Submitted:
02 March 2026
Posted:
03 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2 Methods
2.2.1 Coaxial Jet Mixer
2.2.2 Particle Size Analysis
2.2.3 Curcumin Quantification
2.3 Experimental Workflow
2.3.1 Fluid-Dynamic Characterization
2.3.2 Nanoparticle Production
2.3.3 Liposome PRODUCTION
3. Modeling
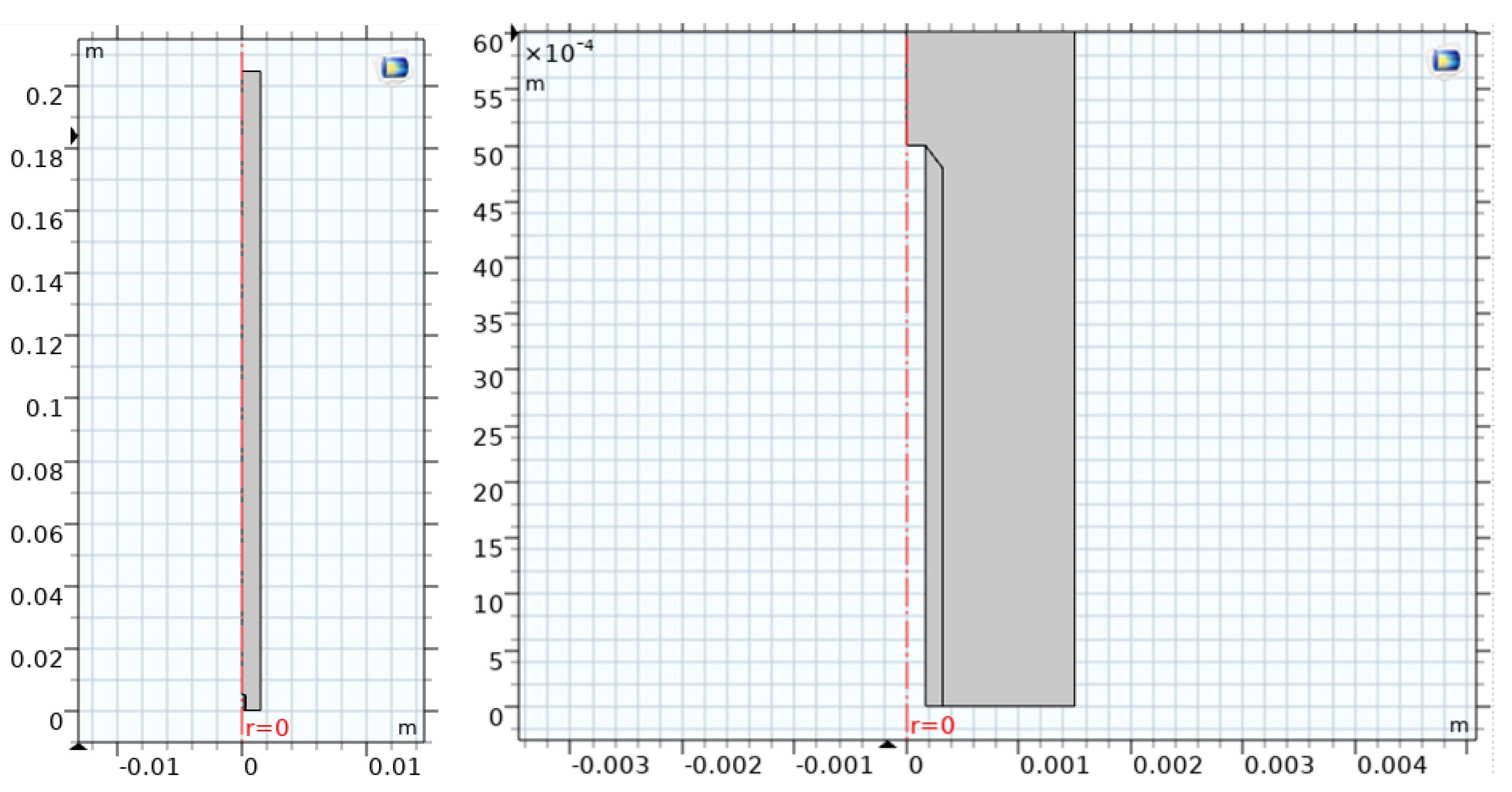
3.1 Geometry and Operating Conditions
3.2 Hydrodynamics and Turbulence Modeling
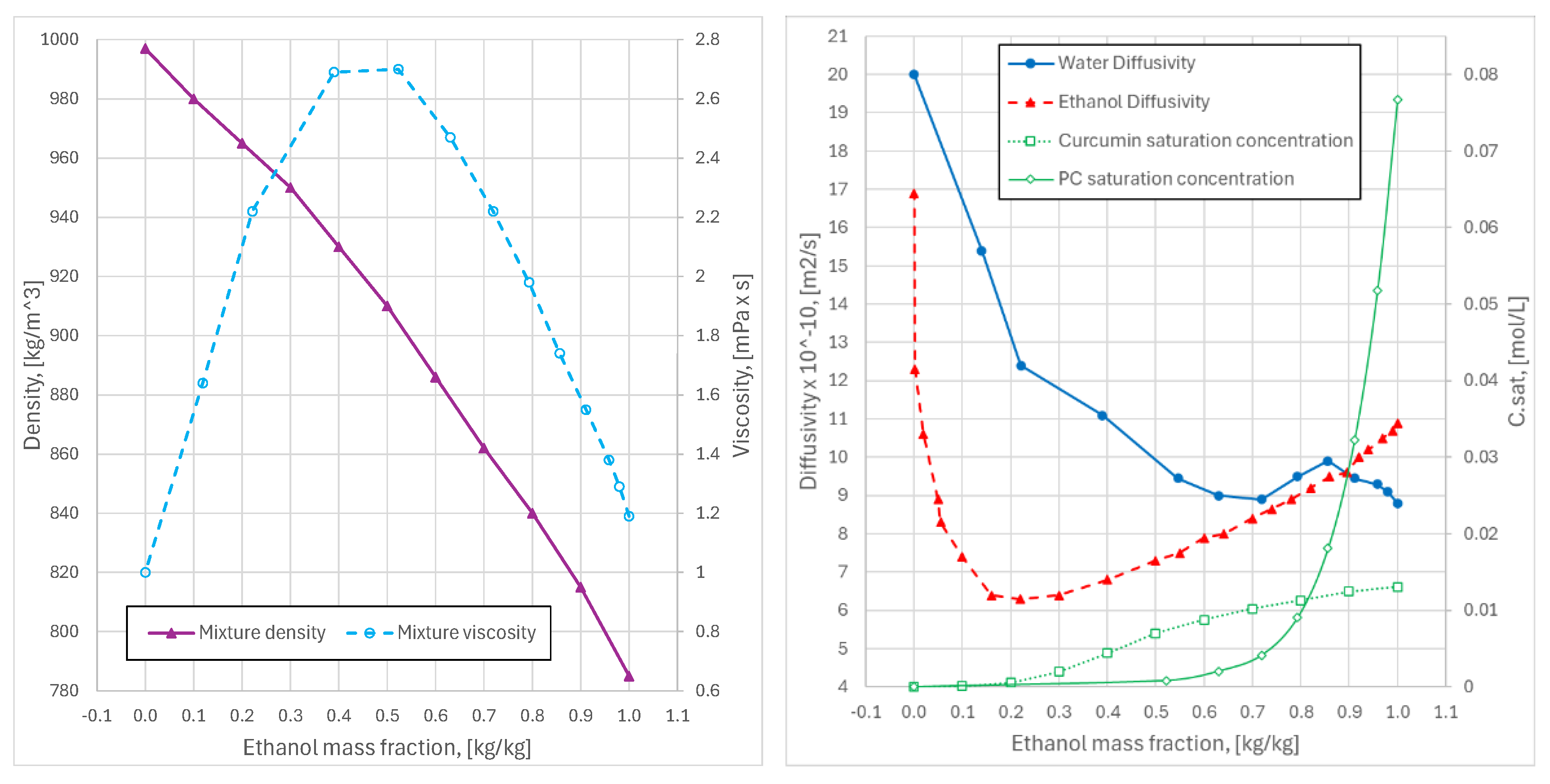
3.3 Ethanol–Water Mixing and Property Correlations
3.4 Population Balance Model and Dissolved Solute Transport Equation
| (16) |
