Submitted:
02 March 2026
Posted:
04 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
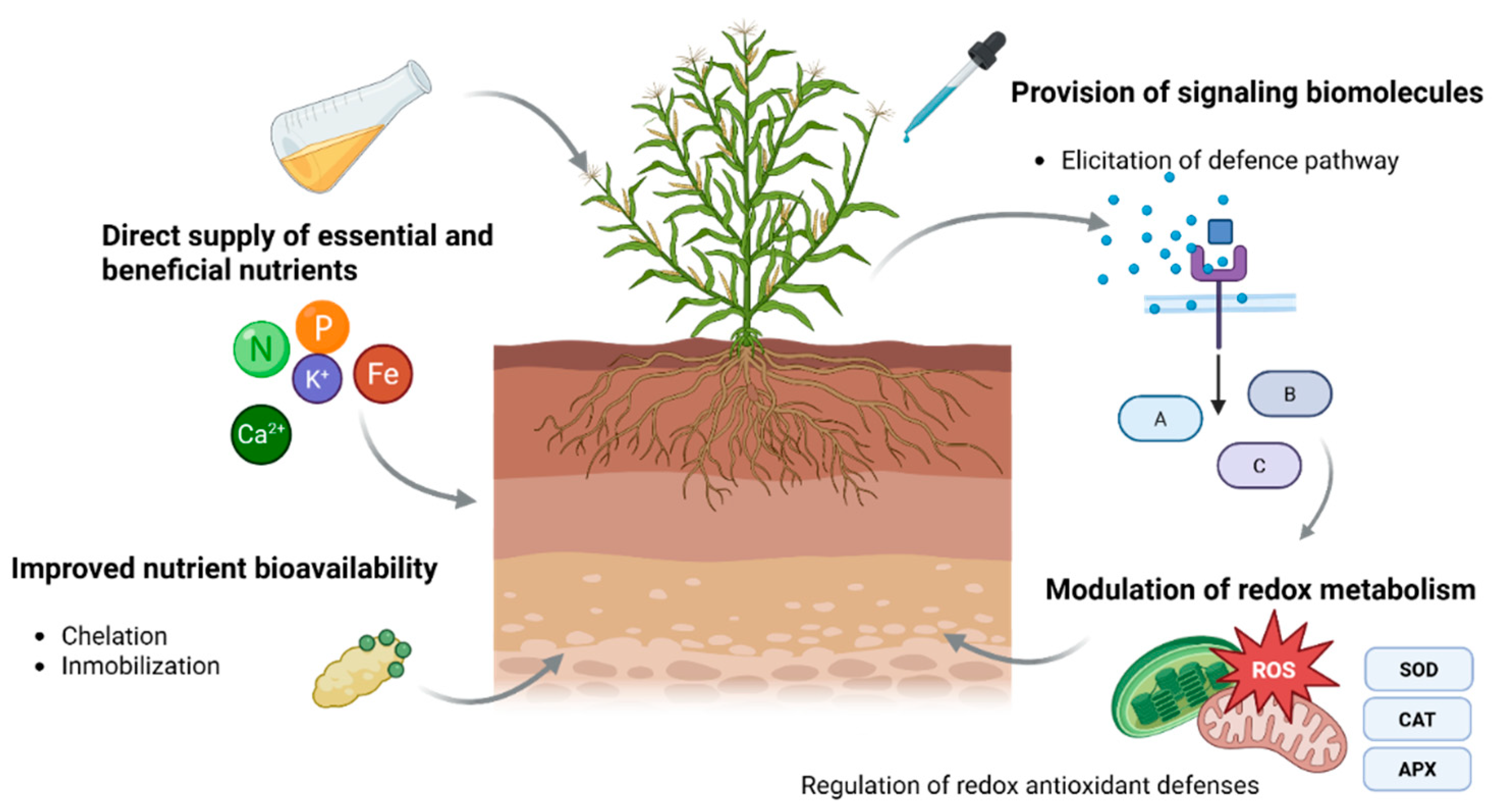
2. Botanical Extracts and Bioactive Constituents Relevant to Redox Responses
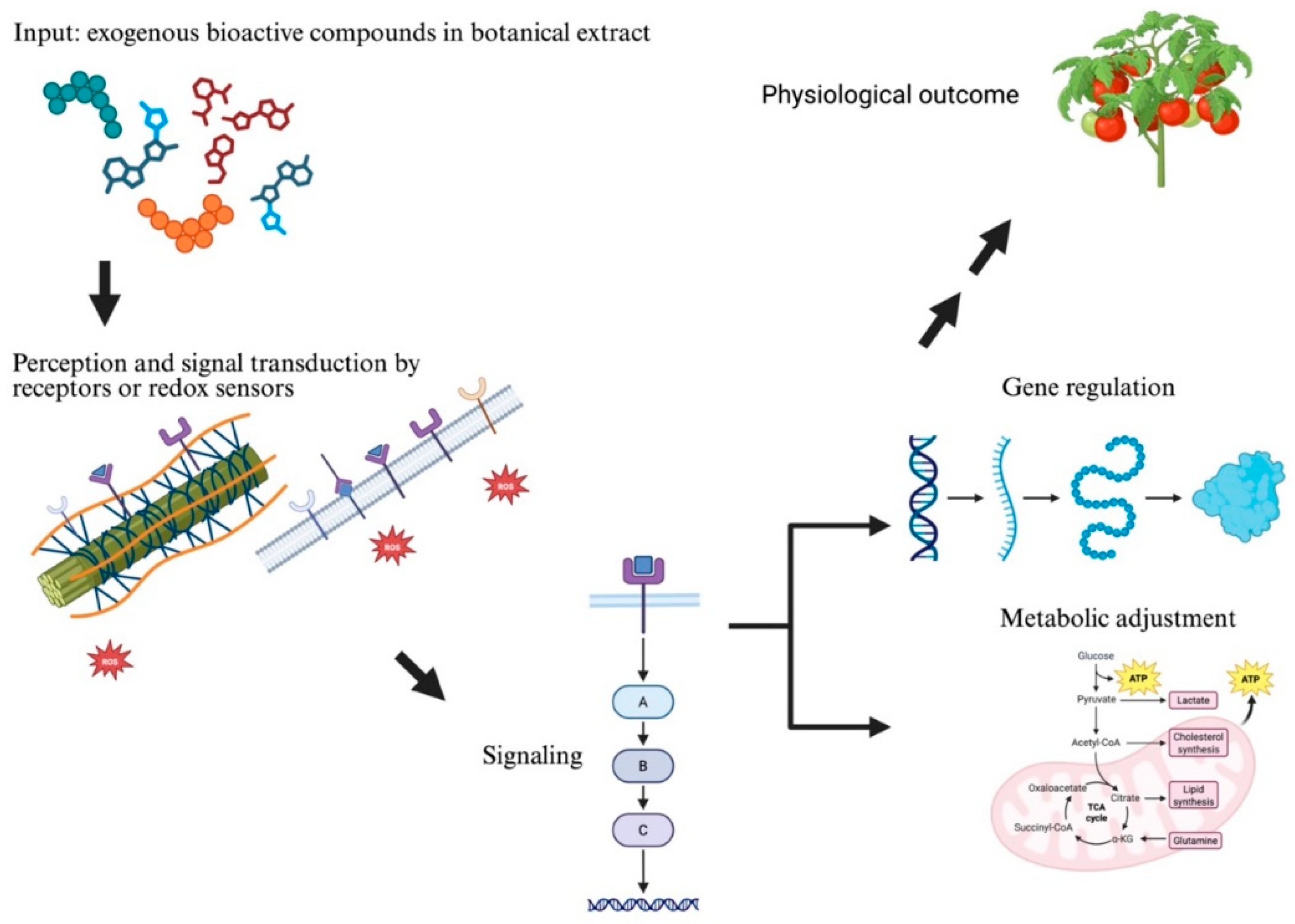
3. Perception, Signaling, and Putative Redox Modulation Responses After Botanical Extract Application
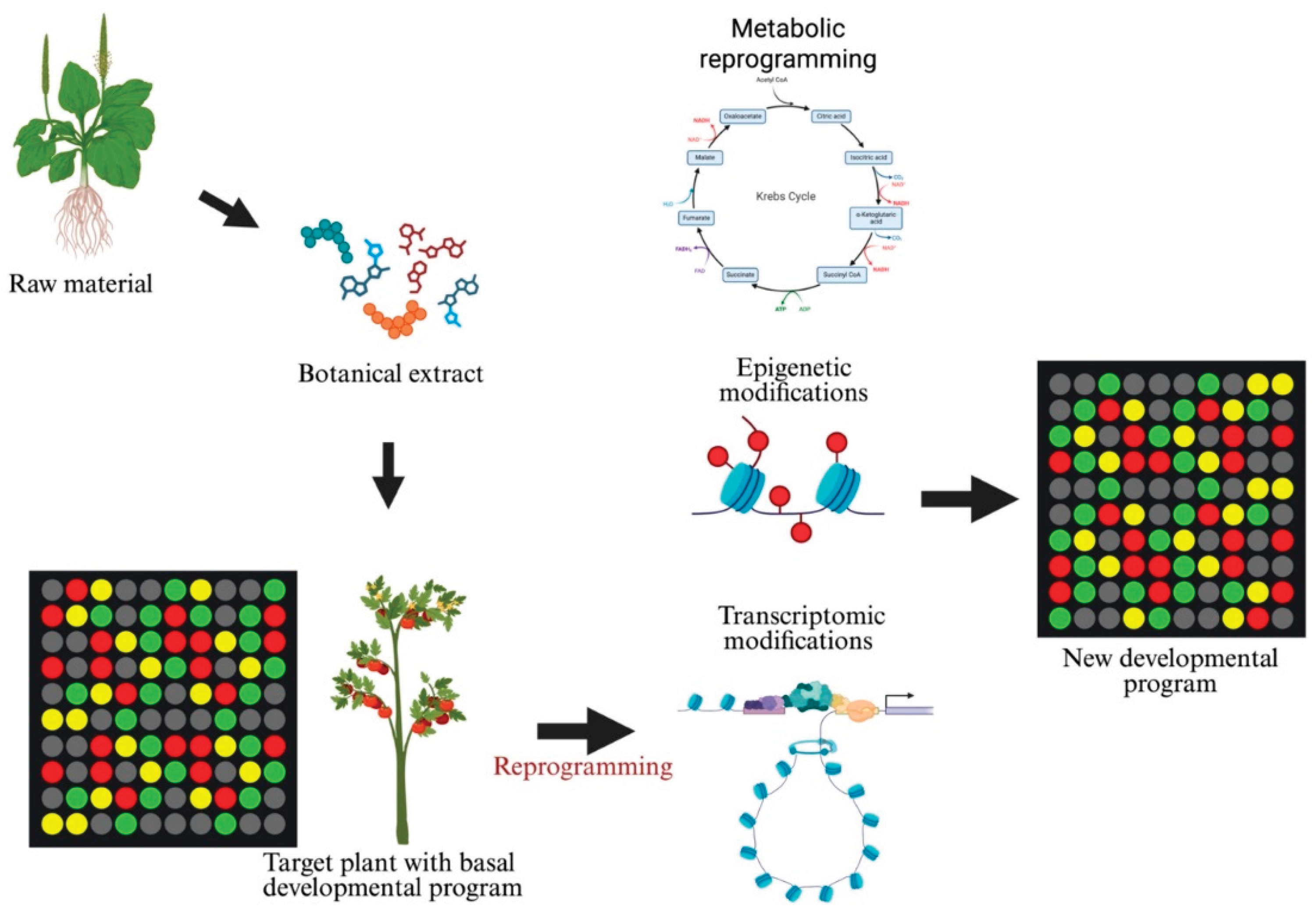
4. Metabolic and Gene Expression Reprogramming by Botanical Extracts
5. Conclusions and Future Directions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| APX | Ascorbate peroxidase |
| CAT | Catalase |
| DHAR | Dehydroascorbate reductase |
| GPX | Glutathione peroxidase |
| GR | Glutathione reductase |
| GSH | Glutathione |
| LED | Light Emitting Diode |
| MDA | Malondialdehyde |
| PAR | Photosynthetically Active Radiation |
| PRX | Peroxidase |
| PrxQ | Peroxiredoxin Q |
| ROS | Reactive oxygen species |
| SOD | Superoxide dismutase |
| DOAJ | Directory of open access journals |
| TLA | Three letter acronym |
| LD | Linear dichroism |
References
- Mansoor, S.; Ali Wani, O.; Lone, J.K.; Manhas, S.; Kour, N.; Alam, P.; Ahmad, A.; Ahmad, P. Reactive Oxygen Species in Plants: From Source to Sink. Antioxidants 2022, 11, 225. [Google Scholar] [CrossRef]
- Mikhailovsky, G.E. Life, Its Definition, Origin, Evolution, and Four-Dimensional Hierarchical Structure. BioSystems 2024, 237, 105158. [Google Scholar] [CrossRef] [PubMed]
- Gomberg, M. An Instance of Trivalent Carbon: Triphenylmethyl. J.Am. Chem. Soc. 1900, 22, 757–771. [Google Scholar] [CrossRef]
- Commoner, B.; Townsend, J.; Pake, G.E. Free Radicals in Biological Materials. Nature 1954, 174, 689–691. [Google Scholar] [CrossRef] [PubMed]
- Ylikahri, R.H.; Hassinen, I.; Kähönen, M.T. Effect of a Transaminase Inhibitor on the Transport of Cytosolic Reducing Equivalents into Mitochondria. Biochemical and Biophysical Research Communications 1971, 44, 150–156. [Google Scholar] [CrossRef]
- Anelli, T.; Sannino, S.; Sitia, R. Proteostasis and “Redoxtasis” in the Secretory Pathway: Tales of Tails from ERp44 and Immunoglobulins. Free Radical Biology and Medicine 2015, 83, 323–330. [Google Scholar] [CrossRef]
- Kunert, K.J.; Foyer, C.H. Chapter Three - The Ascorbate/Glutathione Cycle. In Advances in Botanical Research;Oxidative Stress Response In Plants; Mittler, R., Breusegem, F.V., Eds.; Academic Press, 2023; Vol. 105, pp. 77–112. [Google Scholar]
- Medrano-Macías, J.; Flores-Gallegos, A.C.; Nava-Reyna, E.; Morales, I.; Tortella, G.; Solís-Gaona, S.; Benavides-Mendoza, A. Reactive Oxygen, Nitrogen, and Sulfur Species (RONSS) as a Metabolic Cluster for Signaling and Biostimulation of Plants: An Overview. Plants 2022, 11, 3203. [Google Scholar] [CrossRef]
- Sies, H. Oxidative Stress: Introductory Remarks. In Oxidative stress; Academic Press: London, UK, 1985; pp. 1–8. [Google Scholar]
- Sies, H. Oxidative Stress: Oxidants and Antioxidants. Experimental Physiology 1997, 82, 291–295. [Google Scholar] [CrossRef]
- Baltazar, M.; Correia, S.; Guinan, K.J.; Sujeeth, N.; Bragança, R.; Gonçalves, B. Recent Advances in the Molecular Effects of Biostimulants in Plants: An Overview. Biomolecules 2021, 11, 1096. [Google Scholar] [CrossRef]
- Yakhin, O.I.; Lubyanov, A.A.; Yakhin, I.A.; Brown, P.H. Biostimulants in Plant Science: A Global Perspective. Front Plant Sci 2017, 7. [Google Scholar] [CrossRef]
- Garza-Alonso, C.A.; Olivares-Sáenz, E.; González-Morales, S.; Cabrera-De la Fuente, M.; Juárez-Maldonado, A.; González-Fuentes, J.A.; Tortella, G.; Valdés-Caballero, M.V.; Benavides-Mendoza, A. Strawberry Biostimulation: From Mechanisms of Action to Plant Growth and Fruit Quality. Plants 2022, 11, 3463. [Google Scholar] [CrossRef] [PubMed]
- Ben Mrid, R.; Benmrid, B.; Hafsa, J.; Boukcim, H.; Sobeh, M.; Yasri, A. Secondary Metabolites as Biostimulant and Bioprotectant Agents: A Review. Science of The Total Environment 2021, 777, 146204. [Google Scholar] [CrossRef]
- Han, M.; Kasim, S.; Yang, Z.; Deng, X.; Saidi, N.; Uddin, M.; Shuib, E. Plant Extracts as Biostimulant Agents: A Promising Strategy for Managing Environmental Stress in Sustainable Agriculture. PHYTON 2024, 93, 2149–2166. [Google Scholar] [CrossRef]
- Rodrigues, M.; Baptistella, J.L.C.; Horz, D.C.; Bortolato, L.M.; Mazzafera, P. Organic Plant Biostimulants and Fruit Quality—A Review. Agronomy 2020, 10, 988. [Google Scholar] [CrossRef]
- Yuniati, N.; Kusumiyati, K.; Mubarok, S.; Nurhadi, B. The Role of Moringa Leaf Extract as a Plant Biostimulant in Improving the Quality of Agricultural Products. Plants 2022, 11, 2186. [Google Scholar] [CrossRef]
- Martínez-Lorente, S.E.; Martí-Guillén, J.M.; Pedreño, M.Á.; Almagro, L.; Sabater-Jara, A.B. Higher Plant-Derived Biostimulants: Mechanisms of Action and Their Role in Mitigating Plant Abiotic Stress. Antioxidants 2024, 13, 318. [Google Scholar] [CrossRef]
- Roche, D.; Rickson, J.R.; Pawlett, M. Moving towards a Mechanistic Understanding of Biostimulant Impacts on Soil Properties and Processes: A Semi-Systematic Review. Front. Agron. 2024, 6. [Google Scholar] [CrossRef]
- Di Sario, L.; Boeri, P.; Matus, J.T.; Pizzio, G.A. Plant Biostimulants to Enhance Abiotic Stress Resilience in Crops. International Journal of Molecular Sciences 2025, 26, 1129. [Google Scholar] [CrossRef]
- Hernández-Bolaños, E.; Sánchez-Retuerta, V.; Matías-Hernández, L.; Cuyas, L. Promising Applications on the Use of Medicinal and Aromatic Plants in Agriculture. Discov Agric 2025, 3, 36. [Google Scholar] [CrossRef]
- Espinoza-Antón, A.A.; Hernández-Herrera, R.M.; Pizano-Andrade, J.C.; Salcedo-Perez, E. Botanical Extracts with Biostimulant Potential for Agriculture in Mexico: A Review. Biotecnia 2025, 27, e2633–e2633. [Google Scholar] [CrossRef]
- Tudu, C. keya; Dey, A.; Pandey, D.K.; Panwar, J.S.; Nandy, S. Chapter 8 - Role of Plant Derived Extracts as Biostimulants in Sustainable Agriculture: A Detailed Study on Research Advances, Bottlenecks and Future Prospects. In New and Future Developments in Microbial Biotechnology and Bioengineering; Singh, H.B., Vaishnav, A., Eds.; Elsevier, 2022; pp. 159–179. ISBN 978-0-323-85579-2. [Google Scholar]
- Michalak, I.; Chojnacka, K.; Dmytryk, A.; Wilk, R.; Gramza, M.; Rój, E. Evaluation of Supercritical Extracts of Algae as Biostimulants of Plant Growth in Field Trials. Front. Plant Sci. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, R.C.; Alves, M.F.; Luz, J.M.Q.; Blank, A.F.; Nizio, D.A. de C.; Nogueira, P.C. de L.; Silva, S.M.; Castoldi, R. Organo-Mineral Fertilizer Improves Ocimum Basilicum Yield and Essential Oil. Plants 2025, 14, 997. [Google Scholar] [CrossRef] [PubMed]
- İsfendiyaroğlu, H.; Hanoğlu, A.; Yiğit Hanoğlu, D.; Alkaş, F.B.; Başer, K.H.C.; Özkum Yavuz, D. Chemical Characterization of the Essential Oil Compositions of Mentha Spicata and M. Longifolia Ssp. Cyprica from the Mediterranean Basin and Multivariate Statistical Analyses. Molecules 2024, 29, 1970. [Google Scholar] [CrossRef]
- Pant, P.; Pandey, S.; Dall’Acqua, S. The Influence of Environmental Conditions on Secondary Metabolites in Medicinal Plants: A Literature Review. Chemistry & Biodiversity 2021, 18, e2100345. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Kleuter, M.; Taghian Dinani, S.; Trindade, L.M.; van der Goot, A.J. The Role of Plant Age and Leaf Position on Protein Extraction and Phenolic Compounds Removal from Tomato (Solanum Lycopersicum) Leaves Using Food-Grade Solvents. Food Chemistry 2023, 406, 135072. [Google Scholar] [CrossRef]
- Shiga, T.; Shoji, K.; Shimada, H.; Hashida, S.; Goto, F.; Yoshihara, T. Effect of Light Quality on Rosmarinic Acid Content and Antioxidant Activity of Sweet Basil, Ocimum Basilicum L. Plant Biotechnology 2009, 26, 255–259. [Google Scholar] [CrossRef]
