Submitted:
05 March 2026
Posted:
06 March 2026
You are already at the latest version
Abstract
Keywords:
Highlights
- Seventeen Inga species documented in Imbabura, northern Ecuador.
- Species distributions are structured by altitude and soil properties.
- Ten species exhibit narrow thermal and altitudinal specialization, others are generalists.
- Uplift and volcanic substrates generate fine-scale edaphic mosaics.
- Geodiversity likely amplifies ecological filtering in Andean forests.
1. Introduction
2. Materials and Methods
2.1. Study Area
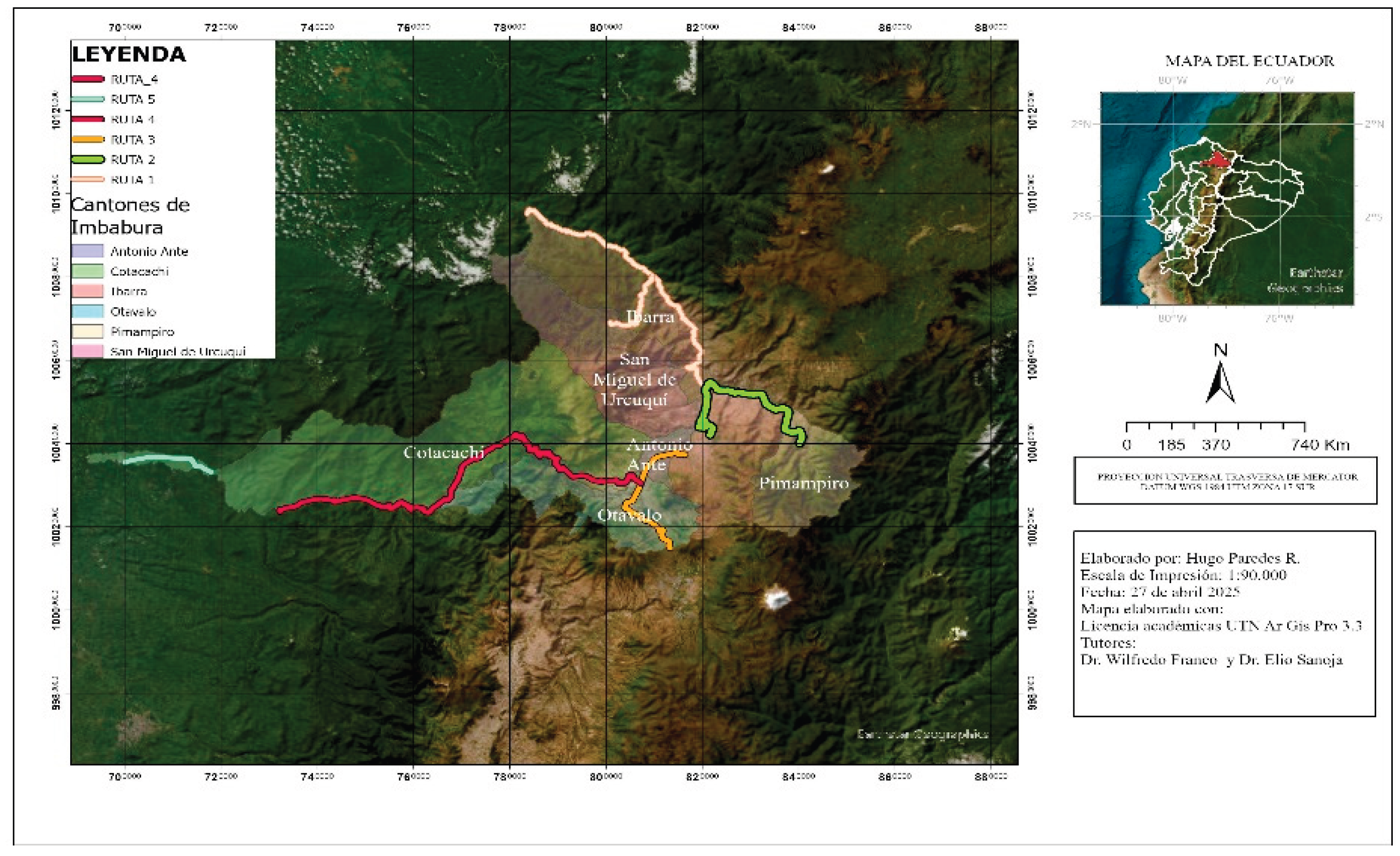
2.2. Data Collection and Sampling Strategy
- 1)
- Ibarra–Lita (73.13 km) and San Gerónimo–Buenos Aires (22.77 km);
- 2)
- Ibarra–Pimampiro (51 km);
- 3)
- Ibarra–Otavalo–Cajas (37.88 km);
- 4)
- Otavalo-Intag (116.7 km);
- 5)
- Las Golondrinas (19.53 km).
