Submitted:
04 March 2026
Posted:
04 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Historical Context and Evolution of Biomaterials Design
1.2. Why Plants? Evolutionary and Functional Rationale for Plant-Based Bioactive Molecules
1.3. Scope and Organization of This Review
2. Natural Plant-Derived Polysaccharides: Structural Diversity, Bioactive Properties, and Therapeutic Applications
2.1. Marine Polysaccharides: Seaweed-Derived Compounds
2.1.1. Fucoidan: Multifunctional Sulfated Polysaccharide with Translational Potential
2.1.2. Alginate, Carrageenan and Ulvan: Structurally Robust Polysaccharides with Distinct Immunological Roles
2.2. Terrestrial Plant Polysaccharides: Traditional Knowledge and Modern Immunoengineering
2.2.1. Lycium barbarum Polysaccharides: Immunomodulation and Microbiota Interactions
2.2.2. Acemannan from Aloe vera: Structure-Dependent Immunoregulation
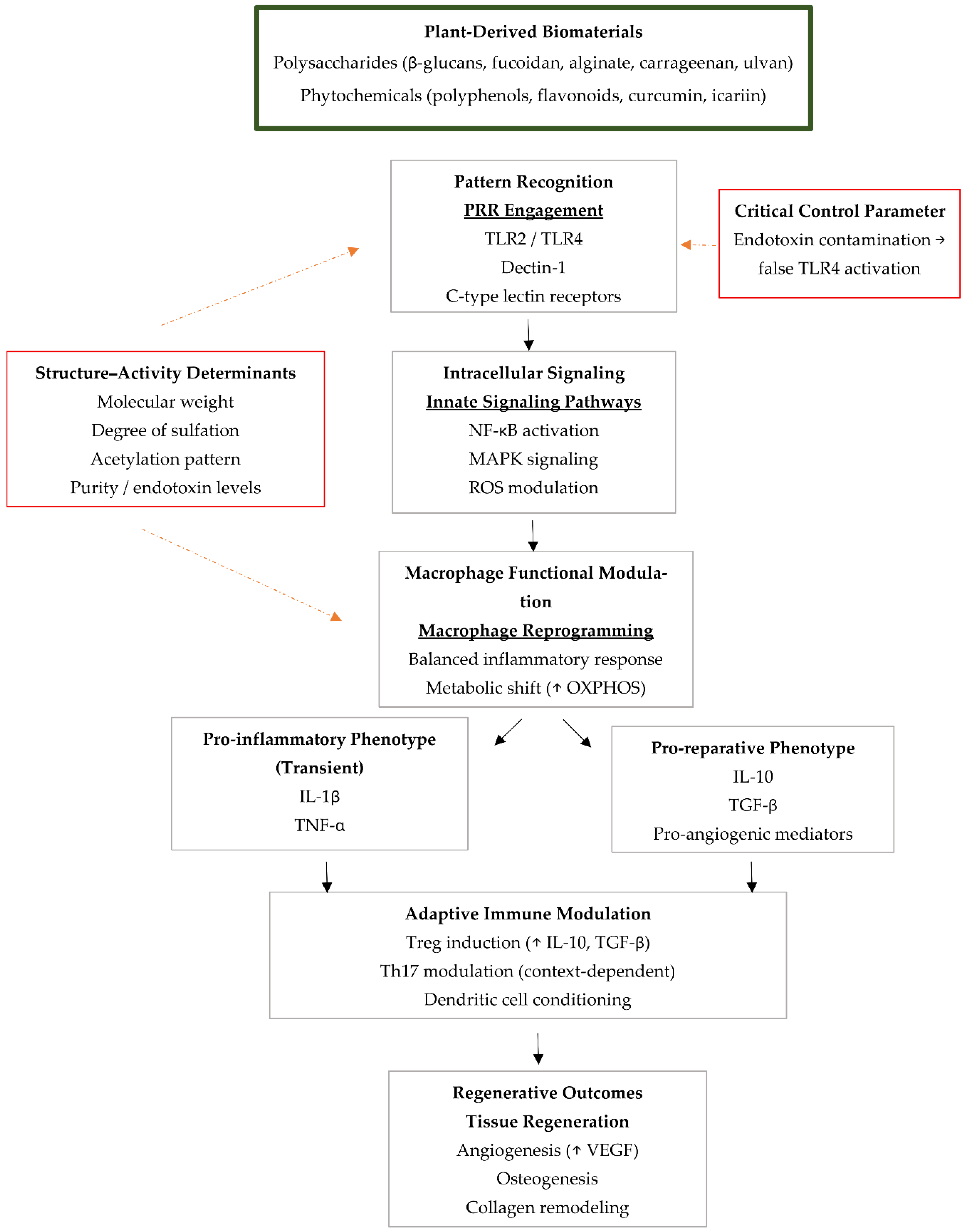
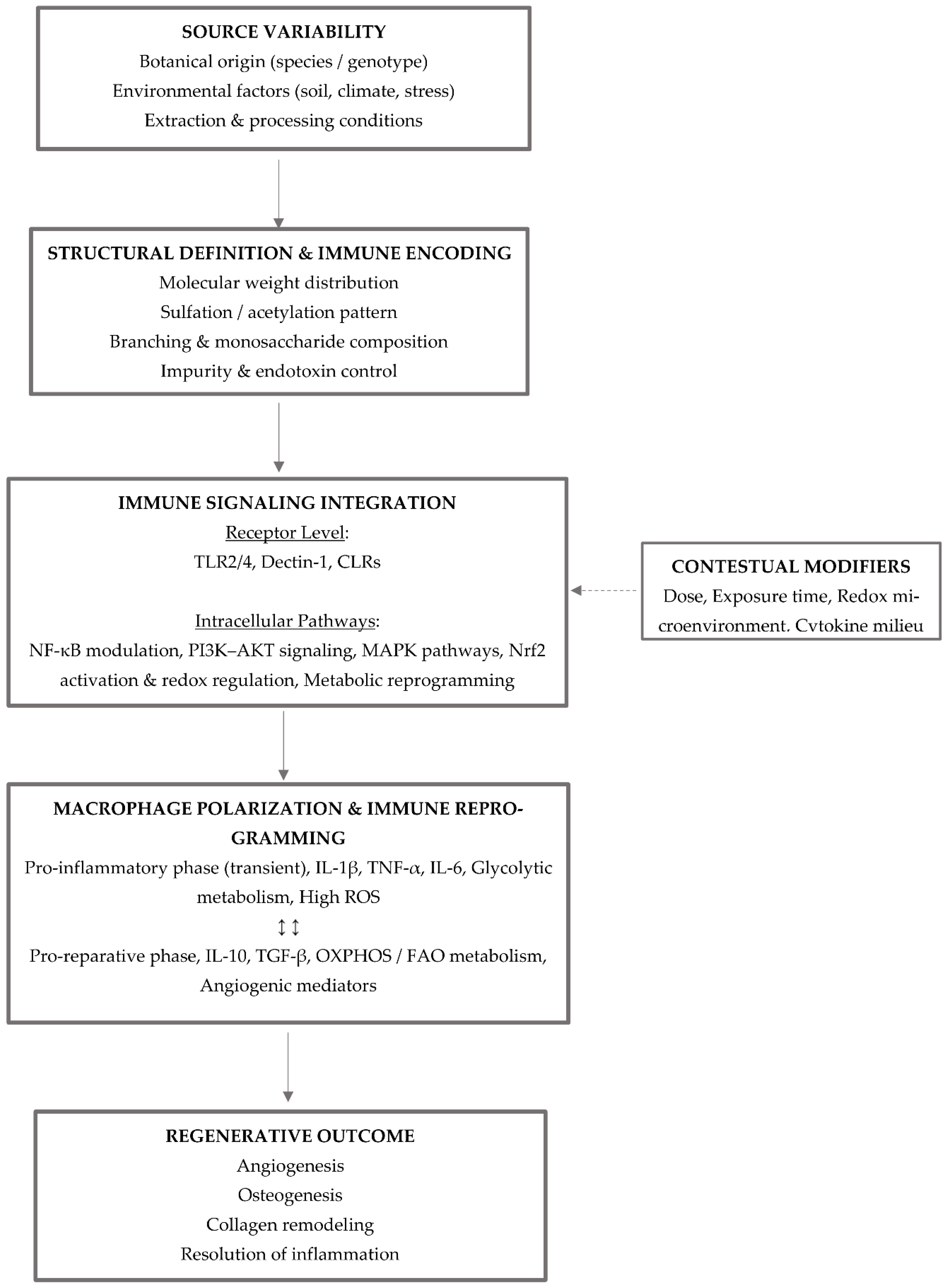
2.3. Structural Determinants and Chemical Modification of Plant Polysaccharides
2.3.1. Physicochemical Parameters Governing Bioactivity
2.3.2. Chemical Modification Strategies for Immunoengineering
3. Plant-Based Phytochemicals and Bioactive Secondary Metabolites: Molecular Mechanisms in Immune Regulation
3.1. Polyphenolic Compounds: From Plant Defense to Therapeutic Agents
3.1.1. Polyphenols in Bone Biology and Immunoregulation
3.1.2. Curcumin: A Model Polyphenol for Immunomodulatory Biomaterials
3.1.3. Icariin: A Multifunctional Flavonoid for Regenerative Immunomodulation
3.2. Plant Lectins: Carbohydrate-Binding Proteins as Immune Recognition Modules
3.3. Phytochemicals in Complex Wound Repair: Integrated Immunomodulatory Pathways
3.3.1. Multicomponent Regulation of Chronic and Diabetic Wounds
3.3.2. Reactive Oxygen Species Modulation as a Central Therapeutic Axis
4. Macrophage Polarization and Immunomodulation: The Central Axis of Bioinstructive Biomaterials
4.1. M1/M2 Macrophage Polarization: A Functional Spectrum Rather Than a Binary State
4.1.1. Macrophage Plasticity and Phenotypic Continuum
4.1.2. Dysregulated Polarization in Pathological Contexts
4.2. Molecular Mechanisms of Macrophage Reprogramming
4.2.1. Pattern Recognition Receptor Engagement and Immune Signal Encoding
4.2.2. Redox Regulation and Metabolic Reprogramming
4.2.3. Integration of PI3K/AKT, NF-κB, and MAPK Signaling Pathways
4.3. Adaptive Immune Modulation and Immune Tolerance
4.3.1. Regulation of Th17/Treg Balance
4.3.2. Tolerogenic Dendritic Cells and Biomaterial-Guided Immune Reprogramming
5. Plant-Based Biomaterials in Tissue Engineering: Tissue-Specific Applications and Mechanistic Considerations
5.1. Wound Healing: From Acute Repair to Chronic Wound Management
5.1.1. Immune Orchestration During Physiological Wound Healing
5.1.2. Advanced Immunomodulatory Hydrogel Systems for Wound Healing
5.1.3. Plant-Derived Extracellular Vesicles in Wound Repair
5.2. Bone Tissue Engineering and Osteoimmunology
5.3. Cartilage and Neural Tissue Engineering
6. Translational Challenges and Pathways Toward Clinical Application
6.1. Advanced Delivery Systems and Bioavailability Enhancement
6.2. Ethical and Regulatory Considerations in Plant-Based Biomaterial Development
6.2.1. Ethnopharmacology, Indigenous Knowledge, and Ethical Translation
6.2.2. Standardization, GMP Compliance, and Regulatory Pathways
6.3. Evidence Quality, Reproducibility, and “Regulatory Readiness”: A Critical Appraisal
| Category | Minimum Requirement | Rationale | Key References |
|---|---|---|---|
| Chemical Characterization | Full compositional profiling (HPLC/MS/NMR); MW distribution; substitution degree | Link structure to function | [224,225,231] |
| Endotoxin Control | LAL or monocyte activation test; defined thresholds | Prevent false PRR activation | [150,181] |
| Immune Profiling | Multiplex cytokine panels; time-course studies; functional macrophage assays | Capture dynamic immune modulation | [134,232] |
| Mechanistic Validation | PRR blocking; receptor engagement assays | Demonstrate causality | [26,143,145] |
| Human-Relevant Models | Primary human macrophages; tissue-relevant co-culture | Improve translational validity | [147,229] |
| EV Characterization (if applicable) | Size distribution; marker profiling; purity assessment | Standardized EV reporting | [191] |
| Stability & Batch Comparability | Cross-batch chemical and biological consistency | Regulatory compliance | [214,226,231] |
7. Conclusion and Future Perspectives
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hench, L.L. Biomaterials. Science 1980, 208, 826–831. [Google Scholar] [CrossRef] [PubMed]
- Rea, S.M.; Brooks, R.A.; Schneider, A.; Best, S.M.; Bonfield, W. Osteoblast-like Cell Response to Bioactive Composites—Surface-topography and Composition Effects. J. Biomed. Mater. Res. B Appl. Biomater. 2004, 70B, 250–261. [Google Scholar] [CrossRef]
- Hench, L.L.; Thompson, I. Twenty-First Century Challenges for Biomaterials. J. R. Soc. Interface 2010, 7. [Google Scholar] [CrossRef] [PubMed]
- Hench, L.L.; Wilson, J. Clinical Performance of Skeletal Prostheses; Springer, 1996; ISBN 0-412-72110-4. [Google Scholar]
- Wink, M. Plant Secondary Metabolism: Diversity, Function and Its Evolution. Nat. Prod. Commun. 2008, 3, 1934578X0800300801. [Google Scholar] [CrossRef]
- Dixon, R.A.; Paiva, N.L. Stress-Induced Phenylpropanoid Metabolism. Plant Cell 1995, 1085–1097. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.D.G.; Dangl, J.L. The Plant Immune System. Nature 2006, 444, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Medzhitov, R. Recognition of Microorganisms and Activation of the Immune Response. Nature 2007, 449, 819–826. [Google Scholar] [CrossRef]
- Spelman, K.; Burns, J.; Nichols, D.; Winters, N.; Ottersberg, S.; Tenborg, M. Modulation of Cytokine Expression by Traditional Medicines: A Review of Herbal Immunomodulators. Altern. Med. Rev. 2006, 11, 128. [Google Scholar]
- Hopkins, A.L. Network Pharmacology: The next Paradigm in Drug Discovery. Nat. Chem. Biol. 2008, 4, 682–690. [Google Scholar] [CrossRef]
- Wagner, H.; Ulrich-Merzenich, G. Synergy Research: Approaching a New Generation of Phytopharmaceuticals. Phytomedicine 2009, 16, 97–110. [Google Scholar] [CrossRef]
- Gertsch, J. Botanical Drugs, Synergy, and Network Pharmacology: Forth and Back to Intelligent Mixtures. Planta Med. 2011, 77, 1086–1098. [Google Scholar] [CrossRef] [PubMed]
- Eming, S.A.; Wynn, T.A.; Martin, P. Inflammation and Metabolism in Tissue Repair and Regeneration. Science 2017, 356, 1026–1030. [Google Scholar] [CrossRef]
- Heinrich, M.; Barnes, J.; Gibbons, S.; Williamson, E. Fundamentals of Pharmacognosy and Phytotherapy. 2nd. Ed. Edinb. Lond. N. Y. Churchhill Livingstone Elsevier 2012.
- Fabricant, D.S.; Farnsworth, N.R. The Value of Plants Used in Traditional Medicine for Drug Discovery. Environ. Health Perspect. 2001, 109, 69–75. [Google Scholar] [CrossRef]
