Submitted:
04 March 2026
Posted:
05 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials and Sample Preparation
2.2. Differential Scanning Calorimetry (DSC)
2.3. Broadband Dielectric Spectroscopy (BDS)
3. Results and Discussion
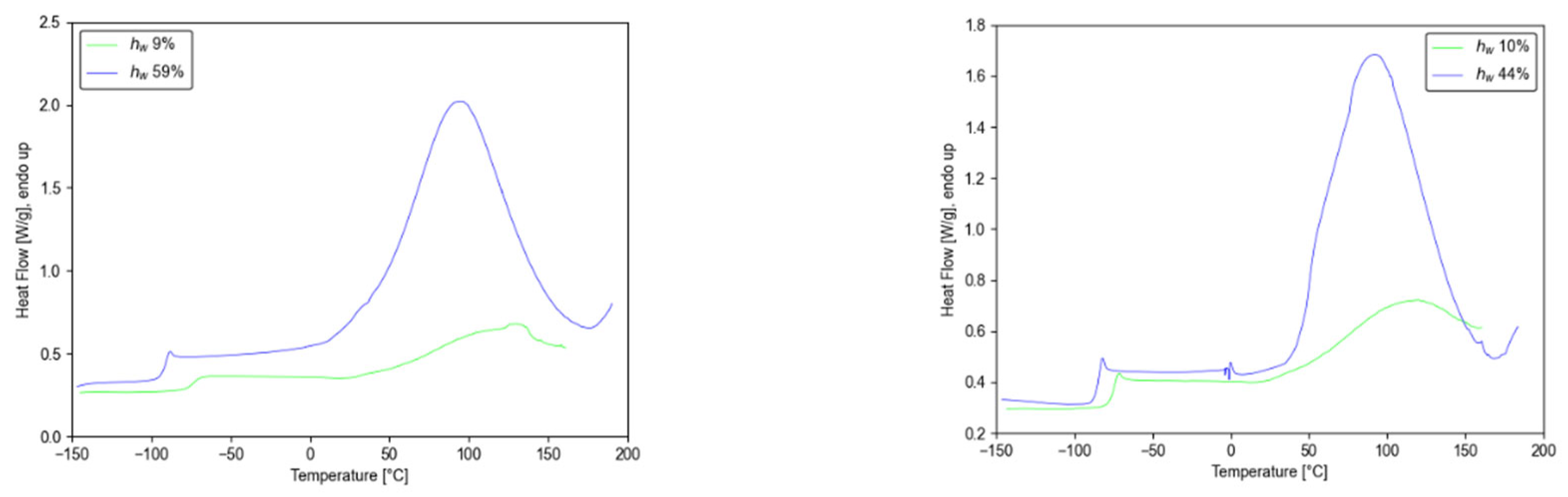
3.1. DSC Results
3.2. DRS Results
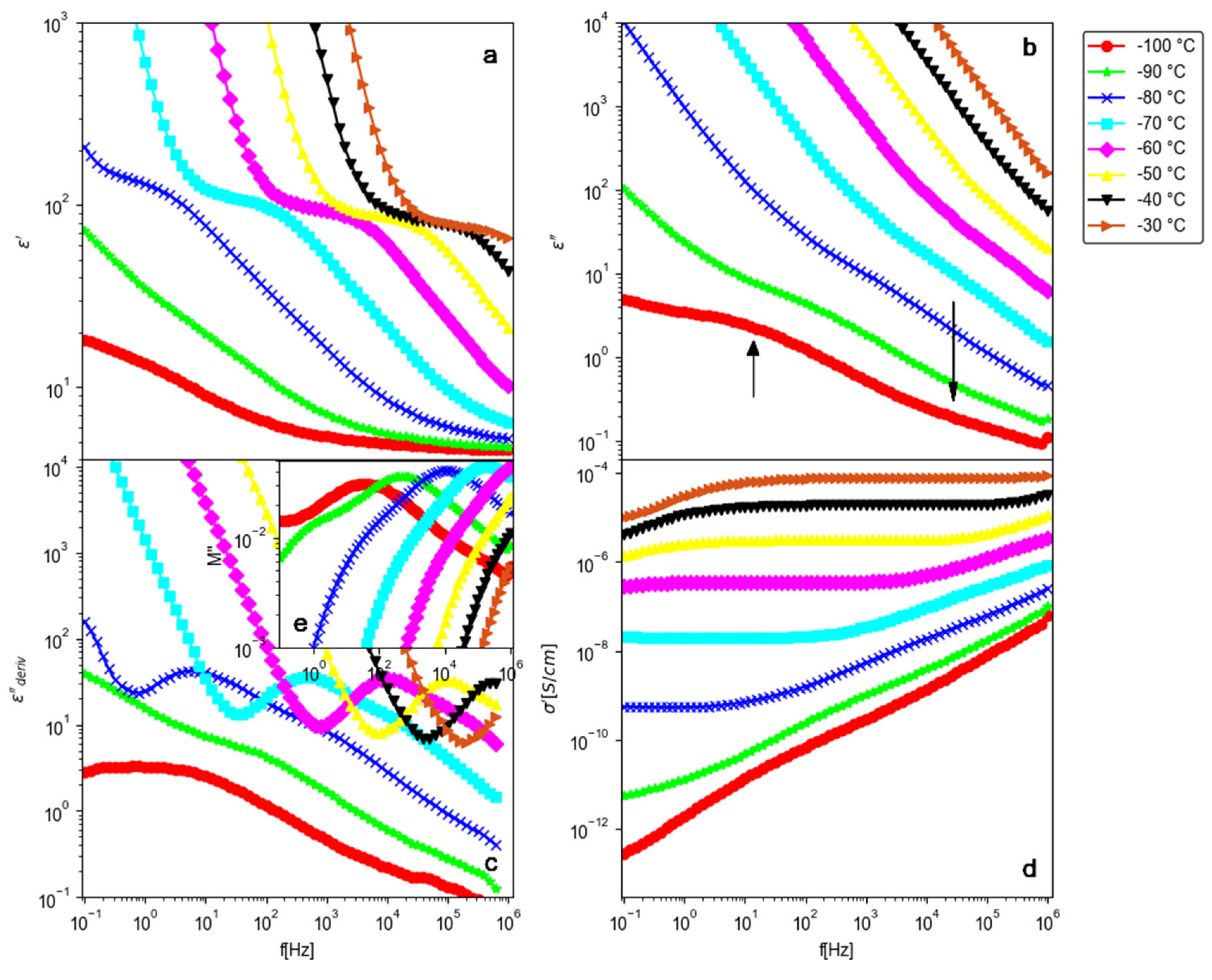
3.2.1. Dielectric Spectra
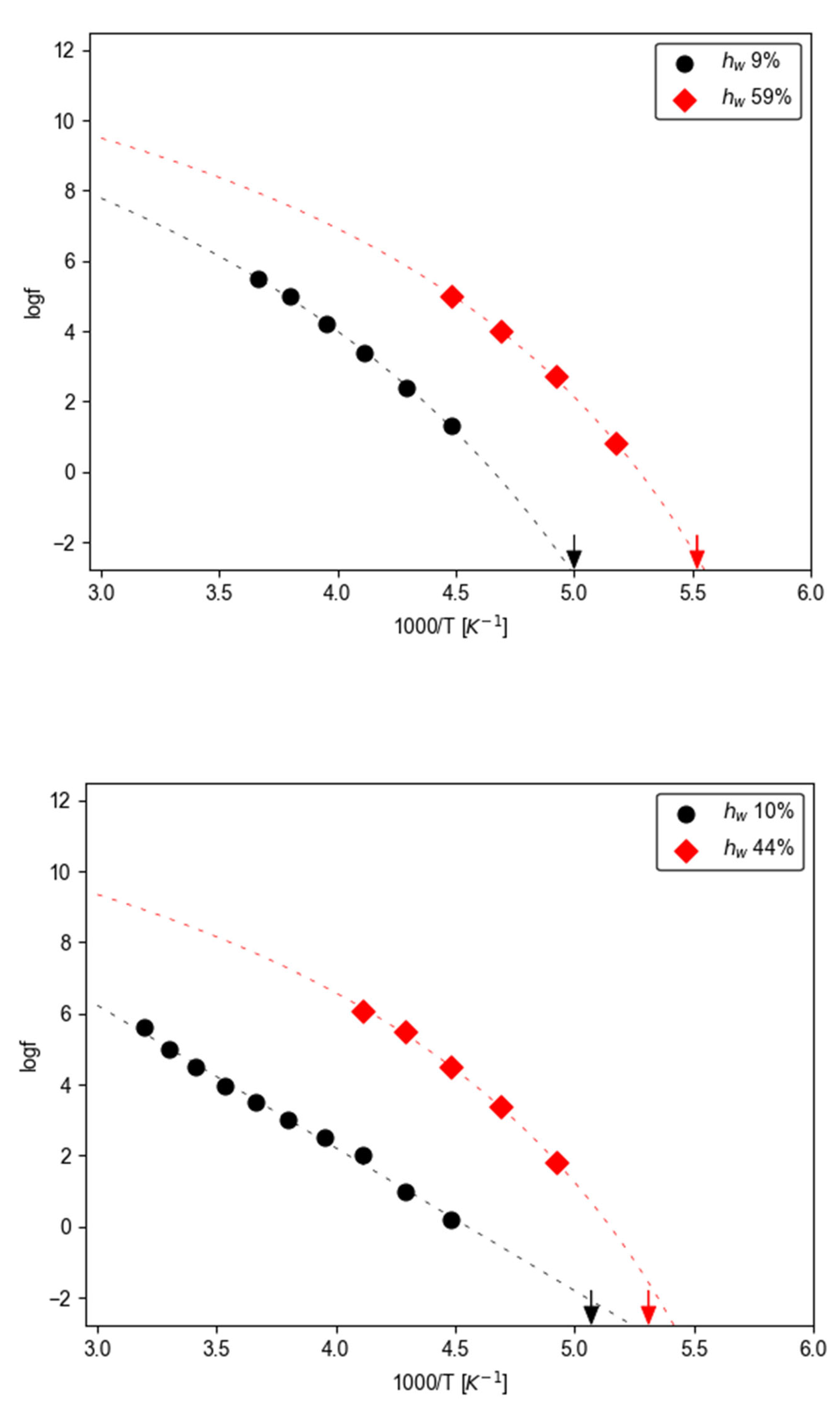
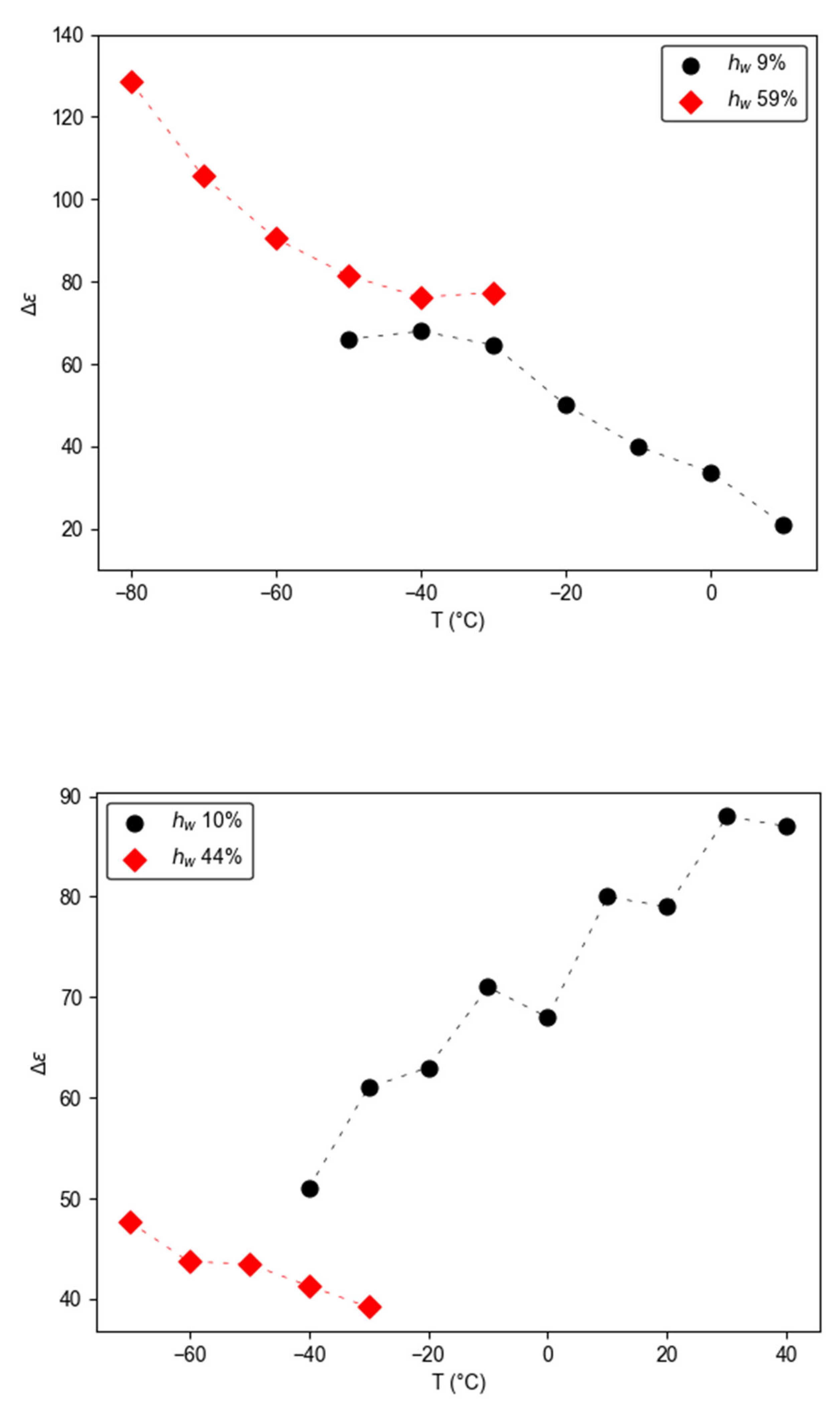
3.2.2. Temperature - Water Dependence of Dynamics and Conductivity
4. Conclusions
Author Contributions
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lei, Z.; Chen, B.; Koo, Y.-M.; MacFarlane, D.R. Introduction: Ionic Liquids. Chem. Rev. 2017, 117, 6633–6635. [Google Scholar] [CrossRef]
- Lei, Z.; Dai, C.; Hallet, J; Shiflett, M. Introduction: Ionic Liquids for Diverse Applications. Chem. Rev. 2024, 124, 7533–7535. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M; Thomas, ML; Zhang, S; Ueno, K; Yasuda, T; Dokko, K. Application of Ionic Liquids to Energy Storage and Conversion Materials. Electrochim. Acta 2017, 235, 633–642. [Google Scholar] [CrossRef]
- Tang, X.; Lv, S.; Jiang, K.; Zhou, G.; Liu, X. Recent Development of Ionic Liquid-Based Electrolytes in Lithium-Ion Batteries. J. Power Sources 2022, 542, 231792. [Google Scholar] [CrossRef]
- Matuszek, K.; Piper, S.L.; Brzeczek-Szafran, A.; Roy, B.; Saher, S.; Pringle, J. M.; MacFarlane, D. R. Unexpected Energy Applications of Ionic Liquids. Adv. Mater. 2024, 36, 2313023. [Google Scholar] [CrossRef]
- Noda, A.; Susan, M. A. B. H.; Kudo, K.; Mitsushima, S.; Hayamizu, K.; Watanabe, M. Brønsted Acid–Base Ionic Liquids as Proton-Conducting Nonaqueous Electrolytes. J. Phys. Chem. B 2003, 107, 4024–4033. [Google Scholar] [CrossRef]
