Submitted:
03 March 2026
Posted:
03 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
Cell Lines
Cell Treatments and Preparation of CM
Senescence-Associated β-Galactosidase Staining
Western Blotting
Invasion Assay
Real-Time PCR
Total ROS Quantification
Prostate and Ovarian Sphere Formation
Cell Transfection
Cell Viability
Determination of GSH/GSSG
Determination of Gln and Ammonium Levels
Gas Chromatography–Mass Spectrometry (GC-MS) Analysis
Confocal Immunofluorescence
Statistical Analysis
3. Results
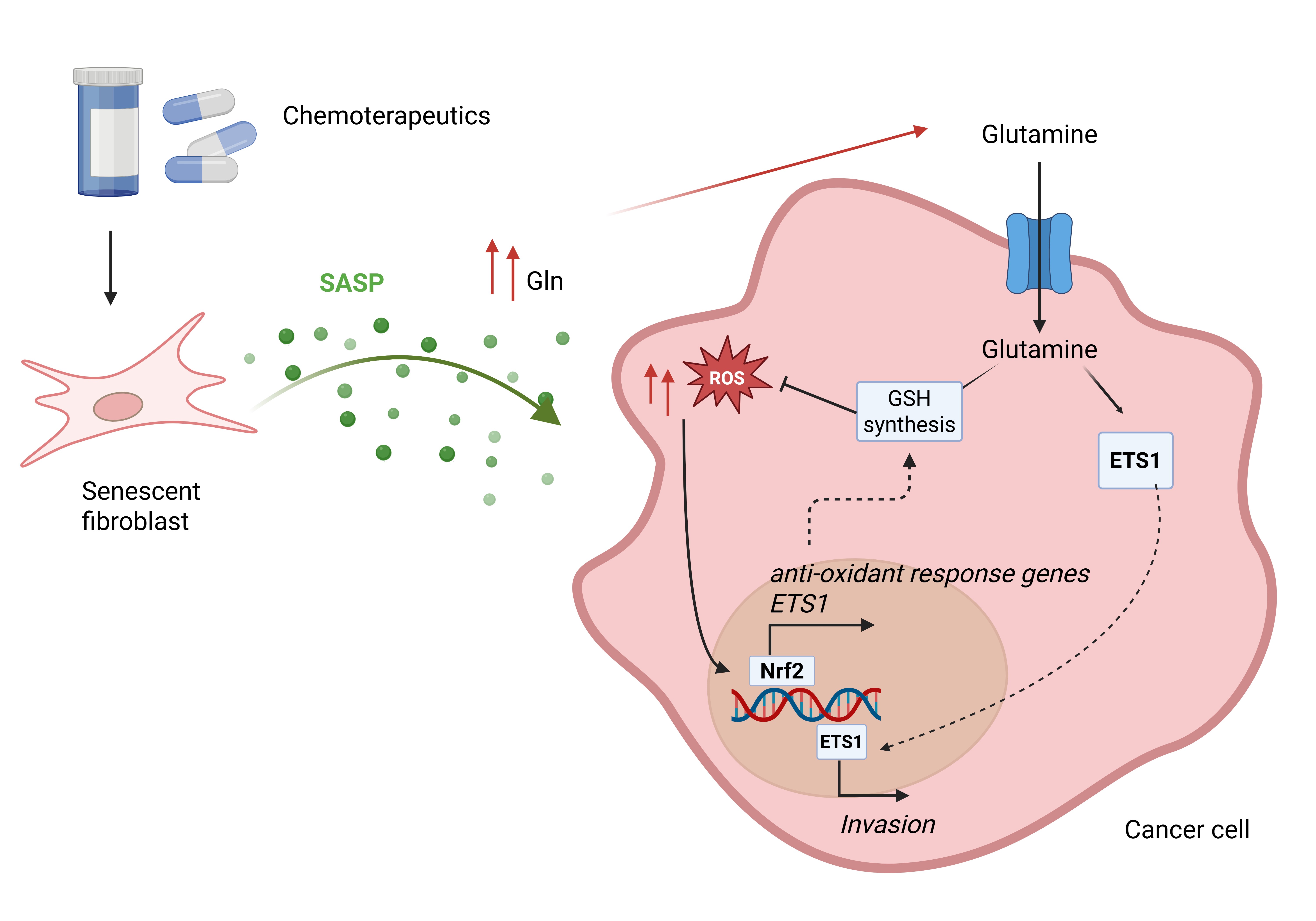
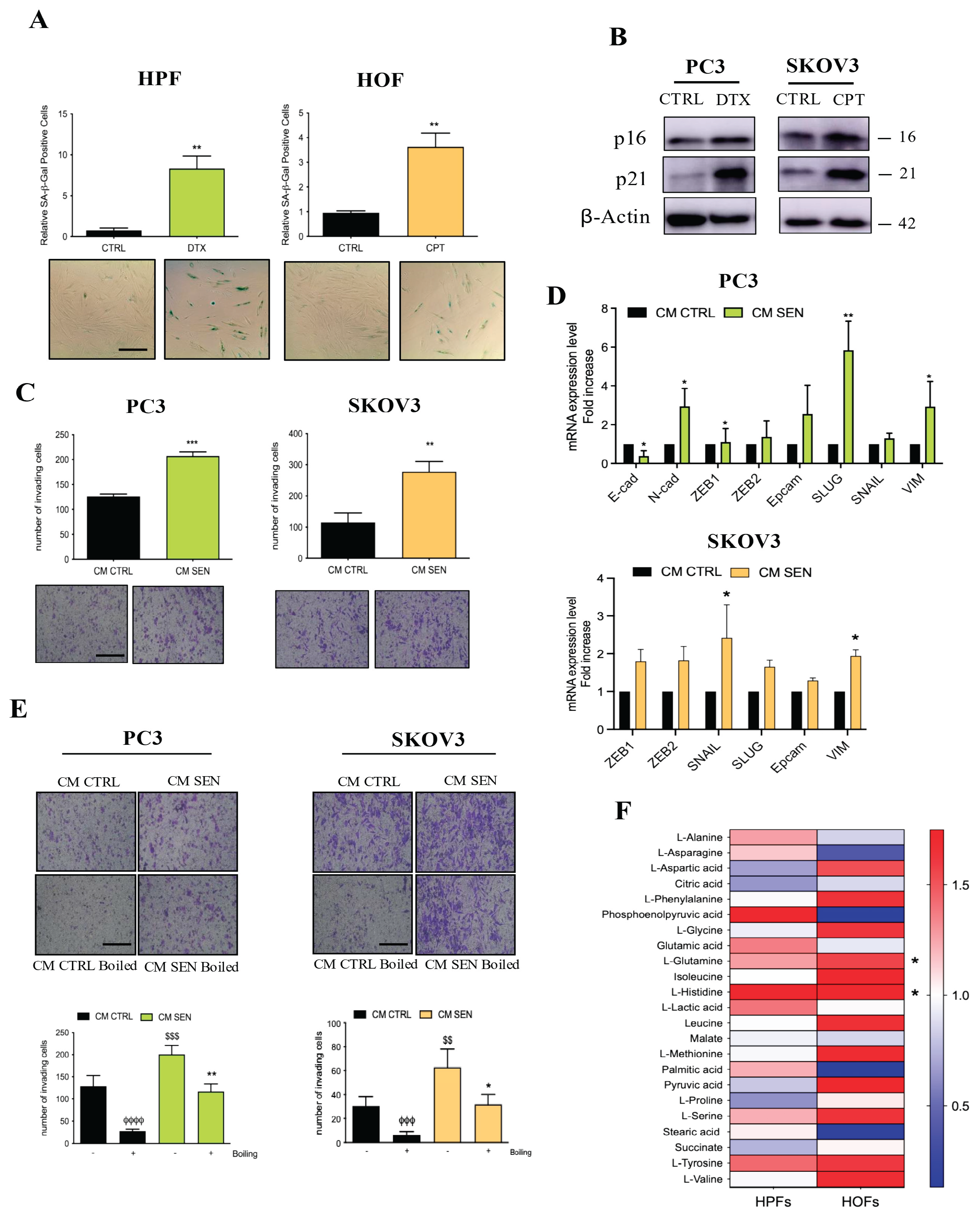
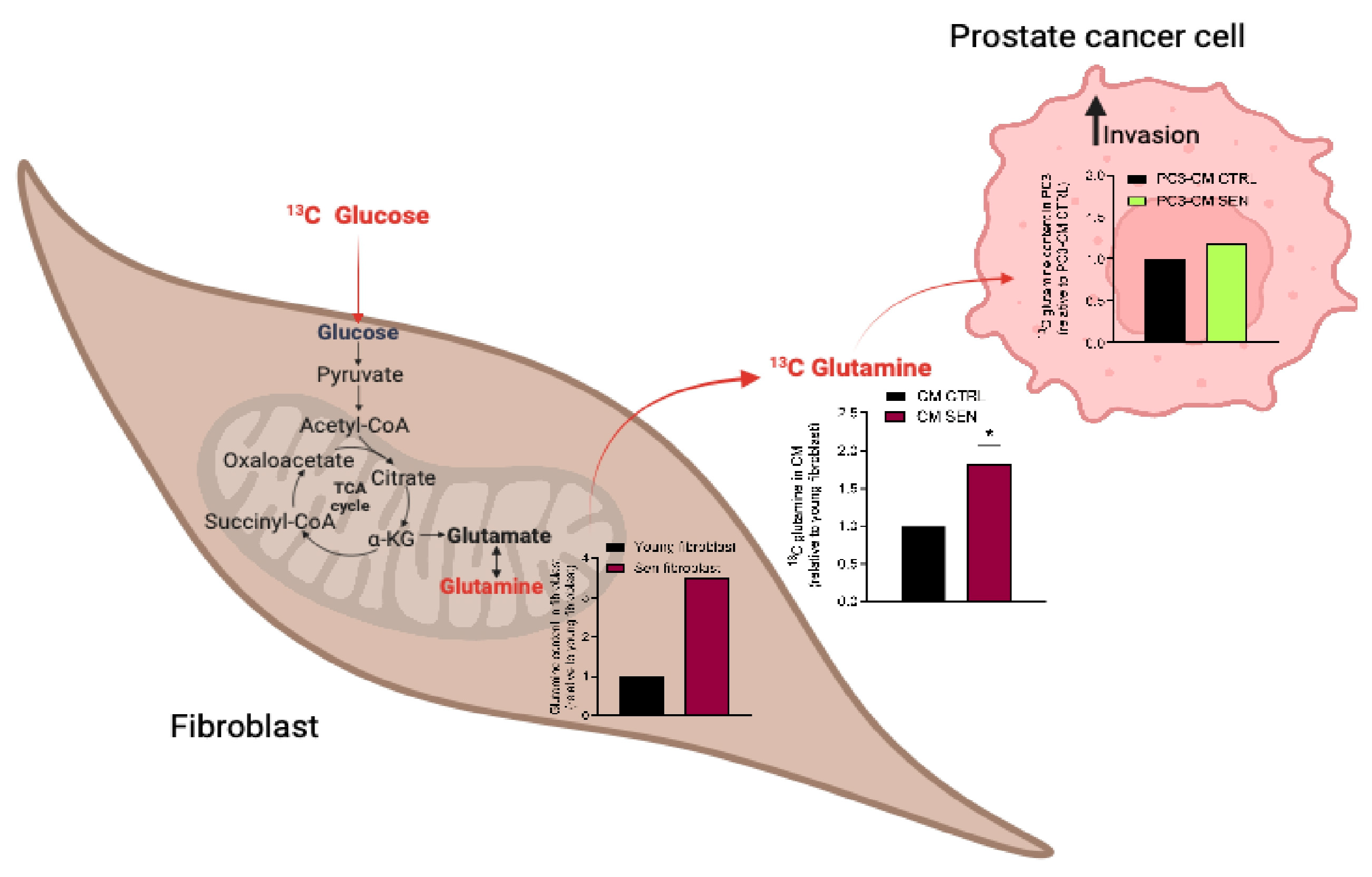
3.1. The Metabolic Composition of CM Derived from Senescent Fibroblasts Supports Ovarian and Prostate Cancer Cell Invasion
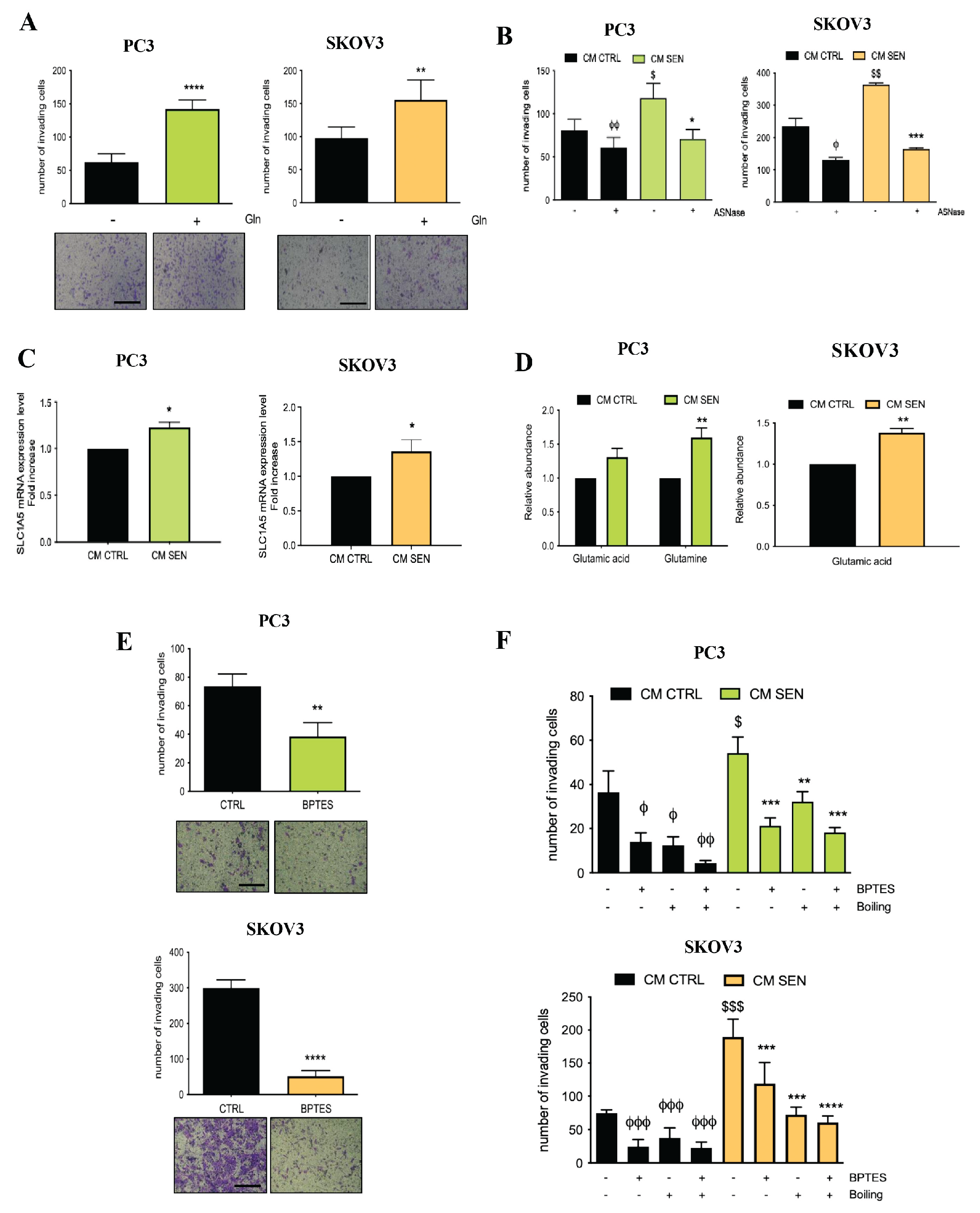
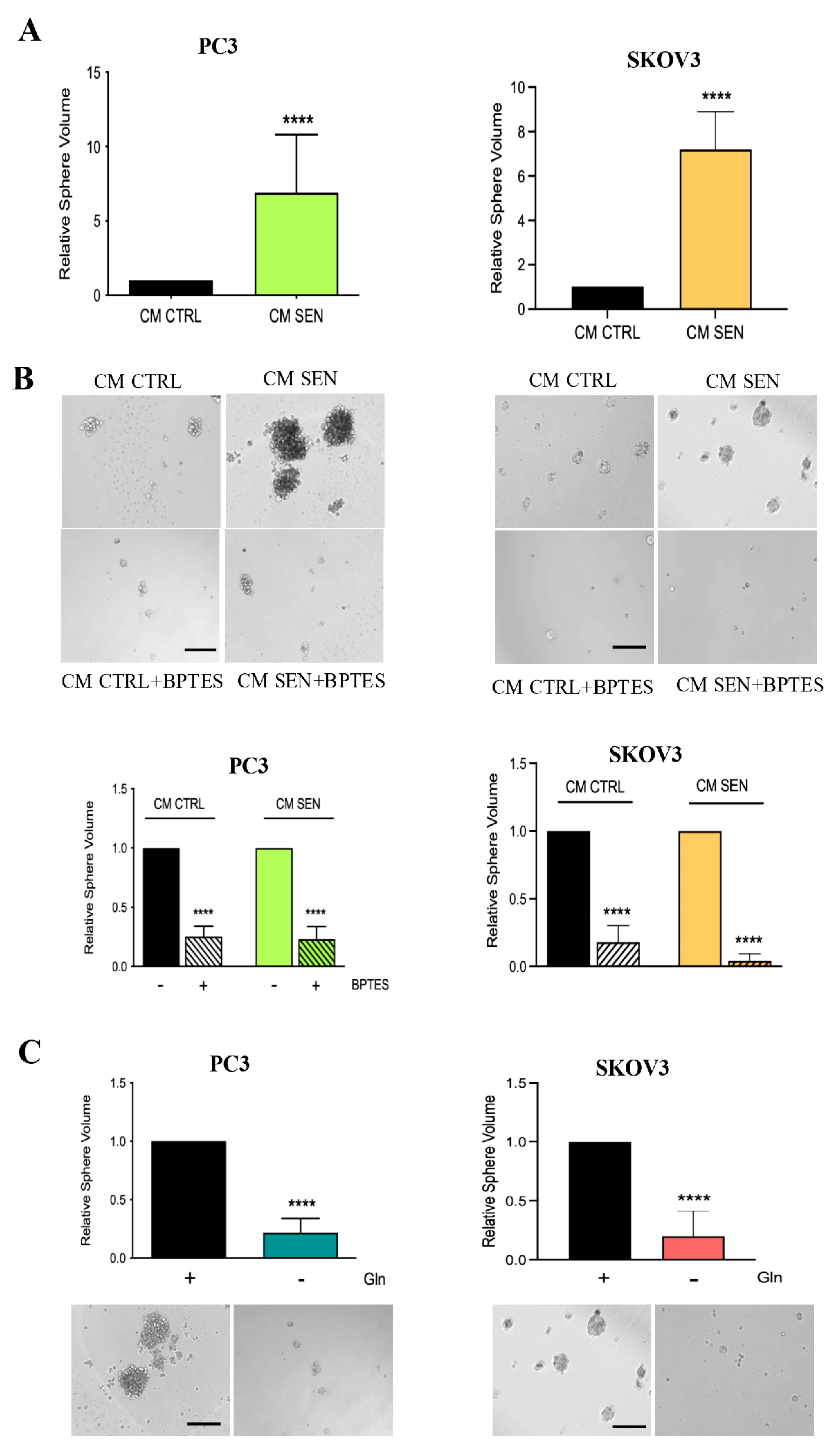
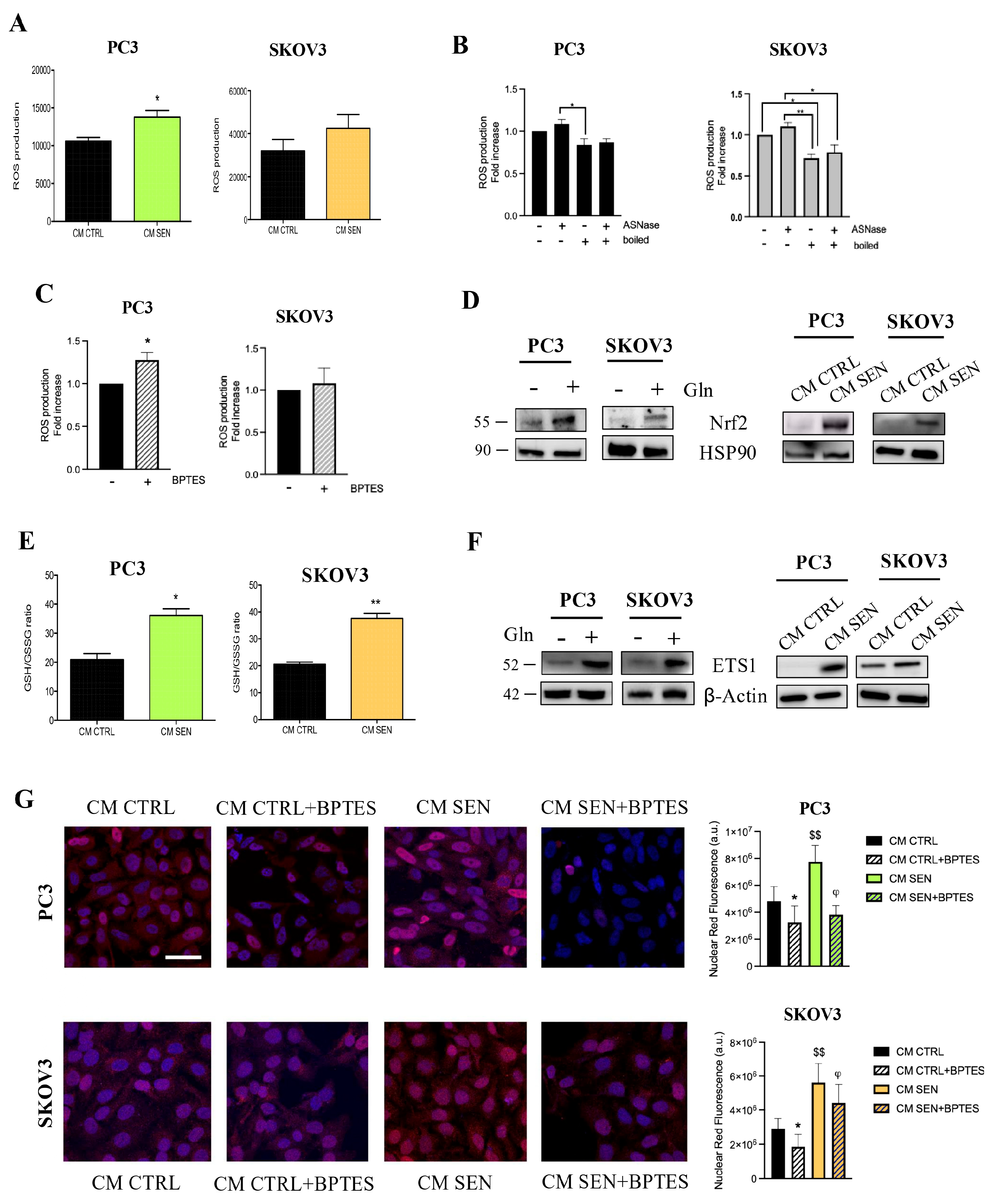
3.2. Availability of Senescent Stroma-Derived Gln Drives Invasive Abilities of PC3 and SKOV3 Cells
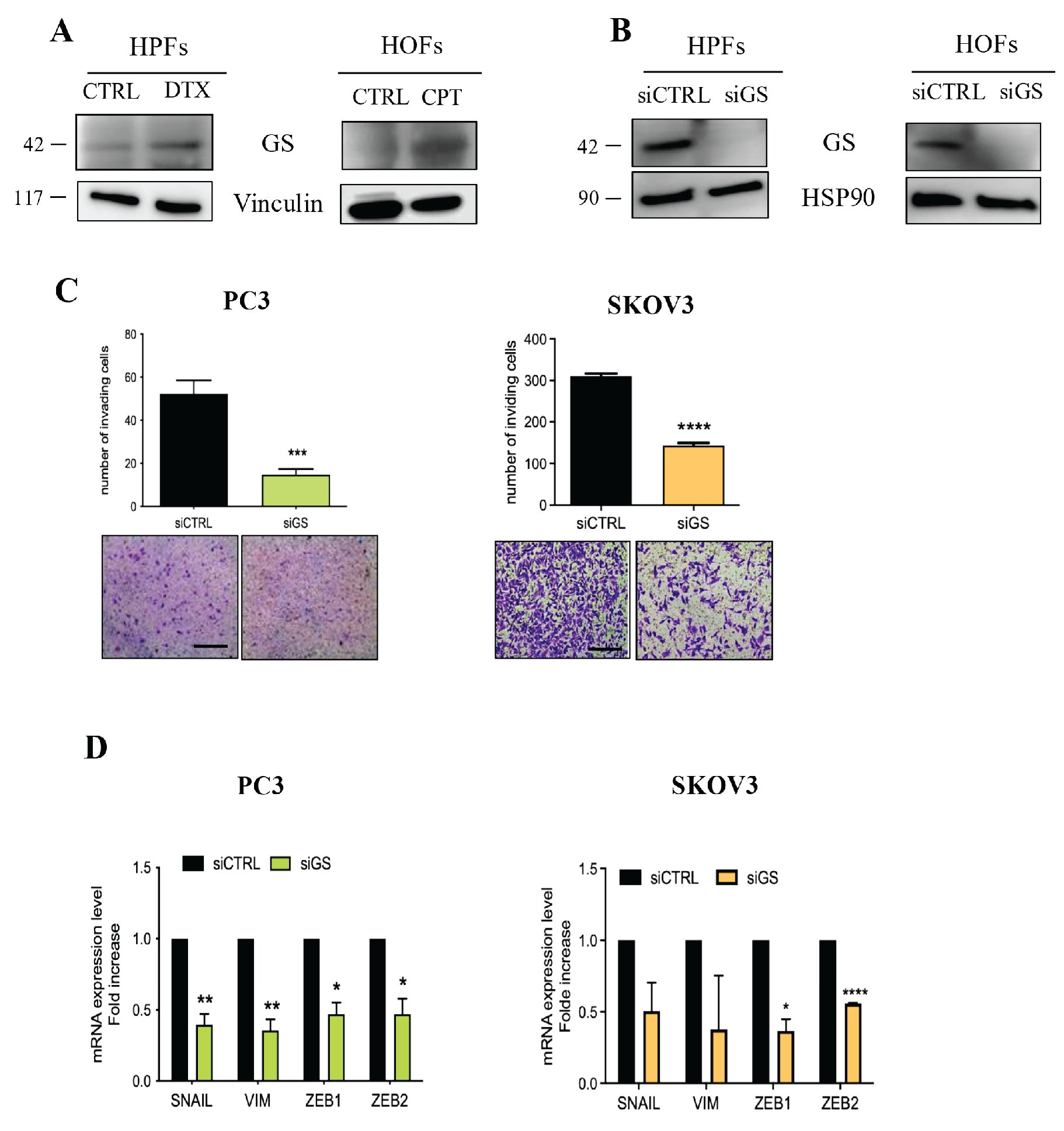
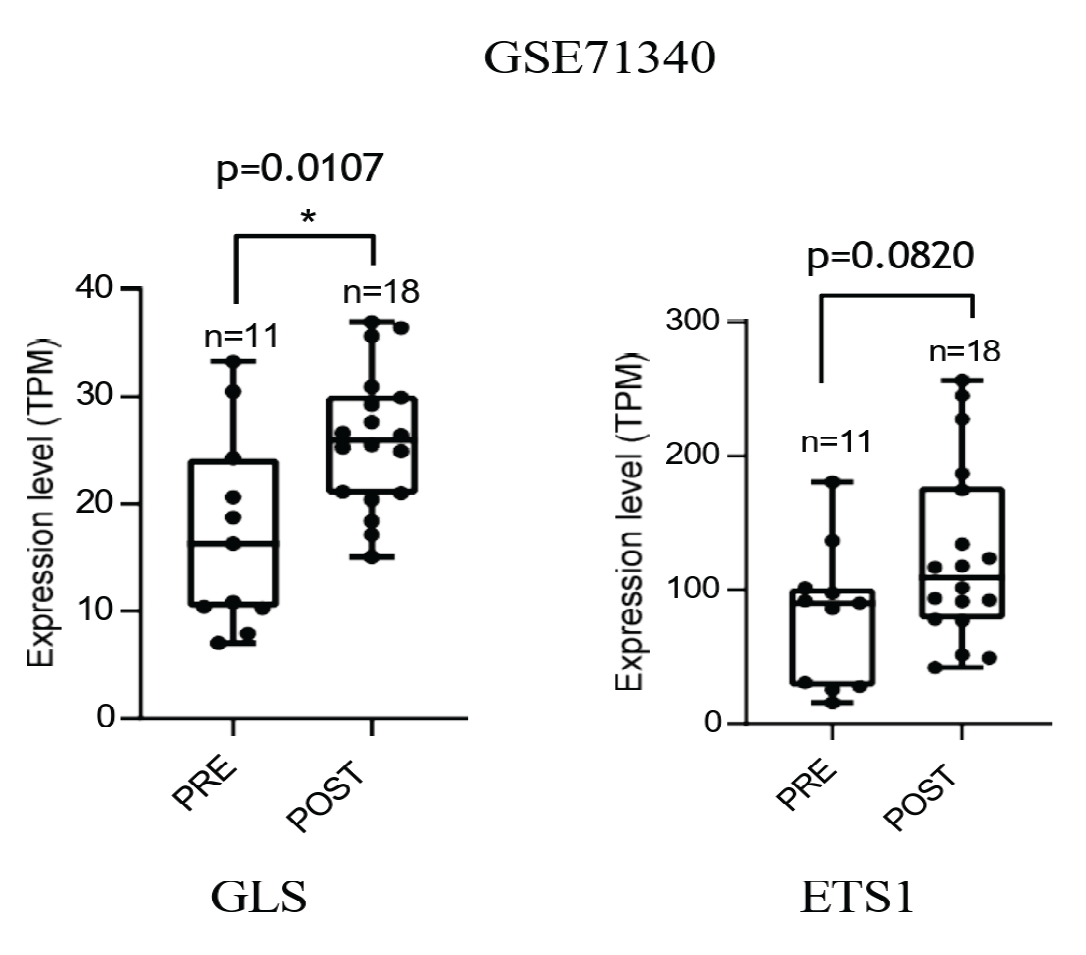
3.3. Senescent Stroma Upregulates Gln Synthetase, Sustaining Gln Metabolism
3.4. Senescent Stroma-Derived Gln Drives the Invasive Abilities of Cancers Cells via a NRF2/ETS1 Axis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ASNase | L-asparaginase |
