Submitted:
04 March 2026
Posted:
06 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
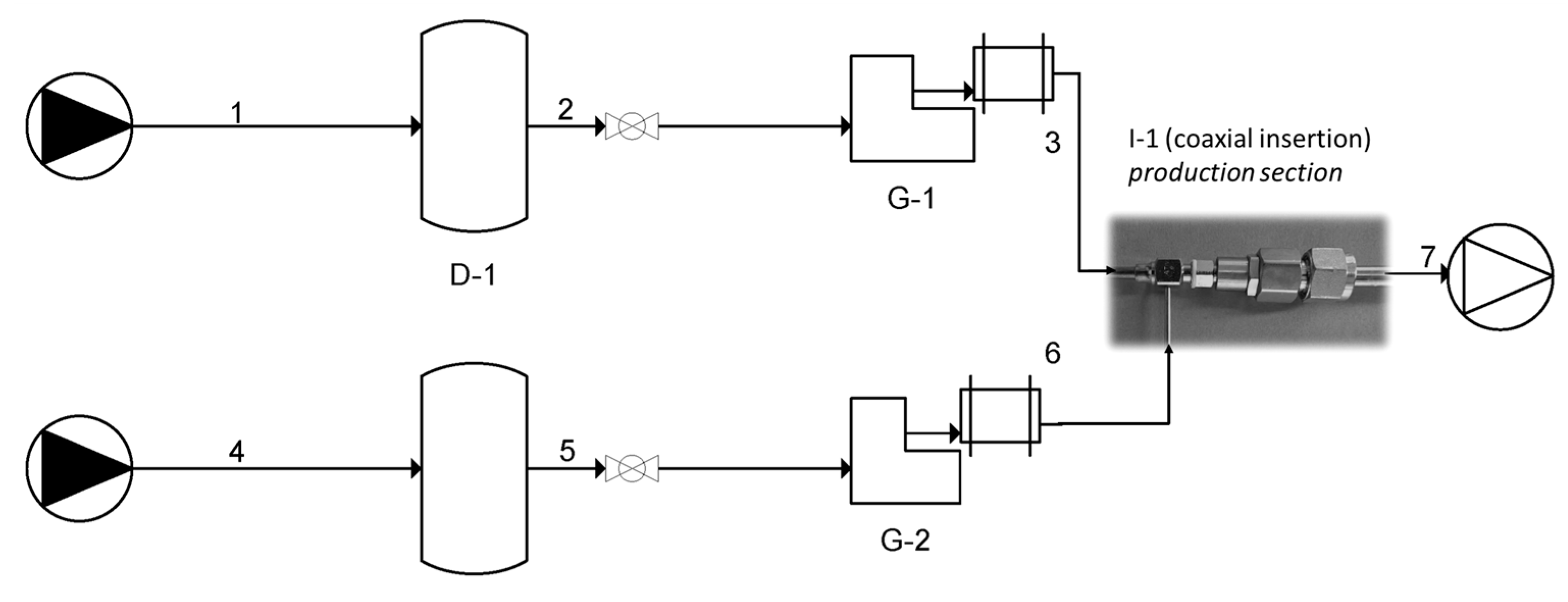
2.2. Setup and Fluido Dynamic Conditions for Invasomes Fabrication
2.2.1. Setup and Fluid Dynamic Insights
2.2.2. Invasomes Fabrication
2.3. Characterization Methods
2.3.1. Separation Steps
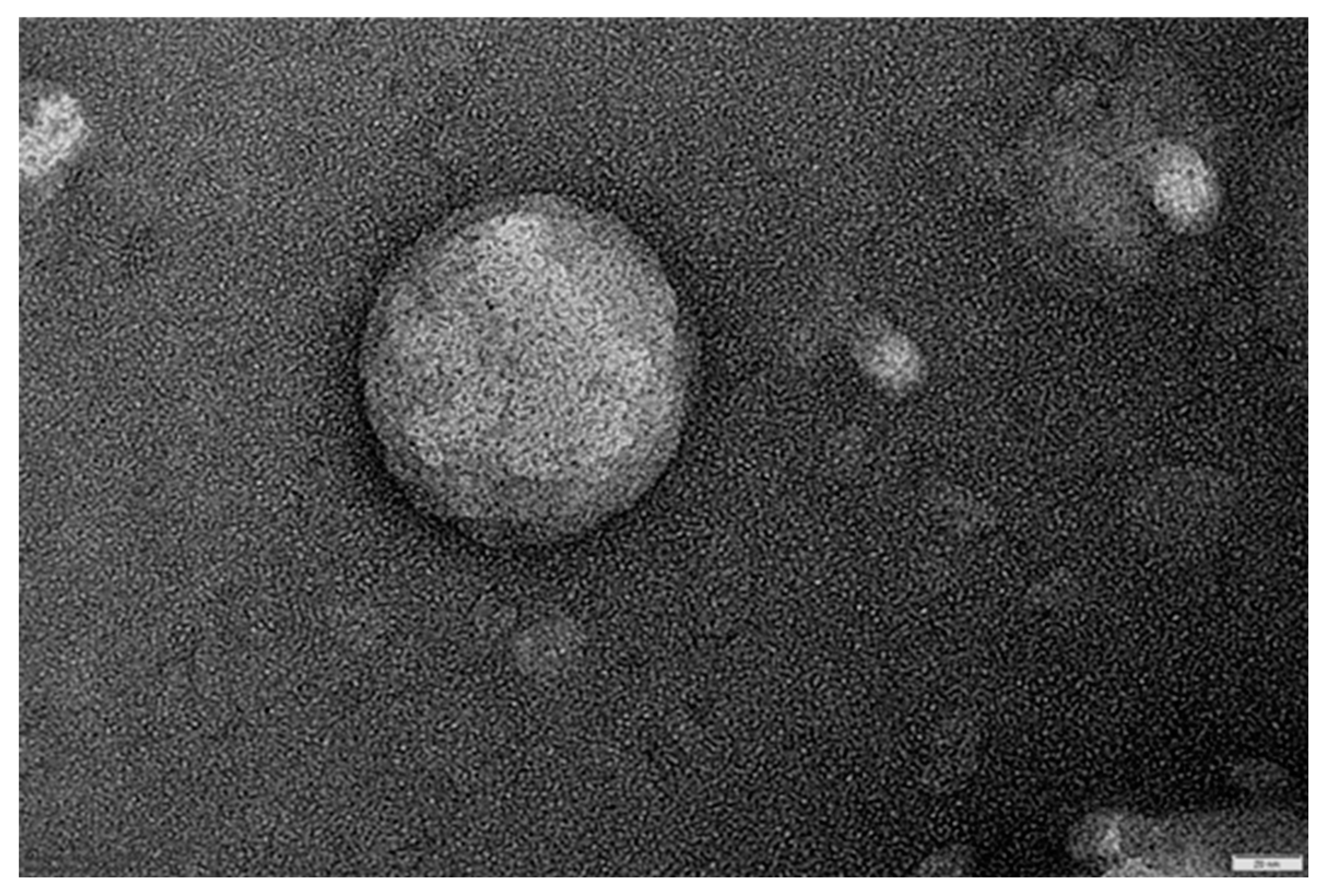
2.3.2. Vesicles Size, Superficial Charge and Morphology Inspections
2.3.3. Encapsulation Efficiency and Effective Load
2.3.4. Short- and Long-Term Stability
2.3.5. Sterile Productions Control and Antimicrobial Assays
2.3. Statistical Analysis
3. Results and Discussion
3.1. Fabrication of Unloaded Liposomes: Effect of Constituents
3.2. Invasomes Production
3.3.1. Effects of Theoretical High Load
3.3.2. Invasomes with Suitable Load
3.3.3. Fabrication Sterility Control
