Submitted:
02 March 2026
Posted:
03 March 2026
You are already at the latest version
Abstract
Keywords:
Introduction
2. Materials and Methods
2.1. Study Design and Ethical Approval
2.2. Study Population and Sample Selection
- -
- Age ≤ 19 years at the time of examination
- -
- Absence of craniofacial syndromes or congenital anomalies
- -
- No history of previous orthodontic treatment
- -
- Availability of high-quality standardized lateral cephalometric radiographs
- -
- Systemic conditions known to affect growth or bone metabolism
- -
- History of craniofacial trauma or surgery
- -
- Radiographs with positioning errors or insufficient diagnostic quality
2.3. Anthropometric Assessment and BMI Classification
- -
- Normal weight: BMI between the 5th and 85th percentile
- -
- Overweight: BMI between the 85th and 95th percentile
- -
- Obesity: BMI ≥ 95th percentile
2.4. Cephalometric Imaging and Analysis
- -
- SNA angle (°)
- -
- SNB angle (°)
- -
- ANB angle (°)
- -
- Maxillary unit length (mm)
- -
- Mandibular unit length (mm)
- -
- Mandibular body length (mm)
- -
- Facial convexity angle (°)
- -
- Lower-to-total facial height ratio (%)
2.5. Measurement Reliability
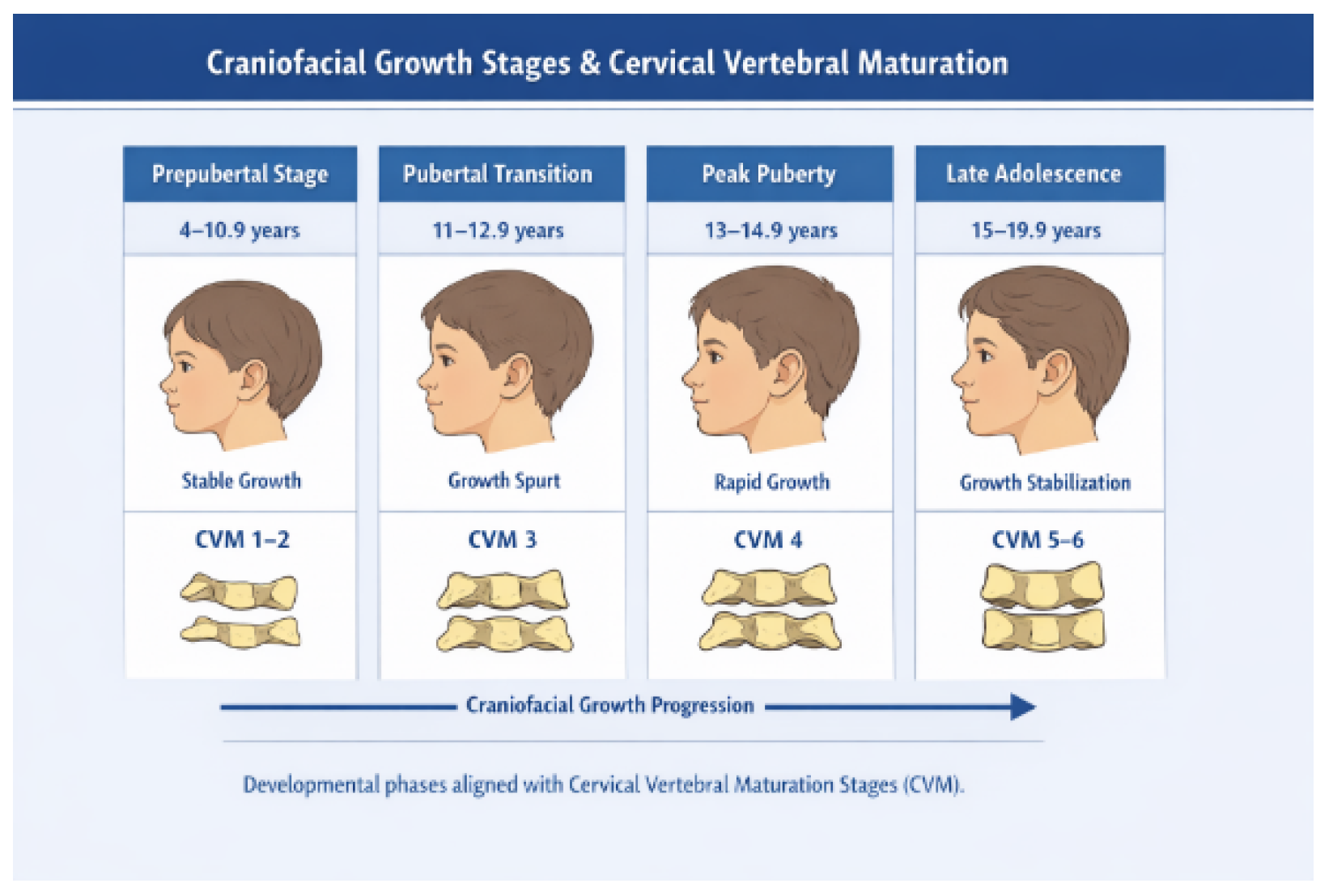
2.6. Age Stratification and Growth Phase Classification
- -
- 4.0–10.9 years: prepubertal growth phase
- -
- 11.0–12.9 years: pubertal onset phase
- -
- 13.0–14.9 years: peak pubertal growth phase
- -
- 15.0–19.9 years: post-pubertal maturation phase
2.7. Statistical Analysis
- Normality assessment using the Shapiro–Wilk test
- Descriptive statistics reported as mean ± standard deviation
- One-way ANOVA to evaluate differences among BMI groups
- Tukey post-hoc testing for pairwise comparisons
- Independent t-tests to assess sex differences
- Two-way ANOVA to examine independent effects of age and BMI
- Multivariate linear regression analysis to determine independent predictors of craniofacial parameters after adjustment for age and sex. Ninety-five percent confidence intervals were calculated for all regression coefficients.
2.8. Sample Size Considerations
3. Results
3.1. Sample Characteristics
3.2. Data Distribution
3.3. Craniofacial Differences According to BMI
3.4. Sex-Related Differences
3.5. Age-Related Craniofacial Changes
3.6. Age–BMI Interaction Results
3.7. Multivariate Regression Analysis
| Dependent Variable | Predictor | β Coefficient | Standard Error | p Value |
|---|---|---|---|---|
| SNB (°) | BMI | 0.18 | 0.05 | <0.001 |
| Age | 0.22 | 0.04 | <0.001 | |
| Sex (Male) | 0.09 | 0.03 | 0.014 | |
