Submitted:
02 March 2026
Posted:
03 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results and Discussion
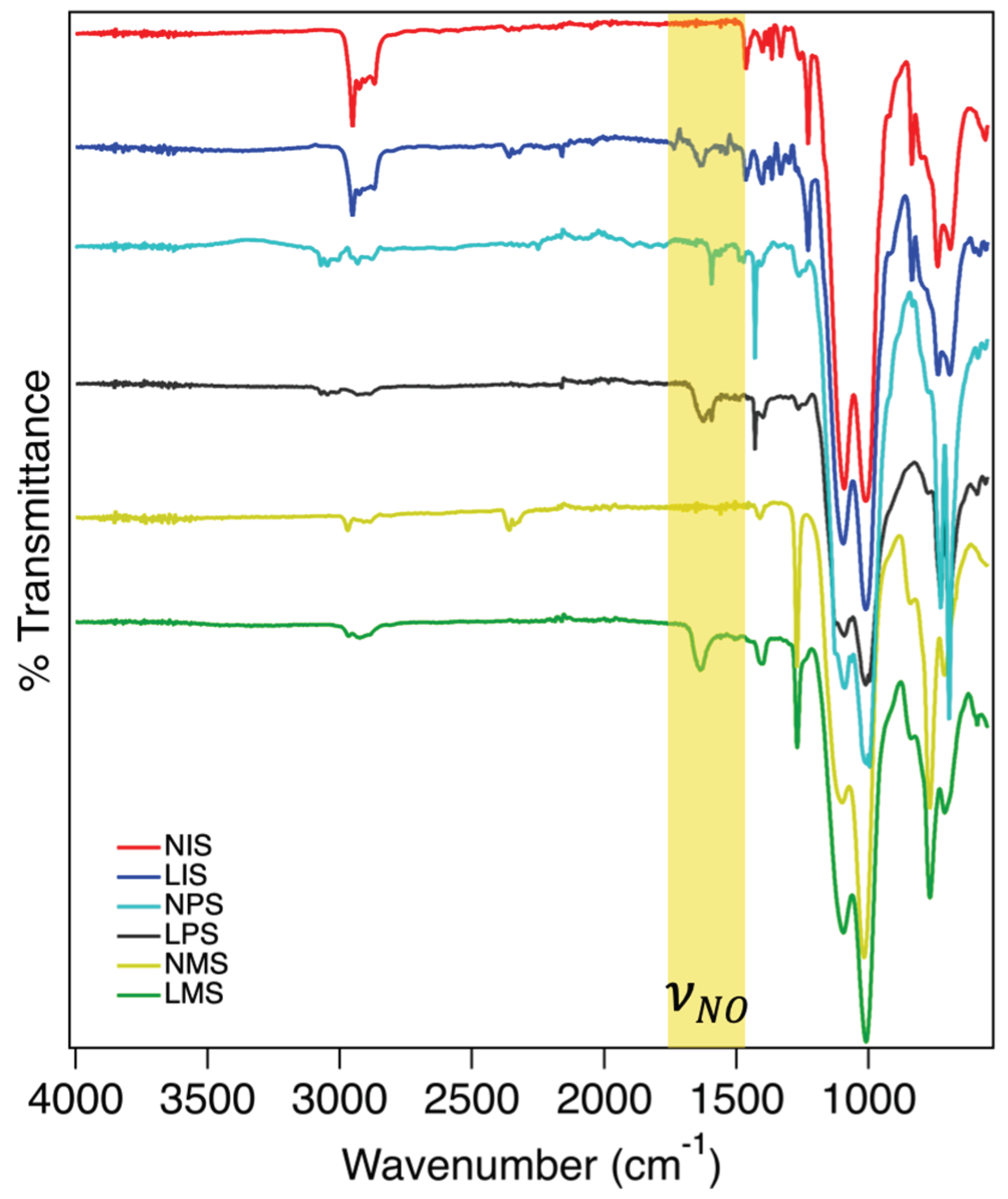
2.1. FTIR Spectral Analysis Confirming Chemical Modifications
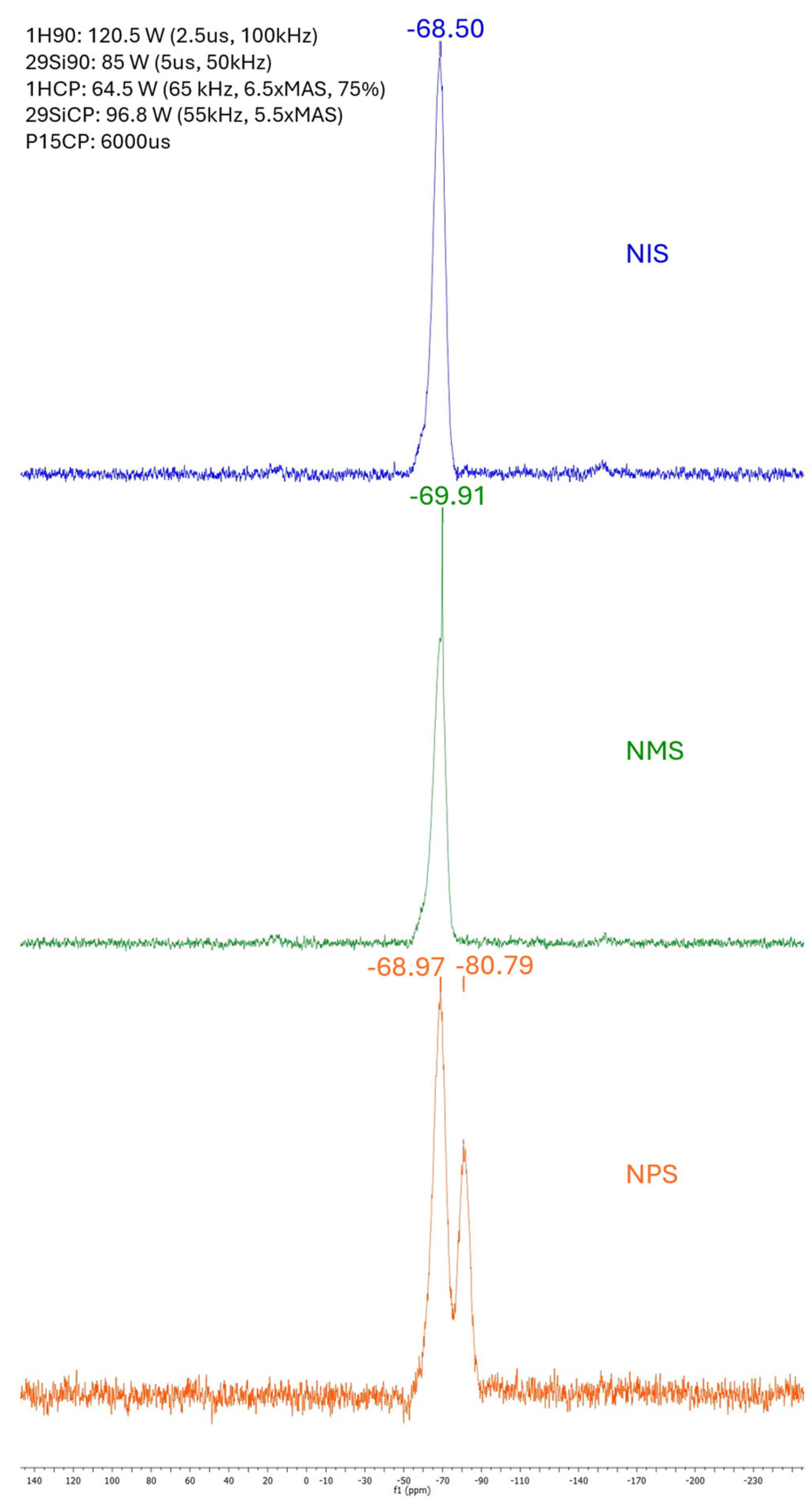
2.2. Solid State MAS 29Si NMR of Organogel Networks
2.3. Characterization of the Thermal Behavior in Modified Elastomeric Samples
2.4. Characterization of Gel Structure via SEM, EDX, and SSA
