Submitted:
04 March 2026
Posted:
06 March 2026
You are already at the latest version
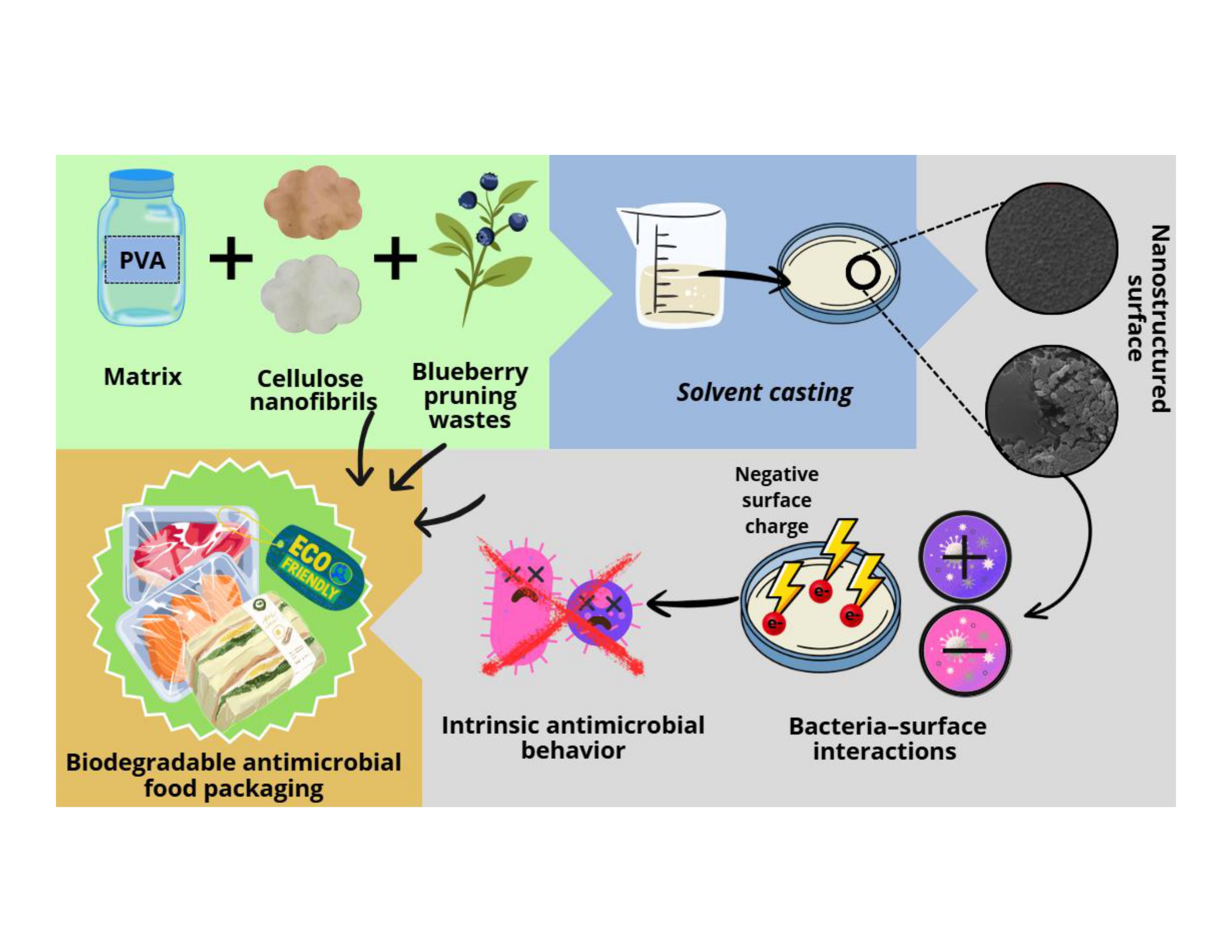
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Removable Extraction from Blueberry Pruning and Corn Husks (Modified TAPPI T204 cm-97)
2.2. CNFs Obtention
2.3. Preparation of PVA/CNFs Nanocomposites
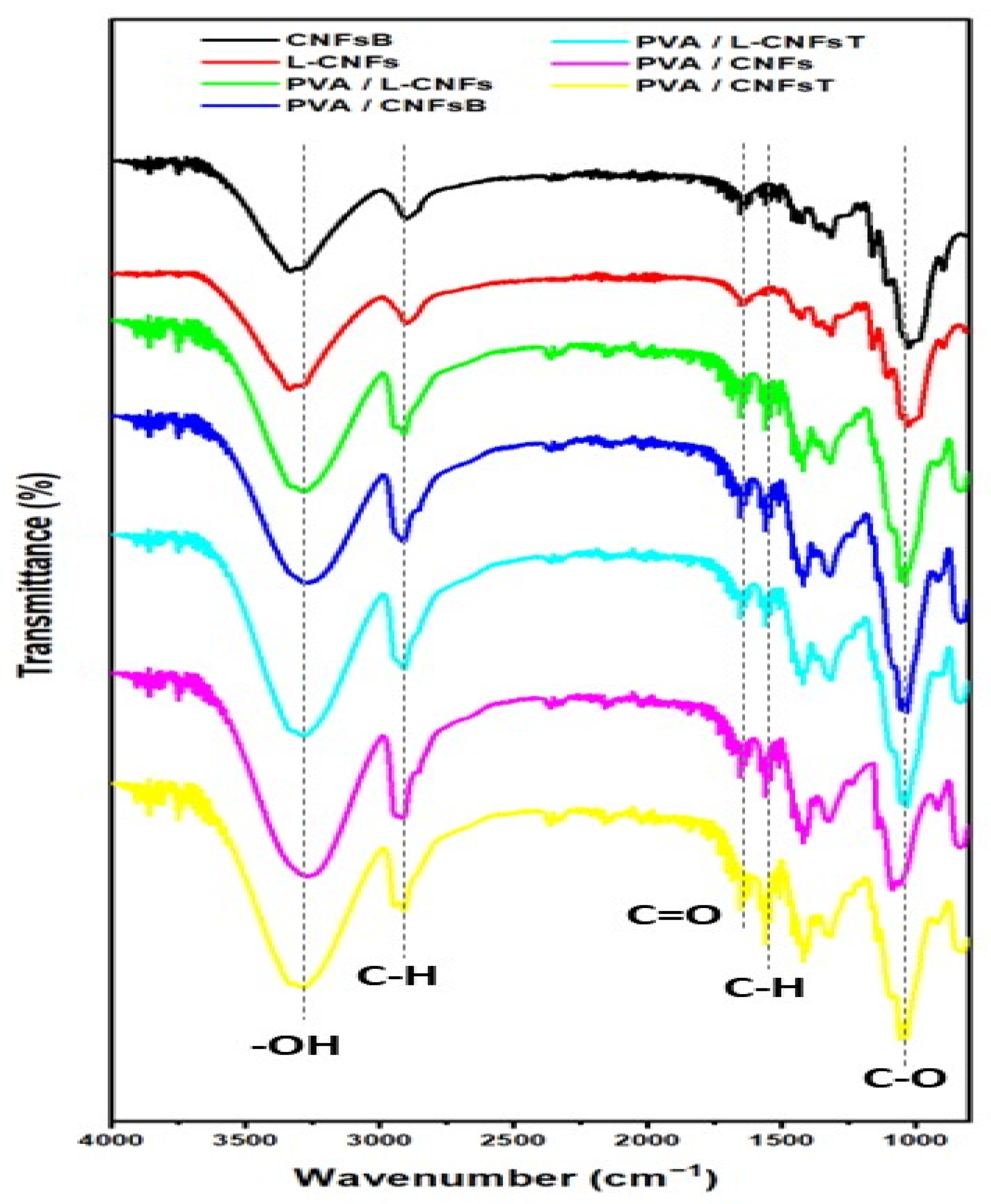
2.4. Fourier Transform Infrared Spectroscopy (FTIR)
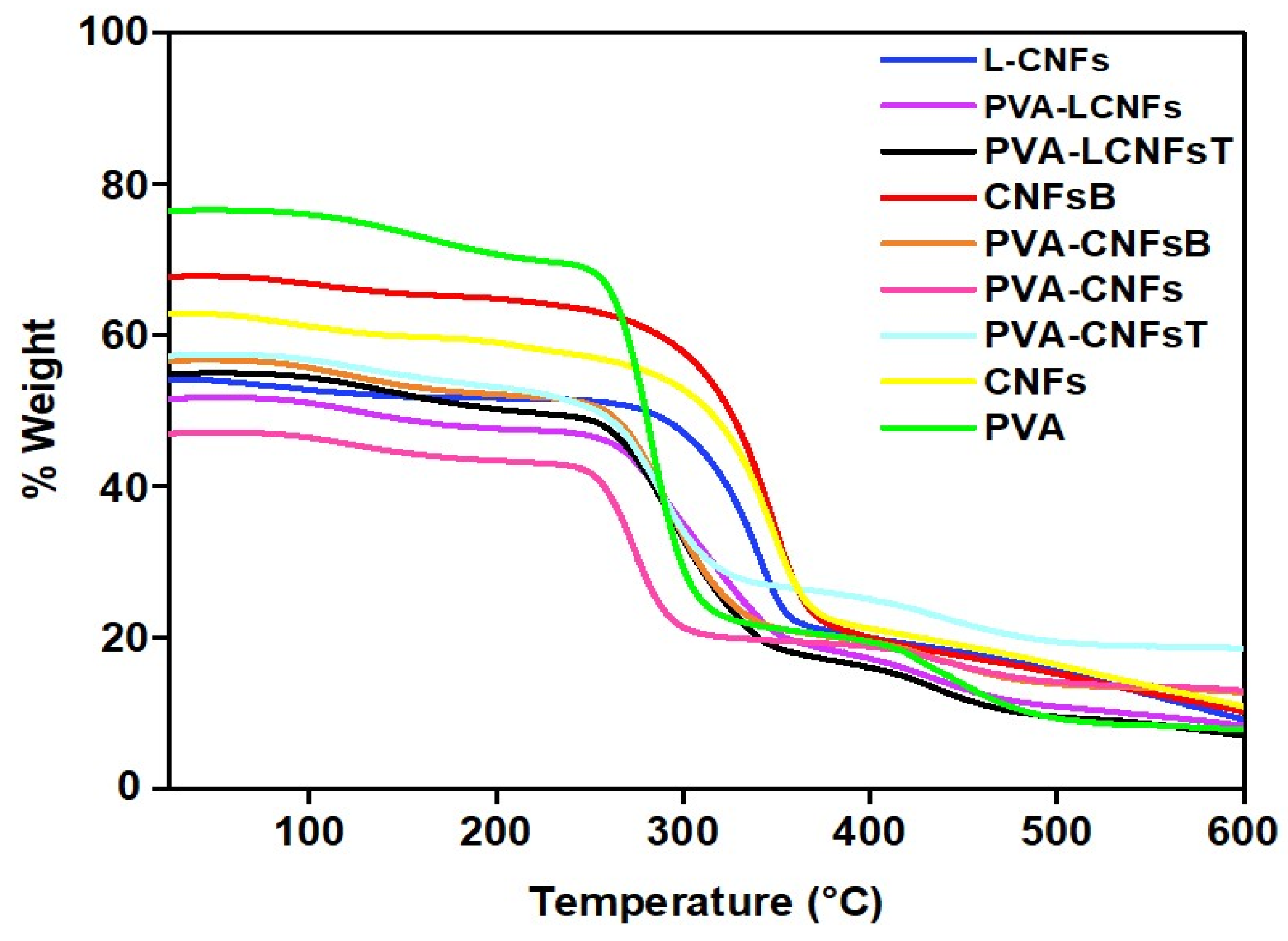
2.5. Thermo-Gravimetric Analysis (TGA)
2.6. Zeta Potential
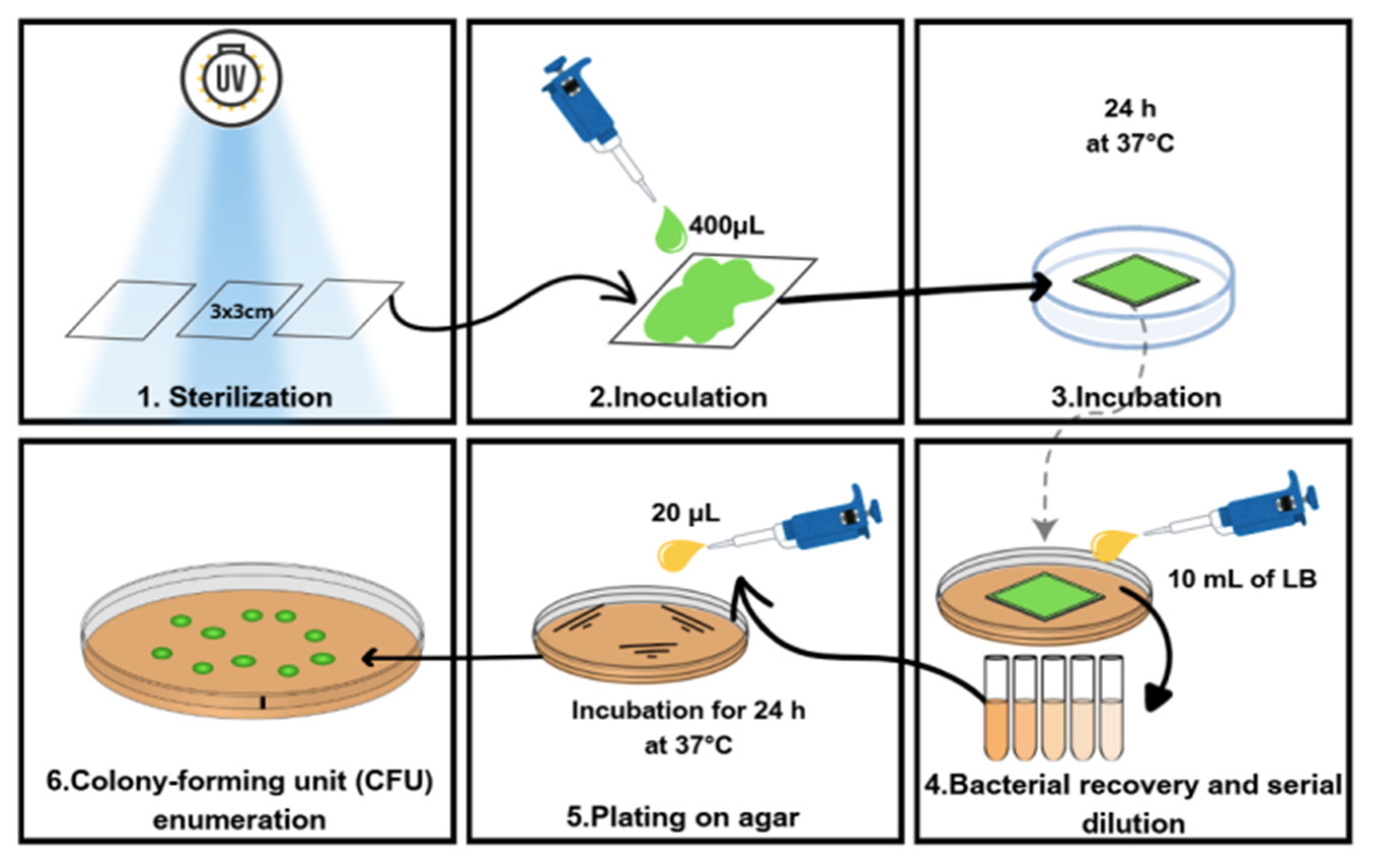
2.7. PVA/CNFs Antimicrobial Activity Nanocomposites
2.8. Scanning Electron Microscopy (SEM) with Energy Dispersive X-Ray Spectroscopy (EDS)
3. Results
3.1. Infrared Spectroscopy Analysis (FTIR)
3.2. PVA/CNFs Nanocomposites Thermo-Gravimetric Analysis (TGA)
3.3. PVA/CNFs Nanocomposites Zeta Potential (mV) Analysis
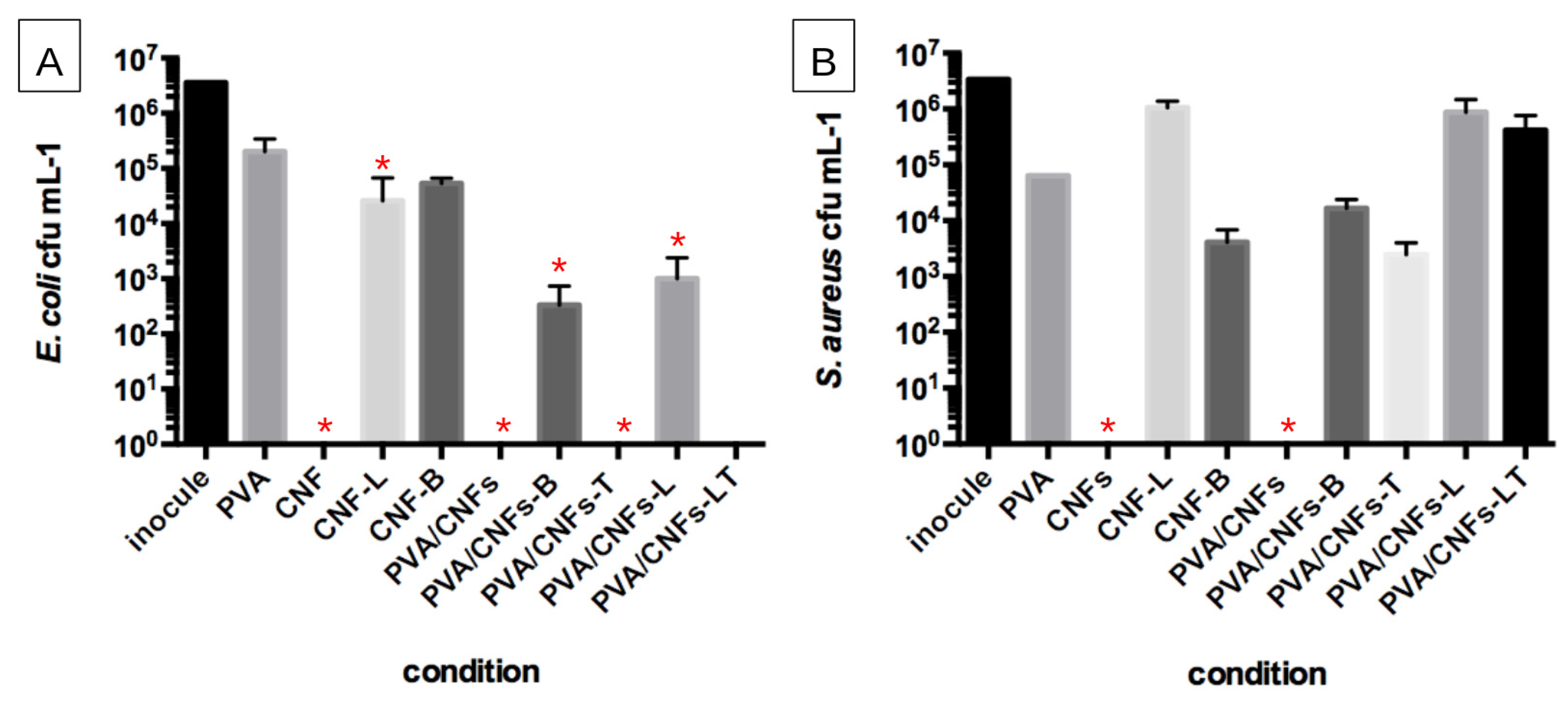
3.4. Antimicrobial Activity of PVA/CNFs Nanocomposites
3.5. Surface Topography and Bacteria–Material Interactions in PVA/CNFs Nanocomposites
4. Discussion
Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nanomaterials from Renewable Resources for Emerging Applications, 1st ed.; Ahankari, S.S., Mohanty, A.K., Misra, M., Eds.; CRC Press, 2023. [Google Scholar] [CrossRef]
- Ghadiri Alamdari, N.; Salmasi, S.; Almasi, H. Tomato Seed Mucilage as a New Source of Biodegradable Film-Forming Material: Effect of Glycerol and Cellulose Nanofibers on the Characteristics of Resultant Films. Food Bioprocess Technol 2021, 14, 2380–2400. [Google Scholar] [CrossRef]
- Plotniece, A. Selected strategies to fight pathogenic bacteria. Journal of Enzyme Inhibition and Medicinal. Chemistry 2023, 38(1). [Google Scholar] [CrossRef]
- Maresca, Diamante; Mauriello, Gianluigi. Development of Antimicrobial Cellulose Nanofiber-Based Films Activated with Nisin for Food Packaging Applications. Foods 2022, 11(no. 19), 3051. [Google Scholar] [CrossRef] [PubMed]
- Mada, T.; Abera, A.; Duraisamy, R. Review on Bio-Nanocomposite Polymers and its Application on Food Packaging Processes. Journal of Earth and Environmental Waste Management 2023, 1(1), 1–8. [Google Scholar]
