Submitted:
05 March 2026
Posted:
06 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
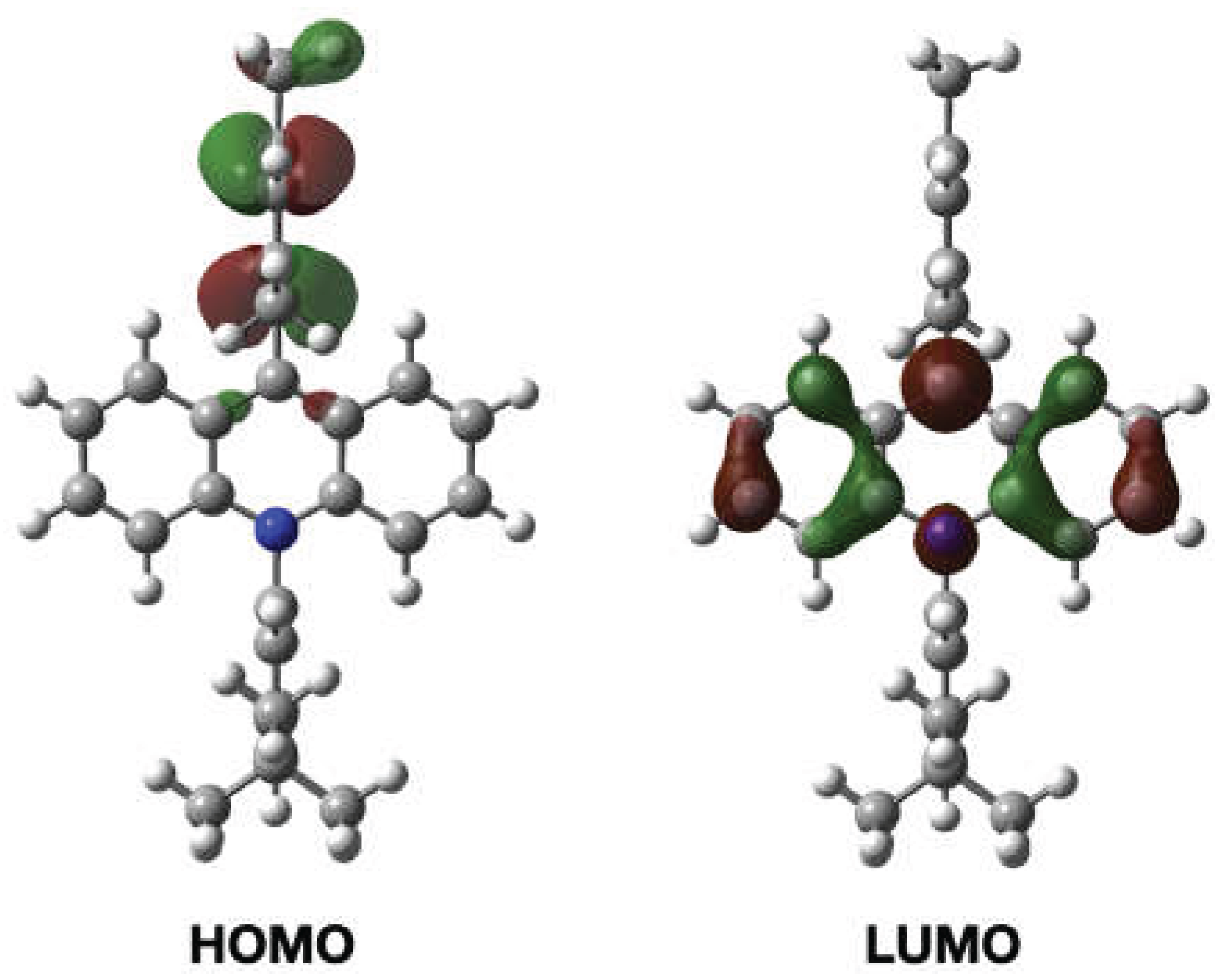
2. Results and Discussion
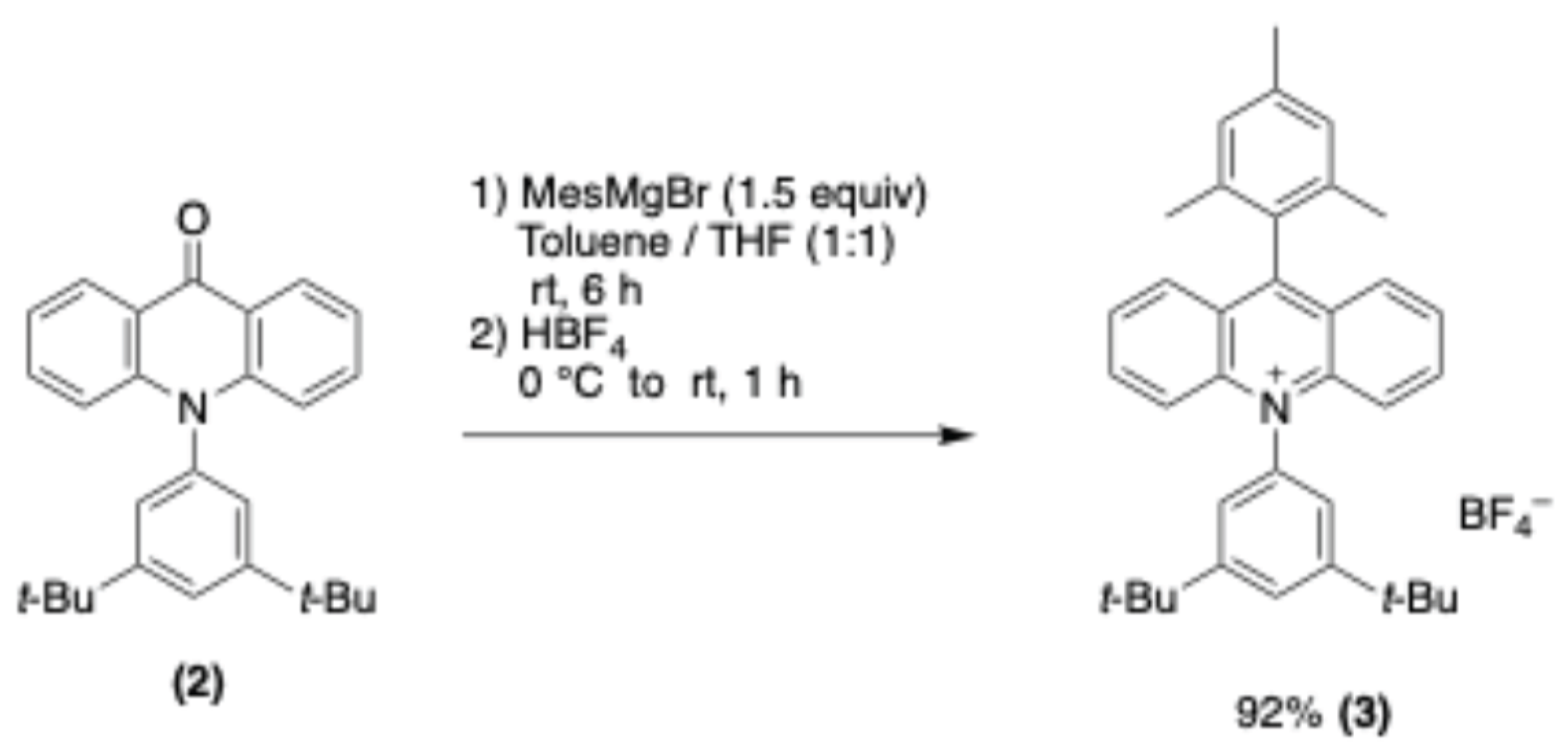
3. Materials and Methods
3.1. Materials
3.2. Synthetic Procedures
3.3. Calculations
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fukuzumi, S.; Ohkubo, K. Organic Synthetic Transformations using Organic Dyes as Photoredox Catalysts. Org. Biomol. Chem. 2014, 12, 6059–6071. [Google Scholar] [CrossRef] [PubMed]
- Fukuzumi, S.; Ohkubo, K. Selective Photocatalytic Reactions with Organic Photocatalysts. Chem. Sci. 2013, 4, 561–574. [Google Scholar] [CrossRef]
- Shaw, M.H.; Twilton, J.; MacMillan, D.W.C. Photoredox Catalysis in Organic Chemistry. J. Org. Chem. 2016, 81, 6898–6926. [Google Scholar] [CrossRef]
- Fukuzumi, S.; Lee, Y.-M.; Nam, W. Bioinspired artificial photosynthesis systems. Tetrahedron 2020, 76, 131024. [Google Scholar] [CrossRef]
- Lee, Y.-M.; Nam, W.; Fukuzumi, S. Redox catalysis via photoinduced electron transfer. Chem. Sci. 2023, 14, 4205–4218. [Google Scholar] [CrossRef] [PubMed]
- Yoshimi, Y. Organic Photoredox Reactions in Two-Molecule Photoredox System. Chem. Rec. 2024, 24, e202300326. [Google Scholar] [CrossRef] [PubMed]
- Fukuzumi, S.; Kuroda, S.; Tanaka, T. Selective Photoalkylation of 10-Methylacridinium Ion with Tetra-alkylstannanes or Diethylmercury Using Visible Irradiation. J. Chem. Soc., Chem. Commun. 1986, 1553–1554. [Google Scholar] [CrossRef]
- Fukuzumi, S.; Kuroda, S.; Tanaka, T. Substrate-Selective Photo-Oxidation of Benzyl Alcohol Derivatives with Oxygen, Catalysed by an NAD⁺ Model Compound. J. Chem. Soc., Chem. Commun. 1987, 120–122. [Google Scholar] [CrossRef]