- Armand, M.; Endres, F.; MacFarlane, D.R.; Ohno, H.; Scrosati, B. Ionic-Liquid Materials for the Electrochemical Challenges of the Future. Nat. Mater. 2009, 8, 621–629. [Google Scholar] [CrossRef] [PubMed]
- MacFarlane, D.R.; Tachikawa, N.; Forsyth, M.; Pringle, J. M.; Howlett, P. C.; Elliott, G. D.; Davis, J. H.; Watanabe, M.; Simon, P.; Angell, C. A. Energy Applications of Ionic Liquids. Energy Environ. Sci. 2014, 7, 232–250. [Google Scholar] [CrossRef]
- Gao, X.; Wu, F.; Mariani, A.; Passerini, S. Concentrated Ionic-Liquid-Based Electrolytes for High-Voltage Lithium Batteries with Improved Performance at Room Temperature. ChemSusChem 2019, 12, 4185–4193. [Google Scholar] [CrossRef]
- Ma, Χ.; Yu, J.; Hu, Y.; Texter, J.; Yan, F. Ionic liquid/poly(ionic liquid)-based electrolytes for lithium batteries. Ind. Chem. Mater. 2023, 1, 39–59. [Google Scholar] [CrossRef]
- Cao, Y.; Chena, Y.; Sun, X.; Zhanga, Z.; Mu, T. Water sorption in ionic liquids: kinetics, mechanisms and hydrophilicity. Phys. Chem. Chem. Phys. 2012, 14, 12252–12262. [Google Scholar] [CrossRef]
- Di Francesco, F.; Calisi, N.; Creatini, M.; Melai, B.; Salvo, P.; Chiappe, C. Water sorption by anhydrous ionic liquids. Green Chem. 2011, 13, 1712–1717. [Google Scholar] [CrossRef]
- Freire, M.G.; Neves, C.M.S.S.; Marrucho, I.M.; Coutinho, J.A.P.; Fernandes, A.M. Hydrolysis of Tetrafluoroborate and Hexafluorophosphate Counterions in Imidazolium-Based Ionic Liquids. J. Phys. Chem. A 2010, 114, 3744–3749. [Google Scholar] [CrossRef]