| Mandibular unit length (mm) | BMI | 0.36 | 0.07 | <0.001 |
| Age | 0.48 | 0.06 | <0.001 | |
| Sex (Male) | 0.21 | 0.05 | 0.002 | |
| ANB (°) | BMI | −0.14 | 0.04 | 0.001 |
| Age | −0.09 | 0.03 | 0.019 | |
| Sex (Male) | −0.05 | 0.02 | 0.087 | |
| Facial convexity angle (°) | BMI | −0.31 | 0.06 | <0.001 |
| Age | −0.28 | 0.05 | <0.001 | |
| Sex (Male) | −0.07 | 0.03 | 0.041 |
Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ebbeling, C.B.; Pawlak, D.B.; Ludwig, D.S. Childhood obesity: Public-health crisis, common-sense cure. Lancet 2002, 360, 473–479. [Google Scholar] [CrossRef]
- Kant, A.K.; Graubard, B.I. Secular trends in regional differences in nutritional biomarkers and self-reported dietary intakes among American adults: National Health and Nutrition Examination Survey (NHANES) 1988–1994 to 2009–2010. Public Health Nutr. 2018, 21, 927–939. [Google Scholar] [CrossRef] [PubMed]
- Thorpe, L.E.; List, D.G.; Marx, T.; May, L.; Helgerson, S.D.; Frieden, T.R. Childhood obesity in New York City elementary school students. Am. J. Public Health 2004, 94, 1496–1500. [Google Scholar] [CrossRef] [PubMed]
- Vourdoumpa, A.; Paltoglou, G.; Charmandari, E. The genetic basis of childhood obesity: A systematic review. Nutrients 2023, 15, 1416. [Google Scholar] [CrossRef]
- Verma, M.; Kapoor, N.; Senapati, S.; Singh, O.; Bhadoria, A.S.; Khetarpal, P.; Kumar, S.; Bansal, K.; Ranjan, R.; Kakkar, R.; et al. Comprehending the epidemiology and aetiology of childhood obesity: Integrating life course approaches for prevention and intervention. Diabetes Ther. 2025, 16, 1177–1206. [Google Scholar] [CrossRef]
- Narang, I.; Mathew, J.L. Childhood obesity and obstructive sleep apnea. J. Nutr. Metab. 2012, 2012, 134202. [Google Scholar] [CrossRef] [PubMed]
- Jebeile, H.; Kelly, A.S.; O’Malley, G.; Baur, L.A. Obesity in children and adolescents: Epidemiology, causes, assessment, and management. Lancet Diabetes Endocrinol. 2022, 10, 351–365. [Google Scholar] [CrossRef]
- Harrist, A.W.; Swindle, T.M.; Hubbs-Tait, L.; Topham, G.L.; Shriver, L.H.; Page, M.C. The social and emotional lives of overweight, obese, and severely obese children. Child Dev. 2016, 87, 1564–1580. [Google Scholar] [CrossRef]
- Verdecchia, A.; Suárez-Fernández, C.; Menéndez Diaz, I.; Sanz, V.G.; Spinas, E.; Cobo, T. Obesity and overweight conditions in children and adolescents (6–18 years) and their impact on craniofacial morphology: A systematic review. Children 2025, 12, 377. [Google Scholar] [CrossRef]