3.6 Numerical Solution and Coupling Strategy
4. Results and Discussion
4.1. Fluid-Dynamics Characterization
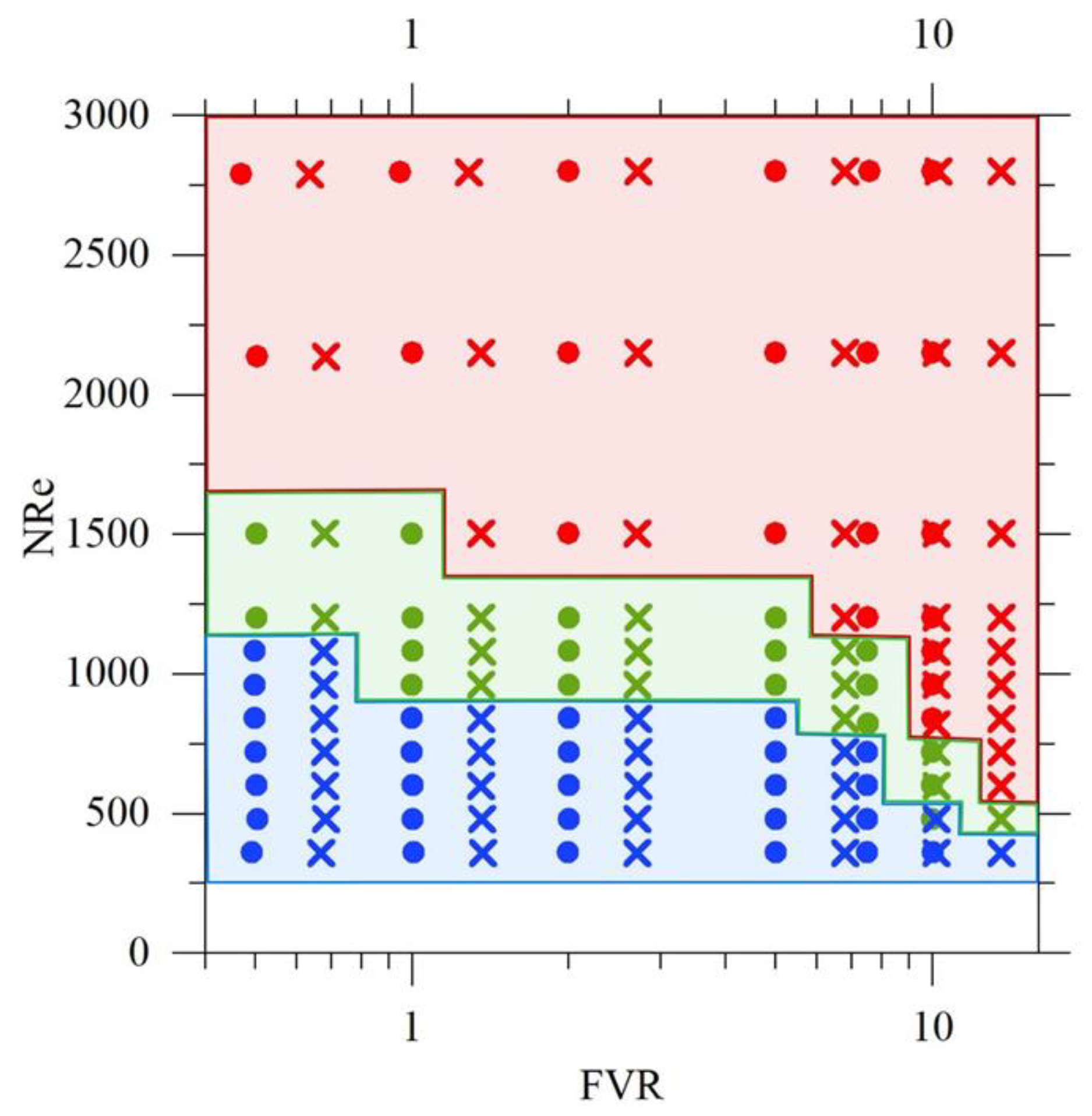
4.1.1. Flow Visualization and Regime Identification
- Laminar focusing – the inner jet retains a coherent core, with limited entrainment and incomplete neutralization along the axial direction.
- Transitional flow – intermittent perturbations appear, producing local deformation of the jet and irregular interfaces.
- Jet-like/turbulent conditions – the colored core rapidly disappears after the needle outlet, indicating intense deformation and fast dilution of the injected stream.
