Submitted:
03 March 2026
Posted:
04 March 2026
You are already at the latest version
Abstract
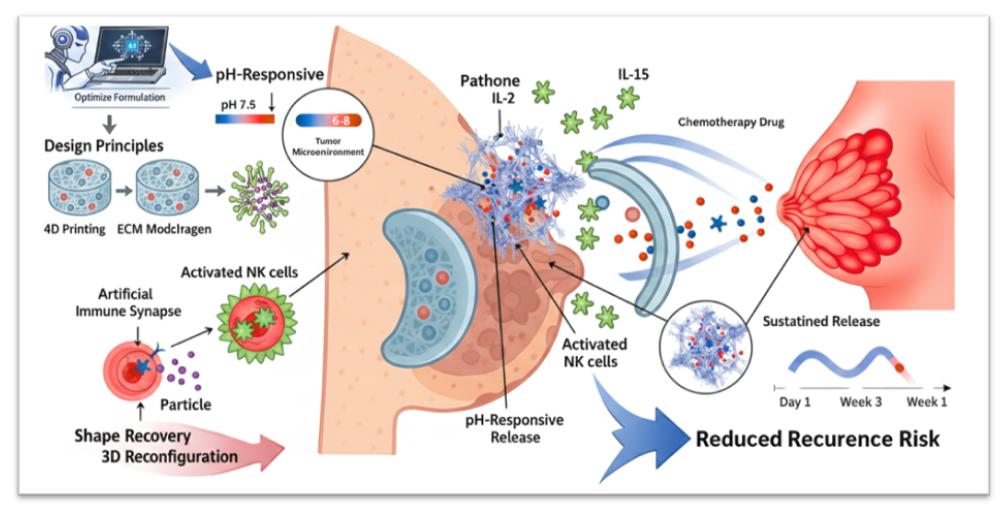
Keywords:
1. Introduction
1.1. Global Burden and Therapeutic Limitations in Breast Cancer
1.2. Tumour Microenvironment as a Therapeutic Barrier and Opportunity
1.3. Emergence of 4D Printing in Drug Delivery
1.4. Rationale for Spatiotemporal Immunotherapy via 4D Nanofiber Implants
1.5. Scope of This Review
1.6. Literature Search Strategy and Study Selection Framework
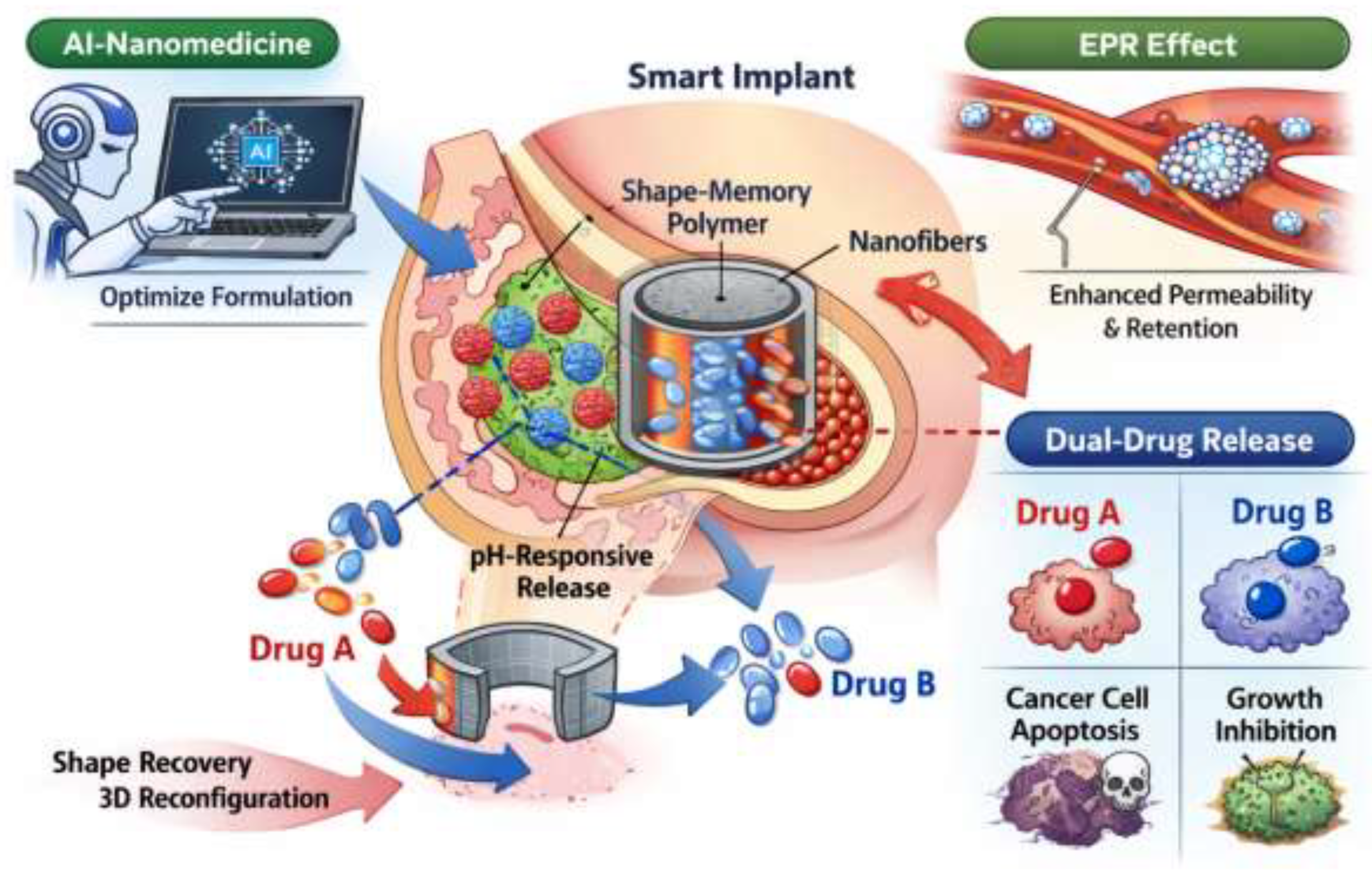
2. Molecular and Metabolic Drivers of Breast Cancer: Implications for Microenvironment-Responsive Therapy
2.1. Carcinogenesis and Oncogenic Signalling in Breast Cancer
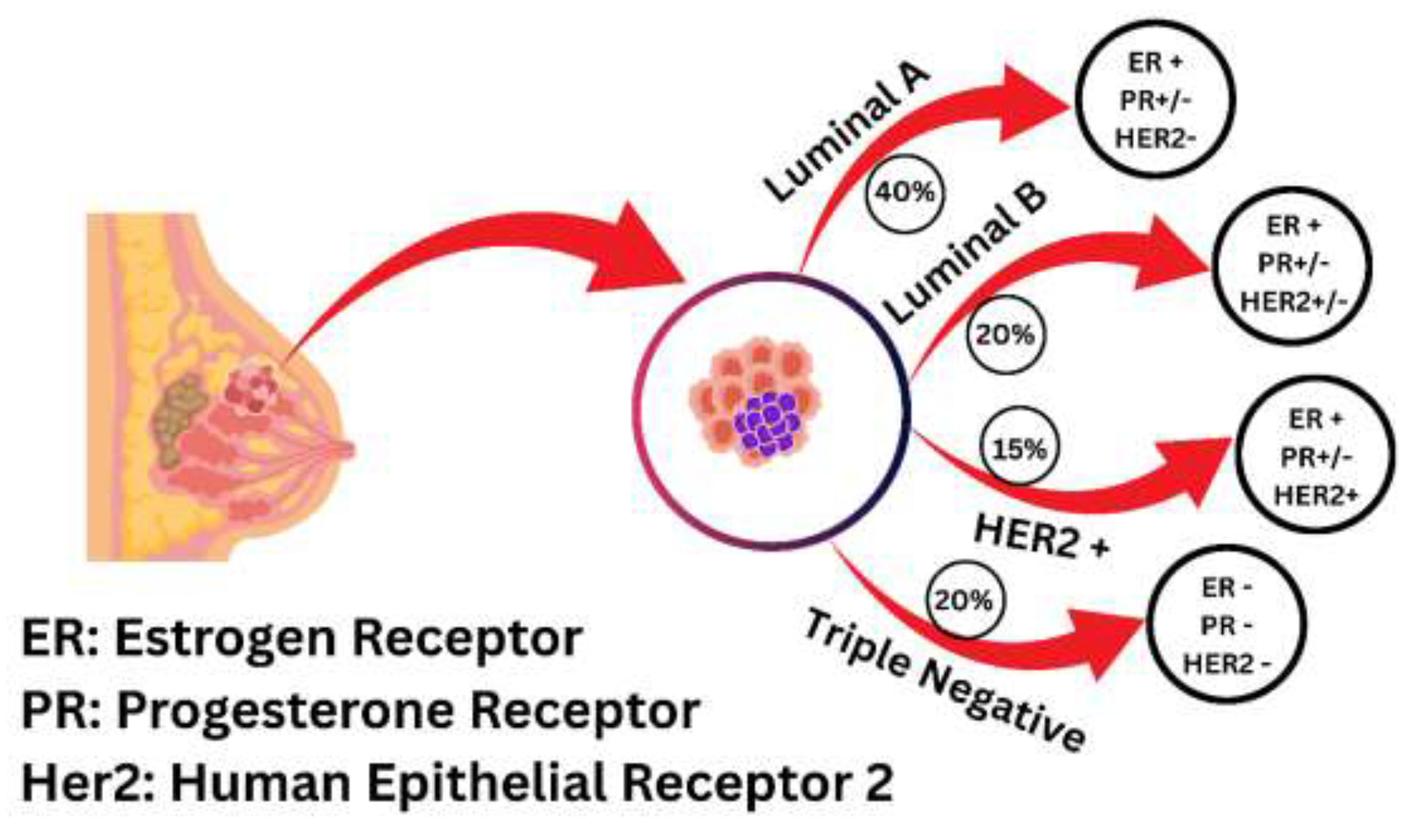
2.2. Metabolic Reprogramming and Acidic Tumour Microenvironment
2.3. Implications for pH-Responsive 4D-Printed Implant Design
3. Fundamentals of 4D Printing in Drug Delivery
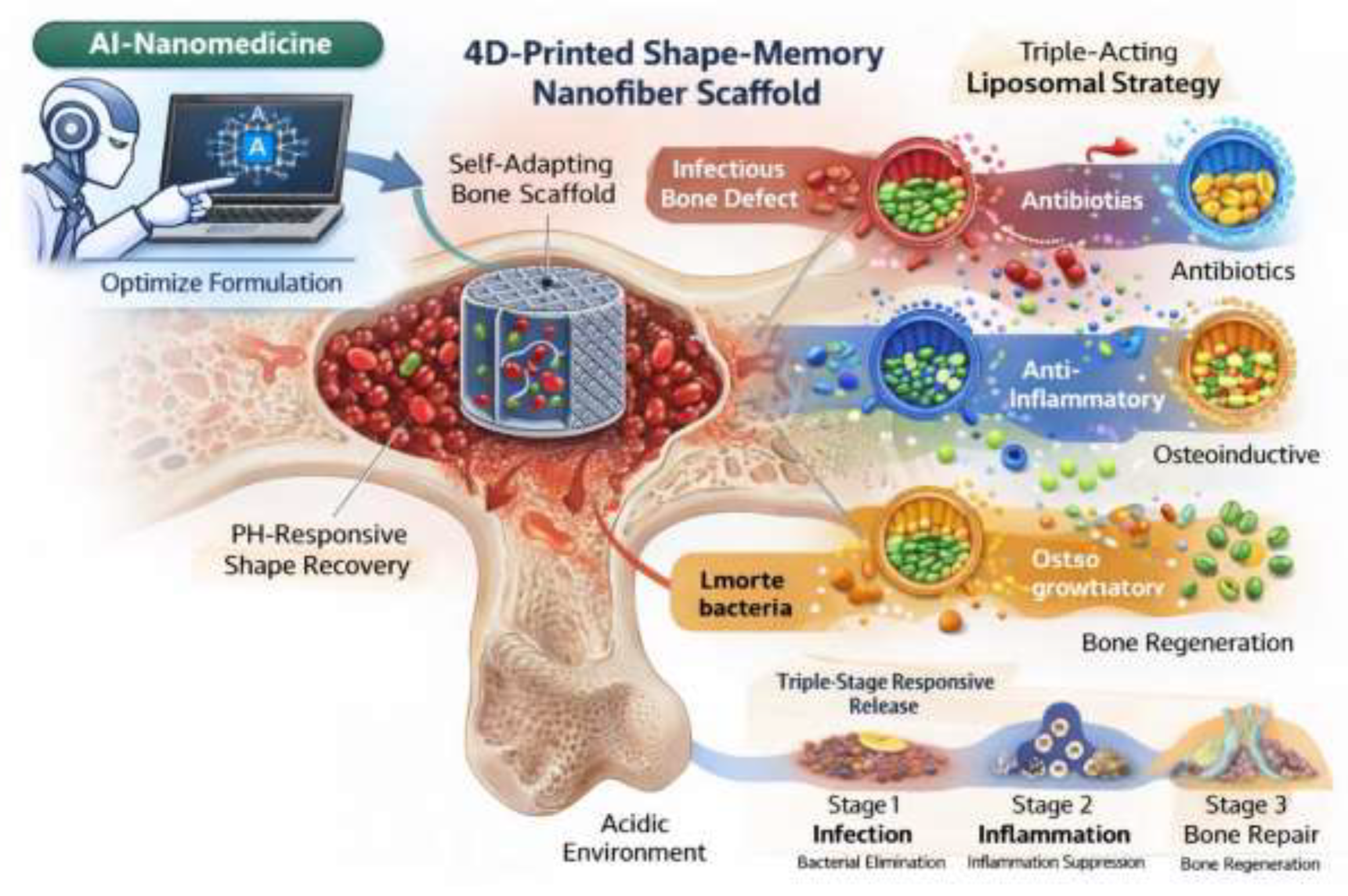
3.1. Shape-Memory Mechanisms and Programming Strategies
| Material type | Mechanism of response | Representative biomedical application | Key advantages | Primary limitations | Reference |
| SMPs | Thermally(or light) activated shape recovery | self-expanding stents, deployable scaffolds, and minimally invasive implants | strong shape recovery, tunable transition temp, printable by FDM/SLA | fatigue over repeated cycles, limited biodegradability for some SMPs | [37] |
| thermo-/photoresponsive hydrogels (e.g., PNIPAM blends) | phase transition (swelling/deswelling) with temp or light (photothermal triggers when loaded with GNRs/UCNPs) | on-demand drug release, wound dressings, and dynamic scaffolds | excellent tissue-like hydration; compatible with cell encapsulation | lower mechanical strength; may require reinforcement | [38,39] |
| pH-responsive hydrogels/polyelectrolytes | Protonation/deprotonation alters swelling or bond cleavage | tumor-targeted DDS, GI tract targeting | site-specific release (tumor/GI) | narrow responsive window; variability in vivo | [40] |
| biopolymers/biodegradable composites (PLA, PCL, bioceramics) | enzyme/hydrolysis mediated degradation → structural change or release | biodegradable implants, bone scaffolds | established biocompatibility; tunable degradation | slower response, mechanical trade-offs | [41] |
| photochromic/photocleavable chemistries (azobenzene, spiropyran, o-nitrobenzyl esters) | photoisomerization or bond cleavage on irradiation | light-triggered release, microactuation in 4D constructs |
high spatial/temporal control (when light accessible) | limited tissue penetration (UV), possible phototoxicity | [38,39] |
| magnetically responsive composites (Fe3O4, etc.) | external magnetic fields induce force/heat → actuation or hyperthermia | remote actuation (implants), magnetically guided carriers | noncontact remote control; reversible actuation | heating risk; long-term biosafety of magnetic NPs | [42] |
