Submitted:
03 March 2026
Posted:
04 March 2026
You are already at the latest version
Abstract
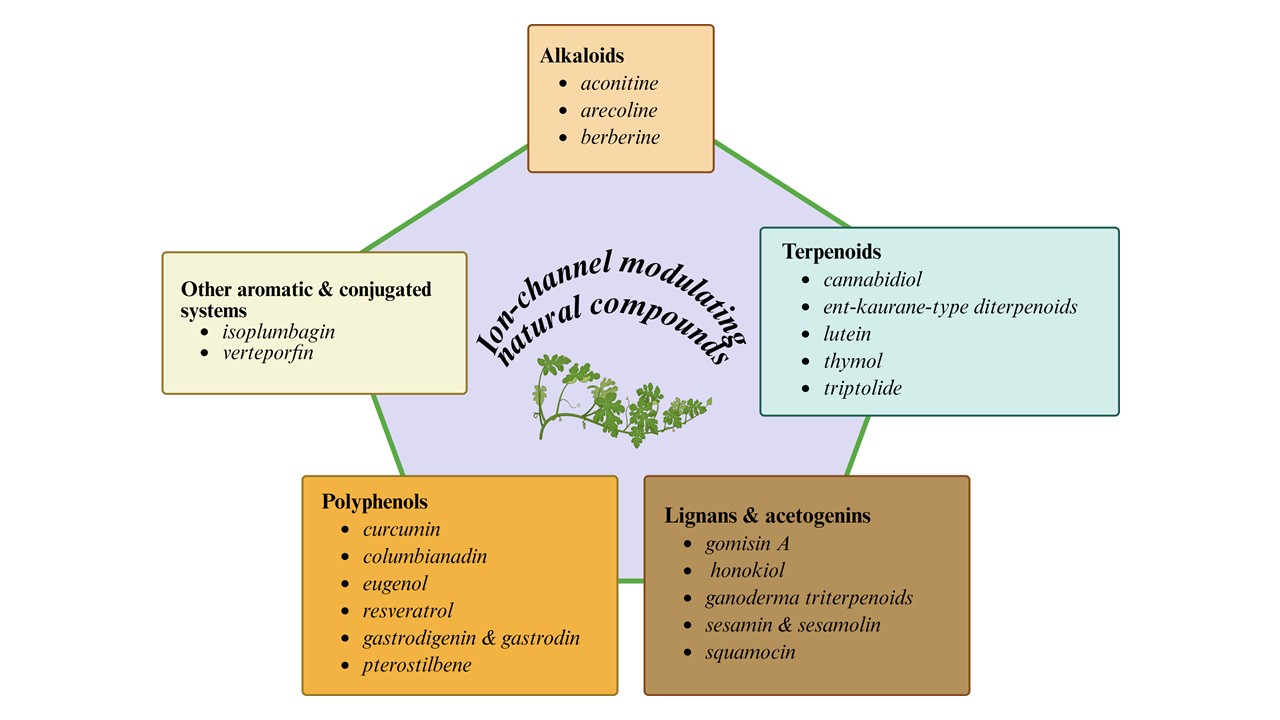
Keywords:
1. Introduction
2. Alkaloids
2.1. Aconitine
2.2. Arecoline
2.3. Berberine
3. Terpenoids
3.1. Cannabidiol
3.2. Ent-Kaurane-Type Diterpenoids
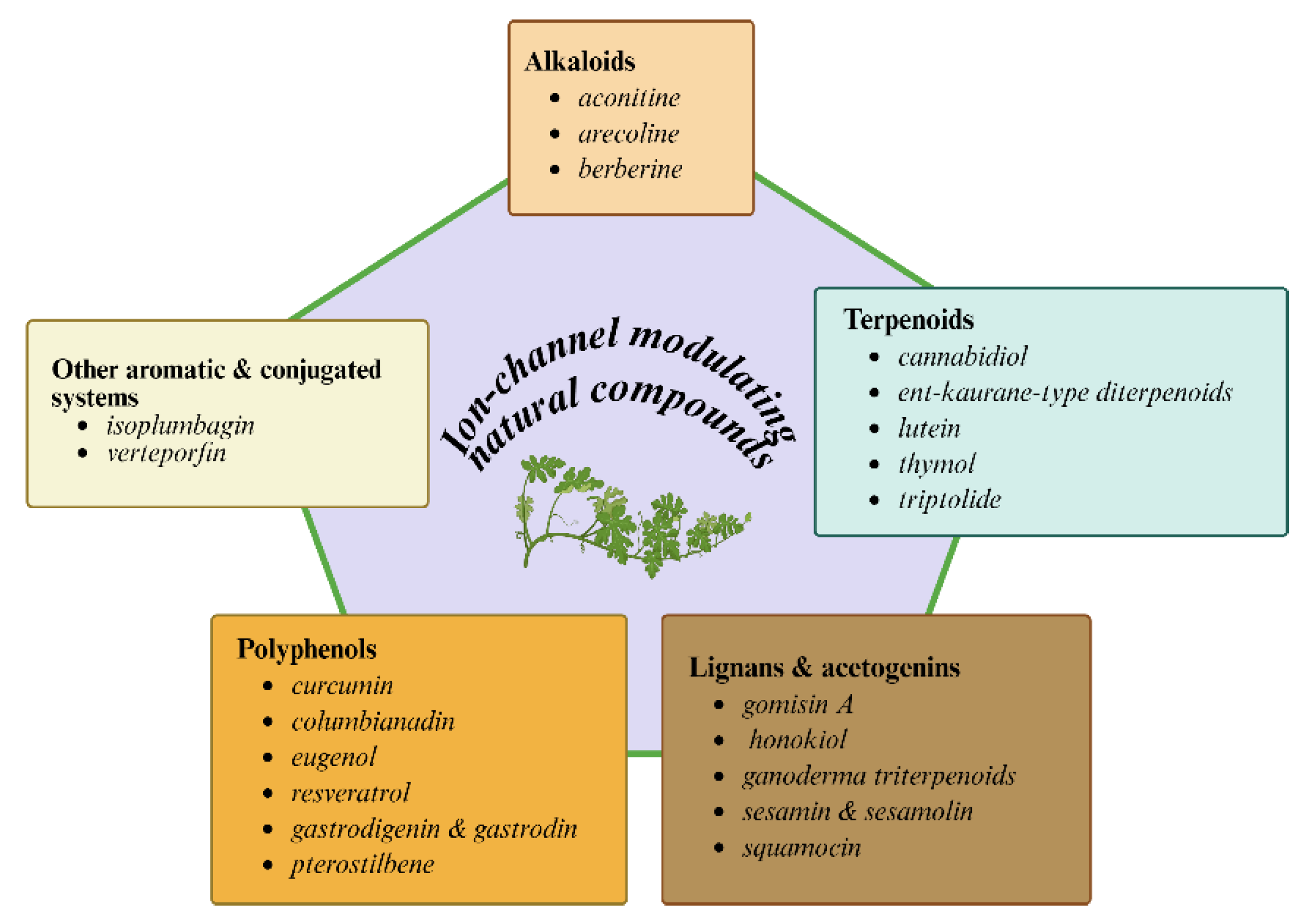
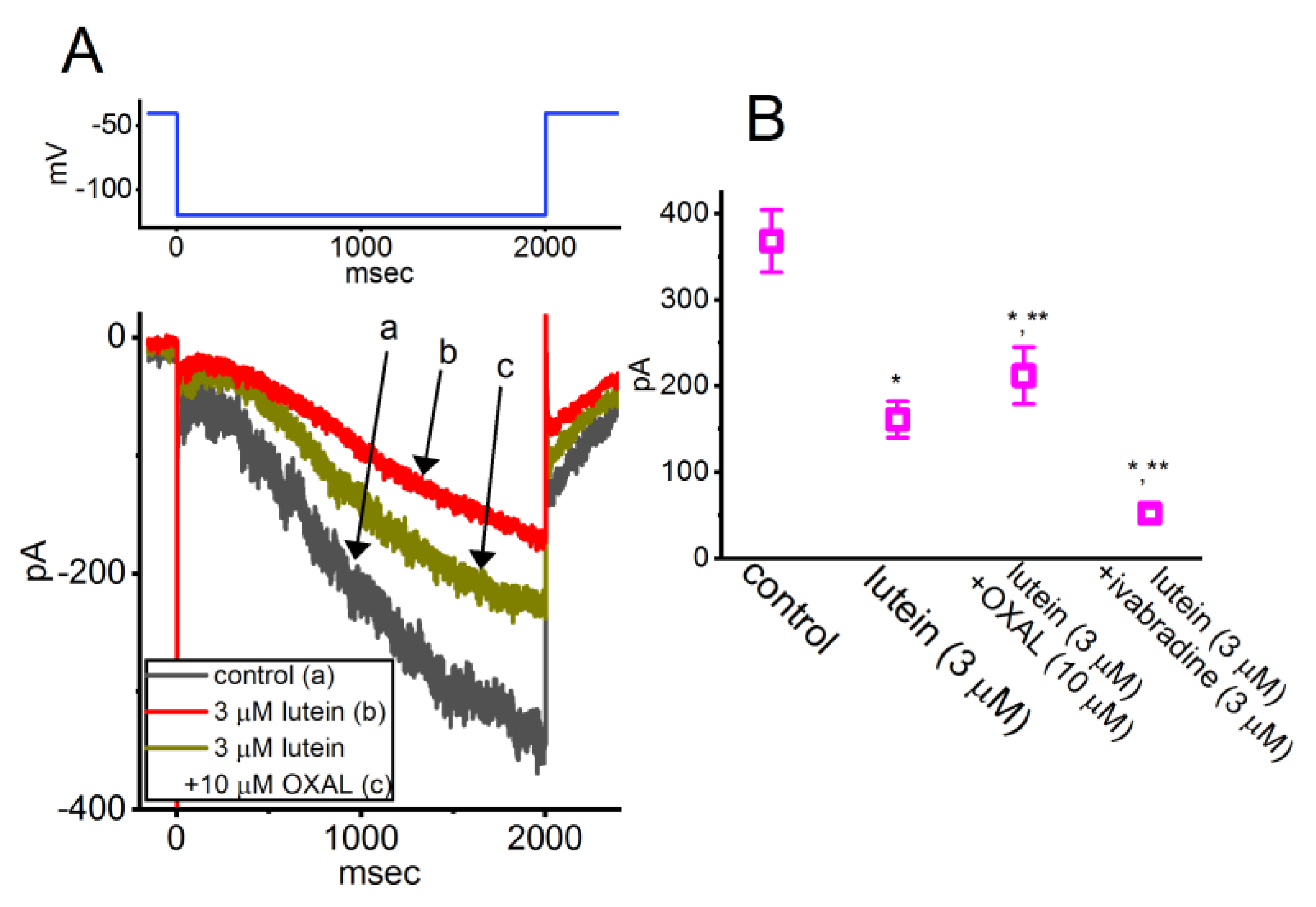
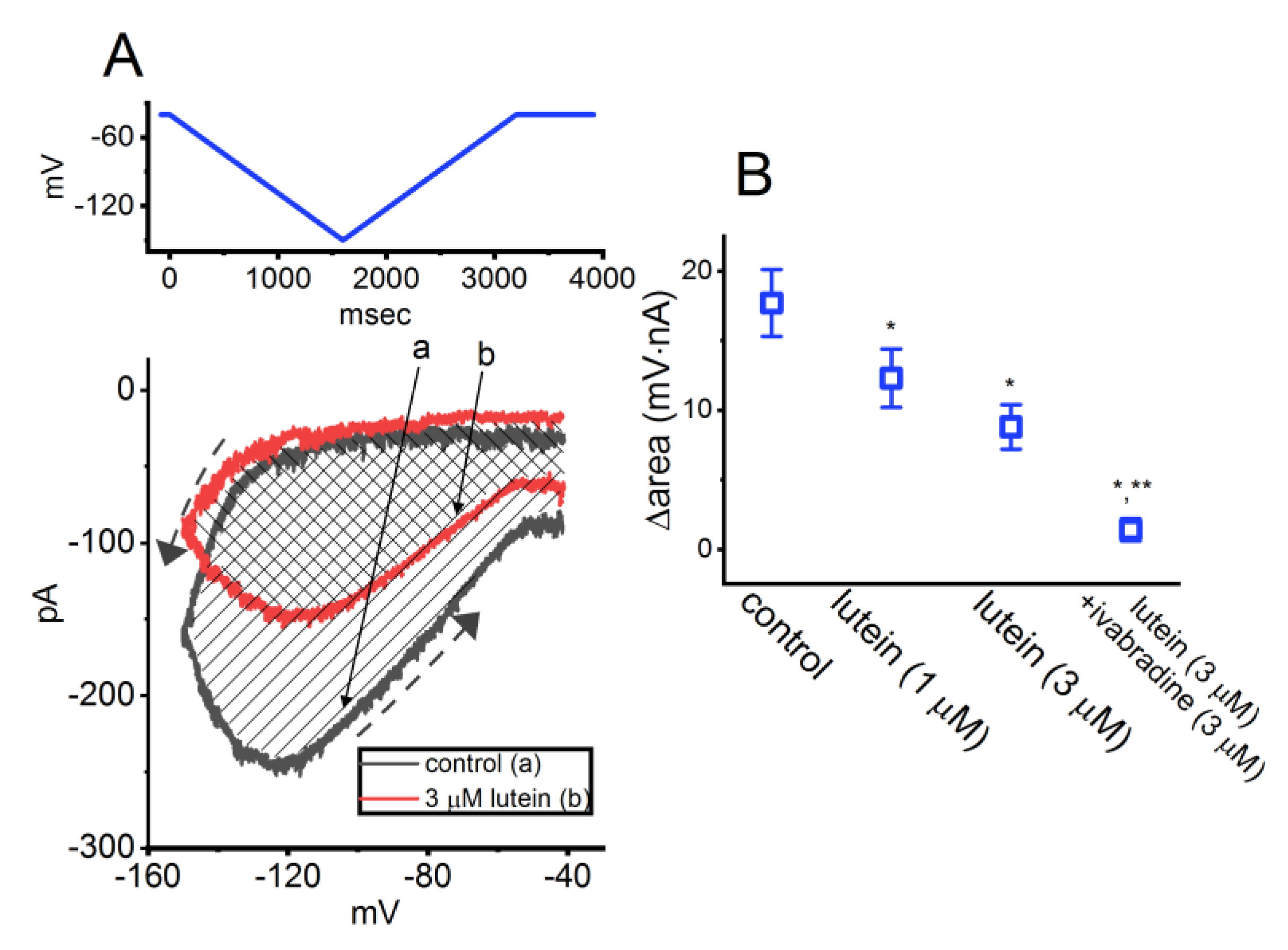
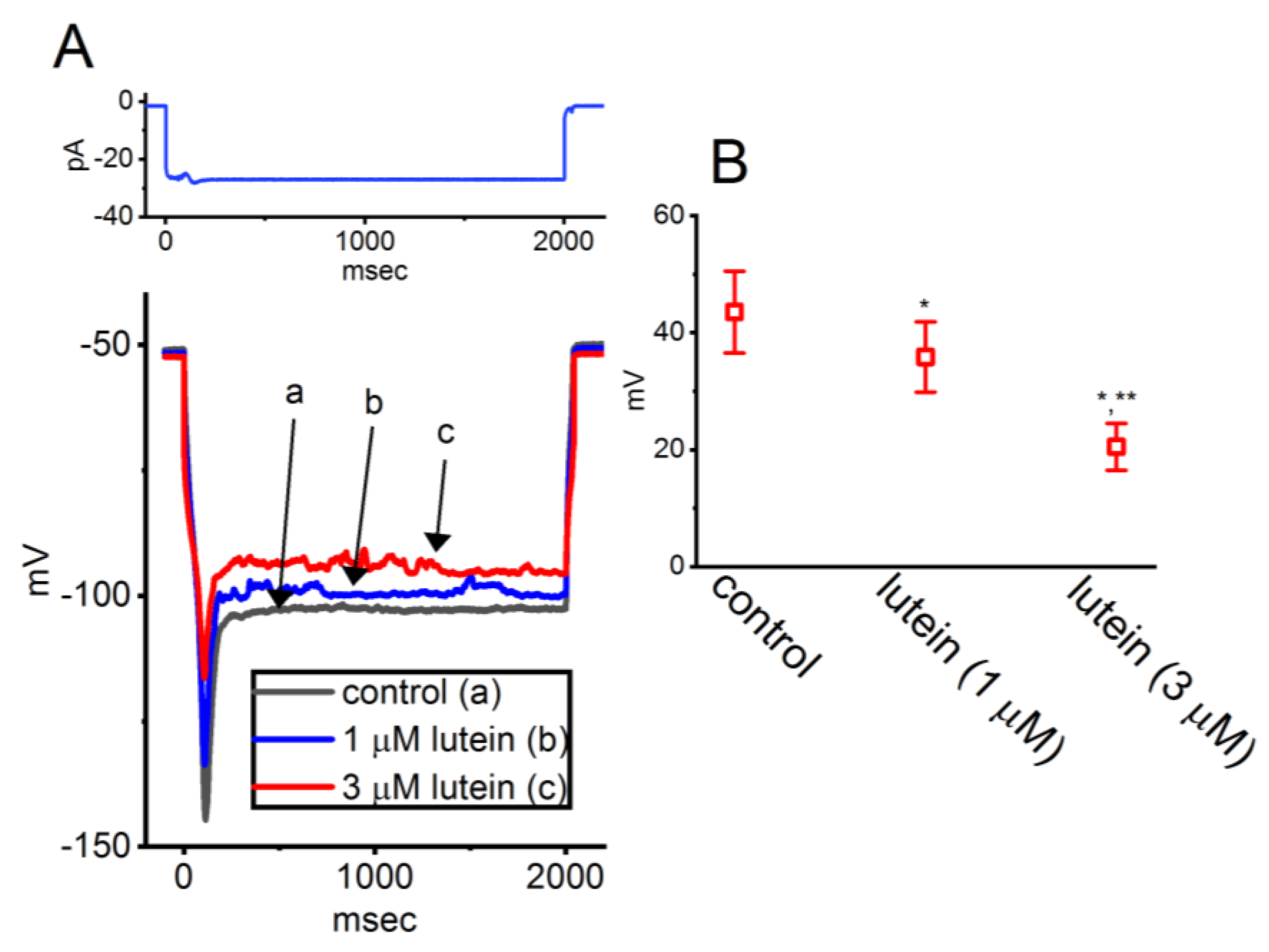
3.3. Lutein
3.4. Thymol
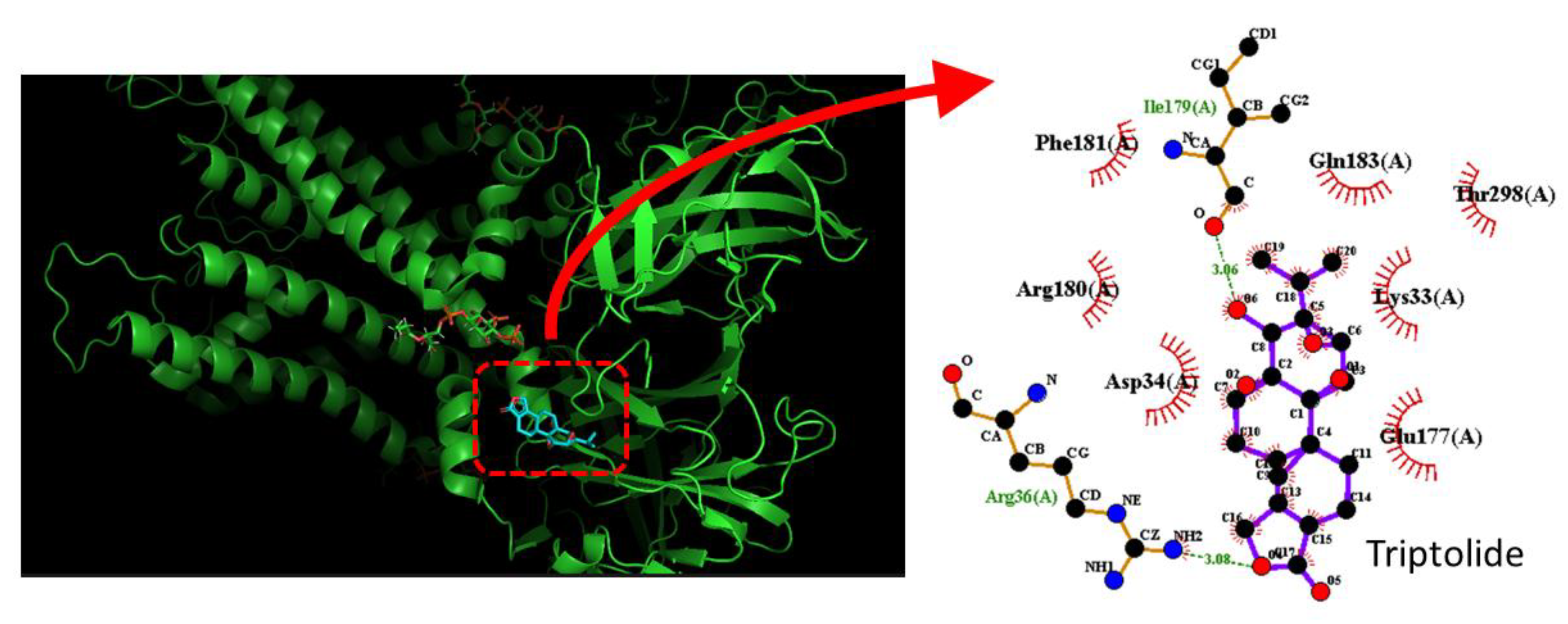
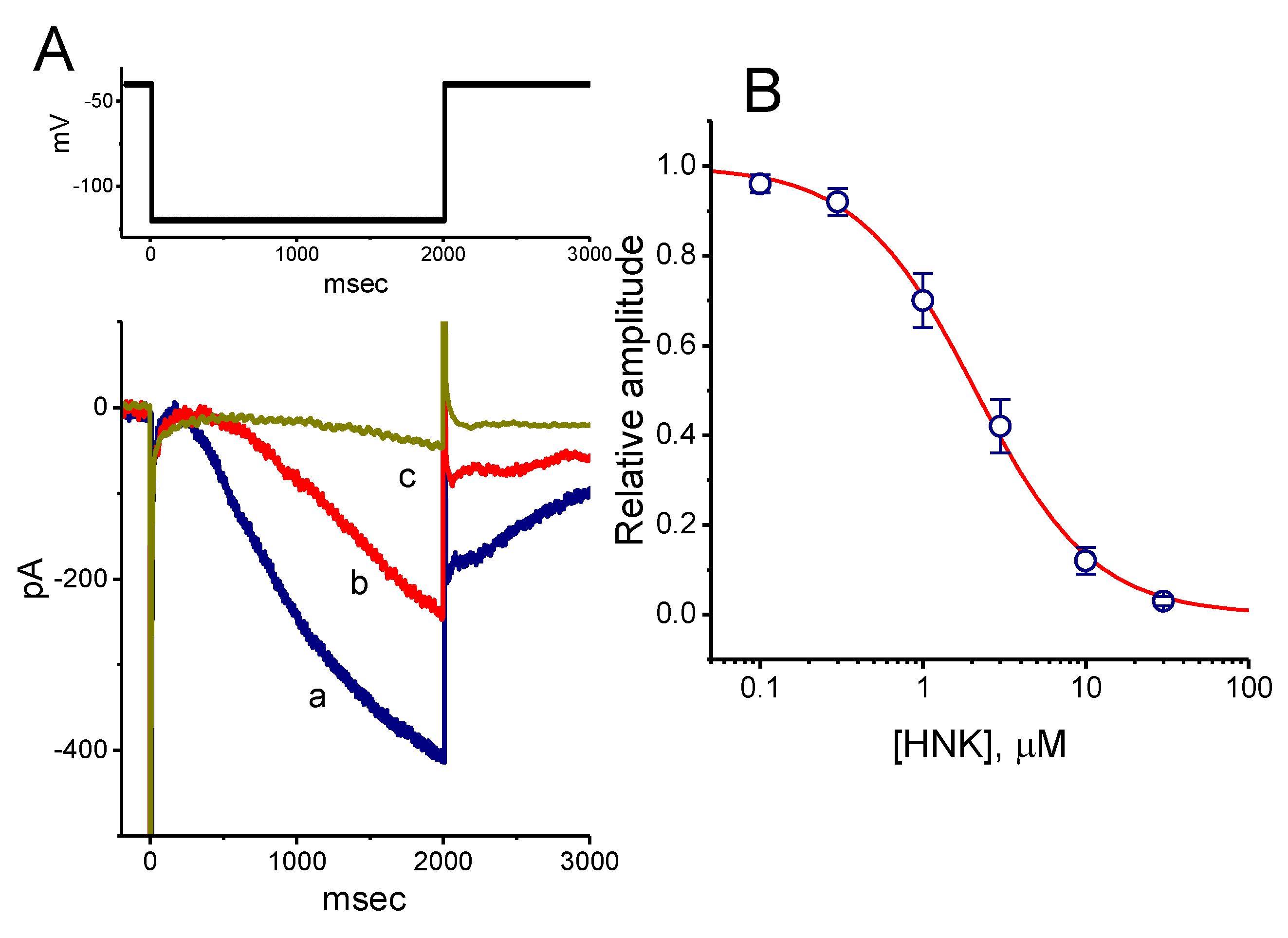
3.5. Triptolide
4. Lignans and Acetogenins
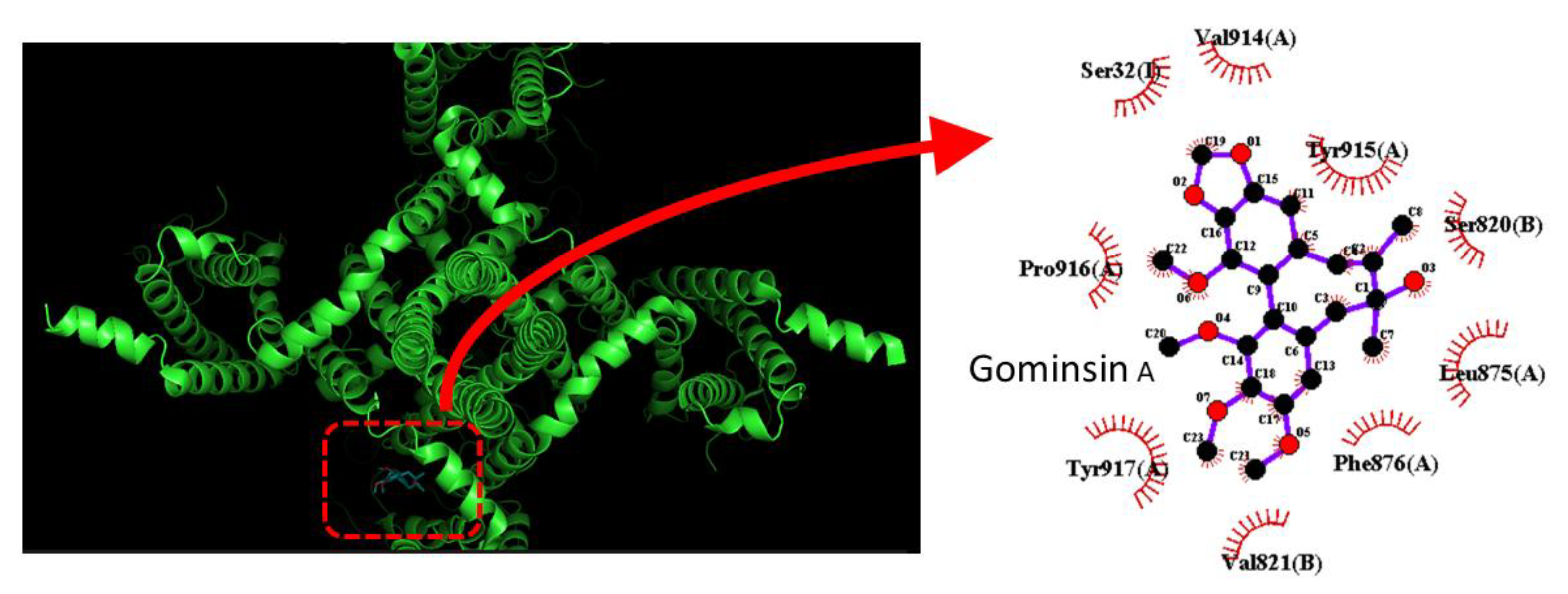
4.1. Gomisin A
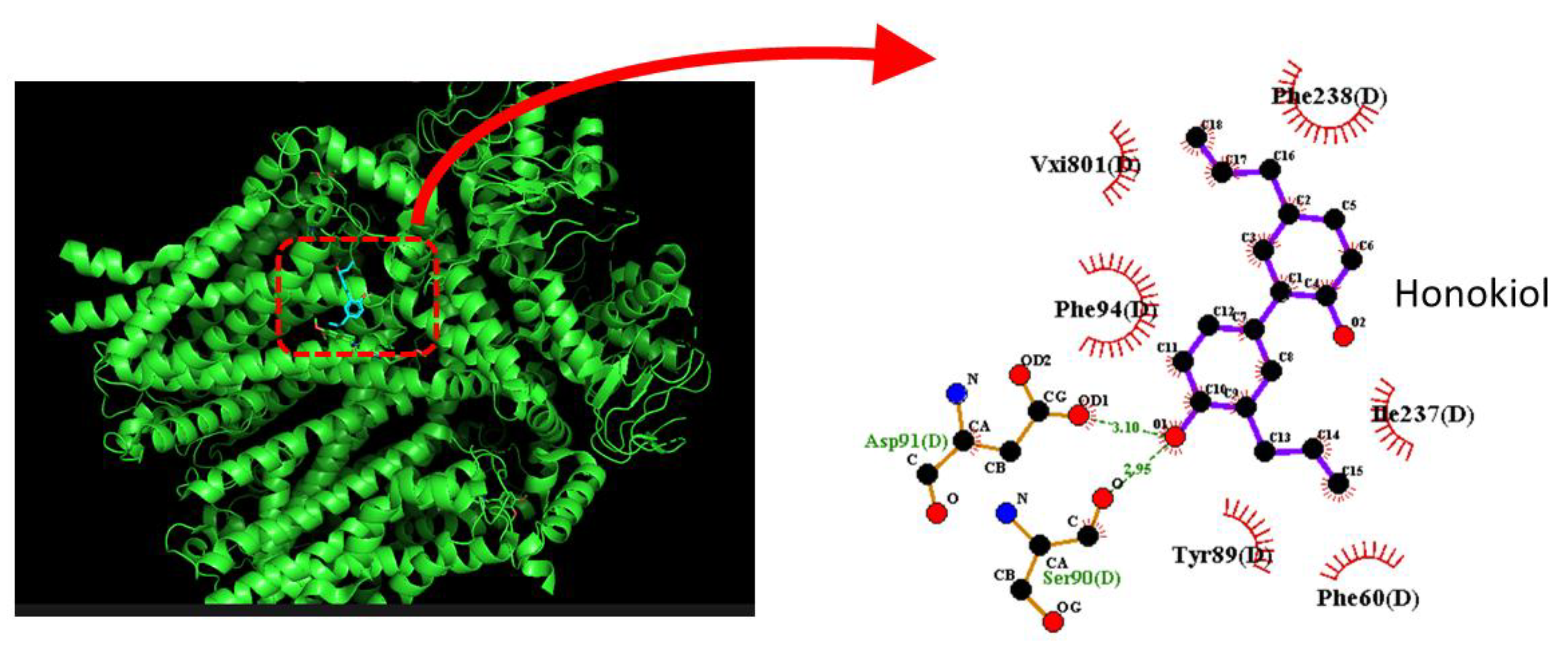
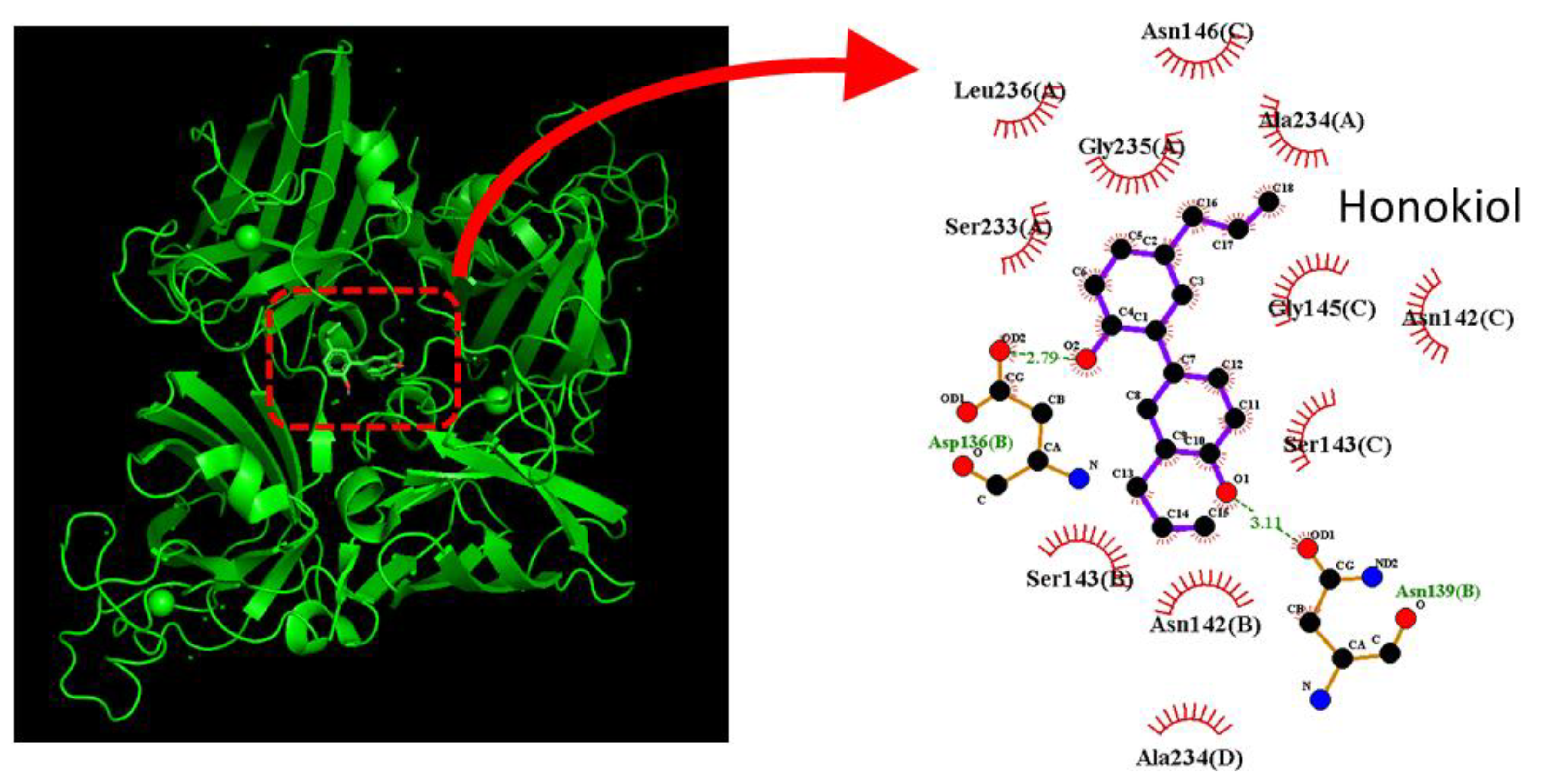
4.2. Honokiol
4.3. Ganoderma Triterpenoids
4.4. Sesamin and Sesamolin
4.5. Squamocin
5. Polyphenols
5.1. Curcumin
5.2. Columbianadin
5.3. Eugenol
5.4. Resveratrol
5.5. Gastrodigenin and Gastrodin
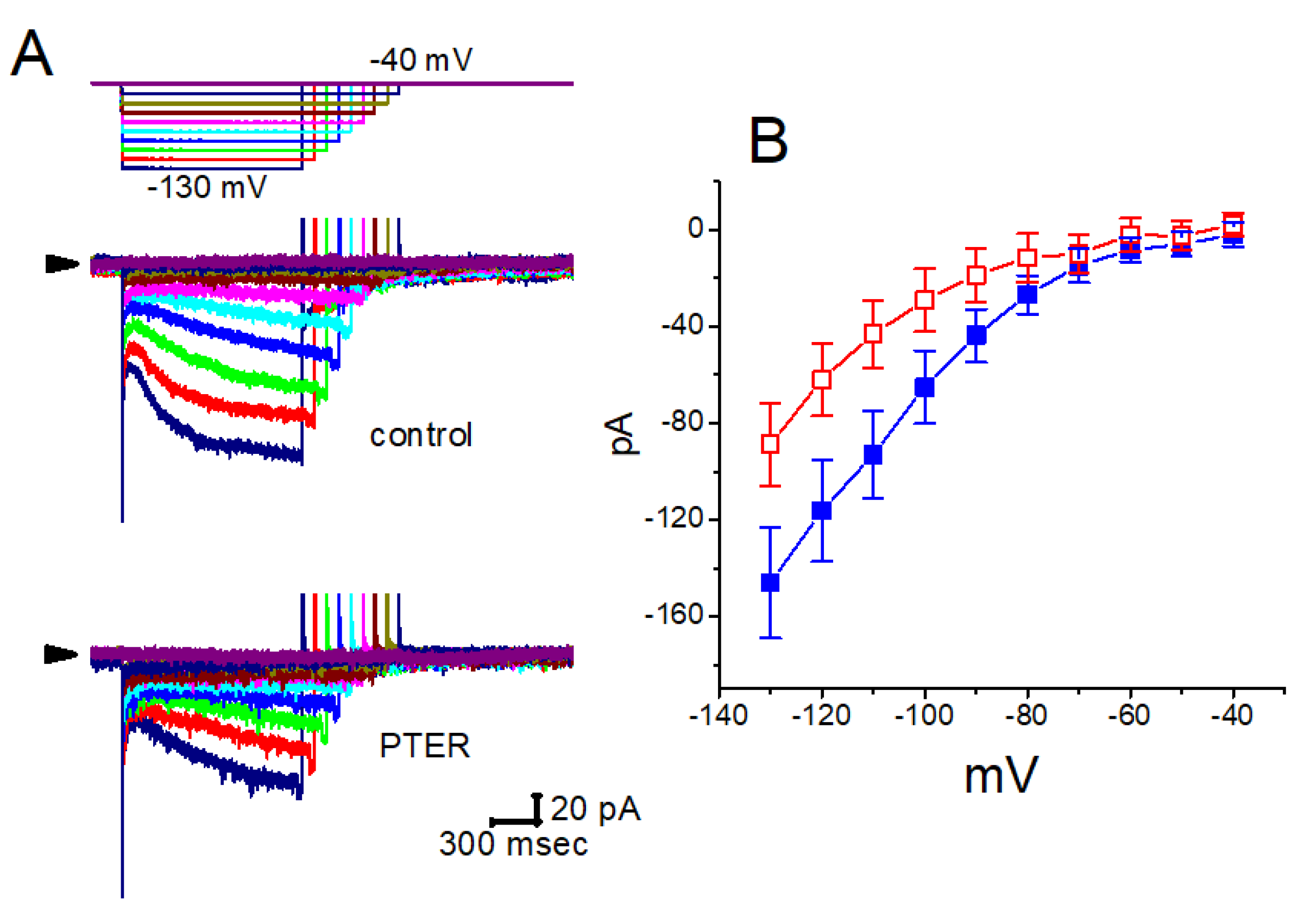
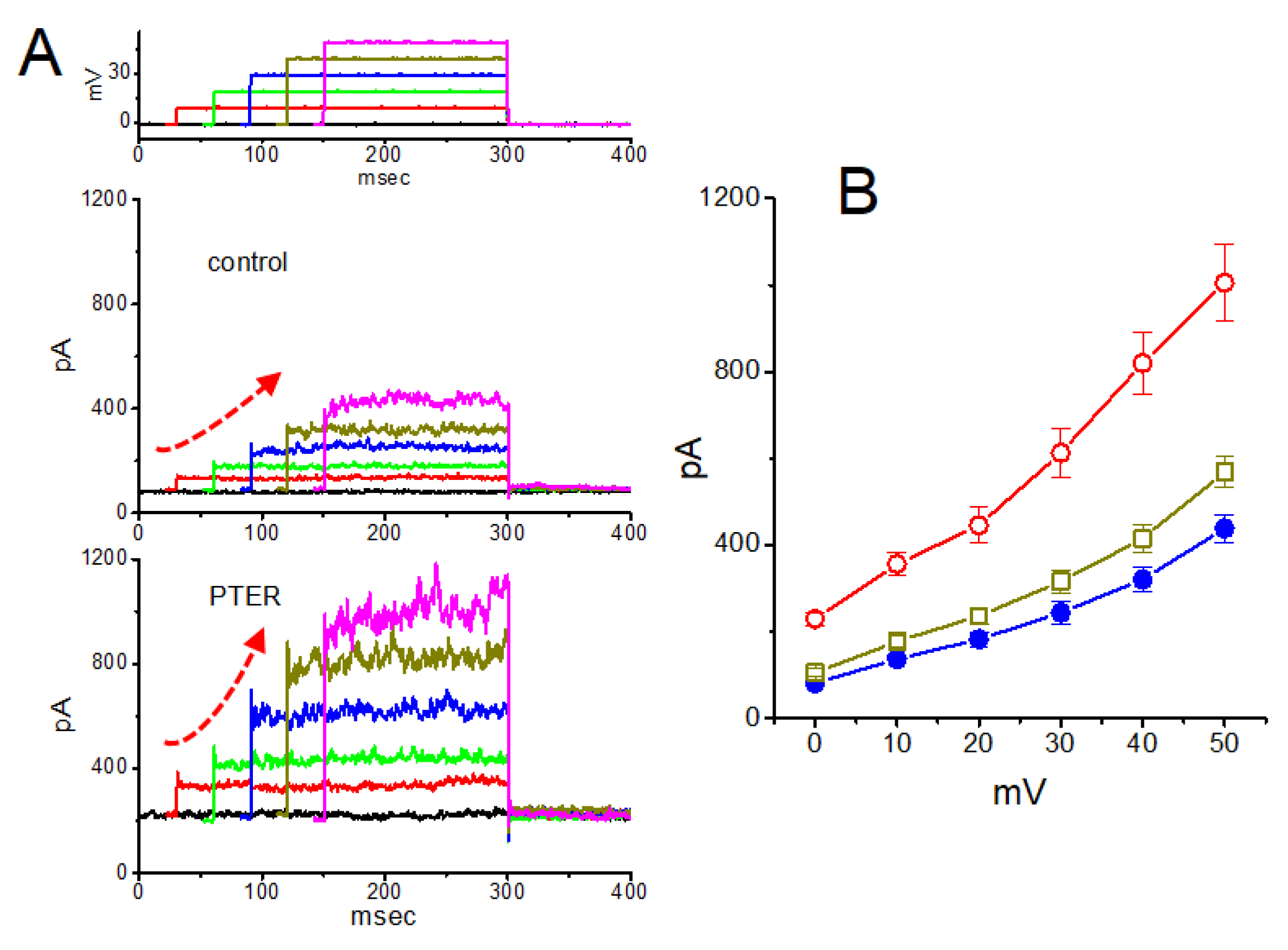
5.6. Pterostilbene
6. Other Aromatic or Conjugated Systems
6.1. Isoplumbagin and Plumbagin
6.2. Verteporfin
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| erg | ether-à-go-go-related gene |
| HCN channel | Hyperpolarization-activated cyclic nucleotide-gated channel |
| IC50 | Concentration required for half-maximal inhibition |
| Ih | Hyperpolarization-activated cation current |
| IK(Ca) | Ca2+-activated K+ current |
| IK(DR) | Delayed-rectifier K+ current |
| IK(erg) | erg-mediated K+ current |
| IK(M) | M-type K+ current |
| INa | Voltage-gated Na+ current |
| NaV (SCN) channel | Voltage-gated Na+ channel |
| I-V relationship | Current versus voltage relationship |
References
- Rao, R.; Mohammed, C.; Alschuler, L.; Pomeranz Krummel, D.A.; Sengupta, S. Phytochemical Modulation of Ion Channels in Oncologic Symptomatology and Treatment. Cancers (Basel) 2024, 16, 1786. [Google Scholar] [CrossRef]
- Ansari, P.; Reberio, A.D.; Ansari, N.J.; Kumar, S.; Khan, J.T.; Chowdhury, S.; Abd El-Mordy, F.M.; Hannan, J.M.A.; Flatt, P.R.; Abdel-Wahab, Y.H.A.; Seidel, V. Therapeutic Potential of Medicinal Plants and Their Phytoconstituents in Diabetes, Cancer, Infections, Cardiovascular Diseases, Inflammation and Gastrointestinal Disorders. Biomedicines 2025, 13, 454. [Google Scholar] [CrossRef] [PubMed]
- Sonnino, R.; Ciccarelli, G.; Moffa, S.; Soldovieri, L.; Di Giuseppe, G.; Brunetti, M.; Cinti, F.; Di Piazza, E.; Gasbarrini, A.; Nista, E.C.; Pontecorvi, A.; Giaccari, A.; Mezza, T. Exploring nutraceutical approaches linking metabolic syndrome and cognitive impairment. iScience 2025, 28, 111848. [Google Scholar] [CrossRef]
- Froldi, G. Bioactivity of Natural Compounds: From Plants to Humans. Molecules 2026, 31, 295. [Google Scholar] [CrossRef]
- Pullela, R.; Young, L.; Gallagher, B.; Avis, S.P.; Randell, E.W. A case of fatal aconitine poisoning by Monkshood ingestion. J. Forensic Sci. 2008, 53, 491–494. [Google Scholar] [CrossRef]
- Gao, Y.; Fan, H.; Nie, A.; Yang, K.; Xing, H.; Gao, Z.; Yang, L.; Wang, Z.; Zhang, L. Aconitine: A review of its pharmacokinetics, pharmacology, toxicology and detoxification. J. Ethnopharmacol. 2022, 293, 115270. [Google Scholar] [CrossRef]
- Wang, S.Y.; Wang, G.K. Voltage-gated sodium channels as primary targets of diverse lipid-soluble neurotoxins. Cell. Signal. 2003, 15, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, Y.P.; Guo, S.; Li, T.T.; Wang, Q.Y.; Zhang, X.; Zheng, Y.M.; Wen, Y.Q.; Meng, F.H.; Zhang, T.J. Novel E-F ring derivatives of aconitine scaffold as potent Hsp90 inhibitors for the treatment of colorectal cancer. Eur. J. Med. Chem. 2025, 296, 117895. [Google Scholar] [CrossRef]
- Lin, M.W.; Wang, Y.J.; Liu, S.I.; Lin, A.A.; Lo, Y.C.; Wu, S.N. Characterization of aconitine-induced block of delayed rectifier K+ current in differentiated NG108-15 neuronal cells. Neuropharmacology 2008, 54, 912–923. [Google Scholar] [CrossRef]
- Wang, Y.J.; Chen, B.S.; Lin, M.W.; Lin, A.A.; Peng, H.; Sung, R.J.; Wu, S.N. Time-dependent block of ultrarapid-delayed rectifier K+ currents by aconitine, a potent cardiotoxin, in heart-derived H9c2 myoblasts and in neonatal rat ventricular myocytes. Toxicol. Sci. 2008, 106, 454–463. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.N.; Chen, B.S.; Lin, M.W.; Liu, Y.C. Contribution of slowly inactivating potassium current to delayed firing of action potentials in NG108-15 neuronal cells: experimental and theoretical studies. J. Theor. Biol. 2008, 252, 711–721. [Google Scholar] [CrossRef]
- Wu, S.N.; Chen, B.S.; Lo, Y.C. Evidence for aconitine-induced inhibition of delayed rectifier K+ current in Jurkat T-lymphocytes. Toxicology 2011, 289, 11–18. [Google Scholar] [CrossRef]
- Chou, C.J.; So, E.C. Effects of aconitine on membrane currents and action potentials in neonatal rat ventricular myocytes and its impact on electrocardiographic changes. Medical Research Archives 2024, 12. [Google Scholar] [CrossRef]
- Lee, C.H.; Chiang, S.L.; Ko, A.M.; Hua, C.H.; Tsai, M.H.; Warnakulasuriya, S.; Ibrahim, S.O.; Sunarjo; Zain, R.B.; Ling, T.Y.; Huang, C.L.; Lane, H.Y.; Lin, C.C.; Ko, Y.C. Betel-quid dependence domains and syndrome associated with betel-quid ingredients among chewers: an Asian multi-country evidence. Addiction 2014, 109, 1194–1204. [Google Scholar] [CrossRef]