- Formica, V.; Leoni, F.; Duce, C.; González-Rivera, J.; Onor, M.; Guarnaccia, P.; Carlesi, S.; Bàrberi, P. Controlled Drought Stress Affects Rosemary Essential Oil Composition with Minimal Impact on Biomass Yield. Industrial Crops and Products 2024, 221, 119315. [Google Scholar] [CrossRef]
- Sah, A.; Naseef, P.P.; Kuruniyan, M.S.; Jain, G.K.; Zakir, F.; Aggarwal, G. A Comprehensive Study of Therapeutic Applications of Chamomile. Pharmaceuticals (Basel) 2022, 15, 1284. [Google Scholar] [CrossRef]
- Rithichai, P.; Jirakiattikul, Y.; Nambuddee, R.; Itharat, A. Effect of Salicylic Acid Foliar Application on Bioactive Compounds and Antioxidant Activity in Holy Basil (Ocimum Sanctum L.). International Journal of Agronomy 2024, 2024, 8159886. [Google Scholar] [CrossRef]
- Liebelt, D.J.; Jordan, J.T.; Doherty, C.J. Only a Matter of Time: The Impact of Daily and Seasonal Rhythms on Phytochemicals. Phytochem Rev 2019, 18, 1409–1433. [Google Scholar] [CrossRef]
- Qaderi, M.M.; Martel, A.B.; Strugnell, C.A. Environmental Factors Regulate Plant Secondary Metabolites. Plants 2023, 12, 447. [Google Scholar] [CrossRef] [PubMed]
- Dou, H.; Niu, G.; Gu, M.; Masabni, J.G. Effects of Light Quality on Growth and Phytonutrient Accumulation of Herbs under Controlled Environments. Horticulturae 2017, 3, 36. [Google Scholar] [CrossRef]
- Sun, Y.; Alseekh, S.; Fernie, A.R. Plant Secondary Metabolic Responses to Global Climate Change: A Meta-Analysis in Medicinal and Aromatic Plants. Global Change Biology 2023, 29, 477–504. [Google Scholar] [CrossRef] [PubMed]
- Ogwu, M.C.; Izah, S.C.; Joshua, M.T. Ecological and Environmental Determinants of Phytochemical Variability in Forest Trees. Phytochem Rev 2025, 24, 5109–5137. [Google Scholar] [CrossRef]
- Krakowska-Sieprawska, A.; Kiełbasa, A.; Rafińska, K.; Ligor, M.; Buszewski, B. Modern Methods of Pre-Treatment of Plant Material for the Extraction of Bioactive Compounds. Molecules 2022, 27, 730. [Google Scholar] [CrossRef]
- Belgacem, I.; Li Destri Nicosia, M.G.; Pangallo, S.; Abdelfattah, A.; Benuzzi, M.; Agosteo, G.E.; Schena, L. Pomegranate Peel Extracts as Safe Natural Treatments to Control Plant Diseases and Increase the Shelf-Life and Safety of Fresh Fruits and Vegetables. Plants 2021, 10, 453. [Google Scholar] [CrossRef]
- Shahbaz, M.U.; Arshad, M.; Mukhtar, K.; Nabi, B.G.; Goksen, G.; Starowicz, M.; Nawaz, A.; Ahmad, I.; Walayat, N.; Manzoor, M.F.; et al. Natural Plant Extracts: An Update about Novel Spraying as an Alternative of Chemical Pesticides to Extend the Postharvest Shelf Life of Fruits and Vegetables. Molecules 2022, 27, 5152. [Google Scholar] [CrossRef]
- Givi, F.; Gholami, M.; Massah, A. Application of Pomegranate Peel Extract and Essential Oil as a Safe Botanical Preservative for the Control of Postharvest Decay Caused by Penicillium Italicum and Penicillium Digitatum on “Satsuma” Mandarin. Journal of Food Safety 2019, 39, e12639. [Google Scholar] [CrossRef]
- Gidagiri, P.; P. R. G., L.; S., S. Nanoparticle-Enriched Edible Coatings with Plant Extracts and Essential Oils for Extending Postharvest Quality of Fruits and Vegetables. Journal of Advances in Biology & Biotechnology 2025, 28, 290–300. [Google Scholar] [CrossRef]
- Pangallo, S.; Li Destri Nicosia, M.G.; Scibetta, S.; Strano, M.C.; Cacciola, S.O.; Belgacem, I.; Agosteo, G.E.; Schena, L. Preharvest and Postharvest Applications of a Pomegranate Peel Extract to Control Citrus Fruit Decay During Storage and Shelf Life. Plant Disease 2021, 105, 1013–1018. [Google Scholar] [CrossRef]
- Gull, A.; Bhat, N.; Wani, S.M.; Masoodi, F.A.; Amin, T.; Ganai, S.A. Shelf Life Extension of Apricot Fruit by Application of Nanochitosan Emulsion Coatings Containing Pomegranate Peel Extract. Food Chemistry 2021, 349, 129149. [Google Scholar] [CrossRef] [PubMed]
- Yasmin, A.; Hossain, M.M.; Hafiz, M.M.H.; Rabeya, T.; Imran, S.; Sayed, S.; Gaber, A.; Hossain, A. Impact of Different Botanical Extracts on the Postharvest Biochemical Attributes and Shelf Life of Mango (Mangifera Indica L.). Qual. Assur. Saf. Crops Foods 2025, 17, 75–85. [Google Scholar] [CrossRef]
- Chitranshi, S.; Dubey, N.; Sajjad, M. Sustainable Botanical Products for Safe Post-Harvest Management of Perishable Produce: A Review. Journal of Horticulture and Postharvest Research 2020, 3, 125–140. [Google Scholar] [CrossRef]
- Rouphael, Y.; Colla, G. Synergistic Biostimulatory Action: Designing the Next Generation of Plant Biostimulants for Sustainable Agriculture. Front. Plant Sci. 2018, 9, 1–7. [Google Scholar] [CrossRef]
- Villanueva-Bermejo, D.; Siles-Sánchez, M. de las N.; Martín Hernández, D.; Rodríguez García-Risco, M.; Jaime, L.; Santoyo, S.; Fornari, T. Theoretical Framework to Evaluate Antioxidant Synergistic Effects from the Coextraction of Marjoram, Rosemary and Parsley. Food Chemistry 2024, 437, 137919. [Google Scholar] [CrossRef]
- Bayram, I.; Decker, E.A. Underlying Mechanisms of Synergistic Antioxidant Interactions during Lipid Oxidation. Trends in Food Science & Technology 2023, 133, 219–230. [Google Scholar] [CrossRef]
- El-Saadony, M.T.; Saad, A.M.; Korma, S.A.; Salem, H.M.; Abd El-Mageed, T.A.; Alkafaas, S.S.; Elsalahaty, M.I.; Elkafas, S.S.; Mosa, W.F.A.; Ahmed, A.E.; et al. Garlic Bioactive Substances and Their Therapeutic Applications for Improving Human Health: A Comprehensive Review. Front. Immunol. 2024, 15. [Google Scholar] [CrossRef]
- Hayat, S.; Ahmad, H.; Ali, M.; Ren, K.; Cheng, Z. Aqueous Garlic Extract Stimulates Growth and Antioxidant Enzymes Activity of Tomato (Solanum Lycopersicum). Scientia Horticulturae 2018, 240, 139–146. [Google Scholar] [CrossRef]
- Ali, M.; Cheng, Z.; Hayat, S.; Ahmad, H.; Ghani, M.I.; Liu, T. Foliar Spraying of Aqueous Garlic Bulb Extract Stimulates Growth and Antioxidant Enzyme Activity in Eggplant (Solanum Melongena L.). Journal of Integrative Agriculture 2019, 18, 1001–1013. [Google Scholar] [CrossRef]
- Khaldoune, K.; Fdil, N.; Ait Ali, M. Exploring Aloe Vera: A Comprehensive Review on Extraction, Chemical Composition, Biological Effects, and Its Utilization in the Synthesis of Metallic Nanoparticles. Biocatalysis and Agricultural Biotechnology 2024, 57, 103052. [Google Scholar] [CrossRef]
- Alkuwayti, M.A.; Aldayel, M.F.; Yap, Y.-K.; El Sherif, F. Exogenous Application of Aloe Vera Leaf Extract Improves Silybin Content in Silybum Marianum L. by Up-Regulating Chalcone Synthase Gene. Agriculture 2022, 12, 1649. [Google Scholar] [CrossRef]
- Melguizo-Melguizo, D.; Diaz-de-Cerio, E.; Quirantes-Piné, R.; Švarc-Gajić, J.; Segura-Carretero, A. The Potential of Artemisia Vulgaris Leaves as a Source of Antioxidant Phenolic Compounds. Journal of Functional Foods 2014, 10, 192–200. [Google Scholar] [CrossRef]
- Del Pino, A.M.; Pannacci, E.; Di Michele, A.; Bravi, E.; Marconi, O.; Tei, F.; Palmerini, C.A. Selective Inhibition of Wild Sunflower Reproduction with Mugwort Aqueous Extract, Tested on Cytosolic Ca2+ and Germination of the Pollen Grains. Plants 2021, 10, 1364. [Google Scholar] [CrossRef] [PubMed]
- Findura, P.; Kocira, S.; Hara, P.; Pawłowska, A.; Szparaga, A.; Kangalov, P. Extracts from Artemisia Vulgaris L. in Potato Cultivation—Preliminary Research on Biostimulating Effect. Agriculture 2020, 10, 356. [Google Scholar] [CrossRef]
- Pannacci, E.; Baratta, S.; Falcinelli, B.; Farneselli, M.; Tei, F. Mugwort (Artemisia Vulgaris L.) Aqueous Extract: Hormesis and Biostimulant Activity for Seed Germination and Seedling Growth in Vegetable Crops. Agriculture 2022, 12, 1329. [Google Scholar] [CrossRef]
- Chang, L.; Liu, Y.; Zhang, W.; Xu, G.; Wang, L.; Yuan, W. Antifungal Activity and Chemical Composition of Neem Seed Extract (Azadirachta Indica A.). | EBSCOhost. BioResources 2022, 17, 592. [Google Scholar] [CrossRef]
- Pretali, L.; Bernardo, L.; Butterfield, T.S.; Trevisan, M.; Lucini, L. Botanical and Biological Pesticides Elicit a Similar Induced Systemic Response in Tomato (Solanum Lycopersicum) Secondary Metabolism. Phytochemistry 2016, 130, 56–63. [Google Scholar] [CrossRef]