- Hancock, S.; Carmack, A.; Kocher, M.; Rezende Silva, E.; Sulkowski, T.; Nanney, E.; Graves, C.; Mitchell, K.; Jacox, L.A. Influence of BMI percentile on craniofacial morphology and development in adolescents. Part II: Elevated BMI is associated with larger final facial dimensions. Eur. J. Orthod. 2024, 46, cjad043. [Google Scholar] [CrossRef]
- Lee, R.W.; Vasudavan, S.; Hui, D.S.; Prvan, T.; Petocz, P.; Darendeliler, M.A.; Cistulli, P.A. Differences in craniofacial structures and obesity in Caucasian and Chinese patients with obstructive sleep apnea. Sleep 2010, 33, 1075–1080. [Google Scholar] [CrossRef]
- Gibas-Stanek, M.; Bilińska, M.; Kaminkova, P.; Direr, P.; Fudalej, P.S. Influence of body mass index on facial soft tissue morphology in growing non-obese children. Eur. J. Orthod. 2025, 47, cjaf069. [Google Scholar] [CrossRef]
- Tentaş, S.; Özden, S. Deep learning-based evaluation of skeletal maturation: A comparative analysis of five hand-wrist methods. Orthod. Craniofac. Res. 2025, 28, 943–954. [Google Scholar] [CrossRef]
- Nokes, B.; Schueler, A.; Darquenne, C.; Schmickl, C.N.; Wojeck, B.S.; Moore, S.; Deyoung, P.; McGinnis, L.; Theilmann, R.J.; Gruenberg, E.; et al. The impact of obesity on upper airway anatomy as assessed by magnetic resonance imaging and obstructive sleep apnea endotypic traits. Front. Physiol. 2025, 16, 1648767. [Google Scholar] [CrossRef]
- Al-Jewair, T.; Marwah, S.; Preston, C.B.; Wu, Y.; Yu, G. Correlation between craniofacial structures, anthropometric measurements, and nasopharyngeal dimensions in black adolescents. Int. Orthod. 2021, 19, 96–106. [Google Scholar] [CrossRef] [PubMed]
- de Onis, M.; Onyango, A.W.; Borghi, E.; Siyam, A.; Nishida, C.; Siekmann, J. Development of a WHO growth reference for school-aged children and adolescents. Bull. World Health Organ. 2007, 85, 660–667. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Baccetti, T.; Franchi, L.; McNamara, J.A. The cervical vertebral maturation (CVM) method for the assessment of optimal treatment timing in dentofacial orthopedics. Semin. Orthod. 2005, 11, 119–129. [Google Scholar] [CrossRef]
- McNamara, J.A., Jr.; Franchi, L. The cervical vertebral maturation method: A user’s guide. Angle Orthod. 2018, 88, 133–143. [Google Scholar] [CrossRef]
- Alfaro, J.M.; Manrique, R.; Santamaría, A.; Álvarez, E.; Manes, C.; Jiménez, M. Effects of endocrine disorders on maxillary and mandibular growth in Colombian children and adolescents: A cross-sectional study. Eur. Arch. Paediatr. Dent. 2024, 25, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Danze, A.; Jacox, L.A.; Bocklage, C.; Whitley, J.; Moss, K.; Hardigan, P.; Garcia-Godoy, C.E.; Jackson, T.H. Influence of BMI percentile on craniofacial morphology and development in children and adolescents. Eur. J. Orthod. 2021, 43, 184–192. [Google Scholar] [CrossRef] [PubMed]