- Wu, Yingji. Advanced nanocellulose-based gas barrier materials: Present status and prospects. Chemosphere, Volume 286, Part 3 2022, 2022, 131891. [Google Scholar] [CrossRef] [PubMed]
- Espinosa, E. PVA/(ligno)nanocellulose biocomposite films. Effect of residual lignin content on structural, mechanical, barrier and antioxidant properties. International Journal of Biological Macromolecules 2019, 141, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Dirpan. A Review on Biopolymer-Based Biodegradable Film for Food Packaging: Trends over the Last Decade and Future Research. Polymers 2023, 15(no. 13), 2781. [Google Scholar] [CrossRef]
- Bose. A Comprehensive Review on Significance and Advancements of Antimicrobial Agents in Biodegradable Food Packaging. Antibiotics 2023, 12(no. 6), 968. [Google Scholar] [CrossRef]
- Prilepskii, Artur. Conductive bacterial cellulose: From drug delivery to flexible electronics. Carbohydrate Polymers,Volume 2023, Volume 313, 120850. [Google Scholar] [CrossRef]
- Chakraborty, G.; et al. Applicability of Fe-CNC/GR/PLA composite as potential sensor for biomolecules. J Mater Sci: Mater Electron 2020, 31, 5984–5999. [Google Scholar] [CrossRef]
- Haghighi, Hossein. Characterization of bio-nanocomposite films based on gelatin/polyvinyl alcohol blend reinforced with bacterial cellulose nanowhiskers for food packaging applications. Food Hydrocolloids,Volume 2021, Volume 113(2021), 106454. [Google Scholar] [CrossRef]
- Hashemzehi, M. A comprehensive review of nanocellulose modification and applications in papermaking and packaging: Challenges, technical solutions, and perspectives. BioResources 2022, 17(2), 3718–3780. [Google Scholar] [CrossRef]
- Nicoleta, Ene. The Production of Biodegradable Polymers-medium-chain-length Polyhydroxyalkanoates(mcl-PHA) in Pseudomonas putida for Biomedical EngineeringApplications. Current Pharmaceutical Biotechnology; Volume 2022, Volume 23(Issue 8, Year), e100821195486. [Google Scholar] [CrossRef]
- Tarrés, Quim. Research on the Strengthening Advantages on Using Cellulose Nanofibers as Polyvinyl Alcohol Reinforcement. Polymers 2020, 12(no. 4), 974. [Google Scholar] [CrossRef]
- Nechita, Petronela; Năstac, Silviu Marian. Overview on Foam Forming Cellulose Materials for Cushioning Packaging Applications. Polymers 2022, 14(no. 10), 1963. [Google Scholar] [CrossRef]