- Michalak, M.; Zagórska-Dziok, M.; Klimek-Szczykutowicz, M.; Szopa, A. Phenolic Profile and Comparison of the Antioxidant, Anti-Ageing, Anti-Inflammatory, and Protective Activities of Borago Officinalis Extracts on Skin Cells. Molecules 2023, 28, 868. [Google Scholar] [CrossRef]
- Zając, M.; Duda, I.; Skoczylas, Ł.; Tabaszewska, M. Potential Use of Hyssopus Officinalis and Borago Officinalis as Curing Ingredients in Pork Meat Formulations. Animals 2020, 10, 2327. [Google Scholar] [CrossRef]
- Franzoni, G.; Ferrante, A. Plant Extract Improves Quality Traits of Green and Red Lettuce Cultivars. Heliyon 2024, 10. [Google Scholar] [CrossRef]
- Brindisi, L.J.; Lyu, W.; Juliani, H.R.; Wu, Q.; Tepper, B.J.; Simon, J.E. Determination of Glucosinolates and Breakdown Products in Brassicaceae Baby Leafy Greens Using UHPLC-QTOF/MS and GC/MS. Food Chemistry Advances 2023, 3, 100389. [Google Scholar] [CrossRef]
- Øverby, A.; Stokland, R.A.; Åsberg, S.E.; Sporsheim, B.; Bones, A.M. Allyl Isothiocyanate Depletes Glutathione and Upregulates Expression of Glutathione S-Transferases in Arabidopsis Thaliana. Front. Plant Sci. 2015, 6. [Google Scholar] [CrossRef]
- Shahane, K.; Kshirsagar, M.; Tambe, S.; Jain, D.; Rout, S.; Ferreira, M.K.M.; Mali, S.; Amin, P.; Srivastav, P.P.; Cruz, J.; et al. An Updated Review on the Multifaceted Therapeutic Potential of Calendula Officinalis L. Pharmaceuticals 2023, 16, 611. [Google Scholar] [CrossRef] [PubMed]
- Sapkota, B.; Kunwar, P. A Review on Traditional Uses, Phytochemistry and Pharmacological Activities of Calendula Officinalis Linn. Natural Product Communications 2024, 19, 1934578X241259021. [Google Scholar] [CrossRef]
- Vella, F.M.; Pignone, D.; Laratta, B. The Mediterranean Species Calendula Officinalis and Foeniculum Vulgare as Valuable Source of Bioactive Compounds. Molecules 2024, 29, 3594. [Google Scholar] [CrossRef] [PubMed]
- Shu, Z.; Ji, Q.; He, T.; Zhou, D.; Zheng, S.; Zhou, H.; He, W. Combined Metabolome and Transcriptome Analyses Reveal That Growing under Red Shade Affects Secondary Metabolite Content in Huangjinya Green Tea. Front. Genet. 2024, 15. [Google Scholar] [CrossRef]
- Ahammed, G.J.; Wu, Y.; Wang, Y.; Guo, T.; Shamsy, R.; Li, X. Epigallocatechin-3-Gallate (EGCG): A Unique Secondary Metabolite with Diverse Roles in Plant-Environment Interaction. Environmental and Experimental Botany 2023, 209, 105299. [Google Scholar] [CrossRef]
- Xu, Y.; He, P.; He, B.; Chen, Z. Bioactive Flavonoids Metabolites in Citrus Species: Their Potential Health Benefits and Medical Potentials. Front. Pharmacol. 2025, 16. [Google Scholar] [CrossRef]
- Arikan, B.; Ozfidan-Konakci, C.; Yildiztugay, E.; Zengin, G.; Alp, F.N.; Elbasan, F. Exogenous Hesperidin and Chlorogenic Acid Alleviate Oxidative Damage Induced by Arsenic Toxicity in Zea Mays through Regulating the Water Status, Antioxidant Capacity, Redox Balance and Fatty Acid Composition. Environmental Pollution 2022, 292, 118389. [Google Scholar] [CrossRef]
- Figueirinha, A.; Paranhos, A.; Pérez-Alonso, J.J.; Santos-Buelga, C.; Batista, M.T. Cymbopogon Citratus Leaves: Characterization of Flavonoids by HPLC–PDA–ESI/MS/MS and an Approach to Their Potential as a Source of Bioactive Polyphenols. Food Chemistry 2008, 110, 718–728. [Google Scholar] [CrossRef]
- Baranová, B.; Kudláčková, B.; Baral, R.P.; Svojanovská, L.; Javed, Q.; Amato, G.; Caputo, L.; De Martino, L.; Francolino, R.; Chaudhary, S.K.; et al. Potential Herbicidal and Insecticidal (Beneficial) Properties of Nepalese Lemongrass Essential Oil. Chemistry & Biodiversity 2025, n/a. [Google Scholar] [CrossRef]
- Purba, R.A.P.; Laosam, P.; Pongsamai, N.; Sangsawad, P. Comparative Evaluation of Takhrai (Cymbopogon Citratus) Leaf Extracts with Commercial Antioxidants for Oxidative Stress Mitigation in Ruminants Under Heat Stress. Veterinary Sciences 2025, 12, 432. [Google Scholar] [CrossRef] [PubMed]
- Petkova, N.; Petrova, A.; Ivanov, I.; Hambarlyiska, I.; Tumbarski, Y.; Dincheva, I.; Ognyanov, M.; Denev, P. Chemical Composition of Different Extracts from Echinacea Purpurea (L.) Moench Roots and Evaluation of Their Antimicrobial Activity. ChemEngineering 2023, 7, 94. [Google Scholar] [CrossRef]
- Hu, C.; Kitts, D.D. Studies on the Antioxidant Activity of Echinacea Root Extract. J. Agric. Food Chem. 2000, 48, 1466–1472. [Google Scholar] [CrossRef]
- Hou, R.; Xu, T.; Li, Q.; Yang, F.; Wang, C.; Huang, T.; Hao, Z. Polysaccharide from Echinacea Purpurea Reduce the Oxidant Stress in Vitro and in Vivo. International Journal of Biological Macromolecules 2020, 149, 41–50. [Google Scholar] [CrossRef]
- ElSayed, A.I.; Rafudeen, M.S.; Ganie, S.A.; Hossain, M.S.; Gomaa, A.M. Seed Priming with Cypress Leaf Extract Enhances Photosynthesis and Antioxidative Defense in Zucchini Seedlings under Salt Stress. Scientia Horticulturae 2022, 293, 110707. [Google Scholar] [CrossRef]
- Wu, H.; Liu, Z.; Zhang, Y.; Gao, B.; Li, Y.; He, X.; Sun, J.; Choe, U.; Chen, P.; Blaustein, R.A.; et al. Chemical Composition of Turmeric (Curcuma Longa L.) Ethanol Extract and Its Antimicrobial Activities and Free Radical Scavenging Capacities. Foods 2024, 13, 1550. [Google Scholar] [CrossRef]
- Zhang, L.; Zengin, G.; Ozfidan-Konakci, C.; Yildiztugay, E.; Arikan, B.; Ekim, R.; Koyukan, B.; Elbasan, F.; Lucini, L. Exogenous Curcumin Mitigates As Stress in Spinach Plants: A Biochemical and Metabolomics Investigation. Plant Physiology and Biochemistry 2024, 211, 108713. [Google Scholar] [CrossRef]
- Desoky, E.-S.M.; EL-Maghraby, L.M.M.; Awad, A.E.; Abdo, A.I.; Rady, M.M.; Semida, W.M. Fennel and Ammi Seed Extracts Modulate Antioxidant Defence System and Alleviate Salinity Stress in Cowpea (Vigna Unguiculata). Scientia Horticulturae 2020, 272, 109576. [Google Scholar] [CrossRef]
- von Eggelkraut-Gottanka, S.G.; Abu Abed, S.; Müller, W.; Schmidt, P.C. Quantitative Analysis of the Active Components and the By-Products of Eight Dry Extracts of Hypericum Perforatum L. (St John’s Wort). Phytochem Anal 2002, 13, 170–176. [Google Scholar] [CrossRef]
- Rady, M.M.; Desoky, E.-S.M.; Elrys, A.S.; Boghdady, M.S. Can Licorice Root Extract Be Used as an Effective Natural Biostimulant for Salt-Stressed Common Bean Plants? South African Journal of Botany 2019, 121, 294–305. [Google Scholar] [CrossRef]
- Liu, W.-Y.; Qiu, H.; Li, H.-M.; Zhang, R.; Pan, Y.-K.; Cao, C.-Y.; Tian, J.-M.; Gao, J.-M. Prenylated Flavonoids from Hypericum Perforatum L. and Their Anti-Neuroinflammatory and Neuroprotective Activities. Industrial Crops and Products 2024, 216, 118792. [Google Scholar] [CrossRef]
- Mišina, I.; Perkons, I.; Siger, A.; Soliven, A.; Górnaś, P. Residues of St. John’s Wort (Hypericum Perforatum) Tea Infusions/Water Extracts as a Valuable Source of Tocotrienols: An Extraction Study. Applied Sciences 2025, 15, 2047. [Google Scholar] [CrossRef]
- Paun, G.; Neagu, E.; Alecu, A.; Albu, C.; Seciu-Grama, A.-M.; Radu, G.L. Evaluating the Antioxidant and Antidiabetic Properties of Medicago Sativa and Solidago Virgaurea Polyphenolic-Rich Extracts. Molecules 2024, 29, 326. [Google Scholar] [CrossRef]
- Król, A.; Railean-Plugaru, V.; Pomastowski, P.; Buszewski, B. Phytochemical Investigation of Medicago Sativa L. Extract and Its Potential as a Safe Source for the Synthesis of ZnO Nanoparticles: The Proposed Mechanism of Formation and Antimicrobial Activity. Phytochemistry Letters 2019, 31, 170–180. [Google Scholar] [CrossRef]
- Zagórska-Dziok, M.; Ziemlewska, A.; Nizioł-Łukaszewska, Z.; Bujak, T. Antioxidant Activity and Cytotoxicity of Medicago Sativa L. Seeds and Herb Extract on Skin Cells. BioResearch Open Access 2020, 9, 229–242. [Google Scholar] [CrossRef]
- Ertani, A.; Schiavon, M.; Muscolo, A.; Nardi, S. Alfalfa Plant-Derived Biostimulant Stimulate Short-Term Growth of Salt Stressed Zea Mays L. Plants. Plant Soil 2013, 364, 145–158. [Google Scholar] [CrossRef]
