Submitted:
02 March 2026
Posted:
04 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Birds Are Specialist- and Extreme Animals
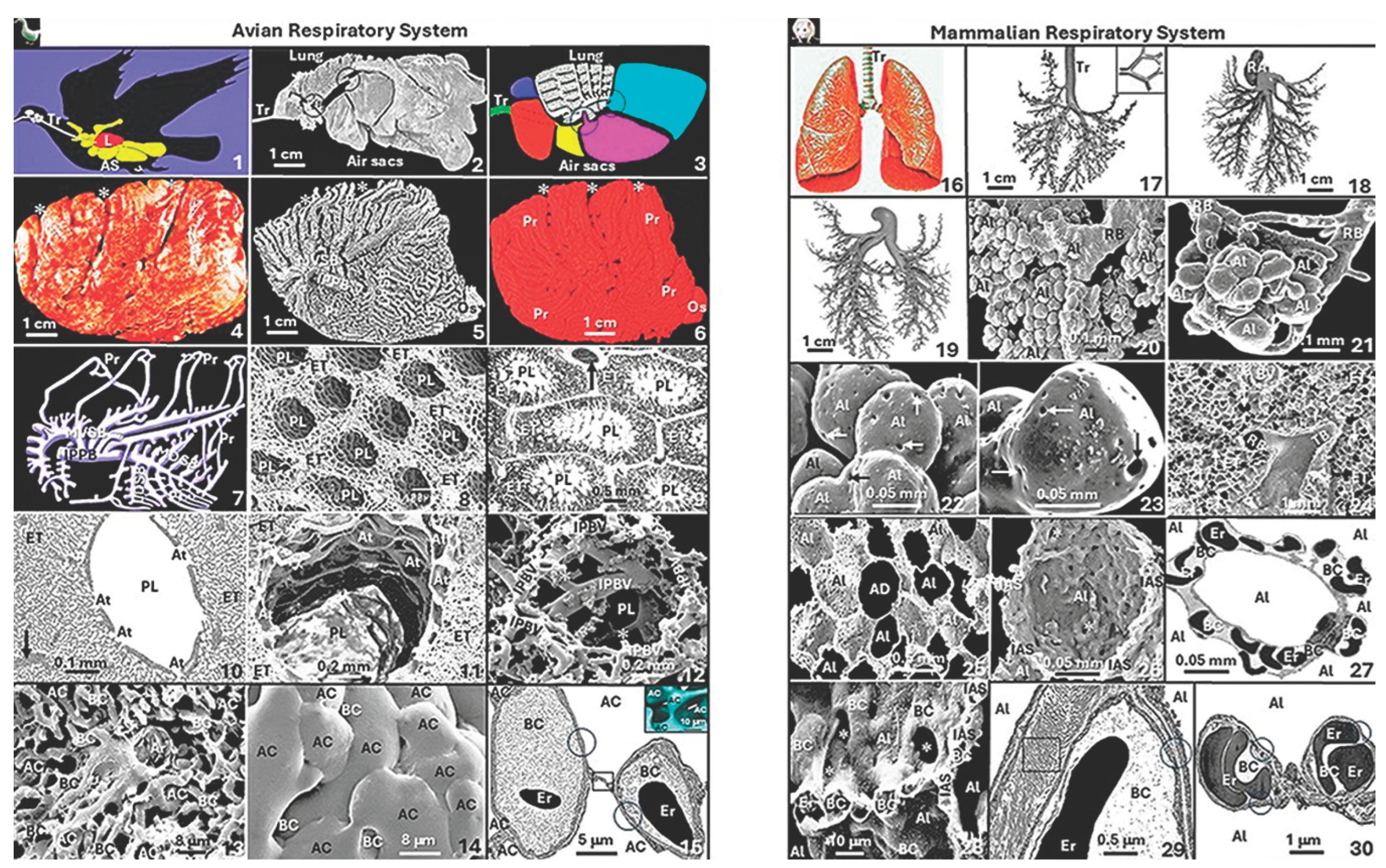
3. Environmental Conditions, Their Impacts on Birds and Aspects Which Suggest Weakening of Their Adaptive Capacities
3.1. Susceptibility of Birds to Diseases and Injuries by Foreign Particulates
3.2. Effects of Climate- and Anthropogenic Habitat Devastation on the Bird Life
3.3. Effects of Environmental Pollution on Birds
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Moczek, A.P. When the end modifies its means: the origins of novelty and the evolution of innovation. Biol. J. Linnean Soc. 2023, 139, 433–440. [Google Scholar] [CrossRef]
- Darwin, C. On the Origin of Species by Means of Natural Selection, or the Preservation of Favoured Races in the Struggle for Life, 2nd ed.; D. Appleton and Company: New York (NY), USA, 1859. [Google Scholar]
- van Wijnen, A.J.; Lewallen, E.A. Natural selection and evolution: evolving concepts. Acad. Biol. 2024, 2. [Google Scholar] [CrossRef] [PubMed]
- Crouch, D.J.M.; Walter, F.B. Evolution by natural selection is a scientific law and not just a theory. Acad. Biol. 2024, 2. [Google Scholar] [CrossRef]
- Mathot, K.J.; Kok, E.M.A.; Burant, J.B.; Dekinga, A.; Manche, P.; Saintonge, D.; Piersma, T. Evolutionary design of a flexible, seasonally migratory, avian phenotype: why trade gizzard mass against pectoral muscle mass? Proc. R. Soc. B 2019, 286, 20190518. [Google Scholar] [CrossRef]
- Jaeger, J. The fourth perspective: evolution and organismal agency. In Organization in Biology. History, Philosophy and Theory of the Life Sciences; Mossio, M., Ed.; Springer: Cham (Switzerland), 2024; Vol. 33, pp. 159–186. [Google Scholar]
- Gregory, T.R. Understanding natural selection: essential concepts and common misconceptions. Evo. Edu. Outreach 2009, 2, 156–175. [Google Scholar] [CrossRef]
- Gardner, A. The purpose of adaptation. Interface Focus 2017, 7, 20170005. [Google Scholar] [CrossRef]
- De Loof, A. The evolution of ‘life’: a metadarwinian integrative approach. Commun. Integrative Biol. 2017, 10, e1301335. [Google Scholar] [CrossRef]
- Phenotypic Integration: Studying the Ecology and Evolution of Complex Phenotypes; Massimo, P., Preston, K., Eds.; Academic Press: Oxford, 2004. [Google Scholar]
- Murren, C.J. The integrated phenotype. Integr. Comp. Biol. 2012, 52, 64–76. [Google Scholar] [CrossRef]
- Olson, E.C; Miller, R.J. Morphological Integration; University of Chicago Press: Chicago (Illinois), 1958. [Google Scholar]
- Chernoff, B.; Magwene, P.M. Afterward - Morphological integration: forty years later. In Morphological Integration; Olson, E.C., Miller, R.L., Eds.; University of Chicago Press: Chicago, Illinois, 1999; pp. 319–353. [Google Scholar]
- Klingenberg, C. Morphological integration and developmental modularity. Annu. Rev. Ecol. Evol. Syst. 2008, 39, 115–132. [Google Scholar] [CrossRef]
- Alexander, R.McN. Evolution of integrated design. Amer. Zool. 1975, 15, 419–425. [Google Scholar] [CrossRef]
- Garland, T.; Huey, R.B. Testing symmorphosis: does structure match functional requirements? Evolution 1987, 41, 1404–1409. [Google Scholar] [CrossRef] [PubMed]
- Principles of Animal Design: The Optimization and Symmorphosis Debate; Weibel, E.R., Taylor, C.R., Bolis, L., Eds.; Cambridge University Press: Cambridge (UK), 2000. [Google Scholar]
- Huber, K. Invited review: resource allocation mismatch as pathway to disproportionate growth in farm animals – prerequisite for a disturbed health. Animal 2018, 12, 528–536. [Google Scholar] [CrossRef]
- Garland, T. Quick guide: trade-offs. Curr. Biol. 2014, 24, R60–R61. [Google Scholar] [CrossRef]
- Garland, T.; Downs, C.J.; Ives, A.R. Trade-offs (and constraints) in organismal biology. Physiol. Biochem. Zool. 2022, 95, 82–112. [Google Scholar] [CrossRef] [PubMed]
- Debelgarric, M.; Récapet, C. Exploring physiological constraints on life-history traits using dynamic energy budgets. Ecol. Model. 2025, 501, 110993. [Google Scholar] [CrossRef]
- Duclos, K.K.; Hendrikse, J.L.; Jamniczky, H.A. Investigating the evolution and development of biological complexity under the framework of epigenetics. Evol. Dev. 2019, 21, 247–264. [Google Scholar] [CrossRef] [PubMed]
- Force, E.; Lamy, D.; Debernard, S.; Savouré, A.; Dacher, M. Developmental transitions involve common biological processes across living beings. Heliyon 2025, 11, e42995. [Google Scholar] [CrossRef]
- Ardia, D.R.; Parmentier, H.K.; Vogel, L.A. The role of constraints and limitation in driving individual variation in immune response. Ecol. Immunol. 2011, 25, 61–73. [Google Scholar] [CrossRef]
- Holekamp, K.E.; Swanson, E.M.; Van Meter, P.E. Developmental constraints on behavioural flexibility. Philos. Trans. R. Soc. Lond. B 2013, 368, 20120350. [Google Scholar] [CrossRef]
- Goldsmith, T.C. Programmed aging, digital genetics, and the evolution of acquisition traits in mammals. Acad. Biol. 2025, 3. [Google Scholar] [CrossRef]
- Chen, L.; Khanna, M. Heterogeneous and long-term effects of a changing climate on bird biodiversity. GEC Advances 2024, 2, 100008. [Google Scholar] [CrossRef]
- Vinton, A.C.; Gascoigne, S.J.L.; Sepil, I.; Salguero-Gómez, R. Plasticity’s role in adaptive evolution depends on environmental change components. Trends Ecol. Evol. 2022, 37, 1067–1078. [Google Scholar] [CrossRef]
- Guo, Y.-W.; Liu, Y.; Huang, P.-C.; Rong, M.; Wei, W.; Xu, Y.-H.; Wei, J.-H. Adaptive changes and genetic mechanisms in organisms under controlled conditions: a review. Int. J. Mol. Sci. 2025, 26, 2130. [Google Scholar] [CrossRef] [PubMed]
- Daniels, R.J.R. Patterns of regional species richness and endemism in Indian birds: a biogeographical perspective. Acad. Biol. 2025, 3. [Google Scholar] [CrossRef]
- Zera, A.J.; Harshman, L.G. The physiology of life history trade-offs in animals. Annu. Rev. Ecol. Syst. 2001, 32, 95–126. [Google Scholar] [CrossRef]
- Bennett, A.F.; Lenski, R.E. An experimental test of evolutionary trade-offs during temperature adaptation. Proc. Natl. Acad. Sci. USA 2007, 104, 8649–8654. [Google Scholar] [CrossRef]
- Garland, T. Quick guide: trade-offs. Curr. Biol. 2014, 24, R60–R61. [Google Scholar] [CrossRef]
- Chu, E.W.; Karr, J.R. Environmental Impact: Concept, Consequences, Measurement. Reference Module in Life Sciences; 2017; B978-0-12-809633-8.02380-3. [Google Scholar]
- Ehrlich, E.; Becks, L.; Gaedke, U. Trait-fitness relationships determine how trade-off shapes affect species coexistence. Ecology 2017, 98, 3188–3198. [Google Scholar] [CrossRef] [PubMed]
- Farahpour, F.; Saeedghalati, M.; Brauer, V.; Hoffmann, D. Trade-off shapes diversity in eco-evolutionary dynamics. eLife 2018, 7, e36273. [Google Scholar] [CrossRef] [PubMed]
- Zinner, M.; Lukonin, I.; Liberali, P. Design principles of tissue organisation: how single cells coordinate across scales. Curr. Opin. Cell Biol. 2020, 67, 37–45. [Google Scholar] [CrossRef]
- Gibert, P.; Debat, V.; Ghalambor, C.K. Phenotypic plasticity, global change, and the speed of adaptive evolution. Curr. Opin. Insect Sci. 2019, 35, 34–40. [Google Scholar] [CrossRef]
- Mauro, A.A.; Ghalambor, C.K. Trade-offs, pleiotropy, and shared molecular pathways: a unified view of constraints on adaptation. Integr. Compar. Biol. 2020, 60, 332–347. [Google Scholar] [CrossRef]
- Lalejini, A.; Ferguson, A.J.; Grant, N.A.; Ofria, C. Adaptive phenotypic plasticity stabilizes evolution in fluctuating environments. Front. Ecol. Evol. 2021, 9, 715381. [Google Scholar] [CrossRef]
- Ancel, L.W. Undermining the Baldwin expediting effect: does phenotypic plasticity accelerate evolution? Theor. Popul. Biol. 2000, 58, 307–319. [Google Scholar] [CrossRef]
- Price, T.D.; Qvarnstrm, A.; Irwin, D.E. The role of phenotypic plasticity in driving genetic evolution. Proc. R. Soc. Lond. Ser. B Biol. Sci. 2003, 270, 1433–1440. [Google Scholar] [CrossRef]
- Paenke, I.; Sendhoff, B.; Kawecki, T. Influence of plasticity and learning on evolution under directional selection. Am. Nat. 2007, 170, E47–E58. [Google Scholar] [CrossRef] [PubMed]
- Futuyma, D.J.; Moreno, G. The evolution of ecological specialization. Annu. Rev. Ecol. Syst. 1988, 19, 207–233. [Google Scholar] [CrossRef]