2.3. Taxonomic Identification
2.4. Geospatial and Ecological Analysis
3. Results
3.1. Floristic Composition and Diversity
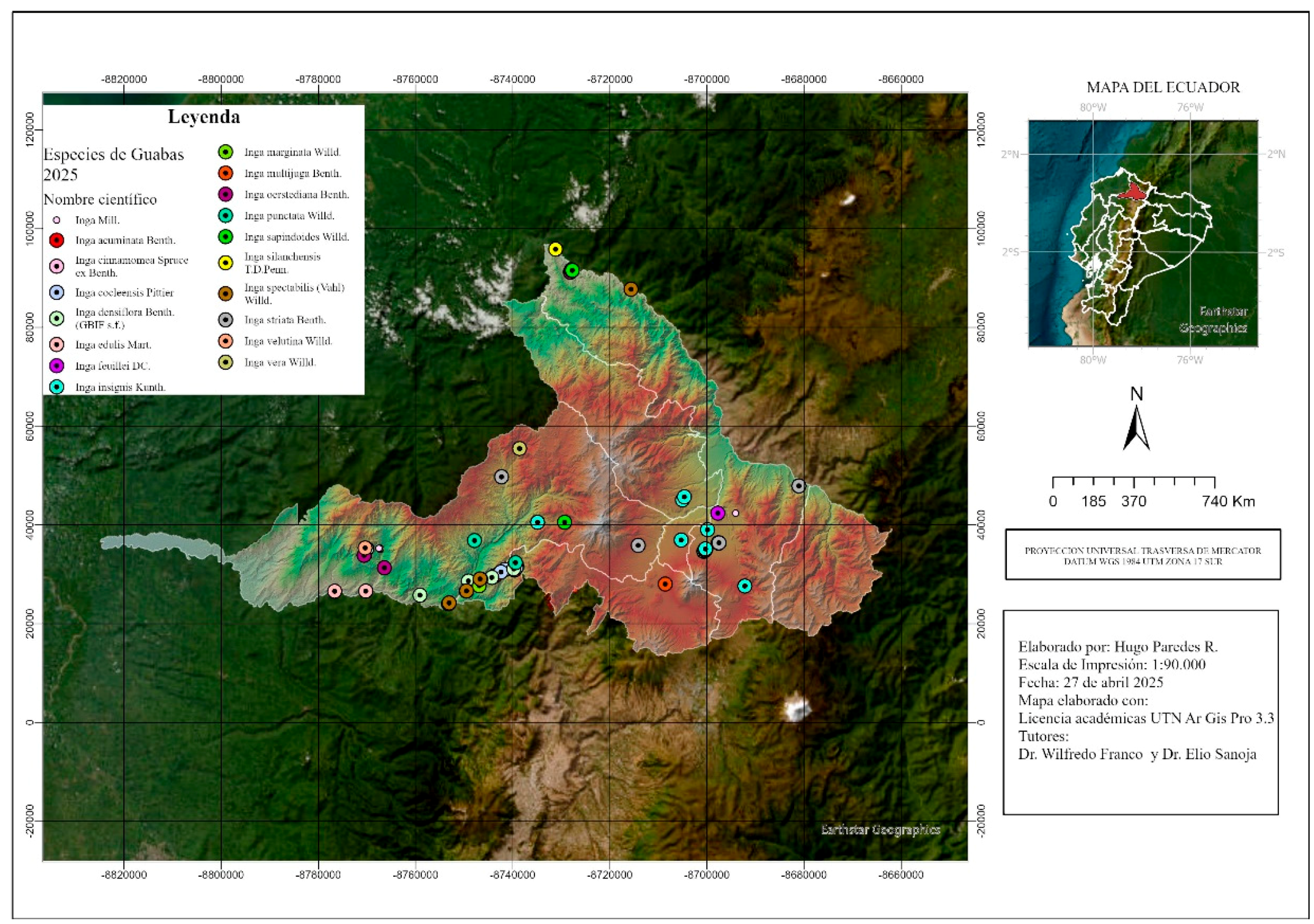
3.2. Thermal and Altitudinal Distribution Patterns
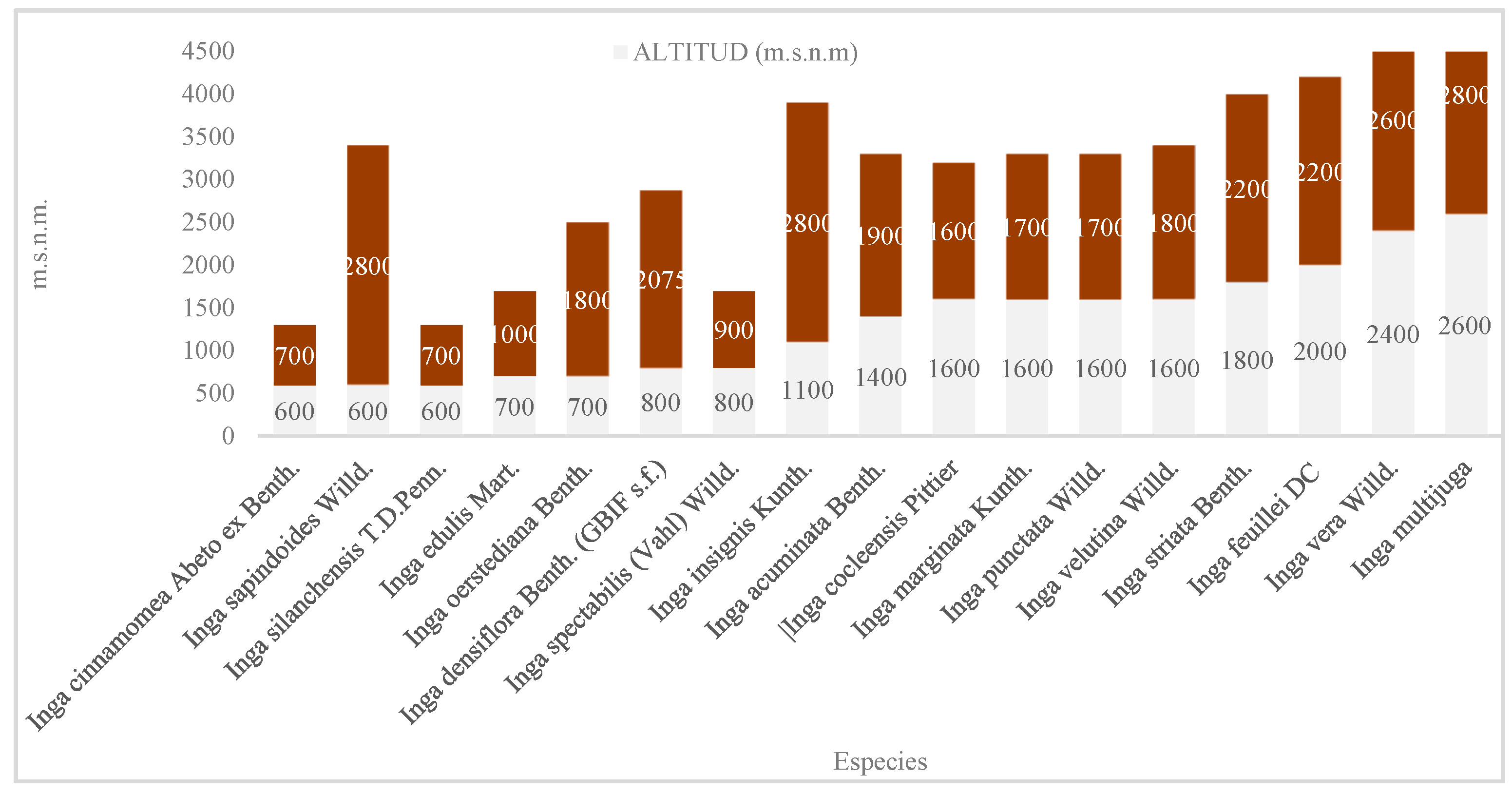
- Specialized species: A group of species, including I. cinnamomea, I. silanchensis, I. edulis, I. spectabilis, I. marginata, I. punctata, I. velutina and I. feuillei, occupy narrow altitudinal ranges with variations of less than 500 m. These species are restricted to specific microhabitats [Table 1; Figure A2].
- Generalist species: In contrast, I. sapindoides, I. oerstediana, I. densiflora, I. insignis and I. striata exhibit extensive plasticity and thrive across broad elevation gradients [Figure 3]. For example, I. densiflora was recorded between 800 and 2,075 m a.s.l., and I. sapindoides between 600 and 2,800 m a.s.l. [Table 1; Figure 3].