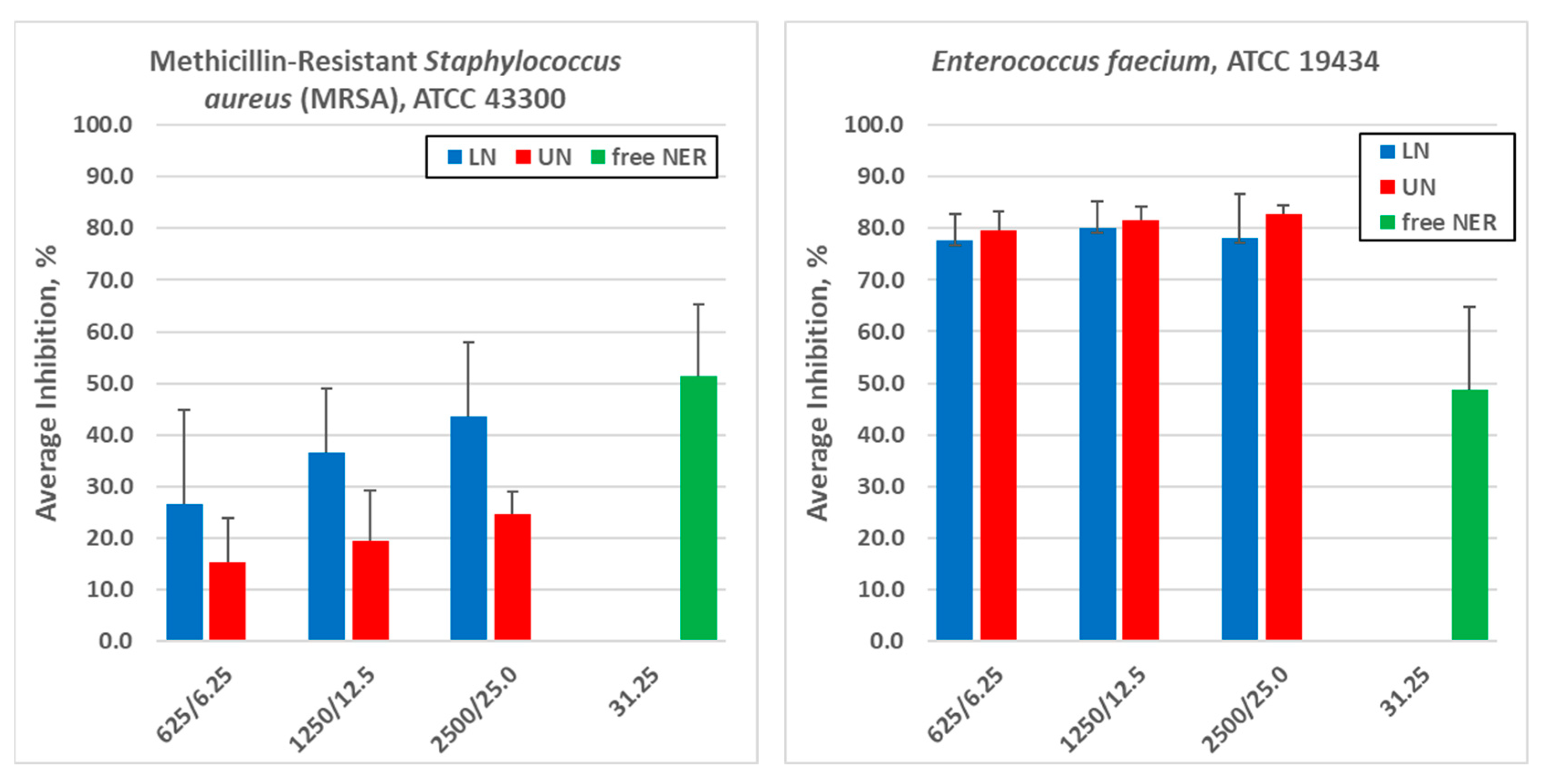
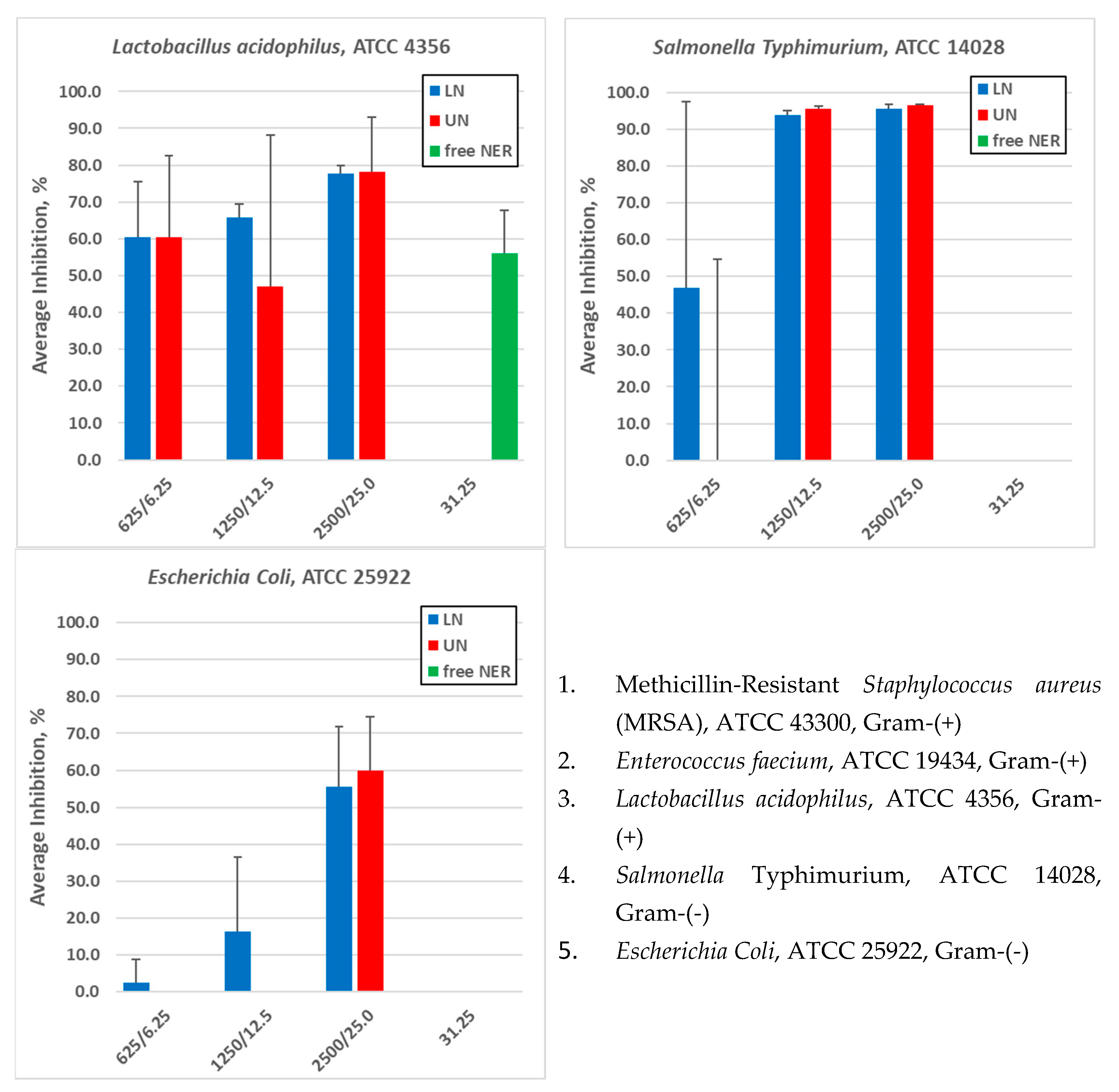
3.3.4. Antimicrobial Assays
- (i)
- antimicrobial responses are threshold-driven rather than linearly dose-dependent;
- (ii)
- meaningful interpretation requires separating the contribution of the lipid matrix from that of Nerolidol; and
- (iii)
- Nerolidol-loaded liposomes should be regarded as delivery platforms whose microbiological behavior depends on both components, rather than as inherently more potent antimicrobial agents. This perspective is essential for rationally designing future applications, particularly in contexts such as animal nutrition, where selectivity toward target microorganisms is critical.
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Rabiee, N.; Ahmadi, S.; Iravani, S.; Varma, R.S. Natural resources for sustainable synthesis of nanomaterials with anticancer applications: A move toward green nanomedicine. Environmental Research 2023, 216, 114803. [Google Scholar] [CrossRef]
- Wang, X.; Izzo, A.A.; Papapetropoulos, A.; Alexander, S.P.H.; Cortese-Krott, M.; Kendall, D.A.; Martemyanov, K.A.; Mauro, C.; Panettieri, R.A., Jr.; Patel, H.H.; et al. Natural product pharmacology: the British Journal of Pharmacology perspective. British Journal of Pharmacology 2024, 181, 3547–3555. [Google Scholar] [CrossRef]
- Theodoridis, S.; Drakou, E.G.; Hickler, T.; Thines, M.; Nogues-Bravo, D. Evaluating natural medicinal resources and their exposure to global change. The Lancet Planetary Health 2023, 7, e155–e163. [Google Scholar] [CrossRef]
- United Nations. THE 17 GOALS. Available online: https://sdgs.un.org/goals (accessed on 03/01/2026).