- Panova, N.; Gerasimova, A.; Tumbarski, Y.; Ivanov, I.; Todorova, M.; Dincheva, I.; Gentscheva, G.; Gledacheva, V.; Slavchev, V.; Stefanova, I.; et al. Metabolic Profile, Antioxidant, Antimicrobial, Contractile, and Anti-Inflammatory Potential of Moringa Oleifera Leaves (India). Life 2025, 15, 583. [Google Scholar] [CrossRef]
- Mei, Y.; Sun, H.; Du, G.; Wang, X.; Lyu, D. Exogenous Chlorogenic Acid Alleviates Oxidative Stress in Apple Leaves by Enhancing Antioxidant Capacity. Scientia Horticulturae 2020, 274, 109676. [Google Scholar] [CrossRef]
- Mir-Cerdà, A.; Granados, M.; Saurina, J.; Sentellas, S. Olive Tree Leaves as a Great Source of Phenolic Compounds: Comprehensive Profiling of NaDES Extracts. Food Chemistry 2024, 456, 140042. [Google Scholar] [CrossRef]
- Benavente-Garcı́a, O.; Castillo, J.; Lorente, J.; Ortuño, A.; Del Rio, J.A. Antioxidant Activity of Phenolics Extracted from Olea Europaea L. Leaves. Food Chemistry 2000, 68, 457–462. [Google Scholar] [CrossRef]
- Hashmi, M.A.; Khan, A.; Hanif, M.; Farooq, U.; Perveen, S. Traditional Uses, Phytochemistry, and Pharmacology of Olea Europaea (Olive). Evidence-Based Complementary and Alternative Medicine 2015, 2015, 541591. [Google Scholar] [CrossRef] [PubMed]
- Wanas, A.; Hamada, M.; Shaimaa, M. Investigation the Ability of Olive Leaf Extract Enhance Growth and Physio-Biochemical Performance of Faba Bean Plants Under Salt Stress Conditions. Damietta Journal of Agricultural Sciences 2025, 4, 35–49. [Google Scholar] [CrossRef]
- Siddiqui, S.A.; Singh, S.; Nayik, G.A. Bioactive Compounds from Pomegranate Peels - Biological Properties, Structure–Function Relationships, Health Benefits and Food Applications – A Comprehensive Review. Journal of Functional Foods 2024, 116, 106132. [Google Scholar] [CrossRef]
- Benchagra, L.; Berrougui, H.; Islam, M.O.; Ramchoun, M.; Boulbaroud, S.; Hajjaji, A.; Fulop, T.; Ferretti, G.; Khalil, A. Antioxidant Effect of Moroccan Pomegranate (Punica Granatum L. Sefri Variety) Extracts Rich in Punicalagin against the Oxidative Stress Process. Foods 2021, 10, 2219. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, M.; Yu, G.; Pu, J.; Tian, K.; Tang, X.; Du, Y.; Wu, H.; Hu, J.; Luo, X.; et al. Comparative Analysis of the Phenolic Contents and Antioxidant Activities of Different Parts of Two Pomegranate (Punica Granatum L.) Cultivars: ‘Tunisia’ and ‘Qingpi.’ Front. Plant Sci. 2023, 14. [Google Scholar] [CrossRef]
- Mena, P.; Cirlini, M.; Tassotti, M.; Herrlinger, K.A.; Dall’Asta, C.; Del Rio, D. Phytochemical Profiling of Flavonoids, Phenolic Acids, Terpenoids, and Volatile Fraction of a Rosemary (Rosmarinus Officinalis L.) Extract. Molecules 2016, 21, 1576. [Google Scholar] [CrossRef]
- Munné-Bosch, S.; Mueller, M.; Schwarz, K.; Alegre, L. Diterpenes and Antioxidative Protection in Drought-Stressed Salvia Officinalis Plants. Journal of Plant Physiology 2001, 158, 1431–1437. [Google Scholar] [CrossRef]
- Gligorić, E.; Igić, R.; Teofilović, B.; Grujić-Letić, N. Phytochemical Screening of Ultrasonic Extracts of Salix Species and Molecular Docking Study of Salix-Derived Bioactive Compounds Targeting Pro-Inflammatory Cytokines. International Journal of Molecular Sciences 2023, 24, 11848. [Google Scholar] [CrossRef]
- Shahzad, A.; Hameed, S.; Qin, M.; Li, H.; Zafar, S.; Siddiqui, S.; Sattar, S.; Mahmood, Z.; Mehwish, S. Cadmium (Cd) Detoxification and Activation of Plant Defense Enzymes in Wheat (Triticum Aestivum) through the Use of Endophytic Bacillus Thuringiensis and Salix Alba Root Powder. Environmental Pollution 2025, 364, 125147. [Google Scholar] [CrossRef]
- Radusiene, J.; Marska, M.; Ivanauskas, L.; Jakstas, V.; Karpaviciene, B. Assessment of Phenolic Compound Accumulation in Two Widespread Goldenrods. Industrial Crops and Products 2015, 63, 158–166. [Google Scholar] [CrossRef]
- Godlewska, K.; Biesiada, A.; Michalak, I.; Pacyga, P. The Effect of Plant-Derived Biostimulants on White Head Cabbage Seedlings Grown under Controlled Conditions. Sustainability 2019, 11, 5317. [Google Scholar] [CrossRef]
- Krüzselyi, D.; Bakonyi, J.; Ott, P.G.; Darcsi, A.; Csontos, P.; Morlock, G.E.; Móricz, Á.M. Goldenrod Root Compounds Active against Crop Pathogenic Fungi. J. Agric. Food Chem. 2021, 69, 12686–12694. [Google Scholar] [CrossRef] [PubMed]
- Jaffar, H.M.; Al-Asmari, F.; Khan, F.A.; Rahim, M.A.; Zongo, E. Silymarin: Unveiling Its Pharmacological Spectrum and Therapeutic Potential in Liver Diseases—A Comprehensive Narrative Review. Food Science & Nutrition 2024, 12, 3097–3111. [Google Scholar] [CrossRef] [PubMed]
- Buzón-Durán, L.; Martín-Gil, J.; Ramos-Sánchez, M. del C.; Pérez-Lebeña, E.; Marcos-Robles, J.L.; Fombellida-Villafruela, Á.; Martín-Ramos, P. Antifungal Activity against Fusarium Culmorum of Stevioside, Silybum Marianum Seed Extracts, and Their Conjugate Complexes. Antibiotics 2020, 9, 440. [Google Scholar] [CrossRef] [PubMed]
- Andrzejewska, J.; Martinelli, T.; Sadowska, K. Silybum Marianum: Non-Medical Exploitation of the Species. Annals of Applied Biology 2015, 167, 285–297. [Google Scholar] [CrossRef]
- Wirngo, F.E.; Lambert, M.N.; Jeppesen, P.B. The Physiological Effects of Dandelion (Taraxacum Officinale) in Type 2 Diabetes. Rev Diabet Stud 2016, 13, 113–131. [Google Scholar] [CrossRef]
- Hu, C.; Kitts, D.D. Antioxidant, Prooxidant, and Cytotoxic Activities of Solvent-Fractionated Dandelion (Taraxacum Officinale) Flower Extracts in Vitro. J. Agric. Food Chem. 2003, 51, 301–310. [Google Scholar] [CrossRef]
- Lis, B.; Jedrejek, D.; Rywaniak, J.; Soluch, A.; Stochmal, A.; Olas, B. Flavonoid Preparations from Taraxacum Officinale L. Fruits—A Phytochemical, Antioxidant and Hemostasis Studies. Molecules 2020, 25, 5402. [Google Scholar] [CrossRef]
- Ivanov, Y.; Atanasova, M.; Godjevargova, T. Nutritional and Functional Values of Grape Seed Flour and Extract for Production of Antioxidative Dietary Supplements and Functional Foods. Molecules 2025, 30, 2029. [Google Scholar] [CrossRef]
- Plaskova, A.; Mlcek, J. New Insights of the Application of Water or Ethanol-Water Plant Extract Rich in Active Compounds in Food. Front. Nutr. 2023, 10. [Google Scholar] [CrossRef]
- Balea, Ş.S.; Pârvu, A.E.; Pârvu, M.; Vlase, L.; Dehelean, C.A.; Pop, T.I. Antioxidant, Anti-Inflammatory and Antiproliferative Effects of the Vitis Vinifera L. Var. Fetească Neagră and Pinot Noir Pomace Extracts. Front. Pharmacol. 2020, 11. [Google Scholar] [CrossRef]
- Mao, Q.-Q.; Xu, X.-Y.; Cao, S.-Y.; Gan, R.-Y.; Corke, H.; Beta, T.; Li, H.-B. Bioactive Compounds and Bioactivities of Ginger (Zingiber Officinale Roscoe). Foods 2019, 8, 185. [Google Scholar] [CrossRef] [PubMed]
- Ezeonu, C.S.; Tatah, S.V.; Imo, C.; Yakubu, O.E.; Garba, Q.H.; Arowora, K.; Umaru, I.J.; Abah, M.A.; Abu, M.S.; Yohanna, E.R.; et al. Antioxidant Potential of Ginger Extract on Metals (Lead, Cadmium, and Boron) Induced Oxidative Stress in Maize Plant. Asian Journal of Tropical Biotechnology 2022, 19. [Google Scholar] [CrossRef]
- Godlewska, K.; Biesiada, A.; Michalak, I.; Pacyga, P. The Effect of Botanical Extracts Obtained through Ultrasound-Assisted Extraction on White Head Cabbage (Brassica Oleracea L. Var. Capitata L.) Seedlings Grown under Controlled Conditions. Sustainability 2020, 12, 1871. [Google Scholar] [CrossRef]
- Juan-Cabot, A.; Carrillo, L.; Fullana-Pericàs, M.; Galmés, J.; Medina, J.; Conesa, M.À. Mediterranean Tomato Landraces Exhibit Genotype-Specific Transcriptomic Responses to Water Stress. Physiologia Plantarum 2025, 177, e70696. [Google Scholar] [CrossRef]
- Aswathi, K.P.R.; Ul-Allah, S.; Puthur, J.T.; Siddique, K.H.M.; Frei, M.; Farooq, M. The Plant Mind: Unraveling Abiotic Stress Priming, Memory, and Adaptation. Physiologia Plantarum 2025, 177, e70372. [Google Scholar] [CrossRef]