- Li, M.; Gao, F.; Zhou, Z.S.; Zhang, H.M.; Zhang, R.; Wu, Y.F.; Bai, M.H.; Li, J.J.; Lin, S.R.; Peng, JY. Arecoline inhibits epithelial cell viability by upregulating the apoptosis pathway: implication for oral submucous fibrosis. Oncol Rep. 2014, 31, 2422–2428. [Google Scholar] [CrossRef]
- Chan, P.K.; Keyes, A.; Papanastasiou, S.; Sarrafpour, B.; Ramaswamy, Y.; Co, S.; Zoellner, H.; Chami, B. Arecoline stimulates the IL-33/13 axis and upregulates pro-fibrotic CTGF: A possible role in oral submucous fibrosis. Food Chem. Toxicol. 2026, 207, 115811. [Google Scholar] [CrossRef]
- Lee, S.C.; Tsai, C.C.; Yao, C.H.; Hsu, Y.M.; Chen, Y.S.; Wu, M.C. Effect of arecoline on regeneration of injured peripheral nerves. Am. J. Chin. Med. 2013, 41, 865–885. [Google Scholar] [CrossRef]
- So, E.C.; Huang, Y.M.; Hsing, C.H.; Liao, Y.K.; Wu, S.N. Arecoline inhibits intermediate-conductance calcium-activated potassium channels in human glioblastoma cell lines. Eur. J. Pharmacol. 2015, 758, 177–187. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Lai, W.; Zhang, Y.; Liu, L.; Luo, X.; Zeng, Y.; Wu, H.; Lan, Q.; Chu, Z. Tumor-associated macrophage-derived IL-6 and IL-8 enhance invasive activity of LoVo cells induced by PRL-3 in a KCNN4 channel-dependent manner. BMC Cancer 2014, 14, 330. [Google Scholar] [CrossRef]
- Bova, S.; Padrini, R.; Goldman, W.F.; Berman, D.M.; Cargnelli, G. On the mechanism of vasodilating action of berberine: possible role of inositol lipid signaling system. J. Pharmacol. Exp. Ther. 1992, 261, 318–323. [Google Scholar] [CrossRef] [PubMed]
- Chi, C.W.; Chang, Y.F.; Chao, T.W.; Chiang, S.H.; P'eng, F.K.; Lui, W.Y.; Liu, T.Y. Flowcytometric analysis of the effect of berberine on the expression of glucocorticoid receptors in human hepatoma HepG2 cells. Life Sci. 1994, 54, 2099–2107. [Google Scholar] [CrossRef]
- Wu, S.N.; Yu, H.S.; Jan, C.R.; Li, H.F.; Yu, C.L. Inhibitory effects of berberine on voltage- and calcium-activated potassium currents in human myeloma cells. Life Sci. 1998, 62, 2283–2294. [Google Scholar] [CrossRef]
- Hsu, Y.Y.; Chen, C.S.; Wu, S.N.; Jong, Y.J.; Lo, Y.C. Berberine activates Nrf2 nuclear translocation and protects against oxidative damage via a phosphatidylinositol 3-kinase/Akt-dependent mechanism in NSC34 motor neuron-like cells. Eur. J. Pharm. Sci. 2012, 46, 415–425. [Google Scholar] [CrossRef]
- Bian, X.; Guo, Q.; Yau, L.F.; Yang, L.; Wang, X.; Zhao, S.; Wu, S.; Qin, X.; Jiang, Z.H.; Li, C. Berberine-inspired ionizable lipid for self-structure stabilization and brain targeting delivery of nucleic acid therapeutics. Nat. Commun. 2025, 16, 2368. [Google Scholar] [CrossRef]
- Zhou, W.; Asif, A.; Situ, C.; Wang, J.; Hao, H. Multiple target and regulatory pathways of berberine. Phytomedicine 2025, 146, 157030. [Google Scholar] [CrossRef] [PubMed]
- Moskalev, A.; Veselova, O. Potential dietary geroprotectors and their impact on key mechanisms of aging. Biogerontology 2026, 27, 8. [Google Scholar] [CrossRef] [PubMed]
- Britch, S.C.; Babalonis, S.; Walsh, S.L. Cannabidiol: pharmacology and therapeutic targets. Psychopharmacology (Berl) 2021, 238, 9–28. [Google Scholar] [CrossRef]
- Huang, C.W.; Lin, P.C.; Chen, J.L.; Lee, M.J. Cannabidiol Selectively Binds to the Voltage-Gated Sodium Channel Na(v)1.4 in Its Slow-Inactivated State and Inhibits Sodium Current. Biomedicines 2021, 9, 1141. [Google Scholar] [CrossRef] [PubMed]
- Ghovanloo, M.R.; Ruben, P.C. Cannabidiol and Sodium Channel Pharmacology: General Overview, Mechanism, and Clinical Implications. Neuroscientist 2022, 28, 318–334. [Google Scholar] [CrossRef]
- Liu, Y.C.; So, E.C.; Wu, S.N. Cannabidiol Modulates M-Type K+ and Hyperpolarization-Activated Cation Currents. Biomedicines 2023, 11, 2651. [Google Scholar] [CrossRef]
- Giang, P.M.; Son, P.T.; Lee, J.J.; Otsuka, H. Four ent-kaurane-type diterpenoids from Croton tonkinensis GAGNEP. Chem. Pharm. Bull. (Tokyo) 2004, 52, 879–882. [Google Scholar] [CrossRef]
- Lee, H.M.; Kuo, P.C.; Chen, W.H.; Chen, P.J.; Lam, S.H.; Su, Y.C.; Chen, C.H. Diterpenoid from Croton tonkinensis as a Potential Radiation Sensitizer in Oral Squamous Cell Carcinoma: An In Vitro Study. Int. J. Mol. Sci. 2024, 25, 11839. [Google Scholar] [CrossRef] [PubMed]
- Hsiao, H.T.; Lee, Y.C.; Liu, Y.C.; Kuo, P.C.; Wu, S.N. Differential suppression of delayed-rectifier and inwardly rectifier K+ currents by a group of ent-kaurane-type diterpenoids from Croton tonkinensis, in microglial cells. Eur. J. Pharmacol. 2019, 856, 172414. [Google Scholar] [CrossRef] [PubMed]
- Kuo, P.C.; Liu, Y.C.; Lo, Y.C.; Wu, S.N. Characterization of Inhibitory Effectiveness in Hyperpolarization-Activated Cation Currents by a Group of ent-Kaurane-Type Diterpenoids from Croton tonkinensis. Int. J. Mol. Sci. 2020, 21, 1268. [Google Scholar] [CrossRef]
- Mitra, S.; Rauf, A.; Tareq, A.M.; Jahan, S.; Emran, T.B.; Shahriar, T.G.; Dhama, K.; Alhumaydhi, F.A.; Aljohani, A.S.M.; Rebezov, M.; Uddin, M.S.; Jeandet, P.; Shah, Z.A.; Shariati, M.A.; Rengasamy, K.R. Potential health benefits of carotenoid lutein: An updated review. Food Chem. Toxicol. 2021, 154, 112328. [Google Scholar] [CrossRef]
- Mrowicka, M.; Mrowicki, J.; Kucharska, E.; Majsterek, I. Lutein and Zeaxanthin and Their Roles in Age-Related Macular Degeneration-Neurodegenerative Disease. Nutrients 2022, 14, 827. [Google Scholar] [CrossRef]
- Chuang, C.W.; Chang, K.P.; Cho, H.Y.; Chuang, T.H.; Yu, M.C.; Wu, C.L.; Wu, S.N. Characterization of Inhibitory Capability on Hyperpolarization-Activated Cation Current Caused by Lutein (β,ε-Carotene-3,3'-Diol), a Dietary Xanthophyll Carotenoid. Int. J. Mol. Sci. 2022, 23, 7186. [Google Scholar] [CrossRef] [PubMed]
