Submitted:
02 March 2026
Posted:
04 March 2026
You are already at the latest version
Abstract
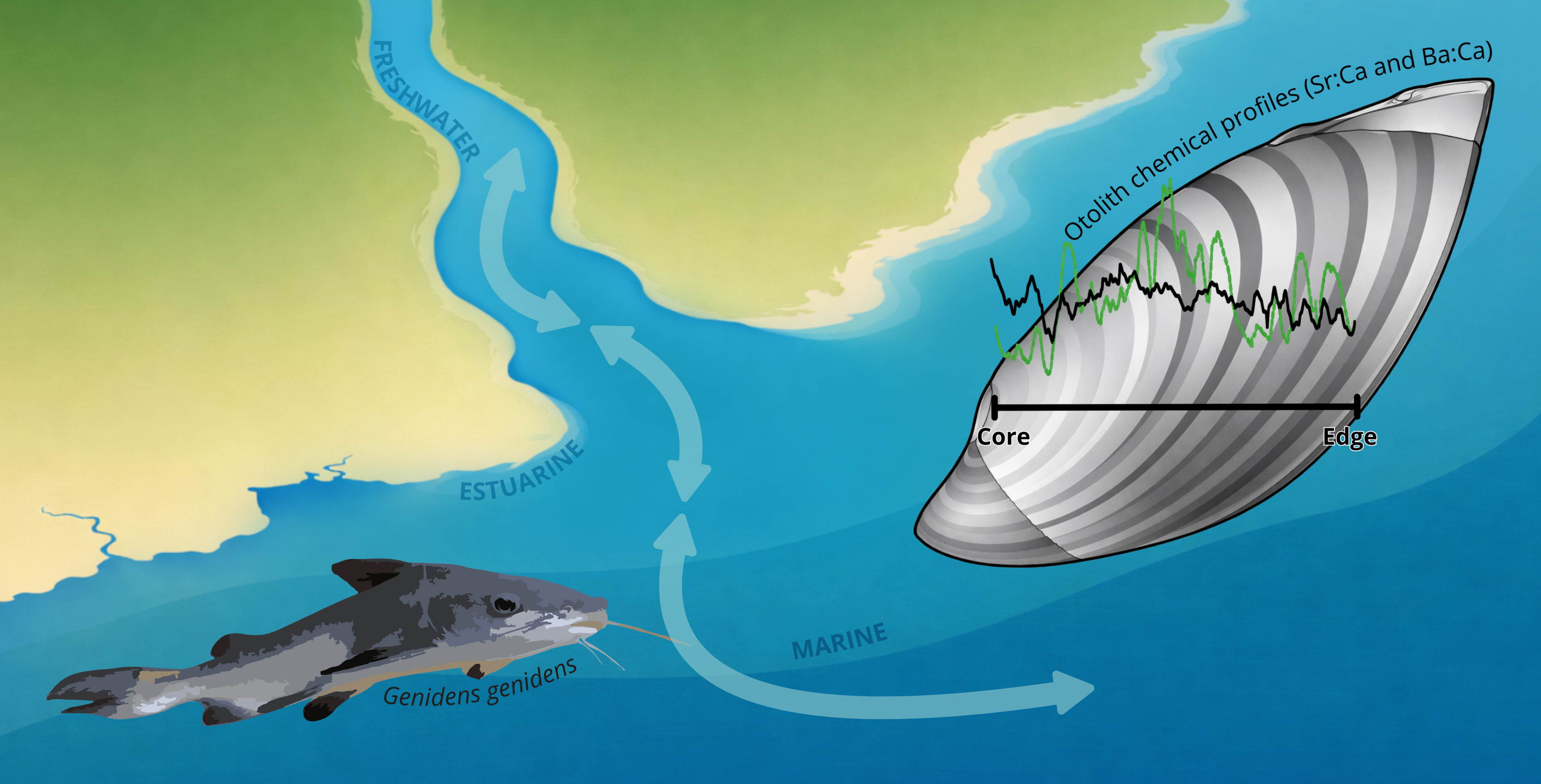
Keywords:
1. Introduction
2. Materials and Methods
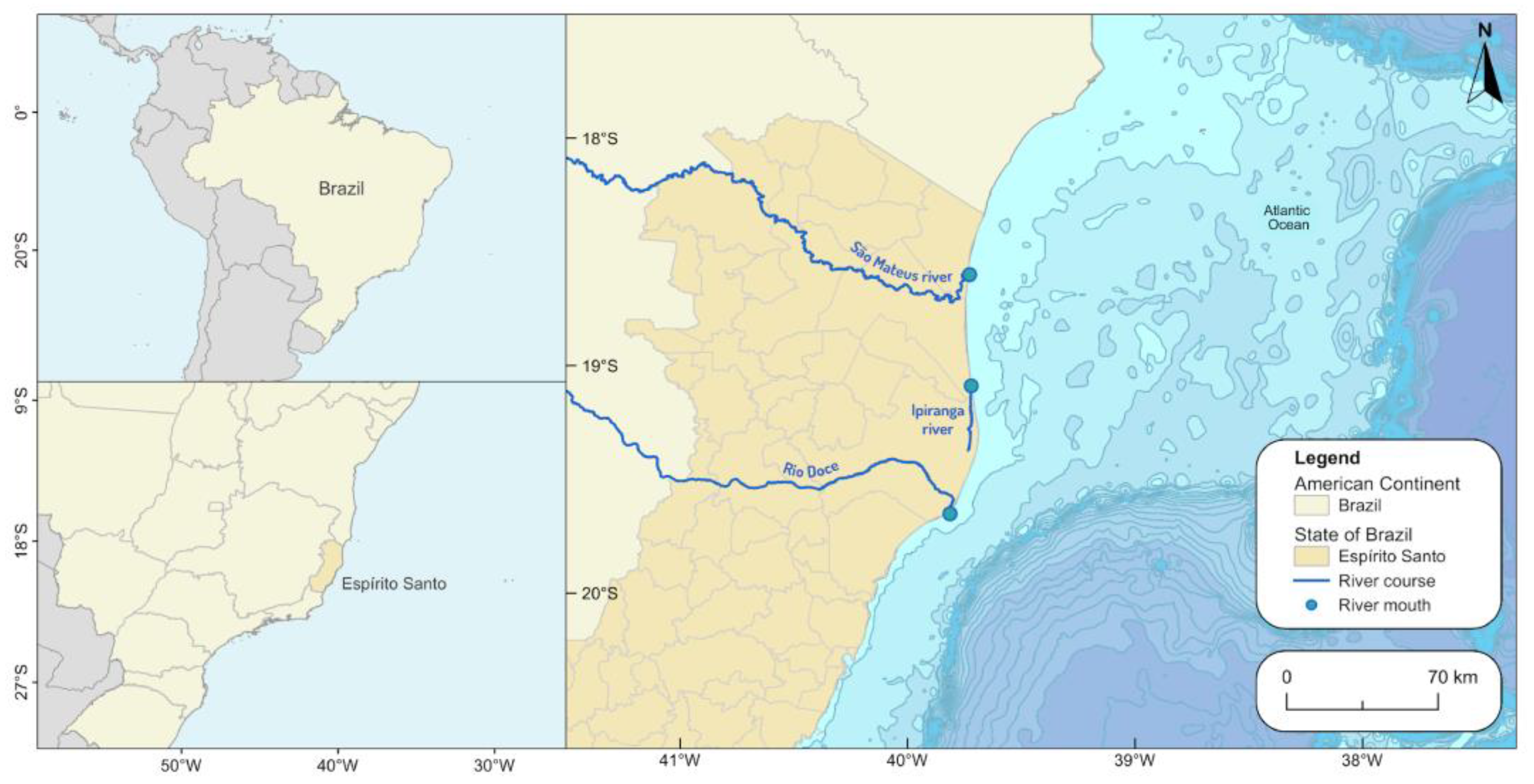
2.1. Study Area
2.2. Local Environmental Conditions
2.3. Sampling and Sample Processing
2.4. Microchemical Analysis
2.5. Age Estimation
2.6. Data Analysis
3. Results
3.1. General Description of the Sample
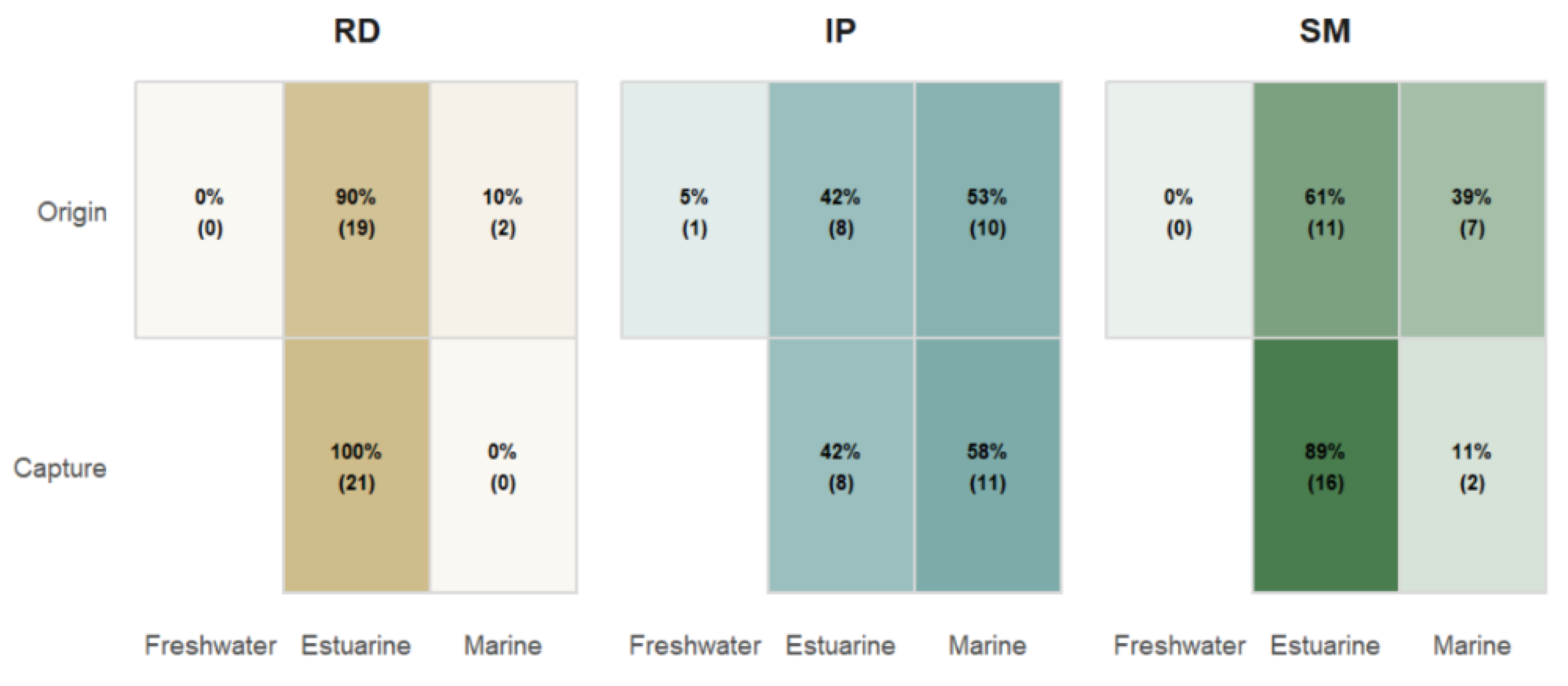
3.2. Environmental Classification Based on Sr:Ca Ratios
3.3. Individual Sr:Ca Profiles and Ontogenetic Habitat-Use Patterns
3.4. Relationship Between Age, Length, and Chemical Patterns
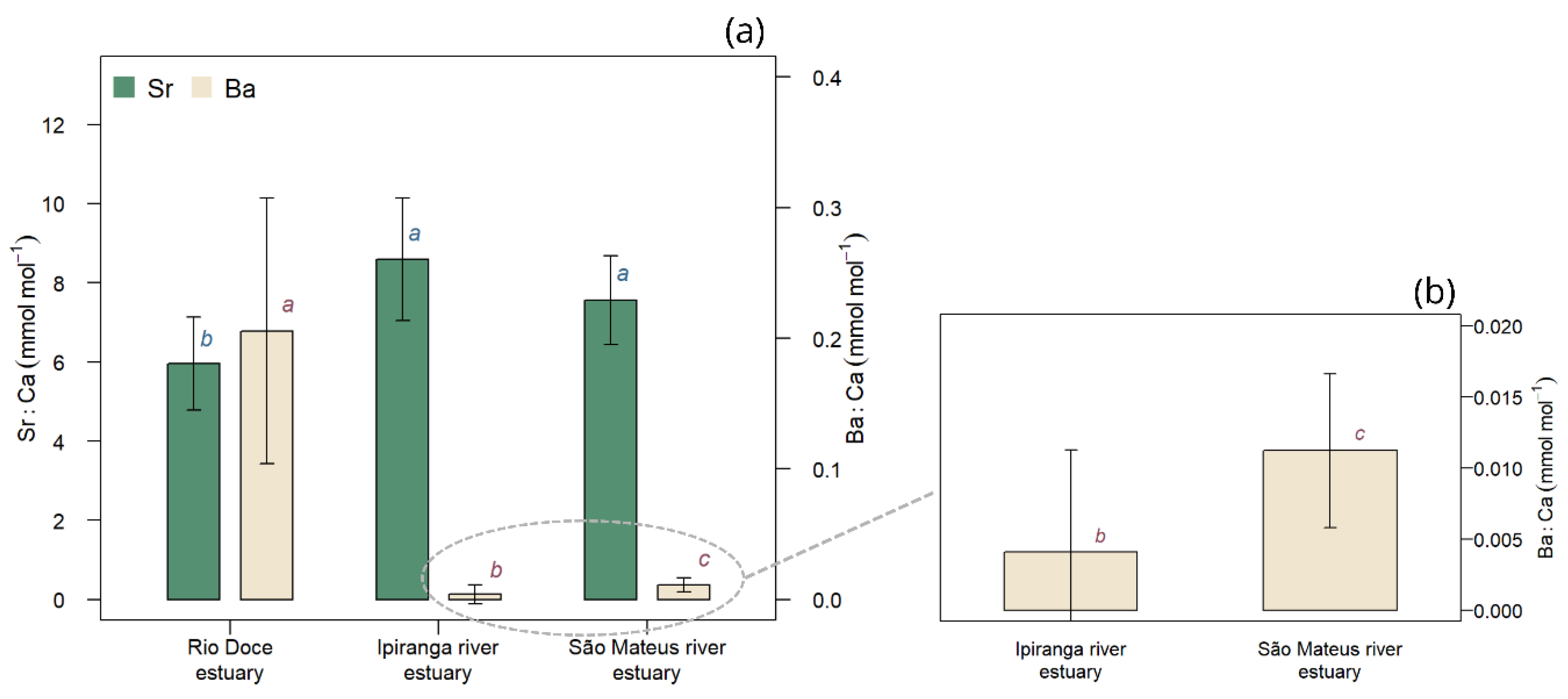
3.5. Differences in Ba:Ca Profiles Among Estuaries
4. Discussion
4.1. General Characteristics of the Sample and Environmental Context
4.2. Spatial Variation in Sr:Ca and Environmental Use
4.3. Ontogenetic Patterns of Habitat Use and Partial Migration
4.4. Ontogenetic Changes in Mobility and Profile Complexity
4.5. Regional Variation in Ba:Ca and Continental Influence
4.6. Ecological Implications, Management Considerations, and Future Directions
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Abbreviations
| ANCOVA | Analysis of Covariance |
| APE | Average Percentage Error |
| Ba:Ca | Barium-to-calcium ratio |
| CP | Change point |
| CPA | Change-point analysis |
| IP | Ipiranga River estuary |
| LA-HR-ICP-MS | Laser ablation high-resolution inductively coupled plasma mass spectrometry |
| RD | Rio Doce estuary |
| SM | São Mateus River estuary |
| Sr:Ca | Strontium-to-calcium ratio |
| TL | Total length |
References
- Potter, I.C.; Warwick, R.M.; Hall, N.G.; Tweedley, J.R. The physico-chemical characteristics, biota and fisheries of estuaries. In: Freshwater Fisheries Ecology, Craig, J.F., Ed.; John Wiley & Sons: Chichester, UK, 2015; Chapter 5. [CrossRef]
