Submitted:
05 March 2026
Posted:
09 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Plant Material and Sample Preparation
2.2. Reference Measurements
2.3. Saffron Corms Preparation and VIS-NIR Spectroscopy
2.4. Spectral Data Pre-Processing
2.5. Feature Engineering: Narrow-Band Ratio Indices
2.6. Machine Learning Techniques for Moisture and Starch Estimating
2.7. Model Validation and Performance Metrics
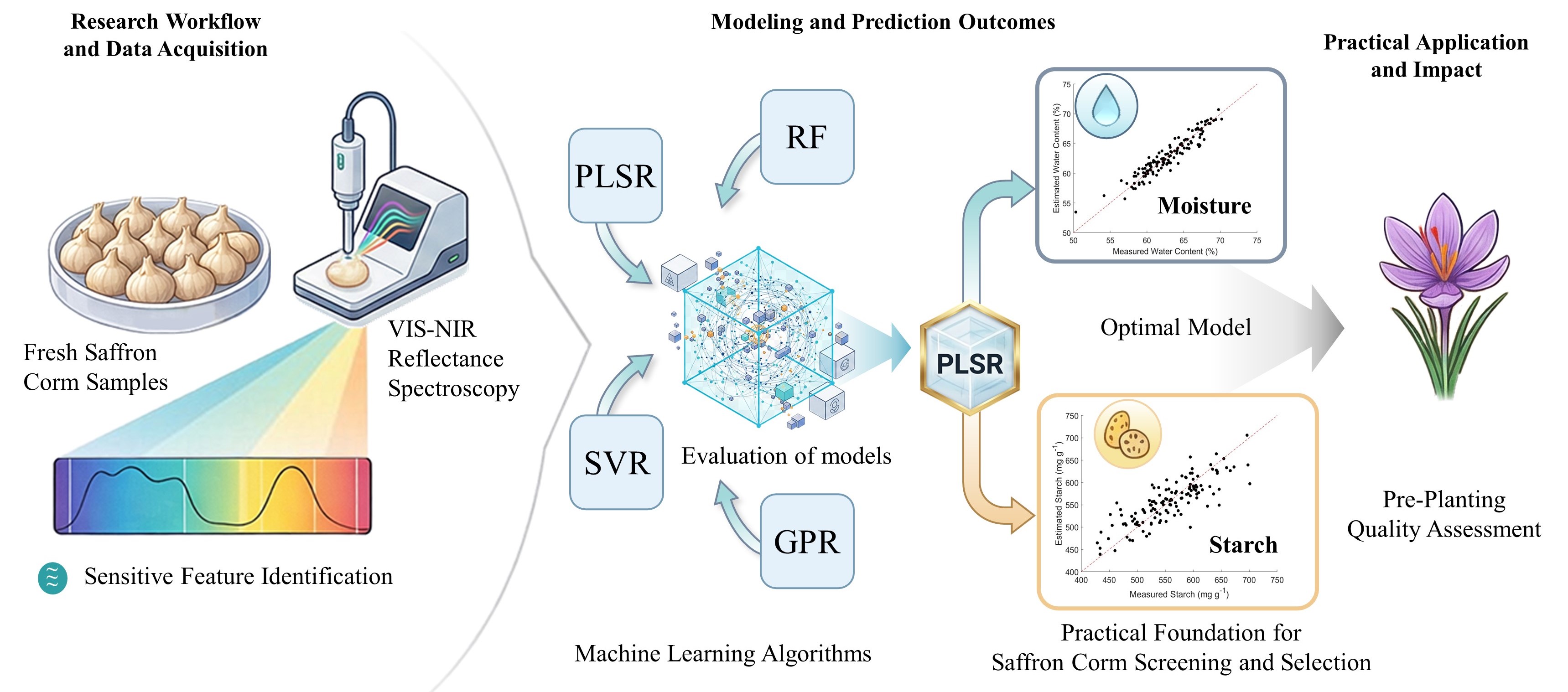
3. Results
3.1. Starch and Moisture Content in Saffron Corms
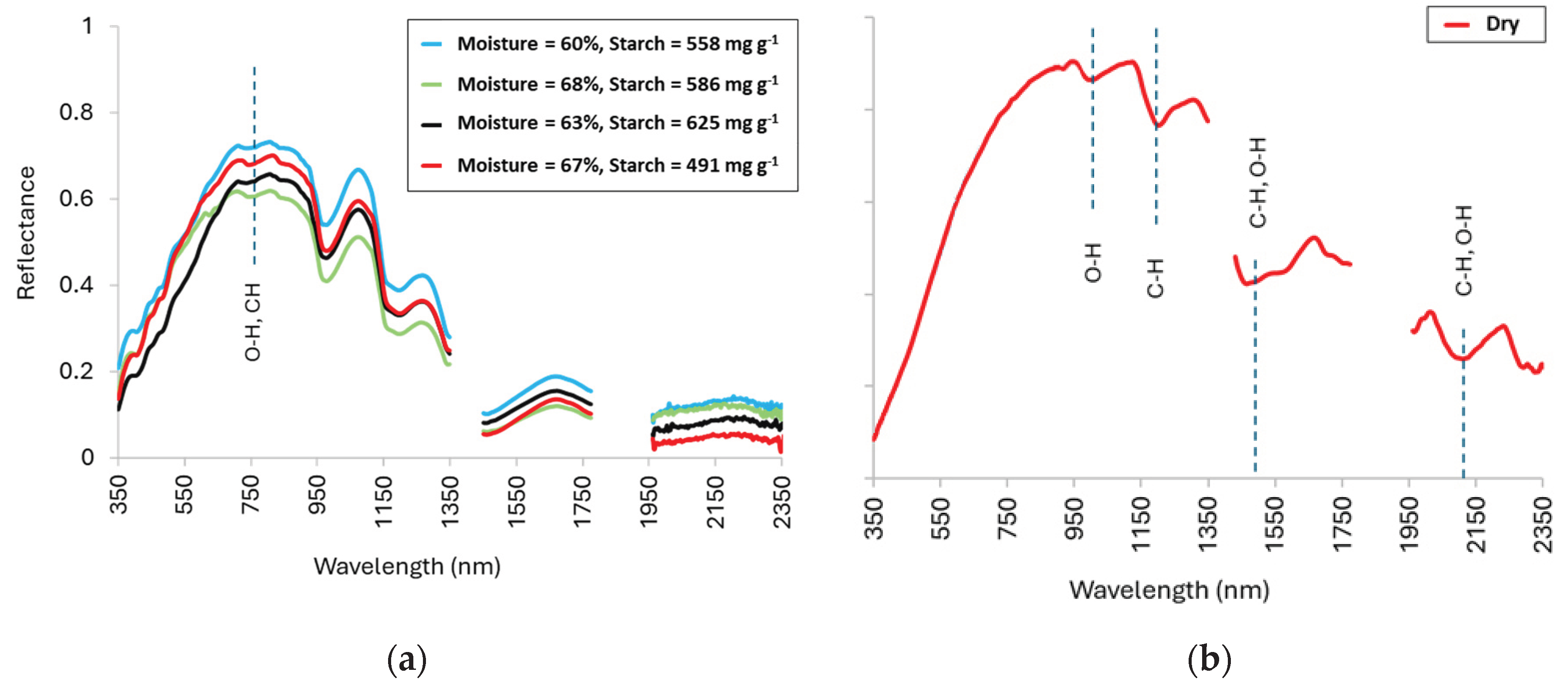
3.2. Spectral Characteristics of Saffron Corm
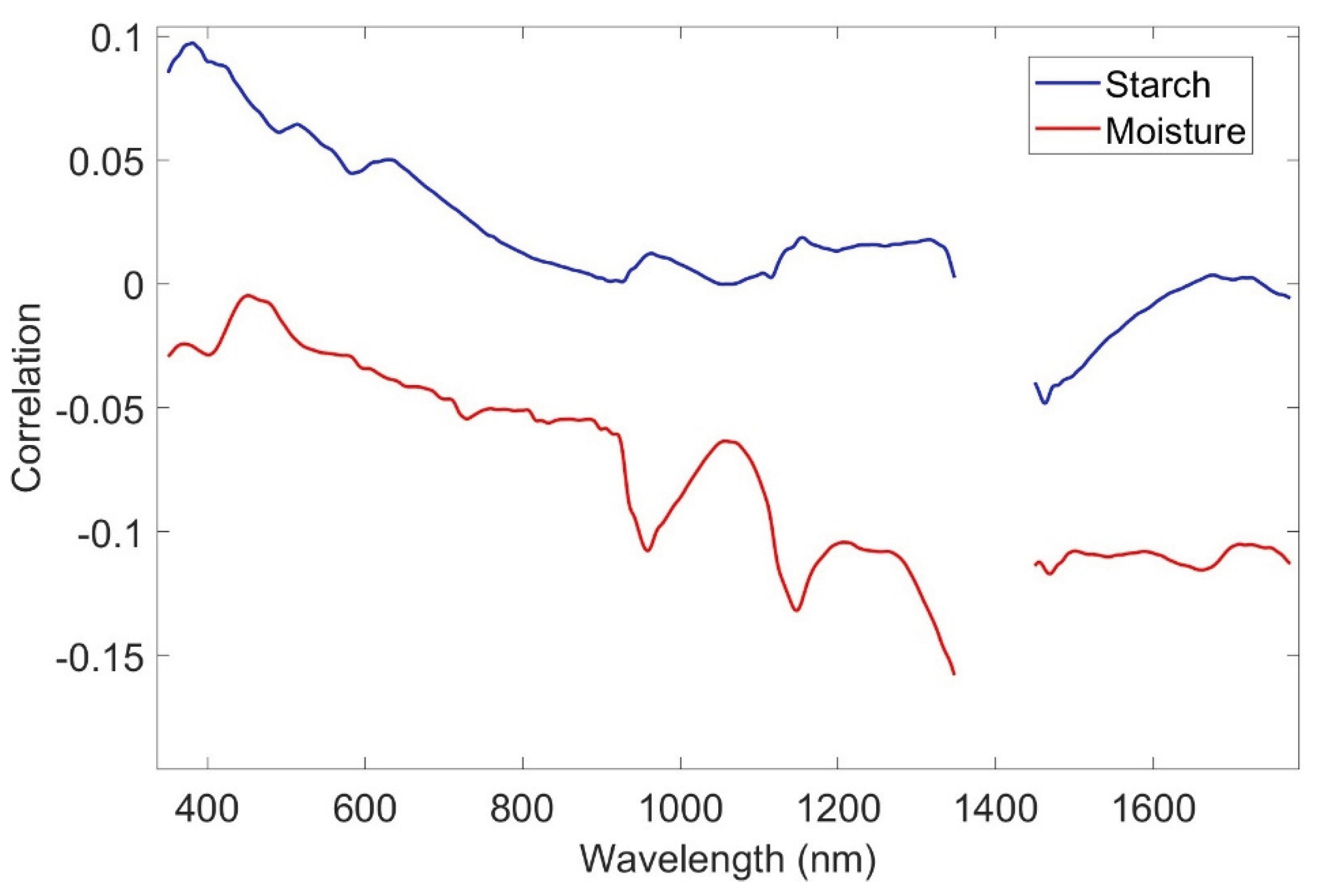
3.3. Wavelength-Specific Correlation with Starch and Moisture
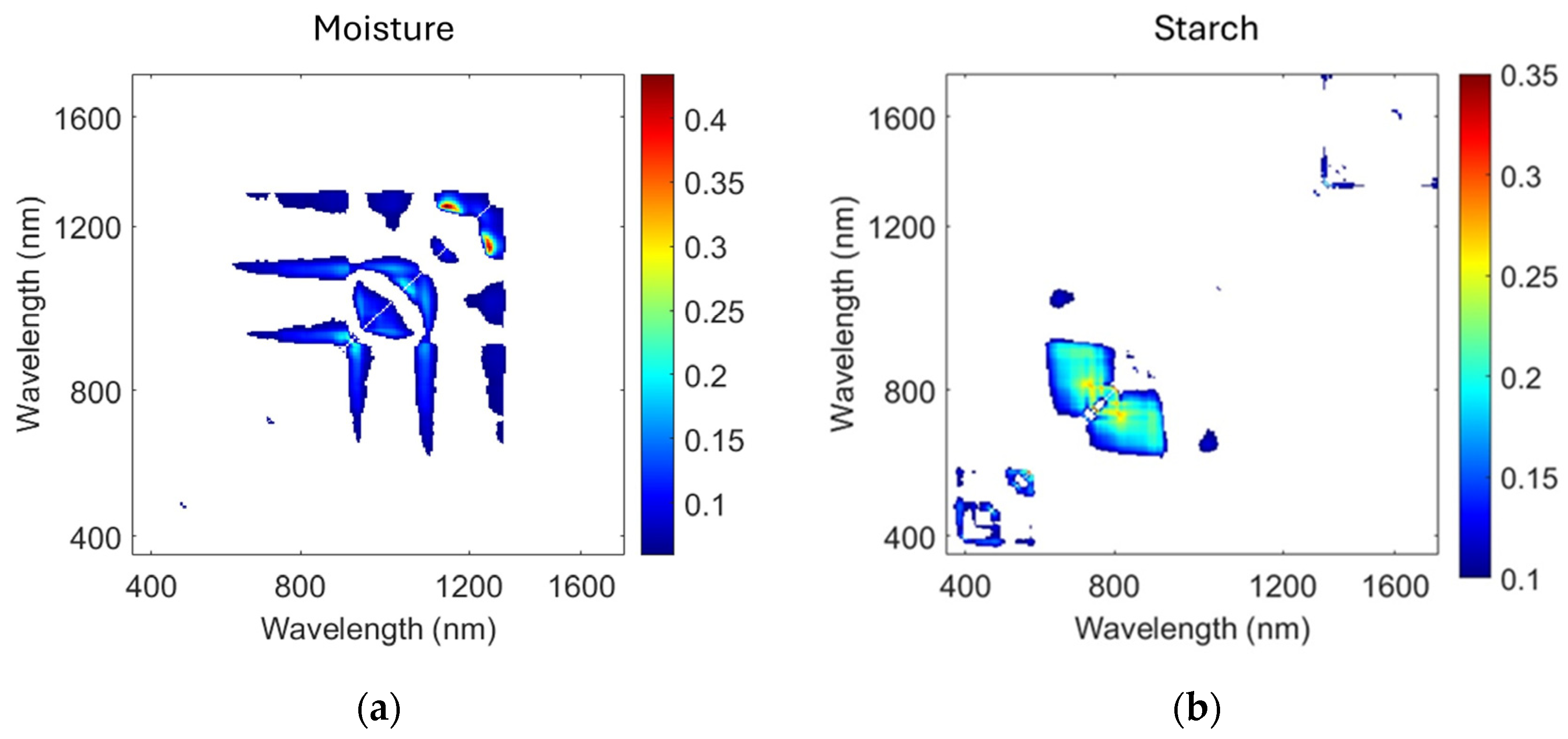
3.4. Optimal Narrow-Band Spectral Indices
3.5. Predictive Performance for Starch and Water Content Estimation
4. Discussion
4.1. Starch and Moisture Content in Saffron Corms
4.2. Spectral Signatures and Their Biochemical Basis
4.3. Spectral Profiles of Fresh and Powdered Saffron Corms Across 400–2350 nm
4.4. Two-Dimensional R2 Maps of RI Band-Pair Indices: Key Wavelengths for Water and Starch
4.5. Machine Learning Algorithms Performance For Starch and Moisture Prediction
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AMG | Amyloglucosidase |
| GC | Gas Chromatography |
| GPR | Gaussian Process Regression |
| GOPOD | Glucose Oxidase/Peroxidase |
| HPLC | High-Performance Liquid Chromatography |
| PLSR | Partial Least Squares Regression |
| RPD | Residual Predictive Deviation |
| RPIQ | Ratio of Performance to Interquartile Range |
| RMSE | Root Mean Square Error |
| RTS | Rapid Total Starch |
| SWIR | Short-Wave Infrared |
| SVR | Support Vector Regression |
| RF | Random Forest |
| VIS-NIR | Visible & Near-Infrared |
References
- Cardone, L.; Castronuovo, D.; Perniola, M.; Cicco, N.; Candido, V. Saffron (Crocus Sativus L.), the King of Spices: An Overview. Sci. Hortic. 2020, 272. [Google Scholar] [CrossRef]