| CM | Conditioned media |
| CPT | Cisplatin |
| DTX | Docetaxel |
| EMT | Epithelial-to-mesenchymal transition |
| Gln | Glutamine |
| GLS1 | Glutaminase-1 |
| GS | Gln synthetase |
| HOFs | Human ovarian fibroblasts |
| HPFs | Human prostate fibroblasts |
| ROS | Reactive oxygen species |
| SASP | Senescence-associated secretory phenotype |
| TIS | Therapy-induced senescence |
| TME | Tumor microenvironment |
References
- Ye, M.; Huang, X.; Wu, Q.; Liu, F. Senescent Stromal Cells in the Tumor Microenvironment: Victims or Accomplices? Cancers (Basel) 2023, 15. [Google Scholar] [CrossRef]
- Campisi, J.; d'Adda di Fagagna, F. Cellular senescence: when bad things happen to good cells. Nat Rev Mol Cell Biol 2007, 8, 729–740. [Google Scholar] [CrossRef]
- Liu, K.; Huang, H.; Zhang, M.; Chen, S.; Yang, Y.; Fang, C.; Zhong, X. When therapy-induced senescence meets tumors: A double-edged sword: A review. Medicine (Baltimore) 2025, 104, e42886. [Google Scholar] [CrossRef]
- Takasugi, M.; Yoshida, Y.; Hara, E.; Ohtani, N. The role of cellular senescence and SASP in tumour microenvironment. FEBS J 2023, 290, 1348–1361. [Google Scholar] [CrossRef] [PubMed]