- Verdecchia, A.; Torre, I.C.; Diaz, I.M.; Sanz, V.G.; Mesa, Y.G.; Cobo, T.; Gallardo, V.P. Analysis of the relationship between body mass index (BMI) and dento-skeletal maturation: A cross-sectional case-control study. Dent. J. 2025, 13, 8. [Google Scholar] [CrossRef]
- Ke, D.; Lu, D.; Cai, G.; Zhang, J.; Wang, X.; Suzuki, K. Accelerated skeletal maturation is associated with overweight and obesity as early as preschool age: A cross-sectional study. BMC Pediatr. 2020, 20, 452. [Google Scholar] [CrossRef]
- Tsutsumi-Arai, C.; Tran, A.; Arai, Y.; Ono, W.; Ono, N. Mandibular condylar cartilage in development and diseases: A PTHrP-centric view. Orthod. Craniofac. Res. 2025, 28, S70–S80. [Google Scholar] [CrossRef]
- Chen, Y.; Ke, J.; Long, X.; Meng, Q.; Deng, M.; Fang, W.; Li, J.; Cai, H.; Chen, S. Insulin-like growth factor-1 boosts the developing process of condylar hyperplasia by stimulating chondrocyte proliferation. Osteoarthr. Cartil. 2012, 20, 279–287. [Google Scholar] [CrossRef]
- Joshi, A.S.; Hatch, N.E.; Hayami, T.; Jheon, A.; Kapila, S. IGF-1 TMJ injections enhance mandibular growth and bone quality in juvenile rats. Orthod. Craniofac. Res. 2022, 25, 183–191. [Google Scholar] [CrossRef]
- Francisco, V.; Pino, J.; Gonzalez-Gay, M.A.; Mera, A.; Lago, F.; Gómez, R.; Mobasheri, A.; Gualillo, O. Adipokines and inflammation: Is it a question of weight? Br. J. Pharmacol. 2018, 175, 1569–1579. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.X.; Yang, T. Roles of leptin in bone metabolism and bone diseases. J. Bone Miner. Metab. 2015, 33, 474–485. [Google Scholar] [CrossRef]
- Alfaro, J.M.; Manrique, R.; Santamaría, A.; Álvarez, E.; Manes, C.; Jiménez, M. Effects of endocrine disorders on maxillary and mandibular growth in Colombian children and adolescents: A cross-sectional study. Eur. Arch. Paediatr. Dent. 2024, 25, 17–25. [Google Scholar] [CrossRef]
- Gordon, L.A.; Miller, S.F.; Caplin, J.; Galang-Boquiren, M.T.; Alrayyes, S.; Nicholas, C.L. Childhood obesity may accelerate timing of human facial growth. Arch. Oral Biol. 2021, 121, 104964. [Google Scholar] [CrossRef] [PubMed]
- Vora, S.R.; Tam, S.; Katsube, M.; Pliska, B.; Heda, K. Craniofacial form differences between obese and nonobese children. Am. J. Orthod. Dentofac. Orthop. 2022, 162, 744–752.e3. [Google Scholar] [CrossRef] [PubMed]