- Oun, Ahmed A. Recent advances in polyvinyl alcohol based composite films and their applications in food packaging. Food Packaging and Shelf Life, Volume 2022, Volume 34, 100991. [Google Scholar] [CrossRef]
- Kouser, S.; et al. In vitro evaluation of modified halloysite nanotubes with sodium alginate-reinforced PVA/PVP nanocomposite films for tissue engineering applications. Appl Nanosci 2022, 12, 3529–3545. [Google Scholar] [CrossRef]
- Zhang, Wanli. Advances in sustainable food packaging applications of chitosan/polyvinyl alcohol blend films. Food Chemistry,Volume 2024, Volume 443, 138506. [Google Scholar] [CrossRef] [PubMed]
- Oreibi, I. Polymer nanocomposites comprising PVA matrix and Ag–BaTiO3 nanofillers: a comparative study of structural, dielectric and optical characteristics for optics and quantum nanoelectronic applications. Opt Quant Electron 2024, 56, 119. [Google Scholar] [CrossRef]
- Deng, Hao. A review of starch/polyvinyl alcohol (PVA) blend film: A potential replacement for traditional plastic-based food packaging film. International Journal of Biological Macromolecules 2024, Volume 273, 132926. [Google Scholar] [CrossRef] [PubMed]
- Ahari, Hamed. Employing Nanosilver, Nanocopper, and Nanoclays in Food Packaging Production: A Systematic Review Coatings. 2021, 11(no. 5), 509. [Google Scholar] [CrossRef]
- Brandelli, Adriano; Lopes, Nathalie Almeida; Pinilla, Cristian Mauricio Barreto. Nanostructured Antimicrobials for Quality and Safety Improvement in Dairy Products Foods. 2023, 12(no. 13), 2549. [Google Scholar] [CrossRef]
- Wang, Mengxia. Effect of Length of Cellulose Nanofibers on Mechanical Reinforcement of Polyvinyl Alcohol. Polymers 2022, 14(no. 1), 128. [Google Scholar] [CrossRef]
- Listyarini, A.; Fauzia, V.; Imawan, C. Mechanical and barrier properties of Tapioca/PVA composite films reinforced with pineapple leaf nanocellulose. Journal of Physics: Conference Series 2020, 1428, 012002. [Google Scholar] [CrossRef]
- Syafrina, D. Improving Physical Properties of Polyvinyl Alcohol Film through The Addition of Nanocellulose Prepared from Palm Oil Solid Waste. IOP Conference Series: Materials Science and Engineering 2020, 845, 012027. [Google Scholar] [CrossRef]