- Atanasov, A.G.; Waltenberger, B.; Pferschy-Wenzig, E.-M.; Linder, T.; Wawrosch, C.; Uhrin, P.; Temml, V.; Wang, L.; Schwaiger, S.; Heiss, E.H.; et al. Discovery and Resupply of Pharmacologically Active Plant-Derived Natural Products: A Review. Biotechnol. Adv. 2015, 33, 1582–1614. [Google Scholar] [CrossRef]
- Laurienzo, P. Marine Polysaccharides in Pharmaceutical Applications: An Overview. Mar. Drugs 2010, 8, 2435–2465. [Google Scholar] [CrossRef] [PubMed]
- Cotas, J.; Lomartire, S.; Gonçalves, A.M.; Pereira, L. From Ocean to Medicine: Harnessing Seaweed’s Potential for Drug Development. Int. J. Mol. Sci. 2024, 25, 797. [Google Scholar] [CrossRef] [PubMed]
- Mano, J.F.; Silva, G.A.; Azevedo, H.S.; Malafaya, P.B.; Sousa, R.A.; Silva, S.S.; Boesel, L.F.; Oliveira, J.M.; Santos, T.C.; Marques, A.P.; et al. Natural Origin Biodegradable Systems in Tissue Engineering and Regenerative Medicine: Present Status and Some Moving Trends. J. R. Soc. Interface 2007, 4, 999–1030. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.Y.; Mooney, D.J. Alginate: Properties and Biomedical Applications. Prog. Polym. Sci. 2012, 37, 106–126. [Google Scholar] [CrossRef]
- Brown, G.D.; Gordon, S. Immune Recognition of Fungal β-Glucans: Immune Recognition of Fungal β-Glucans. Cell. Microbiol. 2005, 7, 471–479. [Google Scholar] [CrossRef]
- Schepetkin, I.A.; Quinn, M.T. Botanical Polysaccharides: Macrophage Immunomodulation and Therapeutic Potential. Int. Immunopharmacol. 2006, 6, 317–333. [Google Scholar] [CrossRef]
- Jayakumar, R.; Prabaharan, M.; Sudheesh Kumar, P.T.; Nair, S.V.; Tamura, H. Biomaterials Based on Chitin and Chitosan in Wound Dressing Applications. Biotechnol. Adv. 2011, 29, 322–337. [Google Scholar] [CrossRef] [PubMed]
- Pomin, V.H. Structure–Function Relationship of Anticoagulant and Antithrombotic Well-Defined Sulfated Polysaccharides from Marine Invertebrates. In Advances in Food and Nutrition Research; Elsevier, 2012; Vol. 65, pp. 195–209. ISBN 978-0-12-416003-3. [Google Scholar]
- Fitton, J.H. Therapies from Fucoidan; Multifunctional Marine Polymers. Mar. Drugs 2011, 9, 1731–1760. [Google Scholar] [CrossRef]
- Takeuchi, O.; Akira, S. Pattern Recognition Receptors and Inflammation. Cell 2010, 140, 805–820. [Google Scholar] [CrossRef]
- Chen, L.; Huang, G. Antitumor Activity of Polysaccharides: An Overview. Curr. Drug Targets 2018, 19. [Google Scholar] [CrossRef]
- Rioux, L.-E.; Turgeon, S.L.; Beaulieu, M. Structural Characterization of Laminaran and Galactofucan Extracted from the Brown Seaweed Saccharina Longicruris. Phytochemistry 2010, 71, 1586–1595. [Google Scholar] [CrossRef]
- Holdt, S.L.; Kraan, S. Bioactive Compounds in Seaweed: Functional Food Applications and Legislation. J. Appl. Phycol. 2011, 23, 543–597. [Google Scholar] [CrossRef]
- Pomin, V.H. Sulfated Glycans in Inflammation. Eur. J. Med. Chem. 2015, 92, 353–369. [Google Scholar] [CrossRef] [PubMed]
- Luthuli, S.; Wu, S.; Cheng, Y.; Zheng, X.; Wu, M.; Tong, H. Therapeutic Effects of Fucoidan: A Review on Recent Studies. Mar. Drugs 2019, 17, 487. [Google Scholar] [CrossRef] [PubMed]
- Fitton, H.; Meyers, S.; Brooks, L.; Mulder, A.; Rolfe, M.; Baker, D.; Robinson, S. Effects of Fucoidan from Fucus Vesiculosus in Reducing Symptoms of Osteoarthritis: A Randomized Placebo-Controlled Trial. Biol. Targets Ther. 2016, 81. [Google Scholar] [CrossRef]
- Fitton, J.; Stringer, D.; Karpiniec, S. Therapies from Fucoidan: An Update. Mar. Drugs 2015, 13, 5920–5946. [Google Scholar] [CrossRef]
- Senthilkumar, K.; Manivasagan, P.; Venkatesan, J.; Kim, S.-K. Brown Seaweed Fucoidan: Biological Activity and Apoptosis, Growth Signaling Mechanism in Cancer. Int. J. Biol. Macromol. 2013, 60, 366–374. [Google Scholar] [CrossRef]
- Yang, J.-Y.; Lim, S.Y. Fucoidans and Bowel Health. Mar. Drugs 2021, 19, 436. [Google Scholar] [CrossRef]
- Pozharitskaya, O.N.; Shikov, A.N.; Obluchinskaya, E.D.; Vuorela, H. The Pharmacokinetics of Fucoidan after Topical Application to Rats. Mar. Drugs 2019, 17, 687. [Google Scholar] [CrossRef]
- Apostolova, E.; Lukova, P.; Baldzhieva, A.; Katsarov, P.; Nikolova, M.; Iliev, I.; Peychev, L.; Trica, B.; Oancea, F.; Delattre, C.; et al. Immunomodulatory and Anti-Inflammatory Effects of Fucoidan: A Review. Polymers 2020, 12, 2338. [Google Scholar] [CrossRef]
- Cumashi, A.; Ushakova, N.A.; Preobrazhenskaya, M.E.; D’Incecco, A.; Piccoli, A.; Totani, L.; Tinari, N.; Morozevich, G.E.; Berman, A.E.; Bilan, M.I.; et al. A Comparative Study of the Anti-Inflammatory, Anticoagulant, Antiangiogenic, and Antiadhesive Activities of Nine Different Fucoidans from Brown Seaweeds. Glycobiology 2007, 17, 541–552. [Google Scholar] [CrossRef] [PubMed]
- Irhimeh, M.R.; Fitton, J.H.; Lowenthal, R.M. Fucoidan Ingestion Increases the Expression of CXCR4 on Human CD34+ Cells. Exp. Hematol. 2007, 35, 989–994. [Google Scholar] [CrossRef]
- Qi, Y.; Wang, L.; You, Y.; Sun, X.; Wen, C.; Fu, Y.; Song, S. Preparation of Low-Molecular-Weight Fucoidan with Anticoagulant Activity by Photocatalytic Degradation Method. Foods 2022, 11, 822. [Google Scholar] [CrossRef]
- Liang, Y.; Zhao, X.; Hu, T.; Chen, B.; Yin, Z.; Ma, P.X.; Guo, B. Adhesive Hemostatic Conducting Injectable Composite Hydrogels with Sustained Drug Release and Photothermal Antibacterial Activity to Promote Full-Thickness Skin Regeneration During Wound Healing. Small 2019, 15, 1900046. [Google Scholar] [CrossRef]
- Sanjeewa, K.K.A.; Herath, K.H.I.N.M.; Yang, H.-W.; Choi, C.S.; Jeon, Y.-J. Anti-Inflammatory Mechanisms of Fucoidans to Treat Inflammatory Diseases: A Review. Mar. Drugs 2021, 19, 678. [Google Scholar] [CrossRef] [PubMed]
- Park, H.Y.; Han, M.H.; Park, C.; Jin, C.-Y.; Kim, G.-Y.; Choi, I.-W.; Kim, N.D.; Nam, T.-J.; Kwon, T.K.; Choi, Y.H. Anti-Inflammatory Effects of Fucoidan through Inhibition of NF-κB, MAPK and Akt Activation in Lipopolysaccharide-Induced BV2 Microglia Cells. Food Chem. Toxicol. 2011, 49, 1745–1752. [Google Scholar] [CrossRef] [PubMed]
- Troy, E.; Tilbury, M.A.; Power, A.M.; Wall, J.G. Nature-Based Biomaterials and Their Application in Biomedicine. Polymers 2021, 13, 3321. [Google Scholar] [CrossRef]
- Zahariev, N.; Katsarov, P.; Lukova, P.; Pilicheva, B. Novel Fucoidan Pharmaceutical Formulations and Their Potential Application in Oncology—A Review. Polymers 2023, 15, 3242. [Google Scholar] [CrossRef]
- Pawar, S.N.; Edgar, K.J. Alginate Derivatization: A Review of Chemistry, Properties and Applications. Biomaterials 2012, 33, 3279–3305. [Google Scholar] [CrossRef] [PubMed]
- Draget, K.I.; Skjåk-Bræk, G.; Smidsrød, O. Alginate Based New Materials. Int. J. Biol. Macromol. 1997, 21, 47–55. [Google Scholar] [CrossRef]
