Submitted:
04 March 2026
Posted:
05 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Sites
2.2. Field Samplings
2.3. Extraction of MPs
2.4. Nile Red Dye Procedure
2.5. Fourier transform Infrared Spectroscopy Procedure
2.6. Detection and Identification of MPs
2.7. Quality Control and Quality Assurance
2.8. Statistical Analysis
3. Results
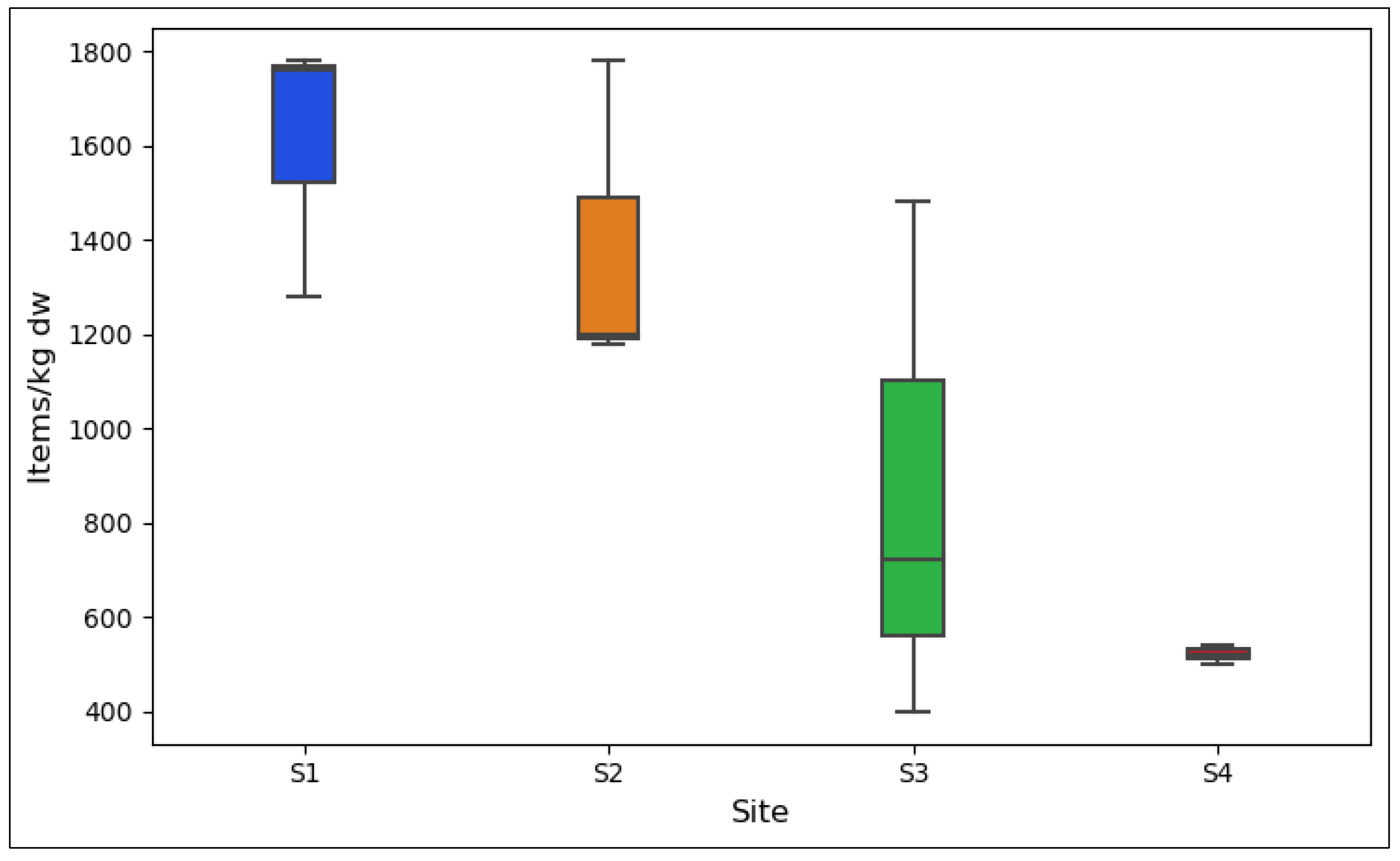
3.1. MPs Concentration in BRS Media
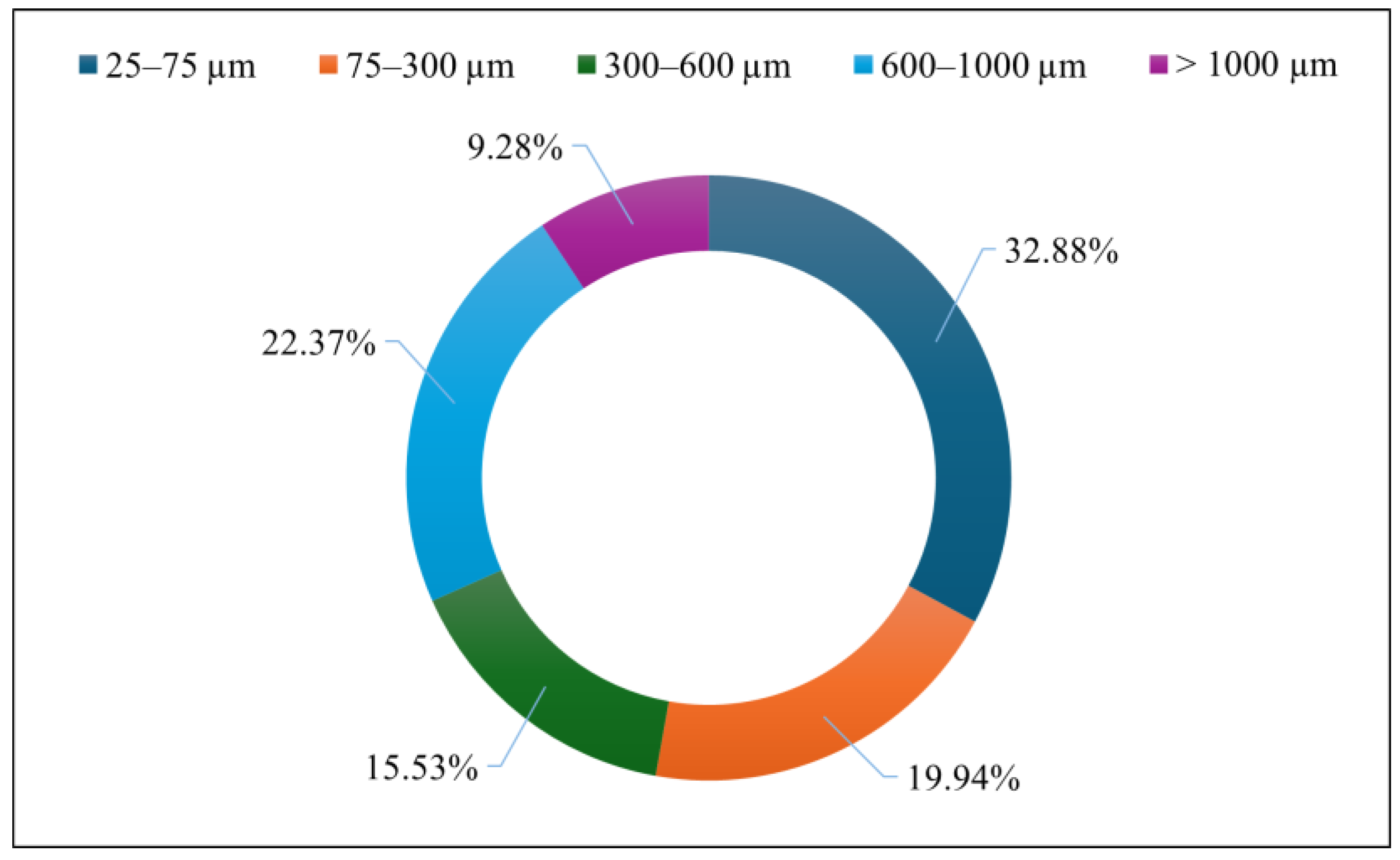
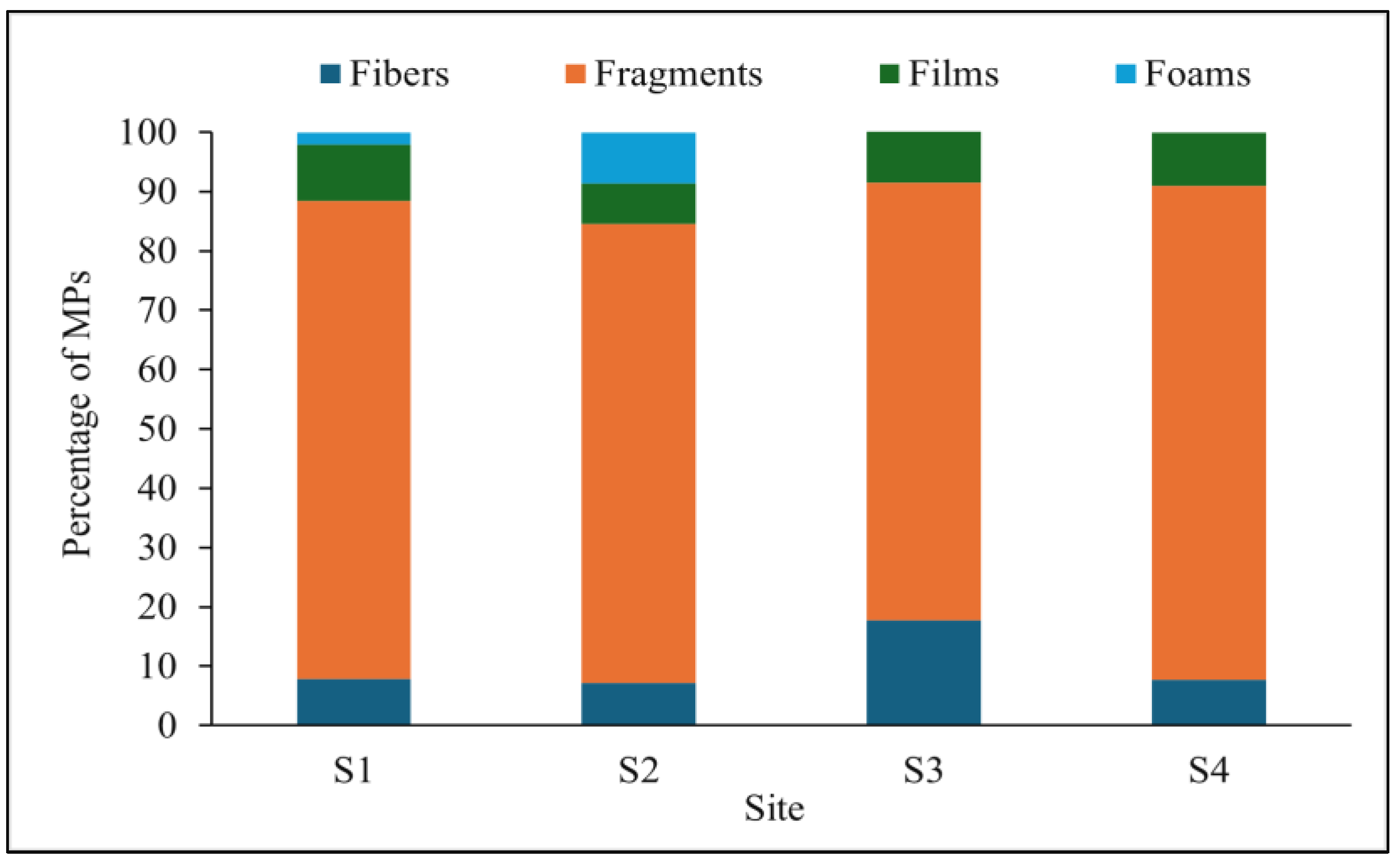
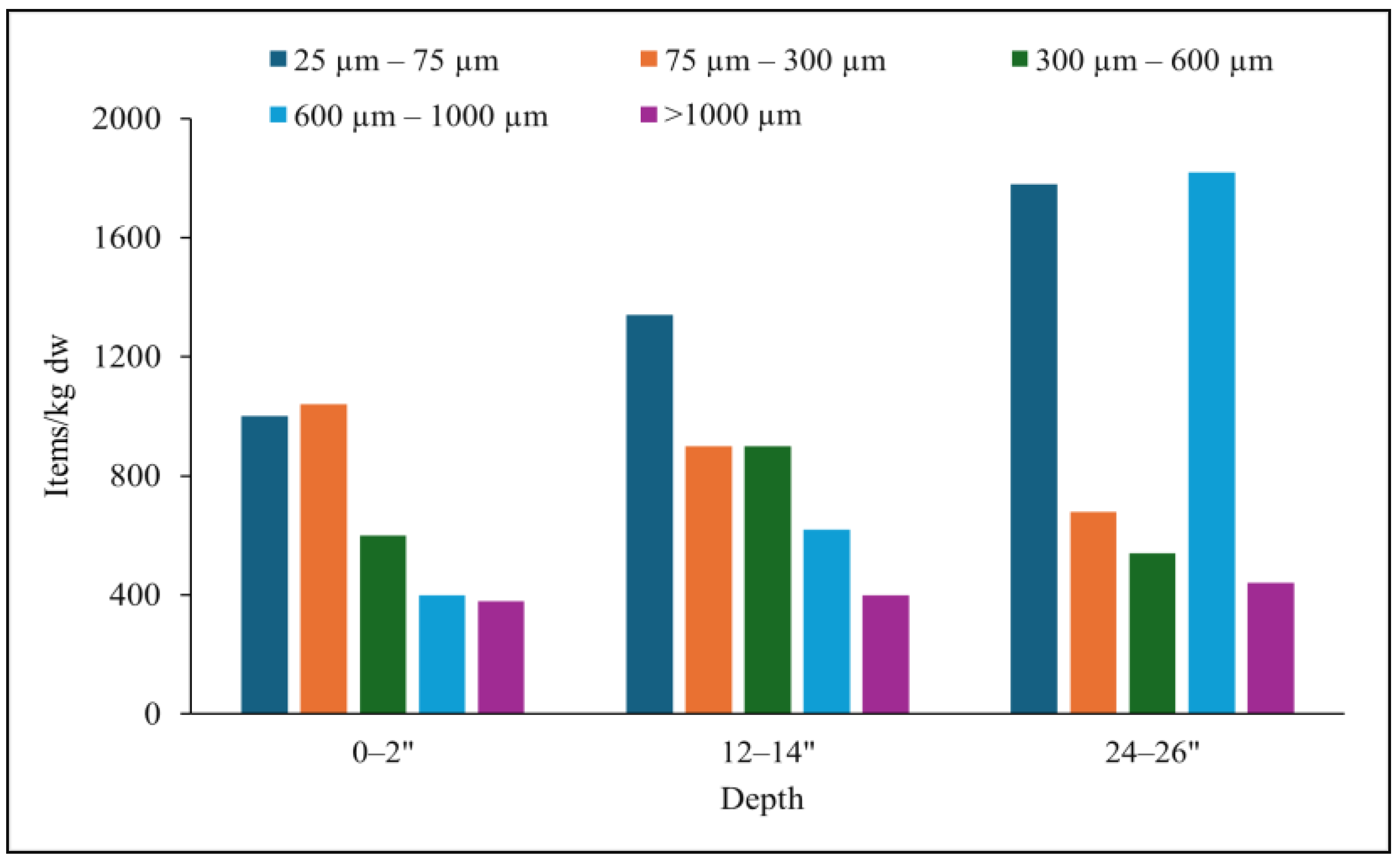
3.2. Size and Shape of MPs in BRS Media
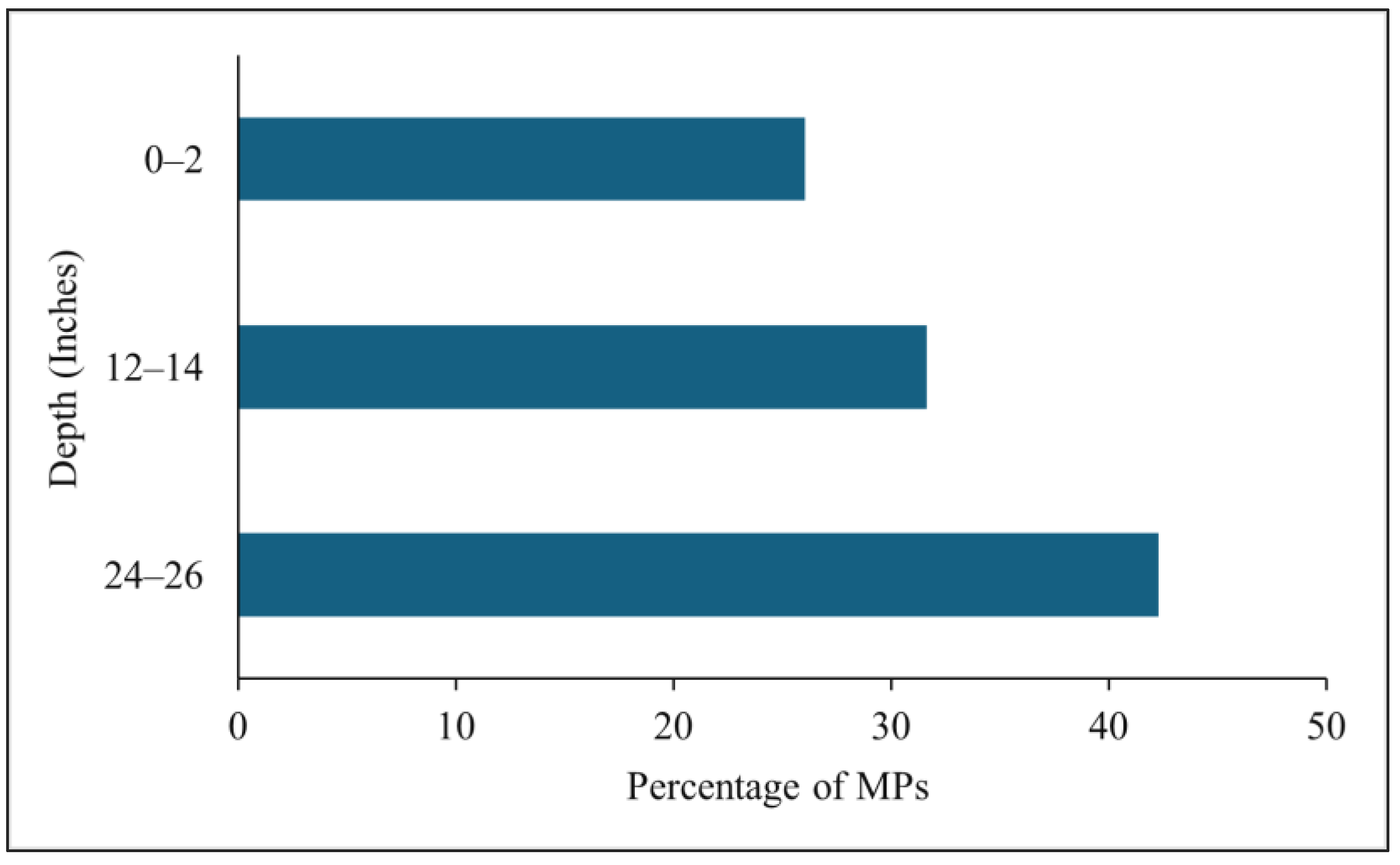
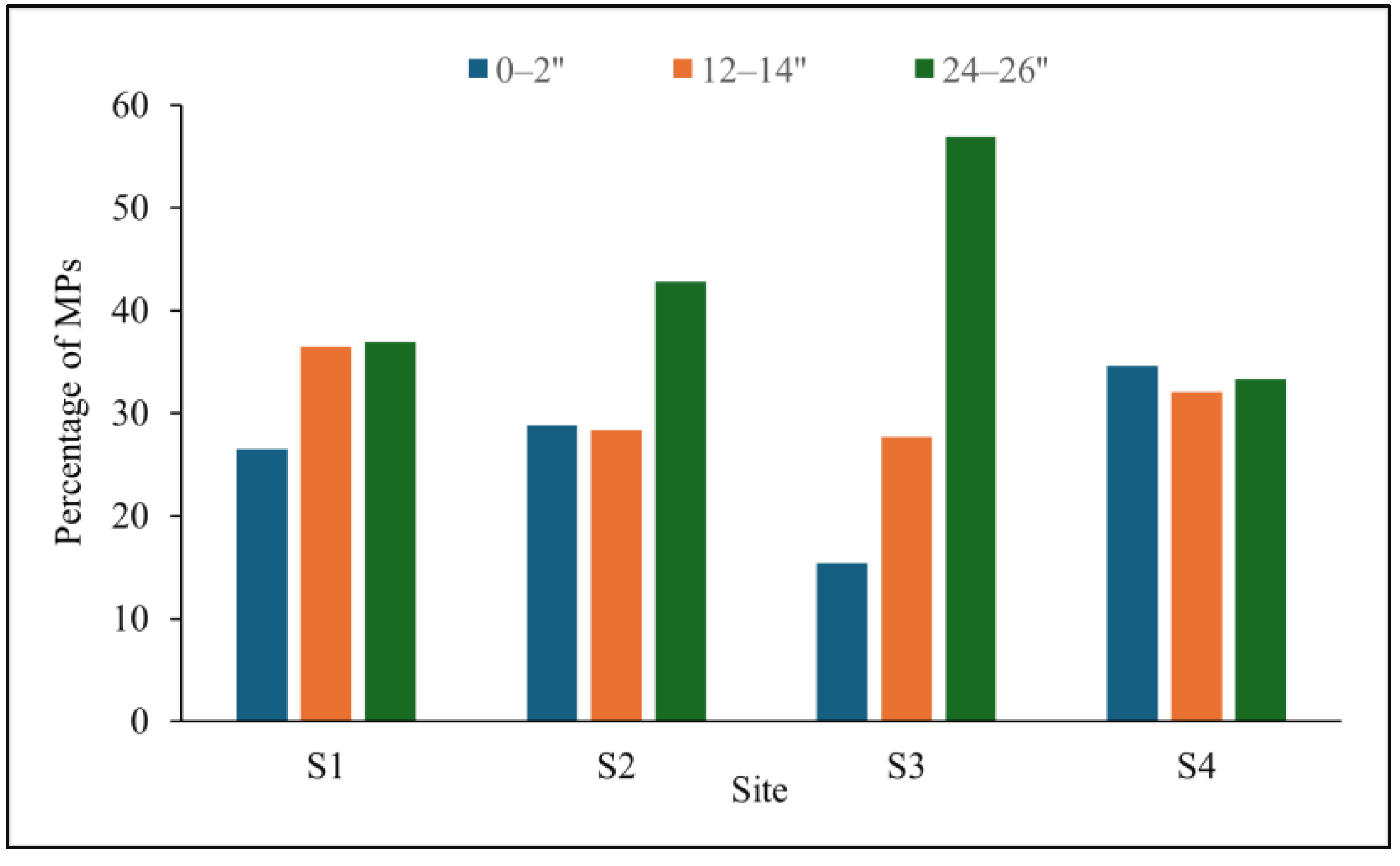
3.3. Vertical Distribution of MPs in BRS Media
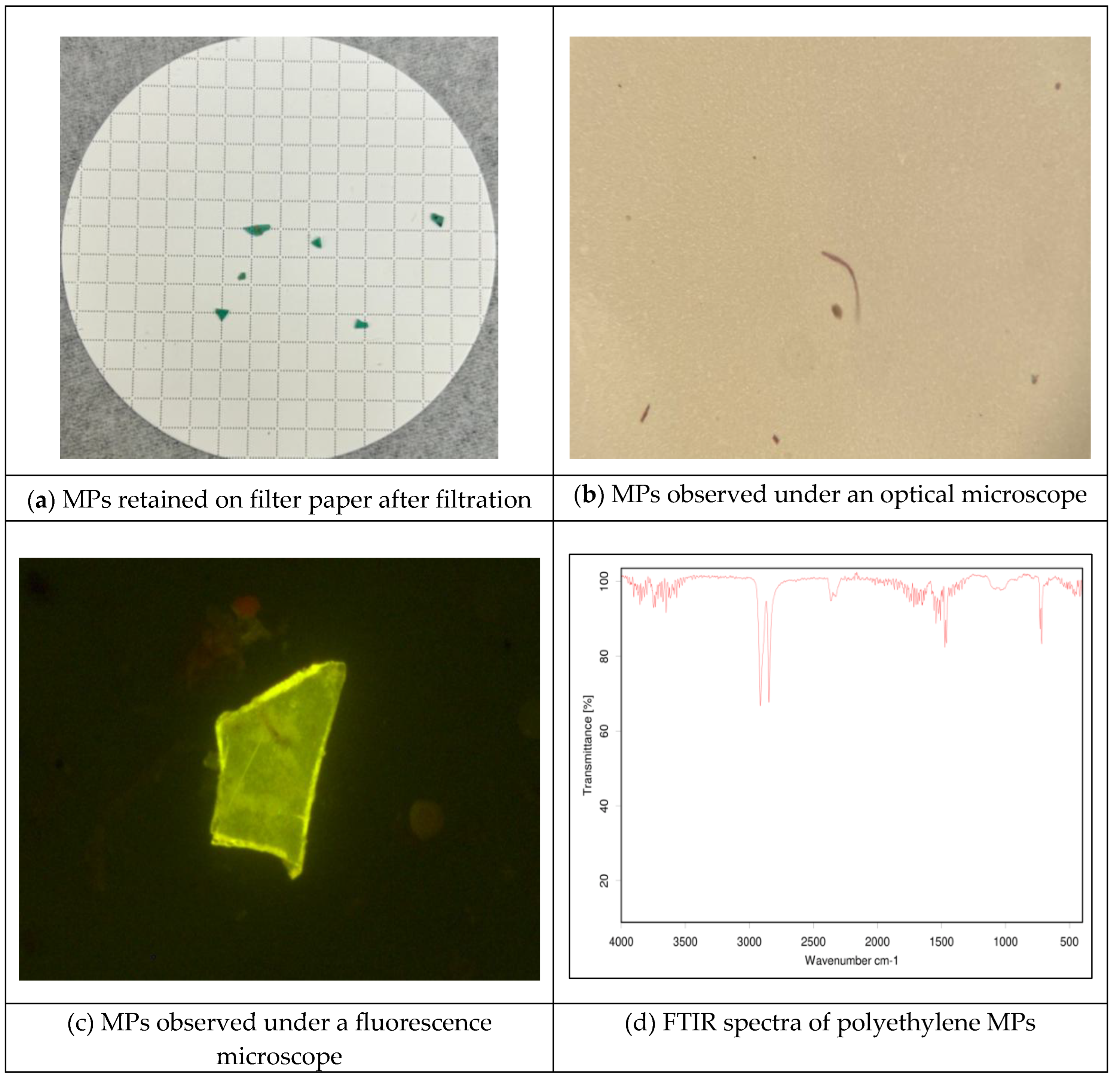
4. Discussion
4.1. Occurrence and Vertical Distribution of MPs in BRS Media
4.2. Field-Installed BRS Media for Trapping MPs from Urban Stormwater
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| MP | Microplastic |
| MPs | Microplastics |
| BRS | Bioretention Systems |
| US | United States |
| USEPA | United States Environmental Protection Agency |
| UTC | The University of Tennessee at Chattanooga |
| ASTM | American Standard Testing and Materials |
| NOAA | National Oceanic and Atmospheric Administration |
| NaCl | Sodium Chloride |
| Fe (II) | Aqueous Ferrous Solution |
| H2O2 | Hydrogen Peroxide |
| WPO | Wet Peroxide Oxidation |
| FTIR | Fourier Transform Infra-red |
| NR | Nile Red |
| ANOVA | Analysis of Variance |
| HSD | Honestly Significant Difference |
| SCMs | Stormwater Control Measures |
| TDEC | Tennessee Department of Environment and Conservation |
| MPCA | Minnesota Pollution Control Agency (MPCA) |
| USDA | United States Department of Agriculture |
| NCDEQ | North Carolina Department of Environmental Quality |
| PP | Polypropylene |
| PES | Polyester |
| PE | Polyethylene |
References
- Bhatt, V.; Chauhan, J. S. Microplastic in freshwater ecosystem: bioaccumulation, trophic transfer, and biomagnification. Environ. Sci. Pollut. Res. 2023, Vol. 30, 9389–9400. [Google Scholar] [CrossRef]