- Avila-Sosa, R.; Nevárez-Moorillón, G.V.; Ochoa-Velasco, C.E.; Navarro-Cruz, A.R.; Hernández-Carranza, P.; Cid-Pérez, T.S. Detection of Saffron’s Main Bioactive Compounds and Their Relationship with Commercial Quality. Foods 2022, 11. [Google Scholar] [CrossRef] [PubMed]
- Cardone, L.; Candido, V.; Castronuovo, D.; Perniola, M.; Cicco, N. Comparing Annual and Biennial Crop Cycle on the Growth, Yield and Quality of Saffron Using Three Corm Dimensions. Sci. Hortic. 2021, 288. [Google Scholar] [CrossRef]
- Naseri, E.; Dalir, N.; Mokhtassi-Bidgoli, A.; Ebadi, M.T.; Rahnemaie, R. Optimizing Saffron Cormlet Production through Substrate Composition Nutrient Concentration and Irrigation Management in Soilless Cultivation. Sci. Rep. 2025, 15. [Google Scholar] [CrossRef]
- Dewir, Y.H.; Alsadon, A.; Ibrahim, A.; El-Mahrouk, M. Effects of Growing Substrate, Mode of Nutrient Supply, and Saffron Corm Size on Flowering, Growth, Photosynthetic Competence, and Cormlet Formation in Hydroponics. Horttechnology 2022, 32, 234–240. [Google Scholar] [CrossRef]
- Ralli, P.E.; Menexes, G.; Dordas, C.A. Corm Size and Planting Density Affect the Yield and Longevity of Saffron (Crocus Sativus L.). Not. Bot. Horti Agrobot. Cluj. Napoca. 2024, 52. [Google Scholar] [CrossRef]
- Chrungoo, N.K.; Farooq, S. Correlative Changes in Carbohydrate Content and Starch Hydrolysing Enzymes in Corms of Saffron Crocus (Crocus Sativus L.) during Dormancy and Sprouting. Biochemie und Physiologie der Pflanzen 1985, 180, 55–61. [Google Scholar] [CrossRef]
- Kamenetsky, R.; Zemah, H.; Ranwala, A.P.; Vergeldt, F.; Ranwala, N.K.; Miller, W.B.; Van As, H.; Bendel, P. Water Status and Carbohydrate Pools in Tulip Bulbs during Dormancy Release. New Phytologist 2003, 158, 109–118. [Google Scholar] [CrossRef]