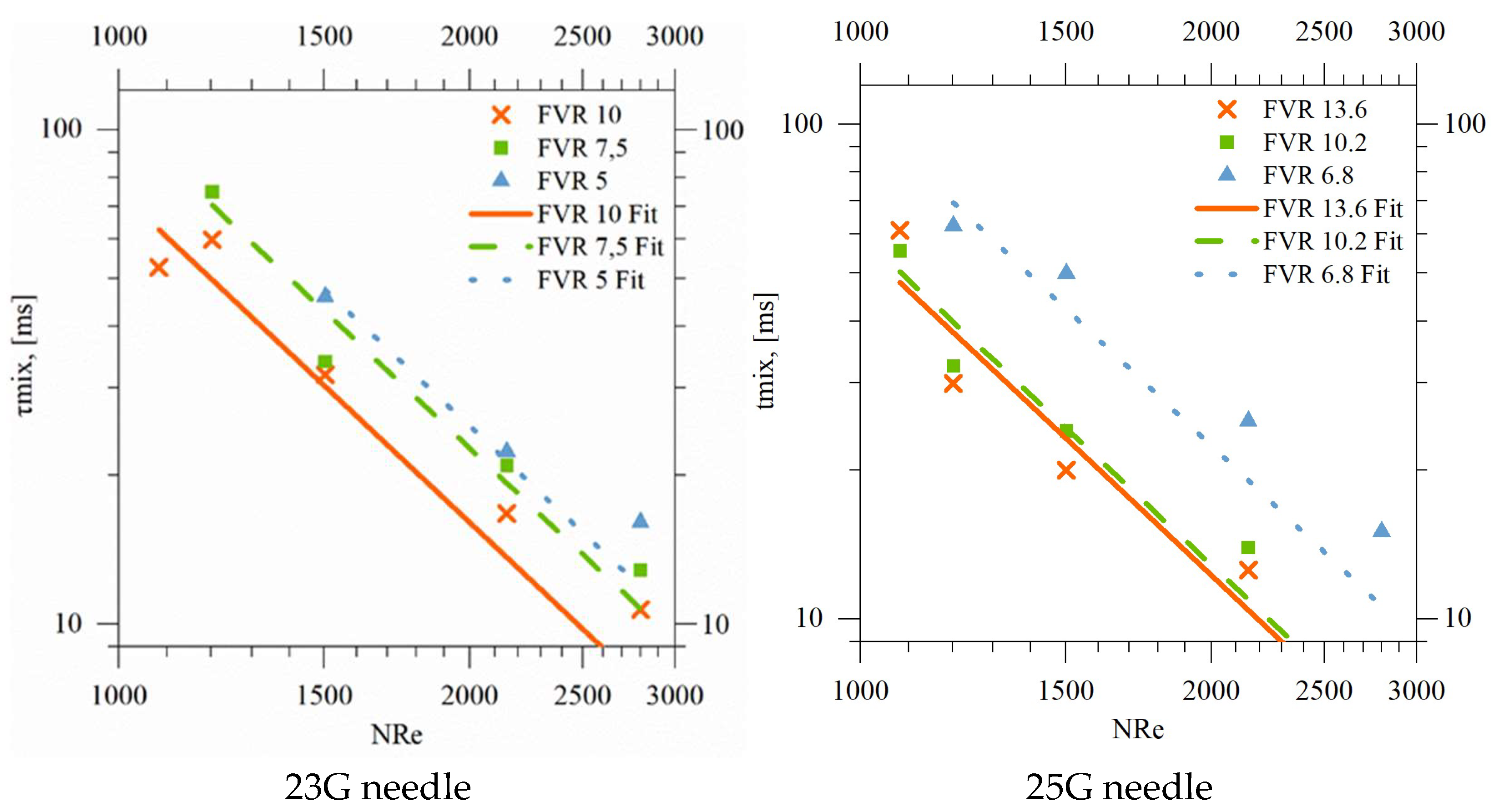
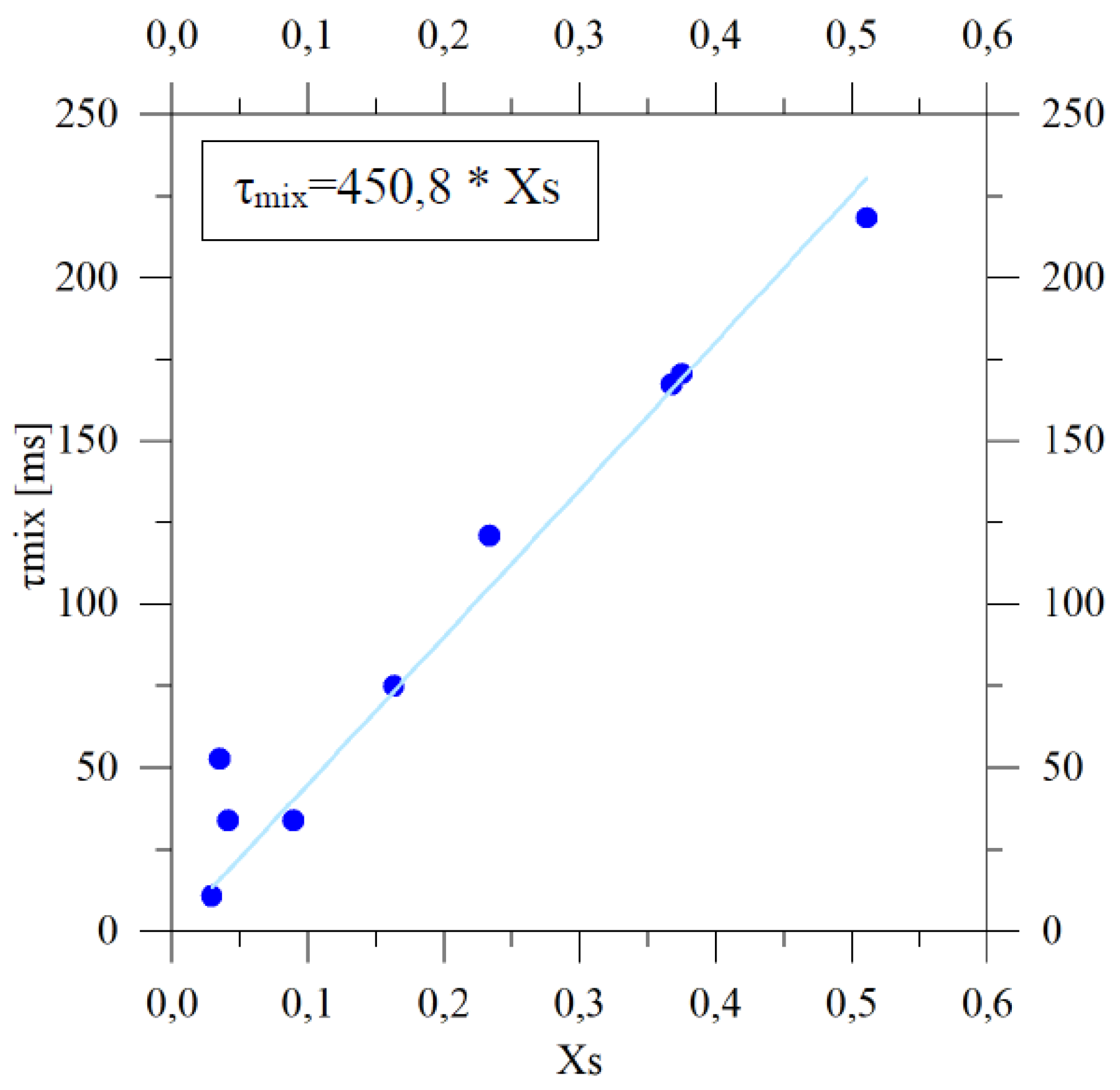
4.1.2. Characteristic Mixing Lengths and Times
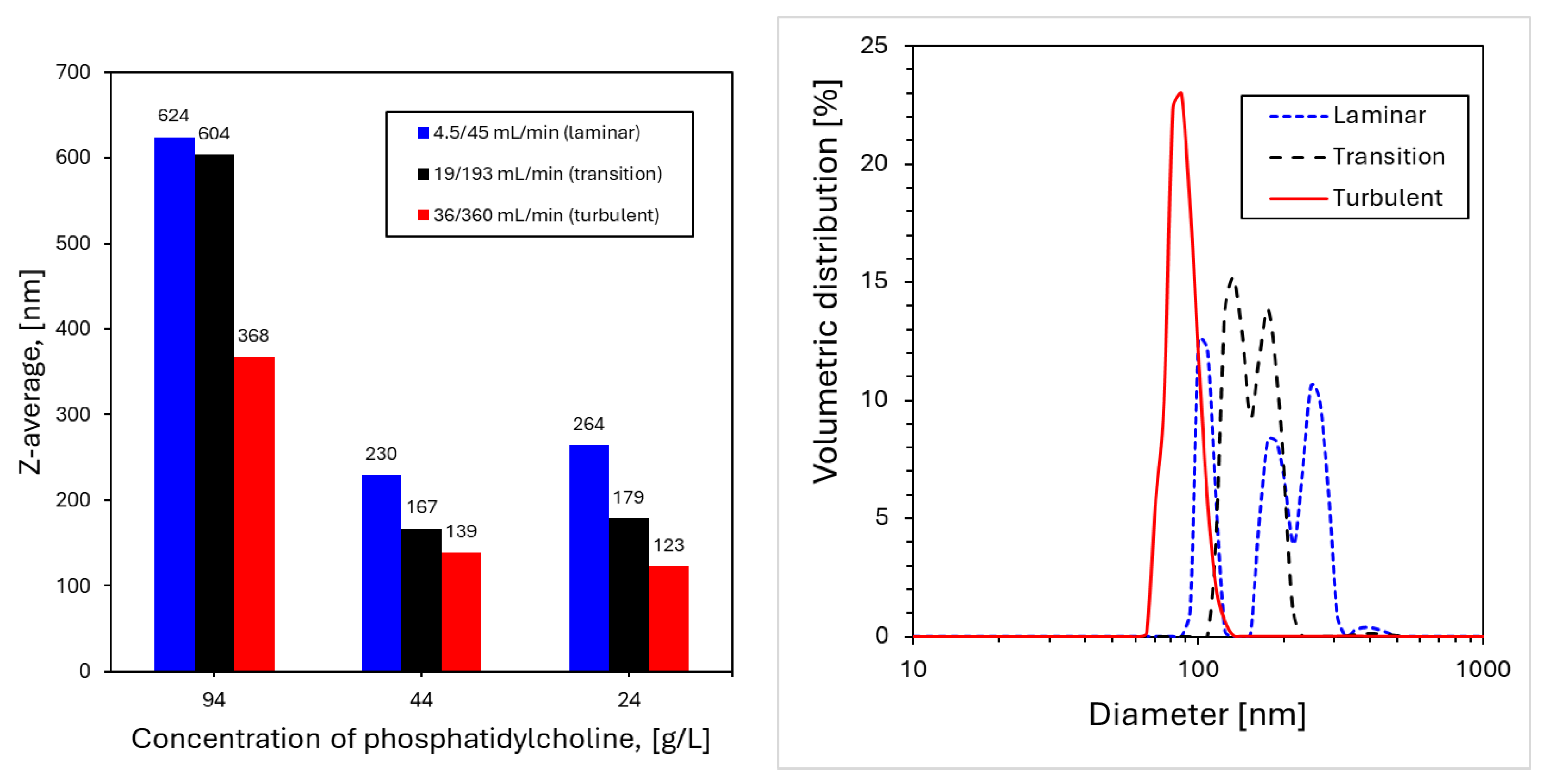
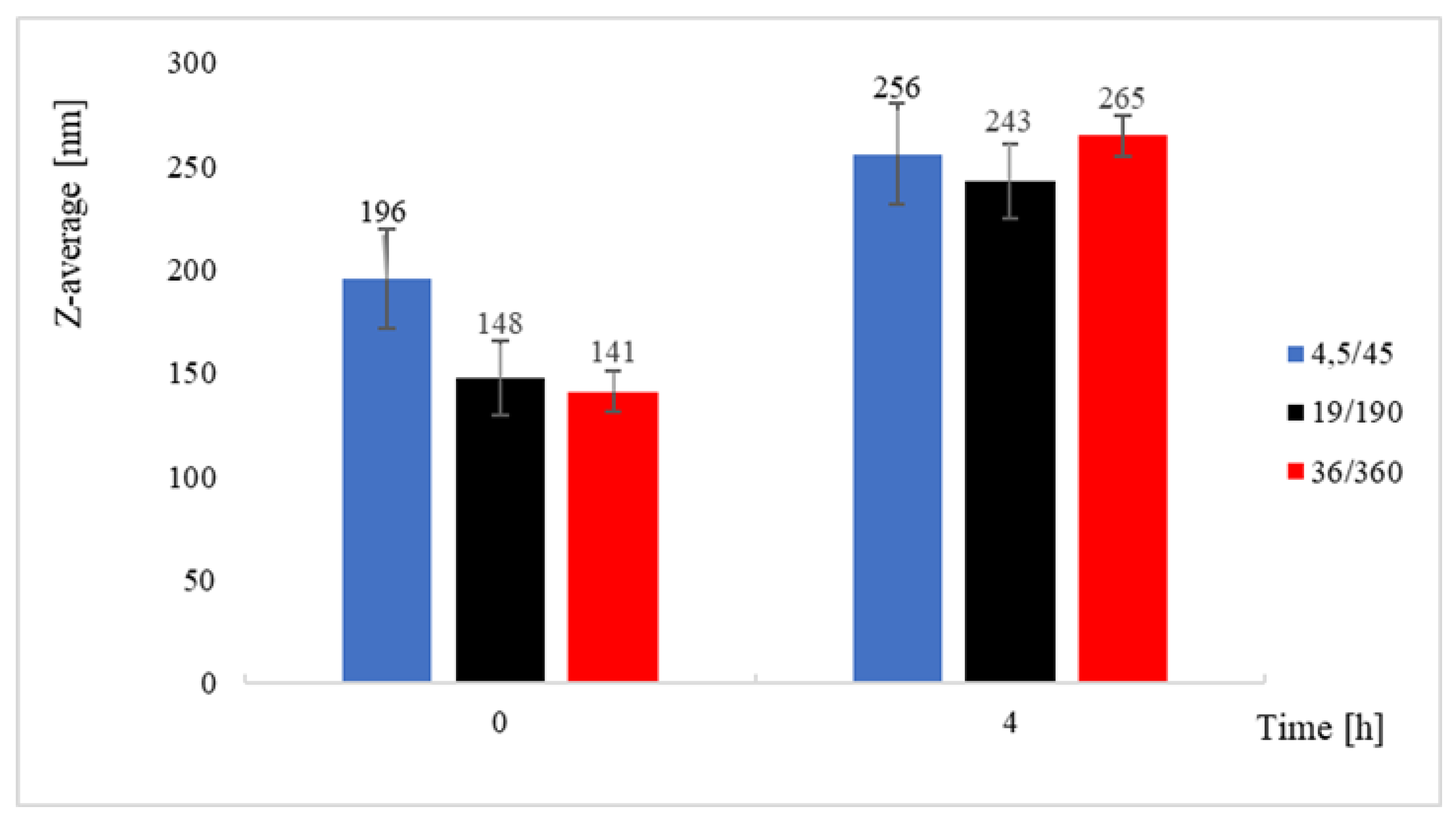
4.2. Nanoparticles Production and Characterization
4.2.1 Nanoliposomes Formation
4.2.2. Curcumin Nanoprecipitation
- shorter operational mixing times correlate with smaller primary structures;
- concentration governs the extent of aggregation during and after formation;
- rapid formation does not automatically guarantee long-term stability.