- Boulan, L.; Milán, M.; Léopold, P. The Systemic Control of Growth. Cold Spring Harb. Perspect. Biol. 2015, 7, a019117. [Google Scholar] [CrossRef]
- Arnott, S.A.; Chiba, S.; Conover, D.O. Evolution of intrinsic growth rate: Metabolic costs drive trade-offs between growth and swimming performance in Menidia menidia. Evolution 2006, 60, 1269–1278. [Google Scholar] [CrossRef] [PubMed]
- Xia, L.; Jiang, W.; Yao, K.; Sun, H.; Lu, X.; Yu, W. Craniofacial, Dental, and Upper Airway Morphologic features of Severely Obese Adults with Obstructive Sleep Apnea. Int. Dent. J. 2025, 75, 1736–1744. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Camastra, S.; Vitali, A.; Anselmino, M.; Gastaldelli, A.; Bellini, R.; Berta, R.; Severi, I.; Baldi, S.; Astiarraga, B.; Barbatelli, G.; et al. Muscle and adipose tissue morphology, insulin sensitivity and beta-cell function in diabetic and nondiabetic obese patients: Effects of bariatric surgery. Sci. Rep. 2017, 7, 9007. [Google Scholar] [CrossRef]
- Saloom, H.F.; Boustan, R.; Seehra, J.; Papageorgiou, S.N.; Carpenter, G.H.; Cobourne, M.T. The impact of obesity on orthodontic treatment outcome in adolescents: A prospective clinical cohort study. Eur. J. Orthod. 2021, 43, 165–172. [Google Scholar] [CrossRef]
- Voltan, C.; Concer, F.; Pecoraro, L.; Pietrobelli, A.; Piacentini, G.; Zaffanello, M. Exploring the Complex Interplay of Obesity, Allergic Diseases, and Sleep-Disordered Breathing in Children. Children 2024, 11, 595. [Google Scholar] [CrossRef]
- Huynh, N.; Zhang, J.; Pliska, B.; Amin, R.; Narang, I.; Chadha, N.; Cholette, M.C.; Kirk, V.; Montpetit, A.; Vezina, K.; et al. Prevalence of Altered Craniofacial Morphology in Children With OSA. J. Sleep Res. 2025, 34, e70060. [Google Scholar] [CrossRef]
- Nahhas, R.W.; Valiathan, M.; Sherwood, R.J. Variation in timing, duration, intensity, and direction of adolescent growth in the mandible, maxilla, and cranial base: The Fels longitudinal study. Anat. Rec. 2014, 297, 1195–1207. [Google Scholar] [CrossRef]
- Kim, N.-H.; Lee, J.-H.; Lee, S.; Choi, Y.J.; Chung, C.J.; Lee, K.-J.; Kim, K.-H. Growth Velocity and the Pubertal Growth Spurt Across Skeletal Maturity Stages in Class II Malocclusion: A Longitudinal Analysis. Children 2025, 12, 1612. [Google Scholar] [CrossRef] [PubMed]
- Gu, T.; Zhang, S.; Xiao, C.; Hu, S.; Xiong, X. Sex differences in craniofacial parameters of children and adolescents: A comparative study with the maturation of cervical vertebrae using a cephalometric method. J. Clin. Pediatr. Dent. 2024, 48, 89–100. [Google Scholar] [CrossRef] [PubMed]