- Dong, Z.; Luo, Y.; Yuan, Z.; Tian, Y.; Jin, T.; Xu, F. Cellular senescence and SASP in tumor progression and therapeutic opportunities. Mol Cancer 2024, 23, 181. [Google Scholar] [CrossRef] [PubMed]
- Coppé, J.P.; Desprez, P.Y.; Krtolica, A.; Campisi, J. The senescence-associated secretory phenotype: the dark side of tumor suppression. Annu Rev Pathol 2010, 5, 99–118. [Google Scholar] [CrossRef] [PubMed]
- Pardella, E.; Pranzini, E.; Nesi, I.; Parri, M.; Spatafora, P.; Torre, E.; Muccilli, A.; Castiglione, F.; Fambrini, M.; Sorbi, F.; et al. Therapy-Induced Stromal Senescence Promoting Aggressiveness of Prostate and Ovarian Cancer. Cells 2022, 11. [Google Scholar] [CrossRef]
- Wiley, C.D.; Campisi, J. The metabolic roots of senescence: mechanisms and opportunities for intervention. Nat Metab 2021, 3, 1290–1301. [Google Scholar] [CrossRef]
- Boroughs, L.K.; DeBerardinis, R.J. Metabolic pathways promoting cancer cell survival and growth. Nat Cell Biol 2015, 17, 351–359. [Google Scholar] [CrossRef]
- Aguilar-Cazares, D.; Perez-Medina, M.; Benito-Lopez, J.J.; Galicia-Velasco, M.; Meneses-Flores, M.; Camarena, A.; Lopez-Gonzalez, J.S. The tumor microenvironment: adding pieces to the puzzle. Front Immunol 2025, 16, 1731338. [Google Scholar] [CrossRef]