- Ma, C.; Laaksonen, A.; Liu, C.; Lu, X.; Ji, X. The peculiar effect of water on ionic liquids and deep eutectic solvents. Chem. Soc. Rev. 2018, 47, 8685–8720. [Google Scholar] [CrossRef] [PubMed]
- Ausín, D.; Parajó, J.J.; Trenzado, J.L.; Varela, L.M.; Cabeza, O.; Segade, L. Influence of Small Quantities of Water on the Physical Properties of Alkylammonium Nitrate Ionic Liquids. Int. J. Mol. Sci. 2021, 22, 7334. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, H.; Brennecke, J.F. Temperature and composition dependence of the density and viscosity of binary mixtures of water plus ionic liquid. J. Chem. Eng. Data 2006, 51, 2145–2155. [Google Scholar] [CrossRef]
- Andanson, J.-M.; Meng, X.; Traïkia, M.; Husson, P. Quantification of the impact of water as an impurity on standard physico-chemical properties of ionic liquids. J. Chem. Thermodyn. 2016, 94, 169–176. [Google Scholar] [CrossRef]
- Grishina, E.P.; Ramenskaya, L.M.; Gruzdev, M.S.; Kraeva, O.V. Water effect on physicochemical properties of 1-butyl-3-methylimidazolium based ionic liquids with inorganic anions. J. Mol. Liq. 2013, 177, 267–272. [Google Scholar] [CrossRef]
- Widegren, J.A.; Laesecke, A.; Magee, J.W. The effect of dissolved water on the viscosities of hydrophobic room-temperature ionic liquids. Chem. Commun. 2005, 1610–1612. [Google Scholar] [CrossRef]
- Domańska, U.; Królikowska, M. Density and viscosity of binary mixtures of thiocyanate ionic liquids plus water as a function of temperature. J. Solut. Chem. 2012, 41, 1422–1445. [Google Scholar] [CrossRef]
- Liu, W.W.; Cheng, L.Y.; Zhang, Y.M.; Wang, H.P.; Yu, M.F. The physical properties of aqueous solution of room-temperature ionic liquids based on imidazolium: database and evaluation. J. Mol. Liq. 2008, 140, 68–72. [Google Scholar] [CrossRef]