- Lee, H. Chemical and physical reinforcement behavior of dialdehyde nanocellulose in PVA composite film: A comparison of nanofiber and nanocrystal. Carbohydrate Polymers 2020, 232, 115771. [Google Scholar] [CrossRef] [PubMed]
- Aprilia, S. Composites polyvinyl alcohol filled with nanocellulose from oil palm waste by formic acid hydrolysis. MATEC Web Conf. 2019, 268, p. 04012. [Google Scholar] [CrossRef]
- Liu, Ming. Chapter 10 - Production of microfibrillated cellulose fibers and their application in polymeric composites. In Micro and Nano Technologies, Nanotechnology in Paper and Wood Engineering; Bhat, Rajeev, Kumar, Ashok, Nguyen, Tuan Anh, Sharma, Swati, Eds.; Elsevier, 2022; pp. Pages 197–229. ISBN 9780323858359. [Google Scholar] [CrossRef]
- Tie, Luna. Nanocellulose fine-tuned poly(acrylic acid) hydrogel for enhanced diclofenac removal. International Journal of Biological Macromolecules 2022, Volume 213, Pages 1029–1036. [Google Scholar] [CrossRef] [PubMed]
- Valdebenito, F. On the nanofibrillation of corn husks and oat hulls fibres. Industrial Crops and Products 2017, 95, 528–534. [Google Scholar] [CrossRef]
- Albornoz-Palma, et al. Effect of lignin and hemicellulose on the properties of lignocellulose nanofibril suspensions. Cellulose 2020, 27, 10631–10647. [Google Scholar] [CrossRef]
- Guimarães, Barbara Maria Ribeiro. Bio-based films/nanopapers of banana tree pseudostem: From lignocellulosic wastes to added-value micro/nanomaterials, 13 May, PREPRINT (Version 1) available at Research Square. 2021. [Google Scholar] [CrossRef]
- Padhi, S.; Singh, A.; Routray, W. Nanocellulose from agro-waste: a comprehensive review of extraction methods and applications. Rev Environ Sci Biotechnol 2023, 22, 1–27. [Google Scholar] [CrossRef]
- Sánchez-Gutiérrez. Production of Cellulose Nanofibers from Olive Tree Harvest—A Residue with Wide Applications. Agronomy 2020, 10(no. 5), 696. [Google Scholar] [CrossRef]
- Zhang, Xiuqiang. Preparation and Characterization of Cellulose Nanofiber/Zinc Oxide Composite Films. Journal of Biobased Materials and Bioenergy 2020, 14, (2)203–208(6). [Google Scholar] [CrossRef]