- Nan, D.; Yao, W.; Huang, L.; Liu, R.; Chen, X.; Xia, W.; Sheng, H.; Zhang, H.; Liang, X.; Lu, Y. Glutamine and cancer: metabolism, immune microenvironment, and therapeutic targets. Cell Commun Signal 2025, 23, 45. [Google Scholar] [CrossRef]
- Mancini, C.; Lori, G.; Mattei, G.; Iozzo, M.; Desideri, D.; Cianchi, F.; Fortuna, L.; Passagnoli, F.; Massi, D.; Ugolini, F.; et al. PHGDH drives 5-FU chemoresistance in colorectal cancer through the Hedgehog signaling. J Exp Clin Cancer Res 2025, 44, 198. [Google Scholar] [CrossRef] [PubMed]
- Heinrich, P.; Kohler, C.; Ellmann, L.; Kuerner, P.; Spang, R.; Oefner, P.J.; Dettmer, K. Correcting for natural isotope abundance and tracer impurity in MS-, MS/MS- and high-resolution-multiple-tracer-data from stable isotope labeling experiments with IsoCorrectoR. Sci Rep 2018, 8, 17910. [Google Scholar] [CrossRef] [PubMed]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: an open-source platform for biological-image analysis. Nat Methods 2012, 9, 676–682. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Peng, X.; Li, Y.; Wei, S.; He, G.; Liu, J.; Yang, S.; Li, D.; Lin, W.; Fang, J.; et al. Glutamine addiction in tumor cell: oncogene regulation and clinical treatment. Cell Commun Signal 2024, 22, 12. [Google Scholar] [CrossRef]