- Rowley, J.A.; Madlambayan, G.; Mooney, D.J. Alginate Hydrogels as Synthetic Extracellular Matrix Materials. Biomaterials 1999, 20, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Bochenek, M.A.; Veiseh, O.; Vegas, A.J.; McGarrigle, J.J.; Qi, M.; Marchese, E.; Omami, M.; Doloff, J.C.; Mendoza-Elias, J.; Nourmohammadzadeh, M.; et al. Alginate Encapsulation as Long-Term Immune Protection of Allogeneic Pancreatic Islet Cells Transplanted into the Omental Bursa of Macaques. Nat. Biomed. Eng. 2018, 2, 810–821. [Google Scholar] [CrossRef]
- Anderson, J.M.; Rodriguez, A.; Chang, D.T. Foreign Body Reaction to Biomaterials. Semin. Immunol. 2008, 20, 86–100. [Google Scholar] [CrossRef] [PubMed]
- Rosiak, P.; Latanska, I.; Paul, P.; Sujka, W.; Kolesinska, B. Modification of Alginates to Modulate Their Physic-Chemical Properties and Obtain Biomaterials with Different Functional Properties. Molecules 2021, 26, 7264. [Google Scholar] [CrossRef]
- Campo, V.L.; Kawano, D.F.; Silva, D.B.D.; Carvalho, I. Carrageenans: Biological Properties, Chemical Modifications and Structural Analysis – A Review. Carbohydr. Polym. 2009, 77, 167–180. [Google Scholar] [CrossRef]
- Necas, J.; Bartosikova, L. Carrageenan: A Review. Veterinární Medicína 2013, 58, 187–205. [Google Scholar] [CrossRef]
- Talarico, L.B.; Damonte, E.B. Interference in Dengue Virus Adsorption and Uncoating by Carrageenans. Virology 2007, 363, 473–485. [Google Scholar] [CrossRef]
- Krylova, N.V.; Kravchenko, A.O.; Iunikhina, O.V.; Pott, A.B.; Likhatskaya, G.N.; Volod’ko, A.V.; Zaporozhets, T.S.; Shchelkanov, M.Y.; Yermak, I.M. Influence of the Structural Features of Carrageenans from Red Algae of the Far Eastern Seas on Their Antiviral Properties. Mar. Drugs 2022, 20, 60. [Google Scholar] [CrossRef] [PubMed]
- Lahaye, M.; Robic, A. Structure and Functional Properties of Ulvan, a Polysaccharide from Green Seaweeds. Biomacromolecules 2007, 8, 1765–1774. [Google Scholar] [CrossRef]
- Tziveleka, L.-A.; Ioannou, E.; Roussis, V. Ulvan, a Bioactive Marine Sulphated Polysaccharide as a Key Constituent of Hybrid Biomaterials: A Review. Carbohydr. Polym. 2019, 218, 355–370. [Google Scholar] [CrossRef]
- Pradhan, B.; Bhuyan, P.; Ki, J.-S. Immunomodulatory, Antioxidant, Anticancer, and Pharmacokinetic Activity of Ulvan, a Seaweed-Derived Sulfated Polysaccharide: An Updated Comprehensive Review. Mar. Drugs 2023, 21, 300. [Google Scholar] [CrossRef] [PubMed]
- D’Ayala, G.G.; Malinconico, M.; Laurienzo, P. Marine Derived Polysaccharides for Biomedical Applications: Chemical Modification Approaches. Molecules 2008, 13, 2069–2106. [Google Scholar] [CrossRef]
- Huang, L.; Shen, M.; Morris, G.A.; Xie, J. Sulfated Polysaccharides: Immunomodulation and Signaling Mechanisms. Trends Food Sci. Technol. 2019, 92, 1–11. [Google Scholar] [CrossRef]
- Wasser, S.P. Medicinal Mushroom Science: History, Current Status, Future Trends, and Unsolved Problems. Int. J. Med. Mushrooms 2010, 12, 1–16. [Google Scholar] [CrossRef]
- Potterat, O. Goji ( Lycium Barbarum and L. Chinense ): Phytochemistry, Pharmacology and Safety in the Perspective of Traditional Uses and Recent Popularity. Planta Med. 2010, 76, 7–19. [Google Scholar] [CrossRef]
- Amagase, H.; Farnsworth, N.R. A Review of Botanical Characteristics, Phytochemistry, Clinical Relevance in Efficacy and Safety of Lycium Barbarum Fruit (Goji). Food Res. Int. 2011, 44, 1702–1717. [Google Scholar] [CrossRef]
- Gan, L.; Zhang, S.-H.; Liu, Q.; Xu, H.-B. A Polysaccharide-Protein Complex from Lycium Barbarum Upregulates Cytokine Expression in Human Peripheral Blood Mononuclear Cells. Eur. J. Pharmacol. 2003, 471, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.; Luo, S.; Luo, X.; Hu, M.; Ma, F.; Wang, Y.; Lai, X.; Zhou, L. Polysaccharides from Chinese Herbal Lycium Barbarum Induced Systemic and Local Immune Responses in H22 Tumor-Bearing Mice. J. Immunol. Res. 2018, 2018, 1–12. [Google Scholar] [CrossRef]
- Feng, L.; Xiao, X.; Liu, J.; Wang, J.; Zhang, N.; Bing, T.; Liu, X.; Zhang, Z.; Shangguan, D. Immunomodulatory Effects of Lycium Barbarum Polysaccharide Extract and Its Uptake Behaviors at the Cellular Level. Molecules 2020, 25, 1351. [Google Scholar] [CrossRef] [PubMed]
- Gong, G.; Dang, T.; Deng, Y.; Han, J.; Zou, Z.; Jing, S.; Zhang, Y.; Liu, Q.; Huang, L.; Wang, Z. Physicochemical Properties and Biological Activities of Polysaccharides from Lycium Barbarum Prepared by Fractional Precipitation. Int. J. Biol. Macromol. 2018, 109, 611–618. [Google Scholar] [CrossRef]
- Rooks, M.G.; Garrett, W.S. Gut Microbiota, Metabolites and Host Immunity. Nat. Rev. Immunol. 2016, 16, 341–352. [Google Scholar] [CrossRef]
- Duan, J.-L.; Liu, M.-Q.; Liu, Y.-N.; Liang, X.-F.; Cao, C.; Yao, A.-N.; Zhao, L.-Q.; Guo, S.; Qian, D.-W.; Bao, C.-J.; et al. Comparative Study on Physicochemical Characterization and Immunomodulatory Activities of Neutral and Acidic Lycium Barbarum Polysaccharides. Biomed. Pharmacother. 2024, 181, 117659. [Google Scholar] [CrossRef]
- Reynolds, T.; Dweck, A.C. Aloe Vera Leaf Gel: A Review Update. J. Ethnopharmacol. 1999, 68, 3–37. [Google Scholar] [CrossRef]
- Ni, Y.; Turner, D.; Yates, K.M.; Tizard, I. Isolation and Characterization of Structural Components of Aloe Vera L. Leaf Pulp. Int. Immunopharmacol. 2004, 4, 1745–1755. [Google Scholar] [CrossRef]
- Tizard, I.; Carpenter, R.; McAnalley, B.; Kemp, M. The Biological Activities of Mannans and Related Complex Carbohydrates. Mol. Biother. 1989, 1, 290–296. [Google Scholar]
- Chithra, P.; Sajithlal, G.B.; Chandrakasan, G. Influence of Aloe Vera on Collagen Characteristics in Healing Dermal Wounds in Rats. Mol. Cell. Biochem. 1998, 181, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Hamman, J.H. Composition and Applications of Aloe Vera Leaf Gel. Molecules 2008, 13, 1599–1616. [Google Scholar] [CrossRef]
- Matei, C.E.; Visan, A.I.; Cristescu, R. Aloe Vera Polysaccharides as Therapeutic Agents: Benefits Versus Side Effects in Biomedical Applications. Polysaccharides 2025, 6, 36. [Google Scholar] [CrossRef]
- Liu, C.; Cui, Y.; Pi, F.; Cheng, Y.; Guo, Y.; Qian, H. Extraction, Purification, Structural Characteristics, Biological Activities and Pharmacological Applications of Acemannan, a Polysaccharide from Aloe Vera: A Review. Molecules 2019, 24, 1554. [Google Scholar] [CrossRef]
- Chen, R.; Xu, J.; Wu, W.; Wen, Y.; Lu, S.; El-Seedi, H.R.; Zhao, C. Structure–Immunomodulatory Activity Relationships of Dietary Polysaccharides. Curr. Res. Food Sci. 2022, 5, 1330–1341. [Google Scholar] [CrossRef] [PubMed]
- Trakoolpolpruek, T.; Moonmangmee, S.; Chanput, W. Structure-Dependent Immune Modulating Activity of Okra Polysaccharide on THP-1 Macrophages. Bioact. Carbohydr. Diet. Fibre 2019, 17, 100173. [Google Scholar] [CrossRef]
- Reakasame, S.; Boccaccini, A.R. Oxidized Alginate-Based Hydrogels for Tissue Engineering Applications: A Review. Biomacromolecules 2018, 19, 3–21. [Google Scholar] [CrossRef]
