acute Liver failure in Pediatric patients (HELP)—An open-label, single-arm Simon’s two stage phase 1 study protocol. PloS one 2023, 18, e0288185. [Google Scholar]
- Mei, J.; Sgroi, A.; Mai, G.; Baertschiger, R.; Gonelle-Gispert, C.; Serre-Beinier, V.; Morel, P.; Bühler, L.H. Improved survival of fulminant liver failure by transplantation of microencapsulated cryopreserved porcine hepatocytes in mice. Cell transplantation 2009, 18, 101–110. [Google Scholar] [CrossRef] [PubMed]
- Soon-Shiong, P.; Heintz, R.E.; Merideth, N.; Yao, Q.X.; Yao, Z.; Zheng, T.; Murphy, M.; Moloney, M.K.; Schmehl, M.; Harris, M. Insulin independence in a type 1 diabetic patient after encapsulated islet transplantation. The Lancet 1994, 343, 950–951. [Google Scholar] [CrossRef]
- Lim, F.; Sun, A.M. Microencapsulated islets as bioartificial endocrine pancreas. Science 1980, 210, 908–910. [Google Scholar] [CrossRef] [PubMed]
- González, M.A.; Gonzalez–Rey, E.; Rico, L.; Büscher, D.; Delgado, M. Adipose-derived mesenchymal stem cells alleviate experimental colitis by inhibiting inflammatory and autoimmune responses. Gastroenterology 2009, 136, 978–989. [Google Scholar] [CrossRef]
- Wang, M.; Liang, C.; Hu, H.; Zhou, L.; Xu, B.; Wang, X.; Han, Y.; Nie, Y.; Jia, S.; Liang, J. Intraperitoneal injection (IP), Intravenous injection (IV) or anal injection (AI)? Best way for mesenchymal stem cells transplantation for colitis. Scientific reports 2016, 6, 30696. [Google Scholar] [CrossRef]
- Hunter, M.M.; Wang, A.; Parhar, K.S.; Johnston, M.J.; Van Rooijen, N.; Beck, P.L.; McKay, D.M. In vitro-derived alternatively activated macrophages reduce colonic inflammation in mice. Gastroenterology 2010, 138, 1395–1405. [Google Scholar] [CrossRef]
- Zani, A.; Cananzi, M.; Fascetti-Leon, F.; Lauriti, G.; Smith, V.V.; Bollini, S.; Ghionzoli, M.; D'Arrigo, A.; Pozzobon, M.; Piccoli, M. Amniotic fluid stem cells improve survival and enhance repair of damaged intestine in necrotising enterocolitis via a COX-2 dependent mechanism. Gut 2014, 63, 300–309. [Google Scholar] [CrossRef]
- Meier, R.P.; Mahou, R.; Morel, P.; Meyer, J.; Montanari, E.; Muller, Y.D.; Christofilopoulos, P.; Wandrey, C.; Gonelle-Gispert, C.; Bühler, L.H. Microencapsulated human mesenchymal stem cells decrease liver fibrosis in mice. Journal of hepatology 2015, 62, 634–641. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.-Y.; Chang, P.-Y.; Chen, J.-Y.; Wu, B.-S.; Yang, A.-H.; Lee, O.K.-S. Adipose-derived mesenchymal stem cells attenuate dialysis-induced peritoneal fibrosis by modulating macrophage polarization via interleukin-6. Stem Cell Research & Therapy 2021, 12, 193. [Google Scholar]
- Wiafe, B.; Kadam, R.; Metcalfe, P.D. Intraperitoneal administration of mesenchymal stem cells is effective at mitigating detrusor deterioration after pBOO. American Journal of Physiology-Renal Physiology 2020, 318, F549–F556. [Google Scholar] [CrossRef]
- Flessner, M.F. The transport barrier in intraperitoneal therapy. American Journal of Physiology-Renal Physiology 2005, 288, F433–F442. [Google Scholar] [CrossRef] [PubMed]
- Solass, W.; Struller, F.; Horvath, P.; Königsrainer, A.; Sipos, B.; Weinreich, F.-J. Morphology of the peritoneal cavity and pathophysiological consequences. Pleura and peritoneum 2016, 1, 193–201. [Google Scholar] [CrossRef]