- Dini, L.; Del Lungo, M.; Resta, F.; Melchiorre, M.; Spinelli, V.; Di Cesare Mannelli, L.; Ghelardini, C.; Laurino, A.; Sartiani, L.; Coppini, R.; Mannaioni, G.; Cerbai, E.; Romanelli, M.N. Selective Blockade of HCN1/HCN2 Channels as a Potential Pharmacological Strategy Against Pain. Front. Pharmacol. 2018, 9, 1252. [Google Scholar] [CrossRef]
- Hsiao, H.T.; Liu, Y.C.; Liu, P.Y.; Wu, S.N. Concerted suppression of Ih and activation of IK(M) by ivabradine, an HCN-channel inhibitor, in pituitary cells and hippocampal neurons. Brain Res. Bull. 2019, 149, 11–20. [Google Scholar] [CrossRef]
- Wu, S.N.; Fang, Y.H.; Liu, P.Y.; Liu, Y.W. Characterization of hyperpolarization-induced cation current in differentiated human embryonic stem cell-derived cardiomyocytes. Journal of the American College of Cardiology (JACC) 2020, 75 (Suppl), P31322–X. [Google Scholar] [CrossRef]
- Nache, V.; Eick, T.; Schulz, E.; Schmauder, R.; Benndorf, K. Hysteresis of ligand binding in CNGA2 ion channels. Nat. Commun. 2013, 4, 2866. [Google Scholar] [CrossRef] [PubMed]
- Barthel, L.; Reetz, O.; Strauss, U. Use Dependent Attenuation of Rat HCN1-Mediated Ih in Intact HEK293 Cells. Cell. Physiol. Biochem. 2016, 38, 2079–2093. [Google Scholar] [CrossRef]
- Xiao, Y.F.; Chandler, N.; Dobrzynski, H.; Richardson, E.S.; Tenbroek, E.M.; Wilhelm, J.J.; Sharma, V.; Varghese, A.; Boyett, M.R.; Iaizzo, P.A.; Sigg, D.C. Hysteresis in human HCN4 channels: a crucial feature potentially affecting sinoatrial node pacemaking. Sheng Li Xue Bao 2010, 62, 1–13. [Google Scholar]
- Datunashvili, M.; Chaudhary, R.; Zobeiri, M.; Lüttjohann, A.; Mergia, E.; Baumann, A.; Balfanz, S.; Budde, B.; van Luijtelaar, G.; Pape, H.C.; Koesling, D.; Budde, T. Modulation of Hyperpolarization-Activated Inward Current and Thalamic Activity Modes by Different Cyclic Nucleotides. Front. Cell. Neurosci. 2018, 12, 369. [Google Scholar] [CrossRef]
- Chang, W.T.; Ragazzi, E.; Liu, P.Y.; Wu, S.N. Effective block by pirfenidone, an antifibrotic pyridone compound (5-methyl-1-phenylpyridin-2[H-1]-one), on hyperpolarization-activated cation current: An additional but distinctive target. Eur. J. Pharmacol. 2020, 882, 173237. [Google Scholar] [CrossRef]
- Kodirov, S.A. Delineation and functions of HCN channels in neurons. Prog. Biophys. Mol. Biol. 2025, 198, 21–31. [Google Scholar] [CrossRef] [PubMed]
- D'Aloisio, R.; Di Antonio, L.; Toto, L.; Rispoli, M.; Di Iorio, A.; Delvecchio, G.; Mastropasqua, R. Choroidal Changes in Blood Flow in Patients with Intermediate AMD after Oral Dietary Supplement Based on Astaxanthin, Bromelain, Vitamin D3, Folic Acid, Lutein, and Antioxidants. Medicina (Kaunas) 2022, 58, 1092. [Google Scholar] [CrossRef]
- Ogaard, B.; Larsson, E.; Glans, R.; Henriksson, T.; Birkhed, D. Antimicrobial effect of a chlorhexidine-thymol varnish (Cervitec) in orthodontic patients. A prospective, randomized clinical trial. J. Orofac. Orthop. 1997, 58, 206−213. [Google Scholar]
- Kasparaviciene, G.; Kalveniene, Z.; Pavilonis, A.; Marksiene, R.; Dauksiene, J.; Bernatoniene, J. Formulation and Characterization of Potential Antifungal Oleogel with Essential Oil of Thyme. Evid. Based Complement. Alternat. Med. 2018, 2018, 9431819. [Google Scholar] [CrossRef]
- Aksoy, T.; Kilimcioğlu, A.A. Thymol's antileishmanial activity and its impact on host cytokine profiles: In vitro and ex vivo studies on Leishmania tropica. Parasitol. Int. 2026, 110, 103139. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Liu, L.; Tang, S.; Li, D.; Dai, C. Antifungal Activity of Natural Thymol: Advances on Molecular Mechanisms and Therapeutic Potential. Biomolecules 2026, 16, 149. [Google Scholar] [CrossRef]
- Hoo, G.W.; Hinds, R.L.; Dinovo, E.; Renner, S.W. Fatal large-volume mouthwash ingestion in an adult: a review and the possible role of phenolic compound toxicity. J. Intensive Care Med. 2003, 18, 150–155. [Google Scholar]
- Dee Manuel, M.P.; Shih, Y.H.; Hsia, S.M.; Wang, T.H.; Tseng, Y.H.; Tu, M.G.; Shieh, T.M. Evaluating thymol vapor for biofilm removal and biocompatibility in curved root canal models in vitro. J. Dent. Sci. 2026, 21, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Shen, A.Y.; Huang, M.H.; Wang, T.S.; Wu, H.M.; Kang, Y.F; Chen, CL. Thymol-evoked Ca2+ mobilization and ion currents in pituitary GH3 cells. Nat. Prod. Commun. 2009, 4, 749–752. [Google Scholar] [PubMed]
- Huang, M.H.; Wu, S.N.; Shen, A.Y. Stimulatory actions of thymol, a natural product, on Ca2+-activated K+ current in pituitary GH3 cells. Planta Med. 2005, 71, 1093–1098. [Google Scholar] [CrossRef]
- Zheng, Y.; Zhang, W.J.; Wang, X.M. Triptolide with potential medicinal value for diseases of the central nervous system. CNS Neurosci. Ther. 2013, 19, 76–82. [Google Scholar]
- Chen, L.W.; Wang, Y.Q.; Wei, L.C.; Shi, M.; Chan, Y.S. Chinese herbs and herbal extracts for neuroprotection of dopaminergic neurons and potential therapeutic treatment of Parkinson's disease. CNS Neurol. Disord. Drug Targets 2007, 6, 273–281. [Google Scholar] [CrossRef]
- Su, Z.; Yuan, Y.; Cao, L.; Zhu, Y.; Gao, L.; Qiu, Y.; He, C. Triptolide promotes spinal cord repair by inhibiting astrogliosis and inflammation. Glia 2010, 58, 901–915. [Google Scholar] [CrossRef]