- Lopes, J.N.C.; Gomes, M.F.C.; Husson, P.; Padua, A.A.H.; Rebelo, L.P.N.; Sarraute, S.; Tariq, M. Polarity, viscosity, and ionic conductivity of liquid mixtures containing [C4C1im][Ntf2] and a molecular component. J. Phys. Chem. B 2011, 115, 6088–6099. [Google Scholar] [CrossRef]
- Danten, Y.; Cabaco, M.I.; Besnard, M. Interaction of water diluted in 1-butyl-3-methyl imidazolium ionic liquids by vibrational spectroscopy modeling. J. Mol. Liq. 2010, 153, 57–66. [Google Scholar] [CrossRef]
- Shekaari, H.; Mousavi, S.S.; Mansoori, Y. Thermophysical properties of ionic liquid, 1-pentyl-3-methylimidazolium chloride in water at different temperatures. Int. J. Thermophys. 2009, 30, 499–514. [Google Scholar] [CrossRef]
- Wojnarowska, Z.; Grzybowska, K.; Hawelek, L.; Święty-Pośpiech, A.; Masiewicz, E.; Paluch, M.; Sawicki, W.; Chmielewska, A.; Bujak, P.; Markowski, J. Molecular Dynamics and Glass Transition Behavior in Ionic Liquids. Mol. Pharm. 2012, 9, 1250–1261. [Google Scholar] [CrossRef]
- Sippel, P.; Dietrich, V.; Reuter, D.; Aumüller, M.; Lunkenheimer, P.; Loidl, A.; Krohns, S. Relaxation Dynamics in Ionic Liquids. J. Mol. Liq. 2016, 223, 635–642. [Google Scholar] [CrossRef]
- Bester-Rogac, M.; Stoppa, A.; Hunger, J.; Hefter, G.; Buchner, R. Electrical Conductivity and Permittivity of Ionic Liquids. Phys. Chem. Chem. Phys. 2011, 13, 17588–17598. [Google Scholar] [PubMed]
- Mero, A.; Guglielmero, L.; D’Andrea, F.; Pomelli, C.S.; Guazzelli, L.; Koutsoumpos, S.; Tsonos, G.; Stavrakas, I.; Moutzouris, K.; Mezzetta, A. Thermophysical Characterization of Ionic Liquids. J. Mol. Liq. 2022, 354, 118850. [Google Scholar] [CrossRef]