- Schluter, D. The Ecology of Adaptive Radiation; Oxford University Press: New York (NY), 2000. [Google Scholar]
- Sintayehu, D.W. Impact of climate change on biodiversity and associated key ecosystem services in Africa: a systematic review. Ecosyst. Health Sustainability (EHS) 2018, 4, 225–239. [Google Scholar] [CrossRef]
- Ardoin, N.M.; Bowers, A.W.; Gaillard, E. Environmental education outcomes for conservation: a systematic review. Biol. Conserv. 2020, 241, 108224. [Google Scholar] [CrossRef]
- Stephenson, P.J.; Londoño-Murcia, M.C.; Borges, P.A.V.; Claassens, L.; Frisch-Nwakanma, H.; Ling, N. Measuring the impact of conservation: the growing importance of monitoring fauna, flora and funga. Diversity 2022, 14, 824. [Google Scholar] [CrossRef]
- Julliard, R.; Jiguet, F.; Couvet, D. Common birds facing global changes: what makes a species at risk? Glob. Chang. Biol. 2003, 10, 148–154. [Google Scholar] [CrossRef]
- Büchi, L.; Vuilleumier, S. Coexistence of specialist and generalist species is shaped by dispersal and environmental factors. Am. Nat. 2014, 183, 612–624. [Google Scholar] [CrossRef]
- Morelli, F.; Benedetti, Y.; Callaghan, C.T. Ecological specialization and population trends in European breeding birds. Glob. Ecol. Conserv. 2020, 22, e00996. [Google Scholar] [CrossRef]
- Morris, J.R.; Allhoff, K.T.; Valdovinos, F.S. Strange invaders increase disturbance and promote generalists in an evolving food web. Sci. Rep. 2021, 11, 21274. [Google Scholar] [CrossRef]
- Biesmeijer, J.C.; Roberts, S.P.M.; Reemer, M.; Ohlemuller, R.; Edwards, M.; Peeters, T.; et al. Parallel declines in pollinators and insect-pollinated plants in Britain and the Netherlands. Science 2006, 313, 351–354. [Google Scholar] [CrossRef]
- Rezende, E.L.; Lavabre, J.E.; Guimaraes, P.R.; Jordano, P.; Bascompte, J. Non-random coextinctions in phylogenetically structured mutualistic networks. Nature 2007, 448, 925–928. [Google Scholar] [CrossRef] [PubMed]
- Colles, A.; Liow, L.H.; Prinzing, A. Are specialists at risk under environmental change? Neoecological, paleoecological and phylogenetic approaches. Ecol. Lett. 2009, 12, 849–863. [Google Scholar] [CrossRef]
- Cooke, R.S.C.; Eigenbrod, F.; Bates, A.E. Ecological distinctiveness of birds and mammals at the global scale. Global Ecol. Conserv. 2020, 22, e00970. [Google Scholar] [CrossRef]
- Tobias, J.A.; Sheard, C.; Pigot, A.L.; Devenish, A.J.M.; Yang, J.; Sayol, F.; et al. AVONET: Morphological, ecological and geographical data for all birds. Ecol. Let. 2022, 25, 581–597. [Google Scholar] [CrossRef]
- Maina, J.N. Current Perspectives on the Functional Design of the Avian Respiratory System; Springer: Cham (Switzerland), 2023. [Google Scholar]
- Maina, J.N. Structure and function of the avian respiratory system. Philos. Trans. R. Soc. Lond. B 2025, 380, 20230435. [Google Scholar] [CrossRef]
- Hernando, M.G.; Roa, I.; Fernandez-Gil, J.; Benito-Fuertes, J.J.; Reguera, B.; Revilla, E. Trends in weather conditions favor generalist over specialist species in rear-edge alpine bird communities. Ecosphere 2022, 13, e3953. [Google Scholar] [CrossRef]
- Germain, R.R.; Feng, S.; Rahbeka, C.; Lein, F.; Buffan, L.; Carmonag, C.P.; et al. Changes in the functional diversity of modern bird species over the last million years. Proc. Natl. Acad. Sci. USA 2023, 120, e2201945119. [Google Scholar] [CrossRef] [PubMed]
- Ashrafzadeh, M.R.; Moradi, M.; Khosravi, R.; Naghipour, A.A.; Chamberlain, D. Impacts of climate change on a high elevation specialist bird are ameliorated by terrain complexity. Glob. Ecol. Conserv. 2024, 56, e03281. [Google Scholar] [CrossRef]
- Mojzsis, S.J.; Arrhenius, G.; McKeegan, K.D.; Harrison, T.M.; Nutman, A.P.; Friend, C.R. Evidence for life on earth before 3,800 million years ago. Nature 1996, 384, 55–59. [Google Scholar] [CrossRef]
- Schopf, J.W.; Kudryavtsev, A.B. Biogenicity of earth’s earliest fossils. In Evolution of Archean Crust and Early Life, Modern Approaches in Solid Earth Sciences; Dilek, Y., Furnes, H., Eds.; Springer: Dordrecht, 2014; Vol. 7, pp. 333–349. [Google Scholar]
- Bell, E.A.; Boehnkea, P.; Harrisona, T.M.; Mao, WL. Potentially biogenic carbon preserved in a 4.1 billion-year-old zircon. Proc. Natl. Acad. Sci. USA 2015, 112, 14518–14521. [Google Scholar] [CrossRef]
- Maina, J.N. What it takes to fly: the structural and functional respiratory refinements in birds and bats. J. Exp. Biol. 2000, 203, 3045–3064. [Google Scholar] [CrossRef]
- Dudley, R. The Biomechanics of Insect Flight: Form, Function. Evolution; Princeton University Press: Princeton (NJ), 2000. [Google Scholar]
- Koroljov, A.V. The flight of pterosaurs. Zh. Obshch. Biol. 2016, 77, 182–238. [Google Scholar] [CrossRef] [PubMed]
- Anderson, S.C.; Ruxton, G.D. The evolution of flight in bats: a novel hypothesis. Mammal Rev. 2020, 50, 426–439. [Google Scholar] [CrossRef]
- Pittman, M.; Barlow, L.A.; Kaye, T.G.; Habib, M.B. Pterosaurs evolved a muscular wing- body junction providing multifaceted flight performance benefits: advanced aerodynamic smoothing, sophisticated wing root control, and wing force generation. Proc. Natl. Acad. Sci. USA 2021, 118, e2107631118. [Google Scholar] [CrossRef]
- Brusatte, S.L.; Lloyd, G.T.; Wang, S.C.; Norell, M.A. Gradual assembly of avian body plan culminated in rapid rates of evolution across the dinosaur-bird transition. Curr. Biol. 2014, 24, 2386–2392. [Google Scholar] [CrossRef]
- Brusatte, S.L.; O’Connor, J.K.; Jarvis, E.D. The origin and diversification of birds. Curr. Biol. 2015, 25, R888–R898. [Google Scholar] [CrossRef]
- Tietze, D.T. Introduction: studying birds in time and space. In Bird Species: How they Arise, Modify and Vanish; Tietze, D.T, Ed.; Springer: Cham (Switzerland), 2018; pp. 1–7. [Google Scholar]
- King, A.S.; King, D.Z. Avian morphology: general principles. In Form and Function in Birds; King, A.S., McLelland, J., Eds.; Academic Press: London, 1979; Vol. 1, pp. 1–38. [Google Scholar]
- King, A.S.; McLelland, J. Birds: Their Structure and Function, 2nd Edtn ed; Bailliere Tindal: London, 1984. [Google Scholar]
- Chatterjee, A. The Rise of Birds: 225 million Years of Evolution; The Johns Hopkins University Press: Baltimore (MD), 1997. [Google Scholar]
- Myers, E. A.; Burbrink, F. T. Ecological opportunity: trigger of adaptive radiation. Nat. Sci. Educ. 2012, 3, 23. [Google Scholar]
- Chaparro-Pedraza, P.C.; Roth, G.; Seehausen, O. The enrichment paradox in adaptive radiations: emergence of predators hinders diversification in resource rich environments. Ecol. Lett. 2022, 25, 802–813. [Google Scholar] [CrossRef]
- Yamasaki, T.; Kobayashi, Y. Evolving dispersal ability causes rapid adaptive radiation. Sci. Rep. 2024, 14, 15734. [Google Scholar] [CrossRef] [PubMed]
- Wingfield, J.C.; Pérez, J.H.; Krause, J.S.; Word, K.R.; González-Gómez, P.L.; Lisovski, S.; Chmura, H.E. How birds cope physiologically and behaviourally with extreme climatic events. Philos. Trans. R. Soc. Lond. B 2017, 2027(372), 20160140. [Google Scholar] [CrossRef] [PubMed]
- Töpfer, T. Morphological variation in birds: plasticity, adaptation, and speciation. In Bird Species: How they Arise, Modify and Vanish; Tietze, D., Ed.; Springer: Cham (Switzerland), 2007; pp. 63–74. [Google Scholar]
- Fragueira, R.; Helfenstein, F.; Fischer, K.; Beaulieu, M. Birds of different morphs use slightly different strategies to achieve similar reproductive performance following heat wave exposure. J. Anim. Ecol. 2021, 90, 2594–2608. [Google Scholar] [CrossRef] [PubMed]
- Swanson, D.L.; Vézina, F.; McKechnie, A.E.; Nord, A. Editorial: Avian behavioral and physiological responses to challenging thermal environments and extreme weather events. Front. Ecol. Evol. 2022, 10, 1034659. [Google Scholar] [CrossRef]
- Norberg, U.M. Energetics of flight. In Avian Energetics and Nutritional Ecology; Carey, C., Ed.; Springer: Boston (MA), 1996; pp. 199–249. [Google Scholar]
- Alexander, R.McN. Principles of Animal Locomotion; Princeton University Press: Princeton (NJ), 2003. [Google Scholar]
- Pennycuick, C.J. Modelling the Flying Bird; Academic Press: London, 2008. [Google Scholar]
- Bishop, C.M.; Butler, P.J. Flight. In Sturkie's Avian Physiology; Scanes, C.G., Ed.; Academic Press: London, 2025; pp. 919–974. [Google Scholar]