- Bagri, J.; Yadav, A.; Anwar, K.; Dkhar, J.; Singla-Pareek, S.L.; Pareek, A. Metabolic Shift in Sugars and Amino Acids Regulates Sprouting in Saffron Corm. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef]
- Renau-Morata, B.; Nebauer, S.G.; Sánchez, M.; Molina, R. V. Effect of Corm Size, Water Stress and Cultivation Conditions on Photosynthesis and Biomass Partitioning during the Vegetative Growth of Saffron (Crocus Sativus L.). Ind. Crops Prod. 2012, 39, 40–46. [Google Scholar] [CrossRef]
- Maleki, F.; Kazemi, H.; Siahmarguee, A.; Kamkar, B. Development of a Land Use Suitability Model for Saffron (Crocus Sativus L.) Cultivation by Multi-Criteria Evaluation and Spatial Analysis. Ecol. Eng. 2017, 106, 140–153. [Google Scholar] [CrossRef]
- Acharya, B.S.; Ghalehgolabbehbahani, A.; Hamido, S.; Zinati, G.; Bozzolo, A.; Archer, L.; Wendelberger, K.; Das, S.; Thapa, R.; Panday, D. Saffron (Crocus Sativus L.): The Golden Spice — Management, Challenges, and Opportunities for Sustainable Production in the United States. J. Agric. Food Res. 2025, 21. [Google Scholar] [CrossRef]
- Ziaei, S.M.; Feizi, H.; Siuki, A.K.; Sahabi, H. Yield and Quality of Saffron (Crocus Sativus L.) in Response to Priming Treatments and Water Deficit. Italian Journal of Agronomy 2024, 19. [Google Scholar] [CrossRef]
- Mena-García, A.; Sanz, M.L.; Díez-Municio, M.; Ruiz-Matute, A.I. A Combined Gas and Liquid Chromatographic Approach for Quality Evaluation of Saffron-Based Food Supplements. Foods 2023, 12. [Google Scholar] [CrossRef]
- Eghbali, S.; Farhadi, F.; Askari, V.R. An Overview of Analytical Methods Employed for Quality Assessment of Crocus Sativus (Saffron). Food Chem. X 2023, 20. [Google Scholar] [CrossRef]
- Nondestructive Determination of Dry Matter and Soluble Solids Content in Dehydrator Onions and Garlic Using a Handheld Visible; Thailand, 2017.
- Ricci, C.; Gadaleta, A.; Gerardino, A.; Didonna, A.; Ferrara, G.; Bertani, F.R. Portable optical spectroscopy and machine learning techniques for quantification of the biochemical content of raw food materials. CABI Agric Biosci 2024, 5, 39. [Google Scholar] [CrossRef]