- Balakrishnan, B.; Joshi, N.; Jayakrishnan, A.; Banerjee, R. Self-Crosslinked Oxidized Alginate/Gelatin Hydrogel as Injectable, Adhesive Biomimetic Scaffolds for Cartilage Regeneration. Acta Biomater. 2014, 10, 3650–3663. [Google Scholar] [CrossRef]
- Wei, Z.; Zhao, J.; Chen, Y.M.; Zhang, P.; Zhang, Q. Self-Healing Polysaccharide-Based Hydrogels as Injectable Carriers for Neural Stem Cells. Sci. Rep. 2016, 6, 37841. [Google Scholar] [CrossRef]
- Chakka, V.P.; Zhou, T. Carboxymethylation of Polysaccharides: Synthesis and Bioactivities. Int. J. Biol. Macromol. 2020, 165, 2425–2431. [Google Scholar] [CrossRef]
- Li, H.; Wang, Y.; Zhao, P.; Guo, L.; Huang, L.; Li, X.; Gao, W. Naturally and Chemically Acetylated Polysaccharides: Structural Characteristics, Synthesis, Activities, and Applications in the Delivery System: A Review. Carbohydr. Polym. 2023, 313, 120746. [Google Scholar] [CrossRef]
- Azagarsamy, M.A.; Anseth, K.S. Bioorthogonal Click Chemistry: An Indispensable Tool to Create Multifaceted Cell Culture Scaffolds. ACS Macro Lett. 2013, 2, 5–9. [Google Scholar] [CrossRef]
- Sletten, E.M.; Bertozzi, C.R. Bioorthogonal Chemistry: Fishing for Selectivity in a Sea of Functionality. Angew. Chem. Int. Ed. 2009, 48, 6974–6998. [Google Scholar] [CrossRef] [PubMed]
- Kolb, H.C.; Finn, M.; Sharpless, K.B. Click Chemistry: Diverse Chemical Function from a Few Good Reactions. Angew. Chem. Int. Ed. 2001, 40, 2004–2021. [Google Scholar] [CrossRef]
- Khodadadi Yazdi, M.; Sajadi, S.M.; Seidi, F.; Rabiee, N.; Fatahi, Y.; Rabiee, M.; Dominic, C.D.M.; Zarrintaj, P.; Formela, K.; Saeb, M.R.; et al. Clickable Polysaccharides for Biomedical Applications: A Comprehensive Review. Prog. Polym. Sci. 2022, 133, 101590. [Google Scholar] [CrossRef]
- Atanasov, A.G.; Zotchev, S.B.; Dirsch, V.M.; the International Natural Product Sciences Taskforce; Orhan, I.E.; Banach, M.; Rollinger, J.M.; Barreca, D.; Weckwerth, W.; Bauer, R.; et al. Natural Products in Drug Discovery: Advances and Opportunities. Nat. Rev. Drug Discov. 2021, 20, 200–216. [Google Scholar] [CrossRef]
- Cragg, G.M.; Newman, D.J. Natural Products: A Continuing Source of Novel Drug Leads. Biochim. Biophys. Acta BBA - Gen. Subj. 2013, 1830, 3670–3695. [Google Scholar] [CrossRef]
- Gaharwar, A.K.; Singh, I.; Khademhosseini, A. Engineered Biomaterials for in Situ Tissue Regeneration. Nat. Rev. Mater. 2020, 5, 686–705. [Google Scholar] [CrossRef]
- Ferreira, C.; Vieira, P.; Sá, H.; Malva, J.; Castelo-Branco, M.; Reis, F.; Viana, S. Polyphenols: Immunonutrients Tipping the Balance of Immunometabolism in Chronic Diseases. Front. Immunol. 2024, 15, 1360065. [Google Scholar] [CrossRef]
- Bai, Y.; Wang, Z.; He, X.; Zhu, Y.; Xu, X.; Yang, H.; Mei, G.; Chen, S.; Ma, B.; Zhu, R. Application of Bioactive Materials for Osteogenic Function in Bone Tissue Engineering. Small Methods 2024, 8, 2301283. [Google Scholar] [CrossRef]
- Pandey, K.B.; Rizvi, S.I. Plant Polyphenols as Dietary Antioxidants in Human Health and Disease. Oxid. Med. Cell. Longev. 2009, 2, 270–278. [Google Scholar] [CrossRef]
- Crozier, A.; Del Rio, D.; Clifford, M.N. Bioavailability of Dietary Flavonoids and Phenolic Compounds. Mol. Aspects Med. 2010, 31, 446–467. [Google Scholar] [CrossRef]
- Santangelo, C.; Vari, R.; Scazzocchio, B.; Di Benedetto, R.; Filesi, C.; Masella, R. Polyphenols, Intracellular Signalling and Inflammation. Ann.-Ist. Super. Sanita 2007, 43, 394. [Google Scholar] [PubMed]
- Takayanagi, H. Osteoimmunology: Shared Mechanisms and Crosstalk between the Immune and Bone Systems. Nat. Rev. Immunol. 2007, 7, 292–304. [Google Scholar] [CrossRef]
- Perrone, P.; De Rosa, C.; D’Angelo, S. Polyphenols and Bone Health: A Comprehensive Review of Their Role in Osteoporosis Prevention and Treatment. Molecules 2025, 30, 4154. [Google Scholar] [CrossRef]
- Trzeciakiewicz, A.; Habauzit, V.; Horcajada, M.-N. When Nutrition Interacts with Osteoblast Function: Molecular Mechanisms of Polyphenols. Nutr. Res. Rev. 2009, 22, 68–81. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.-W.; Chen, S.-C.; Lai, W.-F.T.; Chen, Y.-C.; Tsai, Y.-H. Screening of Flavonoids for Effective Osteoclastogenesis Suppression. Anal. Biochem. 2013, 433, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Domazetovic, V. Oxidative Stress in Bone Remodeling: Role of Antioxidants. Clin. Cases Miner. Bone Metab. 2017, 14, 209. [Google Scholar] [CrossRef]
- Manolagas, S.C. From Estrogen-Centric to Aging and Oxidative Stress: A Revised Perspective of the Pathogenesis of Osteoporosis. Endocr. Rev. 2010, 31, 266–300. [Google Scholar] [CrossRef]
- Xue, S.; Tan, W.; Mao, S.; Pan, H.; Ye, X.; Donlao, N.; Tian, J. Polyphenol-Based Functional Materials: Structural Insights, Composite Strategies, and Biomedical Applications. Adv. Sci. 2025, 12, e08924. [Google Scholar] [CrossRef]
- Faienza, M.F.; Giardinelli, S.; Annicchiarico, A.; Chiarito, M.; Barile, B.; Corbo, F.; Brunetti, G. Nutraceuticals and Functional Foods: A Comprehensive Review of Their Role in Bone Health. Int. J. Mol. Sci. 2024, 25, 5873. [Google Scholar] [CrossRef]
- Aggarwal, B.B.; Harikumar, K.B. Potential Therapeutic Effects of Curcumin, the Anti-Inflammatory Agent, against Neurodegenerative, Cardiovascular, Pulmonary, Metabolic, Autoimmune and Neoplastic Diseases. Int. J. Biochem. Cell Biol. 2009, 41, 40–59. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Wang, J.; Song, Z.; Pei, Y. Regulation Mechanism of Curcumin Mediated Inflammatory Pathway and Its Clinical Application: A Review. Front. Pharmacol. 2025, 16, 1642248. [Google Scholar] [CrossRef]
- Jurenka, J.S. Anti-Inflammatory Properties of Curcumin, a Major Constituent of Curcuma Longa: A Review of Preclinical and Clinical Research. Altern. Med. Rev. 2009, 14. [Google Scholar]
- Wilken, R.; Veena, M.S.; Wang, M.B.; Srivatsan, E.S. Curcumin: A Review of Anti-Cancer Properties and Therapeutic Activity in Head and Neck Squamous Cell Carcinoma. Mol. Cancer 2011, 10, 12. [Google Scholar] [CrossRef]
- Deng, X.; Ratnayake, J.; Ali, A. Curcumin-Loaded Drug Delivery Systems for Acute and Chronic Wound Management: A Review. Bioengineering 2025, 12, 860. [Google Scholar] [CrossRef]
- Anand, P.; Kunnumakkara, A.B.; Newman, R.A.; Aggarwal, B.B. Bioavailability of Curcumin: Problems and Promises. Mol. Pharm. 2007, 4, 807–818. [Google Scholar] [CrossRef]
- Kunnumakkara, A.B.; Bordoloi, D.; Padmavathi, G.; Monisha, J.; Roy, N.K.; Prasad, S.; Aggarwal, B.B. Curcumin, the Golden Nutraceutical: Multitargeting for Multiple Chronic Diseases. Br. J. Pharmacol. 2017, 174, 1325–1348. [Google Scholar] [CrossRef]
- Maiti, K.; Mukherjee, K.; Gantait, A.; Saha, B.P.; Mukherjee, P.K. Curcumin–Phospholipid Complex: Preparation, Therapeutic Evaluation and Pharmacokinetic Study in Rats. Int. J. Pharm. 2007, 330, 155–163. [Google Scholar] [CrossRef]