- Tian, Y.; Demirel, S.E.; Hasan, M.M.F.; Pistikopoulos, E.N. An overview of process systems engineering approaches for process intensification: State of the art. Chemical Engineering and Processing - Process Intensification 2018, 133, 160–210. [Google Scholar] [CrossRef]
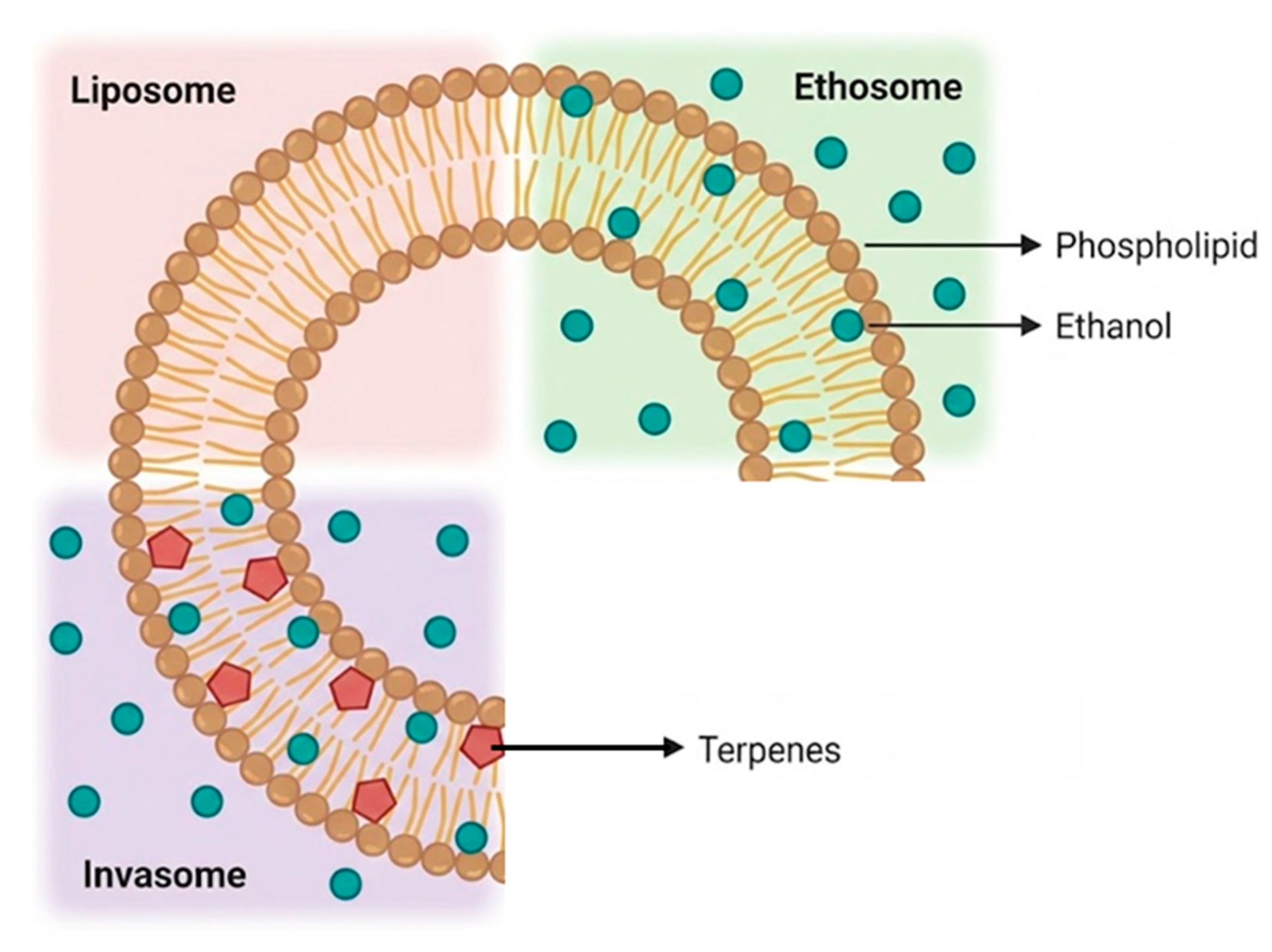
- Jain, S.; Tripathi, S.; Tripathi, P.K. Invasomes: Potential vesicular systems for transdermal delivery of drug molecules. Journal of Drug Delivery Science and Technology 2021, 61, 102166. [Google Scholar] [CrossRef]
- Tripathy, S.; Verma, D.K.; Gupta, A.K.; Srivastav, P.P.; Patel, A.R.; Chávez González, M.L.; Utama, G.L.; Aguilar, C.N. Nanoencapsulation of biofunctional components as a burgeoning nanotechnology-based approach for functional food development: A review. Biocatalysis and Agricultural Biotechnology 2023, 53, 102890. [Google Scholar] [CrossRef]
- Dalmoro, A.; Bochicchio, S.; Lamberti, G.; Bertoncin, P.; Janssens, B.; Barba, A.A. Micronutrients encapsulation in enhanced nanoliposomal carriers by a novel preparative technology. RSC Advances 2019, 9, 19800–19812. [Google Scholar] [CrossRef]
- Bochicchio, S.; Dalmoro, A.; Bertoncin, P.; Lamberti, G.; Moustafine, R.I.; Barba, A.A. Design and production of hybrid nanoparticles with polymeric-lipid shell–core structures: conventional and next-generation approaches. RSC Advances 2018, 8, 34614–34624. [Google Scholar] [CrossRef]
- Caccavo, D.; Lamberti, G.; Barba, A.A. Coaxial Injection Mixer for the Continuous Production of Nanoparticles. Chemical Engineering Transactions 2023, 100, 301–306. [Google Scholar] [CrossRef]
- De Piano, R.; Caccavo, D.; Lamberti, G.; Remaut, K.; Seynaeve, H.; Barba, A.A. A New Productive Approach and Formulative Optimization for Curcumin Nanoliposomal Delivery Systems. Pharmaceutics 2023, 15, 959. [Google Scholar] [CrossRef]
- Bochicchio, S.; Dalmoro, A.; Recupido, F.; Lamberti, G.; Barba, A.A. Nanoliposomes Production by a Protocol Based on a Simil-Microfluidic Approach. In Advances in Bionanomaterials: Selected Papers from the 2nd Workshop in Bionanomaterials, BIONAM 2016, October 4-7, 2016; Salerno, Italy, Piotto, S., Rossi, F., Concilio, S., Reverchon, E., Cattaneo, G., Eds.; Springer International Publishing: Cham, 2018; pp. 3–10. [Google Scholar]
- Chan, W.-K.; Tan, L.T.; Chan, K.-G.; Lee, L.-H.; Goh, B.-H. Nerolidol: A Sesquiterpene Alcohol with Multi-Faceted Pharmacological and Biological Activities. Molecules 2016, 21, 529. [Google Scholar] [CrossRef]
- Guillot, A.J.; Martínez-Navarrete, M.; Garrigues, T.M.; Melero, A. Skin drug delivery using lipid vesicles: A starting guideline for their development. Journal of Controlled Release 2023, 355, 624–654. [Google Scholar] [CrossRef]