- Sturlaugson, A.L.; Fruchey, K.S.; Fayer, M.D. Orientational Dynamics of Room Temperature Ionic Liquid/Water Mixtures: Water-Induced Structure. J. Phys. Chem. B 2012, 116, 1777–1787. [Google Scholar] [CrossRef]
- Jacquemin, J.; Husson, P.; Padua, A.A.H.; Majer, V. Density and Viscosity of Several Pure and Water-Saturated Ionic Liquids. Green Chem. 2006, 8, 172–180. [Google Scholar] [CrossRef]
- Rodriguez, H.; Brennecke, J.F. Temperature and Composition Dependence of the Density and Viscosity of Binary Mixtures of Water Plus Ionic Liquid. J. Chem. Eng. Data 2006, 51, 2145–2155. [Google Scholar] [CrossRef]
- Huddleston, J.G.; Visser, A.E.; Reichert, W.M.; Willauer, H.D.; Broker, G.A.; Rogers, R.D. Characterization and Comparison of Hydrophilic and Hydrophobic Room Temperature Ionic Liquids Incorporating the Imidazolium Cation. Green Chem. 2001, 3, 156–164. [Google Scholar] [CrossRef]
- Thomaz, J.E.; Lawler, CM; Fayeret, MDl. The influence of water on the alkyl region structure in variable chain length imidazolium-based ionic liquid/water mixtures. J. Phys. Chem. B 2016, 120, 10350–10357. [Google Scholar] [CrossRef]
- Wuebbenhorst, M.; van Turnhout, J. Analysis of complex dielectric spectra. i. one-dimensional derivative techniques and three-dimensional modelling. J. Non-Cryst. Solids. 2002, 305, 40–49. [Google Scholar] [CrossRef]
- Johari, G.P.; Goldstein, M. Viscous liquids and the glass transition. II. Secondary relaxations in glasses of rigid molecules. J. Chem. Phys. 1970, 53, 2372–2388. [Google Scholar] [CrossRef]
- Kastner, S.; Köhler, M.; Goncharov, Y.; Lunkenheimer, P.; Loidl, A. High-frequency dynamics of type-B glass formers investigated by broadband dielectric spectroscopy. J. Non-Cryst. Solids. 2011, 357, 510–514. [Google Scholar] [CrossRef]
