Submitted:
18 February 2026
Posted:
05 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
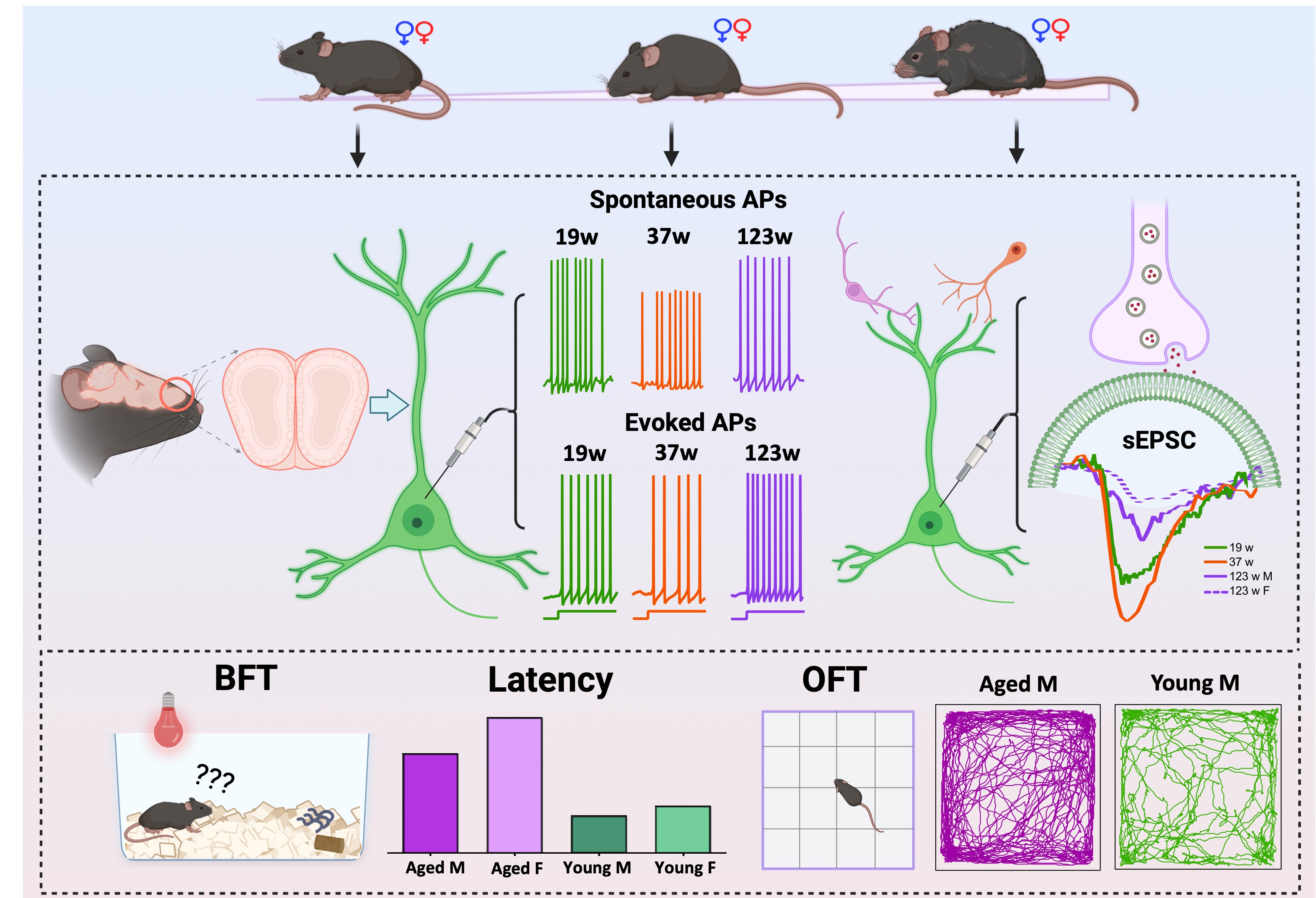
2.1. Animals
2.2. Behavioral tests
2.2.1. Open field test
2.2.2. Buried food and visible food test
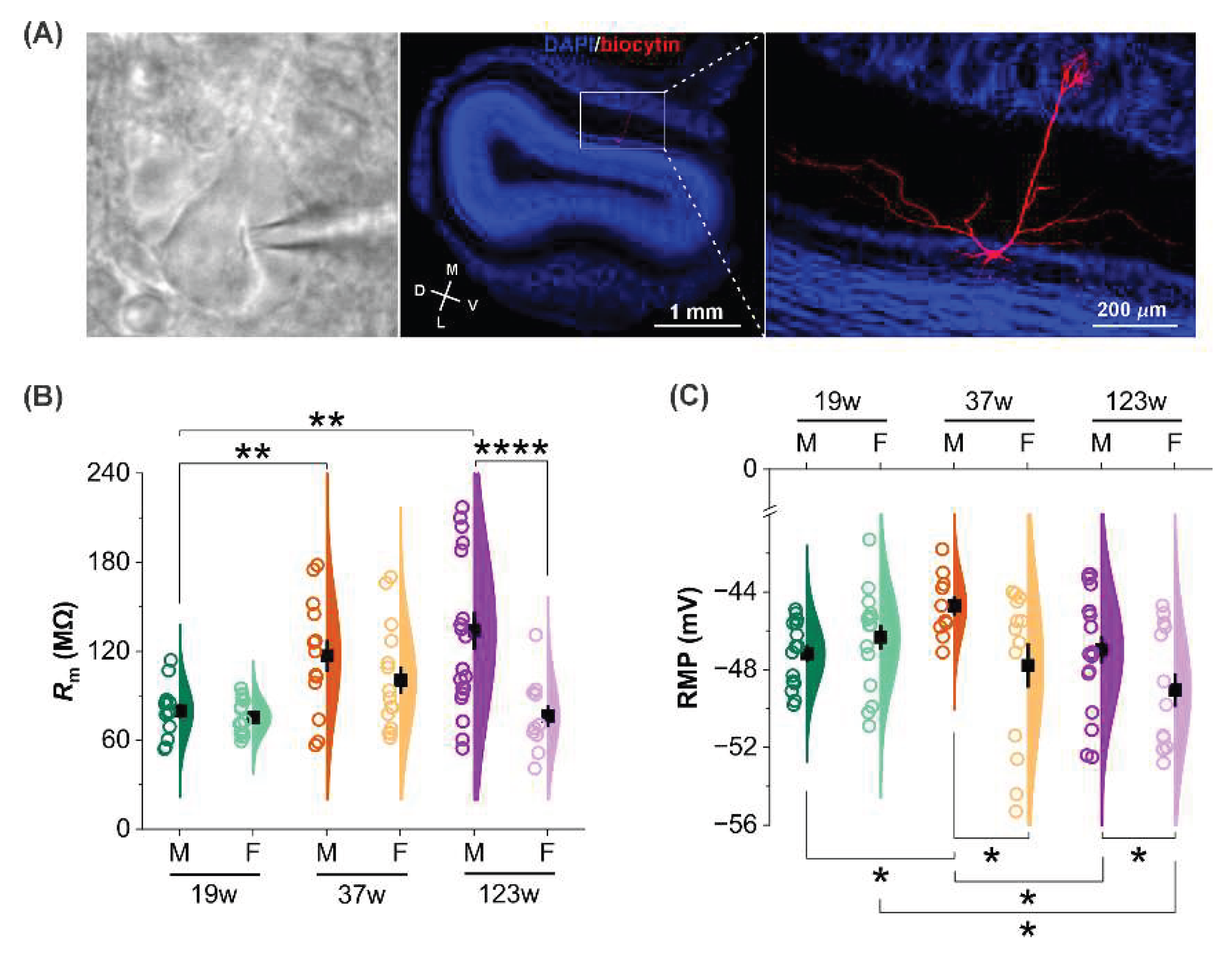
2.3. Slice Preparation
2.4. Electrophysiology
2.5. Immunohistochemical Staining
2.6. Data Analysis
3. Results
3.1. Age- and sex-dependent differences in olfactory behaviors
3.2. Age- and sex-dependent age effects on mitral cell membrane properties
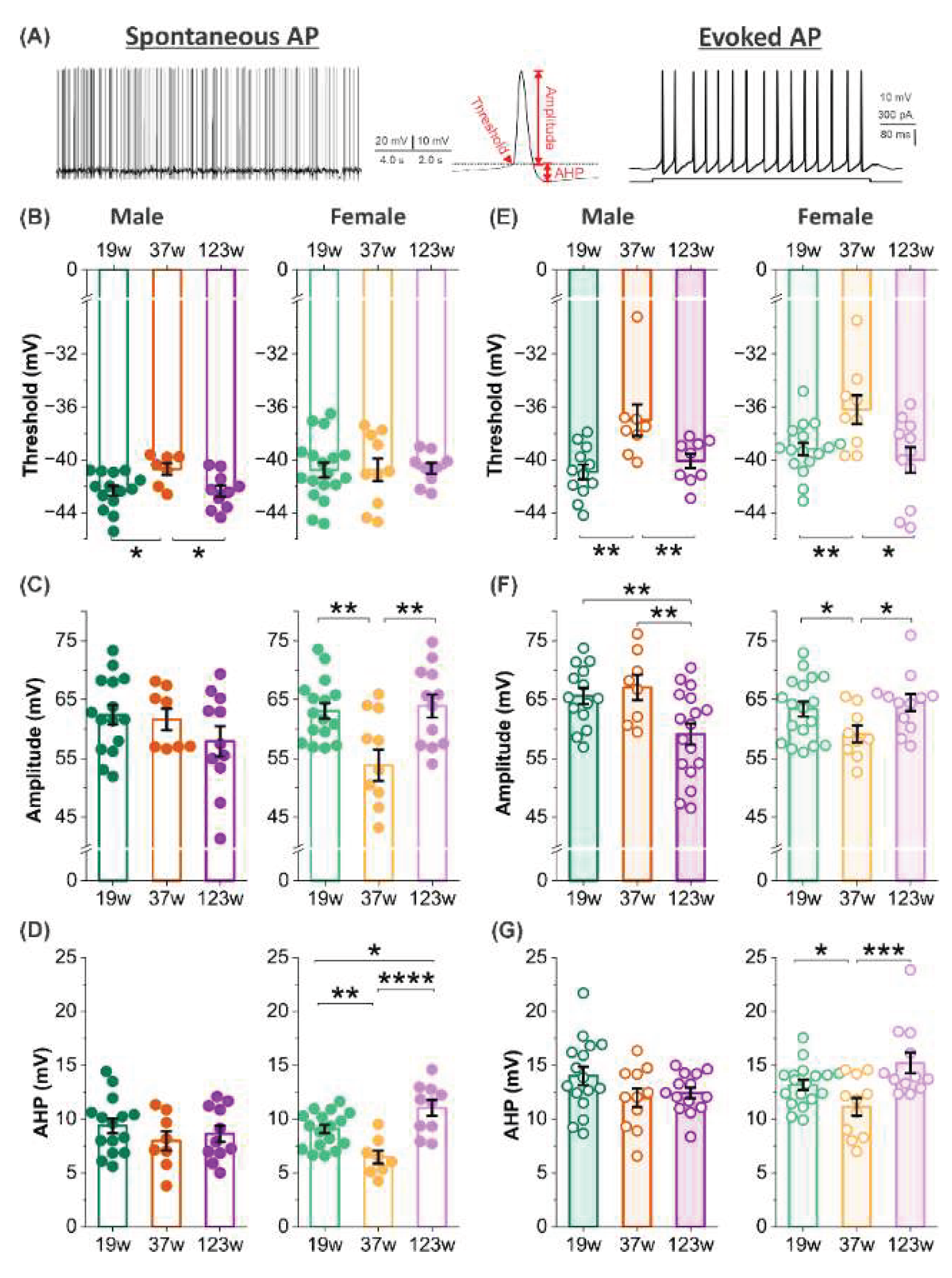
3.3. Sex-specific age effects on mitral cell action potential characteristics
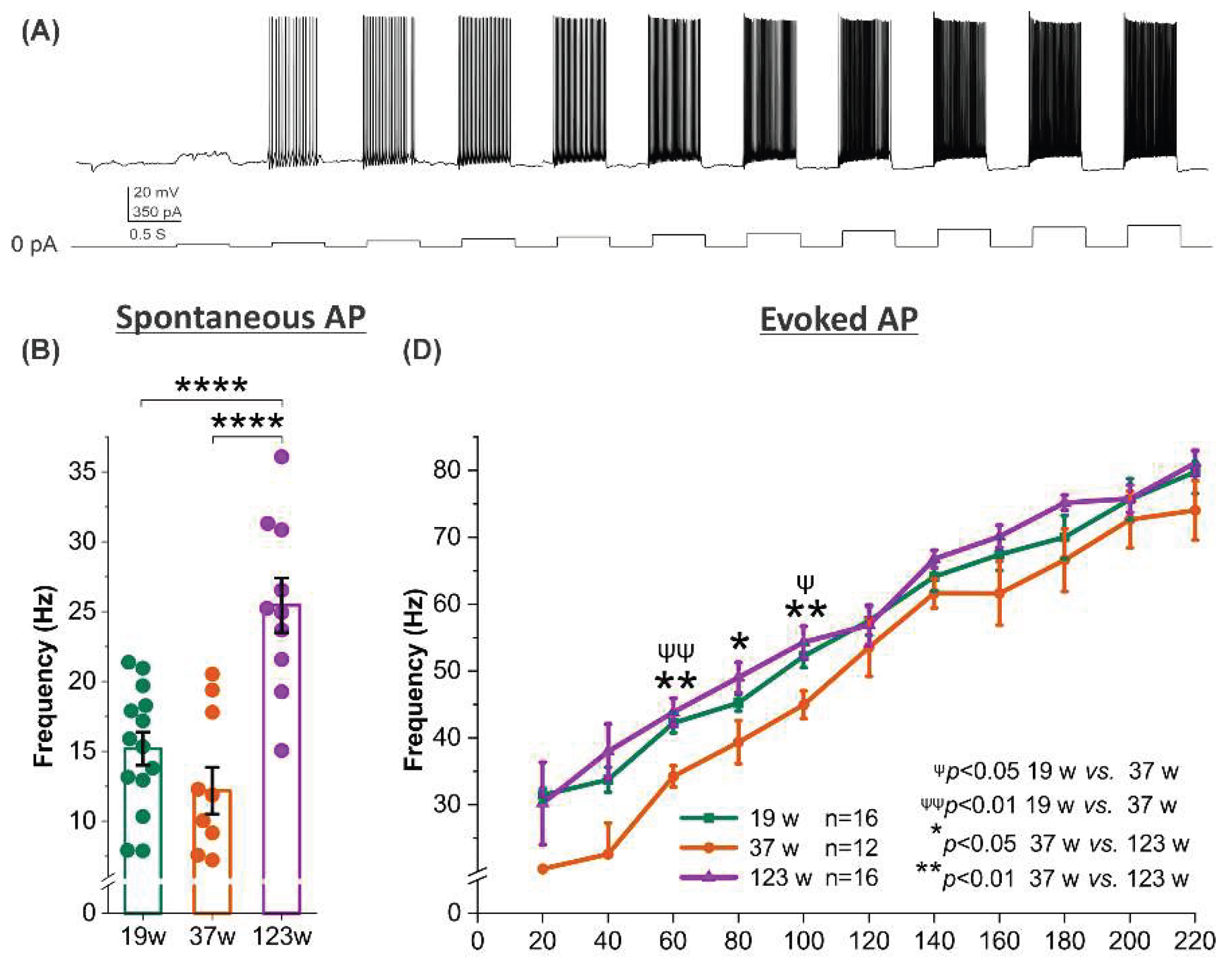
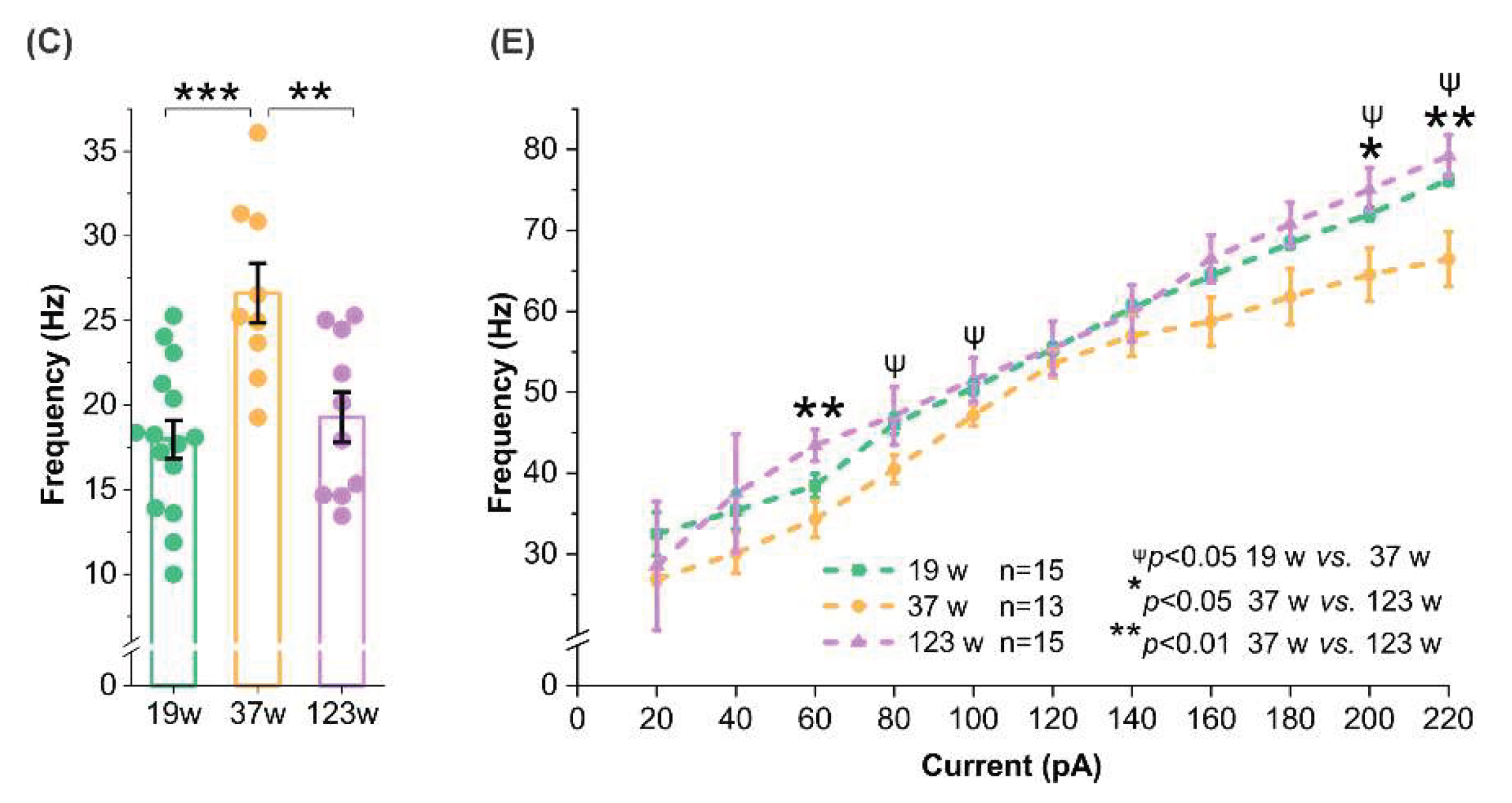
3.4. Opposing Age Effects on Spontaneous Firing in Male and Female Mitral Cells with Consistent Trends in Evoked Excitability

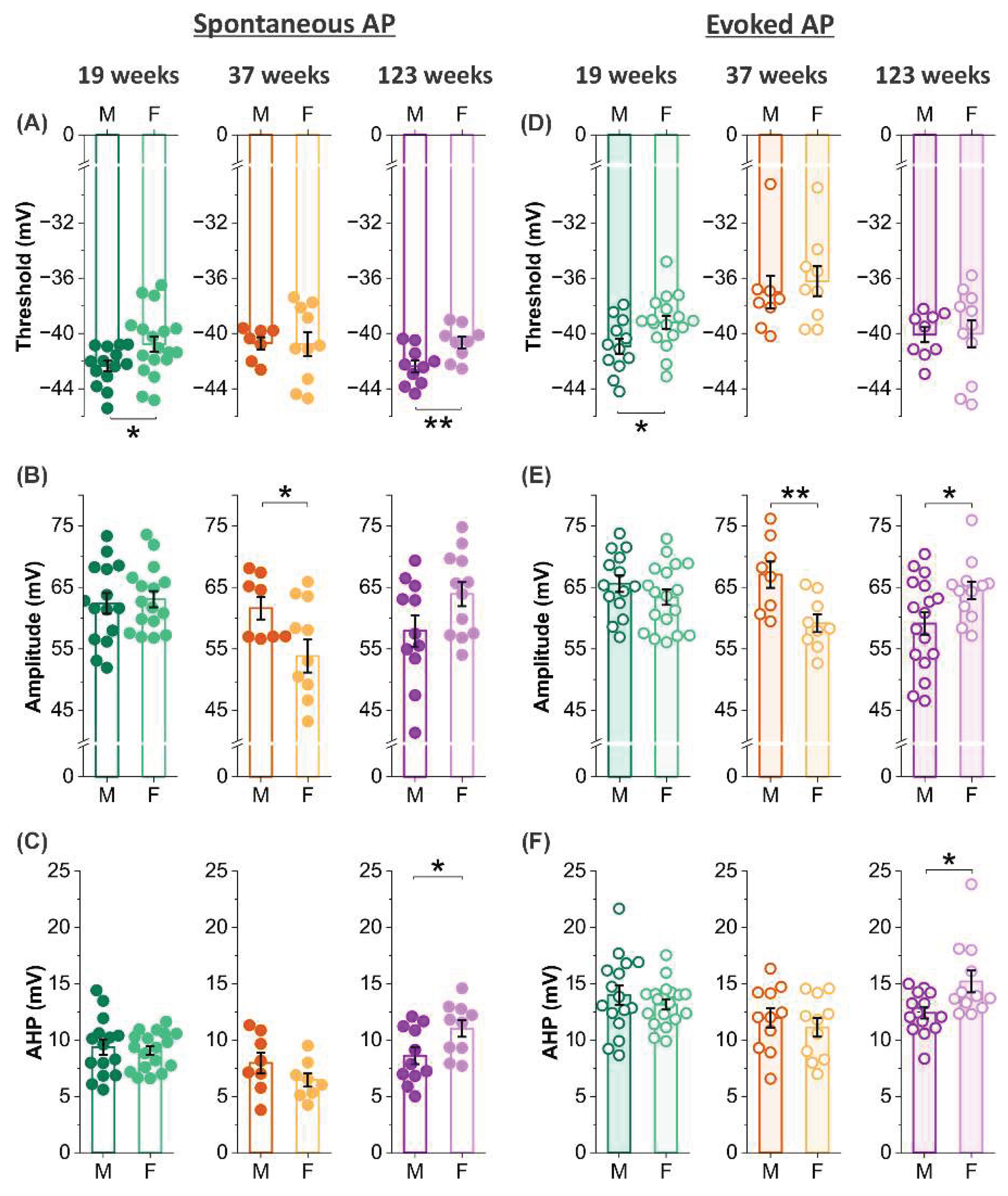
3.5. Sex effects on threshold, amplitude and afterhyperpolarization of action potentials
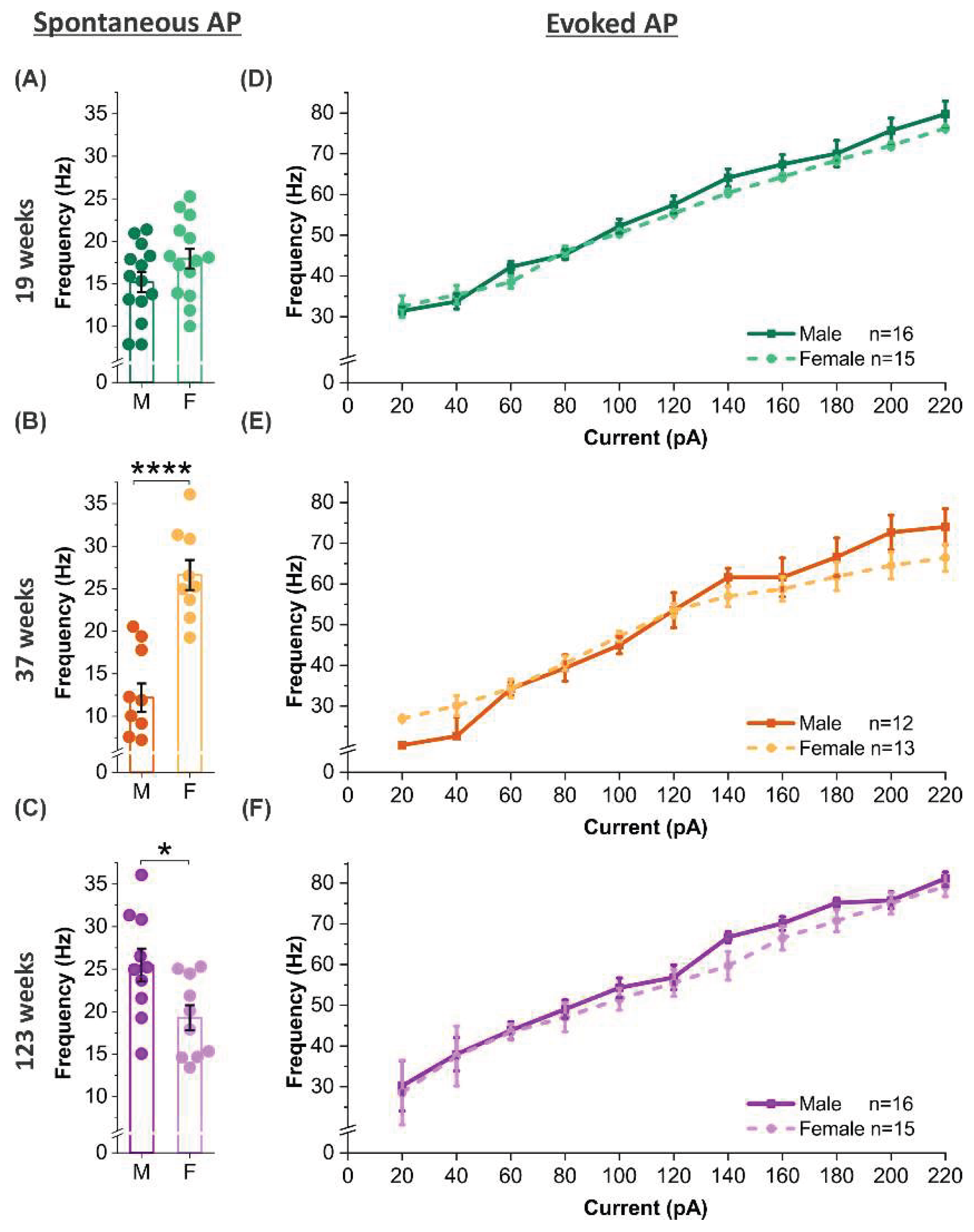
3.6. Age-dependent sex modulation of spontaneous but not evoked firing
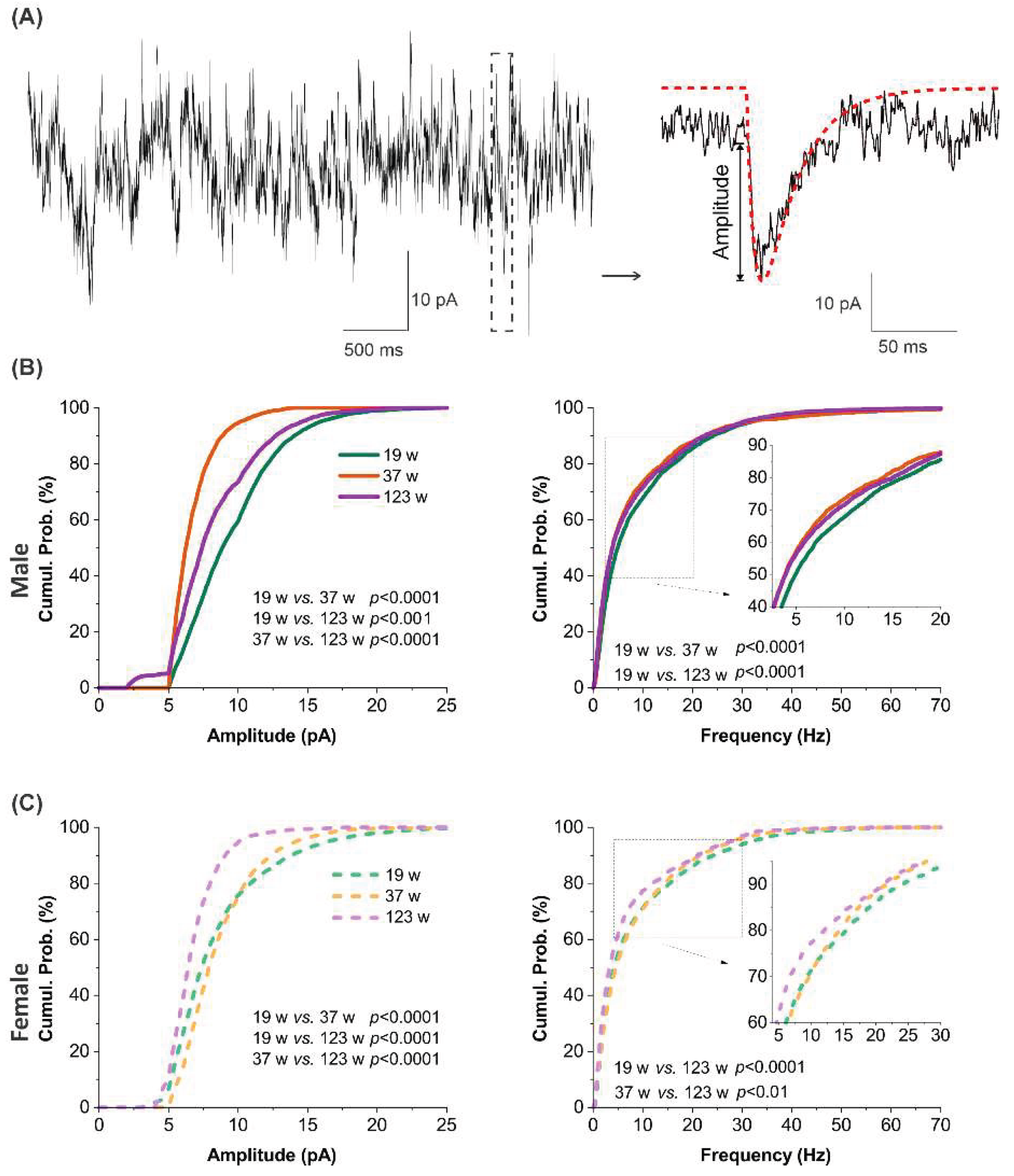
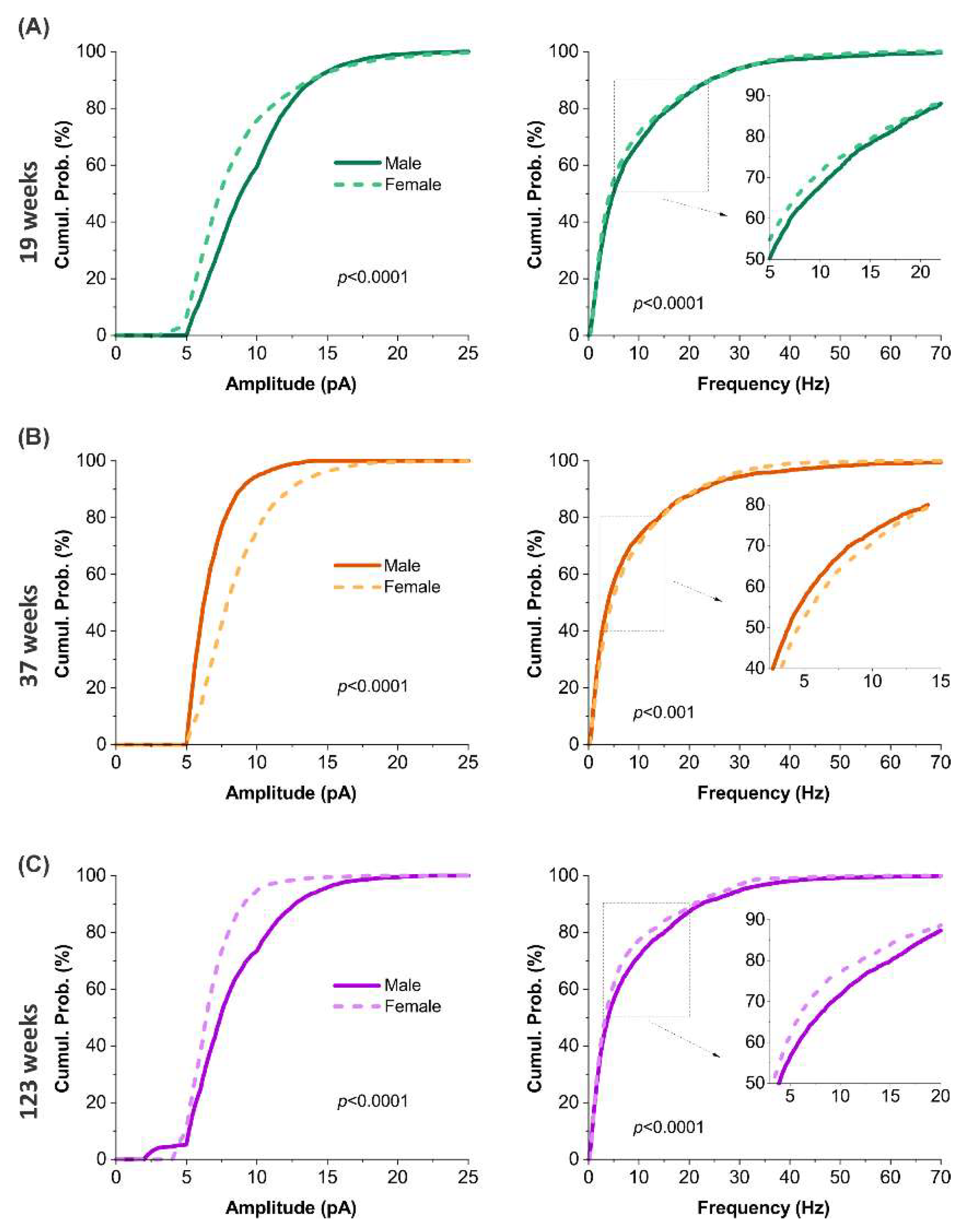
3.7. Sex-dependent age signatures in synaptic excitation
3.8. Sex-dependent alterations in excitatory synaptic input across ages
4. Discussion
5. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
References
- ACKELS, T.; JORDAN, R.; SCHAEFER, A. T.; FUKUNAGA, I. Respiration-locking of olfactory receptor and projection neurons in the mouse olfactory bulb and its modulation by brain state. Frontiers in cellular neuroscience 2020, 14, 220. [Google Scholar] [CrossRef]
- AHNAOU, A.; RODRIGUEZ-MANRIQUE, D.; EMBRECHTS, S.; BIERMANS, R.; MANYAKOV, N.; YOUSSEF, S.; DRINKENBURG, W. Aging alters olfactory bulb network oscillations and connectivity: relevance for aging-related neurodegeneration studies. Neural Plasticity 2020, 2020, 1703969. [Google Scholar] [CrossRef]