- Haque, A.; Holsen, T. M.; Baki, A. B. Distribution and risk assessment of microplastic pollution in a rural river system near a wastewater treatment plant, hydro-dam, and river confluence. Sci. Rep. 2024, Vol. 14, pp. 6006. [Google Scholar] [CrossRef]
- Nisha, N.; Rafizul, I. A CASE STUDY OF AN EDUCATIONAL INSTITUTION INVESTIGATING THE DETERMINANTS OF PEOPLE’S PERCEPTION OF PLASTIC CONSUMPTION.
- Das, T.; Das, N.; Zuthi, M. F. R.; Pal, S. K.; Kraft, E.; Haupt, T.; Kuehlewindt, S. Plastic Waste in Marine Ecosystems: Identification Techniques and Policy Interventions. Water Air Soil Pollut. 2025, Vol. 236, pp. 478. [Google Scholar] [CrossRef]
- Chanda, M.; Bathi, J. R. Transport and Retention of Polyethylene Microplastics in Saturated Porous Media: Effect of Physicochemical Properties. Environmental Pollution and Management 2025, 5. [Google Scholar] [CrossRef]
- Narayanan, M. Origination, fate, accumulation, impact, of microplastics in a marine ecosystem and bio/technological approach for remediation: a review. Process Saf. Environ. Prot 2023, 6. [Google Scholar] [CrossRef]
- Chanda, M.; Bathi, J. R.; Khan, E.; Katyal, D.; Danquah, M. Microplastics in ecosystems: Critical review of occurrence, distribution, toxicity, fate, transport, and advances in experimental and computational studies in surface and subsurface water. J. Environ. Manage. 2024, Vol. 370, pp. 122492. [Google Scholar] [CrossRef]
- Tasnim, N.; Das, N.; Khan, F.; Zuthi, M.; Masum, M. M.; Das, T. ADDRESSING THE CHALLENGES IN TRANSITIONING AWAY FROM PLASTIC: INSIGHTS FROM OUTREACH EVENTS AND WORKSHOPS. In Proceedings of the 7th International Conference on Advances in Civil Engineering (ICACE), Chattogram, Bangladesh, 2024. [Google Scholar]
- Mbachu, O.; Kaparaju, P.; Pratt, C. Plastic pollution risks in bioretention systems: a case study. Environ. Technol. 2023, Vol. 44, 2525–2538. [Google Scholar] [CrossRef] [PubMed]
- Chanda, M. Microplastics in Urban Environments: Distribution, Fate, and Transport. 2025, 10. [Google Scholar]
- Järlskog, I.; Strömvall, A.-M.; Magnusson, K.; Gustafsson, M.; Polukarova, M.; Galfi, H.; Aronsson, M.; Andersson-Sköld, Y. Occurrence of tire and bitumen wear microplastics on urban streets and in sweepsand and washwater. Sci. Total Environ. 2020, Vol. 729, pp. 138950. [Google Scholar] [CrossRef]