3.2. Conceptual Evolution from 3D to 4D Printing
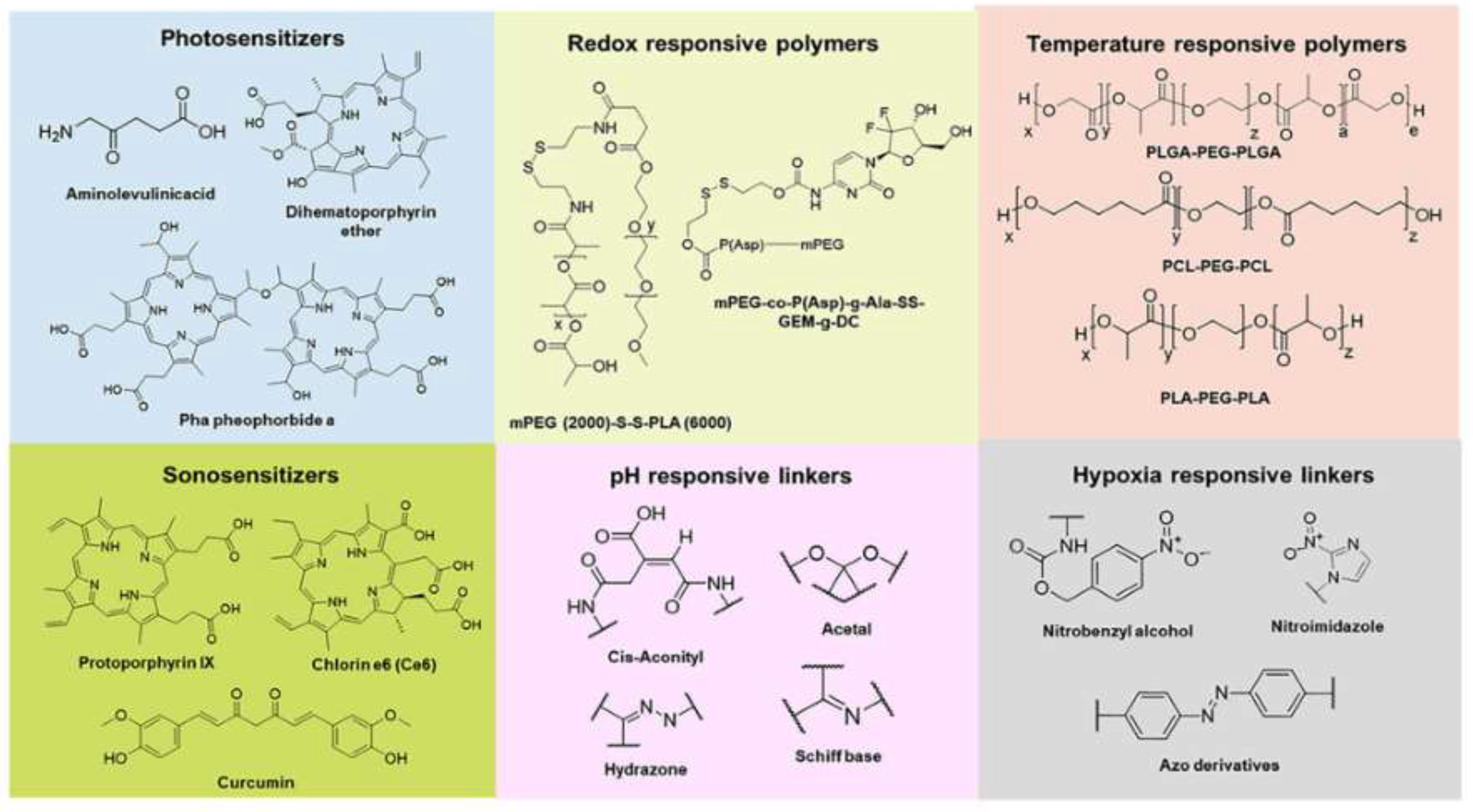
3.3. Stimuli-Responsive Biomaterials for 4D Systems
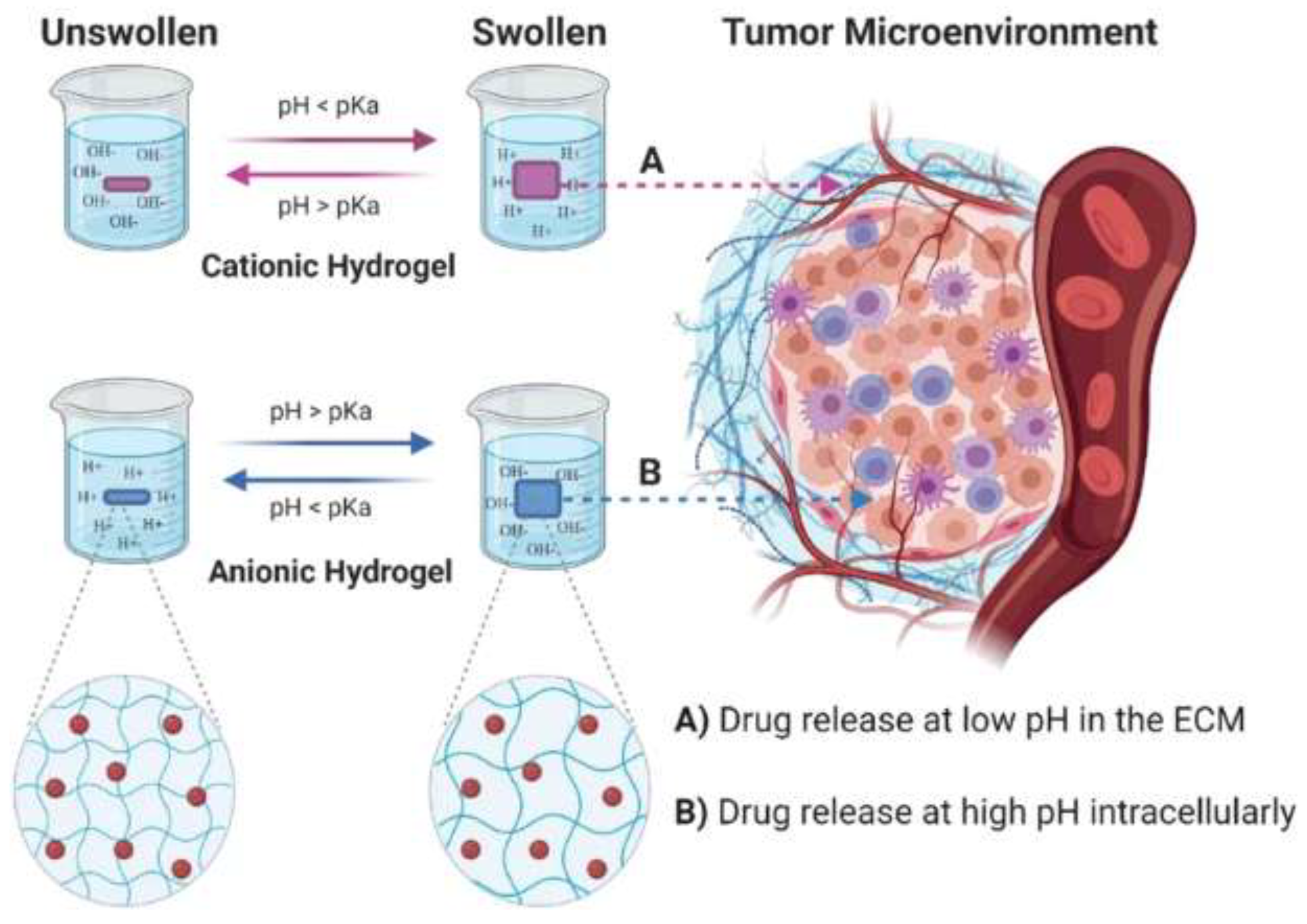
3.3.1. pH-Responsive Polymers
3.4. Mechanisms of Structural Transformation in 4D Implants
3.4.1. Swelling-Induced Porosity Modulation
3.4.2. Acid-Catalysed Hydrolytic Degradation
3.4.3. Shape Memory and Morphological Adaptation
3.4.4. Multistage Release Programming
3.5. Fabrication Technologies for 4D Nanofiber Implants
3.5.1. Extrusion-Based 4D Printing
3.5.2. Electrospinning 4D Hybrid Systems
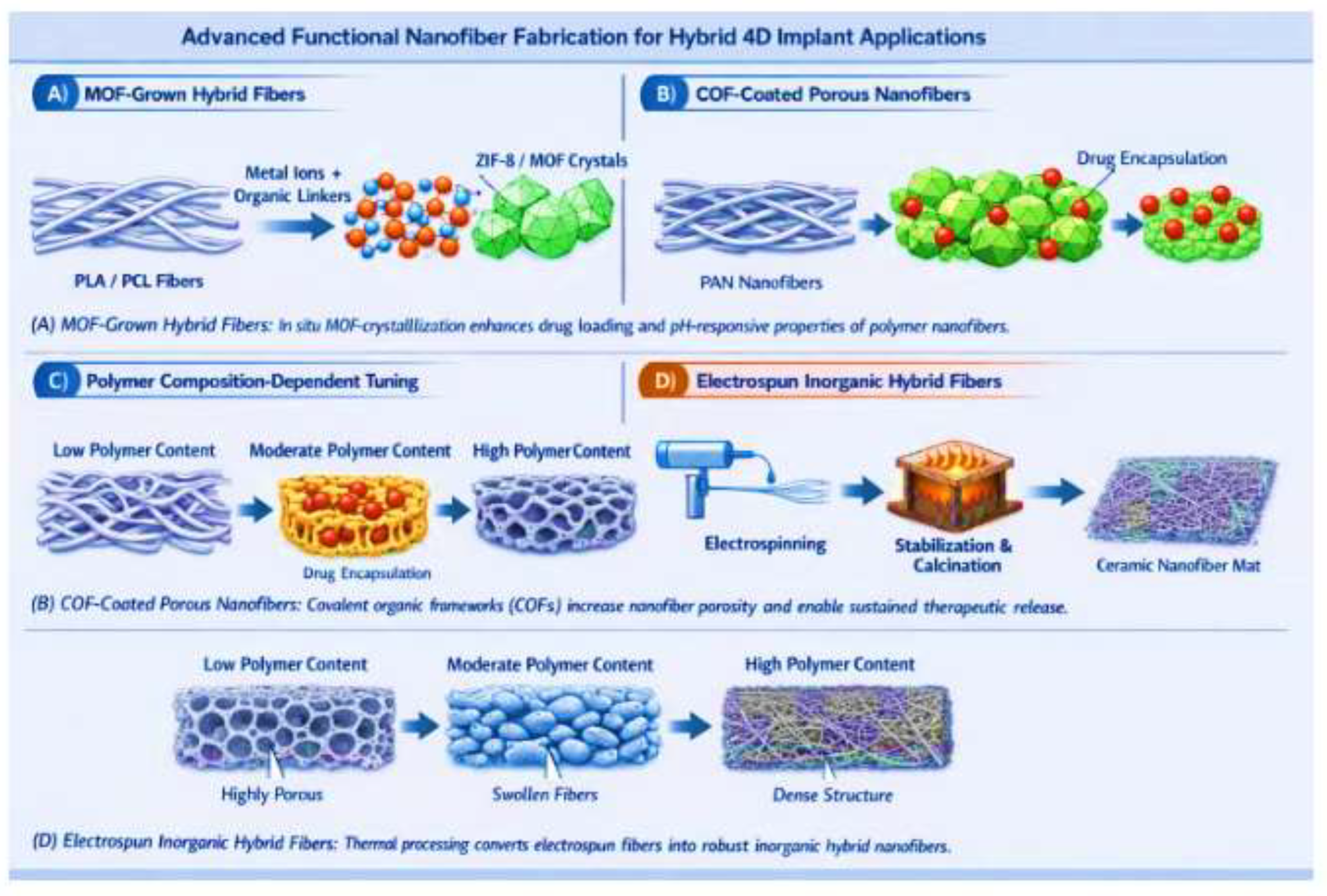
3.5.3. Digital Light Processing (DLP) and Stereolithography
3.6. Advantages of 4D Printing in Oncology-Oriented Drug Delivery
3.7. Limitations and Current Knowledge Gaps
- Limited long-term in vivo data
- Variability in tumour pH among patients
- Reproducibility of transformation kinetics
- Protein stability during printing processes
- Scale-up feasibility
4. Design Principles of pH-Responsive Nanofiber Implants for Breast Cancer Therapy
4.1. Rational Polymer Selection for Microenvironment-Responsive Implants
4.2. Engineering Nanofiber Architecture for Controlled Release
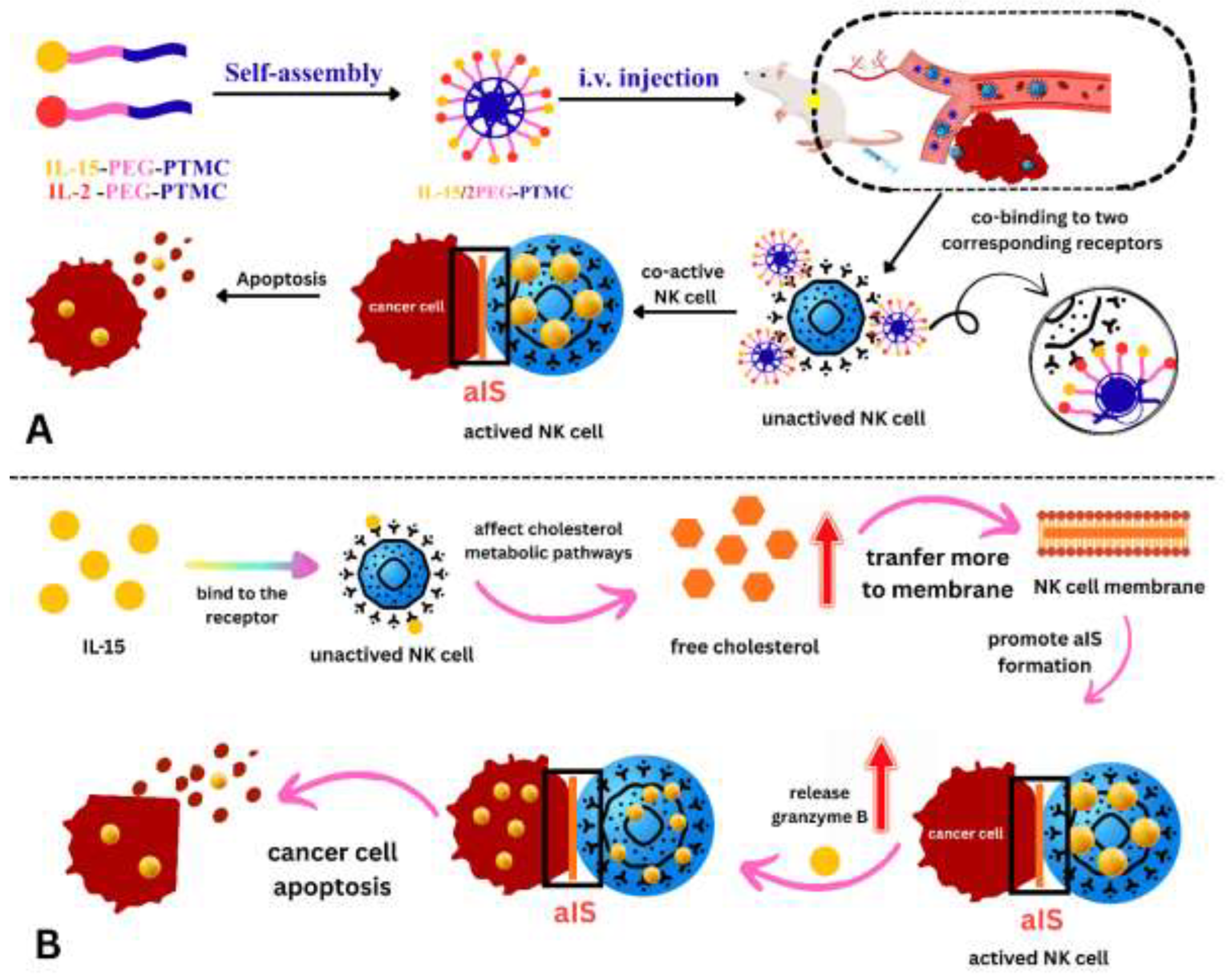
4.3. Cytokine Nano-Assembly and Stabilisation Strategies
4.4. Mechanistic Basis of NK Cell Activation and Artificial Immune Synapse Formation
4.5. Spatiotemporal and Phased Release Strategies in 4D-Printed Implants
4.6. Mechanical and Conformational Adaptation to Surgical Cavities
4.7. Critical Design Considerations and Trade-Offs
5. Tumor Microenvironment Modulation by 4D-Printed pH-Responsive Implants
5.1. The Breast Tumor Microenvironment: A Dynamic Immunosuppressive Niche
5.2. Immune-Suppressive Tumour Microenvironment in Breast Cancer
5.2.1. Antigenicity vs. Immunogenicity in Breast Cancer: Implications for Checkpoint Therapy
5.2.2. T-Cell Exhaustion and Resistance to Immune Checkpoint Blockade
5.2.3. Relevance to 4D-Printed pH-Responsive Nanofiber Implants
5.3. Exploiting Acidic pH as a Therapeutic Activation Signal
5.4. Mechanisms of pH-Responsive Material Activation
5.5. Localised NK Cell Activation and Immune Synapse Enhancement
5.6. Modulation of Tumour-Associated Macrophages and Immune Reprogramming
5.7. ECM Remodelling and Improved Immune Penetration
5.8. Metabolic Rewiring and Cholesterol Dynamics in Immune Activation
5.9. Integrated Microenvironment Reprogramming: From Suppression to Activation
6. Spatiotemporal Immunotherapy Programming and Sequential Release Strategies
6.1. Rationale for Spatiotemporal Control in Cancer Immunotherapy
6.2. Multiphase Release Design: A Three-Stage Programming Model
6.2.1. Phase I: Immune Priming and Microenvironment Conditioning.
6.2.2. Phase II: Sustained NK Cell Activation.
6.2.3. Phase III: Immune Memory Formation and Resolution.
6.3. Sequential Multi-Agent Loading Strategies
6.4. Integration with Immune Checkpoint Blockade
6.5. Mathematical and Engineering Modelling of Release Kinetics
6.6. Avoiding Immune Exhaustion and Cytokine Overload
6.7. Clinical Implications of Spatiotemporal Programming
7. Translational and Regulatory Challenges
7.1. Manufacturing Scalability and GMP Compliance
7.2. Sterilisation of Cytokine-Loaded Implants
7.3. Stability and Storage Considerations
7.4. Regulatory Classification: Combination Product Complexity
7.5. Preclinical Evaluation Challenges
7.6. Safety Considerations and Immune-Related Risks
7.7. Economic and Clinical Implementation Barriers
7.8. Bridging the Gap Between Innovation and Clinical Reality
8. Future Perspectives and Emerging Directions
8.1. Toward Personalised Microenvironment-Responsive Implants
8.2. AI-Guided Optimisation of Polymer and Release Profiles
8.3. Integration of Biosensing and Feedback-Responsive Systems
8.4. Expanding Beyond Cytokines: Gene Editing and Advanced Biologics
8.5. Combination Strategies with Immune Checkpoint Modulation
8.6. Ethical and Long-Term Safety Considerations
| Target Pathway | Representative Agents | Mechanism of TME Modulation | Clinical Status | Main Challenges | Potential Integration with 4D Implants | Reference |
| FAK Signaling | FAK inhibitors ± immune checkpoint inhibitors | Inhibition of focal adhesion kinase reduces stromal fibrosis and enhances immune infiltration. | Preclinical / Early clinical | Limited clinical validation; stromal heterogeneity | Localised co-delivery with cytokines to reduce ECM stiffness and improve immune cell penetration | [163] |
| VEGFR / PDGFR / FGFR | Nintedanib ± anti-PD-1 | Suppresses angiogenesis and collagen deposition, improving CD8⁺ T-cell infiltration. | Preclinical | The translation of breast cancer is unclear | Sequential release to normalise the vasculature before the NK activation phase | [164] |
| TREM2–IL-1β Axis | TREM2 modulation; IL-1β inhibitors | Regulates macrophage-driven inflammatory signalling and TME immunosuppression. | Preclinical | Risk of excessive inflammation | Controlled, localised release to reprogram TAMs while limiting systemic cytokine surge | [165] |
| KRAS / Oncogenic Signalling | KRAS inhibitors (e.g., MRTX-class agents) | Suppresses oncogenic signalling and reverses CAF-mediated drug resistance. | Preclinical / Early clinical | Breast cancer subtype specificity | Combined local immunotherapy with systemic targeted therapy | [166] |
| Hyaluronan / ECM Density | PEGylated hyaluronidase (PEGPH20) | Degrades hyaluronan to reduce interstitial pressure and improve drug penetration. | Clinical (Phase III, other cancers) | Thromboembolic risk; no OS benefit | Localised ECM modulation via pH-triggered enzyme release to enhance immune infiltration | [167] |
| CXCR4–CXCL12 Axis | Motixafortide ± pembrolizumab | Enhances T-cell trafficking and reduces the number of immunosuppressive myeloid cells. | Clinical (Phase II) | Modest survival benefit; biomarker dependence | Incorporation into a multi-phase nanofiber system for immune cell recruitment | [168] |
| VEGFR | Axitinib ± chemotherapy | Inhibits tumour angiogenesis to enhance therapeutic delivery. | Clinical (Phase III) | Resistance to anti-angiogenic therapy | Short-term vascular normalisation phase prior to cytokine release | [169] |
| Mitochondrial Metabolism | CPI-613 (devimistat) | Induces metabolic stress in tumour cells. | Early clinical | Limited breast cancer data | Sequential combination to sensitise tumour cells before immune activation | [170] |