- Dsouza, A.; Dixon, M.; Shukla, M.; Graham, T. Harnessing Controlled-Environment Systems for Enhanced Production of Medicinal Plants. Journal of Experimental Botany 2025, 76, 76–93. [Google Scholar] [CrossRef]
- Punetha, A.; Kumar, D.; Suryavanshi, P.; Padalıa, R.; K.t, V. Environmental Abiotic Stress and Secondary Metabolites Production in Medicinal Plants: A Review. J Agr Sci-Tarim Bili 2022, 28, 351–362. [Google Scholar] [CrossRef]
- Wu, X.; Yuan, J.; Luo, A.; Chen, Y.; Fan, Y. Drought Stress and Re-Watering Increase Secondary Metabolites and Enzyme Activity in Dendrobium Moniliforme. Industrial Crops and Products 2016, 94, 385–393. [Google Scholar] [CrossRef]
- Alami, M.M.; Guo, S.; Mei, Z.; Yang, G.; Wang, X.; Alami, M.M.; Guo, S.; Mei, Z.; Yang, G.; Wang, X. Environmental Factors on Secondary Metabolism in Medicinal Plants: Exploring Accelerating Factors. Med. Plant Biol. 2024, 3, e016. [Google Scholar] [CrossRef]
- Malejane, D.N.; Tinyani, P.; Soundy, P.; Sultanbawa, Y.; Sivakumar, D. Deficit Irrigation Improves Phenolic Content and Antioxidant Activity in Leafy Lettuce Varieties. Food Science & Nutrition 2018, 6, 334–341. [Google Scholar] [CrossRef]
- Nasrallah, A.K.; Atia, M.A.M.; Abd El-Maksoud, R.M.; Kord, M.A.; Fouad, A.S. Salt Priming as a Smart Approach to Mitigate Salt Stress in Faba Bean (Vicia Faba L.). Plants 2022, 11, 1610. [Google Scholar] [CrossRef] [PubMed]
- Ullah, M.S.; Mahmood, A.; Alawadi, H.F.N.; Seleiman, M.F.; Khan, B.A.; Javaid, M.M.; Wahid, A.; Abdullah, F.; Wasonga, D.O. Silicon-Mediated Modulation of Maize Growth, Metabolic Responses, and Antioxidant Mechanisms under Saline Conditions. BMC Plant Biology 2025, 25, 3. [Google Scholar] [CrossRef]
- Skrypnik, L.; Feduraev, P.; Golubkina, N.; Maslennikov, P.; Antipina, M.; Katserov, D.; Nebreeva, S.; Murariu, O.C.; Tallarita, A.V.; Caruso, G. Selenium Improves the Nutritional and Antioxidant Properties of Oregano (Origanum Vulgare L.) Grown in Hydroponics. Horticulturae 2024, 10, 1320. [Google Scholar] [CrossRef]
- Shalaby, O.A. Iodine Application Induces the Antioxidant Defense System, Alleviates Salt Stress, Reduces Nitrate Content, and Increases the Nutritional Value of Lettuce Plants. Functional Plant Biol. 2025, 52, FP24273. [Google Scholar] [CrossRef]
- Medrano-Macías, J.M.; López Caltzontzit, M.G.; Rivas-Martínez, E.N.; Narváez-Ortiz, W.A.; Benavides-Mendoza, A.; Martínez-Lagunes, P. Enhancement to Salt Stress Tolerance in Strawberry Plants by Iodine Products Application. Agronomy 2021, 11, 602. [Google Scholar] [CrossRef]
- García-Fuentes, J.E.; Herrera-Castellanos, B.F.; Rivas-Martínez, E.N.; Narváes-Ortiz, W.A.; Benavides-Mendoza, A.; Medrano-Macías, J. Outcomes of Foliar Iodine Application on Growth, Minerals and Antioxidants in Tomato Plants under Salt Stress. Folia Horticulturae 2022, 34, 1–11. [Google Scholar] [CrossRef]
- Viršilė, A.; Gudžinskaitė, I.; Laužikė, K.; Kudirka, G.; Pukalskas, A.; Samuolienė, G. Light Intensity Effects on Productivity and Post-Harvest Quality in Perilla Frutescens Cultivated in CEA. Agriculture 2024, 14, 2079. [Google Scholar] [CrossRef]
- Doneva, D.; Pál, M.; Szalai, G.; Vasileva, I.; Brankova, L.; Misheva, S.; Janda, T.; Peeva, V. Manipulating the Light Spectrum to Increase the Biomass Production, Physiological Plasticity and Nutritional Quality of Eruca Sativa L. Plant Physiology and Biochemistry 2024, 217, 109218. [Google Scholar] [CrossRef]
- Yeom, M.-S.; Oh, M.-M. Tailoring Red, Green, and Blue LED Spectra to Improve Yield and Quality of Glehnia Littoralis. Sci Rep 2025, 15, 23268. [Google Scholar] [CrossRef]
- Faria de Souza, A.F.; Calderín García, A.; Nivaldo de Oliveira Sátiro, J.; Ribeiro de Lima, B.; Silvestre Fernandes, M.; Louro Berbara, R.L.; Azevedo Santos, L. Humic Acid Regulates Root Growth through ROS-Dependent Pathway and Hormone Signaling in Rice. J. Agric. Food Chem. 2025. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Shahrajabian, M.H.; Petropoulos, S.A.; Shahrajabian, N. Developing Sustainable Agriculture Systems in Medicinal and Aromatic Plant Production by Using Chitosan and Chitin-Based Biostimulants. Plants 2023, 12, 2469. [Google Scholar] [CrossRef] [PubMed]
- Saharkhiz, M.J.; Goudarzi, T. Foliar Application of Salicylic Acid Changes Essential Oil Content and Chemical Compositions of Peppermint (Mentha Piperita L.). Journal of Essential Oil Bearing Plants 2014, 17, 435–440. [Google Scholar] [CrossRef]
- Wen, L.; Zhang, Z.; Sun, D.-W.; Sivagnanam, S.P.; Tiwari, B.K. Combination of Emerging Technologies for the Extraction of Bioactive Compounds. Critical Reviews in Food Science and Nutrition 2020, 60, 1826–1841. [Google Scholar] [CrossRef]
- Usman, I.; Hussain, M.; Imran, A.; Afzaal, M.; Saeed, F.; Javed, M.; Afzal, A.; Ashfaq, I.; Al Jbawi, E.; Saewan, A.S. Traditional and Innovative Approaches for the Extraction of Bioactive Compounds. International Journal of Food Properties 2022, 25, 1215–1233. [Google Scholar] [CrossRef]
- Allegra, M. Redox Systems, Oxidative Stress, and Antioxidant Defences in Health and Disease. Antioxidants 2021, 10, 1955. [Google Scholar] [CrossRef]
- Luo, M.; Zheng, Y.; Tang, S.; Gu, L.; Zhu, Y.; Ying, R.; Liu, Y.; Ma, J.; Guo, R.; Gao, P.; et al. Radical Oxygen Species: An Important Breakthrough Point for Botanical Drugs to Regulate Oxidative Stress and Treat the Disorder of Glycolipid Metabolism. Front. Pharmacol. 2023, 14. [Google Scholar] [CrossRef]
- Stefanson, A.L.; Bakovic, M. Dietary Regulation of Keap1/Nrf2/ARE Pathway: Focus on Plant-Derived Compounds and Trace Minerals. Nutrients 2014, 6, 3777–3801. [Google Scholar] [CrossRef]
- Di Giacomo, M.; Zara, V.; Bergamo, P.; Ferramosca, A. Crosstalk between Mitochondrial Metabolism and Oxidoreductive Homeostasis: A New Perspective for Understanding the Effects of Bioactive Dietary Compounds. Nutrition Research Reviews 2020, 33, 90–101. [Google Scholar] [CrossRef]
- Kaulmann, A.; Bohn, T. Carotenoids, Inflammation, and Oxidative Stress—Implications of Cellular Signaling Pathways and Relation to Chronic Disease Prevention. Nutrition Research 2014, 34, 907–929. [Google Scholar] [CrossRef] [PubMed]
- Juárez-Maldonado, A.; Ortega-Ortíz, H.; Morales-Díaz, A.B.; González-Morales, S.; Morelos-Moreno, Á.; Cabrera-De la Fuente, M.; Sandoval-Rangel, A.; Cadenas-Pliego, G.; Benavides-Mendoza, A. Nanoparticles and Nanomaterials as Plant Biostimulants. International Journal of Molecular Sciences 2019, 20, 162. [Google Scholar] [CrossRef] [PubMed]
- Bhatla, S.C.; Lal, M.A. Signal Perception and Transduction Mechanisms. In Plant Physiology, Development and Metabolism; Bhatla, S.C., Lal, M.A., Eds.; Springer Nature: Singapore, 2023; pp. 517–542. ISBN 978-981-9957-36-1. [Google Scholar]
- Ravi, B.; Foyer, C.H.; Pandey, G.K. The Integration of Reactive Oxygen Species (ROS) and Calcium Signalling in Abiotic Stress Responses. Plant, Cell & Environment 2023, 46, 1985–2006. [Google Scholar] [CrossRef] [PubMed]
- Mansuri, M.L.; Parihar, P.; Solanki, I.; Parihar, M.S. Flavonoids in Modulation of Cell Survival Signalling Pathways. Genes Nutr 2014, 9, 400. [Google Scholar] [CrossRef]
- Ranf, S.; Eschen-Lippold, L.; Pecher, P.; Lee, J.; Scheel, D. Interplay between Calcium Signalling and Early Signalling Elements during Defence Responses to Microbe- or Damage-Associated Molecular Patterns. The Plant Journal 2011, 68, 100–113. [Google Scholar] [CrossRef]
- Mittler, R.; Zandalinas, S.I.; Fichman, Y.; Van Breusegem, F. Reactive Oxygen Species Signalling in Plant Stress Responses. Nat Rev Mol Cell Biol 2022, 23, 663–679. [Google Scholar] [CrossRef]
- Melino, S.; Leo, S.; Toska Papajani, V. Natural Hydrogen Sulfide Donors from Allium Sp. as a Nutraceutical Approach in Type 2 Diabetes Prevention and Therapy. Nutrients 2019, 11, 1581. [Google Scholar] [CrossRef]