- Österlund, H.; Blecken, G.; Lange, K.; Marsalek, J.; Gopinath, K.; Viklander, M. Microplastics in urban catchments: Review of sources, pathways, and entry into stormwater. Sci. Total Environ. 2023, Vol. 858, pp. 159781. [Google Scholar] [CrossRef]
- Shruti, V.; Pérez-Guevara, F.; Elizalde-Martínez, I.; Kutralam-Muniasamy, G. Current trends and analytical methods for evaluation of microplastics in stormwater. Trends Environ. Anal. Chem. 2021, Vol. 30, pp. e00123. [Google Scholar] [CrossRef]
- Wang, C.; O'Connor, D.; Wang, L.; Wu, W.-M.; Luo, J.; Hou, D. Microplastics in urban runoff: Global occurrence and fate. Water Res. 2022, Vol. 225, pp. 119129. [Google Scholar] [CrossRef] [PubMed]
- de Jesus Piñon-Colin, T.; Rodriguez-Jimenez, R.; Rogel-Hernandez, E.; Alvarez-Andrade, A.; Wakida, F. T. Microplastics in stormwater runoff in a semiarid region, Tijuana, Mexico. Sci. Total Environ. 2020, Vol. 704, pp. 135411. [Google Scholar] [CrossRef]
- Werbowski, L. M.; Gilbreath, A. N.; Munno, K.; Zhu, X.; Grbic, J.; Wu, T.; Sutton, R.; Sedlak, M. D.; Deshpande, A. D.; Rochman, C. M. Urban stormwater runoff: a major pathway for anthropogenic particles, black rubbery fragments, and other types of microplastics to urban receiving waters. ACS ES&T Water 2021, Vol. 1, 1420–1428. [Google Scholar] [CrossRef]
- Ahmad, T.; Gul, S.; Peng, L.; Mehmood, T.; Huang, Q.; Ahmad, A.; Ali, H.; Ali, W.; Souissi, S.; Zinck, P. Microplastic mitigation in urban stormwater using green infrastructure: a review. Environ. Chem. Lett. 2025, 1–26. [Google Scholar] [CrossRef]
- Sulistyowati, L.; Riani, E.; Cordova, M. R. The occurrence and abundance of microplastics in surface water of the midstream and downstream of the Cisadane River, Indonesia. Chemosphere 2022, Vol. 291, pp. 133071. [Google Scholar] [CrossRef]
- Stang, C.; Mohamed, B. A.; Li, L. Y. Microplastic removal from urban stormwater: Current treatments and research gaps. J. Environ. Manage. 2022, Vol. 317, pp. 115510. [Google Scholar] [CrossRef] [PubMed]