- Angel Prieto Lage, M.; Barral Martínez, M.; Seyyedimansour, S.; Manikantan, M.R.; Trif, M.; Khaneghah, M.A. Recent Advancements in NIR Spectroscopy for Assessing the Quality and Safety of Horticultural Products: A Comprehensive Review. Front. Nutr. 9. [CrossRef] [PubMed]
- Fodor, M.; Matkovits, A.; Benes, E.L.; Jókai, Z. The Role of Near-Infrared Spectroscopy in Food Quality Assurance: A Review of the Past Two Decades. Foods 2024, 13. [Google Scholar] [CrossRef] [PubMed]
- Darvishi Boloorani, A.; Mirzaei, S.; Bahrami, H.A.; Soleimani, M.; Neysani Samany, N.; Papi, R.; Mahmoudi, M.; Bakhtiari, M.; Huete, A. In vitro plant spectral response reveals dust stress. Agric. For. Meteorol. 2025, 370. [Google Scholar] [CrossRef]
- Darvishi Boloorani, A.; Ranjbar, S.; Mirzaei, S.; Bahrami, H.A.; Mirzapour, F.; Abbaszadeh Tehrani, N. Spectral behavior of Persian oak under compound stress of water deficit and dust storm. Int. J. Appl. Earth Obs. Geoinf. 2020, 88, 102082. [Google Scholar] [CrossRef]
- Rawal, A.; Hartemink, A.; Zhang, Y.; Wang, Y.; Lankau, R.A.; Ruark, M.D. Visible and Near-Infrared Spectroscopy Predicted Leaf Nitrogen Contents of Potato Varieties under Different Growth and Management Conditions. Precis. Agric. 2024, 25, 751–770. [Google Scholar] [CrossRef]
- Bai, S.H.; Tootoonchy, M.; Kämper, W.; Tahmasbian, I.; Farrar, M.B.; Boldingh, H.; Pereira, T.; Jonson, H.; Nichols, J.; Wallace, H.M.; et al. Predicting Carbohydrate Concentrations in Avocado and Macadamia Leaves Using Hyperspectral Imaging with Partial Least Squares Regressions and Artificial Neural Networks. Remote Sens. (Basel). 2024, 16. [Google Scholar] [CrossRef]
- Wang, Z.; Li, J.; Zhang, C.; Fan, S. Development of a General Prediction Model of Moisture Content in Maize Seeds Based on LW-NIR Hyperspectral Imaging. Agriculture (Switzerland) 2023, 13. [Google Scholar] [CrossRef]