- Tobalske, B.W. Evolution of avian flight: Muscles and constraints on performance. Phil. Trans. R. Soc. Lond. B 2016, 371, 20150383. [Google Scholar] [CrossRef]
- Nespolo, R.F.; González-Lagos, C.; Solano-Iguaran, J.J.; Elfwing, M.; Garitano-Zavala, A.; Mañosa, S.; et al. Aerobic power and flight capacity in birds: a phylogenetic test of the heart-size hypothesis. J. Exp. Biol. 2018, 221, jeb162693. [Google Scholar] [CrossRef]
- Guglielmo, C.G. Obese super athletes: fat-fueled migration in birds and bats. J. Exp.Biol. 2018, 221, jeb165753. [Google Scholar] [CrossRef]
- Groom, D.J.E.; Black, B.; Deakin, J.E.; DeSimone, J.G.; Lauzau, M.C.; Pedro, B.P.; et al. Flight muscle size reductions and functional changes following long-distance flight under variable humidity conditions in a migratory warbler. Physiol. Rep. 2023, 11, e15842. [Google Scholar] [CrossRef] [PubMed]
- Wyles, J.S.; Kunkel, J.G.; Wilson, A.C. Birds, behavior, and anatomical evolution. Proc. Nat. Acad. Sci. 1983, 80, 4394–4397. [Google Scholar] [CrossRef]
- O'Malley, B. Clinical Anatomy and Physiology of Exotic Species: Structure and Function of Mammals, Birds, Reptiles and Amphibians; W.B. Saunders: Edinburgh, 2005. [Google Scholar]
- Roff, D.A. The evolution of flightlessness: is history important? Evol. Ecol. 1994, 8, 639–657. [Google Scholar] [CrossRef]
- Iwaniuk, A.N.; Nelson, J.E.; James, H.F.; Olson, S.L. A comparative test of the correlated evolution of flightlessness and relative brain size in birds. J. Zool. 2004, 263, 317–327. [Google Scholar] [CrossRef]
- Wright, N.A.; Steadman, D.W.; Witt, C.C. Predictable evolution toward flightlessness in volant island birds. Proc. Natl. Acad. Sci. USA 2016, 113, 4765–4770. [Google Scholar] [CrossRef]
- Welty, J.C. The Life of Birds, 2nd Edtn ed; Saunders: Philadelphia, 1979. [Google Scholar]
- Maderspacher, F. Flightless birds. Curr. Biol. 2022, 32, R1155–R1162. [Google Scholar] [CrossRef]
- Hieronymus, T.L. Qualitative skeletal correlates of wing shape in extant birds (Aves: Neoaves). BMC Evol. Biol. 2015, 15, 30. [Google Scholar] [CrossRef]
- Chin, D.D.; Matloff, L.Y.; Stowers, A.K.; Tucci, E.R.; Lentink, D. Inspiration for wing design: how forelimb specialization enables active flight in modern vertebrates. J. R. Soc. Interface 2017, 14, 20170240. [Google Scholar] [CrossRef] [PubMed]
- Buhler, P. Light bones in birds. LA Mus. Nat. Hist. Sci. Ser. 1992, 36, 385–394. [Google Scholar]
- Cubo, J.; Casinos, A. Incidence and mechanical significance of pneumatization in the long bones of birds. Zool. J. Linn. Soc. 2000, 130, 499–510. [Google Scholar] [CrossRef]
- Dumont, E.R. Bone density and the light weight skeletons of birds. Proc. Biol. Sci. 2010, 277, 2193–2198. [Google Scholar] [PubMed]
- Hartman, F.A. Heart weight in birds. Condor 1955, 57, 221–238. [Google Scholar] [CrossRef]
- Dzialowski, E.M.; Crossley, D.A. The cardiovascular system. In Sturkie’s Avian Physiology, 7th Edtn. ed; Academic Press: San Diego (CA), 2022; pp. 327–409. [Google Scholar]
- O'Malley, B. Clinical Anatomy and Physiology of Avian Species - From Bird Brains to Pigeon. 2008. Available online: https://www.vin.com/doc/?id=3866642 (accessed on 10-07-25).
- Louchart, A.; Viriot, L. From snout to beak: the loss of teeth in birds. Trends Ecol. Evol. 2011, 26, 663–673. [Google Scholar] [CrossRef]
- Meredith, R.W.; Zhang, G.; Gilbert, M.T.; Jarvis, E.D.; Springer, M.S. Evidence for a single loss of mineralized teeth in the common avian ancestor. Science 2014, 346, 1254390. [Google Scholar] [CrossRef]
- Enoki, Y.; Morimoto, T. Gizzard myoglobin contents and feeding habits in avian species. Comp. Biochem. Physiol. A: Mol.Integr. Physiol. 2000, 125, 33–43. [Google Scholar] [CrossRef]
- McNab, B.K. Ecological factors affect the level and scaling of avian BMR. Comp. Biochem. Physiol. A: Molec. Integr. Physiol. 2009, 152, 22–45. [Google Scholar] [CrossRef]
- Lovegrove, B.G. A phenology of the evolution of endothermy in birds and mammals. Biol. Rev. 2017, 92, 1213–1240. [Google Scholar] [CrossRef] [PubMed]
- Price, E.R.; Dzialowski, E.M. Development of endothermy in birds: patterns and mechanisms. J. Comp. Physiol. B. 2018, 188, 373–391. [Google Scholar] [CrossRef]
- Swanson, D.L.; Stager, M.; Vézina, F.; Liu, J.S.; McKechnie, A.E.; Amirkhiz, R.E. Evidence for a maintenance cost for birds maintaining highly flexible basal, but not summit, metabolic rates. Sci. Rep. 2023, 13, 8968. [Google Scholar] [CrossRef] [PubMed]
- Biewener, A.A. Muscle function in avian flight: achieving power and control. Phil. Trans. R. Soc. Lond. B 2011, 366, 1496–1506. [Google Scholar] [CrossRef]
- Hedrick, T.L.; Tobalske, B.W.; Ros, I.G.; Warrick, D.R.; Biewener, A.A. Morphological and kinematic basis of the hummingbird flight stroke: scaling of flight muscle transmission ratio. Proc Biol Sci. 2012, 279, 1986–1992. [Google Scholar] [CrossRef]
- Scheid, P. Mechanisms of gas exchange in bird lungs. Rev. Physiol. Biochem. Pharmacol. 1979, 86, 137–186. [Google Scholar]
- Avian Medicine and Surgery; Altman, R.B., 117, Clubb, S.L., Dorrestein, G.M., Quesenberry, K., Eds.; Saunders: Philadelphia (PA), 1997. [Google Scholar]
- Sturkie’s Avian Physiology, 7th Edtn; Scanes, C.G., Dridi, S., Eds.; Academic Press: New York, 2022. [Google Scholar]
- Packard, G.C.; Packard, M.J. Evolution of the cleidoic egg among reptilian antecedents of birds. Amer. Zool. 1980, 20, 351–362. [Google Scholar] [CrossRef]
- Blackburn, D.; Evans, H.E. Why are there no viviparous birds? Amer. Natur. 1986, 128, 165–190. [Google Scholar] [CrossRef]
- Lodé, T. Oviparity or viviparity? That is the question. Reprod, Biol. 2012, 12, 259–264. [Google Scholar] [CrossRef] [PubMed]
- Rawal, D.; Rajpurohit, A. Cleidoic eggs: a key to water to land transition. Indian J. Appl. Pure Biol. 2025, 40, 621–627. [Google Scholar] [CrossRef]
- Cockburn, A. Prevalence of different modes of parental care in birds. Proc. R. Soc. B 2006, 273, 1375–1383. [Google Scholar] [CrossRef] [PubMed]
- Mock, D.W. Parental care in birds. Curr. Biol. 2022, 32, R1132–R1136. [Google Scholar] [CrossRef]
- Jacoboski, L.I. Why integrate evolutionary and functional aspects in bird studies inconverted habitats? Internat. Ornithol. 2018, 1, 1–2. [Google Scholar]
- Wiltschko, R.; Wiltschko, W. Magnetoreception in birds. J. R. Soc. Interface 2019, 16, 20190295. [Google Scholar] [CrossRef]
- Demšar, U.; Zein, B.; Long, J.A. A new data-driven paradigm for the study of avian migratory navigation. Mov. Ecol. 2025, 13, 16. [Google Scholar] [CrossRef]
- Rolando, A.; Zunino, M. Observations of tool use in corvids. Ornis Scandinavica 1992, 23, 201–202. [Google Scholar] [CrossRef]
- Hunt, G. Vice-anvil use in nut processing by two Corvus species. New Zealand J. Zool. 2013, 41, 68–76. [Google Scholar] [CrossRef]
- Hunt, G.R.; Sakuma, F.; Shibata, Y. New Caledonian crows drop candle-nuts onto rock from communally used forks on branches. Emu 2002, 102, 283–290. [Google Scholar] [CrossRef]
- Raby, C.R.; Clayton, N.S. The cognition of caching and recovery in food-storing birds. In Advances in the Study of Behavior; Brockmann, H.J., Roper, T.J., Naguib, M., Wynne-Edwards, K.E., Mitani, J.C., Simmons, L.W., Eds.; Elsevier: Amsterdam, 2010; Vol. 41, pp. 1–34. [Google Scholar]
- Applegate, M.C.; Aronov, D. Flexible use of memory by food-caching birds. eLife 2022, 11, e70600. [Google Scholar] [CrossRef]
- Brea, J.; Clayton, N.S.; Gerstner, W. Computational models of episodic-like memory in food-caching birds. Nat. Commun. 2023, 14, 2979. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, G.C. Paradoxical calls: the opposite signaling role of sound frequency across bird species. Behav. Ecol. 2012, 23, 237–241. [Google Scholar] [CrossRef]
- Crisologo, T.L.; Dzielski, S.A.; Purcell, J.R.; Webster, M.S.; Welbergen, J.A.; Dalziell, A.H. Selective alarm call mimicry in the sexual display of the male superb lyrebird (Menura novaehollandiae). Evol. Ecol. 2023, 37, 245–266. [Google Scholar] [CrossRef]
- Brooke, M.L.; Hanley, S.; Laughlin, S.B. The scaling of eye size with body mass in birds. Proc. R. Soc. B Biol. Sci. 1999, 266, 405–412. [Google Scholar] [CrossRef]