4.3. Modeling Results
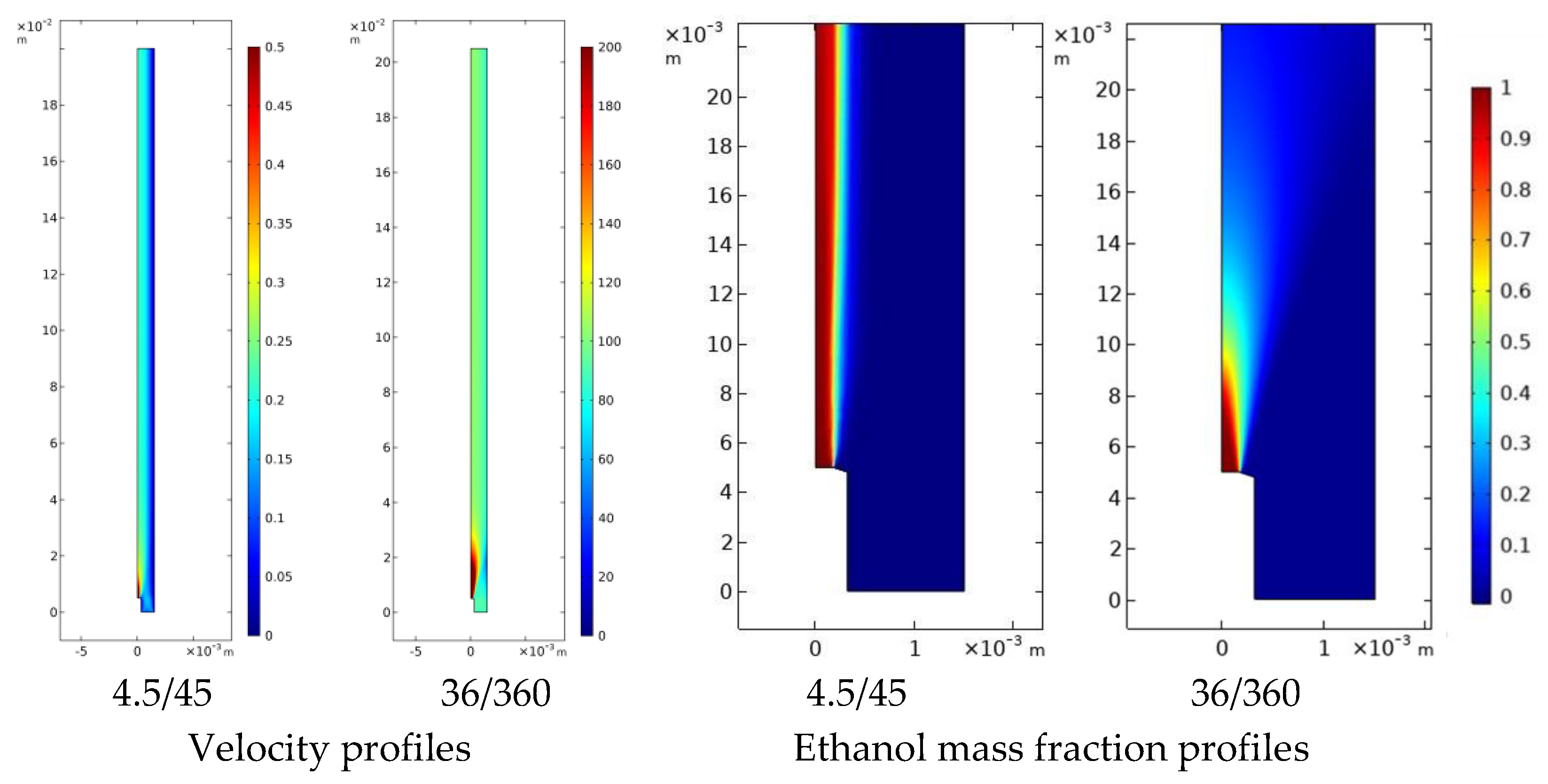
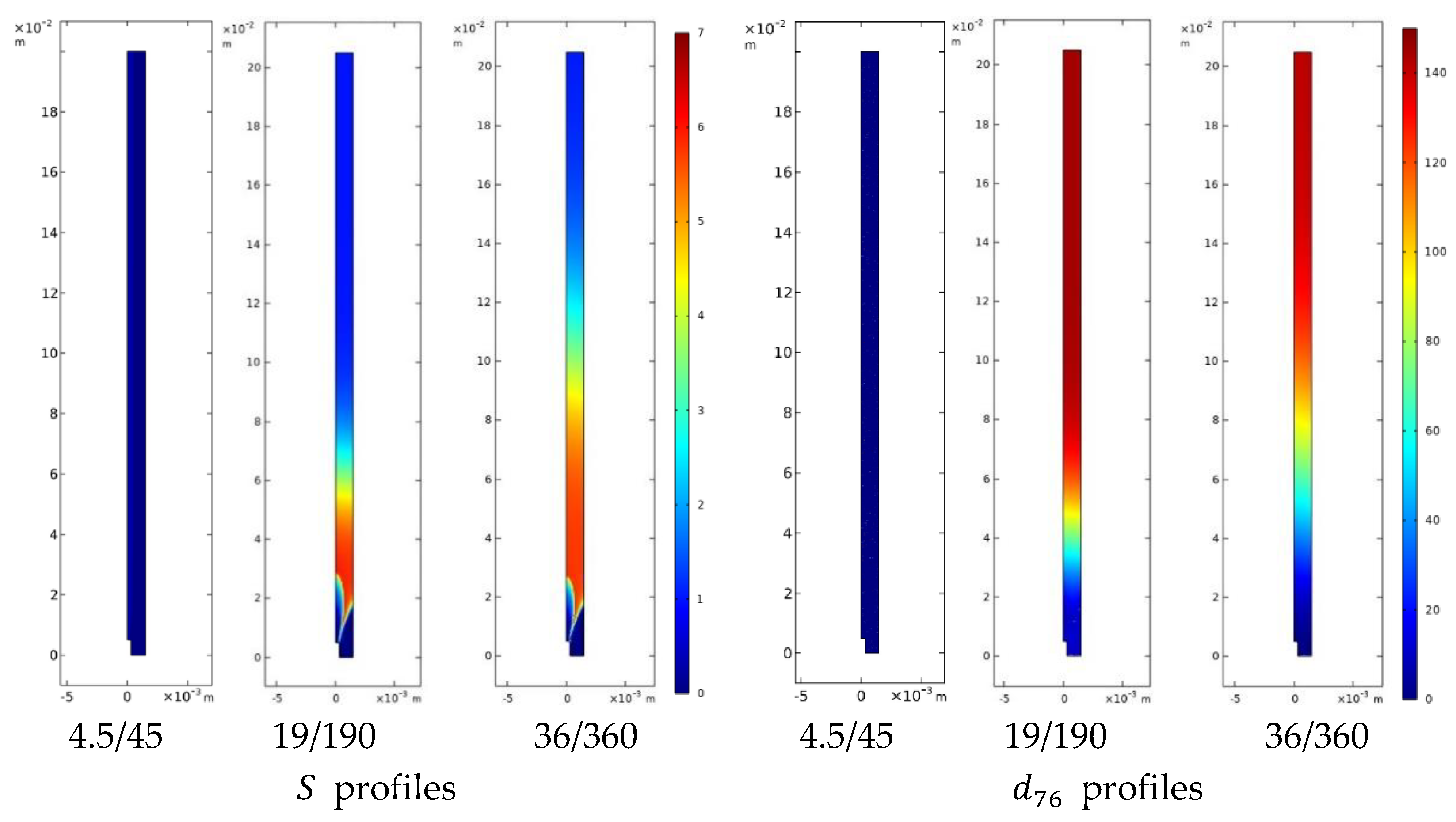
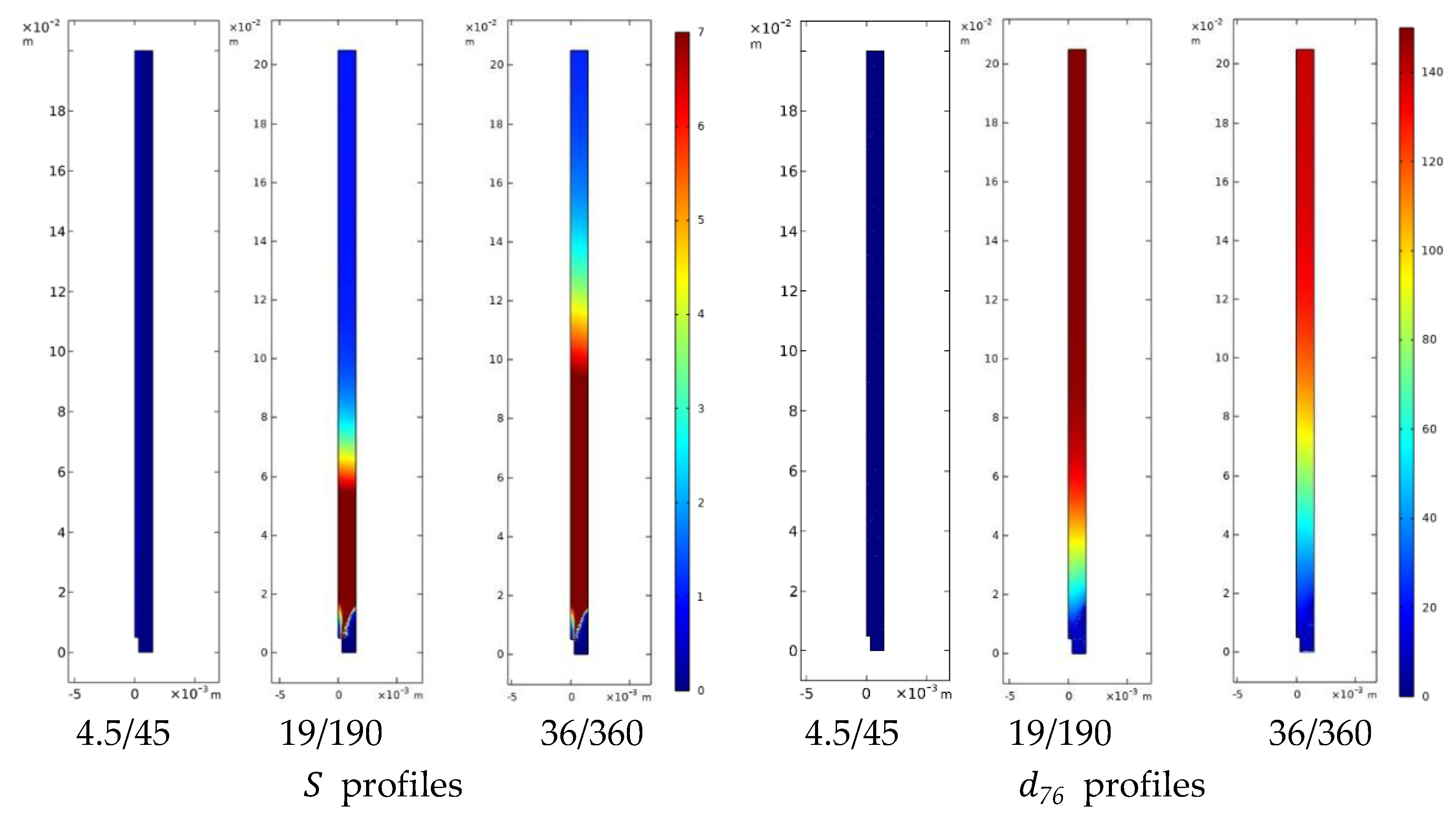
4.3.1 Hydrodynamics and Solvent Displacement Fields
4.3.2 Curcumin Nanoparticle and Liposome Formation
4.3.3 Integrated Interpretation
- shorter operational mixing times → faster solvent displacement → earlier, more homogeneous nucleation
- persistent gradients → extended growth windows → larger and broader particle populations
- hydrodynamics modulates the distribution of supersaturation, rather than imposing a single controlling mechanism
4.4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Poka, M.S.; Milne, M.; Wessels, A.; Aucamp, M. Sugars and Polyols of Natural Origin as Carriers for Solubility and Dissolution Enhancement. Pharmaceutics 2023, 15, 2557. [Google Scholar] [CrossRef]
- Preeti. Exploring LIPIDs for their potential to improves bioavailability of lipophilic drugs candidates: A review. Saudi Pharmaceutical Journal 2023, 31, 101870. [Google Scholar] [CrossRef]
- Baghel, P.; Roy, A.; Verma, S.; Satapathy, T.; Bahadur, S. Amelioration of lipophilic compounds in regards to bioavailability as self-emulsifying drug delivery system (SEDDS). Future Journal of Pharmaceutical Sciences 2020, 6, 21. [Google Scholar] [CrossRef]
- Chen, X.-Q. Oral Delivery of Highly Lipophilic, Poorly Water-Soluble Drugs: Self-Emulsifying Drug Delivery Systems to Improve Oral Absorption and Enable High-Dose Toxicology Studies of a Cholesteryl Ester Transfer Protein Inhibitor in Preclinical Species. Journal of Pharmaceutical Sciences 2018, 107, 1352–1360. [Google Scholar] [CrossRef] [PubMed]
- Gangavarapu, A.; Tapia-Lopez, L.V.; Sarkar, B.; Pena-Zacarias, J.; Badruddoza, A.Z.M.; Nurunnabi, M. Lipid nanoparticles for enhancing oral bioavailability. Nanoscale 2024, 16, 18319–18338. [Google Scholar] [CrossRef]
- Shabatina, T.I.G.Y.A.; Vernaya, O.I.; Soloviev, A.V.; Shabatin, A.V.; Morosov, Y.N.; Astashova, I.V.; Melnikov, M.Y. Pharmaceutical Nanoparticles Formation and Their Physico-Chemical and Biomedical Properties. Pharmaceuticals 2024, 17, 587. [Google Scholar] [CrossRef] [PubMed]
- Ghaly, H.S.A.; Seyedasli, N.; Varamini, P. Enhanced Nanoprecipitation Method for the Production of PLGA Nanoparticles for Oncology Applications. The AAPS Journal 2025, 27, 113. [Google Scholar] [CrossRef]