- Roy-Poirier, A.; Champagne, P.; Filion, Y. Review of bioretention system research and design: past, present, and future. J. Environ. Eng. 2010, Vol. 136, 878–889. [Google Scholar] [CrossRef]
- Davis, A. P.; Hunt, W. F.; Traver, R. G.; Clar, M. Bioretention technology: Overview of current practice and future needs. J. Environ. Eng. 2009, Vol. 135, 109–117. [Google Scholar] [CrossRef]
- Jiang, C.; Li, J.; Li, H.; Li, Y. An improved approach to design bioretention system media. Ecol. Eng. 2019, Vol. 136, 125–133. [Google Scholar] [CrossRef]
- Laurenson, G.; Laurenson, S.; Bolan, N.; Beecham, S.; Clark, I. The role of bioretention systems in the treatment of stormwater. Adv. Agron. 2013, Vol. 120, 223–274. [Google Scholar]
- Spraakman, S.; Van Seters, T.; Drake, J.; Passeport, E. How has it changed? A comparative field evaluation of bioretention infiltration and treatment performance post-construction and at maturity. Ecol. Eng. 2020, Vol. 158, pp. 106036. [Google Scholar] [CrossRef]
- Smyth, K.; Drake, J.; Li, Y.; Rochman, C.; Van Seters, T.; Passeport, E. Bioretention cells remove microplastics from urban stormwater. Water Res. 2021, Vol. 191, pp. 116785. [Google Scholar] [CrossRef]
- Lange, K.; Österlund, H.; Viklander, M.; Blecken, G.-T. Occurrence and concentration of 20–100 μm sized microplastic in highway runoff and its removal in a gross pollutant trap–Bioretention and sand filter stormwater treatment train. Sci. Total Environ. 2022, Vol. 809, pp. 151151. [Google Scholar] [CrossRef]
- Hathaway, J. M.; Brown, R. A.; Fu, J. S.; Hunt, W. F. Bioretention function under climate change scenarios in North Carolina, USA. J. Hydrol. 2014, Vol. 519, 503–511. [Google Scholar] [CrossRef]
- Lange, K.; Magnusson, K.; Viklander, M.; Blecken, G.-T. Removal of rubber, bitumen and other microplastic particles from stormwater by a gross pollutant trap-bioretention treatment train. Water Res. 2021, Vol. 202, pp. 117457. [Google Scholar] [CrossRef] [PubMed]
- USEPA. BIORETENTION DESIGN HANDBOOK Designing Holistic Bioretention for Performance and Longevity. USEPA, Washington, DC, 2023. [Google Scholar]
- Naylor, D.; McClure, R.; Jansson, J. Trends in microbial community composition and function by soil depth. Microorganisms 2022, Vol. 10, pp. 540. [Google Scholar] [CrossRef] [PubMed]