- Fukuzumi, S.; Kitano, T.; Tanaka, T. Reduction of 10-Methylacridinium Ion with Fatty Acids by Photoinduced Electron-Transfer Reactions. Chem. Lett. 1989, 18, 1231–1234. [Google Scholar] [CrossRef]
- Fukuzumi, S.; Kitano, T.; Mochida, K. Photo-Induced One-Electron Reduction of 10-Methylacridinium Ion with Group 14 Dimetallic Compounds Using Visible Irradiation. J. Chem. Soc., Chem. Commun. 1990, 1236–1237. [Google Scholar] [CrossRef]
- Fukuzumi, S.; Yorisue, T. 10,10′-Dimethyl-9,9′-biacridine Acting as a Unique Electron Source Compared with the Corresponding Monomer for the Efficient Reduction of Dioxygen, Catalysed by a Cobalt Porphyrin in the Presence of Perchloric Acid. J. Chem. Soc., Perkin Trans. 2 1991, 1607–1611.
- Fukuzumi, S.; Kotani, H.; Ohkubo, K.; Ogo, S.; Tkachenko, N.V.; Lemmetyinen, H. Electron-Transfer State of 9-Mesityl-10-methylacridinium Ion with a Much Longer Lifetime and Higher Energy Than That of the Natural Photosynthetic Reaction Center. J. Am. Chem. Soc. 2004, 126, 1600–1601. [Google Scholar] [CrossRef] [PubMed]
- Hoshino, M.; Uekusa, H.; Tomita, A.; Koshihara, S.; Sato, T.; Nozawa, S.; Adachi, S.; Ohkubo, K.; Kotani, H.; Fukuzumi, S. Determination of the Structural Features of a Long-Lived Electron-Transfer State of 9-Mesityl-10-methylacridinium Ion. J. Am. Chem. Soc. 2012, 134, 4569–4572. [Google Scholar] [CrossRef]
- Ohkubo, K.; Fujimoto, A.; Fukuzumi, S. Metal-free Oxygenation of Cyclohexane with Oxygen Catalyzed by 9-Mesityl-10-methylacridinium and Hydrogen Chloride under Visible Light Irradiation. Chem. Commun. 2011, 47, 8515–8517. [Google Scholar] [CrossRef] [PubMed]
- Joshi-Pangu, A.; Lévesque, F.; Roth, H.G.; Oliver, S.F.; Campeau, L.-C.; Nicewicz, D.; DiRocco, D.A. Acridinium-Based Photocatalysts: A Sustainable Option in Photoredox Catalysis. J. Org. Chem. 2016, 81, 7244–7249. [Google Scholar] [CrossRef]