- Kiltie, R.A. Scaling of visual acuity with body size in mammals and birds. Funct. Ecol. 2000, 14, 226–234. [Google Scholar] [CrossRef]
- Jetz, W.; Thomas, G.H.; Joy, J.B.; Hartmann, K.; Mooers, A.O. The global diversity of birds in space and time. Nature 2012, 491, 444–448. [Google Scholar] [CrossRef]
- Terrill, R.S.; Shultz, A.J. Feather function and the evolution of birds. Biol. Rev. 2023, 98, 540–566. [Google Scholar] [CrossRef]
- King, A.S. Structural and functional aspects of the avian lung and its air sacs. Intern. Rev. Gen. Exp. Zool 1966, 2, 171–267. [Google Scholar]
- Duncker, H.R. The lung-air sac system of birds. A contribution to the functional anatomy of the respiratory apparatus. Ergeb. Anat. Entwicklung. 1971, 45, 1–171. [Google Scholar]
- Form and Function in Birds; King, A.S., McLelland, J., Eds.; Academic Press: London, 1989; Vol. 4. [Google Scholar]
- McLelland, J. Anatomy of the lungs and air sacs. In Form and Function in Birds, Vol. IV; King, A.S., McLelland, J., Eds.; Academic Press: London, 1989; pp. 221–279. [Google Scholar]
- Ehrlich, P.R.; Dobkin, D.S.; Wheye, D. The Birder’s Handbook: A Field Guide to the Natural History of North American Birds; Simon and Schuster: New York (NY), 1988. [Google Scholar]
- Schwab, R.G.; Schafer, V.F. Avian Thermoregulation and its Significance in Starling Control. In Proceedings of the 5th Vertebrate Pest Conference, 1972; 25. [Google Scholar]
- Mota-Rojas, D.; Titto, C.G.; de Mira Geraldo, A.; Martínez-Burnes, J.; Gómez, J.; Hernández-Ávalos, I. Efficacy and function of feathers, hair, and glabrous skin in the thermoregulation strategies of domestic animals. Animals 2021, 11, 3472. [Google Scholar] [CrossRef]
- Maina, J.N.; King, A.S.; Settle, J.G. An allometric study of the pulmonary morphometric parameters in birds, with mammalian comparison. Philos. Trans. R. Soc. Lond. B 1989, 326, 1–57. [Google Scholar] [CrossRef]
- Maina, J.N. The morphometry of the avian lung. In Form and Function in Birds; King, A.S., McLelland, J., Eds.; Academic Press: London, 1989; Vol. 4, pp. 307–368. [Google Scholar]
- Weibel, E.R. The Pathway for Oxygen: Structure and Function in the Mammalian Respiratory System.; Harvard University Press: Cambridge (MA), 1984. [Google Scholar]
- Fedde, M.R. The structure and gas flow pattern in the avian lung. Poult. Sci. 1980, 59, 2642–2653. [Google Scholar] [CrossRef]
- Stahel, C.D.; Nicol, S.C. Ventilation and oxygen extraction in the little penguin (Eudyptula minor), at different temperatures in air and water. Respir. Physiol. 1988, 71, 387–398. [Google Scholar] [CrossRef] [PubMed]
- Fedde, M.R. Relationship of structure and function of the avian respiratory system to disease susceptibility. Poult. Sci. 1998, 77, 1130–1138. [Google Scholar] [CrossRef]
- Ritchison, G. a Class of their Own: A Detailed Examination of Avian Forms and Functions; Springer: Cham (Switzerland), 2023. [Google Scholar]
- Brent, R.; Rasmussen, J.G.; Bech, C.; Martini, S. Lung ventilation and temperature regulation in the European Coot, Fulica atra. Physiol Zool 1984, 57, 19–25. [Google Scholar] [CrossRef]
- Larcombe, A. Effects of temperature on metabolism, ventilation, and oxygen extraction in the southern brown bandicoot Isoodon obesulus (Marsupialia: Peramelidae). Physiol. Biochem. Zool. 2002, 75, 405–411. [Google Scholar] [CrossRef]
- Chappell, M.A.; Roverud, R.C. Temperature effects on metabolism, ventilation, and oxygen extraction in Neotropical bat. Respir. Physiol. 1990, 81, 401–412. [Google Scholar] [CrossRef]
- Thomas, S.P.; Follette, D.B.; Thomas, G.S. Metabolic and ventilatory adjustments and tolerance of the bat Pteropus poliocephalus to acute hypoxic stress. Comp. Biochem. Physiol. 1995, 112A, 43–54. [Google Scholar] [CrossRef]
- Lasiewski, R.C.; Calder, W.A. A preliminary allometric analysis of respiratory variables in resting birds. Respir. Physiol. 1971, 11, 152–166. [Google Scholar] [CrossRef]
- Meyer, M.; Worth, H.; Scheid, P. Gas-blood CO2 equilibration in parabronchial lungs of birds. J. Appl. Pysiol. 1976, 41, 302–309. [Google Scholar] [CrossRef] [PubMed]
- Scheid, P. Respiration and control of breathing in birds. Physiologist 1978, 2, 60–64. [Google Scholar]
- Monique, P.; Ludders, J.W.; Erb, H.N. Association of partial pressure of carbon dioxide in expired gas and arterial blood at three different ventilation states in apneic chickens (Gallus domesticus) during air sac insufflation anesthesia. Vet. Anaesth. Analg. 2013, 40, 245–256. [Google Scholar]
- Evans, B.R.; Leighton, F.A. A history of One Health. Rev. Sci. Tech. 2014, 33, 413–420. [Google Scholar] [CrossRef] [PubMed]
- Lerner, H.; Berg, C. The concept of health in One Health and some practical implications for research and education: what is One Health? Infect. Ecol. Epidemiol. 2015, 5, 25300. [Google Scholar] [CrossRef]
- Mackenzie, J.S.; Jeggo, M. The One Health Approach - why is it so important? Trop. Med. Infect. Dis. 2019, 4, 88. [Google Scholar] [CrossRef]
- Pitt, S.J.; Gunn, A. The One Health Concept. Br. J. Biomed. Sci. 2024, 81, 12366. [Google Scholar] [CrossRef] [PubMed]
- Messenger, A.M.; Barnes, A.N.; Gray, G.C. Reverse zoonotic disease transmission (zooanthroponosis): a systematic review of seldom-documented human biological threats to animals. PLoS ONE 2014, 9, e89055. [Google Scholar] [CrossRef]
- Wang, L.F.; Crameri, G. Emerging zoonotic viral diseases. Rev. Sci. Tech. 2014, 33, 569–581. [Google Scholar] [CrossRef]
- Rahman, M.T.; Sobur, M.A.; Islam, M.S.; Ievy, S.; Hossain, M.J.; El Zowalaty, M.E.; et al. Zoonotic diseases: etiology, impact, and control. Microorganisms 2020, 8, 1405. [Google Scholar] [CrossRef]
- Dharmarajan, G.; Li, R.; Chanda, E.; Dean, K.R.; Dirzo, R.; Jacobsen, K.S.; et al. The animal origin of major human infectious diseases: what can past epidemics teach us about preventing the next pandemic? Zoonoses 2022, 2, 11. [Google Scholar] [CrossRef]
- Marie, V.; Gordon, M.L. The (re-) emergence and spread of viral zoonotic disease: a perfect storm of human ingenuity and stupidity. Viruses 2023, 15, 1638. [Google Scholar] [CrossRef]
- Boseret, G.; Losson, B.; Mainil, J.G.; Thiry, E.; Saegerman, C. Zoonoses in pet birds: review and perspectives. Vet. Res. 2013, 44, 36. [Google Scholar] [CrossRef]
- Hosseinian, S.A. Zoonotic diseases associated with pet birds. J. Zoonotic Dis. 2022, 6, 91–112. [Google Scholar]
- Qiu, Y.; Chenlong, L.; Chen, J.; Sun, Y.; Tang, T.; Zhang, Y.; et al. The global distribution and diversity of wild-bird-associated pathogens: an integrated data analysis and modeling study. Med. 2025, 6, 100553. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.F.W.; To, K.K.W.; Chen, H.; Yuen, K.Y. Cross-species transmission and emergence of novel viruses from birds. Curr. Opin. Virol. 2015, 10, 63–69. [Google Scholar] [CrossRef]
- Contreras, A.; Gómez-Martín, A.; Paterna, A.; Tatay-Dualde, J.; Prats-van der Ham, M.; Corrales, J.C.; et al. Epidemiological role of birds in the transmission and maintenance of zoonoses. Rev. Sci. Tech. 2016, 35, 845–862. [Google Scholar] [CrossRef]
- Wille, M.; Holmes, E.C. Wild birds as reservoirs for diverse and abundant gamma- and delta coronaviruses. FEMS Microbiol. Rev. 2020, 44, 631–644. [Google Scholar] [CrossRef]
- Softić, A.; Nicevic, M.; Koro-Spahic, A.; Terzic, I.; Goletic, S.; Kapo, N.; et al. The monitoring of emergent zoonotic pathogens in wild and captive birds in Sarajevo Canton, Bosnia and Herzegovina. Front. Vet. Sci. 2025, 2, 1621094. [Google Scholar] [CrossRef]
- Najdenski, H.; Dimova, T.; Zaharieva, M.M.; Nikolov, B.; Petrova-Dinkova, G.; Dalakchieva, S.; et al. Migratory birds along the mediterranean - Black Sea flyway as carriers of zoonotic pathogens. Can. J. Microbiol. 2018, 64, 915–924. [Google Scholar] [CrossRef]
- Rappole, J.H.; Derrickson, S.R.; Hubalek, Z. Migratory birds and spread of West Nile virus in the western hemisphere. Emerg. Infect. Dis. 2000, 6, 319–328. [Google Scholar] [CrossRef] [PubMed]
- Cruz, B. The Impact of Living near Industrial Areas on Bird Microbial Health. 2024. Available online: https://www.midogtest.com/blog/the-impact-of-living-near-industrial-areas-on-bird-microbial-health/ (accessed on 18-09-25).