- Lunkenheimer, P.; Bobnar, V.; Pronin, A.V.; Ritus, A.I.; Volkov, A.A.; Loidl, A. Origin of apparent colossal dielectric constants. Phys. Rev. B 2002, 66, 052105. [Google Scholar] [CrossRef]
- Havriliak, S.; Negami, S. A complex plane representation of dielectric and mechanical relaxation processes in some polymers. Polymer 1967, 8, 161–210. [Google Scholar] [CrossRef]
- Vogel, H. The temperature dependence law of the viscosity of fluids. Phys. Z 1921, 22, 645–646. [Google Scholar]
- Fulcher, G.S. Analysis of Recent Measurements of the Viscosity of Glasses. J. Am. Ceram. Soc. 1925, 8, 339–355. [Google Scholar] [CrossRef]
- Tammann, G; Hesse, W. Die Abhängigkeit der Viscosität von der Temperatur bie unterkühlten Flüssigkeiten. Z. Anorg. Allg. Chem. 1926, 156, 245–257. [Google Scholar] [CrossRef]
- Ito, N; Richert, R. Solvation dynamics and electric field relaxation in an imidazolium-PF6 ionic liquid: from room temperature to the glass transition. J. Phys. Chem. B. 2007, 111, 5016–5022. [Google Scholar] [CrossRef]
- Daguenet, C.; Dyson, J.P.; Krossing, I.; Oleinikova, A.; Slattery, J.; Wakai, C.; Weingaertner, H. Dielectric response of imidazolium-based room-temperature ionic liquids. J. Phys. Chem. B. 2006, 110, 12682–12688. [Google Scholar] [CrossRef]
- Nakamura, K.; Shikata, T. Systematic Dielectric and NMR Study of the Ionic Liquid 1-Alkyl-3-Methyl Imidazolium. ChemPhysChem 2010, 11, 285–294. [Google Scholar] [CrossRef] [PubMed]
- Angell, C.A. Strong and fragile liquids, in: K.L. Ngai, G.B.Wright (Eds.), Relaxations in Complex Systems, NRL, Washington DC 1985, pp. 3–11.