| Immune Activation (Cytokine Axis) | IL-15 + IL-2 nano-assemblies | Enhances NK activation and artificial immune synapse formation. | Investigational | Systemic toxicity if not localised | Core component of 4D implant spatiotemporal immunotherapy | [171] |
9. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bray, F.; Laversanne, M.; Sung, H.; Ferlay, J.; Siegel, R.L.; Soerjomataram, I.; Jemal, A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: a cancer journal for clinicians 2024, 74, 229–263. [Google Scholar] [CrossRef]
- Cai, Y.; Dai, F.; Ye, Y.; Qian, J. The global burden of breast cancer among women of reproductive age: a comprehensive analysis. Scientific Reports 2025, 15, 9347. [Google Scholar] [CrossRef]
- Kim, J.; Harper, A.; McCormack, V.; Sung, H.; Houssami, N.; Morgan, E.; Mutebi, M.; Garvey, G.; Soerjomataram, I.; Fidler-Benaoudia, M.M. Global patterns and trends in breast cancer incidence and mortality across 185 countries. Nature medicine 2025, 31, 1154–1162. [Google Scholar] [CrossRef]
- Deng, T.; Zi, H.; Guo, X.P.; Luo, L.S.; Yang, Y.L.; Hou, J.X.; Zhou, R.; Yuan, Q.Q.; Liu, Q.; Huang, Q. Global, regional, and national burden of breast cancer, 1990–2021, and projections to 2050: A systematic analysis of the global burden of disease study 2021. Thoracic Cancer 2025, 16, e70052. [Google Scholar] [CrossRef]
- Sha, R.; Kong, X.-m.; Li, X.-y.; Wang, Y.-b. Global burden of breast cancer and attributable risk factors in 204 countries and territories, from 1990 to 2021: results from the Global Burden of Disease Study 2021. Biomarker Research 2024, 12, 87. [Google Scholar] [CrossRef]
- Nagpal, D.; Verma, R.; Mittal, V.; Jeandet, P.; Kaushik, D. Targeted therapies against breast cancer: Clinical perspectives, obstacles and new opportunities. Journal of Drug Delivery Science and Technology 2023, 89, 105049. [Google Scholar] [CrossRef]
- Marei, H.E.; Bedair, K.; Hasan, A.; Al-Mansoori, L.; Caratelli, S.; Sconocchia, G.; Gaiba, A.; Cenciarelli, C. Current status and innovative developments of CAR-T-cell therapy for the treatment of breast cancer. Cancer Cell International 2025, 25, 3. [Google Scholar] [CrossRef]
- Harris, M.A.; Savas, P.; Virassamy, B.; O’Malley, M.M.; Kay, J.; Mueller, S.N.; Mackay, L.K.; Salgado, R.; Loi, S. Towards targeting the breast cancer immune microenvironment. Nature Reviews Cancer 2024, 24, 554–577. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Bejarano, O.H.; Parra-López, C.; Patarroyo, M.A. A review concerning the breast cancer-related tumour microenvironment. Critical Reviews in Oncology/Hematology 2024, 199, 104389. [Google Scholar] [CrossRef] [PubMed]
- Monzavi-Karbassi, B.; Kelly, T.; Post, S.R. The tumor microenvironment and immune response in breast cancer. 2024, 25, 914. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Wang, J.; Li, G. 3D/4D printing of stimuli-responsive polymers in biomedical engineering: materials, stimulations, and applications. Materials Science and Engineering: R: Reports 2025, 166, 101071. [Google Scholar] [CrossRef]
- Suryavanshi, P.; Wang, J.; Duggal, I.; Maniruzzaman, M.; Banerjee, S. Four-dimensional printed construct from temperature-responsive self-folding feedstock for pharmaceutical applications with machine learning modeling. Pharmaceutics 2023, 15, 1266. [Google Scholar] [CrossRef]
- Antezana, P.E.; Municoy, S.; Ostapchuk, G.; Catalano, P.N.; Hardy, J.G.; Evelson, P.A.; Orive, G.; Desimone, M.F. 4D printing: the development of responsive materials using 3D-printing technology. Pharmaceutics 2023, 15, 2743. [Google Scholar] [CrossRef]
- Guo, Y.; Li, Y.; Zhang, M.; Ma, R.; Wang, Y.; Weng, X.; Zhang, J.; Zhang, Z.; Chen, X.; Yang, W. Polymeric nanocarrier via metabolism regulation mediates immunogenic cell death with spatiotemporal orchestration for cancer immunotherapy. Nature communications 2024, 15, 8586. [Google Scholar] [CrossRef]
- Xu, Z.; Liu, S.; Li, Y.; Wu, Y.; Tu, J.; Chen, Q.; Sun, C. Engineering strategies of sequential drug delivery systems for combination tumor immunotherapy. Acta Pharmaceutica Sinica B 2025. [Google Scholar] [CrossRef]
- Zhou, H.; Zheng, H.; Yao, W.; Sun, H.; Yang, Y.-G.; Li, Z.; Song, D.; Zhang, Y.; Sun, T. Spatiotemporally controlled delivery of biomacromolecules via injectable hydrogels for precision modulation of the tumor immune microenvironment. Journal of Nanobiotechnology 2025. [Google Scholar] [CrossRef]
- Sajjad, R.; Chauhdary, S.T.; Anwar, M.T.; Zahid, A.; Khosa, A.A.; Imran, M.; Sajjad, M.H. A review of 4D printing–technologies, shape shifting, smart polymer based materials, and biomedical applications. Advanced Industrial and Engineering Polymer Research 2024, 7, 20–36. [Google Scholar] [CrossRef]
- Ramezani, M.; Mohd Ripin, Z. 4D printing in biomedical engineering: advancements, challenges, and future directions. Journal of functional biomaterials 2023, 14, 347. [Google Scholar] [CrossRef] [PubMed]
- Kanaujia, K.A.; Yadav, V.K.; Yadav, S.S.; Talha, M.; Saraf, S.A.; Kumar, S. 4D Printing in Healthcare: Innovations, Challenges, and Future Directions. In ACS Applied Bio Materials; 2025. [Google Scholar]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. 2021, 372. [Google Scholar]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. Bmj 2009, 339. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, D.; Ramar, M.; Lavudi, K.; Sharma, S.; Rajinikanth, P.; Pandey, P. The potential of fucoidans from ocean treasures to biomedical marvels: A review. International Journal of Biological Macromolecules 2025, 148979. [CrossRef]
- Miricescu, D.; Totan, A.; Stanescu-Spinu, I.-I.; Badoiu, S.C.; Stefani, C.; Greabu, M. PI3K/AKT/mTOR signaling pathway in breast cancer: from molecular landscape to clinical aspects. International journal of molecular sciences 2020, 22, 173. [Google Scholar] [CrossRef]
- Panda, V.K.; Mishra, B.; Mahapatra, S.; Swain, B.; Malhotra, D.; Saha, S.; Khanra, S.; Mishra, P.; Majhi, S.; Kumari, K. Molecular insights on signaling cascades in breast cancer: A comprehensive review. Cancers 2025, 17, 234. [Google Scholar] [CrossRef]
- Balkrishna, A.; Kumar, S.; Malik, R.; Mehra, K.S.; Chaturvedi, H.; Mittal, R. Unveiling role of oncogenic signalling pathways in complicating breast cancer. BioMedicine 2025, 15, 13. [Google Scholar] [CrossRef]
- Garg, P.; Singhal, G.; Horne, D.; Salgia, R.; Singhal, S.S. Metabolic reprogramming in breast cancer: Pathways driving progression, drug resistance, and emerging therapeutics. Biochimica et Biophysica Acta (BBA)-Reviews on Cancer 2025, 189396. [CrossRef] [PubMed]