- ALOTAIBI, M. M.; DE MARCO, M.; VENNERI, A. Sex differences in olfactory cortex neuronal loss in aging. Frontiers in human neuroscience 2023, 17, 1130200. [Google Scholar] [CrossRef]
- ARNOLD, A. P.; LUSIS, A. J. Understanding the sexome: measuring and reporting sex differences in gene systems. Endocrinology 2012, 153, 2551–2555. [Google Scholar] [CrossRef]
- ARRUDA, D.; PUBLIO, R.; ROQUE, A. C. The periglomerular cell of the olfactory bulb and its role in controlling mitral cell spiking: a computational model. PLoS One 2013, 8, e56148. [Google Scholar] [CrossRef]
- ATTEMS, J.; WALKER, L.; JELLINGER, K. A. Olfaction and aging: a mini-review. Gerontology 2015, 61, 485–490. [Google Scholar] [CrossRef]
- BARZÓ, P.; SZOTS, I.; TÓTH, M.; CSAJBÓK, É.; MOLNÁR, G.; TAMÁS, G. Electrophysiology and morphology of human cortical supragranular pyramidal cells in a wide age range. bioRxiv Preprint 2025, 13, RP100390. [Google Scholar] [CrossRef]
- BEAM, C. R.; KANESHIRO, C.; JANG, J. Y.; REYNOLDS, C. A.; PEDERSEN, N. L.; GATZ, M. Differences between women and men in incidence rates of dementia and Alzheimer’s disease. Journal of Alzheimer’s disease 2018, 64, 1077–1083. [Google Scholar] [CrossRef] [PubMed]
- BOESVELDT, S.; POSTMA, E. M.; BOAK, D.; WELGE-LUESSEN, A.; SCHÖPF, V.; MAINLAND, J. D.; MARTENS, J.; NGAI, J.; DUFFY, V. B. Anosmia—a clinical review. Chemical senses 2017, 42, 513–523. [Google Scholar] [CrossRef]
- BONTEMPI, P.; RICATTI, M. J.; SANDRI, M.; NICOLATO, E.; MUCIGNAT-CARETTA, C.; ZANCANARO, C. Age-related in vivo structural changes in the male mouse olfactory bulb and their correlation with olfactory-driven behavior. Biology 2023, 12, 381. [Google Scholar] [CrossRef] [PubMed]