- Alighaleh, P.; Pakdel, R.; Ghanei Ghooshkhaneh, N.; Einafshar, S.; Rohani, A.; Saeidirad, M.H. Detection and Classification of Saffron Adulterants by Vis-Nir Imaging, Chemical Analysis, and Soft Computing. Foods 2023, 12. [Google Scholar] [CrossRef] [PubMed]
- Xu, N.; Chen, B.; Sun, H.; Huang, W.; Wu, Y.; Lu, L.; Lou, X.; Xie, C. High-Throughput Phenotyping of Multiple Quantitative Traits for Saffron Corm Rot via NIR Hyperspectral Imaging and Broad Learning System. Microchemical Journal 2026, 220, 116603. [Google Scholar] [CrossRef]
- Verrelst, J.; Malenovský, Z.; Van der Tol, C.; Camps-Valls, G.; Gastellu-Etchegorry, J.P.; Lewis, P.; North, P.; Moreno, J. Quantifying Vegetation Biophysical Variables from Imaging Spectroscopy Data: A Review on Retrieval Methods. Surv. Geophys. 2019, 40, 589–629. [Google Scholar] [CrossRef]
- Basak, D.; Pal, S.; Patranabis, D.C. Support Vector Regression; 2007; Vol. 11. [Google Scholar]
- Smola, A.J.; Sch¨olkopf, B.; Sch¨olkopf, S. A Tutorial on Support Vector Regression; Kluwer Academic Publishers, 2004; Volume 14, pp. 199–222. [Google Scholar]
- Geladi, P.; Kowalski, B.R. Partial Least-Squares Regression: A Tutorial. Anal. Chim. Acta 1986, 185, 1–17. [Google Scholar] [CrossRef]
- Abdel-Rahman, E.M.; Mutanga, O.; Odindi, J.; Adam, E.; Odindo, A.; Ismail, R. Estimating Swiss Chard Foliar Macro- and Micronutrient Concentrations under Different Irrigation Water Sources Using Ground-Based Hyperspectral Data and Four Partial Least Squares (PLS)-Based (PLS1, PLS2, SPLS1 and SPLS2) Regression Algorithms. Comput. Electron. Agric. 2017, 132, 21–33. [Google Scholar] [CrossRef]
- dos Santos Neto, J.P.; de Assis, M.W.D.; Casagrande, I.P.; Cunha Júnior, L.C.; de Almeida Teixeira, G.H. Determination of ‘Palmer’ Mango Maturity Indices Using Portable near Infrared (VIS-NIR) Spectrometer. Postharvest Biol. Technol. 2017, 130, 75–80. [Google Scholar] [CrossRef]