- Liu, S.; Zhang, X.; Wang, W.; Li, X.; Sun, X.; Zhao, Y.; Wang, Q.; Li, Y.; Hu, F.; Ren, H. Metabolic reprogramming and therapeutic resistance in primary and metastatic breast cancer. Molecular cancer 2024, 23, 261. [Google Scholar] [CrossRef] [PubMed]
- Cappellesso, F.; Mazzone, M.; Virga, F. Acid affairs in anti-tumour immunity. Cancer cell international 2024, 24, 354. [Google Scholar] [CrossRef]
- Bhattacharya, S.; Prajapati, B.G.; Singh, S. A critical review on the dissemination of PH and stimuli-responsive polymeric nanoparticular systems to improve drug delivery in cancer therapy. Critical reviews in oncology/hematology 2023, 185, 103961. [Google Scholar] [CrossRef] [PubMed]
- Tan, R.Y.H.; Lee, C.S.; Pichika, M.R.; Cheng, S.F.; Lam, K.Y. PH responsive polyurethane for the advancement of biomedical and drug delivery. Polymers 2022, 14, 1672. [Google Scholar] [CrossRef]
- Liu, Y.; Si, L.; Jiang, Y.; Jiang, S.; Zhang, X.; Li, S.; Chen, J.; Hu, J. Design of pH-responsive nanomaterials based on the tumor microenvironment. International journal of nanomedicine 2025, 705–721. [CrossRef]
- Su, M.; Song, Y. Printable smart materials and devices: strategies and applications. Chemical reviews 2021, 122, 5144–5164. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.; Zhao, J.; Cheng, F.; Chen, J.; Chen, J.; Xu, H.; Shi, K.; Xia, K.; Ding, S.; Wang, K. Silencing circATXN1 in aging nucleus pulposus cell alleviates intervertebral disc degeneration via correcting progerin mislocalization. Research 2024, 7, 0336. [Google Scholar] [CrossRef]
- Behl, M.; Lendlein, A. Shape-memory polymers. Materials today 2007, 10, 20–28. [Google Scholar] [CrossRef]
- Hu, J.; Zhu, Y.; Huang, H.; Lu, J. Recent advances in shape–memory polymers: Structure, mechanism, functionality, modeling and applications. Progress in polymer science 2012, 37, 1720–1763. [Google Scholar] [CrossRef]
- Miranda, A.; Lopez-Blanco, R.; Lopes-Nunes, J.; Melo, A.M.; Campello, M.P.C.; Paulo, A.; Oliveira, M.C.; Mergny, J.-L.; Oliveira, P.A.; Fernandez-Megia, E. Gallic acid–triethylene glycol aptadendrimers synthesis, biophysical characterization and cellular evaluation. Pharmaceutics 2022, 14, 2456. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.; Zhang, L.; Liu, Y.; Liu, L.; Leng, J. 4D printing of personalized shape memory polymer vascular stents with negative Poisson’s ratio structure: A preliminary study. Science China Technological Sciences 2020, 63, 578–588. [Google Scholar] [CrossRef]
- Ghelardini, M.M.; Geisler, M.; Weigel, N.; Hankwitz, J.P.; Hauck, N.; Schubert, J.; Fery, A.; Tracy, J.B.; Thiele, J. 3D-printed hydrogels as photothermal actuators. Polymers 2024, 16, 2032. [Google Scholar] [CrossRef]
- Tran, T.S.; Balu, R.; Mettu, S.; Roy Choudhury, N.; Dutta, N.K. 4D printing of hydrogels: innovation in material design and emerging smart systems for drug delivery. Pharmaceuticals 2022, 15, 1282. [Google Scholar] [CrossRef]
- Parimita, S.; Kumar, A.; Krishnaswamy, H.; Ghosh, P. 4D printing of pH-responsive bilayer with programmable shape-shifting behaviour. European Polymer Journal 2025, 222, 113581. [Google Scholar] [CrossRef]
- Qu, G.; Huang, J.; Gu, G.; Li, Z.; Wu, X.; Ren, J. Smart implants: 4D-printed shape-morphing scaffolds for medical implantation. International Journal of Bioprinting 2023, 9, 764. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Wang, L.; Zheng, Z.; Liu, Y.; Leng, J. Magnetic programming of 4D printed shape memory composite structures. 2019. [Google Scholar] [CrossRef]
- Mandal, A.; Chatterjee, K. 4D printing for biomedical applications. Journal of Materials Chemistry B 2024, 12, 2985–3005. [Google Scholar] [CrossRef]
- Imam, S.S.; Hussain, A.; Altamimi, M.A.; Alshehri, S. Four-dimensional printing for hydrogel: theoretical concept, 4D materials, shape-morphing way, and future perspectives. Polymers 2021, 13, 3858. [Google Scholar] [CrossRef]
- Megdich, A.; Habibi, M.; Laperrière, L. A review on 4D printing: Material structures, stimuli and additive manufacturing techniques. Materials Letters 2023, 337, 133977. [Google Scholar] [CrossRef]
- Osouli-Bostanabad, K.; Masalehdan, T.; Kapsa, R.M.; Quigley, A.; Lalatsa, A.; Bruggeman, K.F.; Franks, S.J.; Williams, R.J.; Nisbet, D.R. Traction of 3D and 4D printing in the healthcare industry: from drug delivery and analysis to regenerative medicine. ACS Biomaterials Science & Engineering 2022, 8, 2764–2797. [Google Scholar] [CrossRef]
- Zhu, Y.; Haghniaz, R.; Hartel, M.C.; Mou, L.; Tian, X.; Garrido, P.R.; Wu, Z.; Hao, T.; Guan, S.; Ahadian, S. Recent advances in bioinspired hydrogels: materials, devices, and biosignal computing. ACS biomaterials science & engineering 2021, 9, 2048–2069. [Google Scholar]
- Basety, S.; Gudepu, R.; Velidandi, A. Smart Nanoformulations for Oncology: A Review on Overcoming Biological Barriers with Active Targeting, Stimuli-Responsive, and Controlled Release for Effective Drug Delivery. Pharmaceutics 2026, 18, 196. [Google Scholar] [CrossRef] [PubMed]
- Singh, J.; Nayak, P. pH-responsive polymers for drug delivery: trends and opportunities. Journal of polymer science 2023, 61, 2828–2850. [Google Scholar] [CrossRef]
- Veres, C.; Tănase, M.; Szabo, D.-A. Polymeric Materials in Biomedical Engineering: A Bibliometric Mapping. Polymers 2025, 17, 2886. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Duan, C.; Widenhoefer, R.A.; Craig, S.L. Improving Catalytic Enantioselectivity of Hydrogenation through Swelling-Induced Molecular Tension in Polymer Networks. Journal of the American Chemical Society 2025, 147, 31085–31090. [Google Scholar] [CrossRef]
- Barmatov, E.; La Terra, F.; Hughes, T. Mechanism of degradation of propargyl alcohol by acid-catalysed hydrolysis and corrosion inhibition efficiency of propargyl alcohol intermediates for carbon steel in hydrochloric acid. Materials Chemistry and Physics 2021, 272, 125048. [Google Scholar] [CrossRef]
- Sun, J.; Lerner, E.; Tighe, B.; Middlemist, C.; Zhao, J. Embedded shape morphing for morphologically adaptive robots. Nature communications 2023, 14, 6023. [Google Scholar] [CrossRef] [PubMed]
- Azizi, V.; Hu, G. A multi-stage stochastic programming model for the multi-echelon multi-period reverse logistics problem. Sustainability 2021, 13, 13596. [Google Scholar] [CrossRef]