- García-Castrillo, Marta. Aqueous Cellulose Nanocrystal-Colloidal Au Inks for 2D Printed Photothermia. ACS Sustainable Chemistry & Engineering 2024, 2024 12(4), 1468–1479. [Google Scholar] [CrossRef]
- Polylactic Acid-Based Nanocellulose and Cellulose Composites, 1st ed.; Parameswaranpillai, J., Siengchin, S., Salim, N.V., George, J.J., Poulose, A., Eds.; CRC Press, 2022. [Google Scholar] [CrossRef]
- Ojagh, S.M.A.; et al. Hairy bacterial nanocellulose (2023): preparation and bioconjugation with an antibacterial agent. Cellulose 30, 10905–10922. [CrossRef]
- Zhang, Shuai. Preparation of amino cellulose nanofiber via ε-poly-L-lysine grafting with enhanced mechanical, anti-microbial and food preservation performance. Industrial Crops and Products 2023, 194, 116288. [Google Scholar] [CrossRef]
- Risks, S.S.C.o.E.a.N.I.H. Nanosilver: safety, health and environmental effects and role in antimicrobial resistance. 2014. [Google Scholar]
- Panáček, A. Bacterial resistance to silver nanoparticles and how to overcome it. Nature Nanotechnology 2018, 13(1), 65–71. [Google Scholar] [CrossRef]
- Seydibeyoğlu, et al. Review on Hybrid Reinforced Polymer Matrix Composites with Nanocellulose, Nanomaterials, and Other Fibers. Polymers 2023, 15(no. 4), 984. [Google Scholar] [CrossRef]
- Vidal, Cristian Patiño. Development of an antibacterial coaxial bionanocomposite based on electrospun core/shell fibers loaded with ethyl lauroyl arginate and cellulose nanocrystals for active food packaging. Food Packaging and Shelf Life 2022, 31, 100802. [Google Scholar] [CrossRef]
- El-Gendy, A. TEMPO-Oxidized Cellulose Nanofibers/Polylactic acid/TiO2 as Antibacterial Bionanocomposite for Active Packaging. Egyptian Journal of Chemistry 2017, 60(6), 1007–1014. [Google Scholar] [CrossRef]
- Meira, S.M.M. A novel active packaging material based on starch-halloysite nanocomposites incorporating antimicrobial peptides. Food Hydrocolloids 2017, 63, 561–570. [Google Scholar] [CrossRef]
- Uranga, J. Citric acid-incorporated fish gelatin/chitosan composite films. Food Hydrocolloids 2019, 86, 95–103. [Google Scholar] [CrossRef]
- Liu, J. Effect of gallic acid grafted chitosan film packaging on the postharvest quality of white button mushroom (Agaricus bisporus). Postharvest Biology and Technology 2019, 147, 39–47. [Google Scholar] [CrossRef]