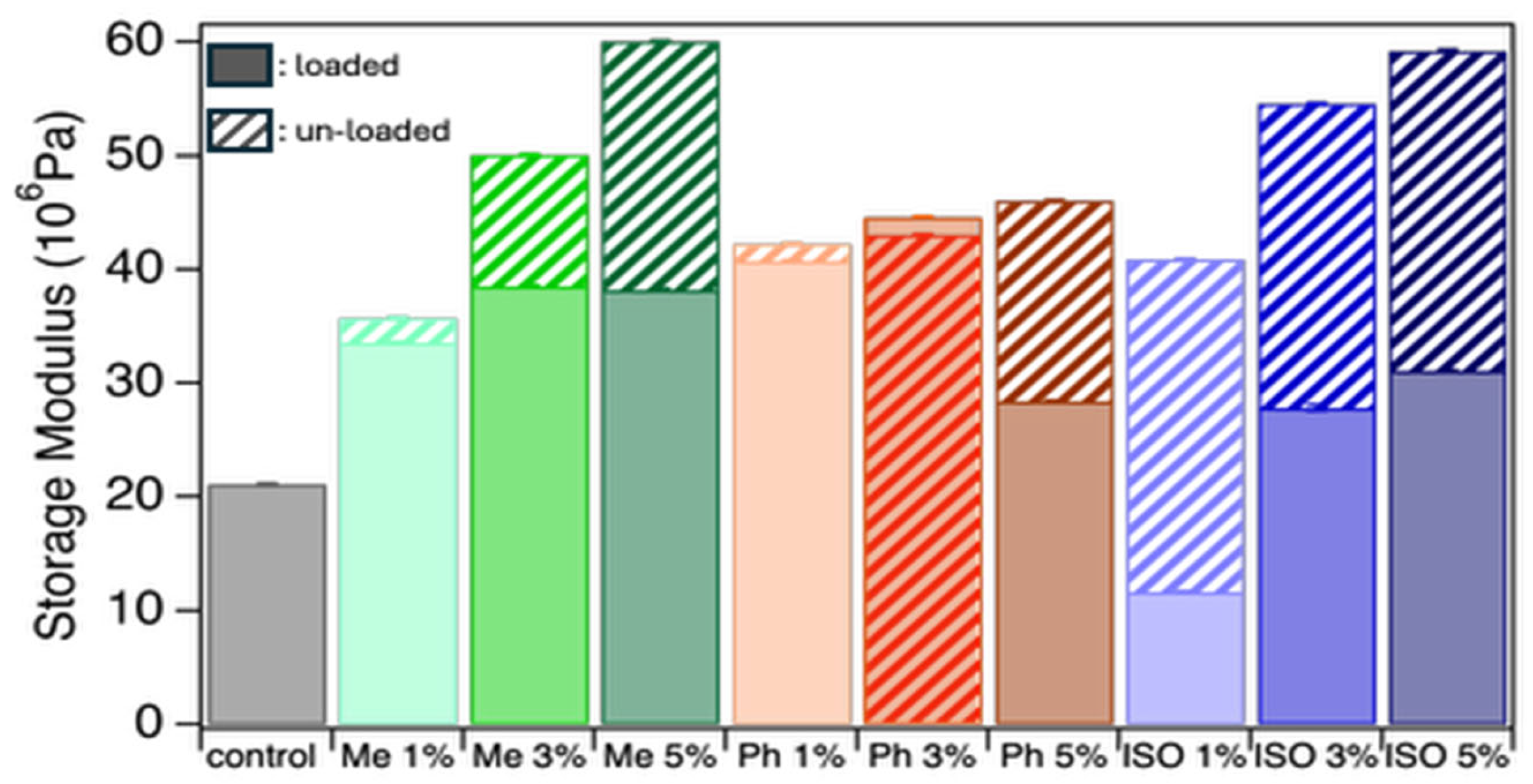
2.5. Dynamic Mechanical Analysis
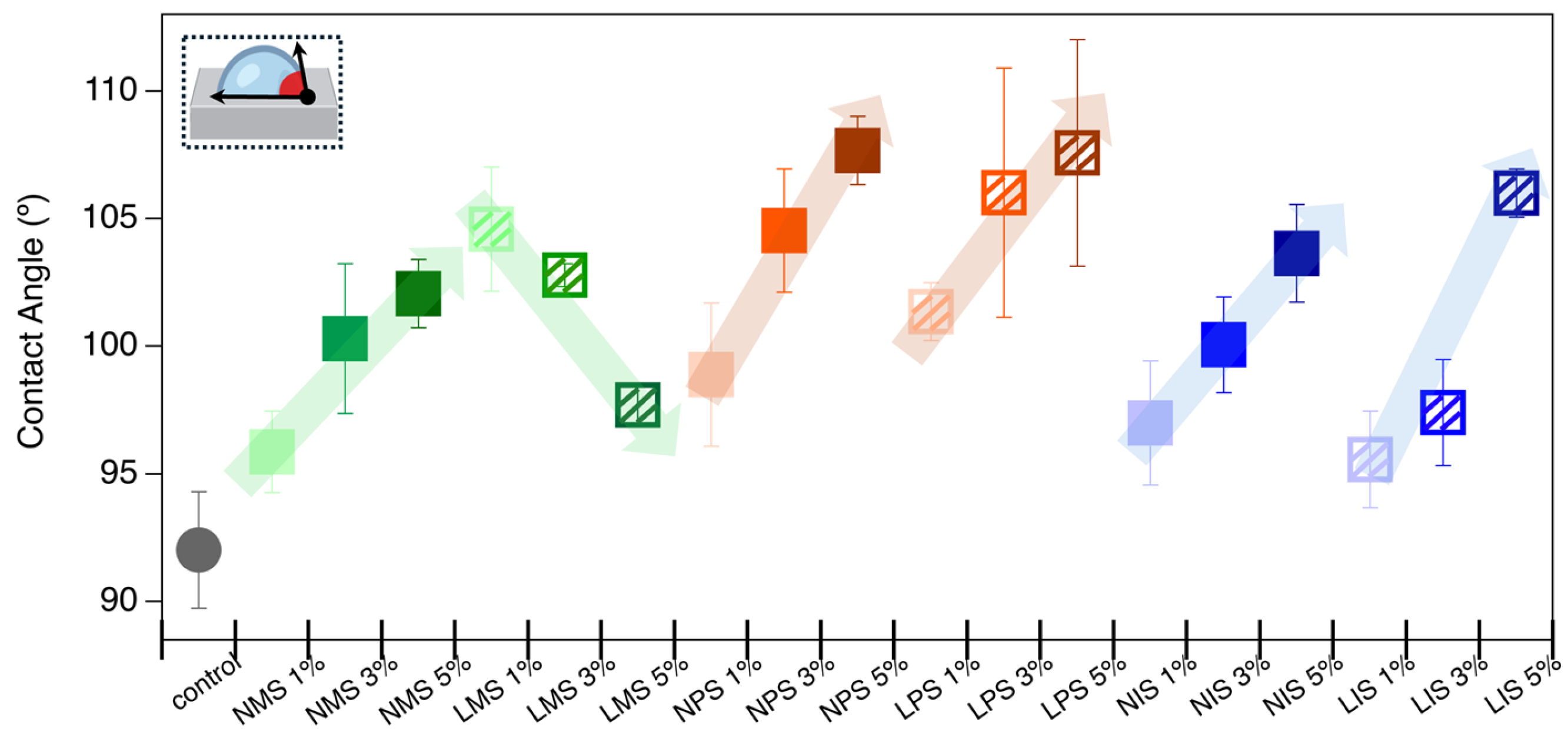
2.6. Hydrophobicity/Hydrophilicity Evaluation
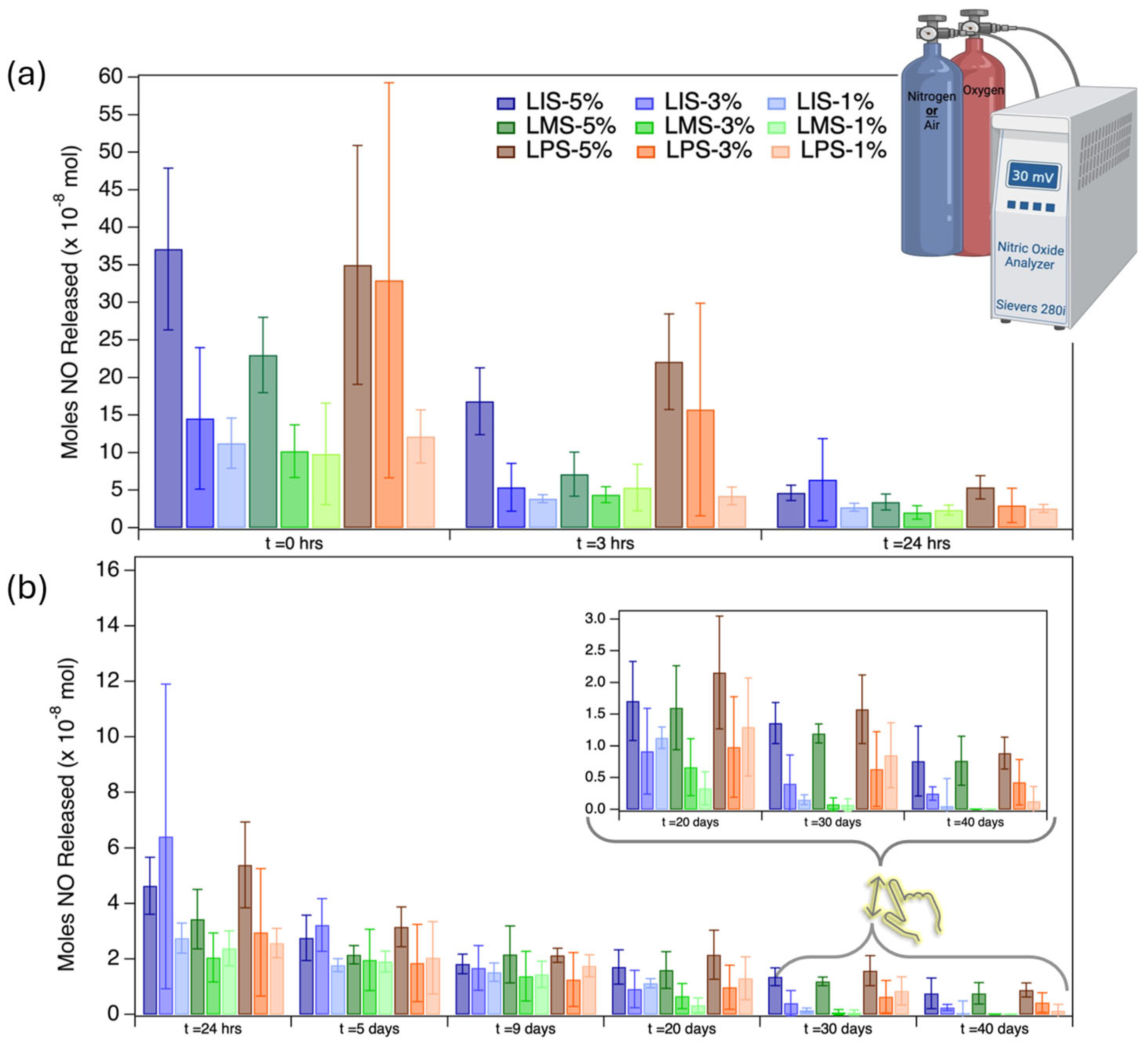
2.7. Nitric Oxide Release: Chemiluminescence Testing
4. Materials and Methods
4.1. Materials
4.2. Synthesis Methods
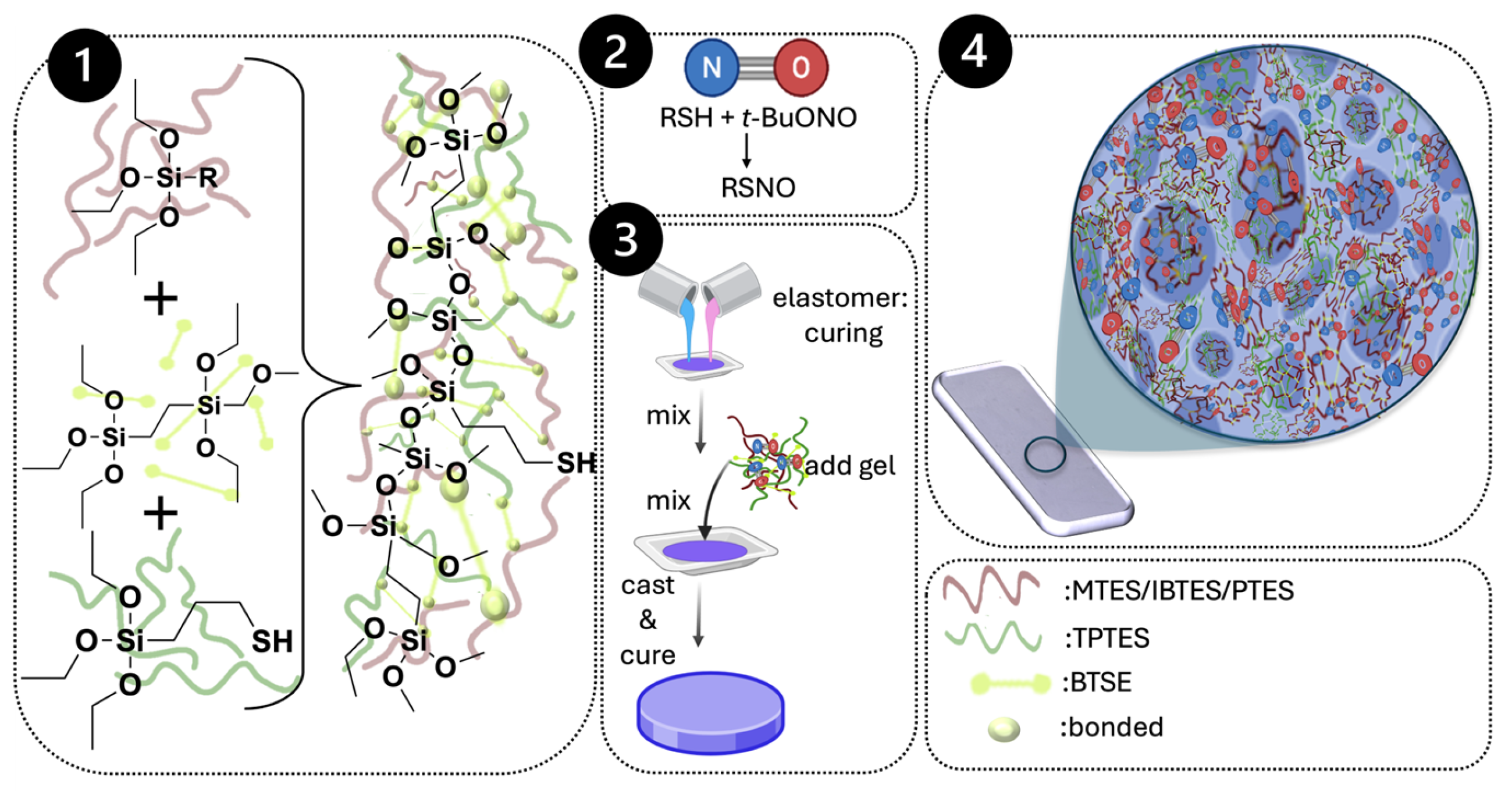
4.2.1. Silsesquioxane Hybrid Organogel Synthesis