- Newton, I. Migration mortality in birds. Ibis 2024, 167, 106–123. [Google Scholar] [CrossRef]
- Klaassen, M.; Hoye, B.J.; Nolet, B.A.; Buttemer, W.A. Ecophysiology of avian migration in the face of current global hazards. Philos. Trans. R. Soc. Lond. B 2012, 367, 1719–1732. [Google Scholar] [CrossRef] [PubMed]
- Ivy, C.M.; Williamson, J.L. On the physiology of high-altitude flight and altitudinal migration in birds. Integr. Comp. Biol. 2024, 64, 1766–1779. [Google Scholar] [CrossRef]
- McPherson, R.A.; Alger, K.E.; Hofmeister, E. Climate-related drivers of migratory bird health in the south-central USA. Biol. Rev. 2025, 100, 1272–1293. [Google Scholar] [CrossRef]
- Dick, M.; Guglielmo, C.G. Flight muscle protein damage during endurance flight is related to energy expenditure but not dietary polyunsaturated fatty acids in a migratory bird. J. Exp. Biol. 2019, 222, jeb187708. [Google Scholar] [CrossRef]
- Eikenaar, C.; Hessler, S.; Hegemann, A. Migrating birds rapidly increase constitutive immune function during stopover. R. Soc. Open Sci. 2020, 7, 192031. [Google Scholar] [CrossRef] [PubMed]
- Melo, A.M.; Stevens, D.A.; Tell, L.A.; Veríssimo, C.; Sabino, R.; Xavier, M.O. Aspergillosis, avian species and the One Health perspective: the possible importance of birds in azole resistance. Microorganisms 2020, 8, 2037. [Google Scholar] [CrossRef] [PubMed]
- Balakin, E.; Yurku, K.; Ivanov, M.; Izotov, A.; Nakhod, V.; Pustovoyt, V. Regulation of stress induced immunosuppression in the context of neuroendocrine, cytokine, and cellular processes. Biology 2025, 14, 76. [Google Scholar] [CrossRef] [PubMed]
- Alispahic, A.; Softic, A.; Kustura, A.; Omeragic, J.; Goletic, T. Clinical and welfare aspects of immunosuppression in poultry farming. In Clinical and Welfare Aspects of Immunosuppression in Poultry Farming; Bakker, J., de la Garza, M.A., Eds.; Intechopen: Slovenia, 2025; pp. 1–24. [Google Scholar]
- Buchmann, K. Evolution of innate immunity: clues from invertebrates via fish to mammals. Front. Immunol. 2014, 5, 459. [Google Scholar] [CrossRef]
- Wigley, P. Immunology of Birds; John Wiley and Sons, Ltd.: Chichester, 2017. [Google Scholar]
- Davison, F. The importance of the avian immune system and its unique features. In Avian Immunology, 2nd edtn.; Schat, K.A., Kaspers, B., Kaiser, P., Eds.; Academic Press: Boston, 2014; pp. 1–9. [Google Scholar]
- Verma, V.K.; Yadav, S.K.; Haldar, C. Influence of environmental factors on avian immunity: an overview. J. Immun. Res. 2017, 4, 1028. [Google Scholar]
- Schat, K.A.; Skinner, M.A. Avian immunosuppressive diseases and immunoevasion. In Avian Immunology, 2nd Edtn.; Schat, K.A., Kaspers, B., Kaiser, P., Eds.; Academic Press: Boston, 2014; pp. 275–297. [Google Scholar]
- Germinal Centers in Immune Responses: Proceedings of a Symposium; Cottier, H., Ed.; Springer-Verlag: Bern (Switzerland).
- Jenni-Eiermann, S; Srygley, R.B. Physiological aeroecology: anatomical and physiological adaptations for flight. In Aeroecology; Chilson, P., Frick, W.F., Kelly, J., Liechti, F., Eds.; Springer: Cham (Switzerland), 2017; pp. 87–118. [Google Scholar]
- Ingle, D.; Bruellman, R.; Espana, E.; Galloway, K.; Anderson, T.; Meredith, T.L.; Porter, M.E. Science behind the lesson: it’s a bird! it’s a plane! it’s biomechanics! 2019 (course). Accessed on. (accessed on 12-12-25). [CrossRef]
- Dutilleul, M.; Réale, D.; Goussen, B.; Lecomte, C.; Galas, S.; Bonzom, J.M. Adaptation costs to constant and alternating polluted environments. Evol. Appl. 2017, 10, 839–851. [Google Scholar] [CrossRef]
- Jansen, M.; Stoks, R.; Coors, A.; van Doorslaer, W.; de Meester, L. Collateral damage: rapid exposure-induced evolution of pesticide resistance leads to increased susceptibility to parasites. Evolution 2011, 65, 2681–2691. [Google Scholar] [CrossRef]
- Lawton, M. Management of respiratory disease in psittacine birds. In Practice 1999, 21, 76–88. [Google Scholar] [CrossRef]
- Kallapura, G.; Hernandez-Velasco, X.; Pumford, N.R.; Bielke, L.R.; Hargis, B.M.; Tellez, G. Evaluation of respiratory route as a viable portal of entry for Salmonella in poultry. Vet. Med. 2014, 5, 59–73. [Google Scholar]
- Yehia, N.; Salem, H.M.; Mahmmod, Y.; Said, D.; Samir, M.; Mawgod, S.; et al. Common viral and bacterial avian respiratory infections: an updated review. Poult. Sci. 2023, 102, 102553. [Google Scholar] [CrossRef]
- Liu, H.; Pan, S.; Wang, C.; Wenwen, Y.; Xiaofang, W.; Yang, H.; et al. Review of respiratory syndromes in poultry: pathogens, prevention, and control measures. Vet. Res. 2025, 56, 101. [Google Scholar] [CrossRef]
- Hill, D.B.; Button, B.; Rubinstein, M.; Boucher, R.C. Physiology and pathophysiology of human airway mucus. Physiol. Rev. 2022, 102, 1757–1836. [Google Scholar] [CrossRef] [PubMed]
- Kia'i, N.; Bajaj, T. Histology, Respiratory Epithelium. 2023. Available online: https://www.ncbi.nlm.nih.gov/books/NBK541061/.
- Dezube, R. Defense Mechanisms of the Respiratory System. 2025. Available online: https://www.msdmanuals.com/home/lung-and-airway-disorders/biology-of-the-lungs-and-airways/defense-mechanisms-of-the-respiratory-system (accessed on 20-08-25).
- Toth, T.E.; Siegel, P.B. Cellular defense for the avian respiratory tract: paucity of free residing macrophages in the normal chicken. Avian Dis. 1986, 30, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Nganpiep, L.N.; Maina, J.N. Composite cellular defense stratagem in the avian respiratory system: functional morphology of the free (surface) macrophages and specialized pulmonary epithelia. J. Anat. 2002, 200, 499–516. [Google Scholar] [CrossRef] [PubMed]
- Kiama, S.G.; Adekunle, J.S.; Maina, J.N. Comparative in vitro study of interactions between particles and respiratory surface macrophages, erythrocytes, and epithelial cells of the chicken and the rat. J. Anat. 2008, 213, 452–463. [Google Scholar] [CrossRef]
- Mutua, P.M.; Gicheru, M.M.; Makanya, A.N.; Kiama, S.G. Comparative quantitative and qualitative attributes of the surface respiratory macrophages in the domestic duck and the rabbit. Int. J. Morphol. 2011, 29, 353–362. [Google Scholar] [CrossRef]
- Mutua, M.P.; Muya, S.; Gicheru, M.M. Protective roles of free avian respiratory macrophages in captive birds. Biol. Res. 2016, 49, 29. [Google Scholar] [CrossRef]
- Lahellec, C.; Colin, P.; Bennejean, G.; Paquin, J.; Guillerm, A.; Debois, J. Influence of resident Salmonella on contamination of broiler flocks. Poult. Sci. 1986, 65, 2034–2039. [Google Scholar] [CrossRef]
- Mitchell, J.R. The number and location of air sacs in broiler chickens and the implication in Escherichia coli infection. J. S.A. Vet. Assoc. 1984, 2, 57–60. [Google Scholar]
- Bezuidenhout, A.J.; Groenewald, H.B.; Soley, J.T. An anatomical study of the respiratory air sacs in ostriches. Onderstepoort J. Vet. Res. 1999, 66, 317–325. [Google Scholar]
- Daoust, P.Y.; Dobbin, G.V.; Ridlington, A.R.C.F.; Dawson, S.D. Descriptive anatomy of the subcutaneous air diverticula in the Northern gannet, Morus bassanus. Seabird 2008, 21, 64–67. [Google Scholar] [CrossRef]
- Casteleyn, C.; Cornillie, P.; Van Cruchten, S.; Van den Broeck, W.; van Ginneken, C.; Simoens, P. Anatomy of the lower respiratory tract in domestic birds, with emphasis on respiration. Anat. Histol. Embryol. 2018, 47, 89–99. [Google Scholar] [CrossRef]
- da Silva Viegas, K.A.; Correia, S.; Padula, K.; Martins, E.H.; de Vasconcelos Silva, J.A.; Cruz dos, L.E.; Filadelpho, A.L. Morphological analysis of air sacs in red-winged tinamou (Rhynchotus rufescens Temminck, 1815). Brazilian Anim. Sci. 2024, 25, 79886E. [Google Scholar]
- Bezuidenhout, A.J. Light and electron microscopic study of the thoracic respiratory air sacs of the fowl. Anat. Histol. Embryol. 2005, 34, 185–191. [Google Scholar] [CrossRef]
- Maina, J.N. The Lung-Air Sac System of Birds: Development, Structure, and Function.; Springer: Berlin, 2005. [Google Scholar]
- Maina, J.N.; Jimoh, S.A. Study of stress induced failure of the blood-gas barrier and the epithelial-epithelial cells connections of the lung of the domestic fowl, Gallus gallus variant domesticus after vascular perfusion. Biomed. Eng. Comput. Biol. 2013, 5, 77–88. [Google Scholar] [CrossRef]
- Maina, J.N.; Jimoh, S.A. Structural failures of the blood-gas barrier and the epithelial-epithelial cell connections in the different vascular regions of the lung of the domestic fowl (Gallus gallus variant domesticus), at rest and during exercise. Biol Open 2013, 2, 267–76. [Google Scholar] [CrossRef]
- Tell, L.A. Aspergillosis in mammals and birds: impact on veterinary medicine. Med. Mycol. 2005, 43, 71–73. [Google Scholar] [CrossRef]
- Beernaert, L.A.; Pasmans, F.; Van Waeyenberghe, L.; Haesebrouck, F.; Martel, A. Aspergillus infections in birds: a review. Avian Pathol. 2010, 39, 325–31. [Google Scholar] [CrossRef]
- Fletcher, O.J. Pathology of the avian respiratory system. Poult. Sci. 1980, 59, 2666–2679. [Google Scholar] [CrossRef] [PubMed]
- Crosta, L. Respiratory diseases of parrots: anatomy, physiology, diagnosis and treatment. Vet. Clin. Exot. Anim. 2021, 24, 397–418. [Google Scholar] [CrossRef] [PubMed]
- Lightfoot, T.L. Lung and Airway Disorders of Pet Birds (Avian Flu, Bird Flu). Merck Veterinary Manual. 2025. Available online: https://www.msdvetmanual.com/bird-owners/disorders-and-.