- Shen, D.; Zhang, M.; Mujumdar, A.S.; Li, J. Advances and application of efficient physical fields in extrusion based 3D food printing technology. Trends in Food Science & Technology 2023, 131, 104–117. [Google Scholar]
- Vaseashta, A.; Demir, D.; Bolgen, N. Synergetic integration of electrospinning and additive 3D/4D printing process for biomedical applications. In Proceedings of the Nordic-Baltic Conference on Biomedical Engineering and Medical Physics, 2023; pp. 129–137. [Google Scholar]
- Li, W.; Wang, M.; Ma, H.; Chapa-Villarreal, F.A.; Lobo, A.O.; Zhang, Y.S. Stereolithography apparatus and digital light processing-based 3D bioprinting for tissue fabrication. Iscience 2023, 26. [Google Scholar] [CrossRef] [PubMed]
- Vlasa, A.; Bocanet, V.I.; Muntean, M.H.; Bud, A.; Dragomir, B.R.; Rosu, S.N.; Lazar, L.; Bud, E. Accuracy of three-dimensional printed dental models based on ethylene di-methacrylate-stereolithography (SLA) vs. digital light processing (DLP). Applied Sciences 2023, 13, 2664. [Google Scholar] [CrossRef]
- Friedman, L.R.; Stepp, H.E.; Lambdin, J.; Luna, A.; Rossi, A.; Lux, S.C.; Wach, M.M.; Luberice, K.; Ruff, S.M.; McDonald, J. An open-source normothermic perfusion system designed for research scientists. Journal of Visualized Experiments (JoVE) 2025, e68722. [CrossRef]
- Zolfagharian, A.; Kaynak, A.; Bodaghi, M.; Kouzani, A.Z.; Gharaie, S.; Nahavandi, S. Control-based 4D printing: Adaptive 4D-printed systems. Applied Sciences 2020, 10, 3020. [Google Scholar] [CrossRef]
- Sadraei, A.; Naghib, S.M.; Rabiee, N. 4D printing chemical stimuli-responsive hydrogels for tissue engineering and localized drug delivery applications–part 2. Expert Opinion on Drug Delivery 2025, 22, 491–510. [Google Scholar] [CrossRef]
- Gawne, P.J.; Ferreira, M.; Papaluca, M.; Grimm, J.; Decuzzi, P. New opportunities and old challenges in the clinical translation of nanotheranostics. Nature Reviews Materials 2023, 8, 783–798. [Google Scholar] [CrossRef]
- Skylaki, S.; Hilsenbeck, O.; Schroeder, T. Challenges in long-term imaging and quantification of single-cell dynamics. Nature Biotechnology 2016, 34, 1137–1144. [Google Scholar] [CrossRef]
- Zhang, J.; Zhuang, Y.; Sheng, R.; Tomás, H.; Rodrigues, J.; Yuan, G.; Wang, X.; Lin, K. Smart stimuli-responsive strategies for titanium implant functionalization in bone regeneration and therapeutics. Materials Horizons 2024, 11, 12–36. [Google Scholar] [CrossRef]
- Shi, Y.; Yu, Q.; Tan, L.; Wang, Q.; Zhu, W.H. Tumor Microenvironment-Responsive Polymer Delivery Platforms for Cancer Therapy. Angewandte Chemie 2025, 137, e202503776. [Google Scholar] [CrossRef]
- Peppicelli, S.; Bianchini, F.; Calorini, L. Extracellular acidity, a “reappreciated” trait of tumor environment driving malignancy: perspectives in diagnosis and therapy. Cancer and Metastasis Reviews 2014, 33, 823–832. [Google Scholar] [CrossRef]
- Kantamneni, D.; Gurbani, S.; Salvatore, M. The Role of Tumor pH in Breast Cancer Imaging: Biology, Diagnostic Applications, and Emerging Techniques. Diagnostics 2025, 16, 76. [Google Scholar] [CrossRef]
- Feng, W.; Wang, Z. Tailoring the swelling-shrinkable behavior of hydrogels for biomedical applications. Advanced Science 2023, 10, 2303326. [Google Scholar] [CrossRef] [PubMed]
- Ogueri, K.S.; Laurencin, C.T. Nanofiber technology for regenerative engineering. ACS nano 2020, 14, 9347–9363. [Google Scholar] [CrossRef] [PubMed]
- Ye, K.; Kuang, H.; You, Z.; Morsi, Y.; Mo, X. Electrospun nanofibers for tissue engineering with drug loading and release. Pharmaceutics 2019, 11, 182. [Google Scholar] [CrossRef] [PubMed]
- Kayali, Y.; Gleadall, A.; Silberschmidt, V.V. Geometric Characterisation of Stochastic Fibrous Networks: A Comprehensive Review. Fibers 2025, 13, 27. [Google Scholar] [CrossRef]
- Itano, K.; Choi, J.; Rubner, M.F. Mechanism of the pH-induced discontinuous swelling/deswelling transitions of poly (allylamine hydrochloride)-containing polyelectrolyte multilayer films. Macromolecules 2005, 38, 3450–3460. [Google Scholar] [CrossRef]
- Bandaru, S.; Arora, D.; Ganesh, K.M.; Umrao, S.; Thomas, S.; Bhaskar, S.; Chakrabortty, S. Recent advances in research from nanoparticle to nano-assembly: a review. Nanomaterials 2024, 14, 1387. [Google Scholar] [CrossRef]
- Zurletti, B.; Andreana, I.; Salaroglio, I.C.; Bincoletto, V.; Manzoli, M.; Rolando, B.; Milla, P.; Riganti, C.; Stella, B.; Arpicco, S. Tailoring the Composition of HA/PEG Mixed Nano-Assemblies for Anticancer Drug Delivery. Molecules 2025, 30, 1349. [Google Scholar] [CrossRef]
- Eer, K.X.; A Rahman, R.; Mat Alewi, N.A. Progress on encapsulation and entrapment of enzymes in electrospun nanofibers. Bioprocess and Biosystems Engineering 2025, 1–32. [CrossRef]
- Orange, J.S. Formation and function of the lytic NK-cell immunological synapse. Nature Reviews Immunology 2008, 8, 713–725. [Google Scholar] [CrossRef]
- Guo, P.; Zhu, B.; Bai, T.; Guo, X.; Shi, D.; Jiang, C.; Kong, J.; Huang, Q.; Shi, J.; Shao, D. Nanomaterial-Interleukin Combination for Boosting NK Cell-Based Tumor Immunotherapy. ACS Biomaterials Science & Engineering 2025, 11, 3126–3144. [Google Scholar]
- Yi, M.; Li, T.; Niu, M.; Zhang, H.; Wu, Y.; Wu, K.; Dai, Z. Targeting cytokine and chemokine signaling pathways for cancer therapy. Signal transduction and targeted therapy 2024, 9, 176. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Yang, S.; Hu, Q.; Pu, Z.; Hu, Y.; Gong, W.; Wu, H.; Gao, Z.; Wang, J.; Yang, J. Spatiotemporal 4D-Printed Shape-Memory Scaffold with a Triple-Acting Liposomal Strategy for the Treatment of Infectious Bone Defects. Biomaterials 2026, 124020. [Google Scholar] [CrossRef] [PubMed]
- Chakrapani, G.; Vijay Kumar, V.; Zare, M. Next-Generation Biomaterials: Design Strategies, Clinical Translation, and the Rise of Intelligent Therapeutic Platforms. ACS omega 2025, 10, 46272–46293. [Google Scholar] [CrossRef]
- Cattapan, C.; Della Barbera, M.; Dedja, A.; Pavan, P.; Di Salvo, G.; Sabatino, J.; Avesani, M.; Padalino, M.; Guariento, A.; Basso, C. Mechanical and structural adaptation of the pulmonary root after Ross operation in a murine model. Journal of Clinical Medicine 2022, 11, 3742. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Chen, Y.; Huang, L.; Cai, M.; Yin, X.; Zhu, Y.Z.; Ye, L. Bioengineered In Situ-Forming Hydrogels as Smart Drug Delivery Systems for Postoperative Breast Cancer Immunotherapy: From Material Innovation to Clinical Translation. Journal of Functional Biomaterials 2025, 16, 381. [Google Scholar] [CrossRef]