- Babaie, S.; Bakhshayesh, A.R.D.; Ha, J.W.; Hamishehkar, H.; Kim, K.H. Invasome: A Novel Nanocarrier for Transdermal Drug Delivery. Nanomaterials 2020, 10, 341. [Google Scholar] [CrossRef]
- Gonçalves, O.; Pereira, R.; Gonçalves, F.; Mendo, S.; Coimbra, M.A.; Rocha, S.M. Evaluation of the mutagenicity of sesquiterpenic compounds and their influence on the susceptibility towards antibiotics of two clinically relevant bacterial strains. Mutation Research/Genetic Toxicology and Environmental Mutagenesis 2011, 723, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Vieira Nunes Cunha, I.; Machado Campos, A.; Passarella Gerola, A.; Caon, T. Effect of invasome composition on membrane fluidity, vesicle stability and skin interactions. International Journal of Pharmaceutics 2023, 646, 123472. [Google Scholar] [CrossRef] [PubMed]
- Kumar, B.; Pandey, M.; Aggarwal, R.; Sahoo, P.K. A comprehensive review on invasomal carriers incorporating natural terpenes for augmented transdermal delivery. Future Journal of Pharmaceutical Sciences 2022, 8, 50. [Google Scholar] [CrossRef]
- Kaltschmidt, B.P.; Ennen, I.; Greiner, J.F.W.; Dietsch, R.; Patel, A.; Kaltschmidt, B.; Kaltschmidt, C.; Hütten, A. Preparation of Terpenoid-Invasomes with Selective Activity against S. aureus and Characterization by Cryo Transmission Electron Microscopy. Biomedicines 2020, 8, 105. [Google Scholar] [CrossRef]
- Amnuaikit, T.; Limsuwan, T.; Khongkow, P.; Boonme, P. Vesicular carriers containing phenylethyl resorcinol for topical delivery system; liposomes, transfersomes and invasomes. Asian Journal of Pharmaceutical Sciences 2018, 13, 472–484. [Google Scholar] [CrossRef]
- Parvez, S.; Karole, A.; Mudavath, S.L. Fabrication, physicochemical characterization and In vitro anticancer activity of nerolidol encapsulated solid lipid nanoparticles in human colorectal cell line. Colloids and Surfaces B: Biointerfaces 2022, 215, 112520. [Google Scholar] [CrossRef] [PubMed]
- de Moura, D.F.; Rocha, T.A.; de Melo Barros, D.; da Silva, M.M.; dos Santos Santana, M.; Neta, B.M.; Cavalcanti, I.M.F.; Martins, R.D.; da Silva, M.V. Evaluation of the antioxidant, antibacterial, and antibiofilm activity of the sesquiterpene nerolidol. Archives of Microbiology 2021, 203, 4303–4311. [Google Scholar] [CrossRef]
- European Commission. Commission Implementing Regulation (EU) 2017/58 of 14 December 2016 concerning the authorisation of alpha-terpineol, nerolidol, 2-(4-methylphenyl) propan-2-ol, terpineol and linalyl acetate as feed additives for all animal species. 2017.