4.2.2. NO Loading and Elastomer Impregnation
4.3. Characterization Methods
4.3.1. Fourier-transform Infrared Spectroscopy Analysis (FTIR)
4.3.2. Thermal Gravimetric Analysis (TGA)
4.3.3. Specific Surface Area (SSA) and Porosity Analysis
4.3.4. Solid State MAS 29Si NMR
4.3.5. Scanning Electron Microscopy Analysis (SEM) with Energy Dispersive X-Ray Analysis (EDX)
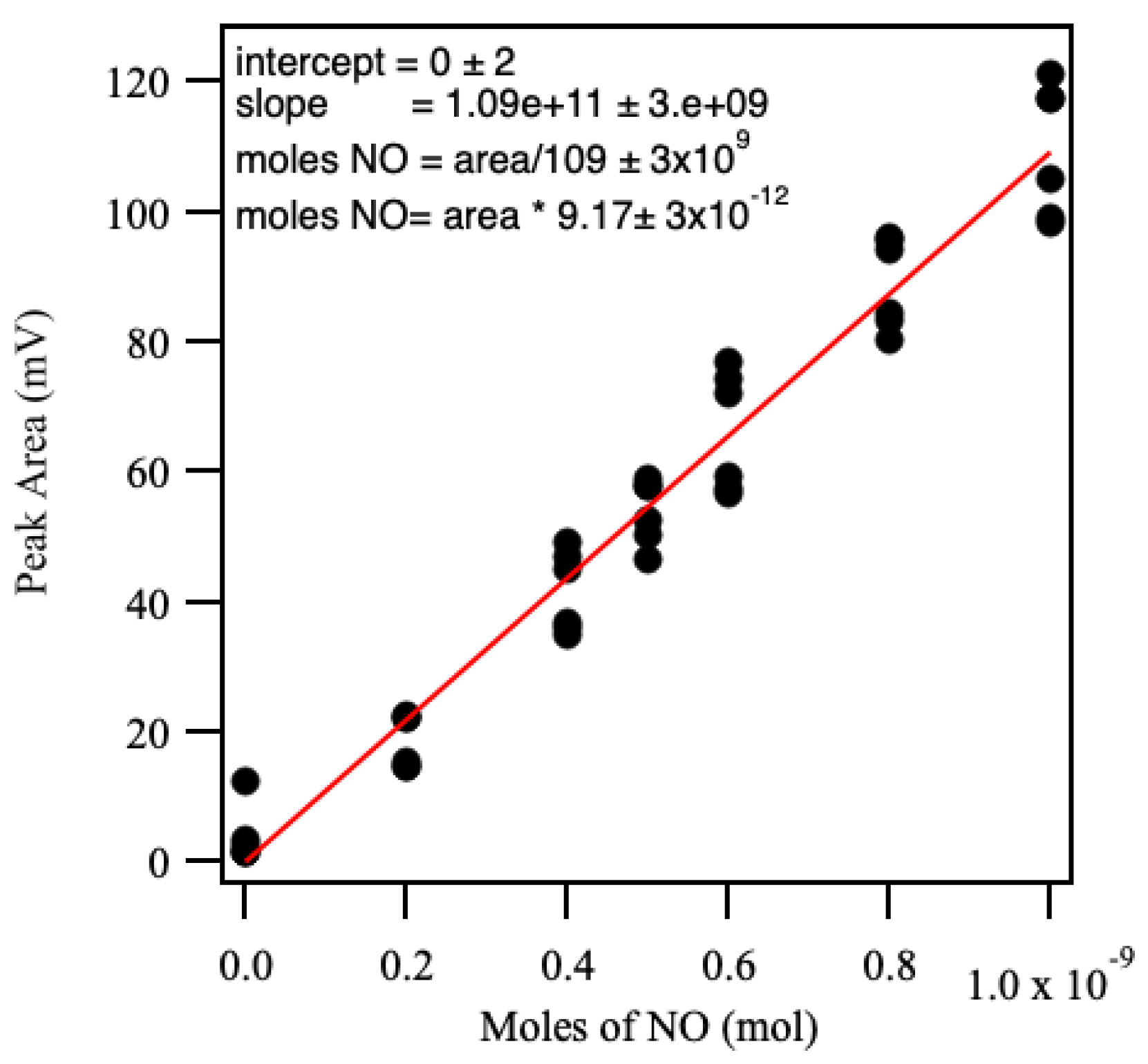
4.3.6. Nitric Oxide Analysis (NOA)-Calibration
4.3.7. Nitric Oxide Analysis (NOA)-Sample Analysis
4.3.8. Dynamic Mechanical Analysis (DMA)
4.3.9. Water Contact Angle Measurement
5. Conclusions
6. Patents
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Haycox, C. L.; Ratner, B. D. In vitro platelet interactions in whole human blood exposed to biomaterial surfaces: Insights on blood compatibility. Journal of Biomedical Materials Research 1993, 27, 1181–1193. [Google Scholar] [CrossRef]
- GORBET, M. B. Biomaterial-associated thrombosis: roles of coagulation factors, complement, platelets and leukocytes. Biomaterials 2004, 25, 5681–5703. [Google Scholar] [CrossRef]
- CDC Guideline for Prevention of Catheter-associated Urinary Tract Infections 2009. Available online: http://www.cdc.gov/hicpac/pdf/CAUTI/CAUTIguideline2009final.pdf.
