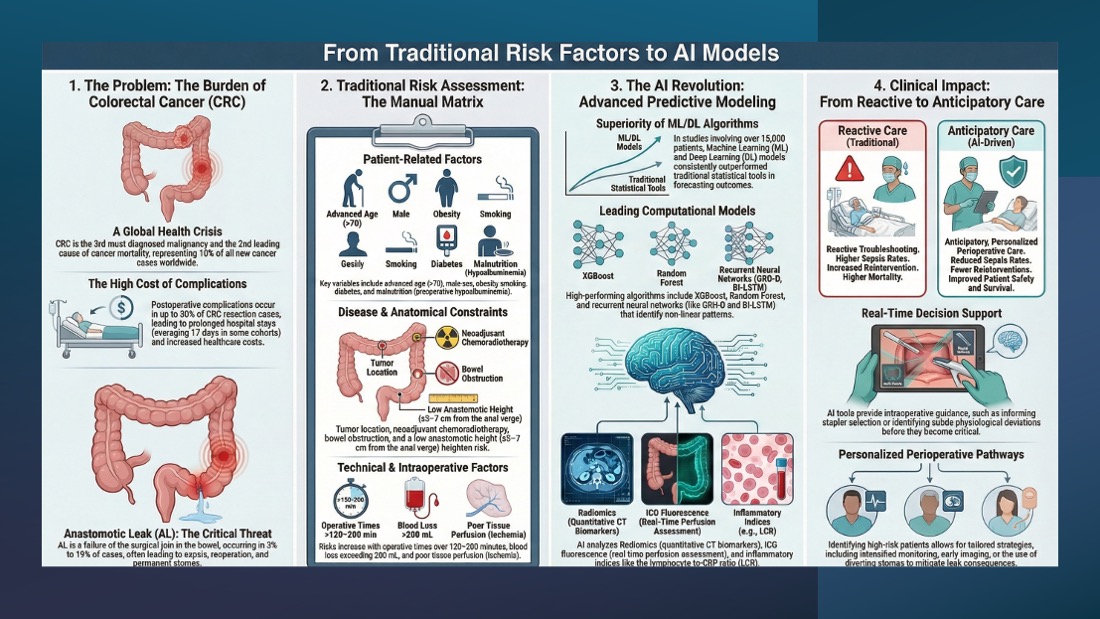
Colorectal cancer (CRC) represents a major global health burden, accounting for roughly 10% of all newly diagnosed cancers and cancer-related deaths worldwide. It is the third most diagnosed malignancy and the second leading cause of cancer mortality according to the World Health Organization. Postoperative complications remain a significant concern after CRC resection, occurring in up to 50% of patients and contributing to increased morbidity, mortality, prolonged hospitalization, and substantial healthcare expenditure. Artificial intelligence (AI) has emerged as a transformative tool in modern healthcare, offering advanced capabilities in predictive analytics, clinical decision support, and personalized perioperative management. The present review systematically evaluates the application of AI, specifically machine learning (ML) and deep learning (DL) algorithms, in predicting anastomotic leak (AL) and other major postoperative complications. AI models aim to refine risk stratification and enhance surgical decision-making. A total of 13 studies were included, encompassing 15,105 patients. Across these studies, ML and DL algorithms consistently outperformed conventional statistical models in forecasting postoperative outcomes. Current evidence suggests that AI has substantial potential to improve perioperative risk prediction, support intraoperative decision-making, and personalize postoperative surveillance in CRC surgery. Methodological limitations including high risk of bias, limited external validation, heterogeneous outcome definitions, and inconsistent reporting necessitate more robust, prospective, multicenter research before widespread clinical adoption can be realized.