- Meza-Perez, S.; Randall, T.D. Immunological functions of the omentum. Trends in immunology 2017, 38, 526–536. [Google Scholar] [CrossRef]
- Liu, M.; Silva-Sanchez, A.; Randall, T.D.; Meza-Perez, S. Specialized immune responses in the peritoneal cavity and omentum. Journal of Leucocyte Biology 2021, 109, 717–729. [Google Scholar] [CrossRef] [PubMed]
- Salas-Benito, D.; Vercher, E.; Conde, E.; Glez-Vaz, J.; Tamayo, I.; Hervas-Stubbs, S. Inflammation and immunity in ovarian cancer. European Journal of Cancer Supplements 2020, 15, 56–66. [Google Scholar] [CrossRef]
- Almeida-Nunes, D.L.; Mendes-Frias, A.; Silvestre, R.; Dinis-Oliveira, R.J.; Ricardo, S. Immune tumor microenvironment in ovarian cancer ascites. International journal of molecular sciences 2022, 23, 10692. [Google Scholar] [CrossRef]
- Mader, E.K.; Butler, G.; Dowdy, S.C.; Mariani, A.; Knutson, K.L.; Federspiel, M.J.; Russell, S.J.; Galanis, E.; Dietz, A.B.; Peng, K.-W. Optimizing patient derived mesenchymal stem cells as virus carriers for a Phase I clinical trial in ovarian cancer. Journal of Translational Medicine 2013, 11, 20. [Google Scholar] [CrossRef]
- Olson, A.; Marini, F.; Westin, S.; Coleman, R.; Thall, P.; Al Jhadhami, V.; Qazilbash, M.H.; Rezvani, K.; Timmons, M.; Heese, L. A phase I trial of mesenchymal stem cells transfected with a plasmid secreting interferon Beta in advanced ovarian Cancer. Biology of Blood and Marrow Transplantation 2018, 24, S473. [Google Scholar] [CrossRef]
- Bazhanov, N.; Ylostalo, J.H.; Bartosh, T.J.; Tiblow, A.; Mohammadipoor, A.; Foskett, A.; Prockop, D.J. Intraperitoneally infused human mesenchymal stem cells form aggregates with mouse immune cells and attach to peritoneal organs. Stem cell research & therapy 2016, 7, 27. [Google Scholar]
- Preda, M.B.; Neculachi, C.A.; Fenyo, I.M.; Vacaru, A.-M.; Simionescu, M.; Burlacu, A. Short lifespan of syngeneic transplanted MSC is a consequence of in vivo apoptosis and immune cell recruitment in mice. Cell death & disease 2021, 12, 566. [Google Scholar]
- Eich, T.; Eriksson, O.; Lundgren, T. Visualization of early engraftment in clinical islet transplantation by positron-emission tomography. New England Journal of Medicine 2007, 356, 2754–2755. [Google Scholar] [CrossRef]
- Bruni, A.; Gala-Lopez, B.; Pepper, A.R.; Abualhassan, N.S.; Shapiro, A.J. Islet cell transplantation for the treatment of type 1 diabetes: recent advances and future challenges. Diabetes, metabolic syndrome and obesity: targets and therapy 2014, 211–223. [Google Scholar]
- Abualhassan, N.; Sapozhnikov, L.; Pawlick, R.L.; Kahana, M.; Pepper, A.R.; Bruni, A.; Gala-Lopez, B.; Kin, T.; Mitrani, E.; Shapiro, A.J. Lung-derived microscaffolds facilitate diabetes reversal after mouse and human intraperitoneal islet transplantation. PloS one 2016, 11, e0156053. [Google Scholar] [CrossRef]
- Gupta, S.; Rajvanshi, P.; Sokhi, R.; Slehria, S.; Yam, A.; Kerr, A.; Novikoff, P.M. Entry and integration of transplanted hepatocytes in rat liver plates occur by disruption of hepatic sinusoidal endothelium. Hepatology 1999, 29, 509–519. [Google Scholar] [CrossRef]
- Lee, C.A.; Sinha, S.; Fitzpatrick, E.; Dhawan, A. Hepatocyte transplantation and advancements in alternative cell sources for liver-based regenerative medicine. Journal of Molecular Medicine 2018, 96, 469–481. [Google Scholar] [CrossRef]