- Cabral, H.N.; Arevalo, E.; Carassou, L. Estuaries: Patterns and trends on ecosystem structure and functioning under global changes. In: Marine Ecology—An Ecosystemic View of Anthropogenic Impacts, Molina, J.M., Blassina, G.E., Eds.; CRC Press: Boca Raton, FL, USA, 2025; Volume 2, pp. 165–200. [CrossRef]
- Gillanders, B.M.; McMillan, M.N.; Reis-Santos, P.; Baumgartner, L.J.; Brown, L.R.; Conallin, J.; Feyrer, F.V.; Henriques, S.; James, N.C.; Jaureguizar, A.J.; Pessanha, A.L.M.; Vasconcelos, R.P.; Vu, A.V.; Walther, B.; Wibowo, A. Climate Change and Fishes in Estuaries. In: Fish and Fisheries in Estuaries, Whitfield, A.K., Able, K.W., Blaber, S.J.M., Elliott, M., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 2022; Volume 1, pp. 380–457. [CrossRef]
- Whitfield, A.K.; Able, K.W.; Blaber, S.J.M.; Elliott, M. Fish and Fisheries in Estuaries: A Global Perspective; John Wiley & Sons: Hoboken, NJ, USA, 2022. [CrossRef]
- Peterson, M.S. A conceptual view of environment-habitat-production linkages in tidal river estuaries. Rev. Fish. Sci. 2003, 11, 291–313. [CrossRef]
- Sheaves, M.; Baker, R.; Nagelkerken, I. et al. True value of estuarine and coastal nurseries for fish: incorporating complexity and dynamics. Estuaries Coasts 2015, 38, 401–414. [CrossRef]
- Elliott, M.; Whitfield, A.K.; Potter, I.C.; Blaber, S.J.M.; Cyrus, D.P.; Nordlie, F.G.; Harrison, T.D. The guild approach to categorizing estuarine fish assemblages: a global review. Fish Fish. 2007, 8, 241–268. [CrossRef]
- Elsdon, T.S.; Gillanders, B.M.; Campana, S.E.; Jones, C.M.; Kerr, L.A.; Secor, D.H.; Thorrold, S.R.; Walther, B.D. Otolith chemistry to describe movements and life-history parameters of fishes: hypotheses, assumptions, limitations and inferences. In: Oceanography and Marine Biology: An Annual Review, Gibson, R.N., Atkinson, R.J.A., Gordon, J.D.M., Eds.; CRC Press: Boca Raton, FL, USA, 2008; Volume 46, pp. 297–330. [CrossRef]
- Campana, S.E. Chemistry and composition of fish otoliths: pathways, mechanisms and applications. Mar. Ecol. Prog. Ser. 1999, 188, 263–297. [CrossRef]