- Seo, Y.L.H.; Park, H.; Yu, J.; An, J.; Yoo, H.Y.; Lee, T. Recent Progress of Lipid Nanoparticles-Based Lipophilic Drug Delivery: Focus on Surface Modifications. Pharmaceutics 2023, 15, 772. [Google Scholar] [CrossRef]
- Giulia, A.; Gustavo, L.; Yang, S.; Yvonne, P. Scalable Manufacturing Processes for Solid Lipid Nanoparticles. Pharmaceutical Nanotechnology 2019, 7, 444–459. [Google Scholar] [CrossRef]
- Webb, C. Using microfluidics for scalable manufacturing of nanomedicines from bench to GMP: A case study using protein-loaded liposomes. International Journal of Pharmaceutics 2020, 582, 119266. [Google Scholar] [CrossRef]
- Elnady, R.E.; Amin, M.M.; Zakaria, M.Y. A review on lipid-based nanocarriers mimicking chylomicron and their potential in drug delivery and targeting infectious and cancerous diseases. AAPS Open 2023, 9, 13. [Google Scholar] [CrossRef]
- Bochicchio, S.; Lamberti, G.; Barba, A.A. Polymer–Lipid Pharmaceutical Nanocarriers: Innovations by New Formulations and Production Technologies. Pharmaceutics 2021, 13, 198. [Google Scholar] [CrossRef]
- De Piano, R.; Caccavo, D.; Lamberti, G.; Remaut, K.; Seynaeve, H.; Barba, A.A. A New Productive Approach and Formulative Optimization for Curcumin Nanoliposomal Delivery Systems. Pharmaceutics 2023, 15, 959. [Google Scholar] [CrossRef]
- Wang, Y.; Pi, C.; Feng, X.; Hou, Y.; Zhao, L.; Wei, Y. The Influence of Nanoparticle Properties on Oral Bioavailability of Drugs. International Journal of Nanomedicine 2020, 15, 6295–6310. [Google Scholar] [CrossRef]
- Mehrdadi, S. Lipid-Based Nanoparticles as Oral Drug Delivery Systems: Overcoming Poor Gastrointestinal Absorption and Enhancing Bioavailability of Peptide and Protein Therapeutics. Adv Pharm Bull 2024, 14, 48–66. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Gao, J.; Fan, R.; Zhang, T.; Tian, Y.; Wang, Z.; Zhang, H.; Zheng, A. The Effect of Particle Size on the Absorption of Cyclosporin A Nanosuspensions. International Journal of Nanomedicine 2022, 17, 1741–1755. [Google Scholar] [CrossRef]
- Desai, J.; Thakkar, H. Effect of particle size on oral bioavailability of darunavir-loaded solid lipid nanoparticles. Journal of Microencapsulation 2016, 33, 669–678. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.T. Absorption Study of Genistein Using Solid Lipid Microparticles and Nanoparticles: Control of Oral Bioavailability by Particle Sizes. Biomolecules & Therapeutics 2017, 25, 452–459. [Google Scholar] [CrossRef] [PubMed]