- Boehmer, R.; Ngai, K.L.; Angell, C.A.; Plazek, D.J. Nonexponential relaxations in strong and fragile glass formers. J. Chem. Phys. 1993, 99, 4201–4209. [Google Scholar] [CrossRef]
- Lunkenheimer P.; Loidl, A. Glassy Dynamics Beyond the α-Relaxation, in Broadband Dielectric Spectroscopy, F. Kremer and A. Schönhals, Eds., Berlin, Heidelberg: Springer, 2003, pp. 131–169.
- C. Gainaru, C.; Stacy, E.W.; Bocharova, V.; Gobet, M.; Holt, A.P.; Saito, T.; Greenbaum, S.; Sokolov, A.P. Mechanism of Conductivity Relaxation in Liquid and Polymeric Electrolytes: Direct Link between Conductivity and Diffusivity. J. Phys. Chem. B 2016, 120, 11074–11083. [Google Scholar] [CrossRef] [PubMed]
- Mizuno, F.; Belieres, J.P.; Kuwata, N.; Pradel, A.; Ribes, A.; Angell, C.A. Highly decoupled ionic and protonic solid electrolyte systems, in relation to other relaxing systems and their energy landscapes. J. Non-Cryst. Solids 2006, 352, 5147–5155. [Google Scholar] [CrossRef]
- Sippel, P.; Dietrich, V.; Reuter, D.; Aumüller, M.; Lunkenheimer, P.; Loidl, A.; Krohns, S. Impact of water on the charge transport of a glass-forming ionic liquid. Journal of Molecular Liquids 2016, 223 635–642. [Google Scholar] [CrossRef]