- Yang, L.; Achreja, A.; Yeung, T.L.; Mangala, L.S.; Jiang, D.; Han, C.; Baddour, J.; Marini, J.C.; Ni, J.; Nakahara, R.; et al. Targeting Stromal Glutamine Synthetase in Tumors Disrupts Tumor Microenvironment-Regulated Cancer Cell Growth. Cell Metab 2016, 24, 685–700. [Google Scholar] [CrossRef]
- Jin, J.; Byun, J.K.; Choi, Y.K.; Park, K.G. Targeting glutamine metabolism as a therapeutic strategy for cancer. Exp Mol Med 2023, 55, 706–715. [Google Scholar] [CrossRef]
- Allgayer, H.; Mahapatra, S.; Mishra, B.; Swain, B.; Saha, S.; Khanra, S.; Kumari, K.; Panda, V.K.; Malhotra, D.; Patil, N.S.; et al. Epithelial-to-mesenchymal transition (EMT) and cancer metastasis: the status quo of methods and experimental models 2025. Mol Cancer 2025, 24, 167. [Google Scholar] [CrossRef]
- Matés, J.M.; Campos-Sandoval, J.A.; de Los Santos-Jiménez, J.; Márquez, J. Glutaminases regulate glutathione and oxidative stress in cancer. Arch Toxicol 2020, 94, 2603–2623. [Google Scholar] [CrossRef]