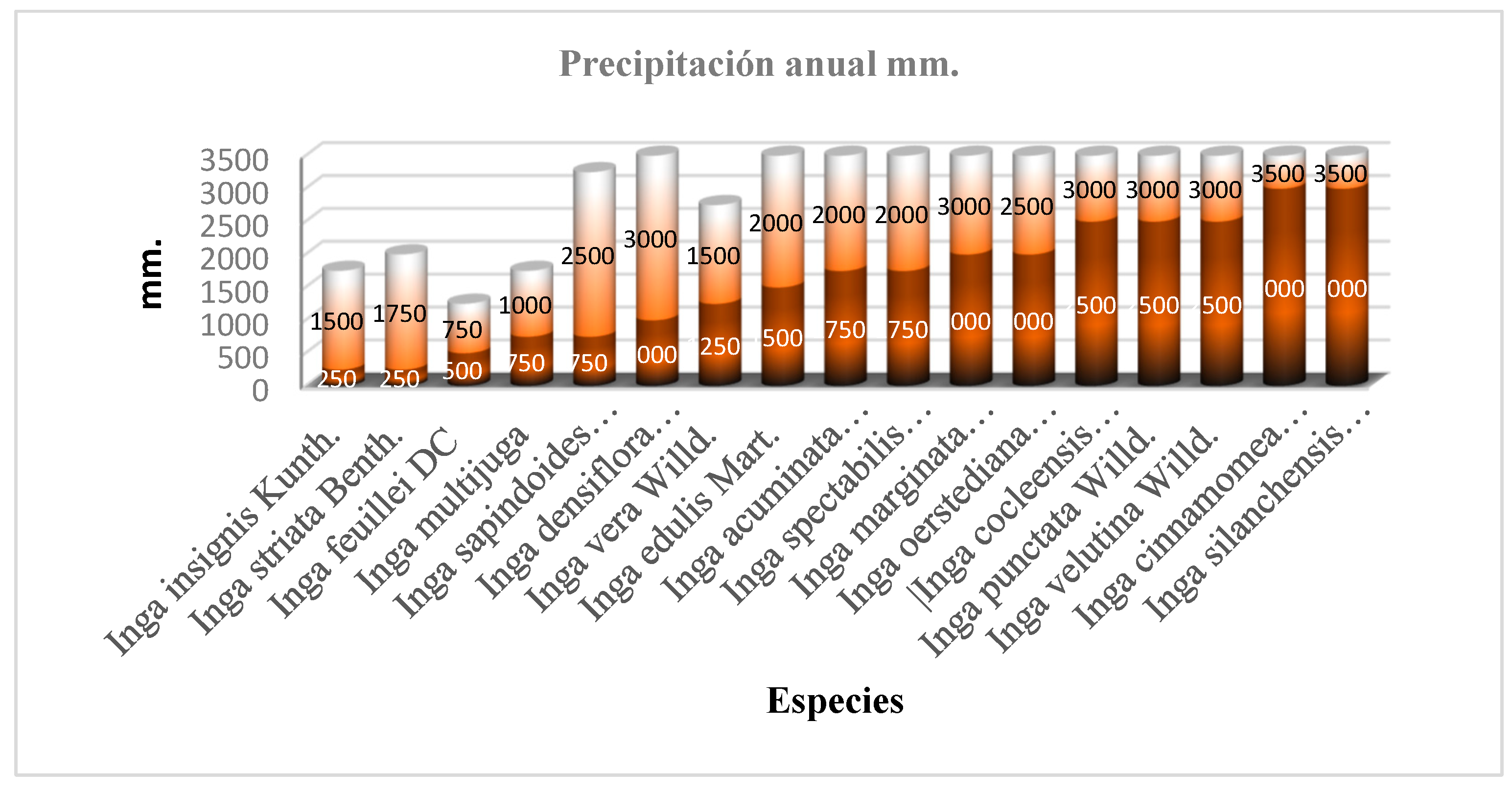
3.3. Soil Preferences and Precipitation Regimes
- Soil texture: Most species prefer sandy loam soils. I. feuillei, I. marginata and I. punctata were predominantly found in these textures, while I. densiflora showed adaptation to sandy clay soils.
- Soil taxonomy: The genus in Imbabura is mainly associated with Inceptisols and Mollisols. I. edulis and I. striata are notably associated with Mollisols, while I. multijuga and I. silanchensis are restricted to Inceptisols.
- pH tolerance: most species (approx. 94%) thrive in neutral to slightly acidic soils (pH ≈ 6.5–7.0). However, I. insignis and I. silanchensis tolerate acidic conditions, while I. feuillei was the only species recorded in alkaline soils.
4. Discussion
4.1. Ecological Filters: Altitude and Climate Differentiation
- Specialists: 58.8% of the species occupy restricted altitudinal floors. This pattern supports recent findings in Andean tropical forests, where the rate of thermal gradient restricts the distribution of trees to specific thermal floors [45]. These limited-range species are particularly susceptible to biotic attrition driven by climate change [30].
- Generalists and Distribution Changes: Species such as I. densiflora and I. sapindoides exhibited extensive plasticity. Notably, our data show that I. densiflora reaches 2075 m a.s.l., an altitude higher than the limit of 1900 m reported in Peru [12]. This upward extension could indicate thermophilization of Andean forests, where lowland species migrate upslope in response to increasing temperatures, a phenomenon widely documented in the tropical Andes [31,32]. Conversely, ecotonal barriers could be preventing the migration of less adaptable species, creating strong barriers to distribution [33].