- Sadraei, A.; Naghib, S.M. 4D printing of physical stimuli-responsive hydrogels for localized drug delivery and tissue engineering. Polymer Reviews 2025, 65, 104–168. [Google Scholar] [CrossRef]
- Nickel, J.; Hurst, A.; Duimering, P.R. Contextual influences on trade-offs in engineering design: A qualitative study. Design Science 2024, 10, e21. [Google Scholar] [CrossRef]
- Moroni, S.; Bingham, R.; Buckley, N.; Casettari, L.; Lamprou, D.A. 4D printed multipurpose smart implants for breast cancer management. International Journal of Pharmaceutics 2023, 642, 123154. [Google Scholar] [CrossRef] [PubMed]
- Wall, I.; Boulat, V.; Shah, A.; Blenman, K.R.; Wu, Y.; Alberts, E.; Calado, D.P.; Salgado, R.; Grigoriadis, A. Leveraging the dynamic immune environment triad in patients with breast cancer: tumour, lymph node, and peripheral blood. Cancers 2022, 14, 4505. [Google Scholar] [CrossRef]
- Piwocka, O.; Piotrowski, I.; Suchorska, W.M.; Kulcenty, K. Dynamic interactions in the tumor niche: how the cross-talk between CAFs and the tumor microenvironment impacts resistance to therapy. Frontiers in Molecular Biosciences 2024, 11, 1343523. [Google Scholar] [CrossRef] [PubMed]
- Dutta, N.; Halder, N.; Chini, E.N.; Kim, S.; Manna, A. Regulating the regulators via targeting CD38 in the tumor microenvironment. Frontiers in Immunology 2026, 17, 1745988. [Google Scholar] [CrossRef]
- Xiong, X.; Zheng, L.-W.; Ding, Y.; Chen, Y.-F.; Cai, Y.-W.; Wang, L.-P.; Huang, L.; Liu, C.-C.; Shao, Z.-M.; Yu, K.-D. Breast cancer: pathogenesis and treatments. Signal transduction and targeted therapy 2025, 10, 49. [Google Scholar] [CrossRef]
- Santana dos Santos, E.; Lallemand, F.; Petitalot, A.; Caputo, S.M.; Rouleau, E. HRness in breast and ovarian cancers. International Journal of Molecular Sciences 2020, 21, 3850. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Guo, Y.; Jia, J.; Cui, W.; Zhang, X.; Wang, Y.; Xie, Z.; Fang, Y.; Ding, X.; Chang, L. Emerging role of cancer-associated fibroblasts in the premetastatic niche. Molecular Cancer 2025, 24, 250. [Google Scholar] [CrossRef]
- Xu, J.; Ding, L.; Mei, J.; Hu, Y.; Kong, X.; Dai, S.; Bu, T.; Xiao, Q.; Ding, K. Dual roles and therapeutic targeting of tumor-associated macrophages in tumor microenvironments. Signal transduction and targeted therapy 2025, 10, 268. [Google Scholar] [CrossRef]
- Bergenfelz, C.; Leandersson, K. The generation and identity of human myeloid-derived suppressor cells. Frontiers in oncology 2020, 10, 109. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Jin, W.; Hardegen, N.; Lei, K.-j.; Li, L.; Marinos, N.; McGrady, G.; Wahl, S.M. Conversion of peripheral CD4+ CD25− naive T cells to CD4+ CD25+ regulatory T cells by TGF-β induction of transcription factor Foxp3. The Journal of experimental medicine 2003, 198, 1875–1886. [Google Scholar] [CrossRef]
- Salven, P.; Mustjoki, S.; Alitalo, R.; Alitalo, K.; Rafii, S. VEGFR-3 and CD133 identify a population of CD34+ lymphatic/vascular endothelial precursor cells. Blood, The Journal of the American Society of Hematology 2003, 101, 168–172. [Google Scholar] [CrossRef]
- Chiossone, L.; Dumas, P.-Y.; Vienne, M.; Vivier, E. Natural killer cells and other innate lymphoid cells in cancer. Nature Reviews Immunology 2018, 18, 671–688. [Google Scholar] [CrossRef]
- Fjørtoft, M.O.; Huse, K.; Rye, I.H. The tumor immune microenvironment in breast cancer progression. Acta Oncologica 2024, 63, 33008. [Google Scholar] [CrossRef]
- Jhunjhunwala, S.; Hammer, C.; Delamarre, L. Antigen presentation in cancer: insights into tumour immunogenicity and immune evasion. Nature Reviews Cancer 2021, 21, 298–312. [Google Scholar] [CrossRef]
- Xue, V.W.; Chung, J.Y.-F.; Córdoba, C.A.G.; Cheung, A.H.-K.; Kang, W.; Lam, E.W.-F.; Leung, K.-T.; To, K.-F.; Lan, H.-Y.; Tang, P.M.-K. Transforming growth factor-β: a multifunctional regulator of cancer immunity. Cancers 2020, 12, 3099. [Google Scholar] [CrossRef]
- Kwiatkowska, I.; Hermanowicz, J.M.; Przybyszewska-Podstawka, A.; Pawlak, D. Not Only Immune Escape—The Confusing Role of the TRP Metabolic Pathway in Carcinogenesis. Cancers 2021, 13, 2667. [Google Scholar] [CrossRef] [PubMed]
- Linderman, S.W.; DeRidder, L.; Sanjurjo, L.; Foote, M.B.; Alonso, M.J.; Kirtane, A.R.; Langer, R.; Traverso, G. Enhancing immunotherapy with tumour-responsive nanomaterials. Nature Reviews Clinical Oncology 2025, 22, 262–282. [Google Scholar] [CrossRef] [PubMed]
- Tseng, T.-Y.; Hsieh, C.-H.; Huang, H.-C.; Wu, Y.-C.; Hsu, C.; Hsu, C.-L.; Ou, D.-L.; Juan, H.-F. Transcriptional dynamics of CD8+ T-cell exhaustion in immune checkpoint inhibitor resistance at single-cell resolution. Molecular Cancer 2025, 24, 306. [Google Scholar] [CrossRef]
- Goff, S.L.; Danforth, D.N. The role of immune cells in breast tissue and immunotherapy for the treatment of breast cancer. Clinical breast cancer 2021, 21, e63–e73. [Google Scholar]
- Ulloa, L.; Doody, J.; Massagué, J. Inhibition of transforming growth factor-β/SMAD signalling by the interferon-γ/STAT pathway. Nature 1999, 397, 710–713. [Google Scholar] [CrossRef]
- Kothawade, S.; Singh, S. Recent Advancements in Stimuli-Responsive Polymeric Implants Fabricated via Additive Manufacturing: A Review. Current Pharmaceutical Design, 2025. [Google Scholar]
- Peppicelli, S.; Calorini, L.; Bianchini, F.; Papucci, L.; Magnelli, L.; Andreucci, E. Acidity and hypoxia of tumor microenvironment, a positive interplay in extracellular vesicle release by tumor cells. Cellular Oncology 2025, 48, 27–41. [Google Scholar] [CrossRef]
- Rahman, M.A.; Yadab, M.K.; Ali, M.M. Emerging role of extracellular pH in tumor microenvironment as a therapeutic target for cancer immunotherapy. Cells 2024, 13, 1924. [Google Scholar] [CrossRef]
- Shah, D.D.; Chorawala, M.R.; Raghani, N.R.; Patel, R.; Fareed, M.; Kashid, V.A.; Prajapati, B.G. Tumor microenvironment: recent advances in understanding and its role in modulating cancer therapies. Medical Oncology 2025, 42, 117. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Guo, Z.; Chen, Z.; Wang, C.; Yang, Y.; Shi, Q.; Gao, Q.; Li, H.; Zhang, D.; Liu, Y. pH-Responsive Materials for Therapy and Precision Biomedical Imaging. Chemical & Biomedical Imaging 2026. [Google Scholar]
- Andrade, F.; Roca-Melendres, M.M.; Duran-Lara, E.F.; Rafael, D.; Schwartz, S., Jr. Stimuli-responsive hydrogels for cancer treatment: The role of pH, light, ionic strength and magnetic field. Cancers 2021, 13, 1164. [Google Scholar] [CrossRef]