- BRINTON, R. D.; YAO, J.; YIN, F.; MACK, W. J.; CADENAS, E. Perimenopause as a neurological transition state. Nature reviews endocrinology 2015, 11, 393–405. [Google Scholar] [CrossRef]
- BRUNERT, D.; ROTHERMEL, M. Extrinsic neuromodulation in the rodent olfactory bulb. Cell and Tissue Research 2021, 383, 507–524. [Google Scholar] [CrossRef] [PubMed]
- CATTANEO, C.; PAGONABARRAGA, J. Sex differences in Parkinson’s disease: a narrative review. Neurology and Therapy 2025, 14, 57–70. [Google Scholar] [CrossRef]
- CONLEY, D. B.; ROBINSON, A. M.; SHINNERS, M. J.; KERN, R. C. Age-related olfactory dysfunction: cellular and molecular characterization in the rat. American journal of rhinology 2003, 17, 169–175. [Google Scholar] [CrossRef]
- CROY, I.; NORDIN, S.; HUMMEL, T. Olfactory Disorders and Quality of Life—An Updated Review. Chemical Senses 2014, 39, 185–194. [Google Scholar] [CrossRef]
- DAN, X.; YANG, B.; MCDEVITT, R. A.; GRAY, S.; CHU, X.; CLAYBOURNE, Q.; FIGUEROA, D. M.; ZHANG, Y.; CROTEAU, D. L.; BOHR, V. A. Loss of smelling is an early marker of aging and is associated with inflammation and DNA damage in C57BL/6J mice. Aging Cell 2023, 22, e13793. [Google Scholar] [CrossRef]
- DAVIS, E. J.; BROESTL, L.; ABDULAI-SAIKU, S.; WORDEN, K.; BONHAM, L. W.; MIÑONES-MOYANO, E.; MORENO, A. J.; WANG, D.; CHANG, K.; WILLIAMS, G. A second X chromosome contributes to resilience in a mouse model of Alzheimer’s disease. Science translational medicine 2020, 12, eaaz5677. [Google Scholar] [CrossRef]
- DEACON, R.; KOROS, E.; BORNEMANN, K.; RAWLINS, J. Aged Tg2576 mice are impaired on social memory and open field habituation tests. Behavioural brain research 2009, 197, 466–468. [Google Scholar] [CrossRef]
- DEJOU, J.; MANDAIRON, N.; DIDIER, A. Olfactory neurogenesis plays different parts at successive stages of life, implications for mental health. Frontiers in Neural Circuits 2024, 18, 1467203. [Google Scholar] [CrossRef] [PubMed]