- Fierer, N.; Schimel, J. P.; Holden, P. A. Variations in microbial community composition through two soil depth profiles. Soil Biol. Biochem. 2003, Vol. 35, 167–176. [Google Scholar] [CrossRef]
- Gelsomino, A.; Azzellino, A. Multivariate analysis of soils: microbial biomass, metabolic activity, and bacterial-community structure and their relationships with soil depth and type. J. Plant Nutr. Soil Sci. 2011, Vol. 174, 381–394. [Google Scholar] [CrossRef]
- ASTM, I. Standard guide for collection, storage, characterization, and manipulation of sediments for toxicological testing. Annual Book of ASTM Standards; ASTM International: West Conshohocken, PA, 2014; Volume 11, pp. 79–85. [Google Scholar]
- Masura, J.; Baker, J. E.; Foster, G. D.; Arthur, C.; Herring, C. Laboratory methods for the analysis of microplastics in the marine environment: recommendations for quantifying synthetic particles in waters and sediments. 2015. [Google Scholar]
- Zhu, Y.; Li, Y.; Huang, J.; Zhang, Y.; Ho, Y. W.; Fang, J. K. H.; Lam, E. Y. Advanced optical imaging technologies for microplastics identification: progress and challenges. Adv. Photonics Res. 2024, Vol. 5, pp. 2400038. [Google Scholar] [CrossRef]
- Shruti, V.; Pérez-Guevara, F.; Roy, P. D.; Kutralam-Muniasamy, G. Analyzing microplastics with Nile Red: Emerging trends, challenges, and prospects. J. Hazard. Mater. 2022, Vol. 423, pp. 127171. [Google Scholar] [CrossRef]
- Sturm, M. T.; Korzin, A.; Ronsse, P.; Myers, E.; Zernikel, O.; Schober, D.; Schuhen, K. Reliable River Microplastic Monitoring Using Innovative Fluorescence Dyes—A Case Study. Microplastics 2025, Vol. 4, pp. 63. [Google Scholar] [CrossRef]
- Prasad, S.; Bennett, A.; Triantafyllou, M. Characterization of Nile red-stained microplastics through fluorescence spectroscopy. J. Mar. Sci. Eng. 2024, Vol. 12, pp. 1403. [Google Scholar] [CrossRef]
- Shim, W. J.; Song, Y. K.; Hong, S. H.; Jang, M. Identification and quantification of microplastics using Nile Red staining. Mar Pollut Bull 2016, Vol. 113, 469–476. [Google Scholar] [CrossRef]