- White, A.R.; Wang, L.; Nicewicz, D.A. Synthesis and Characterization of Acridinium Dyes for Photoredox Catalysis. Synlett 2019, 30, 827–832. [Google Scholar] [CrossRef]
- Lakhdar, S. Acridinium Salts and Cyanoarenes as Powerful Photocatalysts: Opportunities in Organic Synthesis. Chem. Rev. 2021, 121, 19526–19549. [Google Scholar]
- Itabashi, Y.; Asahara, H.; Ohkubo, K. Chlorine-Radical-Mediated C–H Oxygenation Reaction under Light Irradiation. Chem. Commun. 2023, 59, 7506–7517. [Google Scholar] [CrossRef] [PubMed]
- Tanabe, S.; Mitsunuma, H.; Kanai, M. Catalytic Allylation of Aldehydes Using Unactivated Alkenes. J. Am. Chem. Soc. 2020, 142, 12374–12381. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, M.; Uchiyama, M.; Itabashi, Y.; Ohkubo, K.; Kamigaito, M. Acridinium Salts as Photoredox Organocatalysts for Photomediated Cationic RAFT and DT Polymerizations of Vinyl Ethers. Polym. Chem. 2022, 13, 1031–1039. [Google Scholar] [CrossRef]
- Ohkubo, K.; Matsumoto, S.; Asahara, H.; Fukuzumi, S. 9-(4-Halo-2,6-xylyl)-10-methylacridinium Ion as an Effective Photoredox Catalyst for Oxygenation and Trifluoromethylation of Toluene Derivatives. ACS Catal. 2024, 14, 2671–2684. [Google Scholar] [CrossRef]
- Ullmann, F. Ueber eine neue Bildungsweise von Diphenylaminderivaten. Ber. Dtsch. Chem. Ges. 1903, 36, 2382–2384. [Google Scholar] [CrossRef]
- Ullmann, F. Ueber eine neue Darstellungsweise von Phenyläthersalicylsäure. Ber. Dtsch. Chem. Ges. 1904, 37, 853–854. [Google Scholar] [CrossRef]
- Goldberg, I. Ueber Phenylirungen bei Gegenwart von Kupfer als Katalysator. Ber. Dtsch. Chem. Ges. 1906, 39, 1691–1692. [Google Scholar] [CrossRef]
- Ley, S. V.; Thomas, A. W. Modern Synthetic Methods for Copper-Mediated C(aryl)–O, C(aryl)–N, and C(aryl)–S Bond Formation. Angew. Chem. Int. Ed. 2003, 42, 5400–5449. [Google Scholar] [CrossRef] [PubMed]