- Fux, C. A.; Costerton, J. W.; Stewart, P. S.; Stoodley, P. Survival strategies of infectious biofilms. Trends in microbiology (Regular ed.) 2005, 13, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Halpenny, G. M.; Mascharak, P. K. Emerging Antimicrobial Applications of Nitric Oxide (NO) and NO-Releasing Materials. Anti-infective agents in medicinal chemistry 2010, 9, 187–197. [Google Scholar] [CrossRef]
- Goudie, M. J.; Brisbois, E. J.; Pant, J.; Thompson, A.; Potkay, J. A.; Handa, H. Characterization of an S-nitroso-N-acetylpenicillamine-based nitric oxide releasing polymer from a translational perspective. International journal of polymeric materials 2016, 65, 769–778. [Google Scholar] [CrossRef]
- Hopkins, S. P.; Frost, M. C. Synthesis and Characterization of Controlled Nitric Oxide Release from S-Nitroso-N-Acetyl-d-Penicillamine Covalently Linked to Polyvinyl Chloride (SNAP-PVC). Bioengineering 2018, 5. [Google Scholar] [CrossRef]
- Wo, Y.; Li, Z.; Brisbois, E. J.; Colletta, A.; Wu, J.; Major, T. C.; Xi, C.; Bartlett, R. H.; Matzger, A. J.; Meyerhoff, M. E. Origin of Long-Term Storage Stability and Nitric Oxide Release Behavior of CarboSil Polymer Doped with S-Nitroso-N-acetyl-d-penicillamine. ACS Appl. Mater. Interfaces 2015, 7, 22218. [Google Scholar] [CrossRef]
- Carpenter, A. W.; Johnson, J. A.; Schoenfisch, M. H. Nitric oxide-releasing silica nanoparticles with varied surface hydrophobicity. Colloids and surfaces. A, Physicochemical and engineering aspects 2014, 454, 144–151. [Google Scholar] [CrossRef]
- Riccio, D. A.; Schoenfisch, M. H. Nitric oxide release: Part I. Macromolecular scaffolds. Chemical Society reviews 2012, 41, 3731–3741. [Google Scholar] [CrossRef]
- Durão, J.; Vale, N.; Gomes, M. S.; Gomes, P.; Barrias, C. C.; Gales, L. Nitric oxide release from antimicrobial peptide hydrogels for wound healing. Biomolecules (Basel, Switzerland) 2019, 9, 4. [Google Scholar]
- Cheng, J.; He, K.; Shen, Z.; Zhang, G.; Yu, Y.; Hu, J. Nitric Oxide (NO)-Releasing Macromolecules: Rational Design and Biomedical Applications. Frontiers in chemistry 2019, 7, 530. [Google Scholar]
- Zhang, W.; Liu, H.; Qiu, X.; Zuo, F.; Wang, B. Mesoporous silica nanoparticles as a drug delivery mechanism. Open life sciences 2024, 19, 20220867–43. [Google Scholar]
- Lodha, A.; Lodha, M.; Patel, A.; Chaudhuri, J.; Dalal, J.; Edwards, M.; Douroumis, D. Synthesis of mesoporous silica nanoparticles and drug loading of poorly water soluble drug cyclosporin A. Journal of pharmacy & bioallied science 2012, 4, 92–S94. [Google Scholar]
- Sheet, P. S.; Lautner, G.; Meyerhoff, M. E.; Schwendeman, S. P. Mechanistic analysis of the photolytic decomposition of solid-state S-nitroso-N-acetylpenicillamine. Nitric oxide 2024, 142, 38–46. [Google Scholar] [CrossRef]
- Danijel, P.; Nikolina, V.; Ivan, C.; Zeljko, B. J. Effect of nitric oxide donors S-nitroso-N-acetyl-DL-penicillamine, Spermine and PAPA NONOate on intracellular pH in cardiomyocytes. Clinical and experimental pharmacology & physiology 2012, 39, 772–778. [Google Scholar]
- Sheet, P. S.; Lautner, G.; Meyerhoff, M. E.; Schwendeman, S. P. Mechanistic analysis of the photolytic decomposition of solid-state S-nitroso-N-acetylpenicillamine. Nitric oxide 2024, 142, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Hetrick, E. M.; Shin, J. H.; Stasko, N. A.; Johnson, C. B.; Wespe, D. A.; Holmuhamedov, E.; Schoenfisch, M. H. Bactericidal Efficacy of Nitric Oxide-Releasing Silica Nanoparticles. ACS Nano 2008, 2, 235–246. [Google Scholar] [CrossRef]
- Kumar, R.; Chug, M. K.; Brisbois, E. J. Long-Term Storage Stability and Nitric Oxide Release Behavior of (N-Acetyl-S-nitrosopenicillaminyl)-S-nitrosopenicillamine-Incorporated Silicone Rubber Coatings. ACS applied materials & interfaces 2022, 14, 30595–30606. [Google Scholar]
- Vieira de Almeida, H.; da Silva, L. C. E.; Piscelli, B. d. A.; dos Santos, B. R. G.; Guizoni, D. M.; Davel, A. P.; Cormanich, R. A.; Ganzarolli de Oliveira, M. Nitric oxide-releasing PHEMA/polysilsesquioxane photocrosslinked hybrids. RSC advances 2025, 15, 4548–456. [Google Scholar]
- Zhang, W.; Niu, Y.; Zhang, W.; Yang, R. Facile synthesis of polyhedral oligomeric silsesquioxanes with excellent thermosetting, fibrous and crystalline properties. European polymer journal 2024, 221, 113568. [Google Scholar]
- Baney, R. H.; Itoh, M.; Sakakibara, A.; Suzuki, T. Silsesquioxanes. Chemical reviews 1995, 95, 1409–1430. [Google Scholar] [CrossRef]
- Kannan, R. Y.; Salacinski, H. J.; Butler, P. E.; Seifalian, A. M. Polyhedral Oligomeric Silsesquioxane Nanocomposites: The Next Generation Material for Biomedical Applications. Accounts of Chemical Research 2005, 38, 879–884. [Google Scholar] [CrossRef] [PubMed]
- Hu, N.; Lenora, C. U.; May, T. A.; Hershberger, N. C.; Furgal, J. C. In situ formed methyl-co-(bis-R) silsesquioxane based polymer networks with solvent controlled pore size distributions and high surface areas. Mater. Chem. Front. 2020, 4, 851. [Google Scholar]
- Marmo, A. C.; Grunlan, M. A. Biomedical Silicones: Leveraging Additive Strategies to Propel Modern Utility. ACS macro letters 2023, 12, 172–182. [Google Scholar]
- Harris, P. J. F. Sulphur-induced faceting of platinum catalyst particles. Nature 1986, 323, 792–794. [Google Scholar] [CrossRef]
- Yang, T.; Zelikin, A. N.; Chandrawati, R. Progress and Promise of Nitric Oxide-Releasing Platforms. Advanced Science 2018, 5, 1–n/a. [Google Scholar] [CrossRef]
- Zhang, H.; Annich, G. M.; Miskulin, J.; Osterholzer, K.; Merz, S. I.; Bartlett, R. H.; Meyerhoff, M. E. Nitric oxide releasing silicone rubbers with improved blood compatibility: preparation, characterization, and in vivo evaluation. Biomaterials 2002, 23, 1485–1494. [Google Scholar] [CrossRef]
- Hopkins, S.; Frost, M. Synthesis and Characterization of Controlled Nitric Oxide Release from S-Nitroso-N-Acetyl-d-Penicillamine Covalently Linked to Polyvinyl Chloride (SNAP-PVC). Bioengineering (Basel) 2018, 5, 72. [Google Scholar] [CrossRef]
- AnonymousSievers Nitric Oxide Analyzer NOA 280i* Operation and Maintenance Manual. DLM 14291 Rev. A 2006.