- Piragine, E.; Malanima, M.A.; Ceccanti, C.; Guidi, L.; Martelli, A.; Lucenteforte, E.; Calderone, V. Alliaceae versus Brassicaceae for Dyslipidemia: State of the Art and Future Perspectives. Systematic Review and Meta-Analysis of Clinical Studies. Phytotherapy Research 2024, 38, 5765–5781. [Google Scholar] [CrossRef]
- Parra-Espejel, E. de J.; Hipólito-Jiménez, S.J.; Galicia González, M.D.; Montes Rojas, D.; Llera-Rojas, V.G.; Velázquez-Moyado, J.A. Nephroprotective Activity of Eysenhardtia Polystachya through Antioxidant, H2S, and NO Pathways. Naunyn-Schmiedeberg’s Arch Pharmacol 2025. [Google Scholar] [CrossRef]
- Planchet, E.; Kaiser, W.M. Nitric Oxide Production in Plants: Facts and Fictions. Plant Signaling & Behavior 2006, 1, 46–51. [Google Scholar] [CrossRef]
- Muchová, L.; Šranková, M.; Balasubramani, S.; Mehta, P.; Vlachopoulou, D.; Kapoor, A.; Ramundo, A.; Jézéquel, Y.A.; Bożek, I.; Hurtová, M.; et al. Carbon Monoxide-Releasing Activity of Plant Flavonoids. J. Agric. Food Chem. 2025, 73, 1308–1318. [Google Scholar] [CrossRef]
- Khan, R.A.; Abbas, N. Role of Epigenetic and Post-Translational Modifications in Anthocyanin Biosynthesis: A Review. Gene 2023, 887, 147694. [Google Scholar] [CrossRef] [PubMed]
- Abdulraheem, M.I.; Xiong, Y.; Moshood, A.Y.; Cadenas-Pliego, G.; Zhang, H.; Hu, J. Mechanisms of Plant Epigenetic Regulation in Response to Plant Stress: Recent Discoveries and Implications. Plants 2024, 13, 163. [Google Scholar] [CrossRef]
- Cimmino, L.; Staiti, A.; Carputo, D.; Docimo, T.; D’Amelia, V.; Aversano, R. Adaptable Alchemy: Exploring the Flexibility of Specialized Metabolites to Environmental Perturbations Through Post-Translational Modifications (PTMs). Plants 2025, 14, 489. [Google Scholar] [CrossRef] [PubMed]
- Bhuvaneshwari, V.; Paul, P.K. Dual Specific Protein Kinases Mediate Neem Fruit Extract-Induced Defense Gene Expression in Solanum Lycopersicum L. South African Journal of Botany 2023, 155, 16–26. [Google Scholar] [CrossRef]
- Demasi, M.; Augusto, O.; Bechara, E.J.H.; Bicev, R.N.; Cerqueira, F.M.; da Cunha, F.M.; Denicola, A.; Gomes, F.; Miyamoto, S.; Netto, L.E.S.; et al. Oxidative Modification of Proteins: From Damage to Catalysis, Signaling, and Beyond. Antioxidants & Redox Signaling 2021, 35, 1016–1080. [Google Scholar] [CrossRef]
- Feng, D.; Zhang, W.; Zheng, C.; Tang, G.; Adili, Y.; Ge, C.; Xu, W. Mechanisms of Plant Extracts in Alleviating Drought and Saline-Alkali Stress in Plants. Industrial Crops and Products 2025, 233, 121346. [Google Scholar] [CrossRef]
- Pacyga, K.; Pacyga, P.; Boba, A.; Kozak, B.; Wolko, Ł.; Kochneva, Y.; Michalak, I. Potential of Plant-Based Extracts to Alleviate Sorbitol-Induced Osmotic Stress in Cabbage Seedlings. Plants 2024, 13, 843. [Google Scholar] [CrossRef]
- Desoky, E.-S.M.; ElSayed, A.I.; Merwad, A.-R.M.A.; Rady, M.M. Stimulating Antioxidant Defenses, Antioxidant Gene Expression, and Salt Tolerance in Pisum Sativum Seedling by Pretreatment Using Licorice Root Extract (LRE) as an Organic Biostimulant. Plant Physiology and Biochemistry 2019, 142, 292–302. [Google Scholar] [CrossRef]
- Kutasy, B.; Kiniczky, M.; Decsi, K.; Kálmán, N.; Hegedűs, G.; Alföldi, Z.P.; Virág, E. ‘Garlic-Lipo’4Plants: Liposome-Encapsulated Garlic Extract Stimulates ABA Pathway and PR Genes in Wheat (Triticum Aestivum). Plants 2023, 12, 743. [Google Scholar] [CrossRef]
- Alum, E.U.; Udechukwu, C.D.; Obasi, D.C. RNA Signaling in Medicinal Plants: An Overlooked Mechanism for Phytochemical Regulation. Biochemistry and Biophysics Reports 2025, 42, 102032. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Xu, S.; Mei, Y.; Li, J.; Gu, Y.; Zhang, W.; Wang, J. MicroRNAs in Medicinal Plants. International Journal of Molecular Sciences 2022, 23, 10477. [Google Scholar] [CrossRef] [PubMed]
- Kirk, H.; Cefalu, W.T.; Ribnicky, D.; Liu, Z.; Eilertsen, K.J. Botanicals as Epigenetic Modulators for Mechanisms Contributing to Development of Metabolic Syndrome. Metabolism 2008, 57, S16–S23. [Google Scholar] [CrossRef]
- Namkoong, J.; Kern, D.; Knaggs, H.E. Assessment of Human Skin Gene Expression by Different Blends of Plant Extracts with Implications to Periorbital Skin Aging. International Journal of Molecular Sciences 2018, 19, 3349. [Google Scholar] [CrossRef]
- Karwasra, R.; Khanna, K.; Bhardwaj, S.; Sharma, N.; Janakiraman, A.K.; Sundarapandian, R.; Singh, S. Chapter 13 - Harnessing Plant-Derived Biosynthetic Nanomaterials for Epigenetic Modulation in Cancer Therapy. In Cancer Epigenetics and Nanomedicine; Kesharwani, P., Thakur, C., Eds.; Academic Press, 2024; pp. 279–295. ISBN 978-0-443-13209-4. [Google Scholar]
- Nemzer, B.V.; Centner, C.; Zdzieblik, D.; Fink, B.; Hunter, J.M.; König, D. Oxidative Stress or Redox Signalling – New Insights into the Effects of a Proprietary Multifunctional Botanical Dietary Supplement. Free Radical Research 2018, 52, 362–372. [Google Scholar] [CrossRef] [PubMed]
- Trewavas, A. The Foundations of Plant Intelligence. Interface Focus 2017, 7, 20160098. [Google Scholar] [CrossRef]
- Calvo, P.; Friston, K. Predicting Green: Really Radical (Plant) Predictive Processing. Journal of The Royal Society Interface 2017, 14, 20170096. [Google Scholar] [CrossRef]
- EL Boukhari, M.E.M.; Barakate, M.; Bouhia, Y.; Lyamlouli, K. Trends in Seaweed Extract Based Biostimulants: Manufacturing Process and Beneficial Effect on Soil-Plant Systems. Plants 2020, 9, 359. [Google Scholar] [CrossRef]
- Ali, O.; Ramsubhag, A.; Jayaraman, J. Biostimulant Properties of Seaweed Extracts in Plants: Implications towards Sustainable Crop Production. Plants 2021, 10, 531. [Google Scholar] [CrossRef]
- Son, S.; Park, S.R. Plant Translational Reprogramming for Stress Resilience. Front. Plant Sci. 2023, 14. [Google Scholar] [CrossRef]
- Ertani, A.; Schiavon, M.; Nardi, S. Transcriptome-Wide Identification of Differentially Expressed Genes in Solanum lycopersicon L. in Response to an Alfalfa-Protein Hydrolysate Using Microarrays. Front. Plant Sci. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Majeed, A.; Singh, A.; Sharma, R.K.; Jaitak, V.; Bhardwaj, P. Comprehensive Temporal Reprogramming Ensures Dynamicity of Transcriptomic Profile for Adaptive Response in Taxus Contorta. Mol Genet Genomics 2020, 295, 1401–1414. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Fu, X. Reprogramming of Plant Central Metabolism in Response to Abiotic Stresses: A Metabolomics View. International Journal of Molecular Sciences 2022, 23, 5716. [Google Scholar] [CrossRef] [PubMed]
- Sugimoto, K.; Xu, L.; Paszkowski, U.; Hayashi, M. Multifaceted Cellular Reprogramming at the Crossroads Between Plant Development and Biotic Interactions. Plant and Cell Physiology 2018, 59, 651–655. [Google Scholar] [CrossRef]
- Karalija, E.; Macanović, A.; Ibragić, S. Revisiting Traditional Medicinal Plants: Integrating Multiomics, In Vitro Culture, and Elicitation to Unlock Bioactive Potential. Plants 2025, 14, 2029. [Google Scholar] [CrossRef]
- Conrath, U. Chapter 9 Priming of Induced Plant Defense Responses. In Advances in Botanical Research; Advances in Botanical Research; Academic Press, 2009; Vol. 51, pp. 361–395. [Google Scholar]
- Mutlu-Durak, H.; Yildiz Kutman, B. Seed Treatment with Biostimulants Extracted from Weeping Willow (Salix Babylonica) Enhances Early Maize Growth. Plants 2021, 10, 1449. [Google Scholar] [CrossRef]
- Monterisi, S.; Zhang, L.; Garcia-Perez, P.; Alzate Zuluaga, M.Y.; Ciriello, M.; El-Nakhel, C.; Buffagni, V.; Cardarelli, M.; Colla, G.; Rouphael, Y.; et al. Integrated Multi-Omic Approach Reveals the Effect of a Graminaceae-Derived Biostimulant and Its Lighter Fraction on Salt-Stressed Lettuce Plants. Sci Rep 2024, 14, 10710. [Google Scholar] [CrossRef]
- Teklić, T.; Parađiković, N.; Špoljarević, M.; Zeljković, S.; Lončarić, Z.; Lisjak, M. Linking Abiotic Stress, Plant Metabolites, Biostimulants and Functional Food. Annals of Applied Biology 2020, 178, 169–191. [Google Scholar] [CrossRef]