- Ephrem, E.; Najjar, A.; Charcosset, C.; Greige-Gerges, H. Use of free and encapsulated nerolidol to inhibit the survival of Lactobacillus fermentum in fresh orange juice. Food and Chemical Toxicology 2019, 133, 110795. [Google Scholar] [CrossRef] [PubMed]
- Yousefi, S.; Weisany, W.; Hosseini, S.E.; Ghasemlou, M. Mechanisms of nanoencapsulation to boost the antimicrobial efficacy of essential oils: A review. Food Hydrocolloids 2024, 150, 109655. [Google Scholar] [CrossRef]
- Babaie, S.; Taghvimi, A.; Charkhpour, M.; Zarebkohan, A.; Keyhanvar, P.; Hamishehkar, H. Optimization of Influential Variables in the Development of Buprenorphine and Bupivacaine Loaded Invasome for Dermal Delivery. Adv Pharm Bull 2021, 11, 522–529. [Google Scholar] [CrossRef]
- Liu, P.; Chen, G.; Zhang, J. A Review of Liposomes as a Drug Delivery System: Current Status of Approved Products, Regulatory Environments, and Future Perspectives. Molecules 2022, 27, 1372. [Google Scholar] [CrossRef]
- Chen, J.; Jiang, Q.-D.; Chai, Y.-P.; Zhang, H.; Peng, P.; Yang, X.-X. Natural Terpenes as Penetration Enhancers for Transdermal Drug Delivery. Molecules 2016, 21, 1709. [Google Scholar] [CrossRef]
- El-Kattan, A.F.; Asbill, C.S.; Kim, N.; Michniak, B.B. The effects of terpene enhancers on the percutaneous permeation of drugs with different lipophilicities. International Journal of Pharmaceutics 2001, 215, 229–240. [Google Scholar] [CrossRef]
- Zhou, X.; Hao, Y.; Yuan, L.; Pradhan, S.; Shrestha, K.; Pradhan, O.; Liu, H.; Li, W. Nano-formulations for transdermal drug delivery: A review. Chinese Chemical Letters 2018, 29, 1713–1724. [Google Scholar] [CrossRef]
- Mezzasalma, N.; Spadini, C.; Righi, F.; Simoni, M.; Lamberti, G.; Barba, A.A.; Greco, D.; Merelli, A.; Bosio, L.; Cupola, A.; et al. Evaluation of the antimicrobial and cytotoxic activity of nerolidol encapsulated in a nanoliposome system. Frontiers in Veterinary Science 2025, 12–2025. [Google Scholar] [CrossRef]
- Bochicchio, S.; Dalmoro, A.; De Simone, V.; Bertoncin, P.; Lamberti, G.; Barba, A.A. Simil-Microfluidic Nanotechnology in Manufacturing of Liposomes as Hydrophobic Antioxidants Skin Release Systems. Cosmetics 2020, 7, 22. [Google Scholar] [CrossRef]
- Lasic, D.D. Novel applications of liposomes. Trends in Biotechnology 1998, 16, 307–321. [Google Scholar] [CrossRef] [PubMed]
- Iannone, M.; Caccavo, D.; Barba, A.A.; Lamberti, G. A low-cost push–pull syringe pump for continuous flow applications. HardwareX 2022, 11. [Google Scholar] [CrossRef]
- Lu, J.; Frank, E.L. Rapid HPLC Measurement of Thiamine and Its Phosphate Esters in Whole Blood. Clinical Chemistry 2008, 54, 901–906. [Google Scholar] [CrossRef]
- Pavan, B.; Dalpiaz, A.; Marani, L.; Beggiato, S.; Ferraro, L.; Canistro, D.; Paolini, M.; Vivarelli, F.; Valerii, M.C.; Comparone, A.; et al. Geraniol Pharmacokinetics, Bioavailability and Its Multiple Effects on the Liver Antioxidant and Xenobiotic-Metabolizing Enzymes. Frontiers in Pharmacology 2018, 9–2018. [Google Scholar] [CrossRef]