- Hiew, T.N.; Solomos, M.A.; Kafle, P.; Polyzois, H.; Zemlyanov, D.Y.; Punia, A.; Smith, D.; Schenck, L.; Taylor, L.S. The importance of surface composition and wettability on the dissolution performance of high drug loading amorphous dispersion formulations. Journal of Pharmaceutical Sciences 2025, 114, 289–303. [Google Scholar] [CrossRef]
- Dong, Y.; Ng, W.K.; Shen, S.; Kim, S.; Tan, R.B.H. Solid lipid nanoparticles: Continuous and potential large-scale nanoprecipitation production in static mixers. Colloids and Surfaces B: Biointerfaces 2012, 94, 68–72. [Google Scholar] [CrossRef]
- Rathod, P.; Narkhede, M.; Dongare, S. A Recent Review on Nanocrystal Manufacturing Techniques with Pharmaceutical Application. Current Nanomedicine 2024, 14, 4–12. [Google Scholar] [CrossRef]
- Tseng, Y.-C.; Kanthamneni, N.; Valiveti, S.; Patel, M.; Xia, H. Enhanced bioavailability of danazol nanosuspensions by wet milling and high-pressure homogenization. International Journal of Pharmaceutical Investigation 2016, 6, 218. [Google Scholar] [CrossRef]
- Hládek, F.; Chvíla, S.; Navrátil, O.; Balouch, M.; Štěpánek, F. Systematic Investigation of Wet-Milling Kinetics and Colloidal Stability of Pharmaceutical Nanocrystals. Crystal Growth & Design 2022, 22, 6928–6940. [Google Scholar] [CrossRef]
- Prosapio, V.; Reverchon, E.; De Marco, I. Incorporation of liposoluble vitamins within PVP microparticles using supercritical antisolvent precipitation. Journal of CO2 Utilization 2017, 19, 230–237. [Google Scholar] [CrossRef]
- Sierra-Pallares, J.; Marchisio, D.L.; Parra-Santos, M.T.; García-Serna, J.; Castro, F.; Cocero, M.J. A computational fluid dynamics study of supercritical antisolvent precipitation: Mixing effects on particle size. AIChE Journal 2012, 58, 385–398. [Google Scholar] [CrossRef]
- Liu, T.; Yu, X.; Yin, H. Study of Top-down and Bottom-up Approaches by Using Design of Experiment (DoE) to Produce Meloxicam Nanocrystal Capsules. AAPS PharmSciTech 2020, 21, 79. [Google Scholar] [CrossRef]
- Ahn, G.-Y.; Choi, I.; Ryu, T.-K.; Ryu, Y.-H.; Oh, D.-H.; Kang, H.-W.; Kang, M.-H.; Choi, S.-W. Continuous production of lipid nanoparticles by multiple-splitting in microfluidic devices with chaotic microfibrous channels. Colloids and Surfaces B: Biointerfaces 2023, 224, 113212. [Google Scholar] [CrossRef]
- Barba, A.A.; Lamberti, G.; D'Amore, M.; Bochicchio, S.; Dalmoro, A. ENG4LIFE S R L: EP 19710504 A; CONTINUOUS PROCESS FOR COATING LIPOSOMIAL VECTORS WITH POLYMER. 2021.