- Sayin, V.I.; LeBoeuf, S.E.; Singh, S.X.; Davidson, S.M.; Biancur, D.; Guzelhan, B.S.; Alvarez, S.W.; Wu, W.L.; Karakousi, T.R.; Zavitsanou, A.M.; et al. Activation of the NRF2 antioxidant program generates an imbalance in central carbon metabolism in cancer. Elife 2017, 6. [Google Scholar] [CrossRef]
- Dittmer, J. The role of the transcription factor Ets1 in carcinoma. Semin Cancer Biol 2015, 35, 20–38. [Google Scholar] [CrossRef] [PubMed]
- Prasad, P.; Roy, S.S. Glutamine regulates ovarian cancer cell migration and invasion through ETS1. Heliyon 2021, 7, e07064. [Google Scholar] [CrossRef] [PubMed]
- Beier, A.K.; Ebersbach, C.; Siciliano, T.; Scholze, J.; Hofmann, J.; Hönscheid, P.; Baretton, G.B.; Woods, K.; Guezguez, B.; Dubrovska, A.; et al. Targeting the glutamine metabolism to suppress cell proliferation in mesenchymal docetaxel-resistant prostate cancer. Oncogene 2024, 43, 2038–2050. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, D.; Pakhira, S.; Ghosh, D.D.; Roychoudhury, S.; Roy, S.S. Ets1 facilitates EMT/invasion through Drp1-mediated mitochondrial fragmentation in ovarian cancer. iScience 2023, 26, 107537. [Google Scholar] [CrossRef]
- Colucci, M.; Sarill, M.; Maddalena, M.; Valdata, A.; Troiani, M.; Massarotti, M.; Bolis, M.; Bressan, S.; Kohl, A.; Robesti, D.; et al. Senescence in cancer. Cancer Cell 2025, 43, 1204–1226. [Google Scholar] [CrossRef]