- Zhang, H.; Zhu, W.; Su, X.; Wu, S.; Lin, Y.; Li, J.; Wang, Y.; Chen, J.; Zhou, Y.; Qiu, P.; Yan, G.; Zhao, S.; Hu, J.; Zhang, J. Triptolide inhibits proliferation and invasion of malignant glioma cells. J. Neurooncol. 2012, 109, 53–62. [Google Scholar] [CrossRef]
- Lin, J.; Chen, L.Y.; Lin, Z.X.; Zhao, M.L. The effect of triptolide on apoptosis of glioblastoma multiforme (GLM) cells. J. Int. Med. Res. 2007, 35, 637–643. [Google Scholar] [CrossRef] [PubMed]
- So, E.C.; Lo, Y.C.; Chen, L.T.; Kao, C.A.; Wu, S.N. High effectiveness of triptolide, an active diterpenoid triepoxide, in suppressing Kir-channel currents from human glioma cells. Eur. J. Pharmacol. 2014, 738, 332–341. [Google Scholar] [CrossRef] [PubMed]
- Haj-Yasein, N.N.; Jensen, V.; Vindedal, G.F.; Gundersen, G.A.; Klungland, A.; Ottersen, O.P.; Hvalby, O.; Nagelhus, E,A. Evidence that compromised K+ spatial buffering contributes to the epileptogenic effect of mutations in the human Kir4.1 gene (KCNJ10). Glia 2011, 59, 1635–1642. [Google Scholar] [CrossRef]
- Zhou, X.; Zhao, C.; Xu, H.; Xu, Y.; Zhan, L.; Wang, P.; He, J.; Lu, T.; Gu, Y.; Yang, Y.; Xu, C.; Chen, Y.; Liu, Y.; Zeng, Y.; Tian, F.; Chen, Q.; Xie, X.; Liu, J.; Hu, H.; Li, J.; Zheng, Y.; Guo, J.; Gao, Z. Pharmacological inhibition of Kir4.1 evokes rapid-onset antidepressant responses. Nat. Chem. Biol. 2024, 20, 857–866. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Jiang, B.; Shan, Y.; Wang, X.; Lv, X; Mohamed, J; Li, H; Wang, C; Chen, J; Sun, J. Metabolic mapping of Schisandra chinensis lignans and their metabolites in rats using a metaboliomic approach based on HPLC with quadrupole time-of-flight MS/MS spectrometry. J. Sep. Sci. 2020, 43, 378–388. [Google Scholar] [CrossRef]
- Takanche, J.S.; Kim, J.E.; Han, S.H.; Yi, H.K. Effect of gomisin A on osteoblast differentiation in high glucose-mediated oxidative stress. Phytomedicine 2020, 66, 153107. [Google Scholar] [CrossRef]
- Hwang, I.S.; Kim, J.E.; Lee, Y.J.; Kwak, M.H.; Choi, Y.H.; Kang, B.C.; Hong, J.T.; Hwang, D.Y. Protective effects of gomisin A isolated from Schisandra chinensis against CCl4-induced hepatic and renal injury. Int. J. Mol. Med. 2013, 31, 888–898. [Google Scholar] [CrossRef]
- Kim, E.J.; Jang, M.; Lee, M.J.; Choi, J.H.; Lee, S.J.; Kim, S.K.; Jang, D.S.; Cho, I.H. Schisandra chinensis stem ameliorates 3-nitropropionic acid-induced striatal toxicity via activation of the Nrf2 pathway and inhibition of the MAPKs and NF-B pathways. Front. Pharmacol. 2017, 8, 673. [Google Scholar]
- Nam, S.Y.; Kim, K.Y.; Kim, M.H.; Jang, J.B.; Rah, S.Y.; Chae, H.J.; Lee, J.M.; Kim, H.M.; Jeong, H.J. Anti-inflammatory effects of a traditional Korean medicine: Ojayeonjonghwan. Pharm. Bio. 2017, 55, 1856–1862. [Google Scholar]
- Ye, B.H.; Lee, S.J.; Choi, Y.W.; Park, S.Y.; Kim, C.D. Preventive effect of gomisin J from Schisandra chinensis on angiotensin II-induced hypertension via an increased nitric oxide bioavailability. Hypertens. Res. 2015, 38, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Hwang, D.; Shin, S.Y.; Lee, Y.; Hyun, J.; Yong, Y.; Park, J.C.; Lee, Y.H.; Lim, Y. A compound isolated from Schisandra chinensis induces apoptosis. Bioorg. Med. Chem. Lett. 2011, 21, 6054–6057. [Google Scholar] [CrossRef]
- Hong, S.H.; Li, M.; Jeung, E.B.; Lee, G.S.; Hong, E.J.; Choi, Y.W.; An, B.S. Therapeutic effects of Schisandra chinensis on the hyperprolactinemia in rat. Int. J. Oncol. 2017, 50, 1448–1454. [Google Scholar] [CrossRef]
- Chang, W.T.; Wu, S.N. Inhibitory Effectiveness of Gomisin A, a Dibenzocyclooctadiene Lignan Isolated from Schizandra chinensis, on the Amplitude and Gating of Voltage-Gated Na+ Current. Int. J. Mol. Sci. 2020, 21, 8816. [Google Scholar] [CrossRef]
- Fujita, M.; Itokawa, H.; Sashida, Y. Studies on the components of Magnolia obovate Thunb. 3. Occurrence of magnolol and hõnokiol in M. obovate and other allied plants. Yakugaku Zasshi 1973, 93, 429–434. [Google Scholar] [CrossRef]
- Ong, C.P.; Lee, W.L.; Tang, Y.Q.; Yap, W.H. Honokiol: a review of its anticancer potential and mechanisms. Cancers (Basel) 2019, 12, 48. [Google Scholar] [CrossRef]
- Lu, C.H.; Chen, S.H.; Chang, Y.S.; Liu, Y.W.; Wu, J.Y.; Lim, Y.P.; Yu, H.I.; See, Y.R. Honokiol, a potential therapeutic agent, induces cell cycle arrest and program cell death in vitro and in vivo in human thyroid cancer cells. Pharmacol. Res. 2017, 115, 288–298. [Google Scholar] [CrossRef]
- Tachikawa, E.; Takahashi, M.; Kashimoto, T. Effects of extract and ingredients isolated from Magnolia obovata Thunberg on catecholamine secretion from bovine adrenal chromaffin cells. Biochem. Pharmacol. 2000, 60, 433–440. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Gan, D.; Wu, J.; Liao, M.; Liao, X.; Ai, W. Honokiol exerts antidepressant effects in rats exposed to chronic unpredictable mild stress by regulating brain derived neurotrophic factor level and hypothalamus-pituitary-adrenal axis activity. Neurochem. Res. 2018, 43, 1519–1528. [Google Scholar] [PubMed]
- Chan, M.H.; Chen, H.H.; Lo, Y.C.; Wu, S.N. Effectiveness in the Block by Honokiol, a Dimerized Allylphenol from Magnolia Officinalis, of Hyperpolarization-Activated Cation Current and Delayed-Rectifier K+ Current. Int. J. Mol. Sci. 2020, 21, 4260. [Google Scholar] [CrossRef] [PubMed]
- Khodir, S.A.; Sweed, E.M.; El-Haroun, H.; Abd-Elhamid, T.H.; El Derbaly, S.A.; Mahmoud, A.R.; Motawea, S.M. Honokiol ameliorates reserpine-induced fibromyalgia through antioxidant, anti-inflammatory, neurotrophic, and anti-apoptotic mechanisms. Sci. Rep. 2025, 15, 25983. [Google Scholar] [CrossRef]