- Shim, W. J.; Song, Y. K.; Hong, S. H.; Jang, M. Identification and quantification of microplastics using Nile Red staining. Mar. Pollut. Bull. 2016, Vol. 113, 469–476. [Google Scholar] [CrossRef]
- Szkudlarek, M.; Najbar, B.; Jankowiak, Ł. Variation in microplastic characteristics among amphibian larvae: a comparative study across different species and the influence of human activity. Sci. Rep. 2024, Vol. 14, pp. 13574. [Google Scholar] [CrossRef]
- Sheriff, I.; Awang, N. A.; Halim, H. B.; Ikechukwu, O. S.; Jusoh, A. F. Extraction and analytical methods of microplastics in wastewater treatment plants: Isolation patterns, quantification, and size characterization techniques. Desalination and Water Treatment 2024, Vol. 318, pp. 100399. [Google Scholar] [CrossRef]
- Yong, M. M. H.; Leistenschneider, C.; Miranda, J. A.; Paler, M. K.; Legaspi, C.; Germanov, E.; Araujo, G.; Burkhardt-Holm, P.; Erni-Cassola, G. Microplastics in fecal samples of whale sharks (Rhincodon typus) and from surface water in the Philippines. Microplast. Nanoplast. 2021, Vol. 1, pp. 17. [Google Scholar] [CrossRef] [PubMed]
- Vdovchenko, A.; Resmini, M. Mapping microplastics in humans: analysis of polymer types, and shapes in food and drinking water—a systematic review. Int. J. Mol. Sci. 2024, Vol. 25, pp. 7074. [Google Scholar] [CrossRef] [PubMed]
- Lozano, Y. M.; Lehnert, T.; Linck, L. T.; Lehmann, A.; Rillig, M. C. Microplastic shape, polymer type, and concentration affect soil properties and plant biomass. Front. Plant Sci. 2021, Vol. 12, pp. 616645. [Google Scholar] [CrossRef]
- Fiore, L.; Serranti, S.; Mazziotti, C.; Riccardi, E.; Benzi, M.; Bonifazi, G. Classification and distribution of freshwater microplastics along the Italian Po river by hyperspectral imaging. Environ. Sci. Pollut. Res. 2022, Vol. 29, 48588–48606. [Google Scholar] [CrossRef] [PubMed]
- Chanda, M.; Wright, L.; Manning-Berg, A.; Bhattacharyya, D.; Bathi, J. R. Microplastics pollution in urban freshwater sediments: A descriptive assessment of land-use categories. Sci. Total Environ. 2026, Vol. 1014, pp. 181351. [Google Scholar] [CrossRef]