- Yang, L.; Moss, T.; Mangala, L.S.; Marini, J.; Zhao, H.; Wahlig, S.; Armaiz-Pena, G.; Jiang, D.; Achreja, A.; Win, J.; et al. Metabolic shifts toward glutamine regulate tumor growth, invasion and bioenergetics in ovarian cancer. Mol Syst Biol 2014, 10, 728. [Google Scholar] [CrossRef]
- Zou, W.; Han, Z.; Wang, Z.; Liu, Q. Targeting glutamine metabolism as a potential target for cancer treatment. J Exp Clin Cancer Res 2025, 44, 180. [Google Scholar] [CrossRef]
- Yerbes, R.; Mora-Molina, R.; Fernández-Farrán, F.J.; Hiraldo, L.; López-Rivas, A.; Palacios, C. Limiting glutamine utilization activates a GCN2/TRAIL-R2/Caspase-8 apoptotic pathway in glutamine-addicted tumor cells. Cell Death Dis 2022, 13, 906. [Google Scholar] [CrossRef]
- Fang, L.; Gao, D.; Jiang, Z.; Li, G.; Li, M. Glutamine's double-edged sword: fueling tumor growth and offering therapeutic hope. Front Immunol 2025, 16, 1578940. [Google Scholar] [CrossRef]
- Chen, T.; Xu, Y.; Yang, F.; Pan, Y.; Ji, N.; Li, J.; Zeng, X.; Chen, Q.; Jiang, L.; Shen, Y.Q. Crosstalk of glutamine metabolism between cancer-associated fibroblasts and cancer cells. Cell Signal 2025, 133, 111874. [Google Scholar] [CrossRef]