- Kunz, K.; Scholz, U.; Ganzer, D. Renaissance of Ullmann and Goldberg Reactions—Progress in Copper-Catalyzed C–N-, C–O-, and C–S-Coupling. Synlett 2003, 15, 2428–2439. [Google Scholar] [CrossRef]
- Seifinoferest, B.; Tanbakouchian, A.; Larijani, B.; Mahdavi, M. Ullmann–Goldberg and Buchwald–Hartwig C–N Cross Couplings: Synthetic Methods to Pharmaceutically Potential N-Heterocycles. Asian J. Org. Chem. 2021, 10, 1319–1344. [Google Scholar] [CrossRef]
- Buck, E.; Song, Z. J.; Tschaen, D.; Dormer, P. G.; Volante, R. P.; Reider, P. J. Ullmann Diaryl Ether Synthesis: Rate Acceleration by 2,2,6,6-Tetramethylheptane-3,5-dione. Org. Lett. 2002, 4, 1623–1626. [Google Scholar] [CrossRef]
- Gaussian 16, Revision C.02, M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, G. A. Petersson, H. Nakatsuji, X. Li, M. Caricato, A. V. Marenich, J. Bloino, B. G. Janesko, R. Gomperts, B. Mennucci, H. P. Hratchian, J. V. Ortiz, A. F. Izmaylov, J. L. Sonnenberg, D. Williams-Young, F. Ding, F. Lipparini, F. Egidi, J. Goings, B. Peng, A. Petrone, T. Henderson, D. Ranasinghe, V. G. Zakrzewski, J. Gao, N. Rega, G. Zheng, W. Liang, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, K. Throssell, J. A. Montgomery, Jr., J. E. Peralta, F. Ogliaro, M. J. Bearpark, J. J. Heyd, E. N. Brothers, K. N. Kudin, V. N. Staroverov, T. A. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. P. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, J. M. Millam, M. Klene, C. Adamo, R. Cammi, J. W. Ochterski, R. L. Martin, K. Morokuma, O. Farkas, J. B. Foresman, D. J. Fox, Gaussian, Inc. Wallingford CT, 2019.
- Yanai, T.; Tew, D.; Handy, N. A new hybrid exchange-correlation functional using the Coulomb-attenuating method (CAM-B3LYP). Chem. Phys. Lett. 2004, 393, 51–57. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