- Breiman, L. Random Forests. Machine Learning 2001, 45, 5–32. [Google Scholar] [CrossRef]
- Rodriguez-Galiano, V.F.; Ghimire, B.; Rogan, J.; Chica-Olmo, M.; Rigol-Sanchez, J.P. An Assessment of the Effectiveness of a Random Forest Classifier for Land-Cover Classification. ISPRS Journal of Photogrammetry and Remote Sensing 2012, 67, 93–104. [Google Scholar] [CrossRef]
- Pallotti, C.; Renau-Morata, B.; Cardone, L.; Nebauer, S.G.; Albiñana Palacios, M.; Rivas-Sendra, A.; Seguí-Simarro, J.M.; Molina, R. V. Understanding the Saffron Corm Development—Insights into Histological and Metabolic Aspects. Plants 2024, 13. [Google Scholar] [CrossRef] [PubMed]
- Gusain, S.; Joshi, R. Morphological, Physiological, and Transcriptional Changes in Crocus Sativus L. Under In Vitro Polyethylene Glycol-Induced Water Stress. Biology (Basel). 2025, 14. [Google Scholar] [CrossRef]
- Bertoft, E. Understanding Starch Structure: Recent Progress. Agronomy 2017, 7. [Google Scholar] [CrossRef]
- Pérez, S.; Baldwin, P.M.; Gallant, D.J. Structural Features of Starch Granules I. In Starch; Elsevier Inc., 2009; pp. 149–192. ISBN 9780127462752. [Google Scholar]
- Roger, P.; Bello-Perez, L.A.; Colonna, P. Contribution of Amylose and Amylopectin to the Light Scattering Behaviour of Starches in Aqueous Solution. Polymer 1999, 40(25), 6897–6909. [Google Scholar] [CrossRef]
- Zeng, L.; Chen, C. Simultaneous Estimation of Amylose, Resistant, and Digestible Starch in Pea Flour by Visible and near-Infrared Reflectance Spectroscopy. Int. J. Food Prop. 2018, 21, 1129–1137. [Google Scholar] [CrossRef]
- Sims, D.A.; Gamon, J.A. Estimation of Vegetation Water Content and Photosynthetic Tissue Area from Spectral Reflectance: A Comparison of Indices Based on Liquid Water and Chlorophyll Absorption Features. Remote Sensing of Environment 2003, 84, 526–537. [Google Scholar] [CrossRef]