- Mestre-Farrera, A.; Bruch-Oms, M.; Peña, R.; Rodríguez-Morató, J.; Alba-Castellón, L.; Comerma, L.; Quintela-Fandino, M.; Duñach, M.; Baulida, J.; Pozo, Ó.; et al. Glutamine-Directed Migration of Cancer-Activated Fibroblasts Facilitates Epithelial Tumor Invasion. Cancer Res 2021, 81, 438–451. [Google Scholar] [CrossRef]
- Wilson, L.A.; Gemin, A.; Espiritu, R.; Singh, G. ets-1 is transcriptionally up-regulated by H2O2 via an antioxidant response element. FASEB J 2005, 19, 2085–2087. [Google Scholar] [CrossRef]
- Wang, Y.; Huang, Z.; Sun, M.; Huang, W.; Xia, L. ETS transcription factors: Multifaceted players from cancer progression to tumor immunity. Biochim Biophys Acta Rev Cancer 2023, 1878, 188872. [Google Scholar] [CrossRef]
- Ichikawa, M.K.; Endo, K.; Itoh, Y.; Osada, A.H.; Kimura, Y.; Ueki, K.; Yoshizawa, K.; Miyazawa, K.; Saitoh, M. Ets family proteins regulate the EMT transcription factors Snail and ZEB in cancer cells. FEBS Open Bio 2022, 12, 1353–1364. [Google Scholar] [CrossRef]
- Wang, H.; Li, X.; Hu, H.; Gao, Z. Glutaminase 1 inhibitors: Therapeutic potential, mechanisms of action, and clinical perspectives. Biochem Pharmacol 2025, 242, 117275. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