- DÍAZ-GUERRA, E.; PIGNATELLI, J.; NIETO-ESTÉVEZ, V.; VICARIO-ABEJÓN, C. Transcriptional regulation of olfactory bulb neurogenesis. The Anatomical Record 2013, 296, 1364–1382. [Google Scholar] [CrossRef] [PubMed]
- DISTERHOFT, J. F.; WU, W. W.; OHNO, M. Biophysical alterations of hippocampal pyramidal neurons in learning, ageing and Alzheimer's disease. Ageing research reviews 2004, 3, 383–406. [Google Scholar] [CrossRef]
- DOTY, R. L.; CAMERON, E. L. Sex differences and reproductive hormone influences on human odor perception. Physiol Behav 2009, 97, 213–28. [Google Scholar] [CrossRef]
- DOTY, R. L.; KAMATH, V. The influences of age on olfaction: a review. Frontiers in psychology 2014, 5, 20. [Google Scholar] [CrossRef]
- DOTY, R. L.; SHAMAN, P.; APPLEBAUM, S. L.; GIBERSON, R.; SIKSORSKI, L.; ROSENBERG, L. Smell identification ability: changes with age. Science 1984, 226, 1441–3. [Google Scholar] [CrossRef]
- DUMÉNIEU, M.; FOURCAUD-TROCMÉ, N.; GARCIA, S.; KUCZEWSKI, N. Afterhyperpolarization (AHP) regulates the frequency and timing of action potentials in the mitral cells of the olfactory bulb: role of olfactory experience. Physiological reports 2015, 3, e12344. [Google Scholar] [CrossRef]
- EKANAYAKE, A.; PEIRIS, S.; AHMED, B.; KANEKAR, S.; GROVE, C.; KALRA, D.; ESLINGER, P.; YANG, Q.; KARUNANAYAKA, P. A review of the role of estrogens in olfaction, sleep and glymphatic functionality in relation to sex disparity in Alzheimer’s disease. American Journal of Alzheimer's Disease & Other Dementias® 2024, 39, 15333175241272025. [Google Scholar]
- FADOOL, D. A.; TUCKER, K.; PEDARZANI, P. Mitral cells of the olfactory bulb perform metabolic sensing and are disrupted by obesity at the level of the Kv1. 3 ion channel. PloS one 2011, 6, e24921. [Google Scholar] [CrossRef] [PubMed]