- Lin, L.; Zuo, L. Z.; Peng, J. P.; Cai, L. Q.; Fok, L.; Yan, Y.; Li, H. X.; Xu, X. R. Occurrence and distribution of microplastics in an urban river: A case study in the Pearl River along Guangzhou City, China. Sci Total Environ 2018, Vol. 644, 375–381. [Google Scholar] [CrossRef]
- Witzig, C. S.; Földi, C.; Wörle, K.; Habermehl, P.; Pittroff, M.; Müller, Y. K.; Lauschke, T.; Fiener, P.; Dierkes, G.; Freier, K. P. When good intentions go bad—false positive microplastic detection caused by disposable gloves. Environ. Sci. Technol. 2020, Vol. 54, 12164–12172. [Google Scholar] [CrossRef]
- Sturm, M. T.; Myers, E.; Korzin, A.; Polierer, S.; Schober, D.; Schuhen, K. Fast Forward: Optimized Sample Preparation and Fluorescent Staining for Microplastic Detection. Microplastics 2023, Vol. 2, 334–349. [Google Scholar] [CrossRef]
- Koutnik, V. S.; Leonard, J.; Glasman, J.; Brar, J.; Koydemir, H. C.; Novoselov, A.; Bertel, R.; Tseng, D.; Ozcan, A.; Ravi, S. Microplastics retained in stormwater control measures: Where do they come from and where do they go? Water Res. 2022, Vol. 210, pp. 118008. [Google Scholar] [CrossRef]
- Banik, P.; Hossain, M. B.; Nur, A.-A. U.; Choudhury, T. R.; Liba, S. I.; Yu, J.; Noman, M. A.; Sun, J. Microplastics in sediment of Kuakata Beach, Bangladesh: occurrence, spatial distribution, and risk assessment. Front. Mar. Sci. 2022, Vol. 9, pp. 860989. [Google Scholar] [CrossRef]
- Xun, F.; Zhou, J.; Xiong, J.; Hu, T.; Xia, Q. Migration characteristics of polypropylene (PP) microplastics and effects on pollutants removal performance of bioretention cells. J. Environ. Chem. Eng. 2024, Vol. 12, pp. 113833. [Google Scholar] [CrossRef]
- MPCA, Design criteria for bioretention - Minnesota Stormwater Manual, Minnesota 2021.
- TDEC, Tennessee Permanent Stormwater Management and Design Guidance Manual, Tennessee. 2014.
- Waagen, E. N. Quantifying Pollutant Removal Rates of Bioretention Basins as a Stormwater Best Management Practice; Master of Science, Civil & Environmental Engineering, Old Dominion University, ODU Digital Commons, 2017. [Google Scholar]
- NCDEQ, Bioretention Cell, North Carolina Department of Environmental Quality Stormwater Design Manual, North Carolina. 2018.