- Hennessy, A.; Clarke, K.; Lewis, M. Hyperspectral Classification of Plants: A Review of Waveband Selection Generalisability. Remote Sens. (Basel). 2020, 12. [Google Scholar] [CrossRef]
- Li, C.; Czyż, E.A.; Halitschke, R.; Baldwin, I.T.; Schaepman, M.E.; Schuman, M.C. Evaluating Potential of Leaf Reflectance Spectra to Monitor Plant Genetic Variation. Plant Methods 2023, 19. [Google Scholar] [CrossRef]
- Farhadi, R.; Afkari-Sayyah, A.H.; Jamshidi, B.; Mousapour Gorji, A. Prediction of internal compositions change in potato during storage using visible/near-infrared (Vis/NIR) spectroscopy. International Journal of Food Engineering 2020, 16(4), 20190110. [Google Scholar] [CrossRef]
- Daughtry, C.S.T.; Hunt, E.R.; McMurtrey, J.E. Assessing Crop Residue Cover Using Shortwave Infrared Reflectance. Remote Sens. Environ. 2004, 90, 126–134. [Google Scholar] [CrossRef]
- Lamb, B.T.; Dennison, P.E.; Hively, W.D.; Kokaly, R.F.; Serbin, G.; Wu, Z.; Dabney, P.W.; Masek, J.G.; Campbell, M.; Daughtry, C.S.T. Optimizing Landsat Next Shortwave Infrared Bands for Crop Residue Characterization. Remote Sens. (Basel). 2022, 14. [Google Scholar] [CrossRef]
- Sadeghi, A.; Khani, S.; Sabourian, R.; Hajimahmoodi, M.; Ghasemi, J.B. Integrating CNNs and chemometrics for ana-lyzing NIR spectra and RGB images in turmeric adulterant detection. J. Food Compos. Anal. 2025, 141, 107324. [Google Scholar] [CrossRef]
- Cozzolino, D. The Role of Near-Infrared Sensors to Measure Water Relationships in Crops and Plants. Appl. Spectrosc. Rev. 2017, 52, 837–849. [Google Scholar] [CrossRef]
- Sun, Y.; Liu, Y.; Yu, H.; Xie, A.; Li, X.; Yin, Y.; Duan, X. Non-Destructive Prediction of Moisture Content and Freezable Water Content of Purple-Fleshed Sweet Potato Slices during Drying Process Using Hyperspectral Imaging Technique. Food Anal. Methods 2017, 10, 1535–1546. [Google Scholar] [CrossRef]