- Archer, S. L.; Shultz, P. J.; Warren, J. B.; Hampl, V.; DeMaster, E. G. Preparation of Standards and Measurement of Nitric Oxide, Nitroxyl, and Related Oxidation Products. Methods (San Diego, Calif.) 1995, 7, 21–34. [Google Scholar] [CrossRef]
- Boo, Y. C.; Tressel, S. L.; Jo, H. An improved method to measure nitrate/nitrite with an NO-selective electrochemical sensor. Nitric oxide 2007, 16, 306–312. [Google Scholar] [CrossRef]
- Marmo, A. C.; Grunlan, M. A. Biomedical Silicones: Leveraging Additive Strategies to Propel Modern Utility. ACS macro letters 2023, 12, 172–182. [Google Scholar] [CrossRef]
- Zheng, S.; Liu, Y.; Yang, Y.; Zhu, R.; Yu, X.; Cao, Z. Tough, Antibacterial, and Antithrombogenic Hydrogel Coatings for Blood-Contacting Silicone Medical Devices. ACS applied materials & interfaces 2025, 17, 46583–46595. [Google Scholar]
- Goudie, M. J.; Pant, J.; Handa, H. Liquid-infused nitric oxide-releasing (LINORel) silicone for decreased fouling, thrombosis, and infection of medical devices. Sci Rep 2017, 7, 13623–13. [Google Scholar]
- Dudziec, B.; Żak, P.; Marciniec, B. Synthetic Routes to Silsesquioxane-Based Systems as Photoactive Materials and Their Precursors. Polymers 2019, 11, 504. [Google Scholar] [CrossRef] [PubMed]
- Tretbar, C.; Castro, J.; Yokoyama, K.; Guan, Z. Fluoride-Catalyzed Siloxane Exchange as a Robust Dynamic Chemistry for High-Performance Vitrimers. Advanced materials (Weinheim) 2023, 35, e2303280–n/a. [Google Scholar] [CrossRef] [PubMed]
- Ronchi, M.; Sulaiman, S.; Boston, N. R.; Laine, R. M. Fluoride catalyzed rearrangements of polysilsesquioxanes, mixed Me, vinyl T8, Me, vinyl T10 and T12 cages. Applied Organometallic Chemistry 2010, 24, 551–557. [Google Scholar] [CrossRef]
- Fasce, D. P.; Williams, R. J. J.; Méchin, F.; Pascault, J. P.; Llauro, M. F.; Pétiaud, R. Synthesis and Characterization of Polyhedral Silsesquioxanes Bearing Bulky Functionalized Substituents. Macromolecules 1999, 32, 4757–4763. [Google Scholar] [CrossRef]
- Hu, N.; Furgal, J. C. R-Silsesquioxane-Based Network Polymers by Fluoride Catalyzed Synthesis: An Investigation of Cross-Linker Structure and Its Influence on Porosity. Materials 2020, 13, 1849. [Google Scholar] [CrossRef]
- VanWagner, M.; Rhadigan, J.; Lancina, M.; Lebovsky, A.; Romanowicz, G.; Holmes, H.; Brunette, M. A.; Snyder, K. L.; Bostwick, M.; Lee, B. P.; Frost, M. C.; Rajachar, R. M. S-Nitroso-N-acetylpenicillamine (SNAP) Derivatization of Peptide Primary Amines to Create Inducible Nitric Oxide Donor Biomaterials. ACS applied materials & interfaces 2013, 5, 8430–8439. [Google Scholar]
- Sato, Y.; Hayami, R.; Gunji, T. Characterization of NMR, IR, and Raman spectra for siloxanes and silsesquioxanes: a mini review. J Sol-Gel Sci Technol 2022, 104, 36–52. [Google Scholar] [CrossRef]
- Bivona, L. A.; Fichera, O.; Fusaro, L.; Giacalone, F.; Buaki-Sogo, M.; Gruttadauria, M.; Aprile, C. A polyhedral oligomeric silsesquioxane-based catalyst for the efficient synthesis of cyclic carbonates. Catalysis Science & Technology 2015, 5, 5000–5007. [Google Scholar] [CrossRef]
- Zhu, F.; Zhang, G.; Xu, S.; Hong, X.; Dong, J. A novel alkoxysilane-modified high solids hydroxyl acrylic polyurethane: Preparation and surface properties. Journal of applied polymer science 2006, 101, 1866–1871. [Google Scholar]
- Hu, N.; Furgal, J. C. R-Silsesquioxane-Based Network Polymers by Fluoride Catalyzed Synthesis: An Investigation of Cross-Linker Structure and Its Influence on Porosity. Materials 2020, 13, 1849. [Google Scholar] [CrossRef]
- Lang, N. Theœ influence of matrix properties on cell migration in disordered 3-dimensional biopolymer networks. Available online: http://d-nb.info/1075478790/34.