- Fu, Y.; Ge, Y.; Yi, S.; Peng, Q.; Jiang, H.; Zhou, J. Smart Nanoparticle Delivery Systems for Curcumin: A Targeted Strategy to Enhance Anticancer Efficacy and Bioavailability. J. Mater. Sci. Mater. Med. 2026, 37, 38. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Sun, W.; Yan, F.; Liu, H.; Deng, Z.; Cai, L. Icariin-Loaded Porous Scaffolds for Bone Regeneration through the Regulation of the Coupling Process of Osteogenesis and Osteoclastic Activity. Int. J. Nanomedicine 2019, Volume 14, 6019–6033. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.-Y.; Chen, Y.-P.; Zhang, C.; Zhang, X.; Xia, T.; Han, J.; Yang, N.; Song, S.-L.; Xu, C.-H. Icariin Accelerates Fracture Healing via Activation of the WNT1/β-Catenin Osteogenic Signaling Pathway. Curr. Pharm. Biotechnol. 2020, 21, 1645–1653. [Google Scholar] [CrossRef]
- Xie, B.; Liao, X.; Xin, L.; Xie, Z.; Jin, Q.; Li, A.; Li, H.; Li, J. Icaritin Inhibits Osteoclast Differentiation and Reduces Bone Loss by Targeting ESR1 to Inhibit miR503/RANK Pathway. Front. Pharmacol. 2025, 16, 1603333. [Google Scholar] [CrossRef]
- Luo, M.; Lin, X.; Wang, N.; Xie, Q.; Zeng, S.; Li, S.; He, Z.; Lan, X. Icariin Delivery System Based on Covalent Organic Framework Materials: Dual Effects of Immune Modulation and Osteogenesis Promotion. RSC Adv. 2025, 15, 23783–23800. [Google Scholar] [CrossRef] [PubMed]
- Sharon, N.; Lis, H. Lectins as Cell Recognition Molecules. Science 1989, 246, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Van Kooyk, Y.; Rabinovich, G.A. Protein-Glycan Interactions in the Control of Innate and Adaptive Immune Responses. Nat. Immunol. 2008, 9, 593–601. [Google Scholar] [CrossRef]
- Peumans, W.J.; Van Damme, Ejm. Lectins as Plant Defense Proteins. Plant Physiol. 1995, 109, 347–352. [Google Scholar] [CrossRef] [PubMed]
- Frykberg, R.G.; Banks, J. Challenges in the Treatment of Chronic Wounds. Adv. Wound Care 2015, 4, 560–582. [Google Scholar] [CrossRef]
- Sen, C.K.; Gordillo, G.M.; Roy, S.; Kirsner, R.; Lambert, L.; Hunt, T.K.; Gottrup, F.; Gurtner, G.C.; Longaker, M.T. Human Skin Wounds: A Major and Snowballing Threat to Public Health and the Economy. Wound Repair Regen. 2009, 17, 763–771. [Google Scholar] [CrossRef]
- Schafer, M.; Werner, S. Oxidative Stress in Normal and Impaired Wound Repair. Pharmacol. Res. 2008, 58, 165–171. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, X.; Liu, Y.; Jiang, T.; Ren, S.; Chen, J.; Xiong, H.; Yuan, M.; Li, W.; Machens, H.; et al. NRF2 Signalling Pathway: New Insights and Progress in the Field of Wound Healing. J. Cell. Mol. Med. 2021, 25, 5857–5868. [Google Scholar] [CrossRef]
- Ukaegbu, K.; Allen, E.; Svoboda, K.K.H. Reactive Oxygen Species and Antioxidants in Wound Healing: Mechanisms and Therapeutic Potential. Int. Wound J. 2025, 22, e70330. [Google Scholar] [CrossRef]
- Wynn, T.A.; Vannella, K.M. Macrophages in Tissue Repair, Regeneration, and Fibrosis. Immunity 2016, 44, 450–462. [Google Scholar] [CrossRef] [PubMed]
- Spiller, K.L.; Koh, T.J. Macrophage-Based Therapeutic Strategies in Regenerative Medicine. Adv. Drug Deliv. Rev. 2017, 122, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Bai, L.; Xu, D.; Zhou, Y.-M.; Zhang, Y.-B.; Zhang, H.; Chen, Y.-B.; Cui, Y.-L. Antioxidant Activities of Natural Polysaccharides and Their Derivatives for Biomedical and Medicinal Applications. Antioxidants 2022, 11, 2491. [Google Scholar] [CrossRef] [PubMed]
- Murray, P.J.; Wynn, T.A. Protective and Pathogenic Functions of Macrophage Subsets. Nat. Rev. Immunol. 2011, 11, 723–737. [Google Scholar] [CrossRef]
- Murray, P.J.; Allen, J.E.; Biswas, S.K.; Fisher, E.A.; Gilroy, D.W.; Goerdt, S.; Gordon, S.; Hamilton, J.A.; Ivashkiv, L.B.; Lawrence, T.; et al. Macrophage Activation and Polarization: Nomenclature and Experimental Guidelines. Immunity 2014, 41, 14–20. [Google Scholar] [CrossRef]
- Guilliams, M.; Ginhoux, F.; Jakubzick, C.; Naik, S.H.; Onai, N.; Schraml, B.U.; Segura, E.; Tussiwand, R.; Yona, S. Dendritic Cells, Monocytes and Macrophages: A Unified Nomenclature Based on Ontogeny. Nat. Rev. Immunol. 2014, 14, 571–578. [Google Scholar] [CrossRef] [PubMed]
- Lucas, T.; Waisman, A.; Ranjan, R.; Roes, J.; Krieg, T.; Müller, W.; Roers, A.; Eming, S.A. Differential Roles of Macrophages in Diverse Phases of Skin Repair. J. Immunol. 2010, 184, 3964–3977. [Google Scholar] [CrossRef]
- Ferrante, C.J.; Leibovich, S.J. Regulation of Macrophage Polarization and Wound Healing. Adv. Wound Care 2012, 1, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Wynn, T.A.; Ramalingam, T.R. Mechanisms of Fibrosis: Therapeutic Translation for Fibrotic Disease. Nat. Med. 2012, 18, 1028–1040. [Google Scholar] [CrossRef]
- Khanna, S.; Biswas, S.; Shang, Y.; Collard, E.; Azad, A.; Kauh, C.; Bhasker, V.; Gordillo, G.M.; Sen, C.K.; Roy, S. Macrophage Dysfunction Impairs Resolution of Inflammation in the Wounds of Diabetic Mice. PLoS ONE 2010, 5, e9539. [Google Scholar] [CrossRef] [PubMed]
- Mirza, R.; Koh, T.J. Dysregulation of Monocyte/Macrophage Phenotype in Wounds of Diabetic Mice. Cytokine 2011, 56, 256–264. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Klein, T.; Murray, R.Z.; Crawford, R.; Chang, J.; Wu, C.; Xiao, Y. Osteoimmunomodulation for the Development of Advanced Bone Biomaterials. Mater. Today 2016, 19, 304–321. [Google Scholar] [CrossRef]
- Spiller, K.L.; Freytes, D.O.; Vunjak-Novakovic, G. Macrophages Modulate Engineered Human Tissues for Enhanced Vascularization and Healing. Ann. Biomed. Eng. 2015, 43, 616–627. [Google Scholar] [CrossRef]
- Akira, S.; Uematsu, S.; Takeuchi, O. Pathogen Recognition and Innate Immunity. Cell 2006, 124, 783–801. [Google Scholar] [CrossRef]
- Brown, G.D.; Herre, J.; Williams, D.L.; Willment, J.A.; Marshall, A.S.J.; Gordon, S. Dectin-1 Mediates the Biological Effects of β-Glucans. J. Exp. Med. 2003, 197, 1119–1124. [Google Scholar] [CrossRef]
- Kawai, T.; Akira, S. The Role of Pattern-Recognition Receptors in Innate Immunity: Update on Toll-like Receptors. Nat. Immunol. 2010, 11, 373–384. [Google Scholar] [CrossRef]
- Goodridge, H.S.; Wolf, A.J.; Underhill, D.M. Β-glucan Recognition by the Innate Immune System. Immunol. Rev. 2009, 230, 38–50. [Google Scholar] [CrossRef]
- Iwasaki, A.; Medzhitov, R. Control of Adaptive Immunity by the Innate Immune System. Nat. Immunol. 2015, 16, 343–353. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, Q.; Zhang, Z.; Li, Z. Antioxidant Activity of Sulfated Polysaccharide Fractions Extracted from Laminaria Japonica. Int. J. Biol. Macromol. 2008, 42, 127–132. [Google Scholar] [CrossRef]
- Gorbet, M.B.; Sefton, M.V. Endotoxin: The Uninvited Guest. Biomaterials 2005, 26, 6811–6817. [Google Scholar] [CrossRef] [PubMed]
- Matzinger, P.; Kamala, T. Tissue-Based Class Control: The Other Side of Tolerance. Nat. Rev. Immunol. 2011, 11, 221–230. [Google Scholar] [CrossRef]
- Medzhitov, R.; Janeway, C.A. Decoding the Patterns of Self and Nonself by the Innate Immune System. Science 2002, 296, 298–300. [Google Scholar] [CrossRef]