4.2. Edaphic Influence, Nitrogen Fixation and Functional Traits
4.3. Geological and Evolutionary Multi-Scale Drivers of Local Inga Richness in Imbabura
4.4. Biotic Interactions and Human-Mediated Landscapes
5. Conclusions
Author Contributions
Funding
Expressions of gratitude
Conflicts of interest
Appendix A
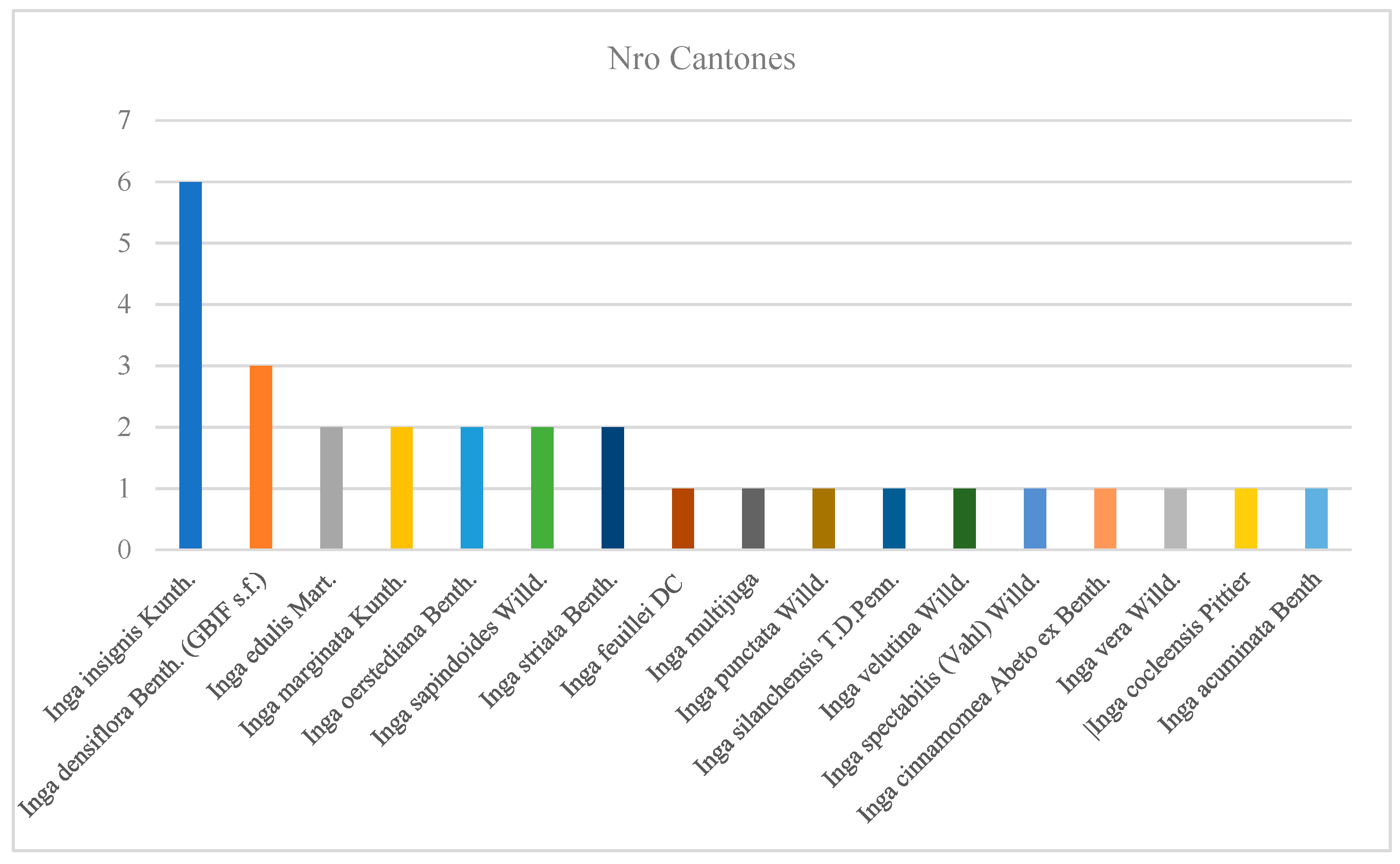
- A1.- Herbariums reviewed in their databases and contained information on the genus Inga in Imbabura.