- Singh, A.; Peppas, N.A. Hydrogels and scaffolds for immunomodulation. Advanced materials 2014, 26, 6530–6541. [Google Scholar] [CrossRef]
- Roy, P.; Mignet, N.; Pocard, M.; Boudy, V. Drug delivery systems to prevent peritoneal metastasis after surgery of digestives or ovarian carcinoma: a review. International Journal of Pharmaceutics 2021, 592, 120041. [Google Scholar] [CrossRef] [PubMed]
- Tibbitt, M.W.; Anseth, K.S. Hydrogels as extracellular matrix mimics for 3D cell culture. Biotechnology and bioengineering 2009, 103, 655–663. [Google Scholar] [CrossRef] [PubMed]
- Geckil, H.; Xu, F.; Zhang, X.; Moon, S.; Demirci, U. Engineering hydrogels as extracellular matrix mimics. Nanomedicine 2010, 5, 469–484. [Google Scholar] [CrossRef] [PubMed]
- Fayzullin, A.; Bakulina, A.; Mikaelyan, K.; Shekhter, A.; Guller, A. Implantable drug delivery systems and foreign body reaction: Traversing the current clinical landscape. Bioengineering 2021, 8, 205. [Google Scholar] [CrossRef] [PubMed]
- Jacobs-Tulleneers-Thevissen, D.; Chintinne, M.; Ling, Z.; Gillard, P.; Schoonjans, L.; Delvaux, G.; Strand, B.L.; Gorus, F.; Keymeulen, B.; Pipeleers, D. Sustained function of alginate-encapsulated human islet cell implants in the peritoneal cavity of mice leading to a pilot study in a type 1 diabetic patient. Diabetologia 2013, 56, 1605–1614. [Google Scholar] [CrossRef]
- Chen, C.-H.; Kuo, C.-Y.; Chen, S.-H.; Mao, S.-H.; Chang, C.-Y.; Shalumon, K.; Chen, J.-P. Thermosensitive injectable hydrogel for simultaneous intraperitoneal delivery of doxorubicin and prevention of peritoneal adhesion. International journal of molecular sciences 2018, 19, 1373. [Google Scholar] [CrossRef]
- Chen, X.; Wei, Y.; Chen, X.; Zheng, L.; Zhao, Y.; You, J.; Yi, C.; Yang, X. Hydrogel-Based intraperitoneal drug delivery platforms for peritoneal metastasis: strategies, advances, and prospects. Drug Delivery 2026, 33, 2576199. [Google Scholar] [CrossRef] [PubMed]
- Annabi, N.; Tamayol, A.; Uquillas, J.A.; Akbari, M.; Bertassoni, L.E.; Cha, C.; Camci-Unal, G.; Dokmeci, M.R.; Peppas, N.A.; Khademhosseini, A. 25th anniversary article: Rational design and applications of hydrogels in regenerative medicine. Advanced materials 2014, 26, 85–124. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Su, X.; Xu, Y.; Kong, B.; Sun, W.; Mi, S. Bioprinting three-dimensional cell-laden tissue constructs with controllable degradation. Scientific reports 2016, 6, 24474. [Google Scholar] [CrossRef] [PubMed]
- Ni, Y.; Qi, H.; Zhang, F.; Jiang, S.; Tang, Q.; Cai, W.; Mo, W.; Miron, R.J.; Zhang, Y. Macrophages modulate stiffness-related foreign body responses through plasma membrane deformation. Proceedings of the National Academy of Sciences 2023, 120, e2213837120. [Google Scholar] [CrossRef]
- Zhuang, Z.; Zhang, Y.; Sun, S.; Li, Q.; Chen, K.; An, C.; Wang, L.; van den Beucken, J.J.; Wang, H. Control of matrix stiffness using methacrylate–gelatin hydrogels for a macrophage-mediated inflammatory response. ACS Biomaterials Science & Engineering 2020, 6, 3091–3102. [Google Scholar]
- Arda, K.; Ciledag, N.; Aktas, E.; Arıbas, B.K.; Köse, K. Quantitative assessment of normal soft-tissue elasticity using shear-wave ultrasound elastography. American Journal of Roentgenology 2011, 197, 532–536. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Zheng, H.; Poh, P.S.P.; Machens, H.-G.; Schilling, A.F. Hydrogels for Engineering of Perfusable Vascular Networks. International Journal of Molecular Sciences 2015, 16, 15997–16016. [Google Scholar] [CrossRef]