- Jordan, A.B.; Gongora, V.; Hartley, D.; Oura, C. A review of eight high-priority, economically important viral pathogens of poultry within the Caribbean region. Vet. Sci. 2018, 5, 14. [Google Scholar]
- Abudabos, A.M.; Samara, E.M.; Hussein, E.O.S.; Al-Ghadi, M.Q.; Al-Atiyat, R.M. Impacts of stocking density on the performance and welfare of broiler chickens. Ital. J. Anim. Sci. 2013, 12, 11. [Google Scholar] [CrossRef]
- Akinyemi, F.T.; Bello, S.F.; Uyanga, V.A.; Oretomiloye, C.; Meng, H. Heat stress and gut microbiota: effects on poultry productivity. Int. J. Poult. Sci. 2020, 19, 294–302. [Google Scholar] [CrossRef]
- Abo Ghanima, M.M.; Abd El-Hack, M.E.; Othman, S.I.; Taha, A.E.; Allam, A.A.; Eid Abdel-Moneim, A.M. Impact of different rearing systems on growth, carcass traits, oxidative stress biomarkers, and humoral immunity of broilers exposed to heat stress. Poult. Sci. 2020, 99, 3070–3078. [Google Scholar] [CrossRef]
- Akinyemi, F.; Adewole, D. Environmental stress in chickens and the potential effectiveness of dietary vitamin supplementation. Front. Anim. Sci. 2021, 2, 775311. [Google Scholar] [CrossRef]
- Hofmann, T.; Schmucker, S.S.; Bessei, W.; Grashorn, M.; Stefanski, V. Impact of housing environment on the immune system in chickens: a review. Animals 2020, 10, 1138. [Google Scholar] [CrossRef]
- Wasti, S.; Sah, N.; Mishra, B. Impact of heat stress on poultry health and performances, and potential mitigation strategies. Animals 2020, 10, 1266. [Google Scholar] [CrossRef] [PubMed]
- Awad, E.A.; Najaa, M.; Zulaikha, Z.A.; Zulkifli, I.; Soleimani, A.F. Effects of heat stress on growth performance, selected physiological and immunological parameters, caecal microflora, and meat quality in two broiler strains. Asian-Australas. J. Anim. Sci 2020, 33, 778–787. [Google Scholar] [CrossRef] [PubMed]
- Abo-Al-Ela, H.G.; El-Kassas, S.; El-Naggar, K.; Abdo, S.E.; Jahejo, A.R.; Al-Wakeel, R.A. Stress and immunity in poultry: light management and nanotechnology as effective immune enhancers to fight stress. Cell Stress Chaperones (CSC) 2021, 26, 457–472. [Google Scholar] [CrossRef] [PubMed]
- Oke, O.E.; Akosile, O.A.; Oni, A.I.; Opowoye, I.O.; Ishola, C.A.; Adebiyi, J.O.; et al. Oxidative stress in poultry production. Poult. Sci. 2024, 103, 104003. [Google Scholar] [CrossRef]
- Ncho, C.J.; Berdos, J.I.; Gupta, V.; Rahman, A.; Mekonnen, K.T.; Bakhsh, A.; 237. Abiotic stressors in poultry production: a comprehensive review. J. Anim. Physiol. Anim. Nutr. 2025, 109, 30–50. [Google Scholar] [CrossRef]
- Elitok, B. Importance of stress factors in poultry. Juniper Online J. Case Stud. 2018, 7, 20–22. [Google Scholar] [CrossRef]
- Eugen, K.V.; Nordquist, R.E.; Zeinstra, E.; Staay, F.J.V. Stocking density affects stress and anxious behavior in the laying hen chick during rearing. Animals 2019, 9, 53. [Google Scholar] [CrossRef] [PubMed]
- Gržinić, G.; Piotrowicz-Cieślak, A.; Klimkowicz-Pawlas, A.; Górny, R.L.; Ławniczek-Wałczyk, A.; Piechowicz, L.; et al. Intensive poultry farming: a review of the impact on the environment and human health. Sci. Total Environ. 2023, 858, 160014. [Google Scholar] [CrossRef] [PubMed]
- Izah, S.C.; Nurmahanova, A.; Ogwu, M.C.; Toktarbay, Z.; Umirbayeva, Z.; Ussen, K.; et al. Public health risks associated with antibiotic residues in poultry food products. J. Agric. Food Res. 2025, 21, 101815. [Google Scholar] [CrossRef]
- Trautmann, S. Climate change impacts on bird species. In Bird Species: How they Arise, Modify and Vanish; Tietze, D.T, Ed.; Springer: Cham (Switzerland), 2018; pp. 217–234. [Google Scholar]
- Freeman, B.G.; Scholer, M.N.; Ruiz-Gutierrez, V.; Fitzpatrick, J.W. Climate change causes upslope shifts and mountain top extirpations in a tropical bird community. Proc. Natl. Acad. Sci. USA 2018, 115, 11982–11987. [Google Scholar] [CrossRef] [PubMed]
- Simmons, R.E.; Barnard, P.; Dean, W.R.J.; Midgley, G.F.; Thuiller, W.; Hughes, G. Climate change and birds: perspectives and prospects from Southern Africa. Ostrich 2005, 75, 295–308. [Google Scholar] [CrossRef]
- Wormworth, J.; Mallon, K. Climate Risk Report - Bird Species and Climate Change. The Global Status Report Version 1.0; Climate Risk Pty Ltd, 2006. Available online: www.climaterisk.net (accessed on 11-09-25).
- Foden, W.B.; Butchart, S.H.M.; Stuart, S.N.; Vié, J.C.; Akcakaya, H.R.; et al. Identifying the world’s most climate change vulnerable species: a systematic trait-based assessment of all birds, amphibians and corals. PLoS ONE 2013, 8, e65427. [Google Scholar] [CrossRef]
- Dutta, H. Insights into the impacts of four current environmental problems on flying birds. Energ. Ecol. Environ. 2017, 2, 329–349. [Google Scholar] [CrossRef]
- Li, B.; Liang, C.; Song, P.; Liu, D.; Qin, W.; Jiang, F.; et al. Threatened birds face new distribution under future climate change on the Qinghai-Tibet Plateau (QTP). Ecol. Indic. 2023, 150, 110217. [Google Scholar] [CrossRef]
- Pottier, P.; Kearney, M.R.; Wu, N.C.; Gunderson, A.R.; Rej, J.E.; Rivera-Villanueva, A.N. Vulnerability of amphibians to global warming. Nature 2025, 639, 954–961. [Google Scholar] [CrossRef] [PubMed]
- Walker, L.A.; Shore, R.F.; Turk, A.; Pereira, M.G.; Best, J. The predatory bird monitoring scheme: identifying chemical risks to top predators in Britain. Ambio 2008, 37, 466–471. [Google Scholar] [CrossRef] [PubMed]
- Wormworth, J.; Şekercioğlu, Ç.H. Winged Sentinels: Birds and Climate Change, 1st Edtn. ed; Cambridge University Press: Cambridge, 2011. [Google Scholar]
- Badry, A.; Krone, O.; Jaspers, V.L.B.; Mateo, R.; García-Fernández, A.; Leivits, M.; Shore, R.F. Towards harmonisation of chemical monitoring using avian apex predators: identification of key species for pan-European biomonitoring. Sci. Total Environ. 2020, 731, 139198. [Google Scholar] [CrossRef]
- Price, T.D.; Hooper, D.M.; Buchanan, C.D.; Johansson, U.S.; Tietze, D.T.; Alstrom, P; et al. Niche filling slows the diversification of Himalayan songbirds. Nature 2014, 509, 222–225. [Google Scholar] [CrossRef]
- Ezard, T.H.; Purvis, A. Environmental changes define ecological limits to species richness and reveal the mode of macroevolutionary competition. Ecol. Lett. 2016, 19, 899–906. [Google Scholar] [CrossRef]
- Boyle, A.W.; Sandercock, B.K.; Martin, K. Patterns and drivers of intraspecific variation in avian life history along elevational gradients: a meta-analysis. Biol. Rev. 2016, 91, 469–82. [Google Scholar] [CrossRef]
- Stronen, A.V.; Norman, A.J.; Vander, W.E.; Paquet, P.C. The relevance of genetic structure in ecotype designation and conservation management. Evol. Appl. 2022, 15, 185–202. [Google Scholar] [CrossRef]
- Cooper, E. Birds and biodiversity: the vital role of birds in ecosystem function. J. Zool. Sci. 2023, 11, 007. [Google Scholar]
- Whelan, C.J.; Şekercioğlu, Ç.H.; Wenny, D.G. Why birds matter: from economic ornithology to ecosystem services. J. Ornithol. 2015, 56, 227–238. [Google Scholar] [CrossRef]
- Why Birds Matter: Avian Ecological Function and Ecosystem Service; Şekercioğlu, Ç.H., Wenny, D.G., Whelan, C.J., Eds.; University of Chicago Press: Chicago (IL), 2016. [Google Scholar]
- Sumasgutner, P.; Cunningham, S.J.; Hegemann, A.; Amar, A.; Watson, H.; Nilsson, J.F.; et al. Interactive effects of rising temperatures and urbanisation on birds across different climate zones: a mechanistic perspective. Glob. Chang. Biol. 2023, 29, 2399–2420. [Google Scholar] [CrossRef] [PubMed]
- Hewitt, G.M. Genetic consequences of climatic oscillations in the quaternary. Phil. Trans. R. Soc. Lond. B 2004, 359, 183–195. [Google Scholar] [CrossRef] [PubMed]
- Prieto-Torres, D.A.; Lira-Noriega, A.; Navarro-Sigüenza, A.G. Climate change promotes species loss and uneven modification of richness patterns in the avifauna associated to neotropical seasonally dry forests. Perspect. Ecol. Conserv. 2010, 18, 19–30. [Google Scholar] [CrossRef]
- Kerr, J.T. Racing against change: understanding dispersal and persistence to improve species' conservation prospects. Proc. Biol. Sci. 2020, 287, 20202061. [Google Scholar] [CrossRef]
- Pfenning-Butterworth, A; Buckley, L.B.; Drake, J.M.; Farner, J.E.; Farrell, M.J.; Gehman, A.M.; et al. Interconnecting global threats: climate change, biodiversity loss, and infectious diseases. Lancet Planet. Health 2024, 8, e270–e283. [Google Scholar] [CrossRef]
- Chen, J.; Wang, Y.L. Effects of habitat fragmentation on bird behavior and extinction mechanisms. Int. J. Mol. Zool. 2024, 14, 97–110. [Google Scholar] [CrossRef]
- Clark, P.; Shakun, J.; Marcott, S.; Mix, A.C.; Eby, M.; Kulp, S.; et al. Consequences of twenty-first-century policy for multi-millennial climate and sea-level change. Nature Clim. Change 2016, 6, 360–369. [Google Scholar] [CrossRef]
- Scafetta, N. Impacts and risks of ‘realistic’ global warming projections for the 21st century. Geosci. Front. 2024, 15, 101774. [Google Scholar] [CrossRef]
- Kaiho, K.; Kaiho, K.; Extinction magnitude of animals in the near future. An animal crisis caused by pollution, deforestation, and warming in the late 21st century and exacerbation by nuclear war. Sci. Rep.;Heliyon 2022, 12 9, 19593.269 e15221. [Google Scholar] [CrossRef]
- Abbass, K.; Qasim, M.Z.; Song, H.; Murshed, M.; Mahmood, H.; Younis, I. A review of the global climate change impacts, adaptation, and sustainable mitigation measures. Environ. Sci. Pollut. Res. 2022, 29, 42539–42559. [Google Scholar] [CrossRef]
- Şekercioğlu, Ç.H.; Primack, R.B.; Wormworth, J. The effects of climate change on tropical birds. Biol. Conserv. 2012, 148, 1–18. [Google Scholar] [CrossRef]