- Kuoppamäki, K.; Pflugmacher Lima, S.; Scopetani, C.; Setälä, H. The ability of selected filter materials in removing nutrients, metals, and microplastics from stormwater in biofilter structures; Wiley Online Library, 2021; pp. 0047–2425. [Google Scholar]
- Koutnik, V. S.; Leonard, J.; Brar, J.; Cao, S.; Glasman, J. B.; Cowger, W.; Ravi, S.; Mohanty, S. K. Transport of microplastics in stormwater treatment systems under freeze-thaw cycles: Critical role of plastic density. Water Res. 2022, Vol. 222, pp. 118950. [Google Scholar] [CrossRef]
- Gunther, H. J.; Das, T. K.; Leonard, J.; Koutnik, V. S.; El Rassi, L. A.; Tang, Z.; Mohanty, S. K. UV exposure to PET microplastics increases their downward mobility in stormwater biofilters undergoing freeze–thaw cycles. Environ. Sci. Water Res. Technol. 2023, Vol. 9, 3136–3145. [Google Scholar] [CrossRef]
- O'Connor, D.; Pan, S.; Shen, Z.; Song, Y.; Jin, Y.; Wu, W.-M.; Hou, D. Microplastics undergo accelerated vertical migration in sand soil due to small size and wet-dry cycles. Environ. Pollut. 2019, Vol. 249, 527–534. [Google Scholar] [CrossRef]
- Li, M.; He, L.; Hsieh, L.; Rong, H.; Tong, M. Transport of plastic particles in natural porous media under freeze–thaw treatment: Effects of porous media property. J. Hazard. Mater. 2023, Vol. 442, pp. 130084. [Google Scholar] [CrossRef] [PubMed]
- Xun, F.; Zhou, J.; Xiong, J.; Hu, T.; Xia, Q. Migration characteristics of polypropylene (PP) microplastics and effects on pollutants removal performance of bioretention cells. Journal of Environmental Chemical Engineering 2024, pp. 113833. [Google Scholar] [CrossRef]
- Lange, K.; Furén, R.; Österlund, H.; Winston, R.; Tirpak, R. A.; Nordqvist, K.; Smith, J.; Dorsey, J.; Viklander, M.; Blecken, G.-T. Abundance, distribution, and composition of microplastics in the filter media of nine aged stormwater bioretention systems. Chemosphere 2023, Vol. 320, pp. 138103. [Google Scholar] [CrossRef] [PubMed]
- Smyth, K.; Tan, S.; Van Seters, T.; Gasperi, J.; Dris, R.; Drake, J.; Passeport, E. Small-Size Microplastics in Urban Stormwater Runoff are Efficiently Trapped in a Bioretention Cell. ACS ES&T Water 2024, Vol. 4, 2522–2531. [Google Scholar] [CrossRef]
- Ronda, A. C.; Menéndez, M. C.; Tombesi, N.; Álvarez, M.; Tomba, J. P.; Silva, L. I.; Arias, A. H. Microplastic levels on sandy beaches: are the effects of tourism and coastal recreation really important? Chemosphere 2023, Vol. 316, pp. 137842. [Google Scholar] [CrossRef]
- Sun, H.; Zhou, S.; Jiang, Y.; Qin, Z.; Fei, J.; Sun, Y.; Wang, J.; Yin, X. Effect of cationic, anionic and non-ionic surfactants on transport of microplastics: role of adhesion of surfactants on the polyethylene surface. J. Hydrol. 2022, Vol. 612, pp. 128051. [Google Scholar] [CrossRef]
- Waldschläger, K.; Schüttrumpf, H. Infiltration behavior of microplastic particles with different densities, sizes, and shapes—from glass spheres to natural sediments. Environmental science & technology 2020, Vol. 54, 9366–9373. [Google Scholar]
- Fan, Y.; Zheng, K.; Zhu, Z.; Chen, G.; Peng, X. Distribution, sedimentary record, and persistence of microplastics in the Pearl River catchment, China. Environmental Pollution 2019, Vol. 251, 862–870. [Google Scholar] [CrossRef]
- Hernandez, L. M.; Grant, J.; Fard, P. S.; Farner, J. M.; Tufenkji, N. Analysis of ultraviolet and thermal degradations of four common microplastics and evidence of nanoparticle release. Journal of Hazardous Materials Letters 2023, Vol. 4, pp. 100078. [Google Scholar] [CrossRef]
- González-Pleiter, M.; Tamayo-Belda, M.; Pulido-Reyes, G.; Amariei, G.; Leganés, F.; Rosal, R.; Fernández-Piñas, F. Secondary nanoplastics released from a biodegradable microplastic severely impact freshwater environments. In Environmental Science: Nano; 2019; Vol. 6, pp. 1382–1392. [Google Scholar]
- Vijayaraghavan, K.; Biswal, B. K.; Adam, M. G.; Soh, S. H.; Tsen-Tieng, D. L.; Davis, A. P.; Chew, S. H.; Tan, P. Y.; Babovic, V.; Balasubramanian, R. Bioretention systems for stormwater management: Recent advances and future prospects. J Environ Manage 2021, Vol. 292, pp. 112766. [Google Scholar] [CrossRef]
- Gilbreath, A.; McKee, L.; Shimabuku, I.; Lin, D.; Werbowski, L. M.; Zhu, X.; Grbic, J.; Rochman, C. Multiyear water quality performance and mass accumulation of PCBs, mercury, methylmercury, copper, and microplastics in a bioretention rain garden. J. Sustainable Water Built Environ. 2019, Vol. 5, pp. 04019004. [Google Scholar] [CrossRef]
- Järlskog, I.; Strömvall, A.-M.; Magnusson, K.; Galfi, H.; Björklund, K.; Polukarova, M.; Garção, R.; Markiewicz, A.; Aronsson, M.; Gustafsson, M. Traffic-related microplastic particles, metals, and organic pollutants in an urban area under reconstruction. Sci. Total Environ. 2021, Vol. 774, pp. 145503. [Google Scholar] [CrossRef]
- Johansson, G.; Fedje, K. K.; Modin, O.; Haeger-Eugensson, M.; Uhl, W.; Andersson-Sköld, Y.; Strömvall, A.-M. Removal and release of microplastics and other environmental pollutants during the start-up of bioretention filters treating stormwater. J. Hazard. Mater. 2024, Vol. 468, pp. 133532. [Google Scholar] [CrossRef] [PubMed]
- Järlskog, I.; Jaramillo-Vogel, D.; Rausch, J.; Gustafsson, M.; Strömvall, A.-M.; Andersson-Sköld, Y. Concentrations of tire wear microplastics and other traffic-derived non-exhaust particles in the road environment. Environ. Int. 2022, Vol. 170, pp. 107618. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, L. A.; Lykkemark, J.; Andersen, T. R.; Vollertsen, J. Permeable pavements: A possible sink for tyre wear particles and other microplastics? Sci. Total Environ. 2023, Vol. 869, pp. 161770. [Google Scholar] [CrossRef] [PubMed]
- Sturm, M. T.; Myers, E.; Schober, D.; Korzin, A.; Schuhen, K. Development of an inexpensive and comparable microplastic detection method using fluorescent staining with novel nile red derivatives. Analytica 2023, Vol. 4, 27–44. [Google Scholar] [CrossRef]
- Erni-Cassola, G.; Gibson, M. I.; Thompson, R. C.; Christie-Oleza, J. A. Lost, but found with Nile red: a novel method for detecting and quantifying small microplastics (1 mm to 20 μm) in environmental samples. Environ. Sci. Technol. 2017, Vol. 51, 13641–13648. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