- Ueno, K.; Zhao, T.; Watanabe, M.; Angell, C.A. Protic ionic liquids based on decahydroisoquinoline: lost superfragility and ionicity-fragility correlation. Phys. Chem. B 2012, 116, 63–70. [Google Scholar] [CrossRef]
- Griffin, P.J.; Agapov, A.L.; Sokolov, A.P. Translation-rotation decoupling and nonexponentiality in room temperature ionic liquids. Phys. Rev. E 2012, 86, 021508. [Google Scholar] [CrossRef]
- Wojnarowska, Z.; Kołodziejczyk, K.; Paluch, K.J.; Tajber, L.; Grzybowska, K.; Ngai, K.L.; Paluch, M. Decoupling of conductivity relaxation from structural relaxation in protic ionic liquids and general properties. Phys. Chem. Chem. Phys. 2013, 15, 9205–9211. [Google Scholar] [CrossRef] [PubMed]
- Sippel, P.; Lunkenheimer, P.; Krohns, S.; Thoms, E.; Loidl, A. Importance of liquid fragility for energy applications of ionic liquids. Sci. Rep. 2015, 5, 13922. [Google Scholar] [CrossRef] [PubMed]
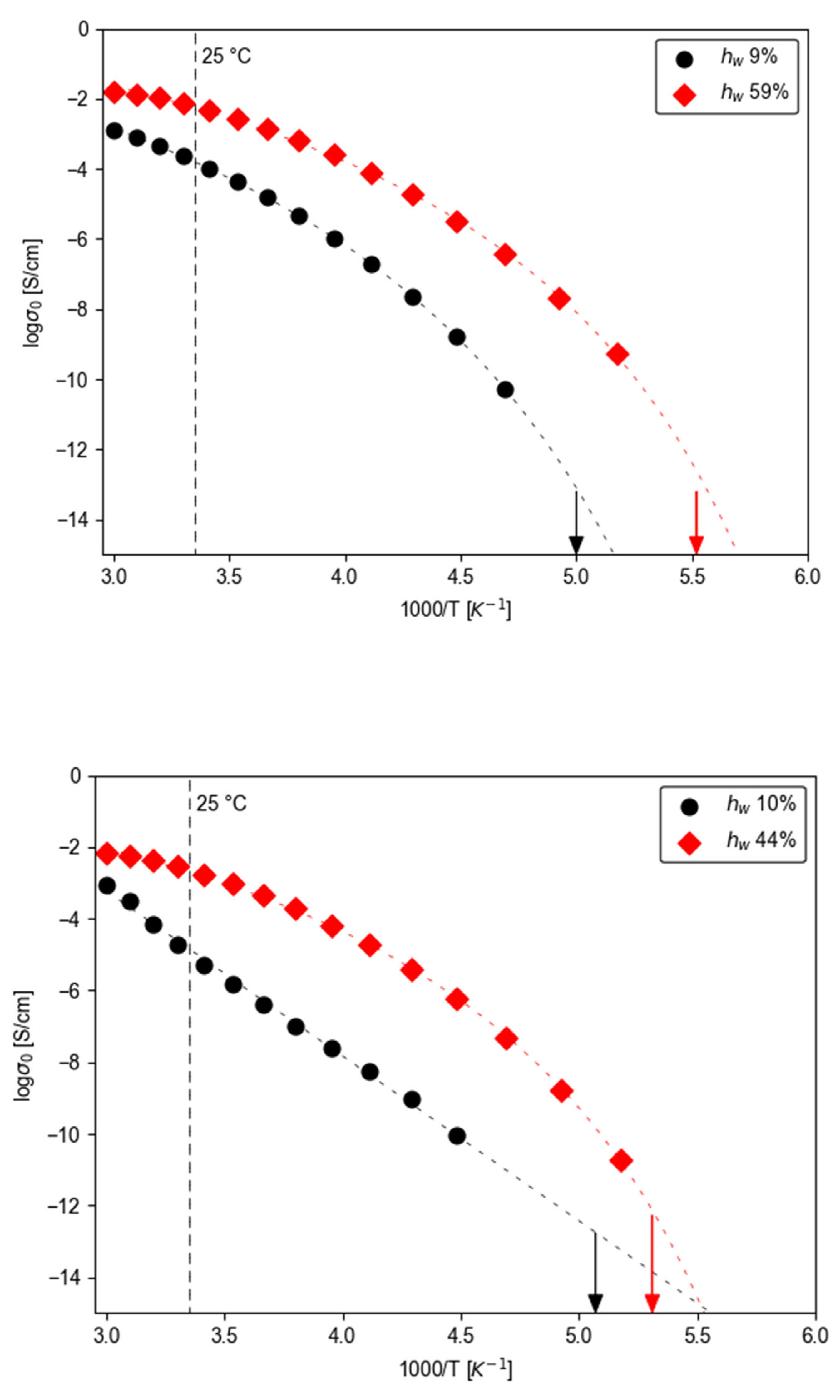
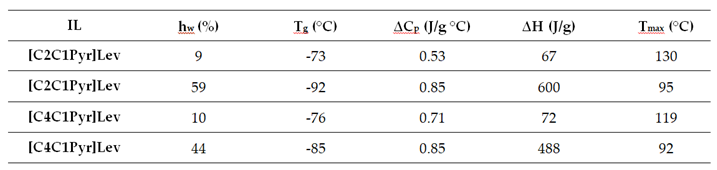 |
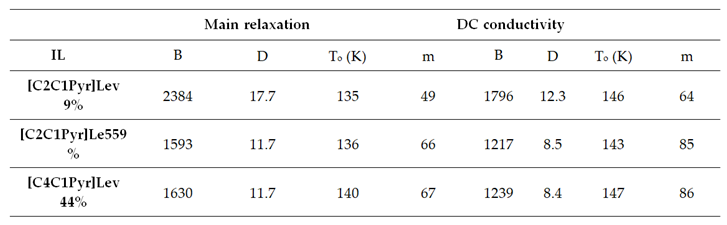 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