- Mann, M.E.; Zhang, Z.; Hughes, M.K.; Bradley, R.S.; Miller, S.K.; Rutherford, S.; Ni, F. Proxy-based reconstructions of hemispheric and global surface temperature variations over the past two millennia. Proc. Natl. Acad. Sci. USA 2008, 105, 13252–13257. [Google Scholar] [CrossRef]
- Warren, R.; VanDerWal, J.; Price, J.; Welbergen, J.A.; Atkinson, I.; Ramirez-Villegas; et al. Quantifying the benefit of early climate change mitigation in avoiding biodiversity loss. Nature Clim. Change 2013, 3, 678–682. [Google Scholar] [CrossRef]
- Brüniche-Olsen, A.; Kellner, K.F.; DeWoody, J.A. Island area, body size and demographic history shape genomic diversity in Darwin's finches and related tanagers. Mol. Ecol. 2019, 28, 4914–4925. [Google Scholar] [CrossRef]
- Ripple, W.J.; Wolf, C.; Newsome, T.M.; Hoffmann, M.; Wirsing, A.J.; McCauley, D.J. Extinction risk is most acute for the world's largest and smallest vertebrates. Proc. Natl. Acad. Sci. USA 2017, 114, 10678. [Google Scholar] [CrossRef] [PubMed]
- Chichorro, F.; Juslén, A.; Cardoso, P. A review of the relation between species traits and extinction risk. Biol. Conserv. 2019, 237, 220–229. [Google Scholar] [CrossRef]
- Owens, I.P.F.; Bennett, P.M. Ecological basis of extinction risk in birds: habitat loss versus human persecution and introduced predators. Proc. Natl. Acad. Sci. USA 2000, 97, 12144–12148. [Google Scholar] [CrossRef]
- Hughes, E.C.; Edwards, D.P.; Bright, J.A.; Capp, E.J.R.; Cooney, C.R.; Varley, Z.K.; Thomas, G.H. Global biogeographic patterns of avian morphological diversity. Ecol. Lett. 2022, 25, 598–610. [Google Scholar] [CrossRef]
- Hughes, E.C.; Edwards, D.P.; Thomas, G.H. The homogenization of avian morphological and phylogenetic diversity under the global extinction crisis. Curr. Biol. 2022, 32, 3830–3837.e3. [Google Scholar] [CrossRef]
- Ali, J.R.; Blonder, B.W.; Pigot, A.L.; Tobias, J.A. Bird extinctions threaten to cause disproportionate reductions of functional diversity and uniqueness. Funct. Ecol. 2023, 37, 162–175. [Google Scholar] [CrossRef]
- Marra, P.P.; Francis, C.M.; Mulvihill, R.S.; Moore, F.R. The influence of climate on the timing and rate of spring bird migration. Oecologia 2005, 142, 307–315. [Google Scholar] [CrossRef]
- Møller, A.P.; Flensted-Jensen, E.; Klarborg, K.; Marda, W.; Nielsen, J.T. Climate change affects the duration of the reproductive season in birds. J. Anim. Ecol. 2010, 79, 777–784. [Google Scholar] [CrossRef] [PubMed]
- Charmantier, A.; Gienapp, P. Climate change and timing of avian breeding and migration: e volutionary versus plastic changes. Evol. Appl. 2014, 7, 15–28. [Google Scholar] [CrossRef]
- Huntley, B.; Collingham, Y.C.; Green, R.E.; Hilton, G.M.; Rahbek, C.; Willis, S. Potential impacts of climate change upon geographical distributions of birds. Ibis 2006, 148, 8. [Google Scholar] [CrossRef]
- Gill, J.A.; Alves, J.A.; Sutherland, W.J.; Appleton, G.F.; Potts, P.M.; Gunnarsson, T.G. Why is timing of bird migration advancing when individuals are not? Proc. R. Soc. B 2014, 281, 20132161. [Google Scholar] [CrossRef]
- Harnos, A.; Fehérvári, P.; Piross, I.S.; Karcza, Z.; Ágh, N.; Kovács, S.; Csörgo, T. Exploratory analyses of migration timing and morphometrics of the pied flycatcher (Ficedula hypoleuca). Ornis Hung 2016, 24, 109–126. [Google Scholar] [CrossRef]
- Both, C.; Bouwhuis, S.; Lessells, C.M.; Visser, M.W. Climate change and population declines in a long-distance migratory bird. Nature 2006, 441, 81. [Google Scholar] [CrossRef]
- Saino, N.; Ambrosini, R.; Rubolini, D.; von Hardenberg, J.; Provenzale, A.; Hüppop, K.; et al. Climate warming, ecological mismatch at arrival and population decline in migratory birds. Proc. R. Soc. B 2011, 278, 835–842. [Google Scholar] [CrossRef]
- Schmaljohann, H. The start of migration correlates with arrival timing, and the total speed of migration increases with migration distance in migratory songbirds: a cross-continental analysis. Mov. Ecol. 2019, 7, 25. [Google Scholar] [CrossRef]
- Grazer, V.M.; Martin, O.Y. Investigating climate change and reproduction: experimental tools from evolutionary biology. Biology 2012, 1, 411–438. [Google Scholar] [CrossRef] [PubMed]
- Haile, W.A. Impact of climate change on animal production and expansion of animal disease: a review on Ethiopia perspective. Amer. J. Pure Appl. Biosci. 2020, 2, 64–76. [Google Scholar]
- Radchuk, V.; Reed, T.; Teplitsky, C.; van de Pol, M.; Charmantier, A.; Hassall, C.; et al. Adaptive responses of animals to climate change are most likely insufficient. Nat. Commun. 2019, 10, 3109. [Google Scholar] [CrossRef] [PubMed]
- Canteri, E.; Brown, S.C.; Post, E.; Schmidt, N.M.; Nogues-Bravo, D.; Fordham, D.A. Mismatch in reindeer resilience to past and future warming signals ongoing declines. Sci. Adv. 2025, 11, eadu0175. [Google Scholar] [CrossRef] [PubMed]
- Evans, D.M.; Che-Castaldo, J.P.; Crouse, D.; Davis, F.W.; Epanchin-Niell, R.; Flather, C.H.; et al. Species recovery in the United States: increasing the effectiveness of the endangered species act. Issues in Ecology 2016, 20. [Google Scholar]
- Taylor, G. Improving the Recovery Efforts of Threatened Species. PhD Thesis, University College London, 2019.
- Kovach, A.I.; Cheeseman, A.E.; Cohen, J.B.; Rittenhouse, C.D.; Whipps, C.M. Separating proactive conservation from species listing decisions. Environ. Manag. 2022, 70, 710–729. [Google Scholar] [CrossRef]
- Cullen, D.; Kingsford, R.T.; Bino, G.; West, R.; Letnic, M.; Pedler, R. Bucking the trend recovery from near continent-wide extinction by a marsupial micro-predator during drought. Biol. Conserv. 2025, 311, 111411. [Google Scholar] [CrossRef]
- Ricklefs, R. E.; Travis, J. A morphological approach to the study of avian community organization. Auk 1980, 97, 321–338. [Google Scholar]
- Ecological Morphology: Integrative Organismal Biology; Wainwright, P.C., Reilly, S.M., Eds.; University of Chicago Press: Chicago, 1994. [Google Scholar]
- Ricklefs, R.E.; Miles, D.B. Ecological and evolutionary inferences from morphology: an ecological perspective. In Ecological Morphology: Integrative Organismal Biology; Wainwright, P.C., Reilly, S.M., Eds.; University of Chicago Press: Chicago, 1994; pp. 13–41. [Google Scholar]
- Acevedo-Whitehouse, K; Duffus, A.L. Effects of environmental change on wildlife health Philos. Trans. R. Soc. Lond. B, Biol. Sci. 2009, 364, 3429–38. [Google Scholar] [CrossRef]
- Richard, F.J.; Southern, I.; Gigauri, M.; Bellini, G.; Rojas, O.; Runde, A. Warning on nine pollutants and their effects on avian communities. Global. Ecol. Conserv. 2021, 32, e01898. [Google Scholar] [CrossRef]
- Steyn, L.; Bouwman, H.; Maina, J.N. Associations between DDT and egg parameters of the house sparrow, Passer domesticus from the Thohoyandou area of South Africa. Chemosphere 2018, 198, 249–256. [Google Scholar] [CrossRef]
- Bouwman, H.; Yohannes, Y.B.; Nakayama, S.M.M.; Motohira, K.; Ishizuka, M.; Humphries, M.S.; et al. Evidence of impacts from DDT in pelican, cormorant, stork, and egret eggs from KwaZulu-Natal, South Africa. Chemosphere 2019, 225, 647–658. [Google Scholar] [CrossRef]
- Mansfield, I.; Reynolds, S.J.; Lynch, I.; Matthews, T.J.; Sadler, J.P. Birds as bioindicators of plastic pollution in terrestrial and freshwater environments: a 30-year review. Environ. Pollut. 2024, 348, 123790. [Google Scholar] [CrossRef]
- Bundle, M.; Hoppeler, H.; Vock, R.; Tester, J.M.; Weyand, P.G. High metabolic rates in running birds. Nature 1999, 397, 31–32. [Google Scholar] [CrossRef]
- Hedenström, A. Extreme endurance migration: what is the limit to non-stop flight? PLoS Biol. 2010, 8, e1000362. [Google Scholar] [CrossRef]
- Conklin, J.R.; Senner, N.R.; Battley, P.F.; Piersma, T. Extreme migration and the individual quality spectrum. J. Avian Biol. 2017, 48, 19–36. [Google Scholar] [CrossRef]
- Costantini, D. Oxidative stress in ecology and evolution: lessons from avian studies. Ecol. Lett. 2008, 11, 1238–1251. [Google Scholar] [CrossRef]
- Jenni-Eiermann, S.; Jenni, L.; Smith, S.; Costantini, D. Oxidative stress in endurance flight: an unconsidered factor in bird migration. PLoS One 2014, 9, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, J.S.; Sabat, P.; Castañeda, L.E.; Contreras, C.; Navarrete, L.; Peña-Villalobos, I.; Navedo, J.G. Oxidative status and metabolic profile in a long-lived bird preparing for extreme endurance migration. Sci. Rep. 2019, 9, 17616. [Google Scholar] [CrossRef]
- Eikenaar, C.; Hessler, S.; Hegemann, A. Migrating birds rapidly increase constitutive immune function during stopover. R. Soc. Open Sci. 2020, 7, 192031. [Google Scholar] [CrossRef]
- Kuo, D.T.F.; Rattner, B.A.; Marteinson, S.C.; Letcher, R.; Fernie, K.J.; Treu, G.; et al. A critical review of bioaccumulation and biotransformation of organic chemicals in birds. Rev. Env. Contamination 2022, 260, 6. [Google Scholar] [CrossRef]
- Sanderfoot, O.V.; Holloway, T. Air pollution impacts on avian species via inhalation exposure and associated outcomes. Environ. Res. Lett. 2017, 12, 083002. [Google Scholar] [CrossRef]
- Egwumah, F.A.; Egwumah, P.O.; Edet, D.I. Paramount roles of wild birds as bioindicators of contamination. Int. J. Avian Wildlife Biol. 2017, 2, 194–200. [Google Scholar]
- Barton, M.G.; Henderson, I.; Border, J.A.; Siriwardena, G. A review of the impacts of air pollution on terrestrial birds. Sci. Total Environ. 2023, 873, 162136. [Google Scholar] [CrossRef] [PubMed]
- Carrasco, L.; Jiménez-Mora, E.; Utrilla, M.J.; Pizarro, I.T.; Reglero, M.M.; Rico-San Román, L.; Martin-Maldonado, B. Birds as bioindicators: revealing the widespread.