- Akbarzadeh, R.; Yousefi, A.M. Effects of processing parameters in thermally induced phase separation technique on porous architecture of scaffolds for bone tissue engineering. Journal of biomedical materials research Part B: applied biomaterials 2014, 102, 1304–1315. [Google Scholar] [CrossRef]
- Zhang, Y.S.; Haghiashtiani, G.; Hübscher, T.; Kelly, D.J.; Lee, J.M.; Lutolf, M.; McAlpine, M.C.; Yeong, W.Y.; Zenobi-Wong, M.; Malda, J. 3D extrusion bioprinting. Nature Reviews Methods Primers 2021, 1, 75. [Google Scholar] [CrossRef]
- McCauley, P.J.; Fromen, C.A.; Bayles, A.V. Cell viability in extrusion bioprinting: the impact of process parameters, bioink rheology, and cell mechanics. Rheologica Acta 2025, 1–19. [CrossRef]
- Tumbleston, J.R.; Shirvanyants, D.; Ermoshkin, N.; Janusziewicz, R.; Johnson, A.R.; Kelly, D.; Chen, K.; Pinschmidt, R.; Rolland, J.P.; Ermoshkin, A. Continuous liquid interface production of 3D objects. Science 2015, 347, 1349–1352. [Google Scholar] [CrossRef]
- Dhand, A.P.; Davidson, M.D.; Burdick, J.A. Lithography-based 3D printing of hydrogels. Nature reviews bioengineering 2025, 3, 108–125. [Google Scholar] [CrossRef] [PubMed]
- Carlberg, L.; Keku, I.; Zhang, Y.; Forbes, J.; Thang, M.; Perry, J.; Hingtgen, S. Characterization of a bioprinted anticancer cell therapy system generated with continuous liquid interface production. Advanced NanoBiomed Research 2025. [Google Scholar] [CrossRef] [PubMed]
- Hakim Khalili, M.; Zhang, R.; Wilson, S.; Goel, S.; Impey, S.A.; Aria, A.I. Additive manufacturing and physicomechanical characteristics of PEGDA hydrogels: recent advances and perspective for tissue engineering. Polymers 2023, 15, 2341. [Google Scholar] [CrossRef] [PubMed]
- Ying, G.; Jiang, N.; Yu, C.; Zhang, Y.S. Three-dimensional bioprinting of gelatin methacryloyl (GelMA). Bio-Design and Manufacturing 2018, 1, 215–224. [Google Scholar] [CrossRef]
- Pepelanova, I.; Kruppa, K.; Scheper, T.; Lavrentieva, A. Gelatin-methacryloyl (GelMA) hydrogels with defined degree of functionalization as a versatile toolkit for 3D cell culture and extrusion bioprinting. Bioengineering 2018, 5, 55. [Google Scholar] [CrossRef]
- Choi, J.R.; Yong, K.W.; Choi, J.Y.; Cowie, A.C. Recent advances in photo-crosslinkable hydrogels for biomedical applications. BioTechniques 2019, 66, 40–53. [Google Scholar] [CrossRef]
- Bagheri, A.; Jin, J. Photopolymerization in 3D printing. ACS Applied Polymer Materials 2019, 1, 593–611. [Google Scholar] [CrossRef]
- Abid, A.R.; Marciniak, B.; Pędziński, T.; Shahid, M. Photo-stability and photo-sensitizing characterization of selected sunscreens’ ingredients. Journal of Photochemistry and Photobiology A: Chemistry 2017, 332, 241–250. [Google Scholar] [CrossRef]
- Xu, H.; Casillas, J.; Krishnamoorthy, S.; Xu, C. Effects of Irgacure 2959 and lithium phenyl-2, 4, 6-trimethylbenzoylphosphinate on cell viability, physical properties, and microstructure in 3D bioprinting of vascular-like constructs. Biomedical Materials 2020, 15, 055021. [Google Scholar] [CrossRef] [PubMed]
- Cao, P.; Mooney, R.; Tirughana, R.; Abidi, W.; Aramburo, S.; Flores, L.; Gilchrist, M.; Nwokafor, U.; Haber, T.; Tiet, P.; et al. Intraperitoneal Administration of Neural Stem Cell-Nanoparticle Conjugates Targets Chemotherapy to Ovarian Tumors. Bioconjug Chem 2017, 28, 1767–1776. [Google Scholar] [CrossRef] [PubMed]