- Dobrovolskaia, M.A.; McNeil, S.E. Understanding the Correlation between in Vitro and in Vivo Immunotoxicity Tests for Nanomedicines. J. Controlled Release 2013, 172, 456–466. [Google Scholar] [CrossRef] [PubMed]
- Sena, L.A.; Chandel, N.S. Physiological Roles of Mitochondrial Reactive Oxygen Species. Mol. Cell 2012, 48, 158–167. [Google Scholar] [CrossRef]
- Mills, E.L.; Kelly, B.; O’Neill, L.A.J. Mitochondria Are the Powerhouses of Immunity. Nat. Immunol. 2017, 18, 488–498. [Google Scholar] [CrossRef] [PubMed]
- West, A.P.; Brodsky, I.E.; Rahner, C.; Woo, D.K.; Erdjument-Bromage, H.; Tempst, P.; Walsh, M.C.; Choi, Y.; Shadel, G.S.; Ghosh, S. TLR Signalling Augments Macrophage Bactericidal Activity through Mitochondrial ROS. Nature 2011, 472, 476–480. [Google Scholar] [CrossRef]
- Kelly, B.; O’Neill, L.A. Metabolic Reprogramming in Macrophages and Dendritic Cells in Innate Immunity. Cell Res. 2015, 25, 771–784. [Google Scholar] [CrossRef] [PubMed]
- Van Den Bossche, J.; O’Neill, L.A.; Menon, D. Macrophage Immunometabolism: Where Are We (Going)? Trends Immunol. 2017, 38, 395–406. [Google Scholar] [CrossRef]
- Ahmed, S.M.U.; Luo, L.; Namani, A.; Wang, X.J.; Tang, X. Nrf2 Signaling Pathway: Pivotal Roles in Inflammation. Biochim. Biophys. Acta BBA - Mol. Basis Dis. 2017, 1863, 585–597. [Google Scholar] [CrossRef] [PubMed]
- Furman, D.; Campisi, J.; Verdin, E.; Carrera-Bastos, P.; Targ, S.; Franceschi, C.; Ferrucci, L.; Gilroy, D.W.; Fasano, A.; Miller, G.W.; et al. Chronic Inflammation in the Etiology of Disease across the Life Span. Nat. Med. 2019, 25, 1822–1832. [Google Scholar] [CrossRef]
- Huang, L.; Su, Y.; Zhang, D.; Zeng, Z.; Hu, X.; Hong, S.; Lin, X. Recent Theranostic Applications of Hydrogen Peroxide-Responsive Nanomaterials for Multiple Diseases. RSC Adv. 2023, 13, 27333–27358. [Google Scholar] [CrossRef]
- Lawrence, T.; Natoli, G. Transcriptional Regulation of Macrophage Polarization: Enabling Diversity with Identity. Nat. Rev. Immunol. 2011, 11, 750–761. [Google Scholar] [CrossRef] [PubMed]
- Orecchioni, M.; Ghosheh, Y.; Pramod, A.B.; Ley, K. Macrophage Polarization: Different Gene Signatures in M1(LPS+) vs. Classically and M2(LPS–) vs. Alternatively Activated Macrophages. Front. Immunol. 2019, 10, 1084. [Google Scholar] [CrossRef] [PubMed]
- Arranz, A.; Doxaki, C.; Vergadi, E.; Martinez De La Torre, Y.; Vaporidi, K.; Lagoudaki, E.D.; Ieronymaki, E.; Androulidaki, A.; Venihaki, M.; Margioris, A.N.; et al. Akt1 and Akt2 Protein Kinases Differentially Contribute to Macrophage Polarization. Proc. Natl. Acad. Sci. 2012, 109, 9517–9522. [Google Scholar] [CrossRef]
- Meng, T.; Xiao, D.; Muhammed, A.; Deng, J.; Chen, L.; He, J. Anti-Inflammatory Action and Mechanisms of Resveratrol. Molecules 2021, 26, 229. [Google Scholar] [CrossRef]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.-C. NF-κB Signaling in Inflammation. Signal Transduct. Target. Ther. 2017, 2, 17023. [Google Scholar] [CrossRef]
- Ahmed, T.; Gilani, A. Therapeutic Potential of Turmeric in Alzheimer’s Disease: Curcumin or Curcuminoids? Phytother. Res. 2014, 28, 517–525. [Google Scholar] [CrossRef]
- Martinez, F.O.; Gordon, S. The M1 and M2 Paradigm of Macrophage Activation: Time for Reassessment. F1000Prime Rep. 2014, 6. [Google Scholar] [CrossRef]
- Yuan, G.; Wahlqvist, M.L.; He, G.; Yang, M.; Li, D. Natural Products and Anti-Inflammatory Activity. Asia Pac. J. Clin. Nutr. 2006, 15. [Google Scholar]
- Pan, M.-H.; Lai, C.-S.; Ho, C.-T. Anti-Inflammatory Activity of Natural Dietary Flavonoids. Food Funct. 2010, 1, 15. [Google Scholar] [CrossRef]
- Korn, T.; Bettelli, E.; Oukka, M.; Kuchroo, V.K. IL-17 and Th17 Cells. Annu. Rev. Immunol. 2009, 27, 485–517. [Google Scholar] [CrossRef] [PubMed]
- Lee, G.R. The Balance of Th17 versus Treg Cells in Autoimmunity. Int. J. Mol. Sci. 2018, 19, 730. [Google Scholar] [CrossRef]
- Josefowicz, S.Z.; Lu, L.-F.; Rudensky, A.Y. Regulatory T Cells: Mechanisms of Differentiation and Function. Annu. Rev. Immunol. 2012, 30, 531–564. [Google Scholar] [CrossRef]
- Noack, M.; Miossec, P. Th17 and Regulatory T Cell Balance in Autoimmune and Inflammatory Diseases. Autoimmun. Rev. 2014, 13, 668–677. [Google Scholar] [CrossRef]
- Zhang, S.; Gang, X.; Yang, S.; Cui, M.; Sun, L.; Li, Z.; Wang, G. The Alterations in and the Role of the Th17/Treg Balance in Metabolic Diseases. Front. Immunol. 2021, 12, 678355. [Google Scholar] [CrossRef]
- Franz, S.; Rammelt, S.; Scharnweber, D.; Simon, J.C. Immune Responses to Implants – A Review of the Implications for the Design of Immunomodulatory Biomaterials. Biomaterials 2011, 32, 6692–6709. [Google Scholar] [CrossRef] [PubMed]
- Maldonado, R.A.; Von Andrian, U.H. How Tolerogenic Dendritic Cells Induce Regulatory T Cells. In Advances in Immunology; Elsevier, 2010; Vol. 108, pp. 111–165. ISBN 978-0-12-380995-7. [Google Scholar]
- Benam, K.H.; Dauth, S.; Hassell, B.; Herland, A.; Jain, A.; Jang, K.-J.; Karalis, K.; Kim, H.J.; MacQueen, L.; Mahmoodian, R.; et al. Engineered In Vitro Disease Models. Annu. Rev. Pathol. Mech. Dis. 2015, 10, 195–262. [Google Scholar] [CrossRef]
- Martin, P.; Leibovich, S.J. Inflammatory Cells during Wound Repair: The Good, the Bad and the Ugly. Trends Cell Biol. 2005, 15, 599–607. [Google Scholar] [CrossRef] [PubMed]
- Lieder, R.; Petersen, P.H.; Sigurjónsson, Ó.E. Endotoxins—the Invisible Companion in Biomaterials Research. Tissue Eng. Part B Rev. 2013, 19, 391–402. [Google Scholar] [CrossRef]
- Gurtner, G.C.; Werner, S.; Barrandon, Y.; Longaker, M.T. Wound Repair and Regeneration. Nature 2008, 453, 314–321. [Google Scholar] [CrossRef] [PubMed]
- Steinman, R.M.; Hawiger, D.; Nussenzweig, M.C. Tolerogenic Dendritic Cells. Annu. Rev. Immunol. 2003, 21, 685–711. [Google Scholar] [CrossRef] [PubMed]
- Shaw, T.J.; Martin, P. Wound Repair at a Glance. J. Cell Sci. 2009, 122, 3209–3213. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Khanna, S.; Nallu, K.; Hunt, T.K.; Sen, C.K. Dermal Wound Healing Is Subject to Redox Control. Mol. Ther. 2006, 13, 211–220. [Google Scholar] [CrossRef]
- Snyder, R.J.; Lantis, J.; Kirsner, R.S.; Shah, V.; Molyneaux, M.; Carter, M.J. Macrophages: A Review of Their Role in Wound Healing and Their Therapeutic Use. Wound Repair Regen. 2016, 24, 613–629. [Google Scholar] [CrossRef]
- Annabi, N.; Tamayol, A.; Uquillas, J.A.; Akbari, M.; Bertassoni, L.E.; Cha, C.; Camci-Unal, G.; Dokmeci, M.R.; Peppas, N.A.; Khademhosseini, A. 25th Anniversary Article: Rational Design and Applications of Hydrogels in Regenerative Medicine. Adv. Mater. 2014, 26, 85–124. [Google Scholar] [CrossRef]
- Qu, J.; Zhao, X.; Liang, Y.; Zhang, T.; Ma, P.X.; Guo, B. Antibacterial Adhesive Injectable Hydrogels with Rapid Self-Healing, Extensibility and Compressibility as Wound Dressing for Joints Skin Wound Healing. Biomaterials 2018, 183, 185–199. [Google Scholar] [CrossRef]