| Species | Number of samples | Collection date |
|---|---|---|
| Inga acuminata Benth. | 2 | 2 025 |
| Inga coclensis | 7 | 2 025 |
| Inga densiflora Benth. (GBIF s.f.) | 10 | 2 025 |
| Inga edulis Mart. | 4 | 2 024; 2 025 |
| Inga insignis Kunth. | 18 | 2 025 |
| Inga oerstediana Benth. | 3 | 2 024 |
| Inga sapindoides Willd. | 2 | 2 024 |
| Inga spectabilis (Vahl) Willd. | 3 | 2 025 |
| Inga striata Benth. | 2 | 2 025 |
| Inga punctata Willd. | 1 | 2 025 |
Appendix B
References
- Tomaseili, M. Introducción a la dendrocronología: Anatomía de la madera en los trópicos. In Proceedings of the II Curso Internacional: Anatomía y dendrocronología tropical, Huancayo, Peru, 2023; p. 5. [Google Scholar]
- Neill, D. La evolución de las especies en el Ecuador: Las Magnoliaceae. In Proceedings of the V Congreso de Bosques de la Amazonía Ecuatoriana; Universidad Estatal Amazónica: Puyo, Ecuador, 2019; p. 5. [Google Scholar]
- Ministerio del Ambiente del Ecuador (MAE). Deforestación del Ecuador continental 2014-2016; MAE: Quito, Ecuador, 2017. [Google Scholar]
- Bruneau, A.; de Queiroz, L.P.; Ringelberg, J.; Borges, L. Advances in Legume Systematics 14. Classification of Caesalpinioideae. Part 2: Higher-level classification. PhytoKeys 2024, 239, 1–552. [Google Scholar] [CrossRef]
- Palacios, W. Árboles del Ecuador: Familias y géneros; Universidad Técnica del Norte: Ibarra, Ecuador, 2016. [Google Scholar]
- Pennington, T.D.; Revelo, N. El género Inga en el Ecuador: Morfología, distribución y usos; Herbario Nacional del Ecuador: Quito, Ecuador, 1997. [Google Scholar]
- Gutiérrez, E. Efecto del cambio climático en la distribución potencial de cinco especies arbóreas de bosque templado en México. Rev. Mex. Biodivers. 2014, 85, 52–55. [Google Scholar] [CrossRef]
- Denevan, W.M. The Pristine Myth: The Landscape of the Americas in 1492. Ann. Assoc. Am. Geogr. 1992, 82, 369–385. [Google Scholar] [CrossRef]
- Sánchez-Albornoz, N. The Population of Latin America: A History; University of California Press: Berkeley, CA, USA, 1984. [Google Scholar]
- Paredes, H.; Rosales, O.; Varela, G.; Carvajal, G.; León, M. Herbario Universidad Técnica del Norte (HUTN), un laboratorio para conocer la diversidad de especies forestales del Ecuador. Ciencia Latina 2023, 7, 1167–1184. [Google Scholar] [CrossRef]
- Paredes, H. KoBoToolbox. Available online: https://kf.kobotoolbox.org/#/projects/home (accessed on 4 May 2024).
- Pennington, T.D. Flora ilustrada peruana – Inga (Leguminosas): Guabas y pacáes del Perú. Rev. For. Perú 2024, 39, 5–453. [Google Scholar] [CrossRef]
- INEC. Límites provinciales y cantonales; Instituto Nacional de Estadística y Censos: Quito, Ecuador, 2024. [Google Scholar]
- MAGAP. Mapa de fertilidad de suelos del Ecuador; Jiménez, J., Ed.; Ministerio de Agricultura, Ganadería, Acuacultura y Pesca: Quito, Ecuador, 2012. [Google Scholar]
- MAATE. Mapa interactivo del Ecuador. Available online: http://ide.ambiente.gob.ec/mapainteractivo/ (accessed on 10 January 2025).
- INAMHI. Visor climático. Available online: http://www.inamhi.gob.ec/visor/ (accessed on 20 April 2025).
- GBIF. Genus Inga. Available online: https://www.gbif.org/species/2965936 (accessed on 28 August 2023).
- Murga, H.; Coronado, M.F.; Abanto-Rodríguez, C.; De Almeida-Lobo, F. Gradiente altitudinal y su influencia en las características edafoclimáticas de los bosques tropicales. Madera Bosques 2021, 27, e271211. [Google Scholar]
- Castro, A. Biogeografía y modelos de distribución de especies. WorldClim 2020. [Google Scholar]
- Maciel, E.A.; Oliveira-Filho, A.T.; Eisenlohr, P.V. Distribution of Inga species in South America. J. Biogeogr. 2015, 42, 1–12. [Google Scholar]
- GBIF. Genus Inga. 2023. Available online: https://www.gbif.org/species/2965936.