- Mooney, R.; Abidi, W.; Batalla-Covello, J.; Ngai, H.W.; Hyde, C.; Machado, D.; Abdul-Majid, A.; Kang, Y.; Hammad, M.; Flores, L. Allogeneic human neural stem cells for improved therapeutic delivery to peritoneal ovarian cancer. Stem Cell Research & Therapy 2021, 12, 1–11. [Google Scholar] [CrossRef]
- Mooney, R.; Majid, A.A.; Batalla-Covello, J.; Machado, D.; Liu, X.; Gonzaga, J.; Tirughana, R.; Hammad, M.; Dellinger, T.H.; Lesniak, M.S. Enhanced delivery of oncolytic adenovirus by neural stem cells for treatment of metastatic ovarian cancer. Molecular Therapy-Oncolytics 2019, 12, 79–92. [Google Scholar] [CrossRef]
- Hammad, M.; Cornejo, Y.R.; Batalla-Covello, J.; Majid, A.A.; Burke, C.; Liu, Z.; Yuan, Y.-C.; Li, M.; Dellinger, T.H.; Lu, J. Neural stem cells improve the delivery of oncolytic chimeric orthopoxvirus in a metastatic ovarian cancer model. Molecular Therapy-Oncolytics 2020, 18, 326–334. [Google Scholar] [CrossRef]
- Kass, L.; Thang, M.; Zhang, Y.; DeVane, C.; Logan, J.; Tessema, A.; Perry, J.; Hingtgen, S. Development of a biocompatible 3D hydrogel scaffold using continuous liquid interface production for the delivery of cell therapies to treat recurrent glioblastoma. Bioengineering & Translational Medicine 2024, 9, e10676. [Google Scholar] [CrossRef]
- Jiang, W.; Yang, Y.; Mercer-Smith, A.R.; Valdivia, A.; Bago, J.R.; Woodell, A.S.; Buckley, A.A.; Marand, M.H.; Qian, L.; Anders, C.K. Development of next-generation tumor-homing induced neural stem cells to enhance treatment of metastatic cancers. Science Advances 2021, 7, eabf1526. [Google Scholar] [CrossRef] [PubMed]
| Disease / Indication | Cell Type in Clinical Trials (Representative Study Number) |
Cell Type in Preclinical Studies (Key Reference) |
|---|---|---|
| Cancer | ||
| Ovarian Cancer | CAR-T (NCT07181720) CAR-M (NCT04660929) NK (NCT06321484) MSC-INFβ (NCT02530047) γδ T Cells (Pilot, UMIN-CTR UMIN000015233[5]) |
NSC-nanoparticle Conjugate[2] Polymer-encapsulated RPE-IL2[6] Cytokine-Induced Killer (CIK) Cells[7] iPSC-Derived NK Cells[8] |
| Colorectal/Colon Cancer | CAR-T (NCT07179692) | CAR-NK cells[9] |
| Gastric Cancer | CAR-T (NCT07179692) γδ T Cells (Pilot, UMIN-CTR UMIN000004130[10]) Tumor-Associated Lymphocyte (TALs)[11] |
Cytokine-Induced Killer (CIK) Cells[12] CAR-NK Cells[13] |
| Esophagogastric Cancer | CAR-T (NCT06623396) CAR-M (NCT04660929) |
|
|
Regenerative Cell Replacement |
||
| Pediatric Acute Liver Failure | Alginate Microbeads Encapsulated Hepatocyte and Mesenchymal Stromal Cells (NCT05491135, “HELP”- first-in-huma trial[14]) | Micro-encapsulated Hepatocyte[15] |
| Type 1 Diabetes | Alginate-Polylysine Microcapsules Encapsulated Human Islet Cells (first-in-human trial[16]) | Encapsulated Islet Cells[17] |
|
Inflammatory & Autoimmune Disease |
||
| Crohn's Disease | Tolerogenic Dendritic Cells (EudraCT number 2007-003469-42) | MSCs[18] |
| Colitis | - | MSCs[19] Alternatively Activated Macrophages[20] |
| Necrotizing Enterocolitis (NEC) | - | Amniotic Fluid Stem Cells (AFSCs)[21] |
| Liver Fibrosis | Microencapsulated MSCs[22] | |
| Peritoneal Fibrosis (Dialysis) | - | MSCs[23] |
| Bladder Fibrosis | - | MSCs[24] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