- Shiao, M.S. Natural products of the medicinal fungus Ganoderma lucidum: occurrence, biological activities, and pharmacological functions. Chem. Rec. 2003, 3, 172–180. [Google Scholar] [CrossRef]
- Cör, D.; Knez, Ž.; Knez Hrnčič, M. Antitumour, antimicrobial, antioxidant and antiacetylcholinesterase effect of Ganoderma lucidum terpenoids and polysaccharides: a review. Molecules 2018, 23, 649. [Google Scholar] [CrossRef]
- Peng, X.; Li, L.; Dong, J.; Lu, S.; Lu, J.; Li, X.; Zhou, L.; Qiu, M. Lanostane-type triterpenoids from the fruiting bodies of Ganoderma applanatum. Phytochemistry 2019, 157, 103–110. [Google Scholar] [CrossRef]
- Kuok, Q.Y.; Yeh, C.Y.; Su, B.C.; Hsu, P.L.; Ni, H.; Liu, M.Y.; Mo, F.E. The triterpenoids of Ganoderma tsugae prevent stress-induced myocardial injury in mice. Mol. Nutr. Food Res. 2013, 57, 1892–1896. [Google Scholar] [CrossRef]
- Ma, B.; Ren, W.; Zhou, Y.; Ma, J.; Ruan, Y.; Wen, C.N. Triterpenoids form the spores of Ganoderma lucidum. N. Am. J. Med. Sci. 2011, 3, 495–498. [Google Scholar] [CrossRef] [PubMed]
- Lou, H.W.; Guo, X.Y.; Zhang, X.C.; Guo, L.Q.; Lin, J.F. Optimization of cultivation conditions of lingzhi or reishi medicinal mushroom, Ganoderma lucidum (Agaricomycetes) for the highest antioxidant activity and antioxidant content. Int. J. Med. Mushrooms 2019, 21, 353–366. [Google Scholar] [CrossRef] [PubMed]
- Liang, C.; Tian, D.; Liu, Y.; Li, H.; Zhu, J.; Li, M.; Xin, M.; Xia, J. Review of the molecular mechanisms of Ganoderma lucidum triterpenoids: ganoderic acids A, C2, D, F, DM, X and Y. Eur. J. Med. Chem. 2019, 174, 130–141. [Google Scholar] [CrossRef]
- Chang, W.T.; Gao, Z.H.; Lo, Y.C.; Wu, S.N. Evidence for Effective Inhibitory Actions on Hyperpolarization-Activated Cation Current Caused by Ganoderma Triterpenoids, the Main Active Constituents of Ganoderma Spores. Molecules 2019, 24, 4256. [Google Scholar] [CrossRef]
- Kim, A.Y.; Yun, C.I.; Lee, J.G.; Kim, Y.J. Determination and daily intake estimation of lignans in sesame seeds and sesame oil products in Korea. Foods 2020, 9, 394. [Google Scholar] [CrossRef]
- Dhar, P.; Chattopadhya, K.; Bhattacharyya, D.; Biswas, A.; Roy, B.; Ghosh, S. Ameliorative influence of sesame lignans on lipid profile and lipid peroxidation in induced diabetic rats. J. Agric. Food Chem. 2007, 55, 5875–5880. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.T.; Chen, J.; Jiao, R.; Peng, C.; Zuo, Y.; Lei, L.; Liu, Y.; Wang, X.; Ma, K.Y.; Huang, Y.; Chen, Z.Y. Cholesterol-lowering activity of sesamin is associated with down-regulation on genes of sterol transporters involved in cholesterol absorption. J. Agri. Food Chem. 2015, 63, 2963–2969. [Google Scholar] [CrossRef]
- Ruankham, W.; Suwanjang, W.; Wongchitrat, P.; Prachayasittikul, V.; Prachayasittikul, S.; Phopin, K. Sesamin and sesamol attenuate H2O2-induced oxidative stress on human neuronal cells via the SIRT1-SIRT3-FOXO3a signaling pathway. Nutr. Neurosci. 2021, 24, 90–101. [Google Scholar] [CrossRef]
- Jayaraj, P.; Narasimhulu, C.A.; Rajagopalan, S.; Parthasarathy, S.; Desikan, R. Sesamol: a powerful functional food ingredient from sesame oil for cardioprotection. Food Funct. 2020, 11, 1198–1210. [Google Scholar] [CrossRef]
- Kuo, P.C.; Kao, Z.H.; Lee, S.W.; Wu, S.N. Effects of Sesamin, the Major Furofuran Lignan of Sesame Oil, on the Amplitude and Gating of Voltage-Gated Na+ and K+ Currents. Molecules 2020, 25, 3062. [Google Scholar] [CrossRef]
- Pan, Y.; Cummins, T.R. Distinct functional alterations in SCN8A epilepsy mutant channels. J. Physiol. 2020, 598, 381–401. [Google Scholar]
- Djamgoz, M.B.A. Stemness of Cancer: A Study of Triple-negative Breast Cancer From a Neuroscience Perspective. Stem Cell Rev. Rep. 2025, 21, 337–350. [Google Scholar] [CrossRef]
- Guadano, A.; Gutierrez, C.; de La Pena, E.; Cortes, D.; Gonzalez-Coloma, A. Insecticidal and mutagenic evaluation of two annonaceous acetogenins. J. Nat. Prod. 2000, 63, 773–776. [Google Scholar] [CrossRef] [PubMed]
- Queiroz, E.F.; Roblot, F.; Duret, P.; Figadere, B.; Gouyette, A.; Laprevote, O.; Serani, L.; Hocquemiller, R. Synthesis, spectroscopy, and cytotoxicity of glycosylated acetogenin derivatives as promising molecules for cancer therapy. J. Med. Chem. 2000, 43, 1604–1610. [Google Scholar] [CrossRef] [PubMed]
- Arndt, S.; Emde, U.; Baurle, S.; Friedrich, T.; Grubert, L.; Koert, U. Quinone-annonaceous acetogenins: synthesis and complex I inhibition studies of a new class of natural product hybrids. Chemistry 2001, 7, 993–1005. [Google Scholar] [CrossRef]
- Shimda, H.; Grutzner, J.B.; Kozlowski, J.F.; McLaughlin, J.L. Membrane conformations and their relation to cytotoxicity of asimicin and its analogues. Biochemistry 1998, 37, 854–866. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.F.; Liu, Z.C.; Xie, B.F.; Li, Z.M.; Feng, G.K.; Xie, H.H.; Wu, S.J.; Yang, R.Z.; Wei, X.Y.; Zeng, Y.X. Involvement of caspase-3 activation in squamocin-induced apoptosis in leukemia cell line HL-60. Life Sci. 2002, 70, 1259–1269. [Google Scholar] [CrossRef]
- Wu, S.N.; Chiang, H.T.; Chang, F.R.; Liaw, C.C.; Wu, Y.C. Stimulatory effects of squamocin, an Annonaceous acetogenin, on Ca2+-activated K+ current in cultured smooth muscle cells of human coronary artery. Chem. Res. Toxicol. 2003, 16, 15–22. [Google Scholar] [CrossRef]
- Priyadarsini, K.I. The chemistry of curcumin: from extraction to therapeutic agent. Molecules 2014, 19, 20091–20112. [Google Scholar] [CrossRef]