- Mazhar, A.; El-Hansi, N.S.; Shafaa, M.W.; Shalaby, M.S. Radiation sterilization of liposomes: A literature review. Radiation Physics and Chemistry 2024, 218, 111592. [Google Scholar] [CrossRef]
- Delma, K.L.; Lechanteur, A.; Evrard, B.; Semdé, R.; Piel, G. Sterilization methods of liposomes: Drawbacks of conventional methods and perspectives. International Journal of Pharmaceutics 2021, 597, 120271. [Google Scholar] [CrossRef]
- Mineart, K.P.; Kelley, E.G. Chapter 19 - Size reduction, purification, sterilization and storage/packaging of liposomes. In Liposomes in Drug Delivery; Antimisiaris, S.G., Ed.; Academic Press, 2024; pp. 489–515. [Google Scholar]
- Dubraska V. Diaz-Campos, D., PhD; Brian V. Lubbers, D., PhD, DACVCP; Stefan Schwarz, D.; Robert Bowden, B.; Claire R. Burbick, D., PhD, DACVM; Merran Govendir, P., BVSc, FHERDSA, MANZCVSc; Scott B. Killian, B.; Sara D. Lawhon, D., PhD, DACVM; Xian-Zhi Li, P.; Kelli Maddock, M., MLS(ASCP)M; et al. CLSI VET01S. Performance Standards for Antimicrobial Disk and Dilution Susceptibility Tests for Bacteria Isolated From Animals. 2024.
- Kaddah, S.; Khreich, N.; Kaddah, F.; Charcosset, C.; Greige-Gerges, H. Cholesterol modulates the liposome membrane fluidity and permeability for a hydrophilic molecule. Food and Chemical Toxicology 2018, 113, 40–48. [Google Scholar] [CrossRef]
- Lakshmi, P.K.K., B.; Prasanthi, D. Invasomes-novel Vesicular Carriers for Enhanced Skin Permeation. Systematic Reviews in Pharmacy 2013, 4, 26–30. [Google Scholar] [CrossRef]
- Emanet, M.; Ciofani, G. Ethosomes as Promising Transdermal Delivery Systems of Natural-Derived Active Compounds. Advanced NanoBiomed Research 2023, 3, 2300020. [Google Scholar] [CrossRef]
- Verma, P.; Pathak, K. Therapeutic and cosmeceutical potential of ethosomes: An overview. Journal of Advanced Pharmaceutical Technology & Research 2010, 1. [Google Scholar] [CrossRef]
- Bernal-Chávez, S.A.; Del Prado-Audelo, M.L.; Caballero-Florán, I.H.; Giraldo-Gomez, D.M.; Figueroa-Gonzalez, G.; Reyes-Hernandez, O.D.; González-Del Carmen, M.; González-Torres, M.; Cortés, H.; Leyva-Gómez, G. Insights into Terminal Sterilization Processes of Nanoparticles for Biomedical Applications. Molecules 2021, 26, 2068. [Google Scholar] [CrossRef]
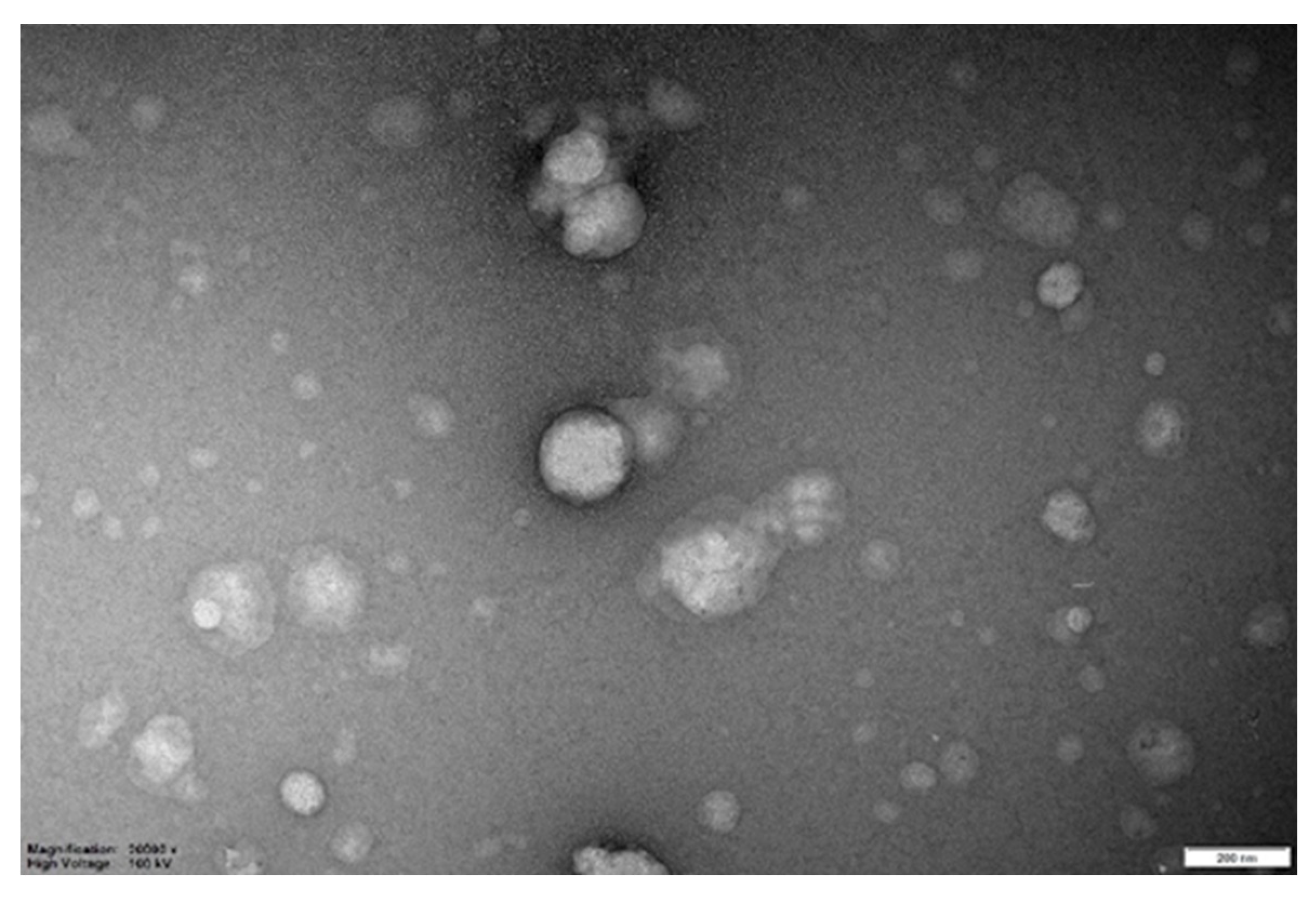

| Used delivery system | NER enhancer to active ingredient | References |
|---|---|---|
| Hydrogel | Curcumin | [6] |
| Hydrogel | Hydrocortisone Hydrochloride Carbamazepine Tamoxifen |
[18,29] |
| Hydrogel | Selegiline hydrochloride | [18] |
| Hydrogel | Terbinafine | [28] |
| Chitosan gel | Ondansetron hydrochloride | [28] |
| Invasome | Buprenorphine hydrochloride Bupivacaine |
[26] |
| Hydrogel | Hydrocortisone | [28] |
| Hydrogel | Propanolol hydrochloride | [28,30] |
| PG gel | Lomerizine dihydrochloride | [28] |
| Property | Value |
|---|---|
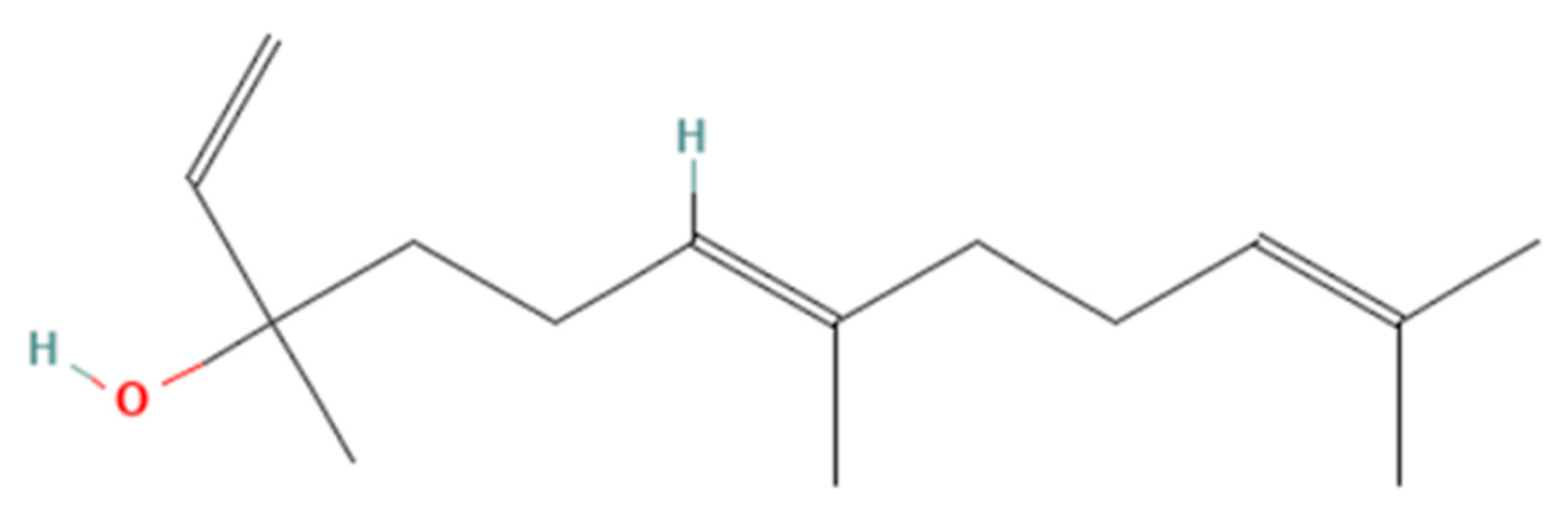
| Molecular Formula | C15H26O |
| State | Liquid |
| Molecular weight (g/mol) | 222.37 |
| CAS number | 7212-44-4 |