- Zhao, X.; Wu, H.; Guo, B.; Dong, R.; Qiu, Y.; Ma, P.X. Antibacterial Anti-Oxidant Electroactive Injectable Hydrogel as Self-Healing Wound Dressing with Hemostasis and Adhesiveness for Cutaneous Wound Healing. Biomaterials 2017, 122, 34–47. [Google Scholar] [CrossRef]
- Guo, S.; DiPietro, L.A. Factors Affecting Wound Healing. J. Dent. Res. 2010, 89, 219–229. [Google Scholar] [CrossRef]
- Liu, Z.; Yu, C.; Li, Z.; Wang, X.; Shang, D.; Dong, W. Matrix Metalloproteinase-Triggered Self-Assembling Peptides for Biomedical Applications. J. Mater. Chem. B 2025, 13, 8298–8334. [Google Scholar] [CrossRef]
- Du, L.; Lin, C.; Hu, H.; Zhao, Y.; Liao, J.; Al-Smadi, F.; Mi, B.; Hu, Y.; Liu, G. Recent Advances and Challenges in Hydrogel-Based Delivery of Immunomodulatory Strategies for Diabetic Wound Healing. Theranostics 2026, 16, 516–544. [Google Scholar] [CrossRef] [PubMed]
- Ju, S.; Mu, J.; Dokland, T.; Zhuang, X.; Wang, Q.; Jiang, H.; Xiang, X.; Deng, Z.-B.; Wang, B.; Zhang, L.; et al. Grape Exosome-like Nanoparticles Induce Intestinal Stem Cells and Protect Mice From DSS-Induced Colitis. Mol. Ther. 2013, 21, 1345–1357. [Google Scholar] [CrossRef]
- Raimondo, S.; Naselli, F.; Fontana, S.; Monteleone, F.; Lo Dico, A.; Saieva, L.; Zito, G.; Flugy, A.; Manno, M.; Di Bella, M.A.; et al. Citrus Limon -Derived Nanovesicles Inhibit Cancer Cell Proliferation and Suppress CML Xenograft Growth by Inducing TRAIL-Mediated Cell Death. Oncotarget 2015, 6, 19514–19527. [Google Scholar] [CrossRef]
- Kim, J.; Li, S.; Zhang, S.; Wang, J. Plant-Derived Exosome-like Nanoparticles and Their Therapeutic Activities. Asian J. Pharm. Sci. 2022, 17, 53–69. [Google Scholar] [CrossRef]
- Hu, Y.; Rao, S.-S.; Wang, Z.-X.; Cao, J.; Tan, Y.-J.; Luo, J.; Li, H.-M.; Zhang, W.-S.; Chen, C.-Y.; Xie, H. Exosomes from Human Umbilical Cord Blood Accelerate Cutaneous Wound Healing through miR-21-3p-Mediated Promotion of Angiogenesis and Fibroblast Function. Theranostics 2018, 8, 169–184. [Google Scholar] [CrossRef] [PubMed]
- Van Niel, G.; D’Angelo, G.; Raposo, G. Shedding Light on the Cell Biology of Extracellular Vesicles. Nat. Rev. Mol. Cell Biol. 2018, 19, 213–228. [Google Scholar] [CrossRef] [PubMed]
- Théry, C.; Witwer, K.W.; Aikawa, E.; Alcaraz, M.J.; Anderson, J.D.; Andriantsitohaina, R.; Antoniou, A.; Arab, T.; Archer, F.; Atkin-Smith, G.K.; et al. Minimal Information for Studies of Extracellular Vesicles 2018 (MISEV2018): A Position Statement of the International Society for Extracellular Vesicles and Update of the MISEV2014 Guidelines. J. Extracell. Vesicles 2018, 7, 1535750. [Google Scholar] [CrossRef]
- Schlundt, C.; El Khassawna, T.; Serra, A.; Dienelt, A.; Wendler, S.; Schell, H.; Van Rooijen, N.; Radbruch, A.; Lucius, R.; Hartmann, S.; et al. Macrophages in Bone Fracture Healing: Their Essential Role in Endochondral Ossification. Bone 2018, 106, 78–89. [Google Scholar] [CrossRef]
- Claes, L.; Recknagel, S.; Ignatius, A. Fracture Healing under Healthy and Inflammatory Conditions. Nat. Rev. Rheumatol. 2012, 8, 133–143. [Google Scholar] [CrossRef]
- Huang, G.; Li, F.; Zhao, X.; Ma, Y.; Li, Y.; Lin, M.; Jin, G.; Lu, T.J.; Genin, G.M.; Xu, F. Functional and Biomimetic Materials for Engineering of the Three-Dimensional Cell Microenvironment. Chem. Rev. 2017, 117, 12764–12850. [Google Scholar] [CrossRef]
- Nahrendorf, M.; Swirski, F.K. Abandoning M1/M2 for a Network Model of Macrophage Function. Circ. Res. 2016, 119, 414–417. [Google Scholar] [CrossRef] [PubMed]
- Veiseh, O.; Doloff, J.C.; Ma, M.; Vegas, A.J.; Tam, H.H.; Bader, A.R.; Li, J.; Langan, E.; Wyckoff, J.; Loo, W.S.; et al. Size- and Shape-Dependent Foreign Body Immune Response to Materials Implanted in Rodents and Non-Human Primates. Nat. Mater. 2015, 14, 643–651. [Google Scholar] [CrossRef] [PubMed]
- Iravani, S. Green Synthesis of Metal Nanoparticles Using Plants. Green Chem. 2011, 13, 2638. [Google Scholar] [CrossRef]
- Hussain, I.; Singh, N.B.; Singh, A.; Singh, H.; Singh, S.C. Green Synthesis of Nanoparticles and Its Potential Application. Biotechnol. Lett. 2016, 38, 545–560. [Google Scholar] [CrossRef]
- Sophia Fox, A.J.; Bedi, A.; Rodeo, S.A. The Basic Science of Articular Cartilage: Structure, Composition, and Function. Sports Health Multidiscip. Approach 2009, 1, 461–468. [Google Scholar] [CrossRef]
- Caldwell, A.S.; Rao, V.V.; Golden, A.C.; Anseth, K.S. Porous Bio-Click Microgel Scaffolds Control hMSC Interactions and Promote Their Secretory Properties. Biomaterials 2020, 232, 119725. [Google Scholar] [CrossRef]
- Rolls, A.; Shechter, R.; Schwartz, M. The Bright Side of the Glial Scar in CNS Repair. Nat. Rev. Neurosci. 2009, 10, 235–241. [Google Scholar] [CrossRef]
- Ruvinov, E.; Cohen, S. Alginate Biomaterial for the Treatment of Myocardial Infarction: Progress, Translational Strategies, and Clinical Outlook. Adv. Drug Deliv. Rev. 2016, 96, 54–76. [Google Scholar] [CrossRef]
- Olasehinde, T.A.; Olaniran, A.O.; Okoh, A.I. Sulfated Polysaccharides of Some Seaweeds Exhibit Neuroprotection via Mitigation of Oxidative Stress, Cholinergic Dysfunction and Inhibition of Zn – Induced Neuronal Damage in HT-22 Cells. BMC Complement. Med. Ther. 2020, 20, 251. [Google Scholar] [CrossRef]
- Walle, T. Bioavailability of Resveratrol. Ann. N. Y. Acad. Sci. 2011, 1215, 9–15. [Google Scholar] [CrossRef]
- Manach, C.; Scalbert, A.; Morand, C.; Rémésy, C.; Jiménez, L. Polyphenols: Food Sources and Bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [CrossRef]
- Chavda, H.; Patel, C.; Anand, I. Biopharmaceutics Classification System. Syst. Rev. Pharm. 2010, 1, 62. [Google Scholar] [CrossRef]
- Blanco, E.; Shen, H.; Ferrari, M. Principles of Nanoparticle Design for Overcoming Biological Barriers to Drug Delivery. Nat. Biotechnol. 2015, 33, 941–951. [Google Scholar] [CrossRef] [PubMed]
- Ventola, C.L. The Nanomedicine Revolution: Part 1: Emerging Concepts. Pharm. Ther. 2012, 37, 512. [Google Scholar]
- Barani, M.; Sangiovanni, E.; Angarano, M.; Rajizadeh, M.A.; Mehrabani, M.; Piazza, S.; Gangadharappa, H.V.; Pardakhty, A.; Mehrbani, M.; Dell’Agli, M.; et al. Phytosomes as Innovative Delivery Systems for Phytochemicals: A Comprehensive Review of Literature. Int. J. Nanomedicine 2021, Volume 16, 6983–7022. [Google Scholar] [CrossRef]
- Semalty, A.; Semalty, M.; Rawat, M.S.M.; Franceschi, F. Supramolecular Phospholipids–Polyphenolics Interactions: The PHYTOSOME® Strategy to Improve the Bioavailability of Phytochemicals. Fitoterapia 2010, 81, 306–314. [Google Scholar] [CrossRef] [PubMed]
- Amrit, P.; Sayyad, N.; Gade, S.; Dudhal, P.; Rasve, V.R.; Kale, A. An Extensive Review of Phytosomes: A Novel Approach to Delivering Phytoconstituents. World J Pharm Res 2024, 26, 346–357. [Google Scholar]
- Ioannidis, J.P.A. Why Most Published Research Findings Are False. PLoS Med. 2005, 2, e124. [Google Scholar] [CrossRef] [PubMed]