- Gillanders, B.M. Using elemental chemistry of fish otoliths to determine connectivity between estuarine and coastal habitats. Estuar. Coast. Shelf Sci. 2005, 64, 47–57. [CrossRef]
- Walther, B.D.; Limburg, K.E.; Jones, C.M.; Schaffler, J.J. Frontiers in otolith chemistry: insights, advances and applications. J. Fish Biol. 2017, 90, 473–479. [CrossRef]
- Campana, S.E.; Thorrold, S.R. Otoliths, increments, and elements: keys to a comprehensive understanding of fish populations? Can. J. Fish. Aquat. Sci. 2001, 58, 30–38. [CrossRef]
- Elsdon, T.S.; Gillanders, B.M. Interactive effects of temperature and salinity on otolith chemistry: challenges for determining environmental histories of fish. Can. J. Fish. Aquat. Sci. 2002, 59, 1796–1808. [CrossRef]
- Sturrock, A.M.; Trueman, C.N.; Darnaude, A.M.; Hunter, E. Can otolith elemental chemistry retrospectively track migrations in fully marine fishes? J. Fish Biol. 2012, 81, 766–795. [CrossRef]
- Walther, B.D.; Limburg, K.E. The use of otolith chemistry to characterize diadromous migrations. J. Fish Biol. 2012, 81, 796–825. [CrossRef]
- Soeth, M.; Spach, H.L.; Daros, F.A.; Castro, J.P.; Correia, A.T. Use of otolith elemental signatures to unravel lifetime movement patterns of Atlantic spadefish, Chaetodipterus faber, in the Southwest Atlantic Ocean. J. Sea Res. 2020, 158, 101873. [CrossRef]
- Menezes, R.; Moura, P.E.; Santos, A.C.; Moraes, L.E.; Condini, M.V.; Rosa, R.S.; Albuquerque, C.Q. Habitat use plasticity by the dog snapper (Lutjanus jocu) across the Abrolhos Bank shelf, eastern Brazil, inferred from otolith chemistry. Estuar. Coast. Shelf Sci. 2021, 263, 107637. [CrossRef]
- Hermann, T.W.; Stewart, D.J.; Limburg, K.E.; Castello, L. Unravelling the life history of Amazonian fishes through otolith microchemistry. R. Soc. Open Sci. 2016, 3, 160206. [CrossRef]