| Water solubility (mg/L) (20°C) | 14.1* [BASF Safety Data Sheet] |
| Melting Point (°C) (1 bar) | -90 [BASF Safety Data Sheet] |
| Boiling Point (°C) (1 bar) | 276 [BASF Safety Data Sheet] |
| Log P | 4.68 [BASF Safety Data Sheet] |
| Solubility | high in ethanol - low in water |
| Topological surface area (Å) | 20.23 |
| Van der Waals Molecular volume (Å3/molecule) | 268.93 |
| *1.532 (25°C) Experimental (https://hmdb.ca/metabolites/HMDB0035662) | |
| Batch # | NER, (g) | Theoretical Load, % |
|---|---|---|
| Prod. 1 (Unl. LIP) | 0 | 0 |
| INV. NER, 1% | 0.0285 | 1 |
| INV. NER, 5% | 0.1480 | 5 |
| INV. NER, 10% | 0.3133 | 10 |
| INV. NER, 15% | 0.4980 | 15 |
| Batch # | NER, (g) | Theoretical Load, % |
|---|---|---|
| INV. NER, 1% | 0.0285 | 1 |
| INV. NER, 2% | 0.0576 | 2 |
| Produced batch, # | Z-Average [nm] | PDI | Turbidity [NTU] |
|---|---|---|---|
| Prod. 1 (Unl. LIP) | 305.08 ± 24.2 | 0.33 ± 0.03 | 318 |
| INV. NER, 1% | 309.99 ± 9.0 | 0.34 ± 0.00 | 238 |
| INV. NER, 5% | 604.00 ± 55.2 | 0.6 ± 0.07 | 589 |
| INV. NER, 10% | 697.00 ± 32.4 | 0.62 ± 0.04 | 912 |
| INV. NER, 15% | 981.00 ± 196.6 | 0.59 ± 0.04 | 1238 |
| Produced batch | Effective load [%] | Efficiency [%] |
|---|---|---|
| INV. NER, 1% | 1.00 ± 0.01 | 99.70 ± 0.42 |
| INV. NER, 5% | 4.93 ± 0.02 | 99.09 ± 1.00 |
| INV. NER, 10% | 9.92 ± 0.08 | 99.27 ± 0.90 |
| INV. NER, 15% | 14.98 ± 0.007 | 99.76 ± 0.27 |
| Produced Batch | Z-Average [nm] | PDI | Z-Average [nm]* | PDI* |
|---|---|---|---|---|
| INV. NER, 1% | 309.99 ± 9.03 | 0.34 ± 0.00 | 329.6 ± 8.99 | 0.69 ± 0.13 |
| INV. NER, 5% | 604.10 ± 55.0 | 0.60 ± 0.07 | 467.7 ± 6.58 | 0.59 ± 0.07 |
| INV. NER, 10% | 697.05 ± 32.5 | 0.62 ± 0.04 | 372.0 ± 20.55 | 0.87 ± 0.03 |
| INV. NER, 15% | 981.00 ± 196.6 | 0.59 ± 0.04 | 477.9 ± 75.56 | 0.84 ± 0.05 |
| Produced Batch* | Z-Average, [nm] | PDI | Z-Potential, [mV] | Effective Load, [%] | Efficiency [%] |
|---|---|---|---|---|---|
| INV. NER, 1% |
298.8 ± 9.84 | 0.489 ± 0.097 | -42.36 ± 5.4 | 1.190 | 100 |
| INV. NER, 2% |
301.8 ± 19.99 | 0.605 ± 0.106 | -52.18 ± 6.4 | 2.02 | 99.34 |
| INV. NER, 1% Aged | 341.7 ± 9.98 | 0.64 ± 0.04 | -49.81 ± 0.8 | 1.08 ± 0.05 | 90.7 |
| INV. NER, 2% Aged |
342.8 ± 8.22 | 0.60 ± 0.11 | -51.41 ± 0.89 | 1.44 ± 0.11 | 71.1 |
| *Since for both the loading ratio the p-values are larger than 0.05, the aging process does not produce any statistical change. | |||||
| Strain | ATCC | Gram | Killing effects | ||
|---|---|---|---|---|---|
| LN | UN | free NER | |||
| Staphylococcus aureus (MRSA) | 43300 | (+) | Yes, dose dependent | Yes, dose dependent | Yes |
| Enterococcus faecium | 19434 | (+) | Yes, slightly dose dependent | Yes, slightly dose dependent | Yes |
| Lactobacillus acidophilus | 4356 | (+) | Yes, dose independent | Yes, dose independent | Yes |
| Salmonella Typhimurium | 14028 | (-) | Yes, slightly dose dependent | Not for all concentrations | No |
| Escherichia Coli | 25922 | (-) | Yes, dose dependent | Not for all concentrations | No |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