- Hwang, S. O.; Lee, J. Y.; Lee, J. Effect of the silsesquioxane structure on the mechanical properties of the silsesquioxane-reinforced polymer composite films. Progress in organic coatings 2019, 137, 105316. [Google Scholar] [CrossRef]
- Alasfar, R. H.; Ahzi, S.; Barth, N.; Kochkodan, V.; Khraisheh, M.; Koç, M. A Review on the Modeling of the Elastic Modulus and Yield Stress of Polymers and Polymer Nanocomposites: Effect of Temperature, Loading Rate and Porosity. Polymers 2022, 14, 360. [Google Scholar] [CrossRef]
- Köglmaier, M.; Caspari, A.; Michel, S.; Auernhammer, G. K.; Kunz, W. Impact of Nitric Oxide on the Surface Properties of Selected Polymers. Applied sciences 2025, 15, 2646. [Google Scholar]
- Mondal, A.; Maffe, P.; Wilson, S. N.; Ghalei, S.; Palacio, R.; Handa, H.; Brisbois, E. J. Catalytic effect of transition metal-doped medical grade polymer on S -nitrosothiol decomposition and its biological response. Materials advances 2023, 4, 3197–3206. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, T. Generalization of the sacrificial bond principle for gel and elastomer toughening. Polym J 2017, 49, 477–485. [Google Scholar]
- Tabish, T. A.; Crabtree, M. J.; Townley, H. E.; Winyard, P. G.; Lygate, C. A. Nitric Oxide Releasing Nanomaterials for Cardiovascular Applications. JACC. Basic to translational science 2024, 9, 691–709. [Google Scholar]
- Kushwaha, M.; Anderson, J. M.; Bosworth, C. A.; Andukuri, A.; Minor, W. P.; Lancaster, J. R.; Anderson, P. G.; Brott, B. C.; Jun, H. A nitric oxide releasing, self assembled peptide amphiphile matrix that mimics native endothelium for coating implantable cardiovascular devices. Biomaterials 2010, 31, 1502–1508. [Google Scholar] [CrossRef] [PubMed]
- Wo, Y.; Li, Z.; Brisbois, E. J.; Colletta, A.; Wu, J.; Major, T. C.; Xi, C.; Bartlett, R. H.; Matzger, A. J.; Meyerhoff, M. E. Origin of Long-Term Storage Stability and Nitric Oxide Release Behavior of CarboSil Polymer Doped with S-Nitroso-N-acetyl-d-penicillamine. ACS Applied Materials & Interfaces 2015, 7, 22218–22227. [Google Scholar]
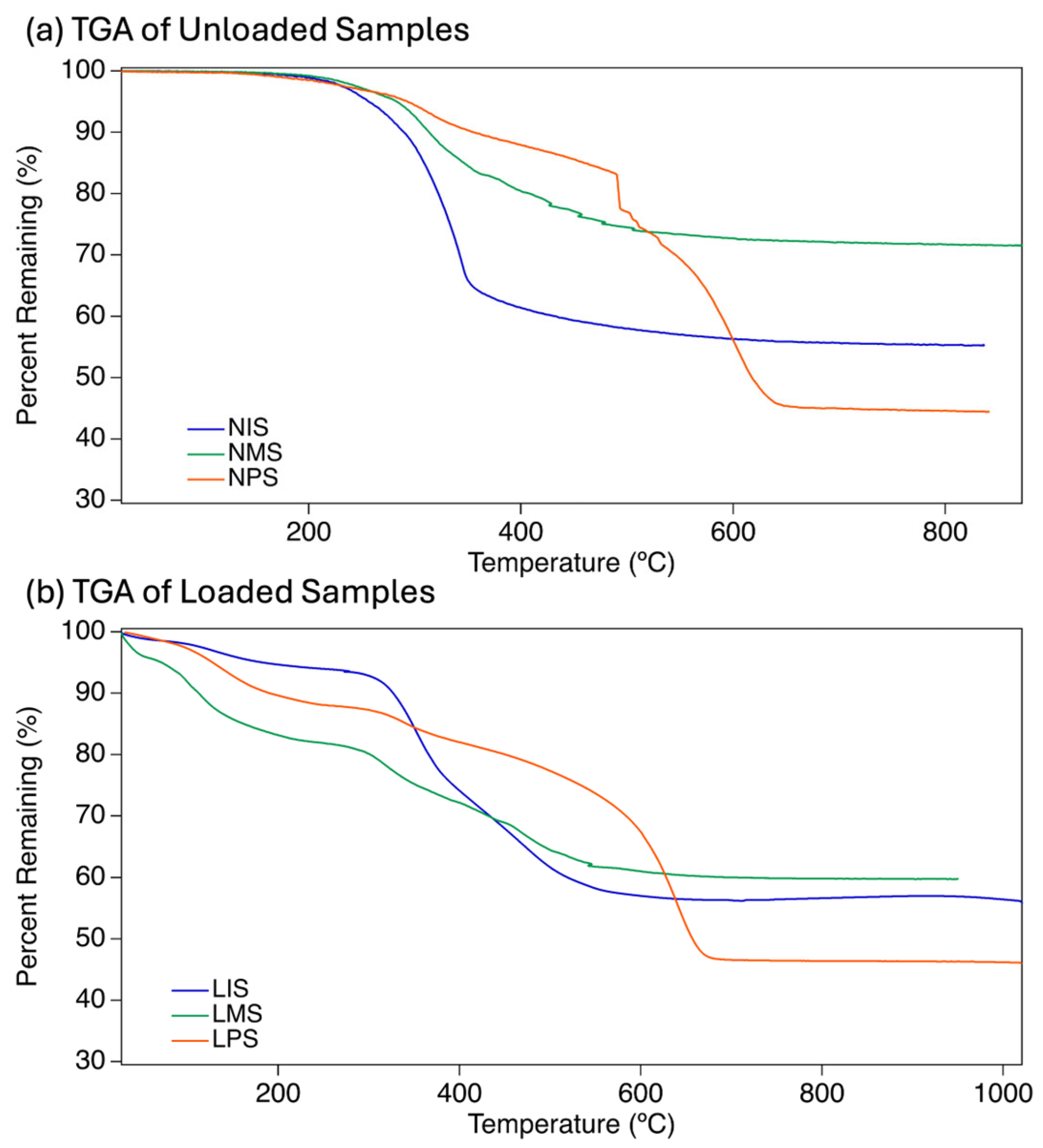

| R-Group | TGA Loading (mg) | Temperature at 5% Mass Loss (T5%) | Residue at 1000 °C (Ceramic Yield) |
| NMS | 8.961 | 298°C | 71.6% |
| LMS | 7.727 | 72°C | 59.8% |
| NPS | 8.378 | 299°C | 44.4% |
| LPS | 7.667 | 126°C | 46.4% |
| NIS | 8.172 | 256°C | 55.3% |
| LIS | 5.276 | 175°C | 56.7% |
| R-Group | Surface Area (m2/g) | Ave. Pore Size (Å) |
| NMS | 160 | 51 |
| NPS | 0.5 | N/A |
| NIS | 559 | 40 |
| R-Group |
RSNO Mass (g) |
R-Group Mass (g) | Bridge Mass (g) | N = non-loaded L = loaded | Abbreviation |
| MTES | 0.245 | 0.890 | 0.361 | N | NMS |
| L | LMS | ||||
| PTES | 0.245 | 1.199 | 0.361 | N | NPS |
| L | LPS | ||||
| IBTES | 0.245 | 1.099 | 0.361 | N | NIS |
| L | LIS | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