- FLURKEY, K.; CURRER, J.; HARRISON, D.; FOX, J. G. The mouse in biomedical research. In American College of Laboratory Animal Medicine series; Elsevier: AP: Amsterdam, 2007; pp. 637–672. [Google Scholar]
- GADIWALLA, S.; GUILLAUME, C.; HUANG, L.; WHITE, S. J. B.; BASHA, N.; PETERSEN, P. H.; GALLIANO, E. Ex Vivo Functional Characterization of Mouse Olfactory Bulb Projection Neurons Reveals a Heterogeneous Continuum. eNeuro 2025, 12. [Google Scholar] [CrossRef]
- GONZALES, M. M.; GARBARINO, V. R.; POLLET, E.; PALAVICINI, J. P.; KELLOGG, D. L.; KRAIG, E.; ORR, M. E. Biological aging processes underlying cognitive decline and neurodegenerative disease. The Journal of clinical investigation 2022, 132. [Google Scholar] [CrossRef] [PubMed]
- HIRATA, T. Olfactory information processing viewed through mitral and tufted cell-specific channels. Frontiers in Neural Circuits 2024, 18, 1382626. [Google Scholar] [CrossRef] [PubMed]
- KENSAKU, M. The Olfactory System: From Odor Molecules to Motivational Behaviors; Springer: Tokyo, 2014. [Google Scholar]
- KHRAMTSOVA, E. A.; DAVIS, L. K.; STRANGER, B. E. The role of sex in the genomics of human complex traits. Nature Reviews Genetics 2019, 20, 173–190. [Google Scholar] [CrossRef]
- KOLLING, L. J.; MARCINKIEWCZ, C. A.; FADOOL, D. A. The superficial tufted and mitral cell output neurons of the mouse olfactory bulb have a dual role in insulin sensing. bioRxiv 2025, 2025.04. 15, 648999. [Google Scholar] [CrossRef]
- KONDO, K.; KIKUTA, S.; UEHA, R.; SUZUKAWA, K.; YAMASOBA, T. Age-related olfactory dysfunction: epidemiology, pathophysiology, and clinical management. Frontiers in aging neuroscience 2020, 12, 208. [Google Scholar] [CrossRef]
- LEE, J.; WANG, Z. M.; MESSI, M. L.; MILLIGAN, C.; FURDUI, C. M.; DELBONO, O. Sex differences in single neuron function and proteomics profiles examined by patch-clamp and mass spectrometry in the locus coeruleus of the adult mouse. Acta Physiologica 2024, 240, e14123. [Google Scholar] [CrossRef] [PubMed]