- Ringsted, T.; Siesler, H.W.; Engelsen, S.B. Monitoring the Staling of Wheat Bread Using 2D MIR-NIR Correlation Spectroscopy. J. Cereal Sci. 2017, 75, 92–99. [Google Scholar] [CrossRef]
- Cozzolino, D.; Degner, S.; Eglinton, J. A Review on the Role of Vibrational Spectroscopy as an Analytical Method to Measure Starch Biochemical and Biophysical Properties in Cereals and Starchy Foods. Foods 2014, 3, 605–621. [Google Scholar] [CrossRef] [PubMed]
- Jin, X.; Shi, C.; Yu, C.Y.; Yamada, T.; Sacks, E.J. Determination of Leaf Water Content by Visible and Near-Infrared Spectrometry and Multivariate Calibration in Miscanthus. Front. Plant Sci. 2017, 8. [Google Scholar] [CrossRef]
- Arefi, A.; Sturm, B.; Hoffmann, T. Explainability of Deep Convolutional Neural Networks When It Comes to NIR Spectral Data: A Case Study of Starch Content Estimation in Potato Tubers. Food Control 2025, 169. [Google Scholar] [CrossRef]
- Frey, L.A.; Baumann, P.; Aasen, H.; Studer, B.; Kölliker, R. A Non-Destructive Method to Quantify Leaf Starch Content in Red Clover. Front. Plant Sci. 2020, 11. [Google Scholar] [CrossRef] [PubMed]

| Property | n | Min | Mean | Max | Standard Deviation |
|---|---|---|---|---|---|
| Starch Content (mg g−1) | 130 | 428 | 558 | 701 | 59 |
| Moisture Content (%) | 130 | 50.3 | 63 | 70.3 | 3.5 |
| Optimal Band Pair | R2 | RMSE | Equation |
|---|---|---|---|
| RI (λ1182, λ1305) | 0.41 | 2.82 | |
| RI (λ735, λ810) | 0.35 | 43.1 | g−1 |
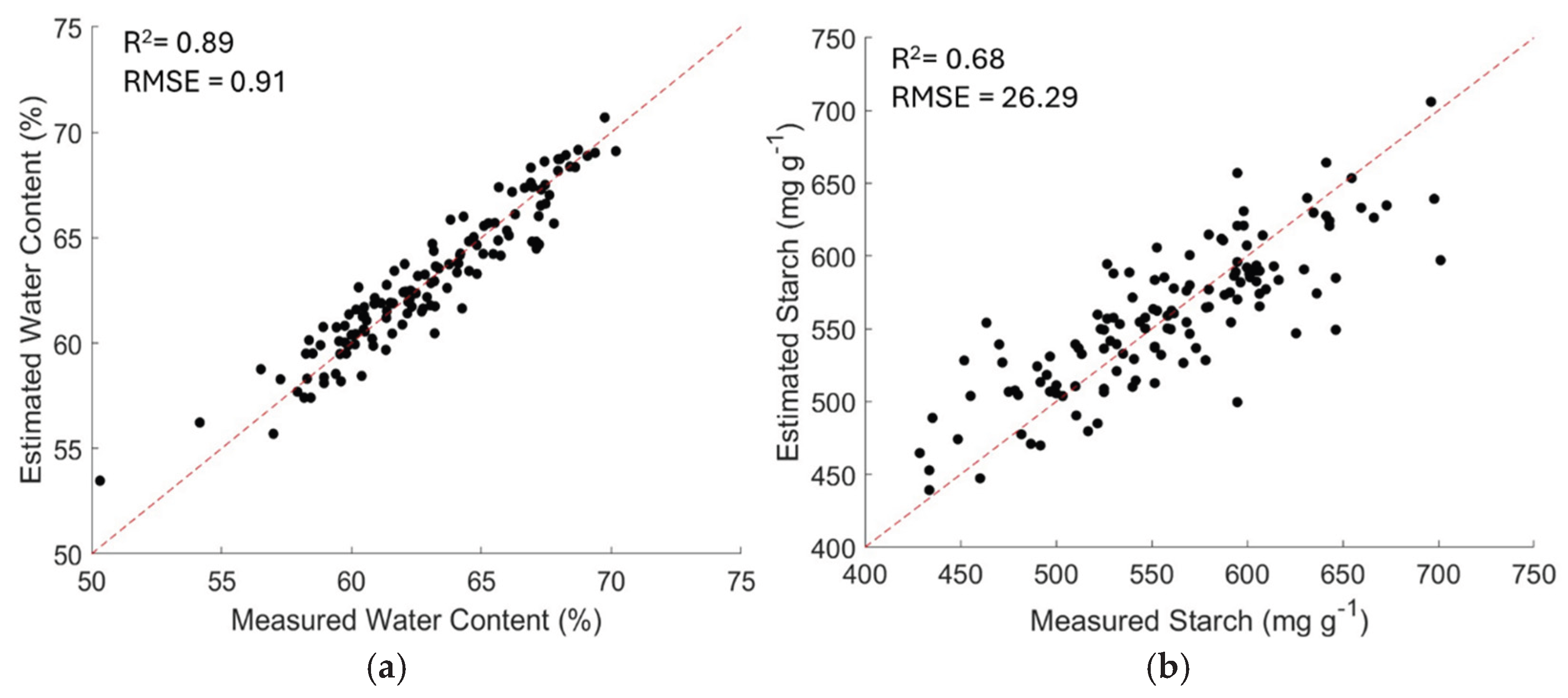
| Property | Algorithm | R2 | RMSE | RPD | RPIQ |
| Starch | GPR | 0.63 | 28.31 | 1. 79 | 2.16 |
| SVR | 0.59 | 32.43 | 1.54 | 1.98 | |
| PLSR | 0.68 | 26.29 | 1.87 | 2.37 | |
| RF | 0.65 | 27.14 | 1.87 | 2.33 | |
| Moisture Content |
GPR | 0.85 | 0.97 | 3.64 | 4.37 |
| SVR | 0.79 | 1.19 | 2.56 | 2.98 | |
| PLSR | 0.89 | 0.91 | 3.67 | 4.91 | |
| RF | 0.80 | 1.04 | 2.98 | 3.28 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