- Austin, C.S.; Bond, M.H.; Smith, J.M.; Lowery, E.D.; Quinn, T.P. Otolith microchemistry reveals partial migration and life history variation in a facultatively anadromous, iteroparous salmonid, bull trout (Salvelinus confluentus). Environ. Biol. Fishes 2019, 102, 95–104. [CrossRef]
- Avigliano, E.; Carvalho, B.; Velasco, G.; Tripodi, P.; Vianna, M.; Volpedo, A.V. Nursery areas and connectivity of the adults anadromous catfish (Genidens barbus) revealed by otolith-core microchemistry in the south-western Atlantic Ocean. Mar. Freshw. Res. 2017, 68, 931–940. [CrossRef]
- Zydlewski, J.; Wilkie, M.P. Freshwater to seawater transitions in migratory fishes. In: Fish Physiology, McCormick, S.D., Farrell, A.P., Brauner, C.J., Eds.; Academic Press: San Diego, CA, USA, 2012; Volume 32, pp. 253–326. [CrossRef]
- Nazir, A.; Khan, M.A. Using otoliths for fish stock discrimination: Status and challenges. Acta Ichthyol. Piscat. 2021, 51, 199–218. [CrossRef]
- Whitfield, A.K.; Able, K.W.; Barletta, M.; Blaber, S.J.M.; Harrison, T.D. Life-history guilds of fishes associated with estuaries: opportunism versus dependency. Estuar. Coast. Shelf Sci. 2023, 292, 108456. [CrossRef]
- Avigliano, E.; Volpedo, A.V. A review of the application of otolith microchemistry toward the study of Latin American fishes. Rev. Fish. Sci. Aquac. 2016, 24, 369–384. [CrossRef]
- Ceni, G.; Fontoura, N.F.; Cabral, H.N. The freshwater artisanal fishery of Patos Lagoon. J. Fish Biol. 2016, 89, 337–354. [CrossRef]
- Maciel, T.R.; Vaz-dos-Santos, A.M.; Caramaschi, E.P.; Vianna, M. Management proposal based on the timing of oral incubation of eggs and juveniles in the sentinel species Genidens genidens (Siluriformes: Ariidae) in a tropical estuary. Neotrop. Ichthyol. 2018, 16, e170119. [CrossRef]
- Daros, F.A.; Condini, M.V.; Altafin, J.P.; de Oliveira Ferreira, F.; Hostim-Silva, M. Fish otolith microchemistry as a biomarker of the world's largest mining disaster. Sci. Total Environ. 2022, 807*, 151780. [CrossRef]