- De Saeger, J.; Van Praet, S.; Vereecke, D.; Park, J.; Jacques, S.; Han, T.; Depuydt, S. Toward the Molecular Understanding of the Action Mechanism of Ascophyllum Nodosum Extracts on Plants. J Appl Phycol 2020, 32, 573–597. [Google Scholar] [CrossRef]
- Hönig, M.; Roeber, V.M.; Schmülling, T.; Cortleven, A. Chemical Priming of Plant Defense Responses to Pathogen Attacks. Front. Plant Sci. 2023, 14. [Google Scholar] [CrossRef]
- Figueroa-Macías, J.P.; García, Y.C.; Núñez, M.; Díaz, K.; Olea, A.F.; Espinoza, L. Plant Growth-Defense Trade-Offs: Molecular Processes Leading to Physiological Changes. International Journal of Molecular Sciences 2021, 22, 693. [Google Scholar] [CrossRef]
- Staszek, P.; Krasuska, U.; Wal, A.; Zak, J.; Gniazdowska, A. 12 - NO and Metabolic Reprogramming under Phytotoxicity Stress. In Nitric Oxide in Plant Biology; Singh, Pratap, Singh, V., Tripathi, S., Romero-Puertas, D.K., Sandalio, M.C.L.M., Eds.; Academic Press, 2022; pp. 297–318. ISBN 978-0-12-818797-5. [Google Scholar]
- Serag, A.; Salem, M.A.; Gong, S.; Wu, J.-L.; Farag, M.A. Decoding Metabolic Reprogramming in Plants under Pathogen Attacks, a Comprehensive Review of Emerging Metabolomics Technologies to Maximize Their Applications. Metabolites 2023, 13, 424. [Google Scholar] [CrossRef] [PubMed]
- Karasov, T.L.; Chae, E.; Herman, J.J.; Bergelson, J. Mechanisms to Mitigate the Trade-Off between Growth and Defense. The Plant Cell 2017, 29, 666–680. [Google Scholar] [CrossRef] [PubMed]
- Marchev, A.S.; Vasileva, L.V.; Amirova, K.M.; Savova, M.S.; Balcheva-Sivenova, Z.P.; Georgiev, M.I. Metabolomics and Health: From Nutritional Crops and Plant-Based Pharmaceuticals to Profiling of Human Biofluids. Cell. Mol. Life Sci. 2021, 78, 6487–6503. [Google Scholar] [CrossRef] [PubMed]
- Panossian, A.; Seo, E.-J.; Efferth, T. Novel Molecular Mechanisms for the Adaptogenic Effects of Herbal Extracts on Isolated Brain Cells Using Systems Biology. Phytomedicine 2018, 50, 257–284. [Google Scholar] [CrossRef]
- R. Dias, T.; L. Bernardino, R.; J. Meneses, M.; Sousa, M.; Sá, R.; G. Alves, M.; M. Silva, B.; Oliveira, F.P. Emerging Potential of Natural Products as an Alternative Strategy to Pharmacological Agents Used Against Metabolic Disorders. Current Drug Metabolism 2016, 17, 582–597. [Google Scholar] [CrossRef]
- Oyenihi, O.R.; Oyenihi, A.B.; Erhabor, J.O.; Matsabisa, M.G.; Oguntibeju, O.O. Unravelling the Anticancer Mechanisms of Traditional Herbal Medicines with Metabolomics. Molecules 2021, 26, 6541. [Google Scholar] [CrossRef]
- Lim, J.-S.; Yu, M.-H.; Choi, D.K.; Kim, H.W.; Park, S.-H.; Sin, S.-I.; Kim, J.-S. Enhanced Antioxidant and Antiproliferative Activities of Apple and Korean Green Chili Pepper Extracts Cultivated with Mineral Supplementation. Foods 2025, 14, 2685. [Google Scholar] [CrossRef]
- Poutaraud, A.; Girardin, P. Improvement of Medicinal Plant Quality: A Hypericum Perforatum Literature Review as an Example. Plant Genetic Resources 2005, 3, 178–189. [Google Scholar] [CrossRef]
- Nazari, M.R.; Abdossi, V.; Hargalani, F.Z.; Larijani, K. Antioxidant Potential and Essential Oil Properties of Hypericum Perforatum L. Assessed by Application of Selenite and Nano-Selenium. Sci Rep 2022, 12, 6156. [Google Scholar] [CrossRef]
| Botanical Extract | Representative Chemical Components | Representative Redox Impact of Chemical Component(s) in Planta |
|---|---|---|
| Allium sativum bulb | Alliin, diallyl sulfide, ajoene, carvacrol, geraniol, quercetin, apigenin, rutin [50] | The aqueous garlic extract enhances the activity of antioxidant enzymes, including SOD and PRX, in tomatoes and eggplant [51,52]. |
| Aloe vera inner-leaf gel | Quercetin, kaempherol, aloesin, rutin, caffeic acid, cinnamic acid, vanillic acid, and acemannan [53] | Application of 60 mL L-1 of Aloe vera extract enhanced the growth and productivity of Silybum marianum, whereas a concentration of 40 mL L-1 A. vera extract increased both silybin content and chalcone synthase gene expression [54]. |
| Artemisia vulgaris leaves | Artemisinic acid, rutin, luteolin, kaempferol-3-O-glucoside, tracheloside [55], hydroxycinnamic acids (caffeic, sinapic, p/m-coumaric, ferulic, homovanillic and chlorogenic) and hydroxybenzoic acids (p-hydroxybenzoic, gallic, syringic, salicylic and gentisic) [56]. | Artemisia vulgaris extract elevated the levels of chlorophyll, carotenoids, proline, and polyphenols in potato plants [57]. A. vulgaris extract (leaves and stems) improved germination and seedling growth in some plant species, while it showed no impact in others [58]. |
| Azadirachta indica (neem) seeds | Azadirachtin, astaxanthin, cinobufagin, anodendroside, marinobufagin [59] | Azadirachtin (2.4% azaridachtin A) application to leaves triggers a defense response in tomatoes comparable to that elicited by Bacillus subtilis through Induced Systemic Resistance (ISR). Foliar treatment with B. subtilis activated ISR via the jasmonic acid signaling pathway and promoted the synthesis of secondary metabolites, including flavonoids, phytoalexins, and auxins. Alterations in sterol and terpene profiles, along with elevated glucosinolate levels, were also detected [60]. |
| Borago officinalis flowers | Rosmarinic acid, astragalin, rutin, linoleic acid [61] | Borago officinalis extracts possess antioxidant properties, enabling their use in food preservation and offering broad therapeutic potential [62]. Its use as a agricultural botanical extract appears to be limited [63]. |
| Brassica juncea seeds | Glucosinolates: 4-hydroxyglucobrassicin, glucobarbarin, glucobrassicanapin, glucoerucin, gluconapin, gluconasturtiin and neoglucobrassicin [64] |
Allyl isothiocyanate (AITC) is a phytochemical associated with plant defense in plants from the Brassicaceae family. AITC has long been recognized as a countermeasure against external threats and is also involved in the onset of defense-related mechanisms, such as the regulation of stomatal aperture. At the level of redox modulation, AITC induces depletion of glutathione and the upregulation of glutathione S-transferases in Arabidopsis thaliana [65]. |
| Calendula officinalis flowers | Lupeol, erythrodiol, calenduloside, rutin, narcissin, esculetin, cubenol, limonene, calenduloside B [66] | Calendula officinalis extracts possess antioxidant properties, enabling their use in food preservation and offering a range of therapeutic benefits [67]. Its use as a agricultural botanical extract appears to be limited [68]. |
| Camellia sinensis leaves (green-tea extract) | Catechins, caffeine, theanine, gallic acid [69] | Exogenous application of catechin enhances photosynthesis, plant growth, leaf expansion, antioxidant defense mechanisms, reactive oxygen species signaling, redox balance, and hormone metabolism under environmental stress conditions [70]. |
| Citrus sinensis peel | Naringin, rutin, hesperidin, melittoside [71] | The physiological and biochemical effects of hesperidin (100 μM) and chlorogenic acid (50 μM) were evaluated in Zea mays under arsenate stress (100 μM). Hesperidin and chlorogenic acid enhanced the activities of SOD, CAT, PRX, glutathione S-transferase, and glutathione peroxidase under stress, effectively reducing H2O2 accumulation and lipid peroxidation [72]. |
| Cymbopogon citratus leaf extract | Luteolin, apigenin, di-C-glycosylflavones, tannins [73] | Cymbopogon citratus extracts possess antioxidant properties, enabling their use in food preservation, as well as herbicide and insecticide, and offering a broad range of medicinal uses [74,75]. The extract use as a agricultural biostimulant appears to be limited. |
| Echinacea purpurea roots | Mannitol, benzoic acid, betulin, campesterol, β-sitosterol [76] | Echinacea purpurea root extracts possess antioxidant properties, enabling their use in food preservation and offering broad therapeutic potential [77,78]. Its use as a plant biostimulant appears to be limited. |