- Alvarado, D.R. A facile strategy for photoactive nanocellulose-based antimicrobial materials. Green Chemistry 2019, 21(12), 3424–3435. [Google Scholar] [CrossRef]
- Fu, H.; Gao, W.; Wang, B.; et al. Effect of lignin content on the microstructural characteristics of lignocellulose nanofibrils. Cellulose 2020, 27, 1327–1340. [Google Scholar] [CrossRef]
- Oliaei, Erfan; Lindström, Tom; Berglund, Lars A. Sustainable Development of Hot-Pressed All-Lignocellulose Composites—Comparing Wood Fibers and Nanofibers. Polymers 2021, 13(no. 16), 2747. [Google Scholar] [CrossRef] [PubMed]
- Albornoz-Palma, Gregory. Effect of lignin on the morphological, rheological, and dielectric characteristics of lignocellulose nanofibrils from Pinus radiata. Industrial Crops and Products 2023, 204, Part B, 117323. [Google Scholar] [CrossRef]
- Liu, Kun. Lignin-containing cellulose nanomaterials: preparation and applications. Green Chem. 2021, 23(24), 9723–9746. [Google Scholar] [CrossRef]
- Behr, Marc; El Jaziri, Mondher; Baucher, Marie. Chapter Three - Glycobiology of the plant secondary cell wall dynamics. In Advances in Botanical Research; Sibout, Richard, Ed.; Academic Press, 2022; Volume 104, pp. Pages 97–131. ISSN 0065-2296, ISBN 9780323912204. [Google Scholar] [CrossRef]
- Priyadarshi, Ruchir. Lignin as a sustainable and functional material for active food packaging applications: A review. Journal of Cleaner Production 2024, 469, 143151. [Google Scholar] [CrossRef]
- Yang, Weijun. Poly(lactic acid)/lignin films with enhanced toughness and anti-oxidation performance for active food packaging. International Journal of Biological Macromolecules 2020, 144, 102–110. [Google Scholar] [CrossRef]
- Tavares, Débora; Woiciechowski, Adenise Lorenci. Lignin from Residual Sawdust of Eucalyptus spp.—Isolation, Characterization, and Evaluation of the Antioxidant Properties. Biomass 2022, 2(no. 3), 195–208. [Google Scholar] [CrossRef]
- Antonino, Leonardo D. Lignin-Based Polyurethanes from the Blocked Isocyanate Approach: Synthesis and Characterization. ACS Omega 2023, 2023 8(30), 27621–27633. [Google Scholar] [CrossRef] [PubMed]