- Guo, J.; Zhang, R.; Zhang, L.; Cao, X. 4D printing of robust hydrogels consisted of agarose nanofibers and polyacrylamide. ACS Macro Letters 2018, 7, 442–446. [Google Scholar] [CrossRef]
- Verron, Q.; Forslund, E.; Brandt, L.; Leino, M.; Frisk, T.W.; Olofsson, P.E.; Önfelt, B. NK cells integrate signals over large areas when building immune synapses but require local stimuli for degranulation. Science Signaling 2021, 14, eabe2740. [Google Scholar] [CrossRef]
- Huang, R.; Kang, T.; Chen, S. The role of tumor-associated macrophages in tumor immune evasion. Journal of cancer research and clinical oncology 2024, 150, 238. [Google Scholar] [PubMed]
- Zhang, Q.; Sioud, M. Tumor-associated macrophage subsets: shaping polarization and targeting. International journal of molecular sciences 2023, 24, 7493. [Google Scholar] [CrossRef]
- Mai, Z.; Lin, Y.; Lin, P.; Zhao, X.; Cui, L. Modulating extracellular matrix stiffness: a strategic approach to boost cancer immunotherapy. Cell death & disease 2024, 15, 307. [Google Scholar]
- Winkler, J.; Abisoye-Ogunniyan, A.; Metcalf, K.J.; Werb, Z. Concepts of extracellular matrix remodelling in tumour progression and metastasis. Nature communications 2020, 11, 5120. [Google Scholar] [CrossRef]
- Zannat, A.; Lopez, A.; Cheng, Y.; Ma, J.; Delemeester, M.; Lin, S.; Jayaraman, K.; Liu, X. Mechanical strain-regulated hydrogel biodegradation for biological scaffolds with programmable lifetime. Journal of Materials Chemistry B 2025, 13, 10485–10499. [Google Scholar] [CrossRef]
- Lee, M.-S.; Bensinger, S.J. Reprogramming cholesterol metabolism in macrophages and its role in host defense against cholesterol-dependent cytolysins. Cellular & molecular immunology 2022, 19, 327–336. [Google Scholar]
- Datta, M.; Coussens, L.M.; Nishikawa, H.; Hodi, F.S.; Jain, R.K. Reprogramming the tumor microenvironment to improve immunotherapy: emerging strategies and combination therapies. In Proceedings of the American Society of Clinical Oncology educational book, 2019; American Society of Clinical Oncology. Annual Meeting; p. 165. [Google Scholar]
- Zhang, H.; Li, S.; Wang, D.; Liu, S.; Xiao, T.; Gu, W.; Yang, H.; Wang, H.; Yang, M.; Chen, P. Metabolic reprogramming and immune evasion: the interplay in the tumor microenvironment. Biomarker research 2024, 12, 96. [Google Scholar] [CrossRef]
- Lazebnik, T. Cell-level spatio-temporal model for a Bacillus Calmette–Guérin-based immunotherapy treatment protocol of superficial bladder cancer. Cells 2022, 11, 2372. [Google Scholar] [CrossRef] [PubMed]
- Huang, K.; Liu, L.; Huang, Y.; Wang, Y.; Zhou, Y.; Han, G. Remote control of cellular immunotherapy. Nature Reviews Bioengineering 2023, 1, 440–455. [Google Scholar] [CrossRef]
- Cheng, J.; Li, B.; Song, X.; Ji, X.; Zhang, Y.; Chen, J.; Xiang, H. Integrated surrogate model-based approach for aerodynamic design optimization of three-stage axial compressor in gas turbine applications. Energies 2025, 18, 4514. [Google Scholar] [CrossRef]
- Jagodinsky, J.C.; Morris, Z.S. Priming and propagating anti-tumor immunity: Focal hypofractionated radiation for in situ vaccination and systemic targeted radionuclide theranostics for immunomodulation of tumor microenvironments. In Proceedings of the Seminars in radiation oncology, 2020; pp. 181–186. [Google Scholar]
- Liu, C.; Yang, X.; Paoli-Bruno, J.; Sikes, D.; Marin-Ruiz, A.V.; Thomas, N.; Shane, R.; Har-Noy, M. Allo-Priming Reverses Immunosenescence and May Restore Broad Respiratory Viral Protection and Vaccine Responsiveness to the Elderly: Results of a Phase I/II Clinical Trial. Vaccines 2025, 13, 463. [Google Scholar] [CrossRef]
- Lee, S.-C.; Shimasaki, N.; Lim, J.S.; Wong, A.; Yadav, K.; Yong, W.P.; Tan, L.K.; Koh, L.P.; Poon, M.L.; Tan, S.H. Phase I trial of expanded, activated autologous NK-cell infusions with trastuzumab in patients with HER2-positive cancers. Clinical Cancer Research 2020, 26, 4494–4502. [Google Scholar]
- Lam, N.; Lee, Y.; Farber, D.L. A guide to adaptive immune memory. Nature Reviews Immunology 2024, 24, 810–829. [Google Scholar] [CrossRef]
- Xydas, E.; Marmaras, C.; Cipcigan, L.M. A multi-agent based scheduling algorithm for adaptive electric vehicles charging. Applied energy 2016, 177, 354–365. [Google Scholar]
- Poggi, A.; Zocchi, M.R. Natural killer cells and immune-checkpoint inhibitor therapy: Current knowledge and new challenges. Molecular Therapy-Oncolytics 2022, 24, 26–42. [Google Scholar] [CrossRef]
- Goswami, S.; Pauken, K.E.; Wang, L.; Sharma, P. Next-generation combination approaches for immune checkpoint therapy. Nature immunology 2024, 25, 2186–2199. [Google Scholar] [CrossRef] [PubMed]
- Mircioiu, C.; Voicu, V.; Anuta, V.; Tudose, A.; Celia, C.; Paolino, D.; Fresta, M.; Sandulovici, R.; Mircioiu, I. Mathematical modeling of release kinetics from supramolecular drug delivery systems. Pharmaceutics 2019, 11, 140. [Google Scholar] [CrossRef] [PubMed]
- Shishir, M.R.I.; Gowd, V.; Suo, H.; Wang, M.; Wang, Q.; Chen, F.; Cheng, K.W. Advances in smart delivery of food bioactive compounds using stimuli-responsive carriers: Responsive mechanism, contemporary challenges, and prospects. Comprehensive Reviews in Food Science and Food Safety 2021, 20, 5449–5488. [Google Scholar] [CrossRef]
- Gao, W.; Zhong, J.; Liu, X.; Bai, D.; Wu, M. Intelligent microstructure materials for diagnosis and treatment of osteoarthritis: progress and AI-enpowered future. Bone Research 2025, 13, 85. [Google Scholar] [CrossRef]
- Wu, Y.; Wu, Y.; Gao, Z.; Yu, W.; Zhang, L.; Zhou, F. Revitalizing T cells: breakthroughs and challenges in overcoming T cell exhaustion. Signal transduction and targeted therapy 2026, 11, 2. [Google Scholar]
- Chen, J.; Liang, R.; Jia, S.; Huang, J.; Liu, P.; Li, W.; Wang, J.; Cui, W.; Lin, J. Spatiotemporal Adaptation in 4D Bioprinting for Dynamic Bone and Cartilage Regeneration. Advanced Functional Materials 2025, e22357.
- Shibue, T.; Weinberg, R.A. EMT, CSCs, and drug resistance: the mechanistic link and clinical implications. Nature reviews Clinical oncology 2017, 14, 611–629. [Google Scholar] [CrossRef] [PubMed]
- Fowell, D.J.; Kim, M. The spatio-temporal control of effector T cell migration. Nature Reviews Immunology 2021, 21, 582–596. [Google Scholar] [CrossRef] [PubMed]
- Joshi, D.; Shukla, P.; Choudhary, N.K. Enhancing Pharmaceutical 3D Printing through Quality-by-Design (QbD): Challenges, Strategies, and Regulatory Insights. International Journal of Newgen Research in Pharmacy & Healthcare 2025, 158–168.