- impact of microplastics. Birds 2025, 6, 10. [CrossRef]
- Souto, H.N.; de Campos Júnior, E.O.; Siqueira, M.V.B.M.; Campos, C.F.; Morais, C.R.; Pereira, B.B.; Morelli, S. Birds as environmental bioindicators of genotoxicity in Brazilian cerrado farmlands: an in situ approach. Animals 2025, 15, 3208. [Google Scholar] [CrossRef] [PubMed]
- Smits, J.E.G.; Fernie, K.J. Avian wildlife as sentinels of ecosystem health. Comp. Immunol. Microbiol. Infect. Dis. 2013, 36, 333–342. [Google Scholar] [CrossRef]
- Brown, R.E.; Brain, J.D.; Wang, N. The avian respiratory system: a unique model for studies of respiratory toxicosis and for monitoring air quality. Environ. Health Perspect. 1997, 105, 188–200. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Ma, J. Waterbirds as bioindicators of wetland heavy metal pollution. Procedia Environ. Sci. 2011, 10, 2769–2774. [Google Scholar] [CrossRef]
- Smith, P. A.; Smith, A. C.; Andres, B.; Francis, C. M.; Harrington, B.; Friis, C.; et al. Accelerating declines of North America’s shorebirds signal the need for urgent conservation action. Ornithol. Appl. 2023, 125, duad003. [Google Scholar] [CrossRef]
- Fry, D.M. Reproductive effects in birds exposed to pesticides and industrial chemicals. Environ. Health Perspect. 1995, 7, 165–171. [Google Scholar]
- Abbasi, N.A.; Jaspers, V.L.B.; Chaudhry, M.J.I.; Ali, S; Malik, R.N. Influence of taxa, trophic level, and location on bioaccumulation of toxic metals in bird’s feathers: a preliminary biomonitoring study using multiple bird species from Pakistan. Chemosphere 2015, 120, 527–537. [Google Scholar] [CrossRef]
- Shore, R.F.; Taggart, M.A. Population-level impacts of chemical contaminants on apex avian species. Curr. Opin. Environ. Sci. Health 2019, 11, 65–70. [Google Scholar] [CrossRef]
- Maznikova, V.N.; Ormerod, S.J.; Gómez-Serrano, M.A. Birds as bioindicators of river pollution and beyond: specific and general lessons from an apex predator. Ecol. Indic. 2024, 158, 111366. [Google Scholar] [CrossRef]
- Castagna, F.; Montano, L.; Lombardi, R.; Pagano, A.; Gigliotti, A.; Bava, R.; et al. Understanding environmental contamination through the lens of the peregrine falcon (Falco peregrinus). Environments 2024, 11, 264. [Google Scholar] [CrossRef]
- Vetere, A.; Di Ianni, F.; Gavezzoli, M.; Cococcetta, C. Avian toxicoses: a review. Front. Vet. Sci. 2025, 12, 1572736. [Google Scholar] [CrossRef]
- Salaberria, C.; Chávez-Zichinelli, C.A.; López-Rull, I.; Romano, M.C.; Schondube, J.E. Physiological status of house sparrows (Passer domesticus) along an ozone pollution gradient. Ecotoxicology 2023, 32, 261–272. [Google Scholar] [CrossRef]
- Zhang, H.; Guo, J.; Peng, P.; Wang, M.; Shen, J.; Sun, X.; et al. Evolution and biological characteristics of the circulated H8N4 avian influenza viruses. J. Integr. Agric. 2025, 24, 23422355. [Google Scholar] [CrossRef]
- Salmón, P.; Stroh, E.; Herrera-Dueñas, A.; von Post, M.; Isaksson, C. Oxidative stress in birds along a NOx (nitrogen oxides) and urbanisation gradient: an interspecific approach. Sci. Total Environ. 2018, 622, 635643. [Google Scholar]
- Liang, Y.; Rudik, I.; Zou, E. Y.; Johnston, A.; Rodewald, A. D.; Kling, C. L. Conservation cobenefits from air pollution regulation: evidence from birds. Proc. Natl. Acad. Sci. USA 2020, 117, 30900–30906. [Google Scholar] [CrossRef]
- Jat, R.; Gurjar, B.R. Contribution of different source sectors and source regions of Indo- Gangetic Plain in India to PM2.5 pollution and its short-term health impacts during peak polluted winter. Atmos. Pollut. Res. 2021, 12, 89–100. [Google Scholar] [CrossRef]
- Hedenström, A.; Hedh, L. Seasonal patterns and processes of migration in a long-distance migratory bird: energy or time minimization? Proc. R. Soc. B 2024, 291, 20240624. [Google Scholar] [CrossRef] [PubMed]
- Cortes-Ramirez, J.; Naish, S.; Sly, P. D.; Jagals, P. Mortality and morbidity in populations in the vicinity of coal mining: a systematic review. BMC Public Health 2018, 18, 1–17. [Google Scholar] [CrossRef]
- Sanderfoot, O.V.; Tingley, M.W.; Bassing, S.B.; Vaughan, J.K.; June, N.A.; Gardner, B. Hazardous wildfire smoke events can alter dawn soundscapes in dry forests of central and eastern Washington, United States. Global Ecol. Conserv. 2024, 54, e03044. [Google Scholar] [CrossRef]
- Saeed, M; Abbas, G.; Alagawany, M.; Kamboh, A.A.; Abd, E.H.; Mohamed, E.; Khafaga, A.F.; Chao, S. Heat stress management in poultry farms: a comprehensive overview. J. Therm. Biol. 2019, 84, 414–425. [Google Scholar] [CrossRef]
- Brugaletta, G.; Teyssier, J.R.; Rochell, S.J.; Dridi, S.; Sirri, F. A review of heat stress in chickens. Part I: insights into physiology and gut health. Front. Physiol. 2022, 13, 934381. [Google Scholar] [CrossRef] [PubMed]
- McKechnie, A.E.; Wolf, B.O. Climate change increases the likelihood of catastrophic avian mortality events during extreme heat waves. Biol Lett. 2010, 6, 253–256. [Google Scholar] [CrossRef] [PubMed]
- Irannezhad, M.; Tahami, M.S.; Ahmadi, B.; Liu, J.; Chen, D. Compound climate extreme events threaten migratory birds’ conservation in western U.S. Sustainable Horizons 2022, 3, 100023. [Google Scholar] [CrossRef]
- Kotz, M.; Amano, T.; Watson, J.E.M. Large reductions in tropical bird abundance attributable to heat extreme intensification. Nat. Ecol. Evol. 2025, 9, 1897–1909. [Google Scholar] [CrossRef]
- Bathiany, S.; Dakos, V.; Scheffer, M.; Lenton, T.M. Climate models predict increasing temperature variability in poor countries. Sci. Adv. 2018, 4, eaar5809. [Google Scholar] [CrossRef]
- McBride, C.M.; Kruger, A.C.; Johnston, C.; Dyson, L. Projected changes in daily temperature extremes for selected locations over South Africa. Weather and Climate Extremes 2025, 47, 100753. [Google Scholar] [CrossRef]
- Liu, Q.; Fu, C.; Xu, Z.; Aijun, D. Global warming intensifies extreme day-to-day temperature changes in mid–low latitudes. Nat. Clim. Chang. 2026, 16, 69–76. [Google Scholar] [CrossRef]
- Gorta, S.B.Z.; Allen, P; Kingsford, R.T.; Berryman, A.J.; Davies, J.; Roderick, M.; et al. Environmental drivers of gadfly petrel vagrancy in the Southwest Pacific. Ibis 2026. [Google Scholar] [CrossRef]
- Phillips, R.A.; Fort, J.; Dias, M.P. Conservation status and overview of threats to seabirds. In Conservation of Marine Birds; Young, L., VanderWerf, E., Eds.; Academic Press: London, 2023; pp. 35–56. [Google Scholar]
- Piironen, A.; Knetter, J.M.; Spragens, K.A.; Dooley, J.L.; Patil, V.; Reed, E.T.; et al. Environmental drivers of productivity explain population patterns of an Arctic-nesting bird across a half-century. Ecol. Appl. 2025, 35, e70067. [Google Scholar] [CrossRef] [PubMed]
- Bressler, S.A.; Diamant, E.S.; Tingley, M.W.; Yeh, P.J. Nests in the cities: adaptive and non-adaptive phenotypic plasticity and convergence in an urban bird. Proc. Biol. Sci. 2020, 287, 20202122. [Google Scholar] [CrossRef] [PubMed]
- Burton, J.F. Birds and Climate Change; Christopher Helm: London, 1995. [Google Scholar]
- Harper, M.; Rytwinski, T.; Creed, I.F.; Helmuth, B.; Smol, J.P.; Bennett, J.R.; et al. A multi-realm perspective on applying potential tipping points to environmental decision-making. Environ. Rev. 2024, 32, 131–144. [Google Scholar] [CrossRef]
- Fattorini, D. Environmental Quality and Global Health; Academia Global and Public Health, 2025. Available online: https://api.semanticscholar.org/CorpusID:283662081.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