- Condini, M.V.; Pereyra, P.E.R.; Garcia, A.M.; Saint’Pierre, T.D.; Ceni, G.; Lugo, R.; Fontoura, N.F.; Vieira, J.P.; Albuquerque, C.Q. Use of fresh water by an estuarine-resident marine catfish: evidence from gonadal and otolith chemistry analyses. J. Mar. Biol. Assoc. U. K. 2019, 99*, 1667–1674. [CrossRef]
- Maciel, T.R.; Moreno, E.; Maichak de Carvalho, B.; Miller, N.; Vianna, M.; Avigliano, E. High-salinity water use of euryhaline catfish Genidens genidens revealed by otolith microchemistry. Austral Ecol. 2024, 49*, e13573. [CrossRef]
- Maciel, T.R.; Avigliano, E.; de Carvalho, B.M.; Miller, N.; Vianna, M. Population structure and habitat connectivity of Genidens genidens (Siluriformes) in tropical and subtropical coasts from Southwestern Atlantic. Estuar. Coast. Shelf Sci. 2020, 242*, 106839. [CrossRef]
- Spalding, M.D.; Fox, H.E.; Allen, G.R.; Davidson, N.; Ferdaña, Z.A.; Finlayson, M.; Halpern, B.S.; Jorge, M.A.; Lombana, A.; Lourie, S.A.; Martin, K.D.; McManus, E.; Molnar, J.; Recchia, C.A.; Robertson, J. Marine Ecoregions of the World: A Bioregionalization of Coastal and Shelf Areas. BioScience 2007, 57, 573–583. [CrossRef]
- Silva, M.A.B.D.; Bernini, E.; Carmo, T.M.S.D. Características estruturais de bosques de mangue do estuário do rio São Mateus, ES, Brasil. Acta Bot. Bras. 2005, 19, 465–471. [CrossRef]
- Tognella, M.M.P.; Falqueto, A.R.; Espinoza, H.D.C.F.; Gontijo, I.; Gontijo, A.B.P.L.; Fernandes, A.A.; Schmidtl, E.R.; Soares, M.L.G.; Chaves, F.O.; Schmidt, A.J.; Lopes, D.M.S.; Barcelos, U.D.; d’Addazio, V.; Lima, K.O.O.; Pascoalini, S.S.; Paris, J.O.; Brites Júnior, N.V.; Porto, L.A.; Almeida Filho, E.; Oliveira, C.P.; Leopoldo, R.V.S.; Leite, S.; Berribilli, M.P.; Vieiras, S.F.R.; Rosa, M.B.; Sá, F.; Rodrigues Neto, R.; Ghisolfi, R.D.; Castro, M.S.M.; Rigo, D.; Tosta, V.C.; Albino, J. Mangroves as traps for environmental damage to metals: The case study of the Fundão Dam. Sci. Total Environ. 2022, 806*, 150452. [CrossRef]