- LLEDO, P.-M.; ALONSO, M.; GRUBB, M. S. Adult neurogenesis and functional plasticity in neuronal circuits. Nature Reviews Neuroscience 2006, 7, 179–193. [Google Scholar] [CrossRef] [PubMed]
- MACHADO, C. F.; REIS-SILVA, T. M.; LYRA, C. S.; FELICIO, L. F.; MALNIC, B. Buried Food-seeking Test for the Assessment of Olfactory Detection in Mice. Bio Protoc 2018, 8, e2897. [Google Scholar] [CrossRef]
- MEI, Z.; LIU, J.; BENNETT, D. A.; SEYFRIED, N.; WINGO, A. P.; WINGO, T. S. Unraveling sex differences in Alzheimer's disease and related endophenotypes with brain proteomes. Alzheimer's & Dementia 2025, 21, e70206. [Google Scholar] [CrossRef]
- MOBLEY, A. S.; RODRIGUEZ-GIL, D. J.; IMAMURA, F.; GREER, C. A. Aging in the olfactory system. Trends in neurosciences 2014, 37, 77–84. [Google Scholar] [CrossRef]
- MORI, K.; SAKANO, H. Olfactory circuitry and behavioral decisions. Annual review of physiology 2021, 83, 231–256. [Google Scholar] [CrossRef] [PubMed]
- NAGAYAMA, S.; HOMMA, R.; IMAMURA, F. Neuronal organization of olfactory bulb circuits. Frontiers in neural circuits 2014, 8, 98. [Google Scholar] [CrossRef]
- OLOFSSON, J. K.; EKSTRÖM, I.; LARSSON, M.; NORDIN, S. Olfaction and Aging: A Review of the Current State of Research and Future Directions. In PsyArXiv; 2020; p. 2. [Google Scholar]
- OLOFSSON, J. K.; EKSTRÖM, I.; LARSSON, M.; NORDIN, S. Olfaction and aging: A review of the current state of research and future directions. i-Perception 2021, 12, 20416695211020331. [Google Scholar] [CrossRef]
- PARK, J.; HO, R. L.; WANG, W.-E.; CHIU, S. Y.; SHIN, Y. S.; COOMBES, S. A. Age-related changes in neural oscillations vary as a function of brain region and frequency band. Frontiers in Aging Neuroscience 2025, 17, 1488811. [Google Scholar] [CrossRef]