- Meng, B.; Li, J.; Cao, H. Antioxidant and antiiflammatory activities of curcumin on diabetes mellitus and its complication. Curr. Pharm. Des. 2013, 19, 2101–2113. [Google Scholar]
- Jiménez-Osorio, A.S.; Monroy, A.; Alavez, S. Curcumin and insulin resistance-Molecular targets and clinical evidences. Biofactors 2016, 42, 561–580. [Google Scholar] [CrossRef]
- Kuo, P.C.; Yang, C.J.; Lee, Y.C.; Chen, P.C.; Liu, Y.C.; Wu, S.N. The comprehensive electrophysiological study of curcuminoids on delayed-rectifier K+ currents in insulin-secreting cells. Eur. J. Pharmacol. 2018, 819, 233–241. [Google Scholar] [CrossRef]
- Gutierres, V.O.; Campos, M.L.; Arcaro, C.A.; Assis, R.P.; Baldan-Cimatti, H.M.; Peccinini, R.G.; Paula-Gomes, S.; Kettelhut, I.C.; Baviera, A.M.; Brunetti, I. L: Curcumin pharmacokinetic and pharmacodynamics evidences in streptozotocin-diabetic rats support the antidiabetic activity to be via metabolite(s). Evid. Based. Complement. Alternat. Med. 2015, 2015, 678218. [Google Scholar]
- Lim, H.J.; Lee, J.H.; Choi, J.S.; Lee, S.K.; Kim, Y.S.; Kim, H.P. Inhibition of airway inflammation by the roots of Angelica decursiva and its constituent, columbianadin. J. Ethnopharmacol. 2014, 155, 1353–1361. [Google Scholar] [CrossRef]
- Yu, J.; Zhong, B.; Xiao, Q.; Du, L.; Hou, Y.; Sun, H.S.; Lu, J.J.; Chen, X. Induction of programmed necrosis: A novel anti-cancer strategy for natural compounds. Pharmacol. Ther. 2020, 214, 107593. [Google Scholar] [CrossRef] [PubMed]
- Chang, W.T.; Wu, S.N. Effectiveness of Columbianadin, a Bioactive Coumarin Derivative, in Perturbing Transient and Persistent INa. Int. J. Mol. Sci. 2021, 22, 621. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.N.; Wu, Y.H.; Chen, B.S.; Lo, Y.C.; Liu, Y.C. Underlying mechanism of actions of tefluthrin, a pyrethroid insecticide, on voltage-gated ion currents and on action currents in pituitary tumor (GH3) cells and GnRH-secreting (GT1-7) neurons. Toxicology 2009, 258, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.H.; Lin, J.F.; Yu, M.C.; Wu, S.N.; Wu, C.L.; Cho, H.Y. Characterization in Potent Modulation on Voltage-Gated Na+ Current Exerted by Deltamethrin, a Pyrethroid Insecticide. Int. J. Mol. Sci. 2022, 23, 14733. [Google Scholar] [CrossRef]
- Huang, H.Y.; Huang, Y.B.; Wu, C.L.; Wu, S.N. Modulatory Impact of Tefluthrin, Telmisartan, and KB-R7943 on Voltage-Gated Na+ Currents. Biophysica 2024, 4, 488–506. [Google Scholar] [CrossRef]
- Hou, S.M.; Hsia, C.W.; Tsai, C.L.; Hsia, C.H.; Jayakumar, T.; Velusamy, M.; Sheu, J.R. Modulation of human platelet activation and in vivo vascular thrombosis by columbianadin: regulation by integrin IIb3 inside-out but not outside-in signals. J. Biomed. Sci. 2020, 27, 60. [Google Scholar] [CrossRef]
- Kang, J.I.; Hong, J.Y.; Choi, J.S.; Lee, S.K. Columbianadin Inhibits Cell Proliferation by Inducing Apoptosis and Necroptosis in HCT116 Colon Cancer Cells. Biomol. Ther. (Seoul) 2016, 24, 320–327. [Google Scholar] [CrossRef] [PubMed]
- Köseoğlu, B.G.; Tanrikulu, S.; Sübay, R.K.; Sencer, S. Anesthesia following overfilling of a root canal sealer into the mandibular canal: a case report. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2006, 101, 803–806. [Google Scholar] [CrossRef] [PubMed]
- Irie, Y.; Keung, W.M. Rhizoma acori graminei and its active principles protect PC-12 cells from the toxic effect of amyloid- peptide. Brain Res. 2003, 963, 282–289. [Google Scholar] [CrossRef]
- Li, J.; Liu, X.; Liu, X.; Zhou, Y.; Wang, L. Interplay between olfactory inputs and seizure regulation: Mechanisms and evidence. Epilepsy Behav. 2026, 177, 110897. [Google Scholar] [CrossRef]
- Müller, M.; Pape, H.C.; Speckmann, E.J.; Gorji, A. Effect of eugenol on spreading depression and epileptiform discharges in rat neocortical and hippocampal tissues. Neuroscience 2006, 140, 743–751. [Google Scholar] [CrossRef]
- Huang, C.W.; Chow, J.C.; Tsai, J.J.; Wu, S.N. Characterizing the effects of Eugenol on neuronal ionic currents and hyperexcitability. Psychopharmacology (Berl) 2012, 221, 575–587. [Google Scholar] [CrossRef]
- Teixeira-Fonseca, J.L.; Santos-Miranda, A.; da Silva, J.B.; Marques, L.P.; Joviano-Santos, J.V.; Nunes, P.I.C.; Roman-Campos, D.; Gondim, A.N.S. Eugenol interacts with cardiac sodium channel and reduces heart excitability and arrhythmias. Life Sci. 2021, 282, 119761. [Google Scholar] [CrossRef]
- Yang, B.H.; Piao, Z.G.; Kim, Y.B.; Lee, C.H.; Lee, J.K.; Park, K.; Kim, J.S.; Oh, S.B. Activation of vanilloid receptor 1 (VR1) by eugenol. J. Dent. Res. 2003, 82, 781–785. [Google Scholar] [CrossRef]
- Chung, G.; Rhee, J.N.; Jung, S.J.; Kim, J.S.; Oh, S.B. Modulation of CaV2.3 calcium channel currents by eugenol. J. Dent. Res. 2008, 87, 137–141. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.W.; Chow, J.C.; Tsai, J.J.; Wu, S.N. Characterizing the effects of Eugenol on neuronal ionic currents and hyperexcitability. Psychopharmacology (Berl) 2012, 221, 575–587. [Google Scholar] [CrossRef] [PubMed]
- Li, H.F.; Chen, S.A.; Wu, S.N. Evidence for the stimulatory effect of resveratrol on Ca2+-activated K+ current in vascular endothelial cells. Cardiovasc. Res. 2000, 45, 1035–45. [Google Scholar] [CrossRef]
- Zhang, L.X.; Li, C.X.; Kakar, M.U.; Khan, M.S.; Wu, P.F.; Amir, R.M.; Dai, D.F.; Naveed, M.; Li, Q.Y.; Saeed, M.; Shen, J.Q.; Rajput, S.A.; Li, J.H. Resveratrol (RV): A pharmacological review and call for further research. Biomed. Pharmacother. 2021, 143, 112164. [Google Scholar] [CrossRef]