- Gonçalves, A.M.; Moreira, A.; Weber, A.; Williams, G.R.; Costa, P.F. Osteochondral tissue engineering: the potential of electrospinning and additive manufacturing. Pharmaceutics 2021, 13, 983. [Google Scholar] [CrossRef]
- Thakur, K.; Kaur, S.D.; Kak, D.; Kapoor, D.N. Quality by Design in parenteral drug development: Addressing formulation challenges and industrial insights. Drug Development and Industrial Pharmacy 2026, 1–23. [Google Scholar] [CrossRef]
- Hachim, D.; Iftikhar, A.; LoPresti, S.T.; Nolfi, A.L.; Ravichandar, S.; Skillen, C.D.; Brown, B.N. Distinct release strategies are required to modulate macrophage phenotype in young versus aged animals. Journal of Controlled Release 2019, 305, 65–74. [Google Scholar] [CrossRef]
- Zhou, Y.; Li, M.; Jin, K.; Wen, M.; Qin, H.; Xu, Y.; Wang, C.; Zhang, X.; Cao, X. The RNA-binding protein RRP1 brakes macrophage one-carbon metabolism to suppress autoinflammation. Nature Communications 2025, 16, 6880. [Google Scholar] [CrossRef]
- Moroni, S.; Casettari, L.; Lamprou, D.A. 3D and 4D Printing in the Fight against Breast Cancer. Biosensors 2022, 12, 568. [Google Scholar] [CrossRef]
- Ojha, A.; Bhargava, S. International council for harmonisation (ICH) guidelines. In Regulatory affairs in the pharmaceutical industry; Elsevier, 2022; pp. 47–74. [Google Scholar]
- Yuan, Z.; Bai, X.; Li, S.; Fu, Y.; Wan, Z.; Guo, X.; Zhai, M.; Yi, J.; Liu, Y.; Zhou, Y. Multimaterial and multidimensional bioprinting in regenerative medicine: advances, limitations, and future directions. Advanced Healthcare Materials 2025, 14, 2500475. [Google Scholar] [CrossRef]
- Leonard, W.J.; Lin, J.-X. Strategies to therapeutically modulate cytokine action. Nature Reviews Drug Discovery 2023, 22, 827–854. [Google Scholar] [CrossRef]
- Gutiérrez, J.M.; Solano, G.; Pla, D.; Herrera, M.; Segura, Á.; Vargas, M.; Villalta, M.; Sánchez, A.; Sanz, L.; Lomonte, B. Preclinical evaluation of the efficacy of antivenoms for snakebite envenoming: state-of-the-art and challenges ahead. Toxins 2017, 9, 163. [Google Scholar] [CrossRef] [PubMed]
- Parham, P.; Moffett, A. Variable NK cell receptors and their MHC class I ligands in immunity, reproduction and human evolution. Nature Reviews Immunology 2013, 13, 133–144. [Google Scholar] [CrossRef] [PubMed]
- Deckers, J.; Anbergen, T.; Hokke, A.M.; de Dreu, A.; Schrijver, D.P.; de Bruin, K.; Toner, Y.C.; Beldman, T.J.; Spangler, J.B.; de Greef, T.F. Engineering cytokine therapeutics. Nature Reviews Bioengineering 2023, 1, 286–303. [Google Scholar] [CrossRef]
- Ball, K.; Dovedi, S.J.; Vajjah, P.; Phipps, A. Strategies for clinical dose optimization of T cell-engaging therapies in oncology. In Proceedings of the MAbs, 2023; p. 2181016. [Google Scholar]
- Eisman, A.B.; Kilbourne, A.M.; Dopp, A.R.; Saldana, L.; Eisenberg, D. Economic evaluation in implementation science: making the business case for implementation strategies. Psychiatry research 2020, 283, 112433. [Google Scholar] [CrossRef]
- Bergkvist, L.; Karlsson, J. Bridging the Gap—From Great Ideas to Realized Innovations. In Service innovation for sustainable business: Stimulating, realizing and capturing the value from service innovation; World Scientific, 2019; pp. 225–252. [Google Scholar]
- Rodriguez-Manzano, J.; Subramaniam, S.; Uchea, C.; Szostak-Lipowicz, K.M.; Freeman, J.; Rauch, M.; Tinto, H.; Zar, H.J.; D'Alessandro, U.; Holmes, A.H. Innovative diagnostic technologies: navigating regulatory frameworks through advances, challenges, and future prospects. The Lancet Digital Health 2024, 6, e934–e943. [Google Scholar] [CrossRef]
- Keihan Shokooh, M.; Emami, F.; Jeong, J.-H.; Yook, S. Bio-inspired and smart nanoparticles for triple negative breast cancer microenvironment. Pharmaceutics 2021, 13, 287. [Google Scholar] [CrossRef] [PubMed]
- Yarali, E.; Mirzaali, M.J.; Ghalayaniesfahani, A.; Accardo, A.; Diaz-Payno, P.J.; Zadpoor, A.A. 4D printing for biomedical applications. Advanced Materials 2024, 36, 2402301. [Google Scholar] [CrossRef]
- Pugliese, R.; Regondi, S. Artificial intelligence-empowered 3D and 4D printing technologies toward smarter biomedical materials and approaches. Polymers 2022, 14, 2794. [Google Scholar] [CrossRef]
- Joo, J.; Kim, W.; Kim, D.H.; Lee, G.; Yoo, J.-Y.; Won, S.M. Materials and Systems for Closed-Loop Optical Actuation with Integrated Sensing Pathways. ACS Applied Optical Materials 2025, 4, 1–12. [Google Scholar] [CrossRef]
- Arias-Peregrino, V.M.; Tenorio-Barajas, A.Y.; Mendoza-Barrera, C.O.; Román-Doval, J.; Lavariega-Sumano, E.F.; Torres-Arellanes, S.P.; Román-Doval, R. 3D printing for tissue engineering: printing techniques, biomaterials, challenges, and the emerging role of 4D bioprinting. Bioengineering 2025, 12, 936. [Google Scholar] [CrossRef]
- Wu, Q.-X.; De Isla, N.; Zhang, L. Biomaterial-Based nucleic acid delivery systems for In situ tissue engineering and regenerative medicine. International Journal of Molecular Sciences 2025, 26, 7384. [Google Scholar] [CrossRef] [PubMed]
- Mc Neil, V.; Lee, S.W. Advancing cancer treatment: A review of immune checkpoint inhibitors and combination strategies. Cancers 2025, 17, 1408. [Google Scholar] [CrossRef] [PubMed]
- PEDARD, M.; Cantero, L.C.; Davanture, S.; Pernot, J.; Widmer, V.; Ghasemi, A.; Dutoit, V.; Migliorini, D. Adapting CAR T-cell therapy for glioblastoma to the post-surgical immunosuppressive microenvironment. 2026. [Google Scholar]
- Wang, J.; Arthanat, S.; Opuda, E.; LaRoche, D.; Hamilton, S.; Li, A.; Mitchell, C.; Woodward, A.; Li, G.; Begum, M. Ethical considerations in home monitoring technologies for persons living with cognitive impairment: a scoping review. The Gerontologist 2026, 66, gnaf261. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Sun, X.; Wang, H.; Wistuba, I.I.; Wang, H.; Maitra, A.; Chen, Y. TREM2 depletion in pancreatic cancer elicits pathogenic inflammation and accelerates tumor progression via enriching IL-1β+ macrophages. Gastroenterology 2025, 168, 1153–1169. [Google Scholar] [CrossRef]
- Liu, X.; Yang, J.; Huang, S.; Hong, Y.; Zhu, Y.; Wang, J.; Wang, Y.; Liang, T.; Bai, X. Pancreatic cancer-derived extracellular vesicles enhance chemoresistance by delivering KRASG12D protein to cancer-associated fibroblasts. Molecular Therapy 2025, 33, 1134–1153. [Google Scholar] [CrossRef]
- Tempero, M.A.; Van Cutsem, E.; Sigal, D.; Oh, D.-Y.; Fazio, N.; Macarulla, T.; Hitre, E.; Hammel, P.; Hendifar, A.E.; Bates, S.E. HALO 109-301: A randomized, double-blind, placebo-controlled, phase 3 study of pegvorhyaluronidase alfa (PEGPH20)+ nab-paclitaxel/gemcitabine (AG) in patients (pts) with previously untreated hyaluronan (HA)-high metastatic pancreatic ductal adenocarcinoma (mPDA). 2020. [Google Scholar]
- Suzuki, N.; Hazama, S.; Iguchi, H.; Uesugi, K.; Tanaka, H.; Hirakawa, K.; Aruga, A.; Hatori, T.; Ishizaki, H.; Umeda, Y. Phase II clinical trial of peptide cocktail therapy for patients with advanced pancreatic cancer: VENUS-PC study. Cancer science 2017, 108, 73–80. [Google Scholar] [CrossRef]
- Bockorny, B.; Semenisty, V.; Macarulla, T.; Borazanci, E.; Wolpin, B.M.; Stemmer, S.M.; Golan, T.; Geva, R.; Borad, M.J.; Pedersen, K.S. BL-8040, a CXCR4 antagonist, in combination with pembrolizumab and chemotherapy for pancreatic cancer: the COMBAT trial. Nature Medicine 2020, 26, 878–885. [Google Scholar] [CrossRef]
- Bockorny, B.; Macarulla, T.; Semenisty, V.; Borazanci, E.; Feliu, J.; Ponz-Sarvise, M.; Abad, D.G.; Oberstein, P.; Alistar, A.; Munoz, A. Motixafortide and pembrolizumab combined to nanoliposomal irinotecan, fluorouracil, and folinic acid in metastatic pancreatic cancer: the COMBAT/KEYNOTE-202 trial. Clinical Cancer Research 2021, 27, 5020–5027. [Google Scholar] [CrossRef]
- Kindler, H.L.; Ioka, T.; Richel, D.J.; Bennouna, J.; Létourneau, R.; Okusaka, T.; Funakoshi, A.; Furuse, J.; Park, Y.S.; Ohkawa, S. Axitinib plus gemcitabine versus placebo plus gemcitabine in patients with advanced pancreatic adenocarcinoma: a double-blind randomised phase 3 study. The lancet oncology 2011, 12, 256–262. [Google Scholar] [CrossRef] [PubMed]
- Alistar, A.; Morris, B.B.; Desnoyer, R.; Klepin, H.D.; Hosseinzadeh, K.; Clark, C.; Cameron, A.; Leyendecker, J.; D'Agostino, R.; Topaloglu, U. Safety and tolerability of the first-in-class agent CPI-613 in combination with modified FOLFIRINOX in patients with metastatic pancreatic cancer: a single-centre, open-label, dose-escalation, phase 1 trial. The lancet oncology 2017, 18, 770–778. [Google Scholar] [CrossRef] [PubMed]
- Patnaik, A.; Pelster, M.; Hong, D.S.; Strickler, J.H.; Garrido-Laguna, I.; Aguirre, A.; Curran, D.; Woo, T.; Spira, A.I. A phase 1 trial evaluating the safety, tolerability, PK, and preliminary efficacy of QTX3034, an oral G12D-preferring multi-KRAS inhibitor, in patients with solid tumors with KRAS G12D mutation. 2024. [Google Scholar] [CrossRef]
| Cell Type | Origin | Key Markers | Major Secreted Factors | Functional Role in Breast Cancer | Potential Relevance to 4D Implants |
Reference |
| Breast Cancer Cells (TNBC / HR+) | Epithelial | HER2, ER, PR, p53, BRCA1/2 | TGF-β, IL-6, lactate, exosomes | Proliferation, EMT, immune evasion, metabolic reprogramming | Targeted cytokine-triggered NK-mediated apoptosis | [90] |
| Cancer-Associated Fibroblasts (CAFs) | Mesenchymal | FAP, α-SMA, PDPN | Collagen, CXCL12, IL-6, ECM proteins | Desmoplasia, ECM stiffening, chemoresistance, and immune exclusion | Local ECM modulation via pH-responsive degradation | [91] |
| Tumour-Associated Macrophages (M2-like) | Monocytes | CD163, CD206 | IL-10, TGF-β, VEGF | Immune suppression, angiogenesis, and metastasis promotion | Cytokine-mediated macrophage reprogramming | [92] |
| Myeloid-Derived Suppressor Cells (MDSCs) | Myeloid lineage | CD11b, CD33 | Arginase, ROS, nitric oxide | T-cell suppression, metabolic inhibition | Local immune reactivation to overcome suppression | [93] |
| Regulatory T Cells (Tregs) | CD4⁺ T cells | FoxP3, CD25 | IL-10, TGF-β | Immune tolerance, IL-2 sequestration | Controlled IL-2 delivery to favour NK over Treg expansion | [94] |
| Endothelial Cells | Vascular | VEGFR, CD31 | VEGF, angiopoietins | Tumour angiogenesis, nutrient supply | Sequential vascular normalisation before cytokine release | [95] |
| NK Cells | Innate lymphoid cells | CD56, NKG2D | IFN-γ, perforin, granzyme B | Direct tumor cytotoxicity | The implant activates primary effector cells | [96] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