| Cupressus macrocarpa leaf extract | Secondary metabolites, phenolics, flavonoids, saponins, tannins, terpenes, and essential oils | Cypress leaf extract and salicylic acid were applied to seeds of zucchini and the seedlings were subjected to salinity stress. Pretreatment with the biostimulants enhanced growth and photosynthetic performance; elevated SOD, CAT, APX, GPX, GR, and DHAR activities; and increased ascorbate, glutathione, and proline compared with untreated stressed plants. Both treatments mitigated declines in CO₂ assimilation and significantly stimulated Rubisco activity. The extract also upregulated stress-inducible antioxidant genes (CuZnSOD2, CAT1, APX, GR, DHAR, PrxQ) and outperformed salicylic acid under both saline and nonsaline conditions [79]. |
| Curcuma longa rhizome | Curcumin and derivatives, calebin A, gallic acid, rutin [80] | Spinach was used as a model to study the toxicity of arsenic on physico-biochemical processes and the mitigating effect of exogenous curcumin at concentrations of 1, 10, and 20 μM. Curcumin reduced oxidative stress markers (H2O2 and MDA) and boosted nonenzymatic antioxidant capacity. It also enhanced the accumulation of glucosinolate and phenolic compounds and increased glutathione redox cycle activity, indicating the activation of secondary metabolism. Moreover, curcumin promoted crosstalk between ROS signaling and phytohormones, especially melatonin and serotonin, to alleviate arsenic-induced oxidative stress [81]. |
| Foeniculum vulgare and Ammi visnaga seed extract | Osmoprotectants, antioxidants and trace nutrients | The seed extracts—rich in macro- and micronutrients, α-tocopherol, phenolics, and glutathione GSH—strengthened salt-stressed cowpea plants’ antioxidant defenses by modulating osmoprotectants, such as proline and soluble sugars, and enhancing CAT, PRX, APX, and SOD enzymes and nonenzymatic systems, including carotenoid and glutathione levels [82]. |
| Glycyrrhiza glabra root | Glycyrrhizin, glycyrrhizic acid, isoliquiritigenin, licochalcone A, 18-β-glycyrrhetinic acid, glabrene [83] | Two field trials assessed the effects of licorice root extract (0.5%; 5 g L-1 in distilled water), applied as seed priming and/or foliar spray, on growth, yield, physiological, biochemical, and antioxidant traits of Phaseolus vulgaris under saline soil (EC = 7.15 dS m-1). Licorice treatments significantly enhanced growth, yield, photosynthetic pigments, proline, soluble carbohydrates and sugars, nutrient content, K+/Na+ ratio, relative water content, membrane stability, antioxidant enzyme activity, and anatomical features. Concurrently, they reduced electrolyte leakage, malondialdehyde levels, Na+ levels, H2O2 levels, and superoxide levels compared to untreated controls under salt stress [84]. |
| Hypericum perforatum leaves and flowers | Hyperforin, adhyperforin hyperoside, rutin, isoquercitrin, quercitrin [83] | Hypericum perforatum extracts possess antioxidant properties, enabling their use in food preservation and offering a range of therapeutic benefits [85,86]. Its use as a plant biostimulant appears to be limited. |
| Medicago sativa leaves | Chlorogenic acid, rutin, quercetin, kaempferol, genistein, vitexin [87] | Medicago sativa extracts possess antioxidant properties, which enable their potential use in food preservation and offer a range of therapeutic and cosmetic applications [88,89]. The use of botanical extracts as an agricultural biostimulant appears to be limited [90]. |
| Moringa oleifera leaves | Phenolic acids (chlorogenic, caffeic), fatty acids, amino acids, flavonoids (rutin, quercetin, kaempferol), glucosinolates, tocopherols [91] | Under oxidative stress, the exogenous application of chlorogenic acid to apple leaves mitigated chlorophyll loss, the decline in photosystem II efficiency, membrane damage, and lipid oxidation, while enhancing the activity of antioxidant enzymes. Phenolic concentrations significantly increased, and the expression of genes involved in antioxidant defense was modulated [92]. |
| Olea europaea leaves | Homogentisic acid, hydroxybenzoic acids, caffeic acid, vicenin, and luteolin [93] | Olea europaea leaf extracts possess antioxidant properties, enabling their use in food preservation and offering a broad therapeutic potential [94,95,96]. Its use as a plant biostimulant appears to be limited. |
| Punica granatum peel | Punicalagin, rutin, ellagic acid, gallic acid, and anthocyanins [97] | Punica granatum peel extracts possess antioxidant properties, enabling their use in food preservation and offering broad therapeutic potential [98,99]. Its use as a plant biostimulant appears to be limited. |
| Rosmarinus officinalis leaves | Carnosic acid, rosmarinic acid, luteolin, apigenin, caffeic acid [100] | The study measured endogenous levels of carnosic acid and α-tocopherol—lipophilic antioxidants in the chloroplasts of Salvia officinalis during a drought-recovery cycle. Drought significantly reduced salvia leaf water content, and as stress intensified, α-tocopherol and carnosic acid levels declined while the oxidation products, rosmanol and isorosmanol, increased. Carnosic acid serves a similar antioxidative role in both rosemary and salvia, emphasizing that drought resistance relies on the combined action of multiple antioxidants rather than a single mechanism [101]. |
| Salix alba bark | Salicin, chlorogenic acid, rutin, epicatechin [102] | Application of Salix alba root powder and Bacillus thuringiensis to wheat significantly increased shoot dry weight, root fresh weight, and the levels of CAT and APX. The combined use of B. thuringiensis and S. alba root powder promoted plant growth and defense responses under elevated soil cadmium concentrations [103]. |
| Solidago gigantea , S. canadensis, S. virgaurea, S. graminifolia, S. speciosa leaves and flowers | Chlorogenic acid, rutin, hyperoside, quercitrin, isoquercitrin [104] | Its use as a biostimulant has been reported [105], as well as its applications in controlling plant pathogens [106]. |
| Silybum marianum seeds | Silybin A, silydianin, taxifolin, quercetin [107] | Silybum marianum seed extracts possess antioxidant properties, offering a range of therapeutic uses for both humans and plants [108]. However, its use as a botanical extract in agriculture appears to be limited [109]. |
| Taraxacum officinale leaves, flowers, and fruits | Chicoric acid, chlorogenic acid, luteolin, quercetin glycosides [110] | Taraxacum officinale extracts possess antioxidant properties [111], enabling their use in food preservation and offering therapeutic potential [112]. Its use as a plant biostimulant appears to be limited. |
| Vitis vinifera seeds | Polyphenolics, flavonoids, procyanidins [113] | Vitis vinifera extracts possess antioxidant properties, enabling their use in food preservation [114] and offering medicinal uses [115]. Its use as a crop biostimulant appears to be limited. |
| Zingiber officinale rhizome | Gingerols (6-shogaol, 6-gingerol, 8-gingerol, 10-gingerol), ginger phenylpropanoids [116] | The inhibitory effect of ginger on lead-, cadmium-, and boron-induced oxidative stress in maize seedlings was examined, revealing an increase in inhibition with higher concentrations of ginger extract. Maize grown for 10-40 days with 1 g each of lead, cadmium, and boron exhibited significantly higher lipid peroxidation than the controls. Ginger extract notably reduced lipid peroxide levels and increased antioxidant enzyme activities in contaminated soil. These results suggest that ginger extract stimulates antioxidant enzymes, which can help mitigate metal-induced oxidative stress in maize [117]. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