- GBIF. Género Inga en Imbabura [Conjunto de datos] 2025. [CrossRef]
- Antonelli, A.; Nylander, J.A.A.; Persson, C.; Sanmartín, I. Tracing the impact of Andean uplift on Neotropical plant evolution. Proc. Natl. Acad. Sci. USA 2009, 106, 9749–9754. [Google Scholar] [CrossRef]
- Hoorn, C.; Wesselingh, F.P.; Ter Steege, H.; et al. Amazonia through time: Andean uplift, climate change, and biodiversity. Science 2010, 330, 927–931. [Google Scholar] [CrossRef] [PubMed]
- Hall, M.L.; Mothes, P. Volcanic history of the Ecuadorian Andes. J. Volcanol. Geotherm. Res. 2008, 176, 1–17. [Google Scholar] [CrossRef]
- Robin, C.; et al. Late Pleistocene volcanic activity in northern Ecuador. Bull. Volcanol. 2009, 71, 1043–1061. [Google Scholar]
- Vitousek, P.M.; Porder, S.; Houlton, B.Z.; Chadwick, O.A. Terrestrial phosphorus cycling: Regional variations and global disparities. Biogeochemistry 2010, 100, 227–239. [Google Scholar]
- Permiso de investigación No. MAATE-ARSFC-2023-0036, otorgado por el Ministerio de Ambiente, Agua y Transición Ecológica del Ecuador (MAATE).
- Richardson, J. E.; et al. Rapid diversification of a species-rich genus of neotropical rain forest trees. In Science; 2001. [Google Scholar]
- Kursar, T.A.; Dexter, K.G.; Lokvam, J.; Pennington, R.T.; Richardson, J.E.; Weber, M.G.; Murakami, E.T.; Drake, S.T.; McGregor, R.; Coley, P.D. The evolution of antiherbivore defenses and their role in binary risk of herbivory in Inga. Proc. Natl. Acad. Sci. USA 2009, 106, 18073–18078. [Google Scholar] [CrossRef]
- Dexter, K.G.; Terborgh, J.W.; Cunningham, C.W. Convergence in habitat specialization in Inga (Fabaceae). Systematics and Biodiversity 2012, 10, 235–244. [Google Scholar]
- Dexter, K.G.; Chave, J. Evolutionary patterns of range size, niche breadth and species survival in Inga. J. Biogeogr. 2010, 37, 2011–2020. [Google Scholar]
- Schley, R.J.; de la Estrella, M.; Pérez-Escobar, O.A.; Bruneau, A.; Barraclough, T.G.; Forest, F.; Klitgård, B.B. The genome sequence of Inga oerstediana Benth., a nitrogen-fixing tree used in agroforestry. Wellcome Open Res. 2024. [Google Scholar] [CrossRef]
- Nichols, J.D.; Carpenter, F.L. Interplanting Inga edulis with Terminalia amazonia in Costa Rica. Agrofor. Syst. 2006, 66, 1–7. [Google Scholar]
- Colwell, R.K.; Brehm, G.; Cardelús, C.L.; Gilman, A.C.; Longino, J.T. Global warming, elevational range shifts, and lowland biotic attrition. Science 2008, 322, 258–261. [Google Scholar] [CrossRef]
- Feeley, K.J.; Silman, M.R.; Bush, M.B.; Farfan, W.; Garcia, K.G.; Malhi, Y.; Meir, P.; Revilla, N.S.; Quispe, M.N.R.; Miles, L. Upslope migration of Andean trees. J. Biogeogr. 2011, 38, 783–791. [Google Scholar] [CrossRef]
- Fadrique, B.; Homeier, J.; Baez, S.; Duque, A.; Tello, J.S.; Renner, S.S.; Feeley, K.J. Widespread but heterogeneous responses of Andean forests to climate change. Nature 2018, 564, 407–411. [Google Scholar] [CrossRef] [PubMed]
- Lutz, D.A.; Powell, R.L.; Silman, M.R. Andean timberline migration and biodiversity loss with optimization of land management. PLoS ONE 2013, 8, e76101. [Google Scholar] [CrossRef] [PubMed]
- Gehring, C.; Vlek, P.L.G.; de Souza, L.A.G.; Denich, M. Biological nitrogen fixation in secondary regrowth in the central Amazon. Agric. Ecosyst. Environ. 2005, 111, 239–252. [Google Scholar] [CrossRef]
- Condit, R.; Engelbrecht, B.M.J.; Pino, D.; Pérez, R.; Turner, B.L. Species distributions in response to individual soil nutrients and seasonal drought across a tropical forest landscape. Proc. Natl. Acad. Sci. USA 2013, 110, 5064–5068. [Google Scholar] [CrossRef]