- Ndaba, Busiswa. Influence of extraction methods on antimicrobial activities of lignin-based materials: A review. Sustainable Chemistry and Pharmacy 2020, 18, 100342. [Google Scholar] [CrossRef]
- Nakason, Kamonwat. Antimicrobial and antioxidant activities of lignin by-product from sugarcane leaf conversion to levulinic acid and hydrochar. Sustainable Materials and Technologies 2024, 40, e00973. [Google Scholar] [CrossRef]
- Ruwoldt, Jost. Functional surfaces, films, and coatings with lignin – a critical review 12529 – 12553. RSC Advances 2023, 13((18)). [Google Scholar] [CrossRef]
- Basbasan; Angel, Jr. Lignin Nanoparticles for Enhancing Physicochemical and Antimicrobial Properties of Polybutylene Succinate/Thymol Composite Film for Active Packaging Polymers. 2023, 15(no. 4), 989. [Google Scholar] [CrossRef]
- Zuluaga, R; Putaux, R; Cruz, J; Vélez, J; Mondragon, I; Gañán, P. Cellulose microfibrils from banana rachis: Effect of alkaline treatments on structural and morphological features. Carbohydrate Polymers 2009, 76(1), 51–59. [Google Scholar] [CrossRef]
- Valdebenito, F.; Albornoz, C.; Rivera, V.; Elgueta, E.; Nisar, M.; Lira, S.; Valerio, O.; Narváez, A.; Quezada, C.; Muñoz, R.; Azócar, L.; Sandoval, F. Stable Reusability of Nanocellulose Aerogels with Amino Group Modification in Adsorption/Desorption Cycles for CO2 Capture. Materials 2025, 18(2), 243. [Google Scholar] [CrossRef]
- Chao, Y.; Zhang, T. Optimization of fixation methods for observation of bacterial cell morphology and surface ultrastructures by atomic force microscopy. Appl Microbiol Biotechnol 2011, 92, 381–392. [Google Scholar] [CrossRef]
- Valdebenito, F. Agricultural-Waste Cellulose Nanofibrils to develop a CO2 adsorbent material. In Programa de doctorado en ciencias de recursos naturales; Universidad de la Frontera, 2017. [Google Scholar]
- Saito, T. Homogeneous Suspensions of Individualized Microfibrils from TEMPO Catalyzed Oxidation of Native Cellulose. Biomacromolecules 2006, 7(6), 1687–1691. [Google Scholar] [CrossRef] [PubMed]
- Dohlen, S. Effect of different packaging materials containing poly-[2-(tert-butylamino) methylstyrene] on the growth of spoilage and pathogenic bacteria on fresh meat. International Journal of Food Microbiology 2017, 257, 91–100. [Google Scholar] [CrossRef]