- Caiati, C; Jirillo, E. Cellular and Molecular Bases for the Application of Polyphenols in the Prevention and Treatment of Cardiovascular Disease. Diseases 2025, 13, 221. [Google Scholar] [CrossRef]
- Rao, Y.L.; Ganaraja, B.; Joy, T.; Pai, M.M.; Ullal, S.D.; Murlimanju, B.V. Neuroprotective effects of resveratrol in Alzheimer's disease. Front. Biosci. (Elite Ed) 2020, 12, 139–149. [Google Scholar] [CrossRef]
- Kim, H.J.; Kim, I.K.; Song, W.; Lee, J.; Park, S. The synergic effect of regular exercise and resveratrol on kainate-induced oxidative stress and seizure activity in mice. Neurochem. Res. 2013, 38, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Almeida, E.R.; Lima-Rezende, C.A.; Schneider, S.E.; Garbinato, C.; Pedroso, J.; Decui, L.; Aguiar, G.P.S.; Müller, L.G.; Oliveira, J.V.; Siebel, A.M. Micronized Resveratrol Shows Anticonvulsant Properties in Pentylenetetrazole-Induced Seizure Model in Adult Zebrafish. Neurochem. Res. 2021, 46, 241–251. [Google Scholar] [CrossRef]
- Wang, Y.J.; Chan, M.H.; Chen, L.; Wu, S.N.; Chen, H.H. Resveratrol attenuates cortical neuron activity: roles of large conductance calcium-activated potassium channels and voltage-gated sodium channels. J. Biomed. Sci. 2016, 23, 47. [Google Scholar] [CrossRef]
- Granados-Soto, V.; Arguelles, C.F.; Ortiz, M.I. The peripheral antinociceptive effect of resveratrol is associated with activation of potassium channels. Neuropharmacology 2002, 43, 917–923. [Google Scholar] [CrossRef]
- Tang, C.; Wu, B.; Wu, J.; Zhang, Z.; Yu, B. Novel strategies using total gastrodin and gastrodigenin, or total gastrodigenin for quality control of Gastrodia elata. Molecules 2018, 23, 270. [Google Scholar] [CrossRef]
- Liu, Y.; Gao, J.; Peng, M.; Meng, H.; Ma, H.; Cai, P.; Xu, Y.; Zhao, Q.; Si, G. A review on central nervous system effect of gastrodin. Front. Pharmacol. 2018, 9, 24. [Google Scholar] [CrossRef]
- Yang, C.S.; Chiu, S.C.; Liu, P.Y.; Wu, S.N.; Lai, M.C.; Huang, C.W. Gastrodin alleviates seizure severity and neuronal excitotoxicities in the rat lithium-pilocarpine model of temporal lobe epilepsy via enhancing GABAergic transmission. J. Ethnopharmacol. 2021, 269, 113751. [Google Scholar] [CrossRef] [PubMed]
- Dai, J.; Shen, H.L.; Li, J.; Zhou, Y.; Dong, Z.X.; Zhu, X.Y. Gastrodin Attenuates Neuroinflammation and Injury in Young Rats with LiCl/Pilocarpine-Induced Status Epilepticus. Biochem. Genet. 2025, 63, 5067–5082. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Liu, W.; Lu, M.; Xu, Q. Neuropharmacological effects of Gastrodia elata Blume and its active ingredients. Front. Neurol. 2025, 16, 1574277. [Google Scholar] [CrossRef]
- Yang, C.S.; Lai, M.C.; Liu, P.Y.; Lo, Y.C.; Huang, C.W.; Wu, S.N. Characterization of the Inhibitory Effect of Gastrodigenin and Gastrodin on M-type K+ Currents in Pituitary Cells and Hippocampal Neurons. Int. J. Mol. Sci. 2019, 21, 117. [Google Scholar] [CrossRef]
- Ahmad, H.; Rajagopal, K. Pharmacology of Pterocarpus marsupium Roxib. Medicinal Plant Research 2015, 5, 1–6. [Google Scholar]
- Tan, K.; Chen, P.W.; Li, S.; Ke, T.M.; Lin, S.H.; Yang, C.C. Pterostilbene inhibits lung squamous cell carcinoma growth in vitro and in vivo by inducing S phase arrest and apoptosis. Oncol. Lett. 2019, 18, 1631–1640. [Google Scholar] [CrossRef]
- Yu, C.L.; Yang, S.F.; Hung, T.W.; Lin, C.L.; Hsieh, Y.H.; Chiou, H.L. Inhibition of eIF2alpha dephosphorylation accelerates pterostilbene-induced cell death in human hepatocellular carcinoma cells in an ER stress and autophagy-dependent manner. Cell Death Dis. 2019, 10, 418. [Google Scholar] [CrossRef]
- Zhang, T.; Li, B.; Feng, Q.; Xu, Z.; Huang, C.; Wu, H.; Chen, Z.; Hu, L.; Gao, L.; Liu, P.; Yang, G.; Zhang, H.; Lu, K.; Li, T.; Tao, Y.; Wu, X.; Shi, J.; Zhu, W. DCZ0801, a novel compound, induces cell apoptosis and cell cycle arrest via MAPK pathway in multiple myeloma. Acta Biochim. Biophys. Sin. (Shanghai) 2019, 51, 517–523. [Google Scholar] [CrossRef] [PubMed]
- So, E.C.; Gao, Z.H.; Ko, S.Y.; Wu, S.N. Characterization of Effectiveness in Concerted Ih Inhibition and IK(Ca) Stimulation by Pterostilbene (Trans-3,5-dimethoxy-4'-hydroxystilbene), a Stilbenoid. Int. J. Mol. Sci. 2020, 21, 357. [Google Scholar] [CrossRef]
- Wu, S.N.; Chern, J.H.; Shen, S.; Chen, H.H.; Hsu, Y.T.; Lee, C.C.; Chan, M.H.; Lai, M.C.; Shie, F.S. Stimulatory actions of a novel thiourea derivative on large-conductance, calcium-activated potassium channels. J. Cell. Physiol. 2017, 232, 3409–3421. [Google Scholar] [CrossRef]
- Sobhani, M.; Abbas-Mohammadi, M.; Ebrahimi, S.N.; Aliahmadi, A. Tracking leading anti-Candida compounds in plant samples; Plumbago europaea. Iran. J. Microbiol. 2018, 10, 187–193. [Google Scholar]
- Liu, Y.; Cai, Y.; He, C.; Chen, M.; Li, H. Anticancer Properties and Pharmaceutical Applications of Plumbagin: A Review. Am. J. Chin. Med. 2017, 45, 423–441. [Google Scholar] [CrossRef]
- Roy, R.; Mandal, S.; Chakrabarti, J.; Saha, P.; Panda, C.K. Downregulation of Hyaluronic acid-CD44 signaling pathway in cervical cancer cell by natural polyphenols Plumbagin, Pongapin and Karanjin. Mol. Cell. Biochem. 2021, 476, 3701–3709. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Zhao, S.; Zheng, W.; Fu, H.; Wu, T.; Hu, F. Plumbagin attenuated oxygen-glucose deprivation/reoxygenation-induced injury in human SH-SY5Y cells by inhibiting NOX4-derived ROS-activated NLRP3 inflammasome. Biosci. Biotechnol. Biochem. 2020, 84, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Thakor, N.; Janathia, B. Plumbagin: A Potential Candidate for Future Research and Development. Curr. Pharm. Biotechnol. 2022, 23, 1800–1812. [Google Scholar] [CrossRef]