- Begley, C.G.; Ellis, L.M. Raise Standards for Preclinical Cancer Research. Nature 2012, 483, 531–533. [Google Scholar] [CrossRef]
- World Health Organization. WHO Guidelines on Good Manufacturing Practices (GMP) for Herbal Medicines; World Health Organization, 2007; ISBN 92-4-154716-2. [Google Scholar]
- Khan, I.A.; Smillie, T. Implementing a “Quality by Design” Approach to Assure the Safety and Integrity of Botanical Dietary Supplements. J. Nat. Prod. 2012, 75, 1665–1673. [Google Scholar] [CrossRef]
- Booker, A.; Johnston, D.; Heinrich, M. Value Chains of Herbal Medicines—Research Needs and Key Challenges in the Context of Ethnopharmacology. J. Ethnopharmacol. 2012, 140, 624–633. [Google Scholar] [CrossRef]
- Heinrich, M.; Edwards, S.; Moerman, D.E.; Leonti, M. Ethnopharmacological Field Studies: A Critical Assessment of Their Conceptual Basis and Methods. J. Ethnopharmacol. 2009, 124, 1–17. [Google Scholar] [CrossRef]
- Oguamanam, C. Genetic Resources & Access and Benefit Sharing: Politics, Prospects and Opportunities for Canada after Nagoya. Prospects Oppor. Can. Nagoya 2011, 22, 87–201. [Google Scholar]
- World Health Organization. WHO Guidelines on Good Agricultural and Collection Practices [GACP] for Medicinal Plants; World Health Organization, 2003; ISBN 92-4-154627-1. [Google Scholar]
- Robinson, D. Confronting Biopiracy: Challenges, Cases and International Debates; Routledge, 2010; ISBN 1-84977-471-4. [Google Scholar]
- Ethics Dumping: Case Studies from North-South Research Collaborations; Schroeder, D., Cook, J., Hirsch, F., Fenet, S., Muthuswamy, V., Eds.; SpringerBriefs in Research and Innovation Governance; Springer International Publishing: Cham, 2018; ISBN 978-3-319-64730-2. [Google Scholar]
- Ramsay, J.R.; Suhrbier, A.; Aylward, J.H.; Ogbourne, S.; Cozzi, S.-J.; Poulsen, M.G.; Baumann, K.C.; Welburn, P.; Redlich, G.L.; Parsons, P.G. The Sap from Euphorbia Peplus Is Effective against Human Nonmelanoma Skin Cancers: Euphorbia Peplus Sap Is Effective against Skin Cancers. Br. J. Dermatol. 2011, no–no. [Google Scholar] [CrossRef]
- El Enshasy, H.A. Cordyceps-Based Bioactives: Product Development and Marketing. In Cordyceps and Allied Species; Springer, 2025; pp. 379–394. [Google Scholar]
- Sarker, S.D.; Nahar, L. An Introduction to Natural Products Isolation. In Natural Products Isolation; Sarker, S.D., Nahar, L., Eds.; Methods in Molecular Biology; Humana Press: Totowa, NJ, 2012; Vol. 864, pp. 1–25. ISBN 978-1-61779-623-4. [Google Scholar]
- Wolfender, J.-L.; Rudaz, S.; Hae Choi, Y.; Kyong Kim, H. Plant Metabolomics: From Holistic Data to Relevant Biomarkers. Curr. Med. Chem. 2013, 20, 1056–1090. [Google Scholar] [PubMed]
- FDA, U. Guidance for Industry CGMP for Phase 1 Investigational Drugs. 2008.
- Kamisetti, R.R. Strategies Followed for the Assessment of the Advanced Therapies by European Medicines Agency. DOI 2023, 10, 2165–7920. [Google Scholar]
- Tian, J.; Song, X.; Wang, Y.; Cheng, M.; Lu, S.; Xu, W.; Gao, G.; Sun, L.; Tang, Z.; Wang, M.; et al. Regulatory Perspectives of Combination Products. Bioact. Mater. 2022, 10, 492–503. [Google Scholar] [CrossRef]
- Seok, J.; Warren, H.S.; Cuenca, A.G.; Mindrinos, M.N.; Baker, H.V.; Xu, W.; Richards, D.R.; McDonald-Smith, G.P.; Gao, H.; Hennessy, L.; et al. Genomic Responses in Mouse Models Poorly Mimic Human Inflammatory Diseases. Proc. Natl. Acad. Sci. 2013, 110, 3507–3512. [Google Scholar] [CrossRef] [PubMed]
- Hannon, G.; Prina-Mello, A. Endotoxin Contamination of Engineered Nanomaterials: Overcoming the Hurdles Associated with Endotoxin Testing. WIREs Nanomedicine Nanobiotechnology 2021, 13, e1738. [Google Scholar] [CrossRef]
- Reay, S.L.; Jackson, E.L.; Salthouse, D.; Ferreira, A.M.; Hilkens, C.M.U.; Novakovic, K. Effective Endotoxin Removal from Chitosan That Preserves Chemical Structure and Improves Compatibility with Immune Cells. Polymers 2023, 15, 1592. [Google Scholar] [CrossRef]
- Delavary, B.M.; Van Der Veer, W.M.; Van Egmond, M.; Niessen, F.B.; Beelen, R.H.J. Macrophages in Skin Injury and Repair. Immunobiology 2011, 216, 753–762. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, S.N.; Ingber, D.E. Microfluidic Organs-on-Chips. Nat. Biotechnol. 2014, 32, 760–772. [Google Scholar] [CrossRef] [PubMed]
| Material | Key Structural Features | Main PRRs Involved | Dominant Immune Effects | Evidence Level | Key References |
|---|---|---|---|---|---|
| Fucoidan | Sulfated fucose-rich polysaccharide; MW variability; sulfation degree critical | TLR2/4, scavenger receptors | Anti-inflammatory, macrophage modulation, angiogenic support | In vitro, in vivo, limited clinical | [31,33,38,39,43] |
| Alginate | Mannuronic/guluronic acid blocks; tunable oxidation | Limited direct PRR activation; foreign body modulation | Immune shielding; fibrosis modulation (when purified) | Extensive in vivo biomaterials | [20,47,48,49,50,51] |
| Carrageenan | Sulfated galactans (κ, ι, λ isoforms) | TLR4 (context-dependent) | Context-dependent pro/anti-inflammatory | In vitro, inflammatory models | [53,54,55] |
| Ulvan | Sulfated rhamnose-rich polysaccharide | Proposed TLR-mediated signaling | Antioxidant and immunomodulatory | Preclinical | [57,58,59] |
| Lycium barbarum polysaccharides | Polysaccharide–protein complexes | TLR2/4 | Macrophage activation; cytokine modulation | Preclinical | [65,66,67,68,69] |
| Acemannan (Aloe vera) | Acetylated mannan | TLR4 | Macrophage activation; wound healing support | Preclinical + topical clinical | [73,74,75,76] |
| Curcumin | Hydrophobic polyphenol; pleiotropic signaling | NF-κB modulation (indirect) | Anti-inflammatory; antioxidant; immunometabolic effects | Extensive preclinical; delivery challenges | [108,109,110,111,112,113,114] |
| Plant-derived EV-like nanoparticles | Lipid bilayer vesicles; miRNA/protein cargo | Uptake via endocytosis; PRR-independent pathways | Inflammatory modulation; tissue repair | Early-stage preclinical | [186,187,188,189,190,191] |
| Development Stage | Required Actions | Critical Quality Attributes (CQA) | Key References |
|---|---|---|---|
| Raw Material Sourcing | Botanical authentication; GACP compliance | Species identity; origin; harvest timing | [220,224,229] |
| Extraction & Processing | Standardized protocols; solvent and temperature control | MW distribution; sulfation/acetylation degree | [224,231] |
| Purification | Endotoxin removal; impurity control | Endotoxin thresholds; residual solvents | [150,181] |
| Biological Testing | Multiparametric immune assays; dose–response validation | Mechanism-linked potency assays | [134,232] |
| GMP Manufacturing | SOP documentation; traceability; scale-up validation | Reproducibility; stability metrics | [214,226] |
| Regulatory Classification | Early agency consultation | Device vs biologic vs combination product | [227,228] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