- Vitousek, P.M.; Porder, S.; Houlton, B.Z.; Chadwick, O.A. Terrestrial phosphorus cyclying: Regional variations and global disparities. Biogeochemistry 2010, 100, 227–239. [Google Scholar]
- Clement, C.R. 1492 and the loss of Amazonian crop genetic resources. I. The relation between domestication and population decline. Econ. Bot. 1999, 53, 188–202. [Google Scholar] [CrossRef]
- Lojka, B.; Dumas, L.; Preininger, D.; Polesny, Z.; Banout, J. The use and integration of Inga edulis in agroforestry systems in the Peruvian Amazon. Agrofor. Syst. 2011, 83, 67–81. [Google Scholar]
- Beer, J.; Muschler, R.; Kass, D.; Somarriba, E. Shade management in coffee and cacao plantations. Agrofor. Syst. 1998, 38, 139–164. [Google Scholar] [CrossRef]
- Somarriba, E.; Harvey, C.A.; Samper, M.; Anthony, F.; González, J.; Staver, C.; Rice, R.A. Biodiversity conservation in neotropical coffee (Coffea arabica) plantations. In Agroforestry and Biodiversity Conservation in Tropical Landscapes; Schroth, G., Ed.; Island Press: Washington, DC, USA, 2004; pp. 198–226. [Google Scholar]
- Holl, K.D.; Aide, T.M. When and where to actively restore ecosystems? For. Ecol. Manag. 2011, 261, 1558–1563. [Google Scholar] [CrossRef]
- Coley, P.D.; Lokvam, J.; Rudolph, K.; Pruitt, K.; Seger, J.; Kursar, T.A. Divergent defensive strategies of young leaves in two species of Inga. Ecology 2005, 86, 2633–2643. [Google Scholar] [CrossRef]
- Endara, M.J.; Weinhold, A.; Cox, J.E.; Wiggins, N.L.; Coley, P.D.; Kursar, T.A. Coevolutionary arms race versus host defense chase in a tropical herbivore–plant system. Proc. Natl. Acad. Sci. USA 2015, 112, E4569–E4576. [Google Scholar] [CrossRef]
- Duque, A.; Stevenson, P.R.; Feeley, K.J. Thermophilization of adult and juvenile tree communities in the northern Tropical Andes. Proc. Natl. Acad. Sci. USA 2015, 112, 10744–10749. [Google Scholar] [CrossRef]
- KoboToolbox. Available online: https://www.kobotoolbox.org/ (accessed on 23 January 2026).
| Species | Altitude (m a.s.l.) | Temp. (°C) | Precip. (mm) | Soil Order | Soil Texture | pH |
|---|---|---|---|---|---|---|
| I. edulis | 700–1000 | 21–22 | 1500–2000 | Mollisol | Clay-Loam | Slightly Acidic |
| I. feuillei | 2000–2200 | 15–16 | 500–750 | Mollisol | Sandy Loam | Alkaline |
| I. multijuga | 2600–2800 | 12–13 | 750–1000 | Inceptisol | Loam | Neutral |
| I. marginata | 1600–1700 | 17–19 | 1750–3000 | Inceptisol | Sandy Loam | Neutral |
| I. oerstediana | 700–1800 | 18–23 | 2000–2500 | Incept.+Entisol | Loam | Med. Acidic |
| I. insignis | 1100–2800 | 11–21 | 500–1500 | Incept.+Mollisol | Loam/Sandy | Variable |
| I. sapindoides | 600–2800 | 12–23 | 750–2500 | Inceptisol | Clay-Loam | Slightly Acidic |
| I. striata | 1800–2200 | 15–17 | 250–1750 | Incept.+Mollisol | Sandy Loam | Neutral |
| I. silanchensis | 600–700 | 22–23 | 3000–3500 | Inceptisol | Loam | Acidic |
| I. spectabilis | 800–900 | 21–22 | 1750–2000 | Mollisol | N/A | N/A |
| I. densiflora | 800–2075 | 14–21 | 1000–3000 | Incept.+Mollisol | Clay-Sandy | Slightly Acidic |
| I. cinnamomea | 600 | 22–23 | 3000–3500 | Inceptisol | Loam | Acidic |
| I. vera | 2400–2600 | 13–14 | 1250–1500 | Incept.+Entisol | Clay-Loam | Slightly Acidic |
| I. cocleensis | 1600 | 17–18 | 2500–3000 | Inceptisol | Sandy Loam | Neutral |
| I. acuminata | 1400–1900 | 16–18 | 1750–2000 | Incept.+Entisol | Sandy Loam | Med. Acidic |
| I. velutina | 1600-1800 | 17-18 | 2500-3000 | Incept.+Entisol | Clay-Loam | Slightly Acidic |
| I. punctata | 1600-1700 | 17-18 | 2500-3000 | Inceptisol | Sandy Loam | Neutral |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