- Bhargava, S.K. Effect of testosterone replacement therapy on quantitative spermatogenesis following plumbagin treatment in immature rats. Acta Eur. Fertil. 1986, 17, 217–219. [Google Scholar]
- Shen, A.Y.; Huang, M.H.; Teng, C.M.; Wang, J.S. Inhibition of 2-P-mercaptophenyl-1,4-naphthoquinone on human platelet function. Life Sci. 1999, 65, 45–53. [Google Scholar] [CrossRef]
- Huang, M.H.; Wu, S.N.; Chen, C.P.; Shen, A.Y. Inhibition of Ca2+-activated and voltage-dependent K+ currents by 2-mercaptophenyl-1,4-naphthoquinone in pituitary GH3 cells: contribution to its antiproliferative effect. Life Sci. 2002, 70, 1185–1203. [Google Scholar] [CrossRef]
- Chen, L.; Cho, H.Y.; Chuang, T.H.; Ke, T.L.; Wu, S.N. The Effectiveness of Isoplumbagin and Plumbagin in Regulating Amplitude, Gating Kinetics, and Voltage-Dependent Hysteresis of erg-mediated K+ Currents. Biomedicines 2022, 10, 780. [Google Scholar] [CrossRef] [PubMed]
- Tsao, Y.C.; Chang, Y.J.; Wang, C.H.; Chen, L. Discovery of Isoplumbagin as a Novel NQO1 Substrate and Anti-Cancer Quinone. Int. J. Mol. Sci. 2020, 21, 4378. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.Y.; Chang, W.T.; Wu, S.N. Characterization of the synergistic inhibition of IK(erg) and IK(DR) by ribociclib, a cyclin-dependent kinase 4/6 inhibitor. Int. J. Mol. Sci. 2020, 21, 8078. [Google Scholar] [CrossRef]
- Stojilkovic, S.S.; Tabak, J.; Bertram, R. Ion channels and signaling in the pituitary gland. Endocr. Rev. 2010, 31, 845–915. [Google Scholar] [CrossRef]
- Vandenberg, J.I.; Perry, M.D.; Perrin, M.J.; Mann, S.A.; Ke, Y.; Hill, A.P. hERG K+ channels: structure, function, and clinical significance. Physiol. Rev. 2012, 92, 1393–1478. [Google Scholar] [CrossRef] [PubMed]
- Martinson, A.S.; Van Rossum, D.B.; Diatta, F.H.; Layden, M.J.; Rhodes, S.A.; Martindale, M.Q.; Jegla, T.J. Functional evolution of Erg potassium channel gating reveals an ancient origin for IKr. Proc. Natl. Acad. Sci. U.S.A. 2014, 111, 5712–5717. [Google Scholar] [CrossRef]
- Min, J.Y.; Lv, Y.; Mao, L.; Gong, Y.Y.; Gu, Q.; Wei, F. A rodent model of anterior ischemic optic neuropathy (AION) based on laser photoactivation of verteporfin. BMC Ophthalmol. 2018, 18, 304. [Google Scholar] [CrossRef]
- Iacono, P.; Toto, L.; Eliana, C.; Varano, M.; Parravano, M.C. Pharmacotherapy of central serous chorioretinopathy: review of the current treatments. Curr. Pharm. Des. 2019, 24, 4864–4873. [Google Scholar] [CrossRef]
- Isildak, H.; Schwartz, S.G.; Flynn, H.W. Pharmacotherapy of myopic choroidal neovascularization. Curr. Pharm. Des. 2019, 24, 4853–4859. [Google Scholar] [CrossRef]
- Tekrony, A.D.; Kelly, N.M.; Fage, B.A.; Cramb, D.T. Photobleaching kinetics of verteporfin and lemuteporfin in cells and optically trapped multilamellar vesicles using two-photon excitation. Photochem. Photobiol. 2011, 87, 853–861. [Google Scholar] [CrossRef]
- Nemes, A.; Fortmann, T.; Poeschke, S.; Greve, B.; Prevedello, D.; Santacroce, A.; Stummer, W.; Senner, V.; Ewelt, C. 5-ALA fluorescence in native pituitary adenoma cell lines: resection control and basis for photodynamic therapy (PDT)? PLoS One 2016, 11, e0161364. [Google Scholar] [CrossRef]
- Valenzeno, DP; Tarr, M. Calcium as a modulator of photosensitized killing of H9c2 cardiac cells. Phtochem. Photobiol. 2001, 74, 605–610. [Google Scholar] [CrossRef] [PubMed]
- Karacorlu, M.; Karacorlu, S.; Ozdemir, H. Nonarteritic anterior ischemic optic neuropathy after photodynamic therapy for choroidal neovascularization. Jpn. J. Ophthalmol. 2004, 48, 424–426. [Google Scholar] [CrossRef]
- Huang, M.H.; Liu, P.Y.; Wu, S.N. Characterization of Perturbing Actions by Verteporfin, a Benzoporphyrin Photosensitizer, on Membrane Ionic Currents. Front. Chem. 2019, 7, 566. [Google Scholar] [CrossRef] [PubMed]
- Gibault, F.; Corvaisier, M.; Bailly, F.; Huet, G.; Melnyk, P.; Cotelle, P. Non-photoinduced biological properties of verteporfin. Curr. Med. Chem. 2016, 23, 1171–1184. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Hu, Y. Photodynamic therapy for an iris metastasis from pulmonary adenocarcinoma. Photodiagnosis Photodyn. Ther. 2017, 20, 246–247. [Google Scholar] [CrossRef] [PubMed]
- Marks, P.V.; Belchetz, P.E.; Saxena, A.; Igbaseimokumo, U.; Thomson, S.; Nelson, M.; Stringer, M.R.; Holroyd, J.A.; Brown, S.B. Effect of photodynamic therapy on recurrent pituitary adenomas: clinical phase I/II trial--an early report. Br. J. Neurosurg. 2000, 14, 317–325. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.N.; Wang, Y.J.; Gao, Z.H.; Liutkevičienė, R.; Rovite, V. Recent Advances in Ionic Mechanisms in Pituitary Cells: Implications for Electrophysiological and Electropharmacological Research. J. Clin. Med. 2025, 14, 3117. [Google Scholar] [CrossRef]
- Lin, C.Y.; Gao, Z.H.; Cheung, C.W.; So, E.C.; Wu, S.N. Modulation of Voltage-Gated Na+ Channel Currents by Small Molecules: Effects on Amplitude and Gating During High-Frequency Stimulation. Sci. Pharma 2025, 93, 33. [Google Scholar] [CrossRef]
- Tsuchiya, H. Membrane Interactions of Phytochemicals as Their Molecular Mechanism Applicable to the Discovery of Drug Leads from Plants. Molecules 2015, 20, 18923–66. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