- Dudnik, O.V.; Mamedov, A.A.; Dybov, A.M.; Kharke, V.V.; Timoshenko, T.V.; Skakodub, A.A.; Maclennan, A.B.; Bille, D.S. Application of additional anthropometric and functional methods in children undergoing orthodontic treatment using braces. Saudi Dent. J. 2021, 33, 222–228. [Google Scholar] [CrossRef] [PubMed]
| Variable | Category | n |
|---|---|---|
| BMI | Normal Weight | 95 |
| Overweight | 18 | |
| Obese | 17 | |
| Sex | Male | 53 |
| Female | 77 | |
| Age group | 4–10.9 yrs | 26 |
| 11–12.9 yrs | 39 | |
| 13–14.9 yrs | 42 | |
| 15–19.9 yrs | 23 |
| Parameter | W value | p Value | Distribution |
|---|---|---|---|
| SNA | 0.972 | 0.118 | Normal |
| SNB | 0.968 | 0.094 | Normal |
| ANB | 0.955 | 0.061 | Normal |
| Maxillary unit length | 0.979 | 0.164 | Normal |
| Mandibular unit length | 0.966 | 0.083 | Normal |
| Mandibular body length | 0.971 | 0.107 | Normal |
| Facial convexity angle | 0.963 | 0.072 | Normal |
| Facial height ratio | 0.976 | 0.149 | Normal |
| Parameter | Normal Weight | Overweight | Obese | p Value |
|---|---|---|---|---|
| SNA (°) | 82.4 ± 3.0 | 85.0 ± 3.3 | 84.1 ± 3.1 | 0.010 |
| SNB (°) | 79.2 ± 2.9 | 81.5 ± 3.0 | 82.4 ± 3.2 | 0.003 |
| ANB (°) | 3.2 ± 1.8 | 3.5 ± 1.7 | 1.7 ± 1.6 | 0.012 |
| Maxillary unit length (mm) | 97.0 ± 4.1 | 98.6 ± 4.4 | 100.1 ± 4.3 | 0.017 |
| Mandibular unit length (mm) | 123.0 ± 6.6 | 126.4 ± 7.0 | 129.6 ± 6.7 | 0.002 |
| Mandibular body length (mm) | 74.0 ± 5.1 | 76.5 ± 5.0 | 78.2 ± 5.3 | 0.013 |
| Facial convexity angle (°) | 15.3 ± 4.1 | 12.8 ± 3.8 | 10.1 ± 3.4 | 0.005 |
| Facial height ratio (%) | 95.4 ± 3.6 | 91.1 ± 4.0 | 87.2 ± 4.3 | 0.007 |
| Parameter | 4–10.9 yrs | 11–12.9 yrs | 13–14.9 yrs | 15–19.9 yrs | Age Effect (p) | BMI Effect (p) |
|---|---|---|---|---|---|---|
| SNA (°) | 81.2 ± 2.9 | 82.5 ± 3.1 | 83.2 ± 3.3 | 83.6 ± 3.5 | 0.039 | 0.010 |
| SNB (°) | 77.8 ± 3.1 | 79.6 ± 3.2 | 80.9 ± 3.4 | 81.3 ± 3.6 | <0.001 | <0.001 |
| ANB (°) | 3.4 ± 1.9 | 2.9 ± 1.8 | 2.3 ± 1.7 | 2.1 ± 1.6 | 0.021 | 0.012 |
| Maxillary unit length (mm) | 93.5 ± 4.6 | 96.1 ± 4.4 | 98.0 ± 4.8 | 99.0 ± 5.1 | <0.001 | 0.017 |
| Mandibular unit length (mm) | 118.3 ± 6.2 | 123.5 ± 6.6 | 127.3 ± 7.1 | 129.0 ± 7.4 | <0.001 | <0.001 |
| Mandibular body length (mm) | 70.0 ± 4.2 | 73.5 ± 4.6 | 76.1 ± 5.0 | 76.9 ± 5.2 | <0.001 | 0.013 |
| Facial convexity angle (°) | 16.4 ± 4.6 | 14.2 ± 4.4 | 11.9 ± 4.2 | 11.0 ± 4.3 | <0.001 | 0.005 |
| Facial height ratio (%) | 96.6 ± 5.2 | 92.1 ± 4.9 | 89.3 ± 4.7 | 87.7 ± 4.5 | <0.001 | 0.007 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