- Bernini, E.; da Silva, M.A.; Carmo, T.; Cuzzuol, G.R. Spatial and temporal variation of the nutrients in the sediment and leaves of two Brazilian mangrove species and their role in the retention of environmental heavy metals. Braz. J. Plant Physiol. 2010, 22, 177–187. [CrossRef]
- Bolzan, M.S.; Andrades, R.; Spach, H.L.; Hostim-Silva, M. The influence of selected environmental parameters and habitat mosaics on fish assemblages in a South American estuary. J. Mar. Biol. Assoc. U. K. 2018, 99*, 249–257. [CrossRef]
- Castro, D.F.; Rossetti, D.F.; Cohen, M.C.L.; Pessenda, L.C.R.; Lorente, F.L. The growth of the Doce River Delta in northeastern Brazil indicated by sedimentary facies and diatoms. Diatom Res. 2013, 28, 455–466. [CrossRef]
- Kütter, V.T.; Martins, G.S.; Brandini, N.; Cordeiro, R.C.; Almeida, J.P.A.; Marques, E.D. Impacts of a tailings dam failure on water quality in the Doce River: The largest environmental disaster in Brazil. J. Trace Elem. Miner. 2023, 5, 100084. [CrossRef]
- Condini, M.V.; Pichler, H.A.; Oliveira-Filho, R.R.; Cattani, A.P.; Andrades, R.; Vilar, C.C.; Joyeux, J.-C.; Soeth, M.; de Biasi, J.B.; Eggertsen, L.; Dias, R.; Hackradt, C.W.; Félix-Hackradt, F.C.; Chiquieri, J.; Garcia, A.M.; Hostim-Silva, M. Marine fish assemblages of Eastern Brazil: An update after the world’s largest mining disaster and suggestions of functional groups for biomonitoring long-lasting effects. Sci. Total Environ. 2022, 807*, 150987. [CrossRef]
- Oliveira, R.L.; Camara, E.M.; Condini, M.V.; Oliveira-Filho, R.R.; Pichler, H.A.; Andrades, R.; Vilar, C.C.; Spach, H.L.; Joyeux, J.-C.; Hostim-Silva, M. Ecological uniqueness of fish assemblages in tropical estuarine and coastal systems: Assessing environmental and spatial drivers. Estuar. Coast. Shelf Sci. 2025, 313, 109111. [CrossRef]
- Schettini, C.A.F.; Hatje, V. The Suspended Sediment and Metals Load from the Mariana’s Tailing Dam Failure to the Coastal Sea. Integr. Environ. Assess. Manag. 2020, 16, 661–668. [CrossRef]