- POWER, J. M.; WU, W. W.; SAMETSKY, E.; OH, M. M.; DISTERHOFT, J. F. Age-related enhancement of the slow outward calcium-activated potassium current in hippocampal CA1 pyramidal neurons in vitro. Journal of Neuroscience 2002, 22, 7234–7243. [Google Scholar] [CrossRef]
- RICHARD, M. B.; TAYLOR, S. R.; GREER, C. A. Age-induced disruption of selective olfactory bulb synaptic circuits. Proceedings of the National Academy of Sciences 2010, 107, 15613–15618. [Google Scholar] [CrossRef] [PubMed]
- ROSENTHAL, M.; VARELA, M.; GARCIA, A.; BRITTON, D. Age-related changes in the motor response to environmental novelty in the rat. Experimental gerontology 1989, 24, 149–157. [Google Scholar] [CrossRef]
- RUSSELL, J. K.; JONES, C. K.; NEWHOUSE, P. A. The role of estrogen in brain and cognitive aging. Neurotherapeutics 2019, 16, 649–665. [Google Scholar] [CrossRef] [PubMed]
- SCHAFFNER, S. L.; TOSEFSKY, K. N.; INSKTER, A. M.; APPEL-CRESSWELL, S.; SCHULZE-HENTRICH, J. M. Sex and gender differences in the molecular etiology of Parkinson’s disease: considerations for study design and data analysis. Biology of Sex Differences 2025, 16, 7. [Google Scholar] [CrossRef]
- SEIBENHENER, M. L.; WOOTEN, M. C. Use of the open field maze to measure locomotor and anxiety-like behavior in mice. Journal of visualized experiments: JoVE 2015, 52434. [Google Scholar]
- SMITH, S.; HOPP, S. C. The 5XFAD mouse model of Alzheimer’s disease displays age-dependent deficits in habituation to a novel environment. Aging Brain 2023, 3, 100078. [Google Scholar] [CrossRef]
- SOROKOWSKI, P.; KARWOWSKI, M.; MISIAK, M.; MARCZAK, M. K.; DZIEKAN, M.; HUMMEL, T.; SOROKOWSKA, A. Sex differences in human olfaction: a meta-analysis. Frontiers in psychology 2019, 10, 242. [Google Scholar] [CrossRef]
- STARR, E.; BUDHATHOKI, R.; GILHOOLY, D.; CASTILLO, L.; HU, M.; ZHAO, D.; LI, Y.; LIU, S. CCKergic Tufted Cells Regulate Odor Sensitivity by Controlling Mitral Cell Output in the Mouse Olfactory Bulb. Journal of Neuroscience 2025, 45. [Google Scholar] [CrossRef]