- Hüwe, C. Potential of antimicrobial treatment of linear low-density polyethylene with poly((tert-butyl-amino)-methyl-styrene) to reduce biofilm formation in the food industry. Biofouling 2018, 34(4), 378–387. [Google Scholar] [CrossRef] [PubMed]
- Safari, S.; Sheikhi, A.; van de Ven, T.G. Electroacoustic characterization of conventional and electrosterically stabilized nanocrystalline celluloses. J. Colloid Interface Sci. 2014, 432(1), 151–157. [Google Scholar] [CrossRef] [PubMed]
- Powell, L.C.; Khan, S.; Chinga-Carrasco, G.; Wright, C.J.; Hill, K.E.; Thomas, D.W. An investigation of Pseudomonas aeruginosa biofilm growth on novel nanocellulose fibre dressings. Carbohydrate Polymers 2016, 137, 191–197. [Google Scholar] [CrossRef]
- Chinga-Carrasco, G.; Pasquier, E.; Solberg, A.; Leirset, I.; Stevanic, J.S. Structure and surface chemistry of nanocellulose films relevant for antibacterial applications. Carbohydrate Polymers 2017, 157, 1955–1962. [Google Scholar]
- Knutsen, M.F.; Agrenius, K.; Ugland, H.; Petronis, S.; Haglerod, C.; Håkansson, J.; Chinga-Carrasco, G. Oxygenated nanocellulose: A material platform for antibacterial wound dressing devices. ACS Applied Bio Materials 2021, 4, 7554–7562. [Google Scholar] [CrossRef] [PubMed]
- Chinga-Carrasco, G.; Pasquier, E.; Solberg, A.; Leirset, I.; Stevanic, J.S. Oxygenated nanocellulose for wound healing applications: Increase of washing efficiency after chemical pre-treatment and stability of homogenized gels over 10 months. Carbohydrate Polymers 2023, 314, 120923. [Google Scholar] [CrossRef] [PubMed]


| Nomenclature | Description |
| CNFs | Krat CNFs film |
| CNFs-L | Lignin-CNFs film |
| CNFs-B | CNFs film additivity with blueberry pruning extractable. |
| PVA | Polyvinyl alcohol film |
| PVA/CNFs | PVA/CNFs nanocomposite |
| PVA/CNFs-L | PVA/lignin-CNFs nanocomposite |
| PVA/CNFs-T | PVA/TEMPO-CNFs nanocomposite |
| PVA/CNFs-LT | PVA/lignin-CNFs-TEMPO nanocomposite |
| PVA/CNFs-B | PVA/CNFs additivity with blueberry pruning extractable nanocomposite. |
| Sample | Zeta potential (mV) |
| PVA | -1.307 |
| CNFs | -35.29 |
| PVA/CNFs | -3.766 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