- Vilar, C.C.; Andrades, R.; Szab lak, F.T.; Guabiroba, H.C.; Pichler, H.A.; Bastos, K.V.; Lima, L.R.S.; Bastos, P.G.P.; Martins, R.F.; Rodrigues, V.L.A.; Hostim-Silva, M.; Joyeux, J.-C. Variability in nearshore fish biodiversity indicators after a mining disaster in eastern Brazil. Mar. Environ. Res. 2022, 175, 105565. [CrossRef]
- Webb, S.D.; Woodcock, S.H.; Gillanders, B.M. Sources of otolith barium and strontium in estuarine fish and the influence of salinity and temperature. Mar. Ecol. Prog. Ser. 2012, 453, 189–199. [CrossRef]
- Sirot, C.; Ferraton, F.; Panfili, J.; Childs, A.-R.; Guilhaumon, F.; Darnaude, A.M. elementr: An R package for reducing elemental data from LA-ICPMS analysis of biological calcified structures. Methods Ecol. Evol. 2017, 8, 1659–1667. [CrossRef]
- Ferreira, I.; Daros, F.A.; Moreira, C.; Feijó, D.; Rocha, A.; Mendez-Vicente, A.; Pisonero, J.; Correia, A.T. Is Chelidonichthys lucerna (Linnaeus, 1758) a Marine Estuarine-Dependent Fish? Insights from Saccular Otolith Microchemistry. Fishes 2023, 8, 383. [CrossRef]
- Giraudoux, P.; Antonietti, J.P.; Beale, C.; Pleydell, D.; Treglia, M. pgirmess: Spatial analysis and data mining for field ecologists. R package version 1.9.678, 2018.
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2025. Available online: https://www.R-project.org/.
- Killick, R.; Eckley, I.A. changepoint: An R Package for Changepoint Analysis. J. Stat. Softw. 2014, 58, 1–19. [CrossRef]
- Gomes, L.E.de O.; Correa, L.B.; Sá, F.; Neto, R.R.; Bernardino, A.F. The impacts of the Samarco mine tailing spill on the Rio Doce estuary, Eastern Brazil. Mar. Pollut. Bull. 2017, 120*, 28–36. [CrossRef]
- Daros, F.A.; Spach, H.L.; Correia, A.T. Habitat residency and movement patterns of Centropomus parallelus juveniles in a subtropical estuarine complex. J. Fish Biol. 2016, 88, 1796–1810. [CrossRef]
- Quaresma, V.D.S.; Catabriga, G.; Bourguignon, S.N.; Godinho, E.; Bastos, A.C. Modern sedimentary processes along the Doce river adjacent continental shelf. Braz. J. Geol. 2015, 45, 635–644. [CrossRef]