- TZENG, W.-Y.; FIGARELLA, K.; GARASCHUK, O. Olfactory impairment in men and mice related to aging and amyloid-induced pathology. Pflügers Archiv-European Journal of Physiology 2021, 473, 805–821. [Google Scholar] [CrossRef]
- VILLA, A.; VEGETO, E.; POLETTI, A.; MAGGI, A. Estrogens, neuroinflammation, and neurodegeneration. Endocrine reviews 2016, 37, 372–402. [Google Scholar] [CrossRef] [PubMed]
- VINOGRAD, A.; TASAKA, G.-I.; KREINES, L.; WEISS, Y.; MIZRAHI, A. The pre-synaptic landscape of mitral/tufted cells of the main olfactory bulb. Frontiers in Neuroanatomy 2019, 13, 58. [Google Scholar] [CrossRef]
- XU, L.; LIU, J.; WROBLEWSKI, K. E.; MCCLINTOCK, M. K.; PINTO, J. M. Odor sensitivity versus odor identification in older US adults: associations with cognition, age, gender, and race. Chemical Senses 2020, 45, 321–330. [Google Scholar] [CrossRef] [PubMed]
- YAMAGUCHI, M. Interneurons in the Olfactory Bulb: Roles in the Plasticity of Olfactory Information Processing. In The Olfactory System: From Odor Molecules to Motivational Behaviors.; MORI, K., Ed.; Springer Japan: Tokyo, 2014. [Google Scholar]
- YANAI, S.; ENDO, S. Functional aging in male C57BL/6J mice across the life-span: a systematic behavioral analysis of motor, emotional, and memory function to define an aging phenotype. Frontiers in aging neuroscience 2021, 13, 697621. [Google Scholar] [CrossRef]
- YANG, J.; PINTO, J. M. The epidemiology of olfactory disorders. Current otorhinolaryngology reports 2016, 4, 130–141. [Google Scholar] [CrossRef]
- ZHU, D.; MONTAGNE, A.; ZHAO, Z. Alzheimer’s pathogenic mechanisms and underlying sex difference. Cellular and Molecular Life Sciences 2021, 78, 4907–4920. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