- Avigliano, E.; Velasco, G.; Volpedo, A.V. Use of lapillus otolith microchemistry as an indicator of the habitat of Genidens barbus from different estuarine environments in the southwestern Atlantic Ocean. Environ. Biol. Fishes 2015, 98, 1623–1632. [CrossRef]
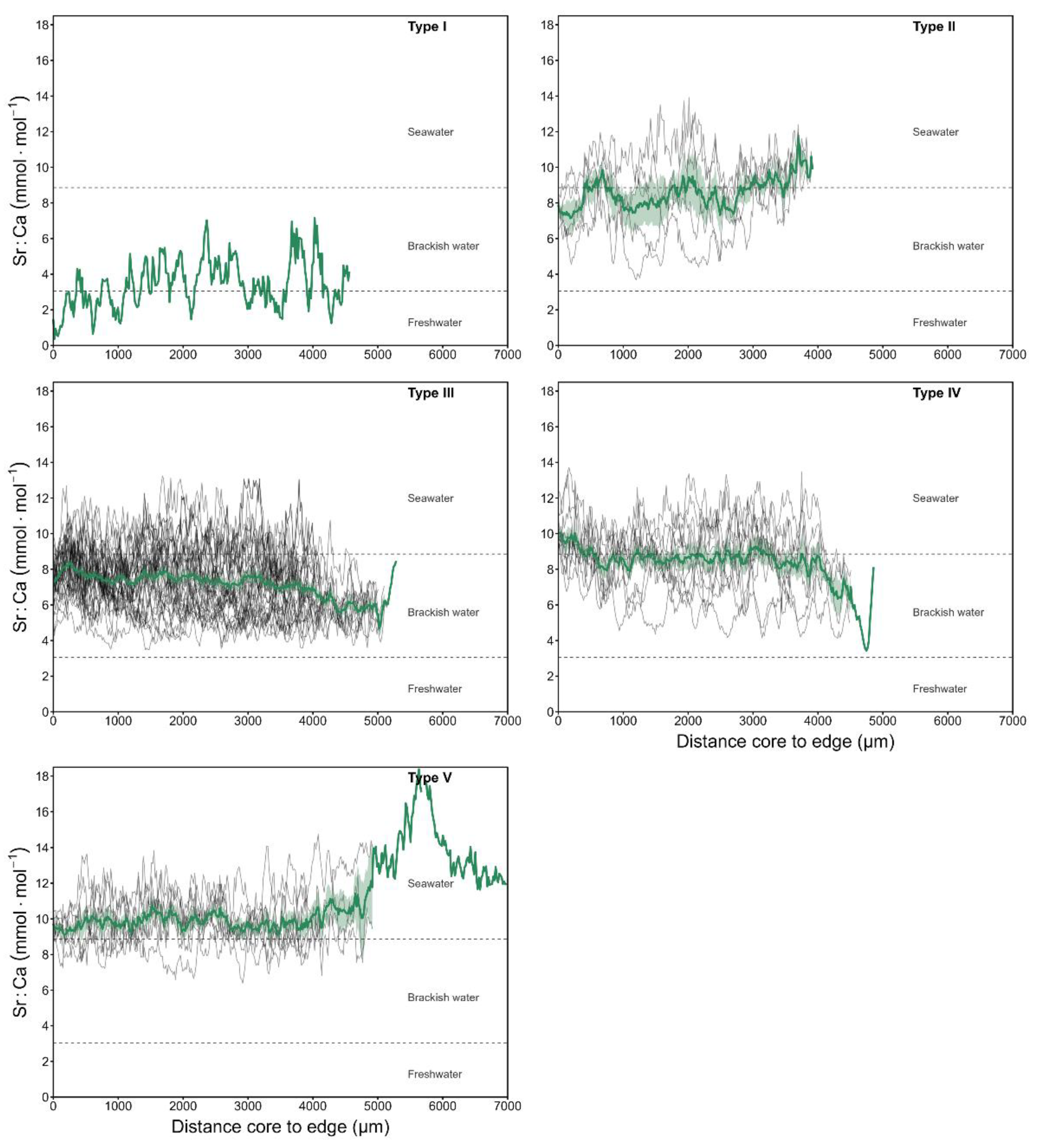
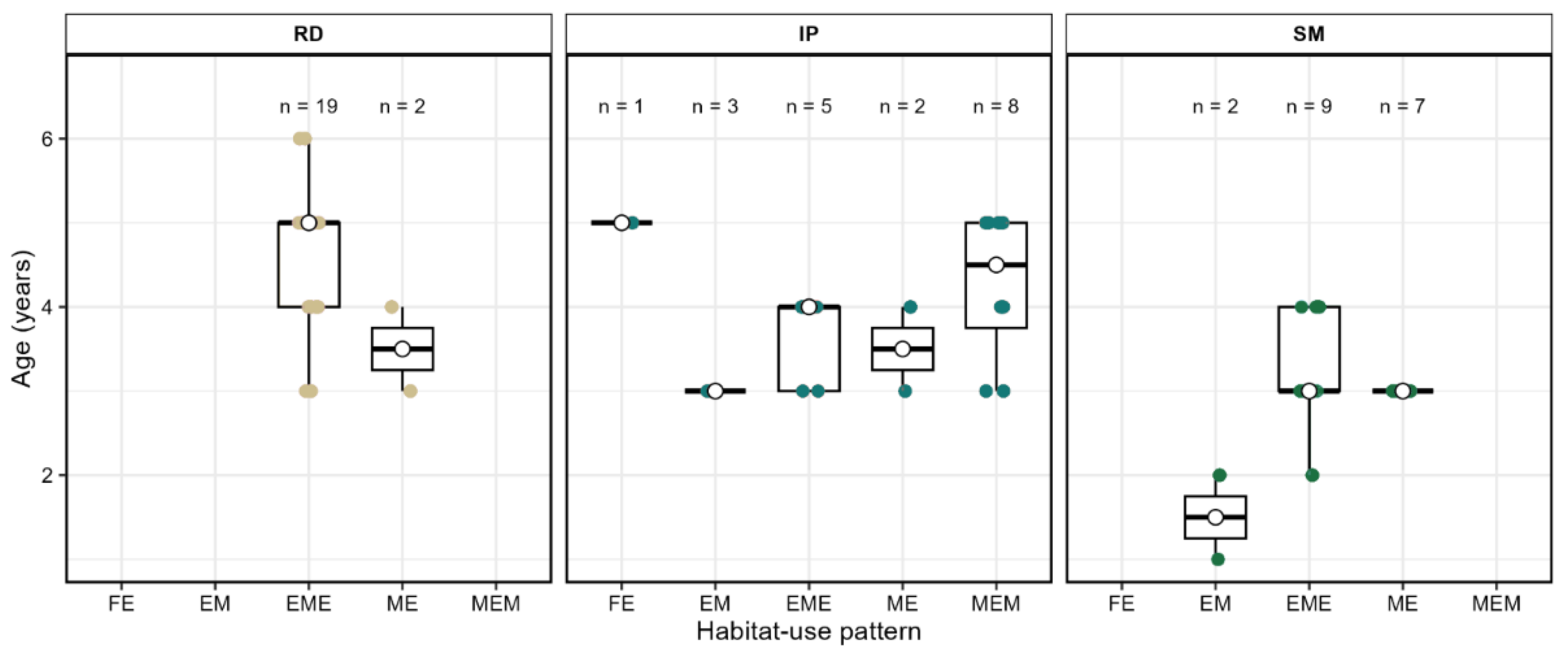
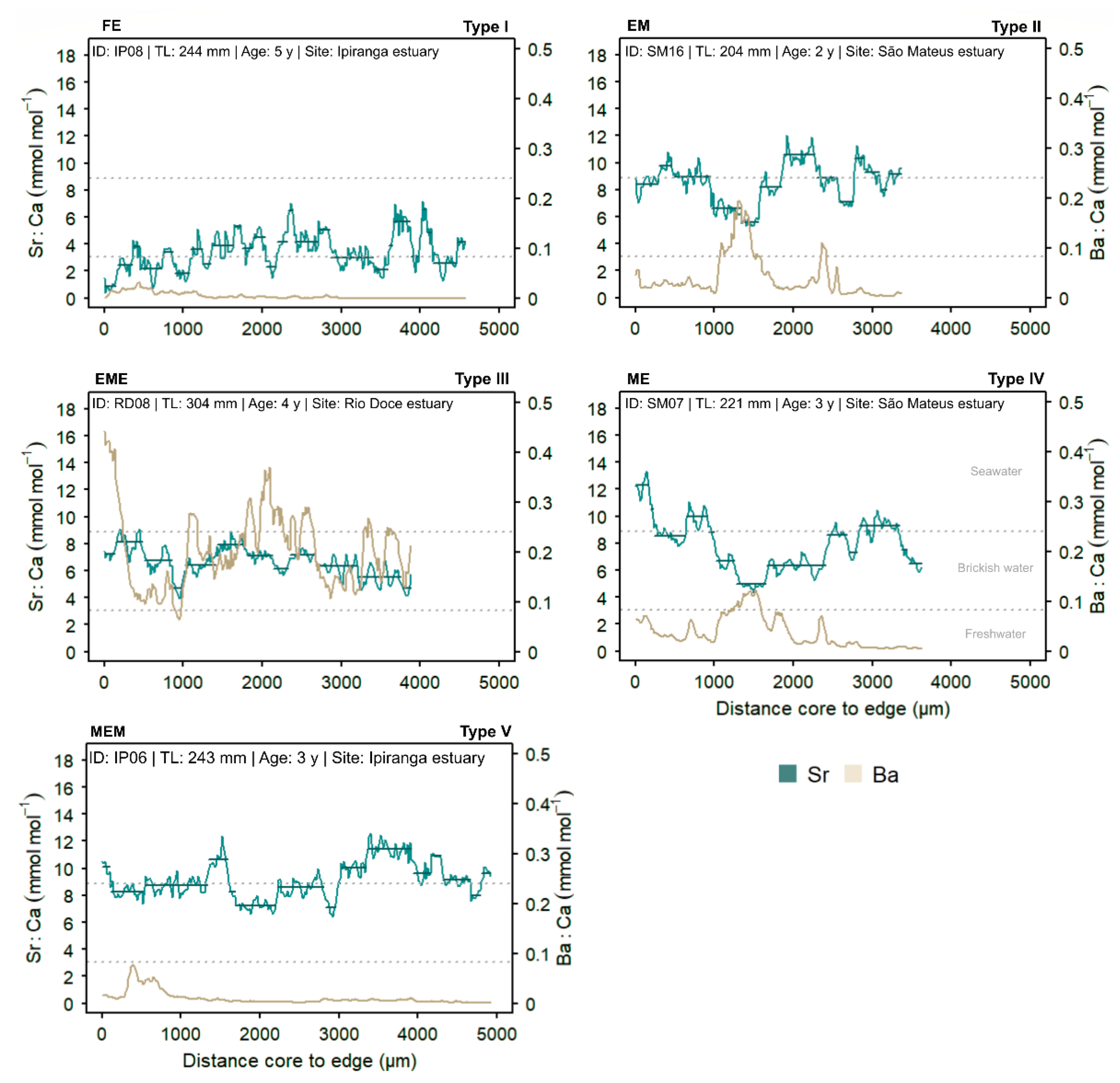
| Type | Symbol | Trajectory | Description |
| I | FE | Freshwater → Estuarine | Individuals born in freshwater that migrate to the estuary. |
| II | EM | Estuarine → Marine | Born in estuarine waters and moved to the marine environment. |
| III | EME | Estuarine → Marine → Estuarine | Origin and pre-capture in estuary, with intermediate incursions into marine waters. |
| IV | ME | Marine → Estuarine | Individuals of marine origin that enter the estuary during growth. |
| V | MEM | Marine → Estuarine → Marine | Fish of marine origin that made temporary movements to the estuary and later returned to the sea. |
| Site | n | TL (mm) | Age (Years) | Ba:Ca (mmol mol-1) | Sr:Ca (mmol mol-1) |
| Rio Doce estuary | 21 | 289.7 ± 18.2a (250 – 315) |
4.5 ± 0.9a (3 - 6) |
0.21 ± 0.11a (0.000009 - 0.75) |
6.62 ± 1.57b (0.0044 – 13.1) |
| Ipiranga estuary | 19 | 252.8 ± 33.3b (232 – 382) |
3.9 ± 0.8a (3 - 5) |
0.02 ± 0.05b (0.000001 - 0.91) |
9.12 ± 2.28a (0.0044 – 18.9) |
| São Mateus estuary | 18 | 222.2 ± 16.5c (199 - 251) |
2.9 ± 0.7b (1 - 4) |
0.03 ± 0.03c (0.000008 - 0.24) |
8.43 ± 1.75a (0.0054 – 13.7) |
| Site | n | Type I (FE) | Type II (EM) | Type III (EME) | Type IV (ME) | Type V (MEM) |
| Rio Doce estuary | 21 | 0 | 0 | 19 (90.5%) | 2 (9.52%) | 0 |
| Ipiranga estuary | 19 | 1 (5.3%) | 3 (15.8%) | 3 (15.8%) | 2 (10.5%) | 8 (42.1%) |
| São Mateus estuary | 18 | 0 | 1 (5.6%) | 9 (50.0%) | 7 (38.9%) | 1 (5.6%) |
| Total | 58 | 1 (1.7%) | 5 (8.6%) | 31 (56.9%) | 11 (19.0%) | 8 (13.8%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