- Vogler, J.; BrÄUer, C.; ClÉMent, P.; Stieneker, F. PREPARATION OF DRUG-LOADED MICRO- AND NANOPARTICLES BY JET IMPINGEMENT. 2024. [Google Scholar]
- Ramstack, J.M.; Podobinski, J. Method for fabricating nanoparticles. CERULEAN PHARMA INC, 2014.
- Franco, P.; De Marco, I. Supercritical Antisolvent Process for Pharmaceutical Applications: A Review. Processes 2020, 8, 938. [Google Scholar] [CrossRef]
- Caccavo, D.; Lamberti, G.; Barba, A.A. Coaxial Injection Mixer for the Continuous Production of Nanoparticles. CHEMICAL ENGINEERING TRANSACTIONS 2023, 100, 301–306. [Google Scholar] [CrossRef]
- Lim, J.-M.; Swami, A.; Gilson, L.M.; Chopra, S.; Choi, S.; Wu, J.; Langer, R.; Karnik, R.; Farokhzad, O.C. Ultra-High Throughput Synthesis of Nanoparticles with Homogeneous Size Distribution Using a Coaxial Turbulent Jet Mixer. ACS Nano 2014, 8, 6056–6065. [Google Scholar] [CrossRef]
- Costa, A.P.; Xu, X.; Khan, M.A.; Burgess, D.J. Liposome Formation Using a Coaxial Turbulent Jet in Co-Flow. Pharmaceutical Research 2016, 33, 404–416. [Google Scholar] [CrossRef]
- Machmudah, S.; Winardi, S.; Wahyudiono; Kanda, H.; Goto, M. Formation of Fine Particles from Curcumin/PVP by the Supercritical Antisolvent Process with a Coaxial Nozzle. ACS Omega 2020, 5, 6705–6714. [Google Scholar] [CrossRef]
- Middha, E.; Duan, Y.; Liu, B.I.N. SYSTEMS AND METHODS FOR FABRICATING NANOPARTICLES; NAT UNIV SINGAPORE: SG 2020050350 W, 2020. [Google Scholar]
- Seaman, P.; Davies, C.; King, L. Microparticle Production Platform, Method of Producing Microparticles and a Pharmaceutical Composition; MIDATECH PHARMA WALES LTD; US 202117801496 A, 2023. [Google Scholar]
- Wang, Y.; Chu, D.-J.; Li, J.-L.; Xie, X.-Q.; Ren, G.-B.; Qi, M.-H. Efficient Production of Inhalable Micro-Nanoparticles: Mechanism, Process Optimization, and Modular Continuous Micro-Crystallizer Design. Organic Process Research & Development 2025, 29, 1264–1278. [Google Scholar] [CrossRef]
- VandenBerg, M.A.; Dong, X.; Smith, W.C.; Tian, G.; Stephens, O.; O’Connor, T.F.; Xu, X. Learning from the future: towards continuous manufacturing of nanomaterials. AAPS Open 2025, 11, 7. [Google Scholar] [CrossRef]
- Caccavo, D.; De Piano, R.; Broegg, L.; Barba, A.A.; Lamberti, G. Optimization of Nanoliposomes Production using a Coaxial Jet Mixer: a Response Surface Modeling Approach. Chemical Engineering Transactions 2025, 118, 295–300. [Google Scholar] [CrossRef]
- Iannone, M.; Caccavo, D.; Barba, A.A.; Lamberti, G. A low-cost push–pull syringe pump for continuous flow applications. HardwareX 2022, 11, e00295. [Google Scholar] [CrossRef]
- Della Vecchia, M. Analysis of the impact of fluid dynamics conditions on nanoparticles production process, in Department of Industrial Engineering; University of Salerno: Fisciano (SA), 2022. [Google Scholar]
- De Caro, C. Characterization of a coaxial injection mixer for continuous production of nanoparticles, in Department of Industrial Engineering; University of Salerno: Fisciano (SA), 2023. [Google Scholar]
- Commenge, J.-M.; Falk, L. Villermaux–Dushman protocol for experimental characterization of micromixers. Chemical Engineering and Processing: Process Intensification 2011, 50, 979–990. [Google Scholar] [CrossRef]
- Landi, F. Produzione e studio del comportamento di nanoparticelle mediante mixer a getto coassiale. In Department of Industrial Engineering; University of Salerno: Fisciano (SA), 2023. [Google Scholar]
- Cupolo, A. Curcumin nanoprecipitation in coaxial jet mixer in Department of Industrial Engineering; University of Salerno: Fisciano (SA), 2024. [Google Scholar]
- Bird, R.B.; Stewart, W.E.; Lightfoot, E.N. Transport Phenomena. In Transport Phenomena; Wiley, 2006. [Google Scholar]
- Pope, S.B. Turbulent Flows; Cambridge University Press, 2000. [Google Scholar]
- Wilcox, D.C. Turbulence Modeling for CFD. Turbulence Modeling for CFD; DCW Industries, 2006. [Google Scholar]
- Launder, B.E.; Spalding, D.B. The numerical computation of turbulent flows. Computer Methods in Applied Mechanics and Engineering 1974, 3, 269–289. [Google Scholar] [CrossRef]
- Khattab, I.S.; Bandarkar, F.; Fakhree, M.A.A.; Jouyban, A. Density, viscosity, and surface tension of water+ethanol mixtures from 293 to 323K. Korean Journal of Chemical Engineering 2012, 29, 812–817. [Google Scholar] [CrossRef]
- González, B.; Calvar, N.; Gómez, E.; Domínguez, Á. Density, dynamic viscosity, and derived properties of binary mixtures of methanol or ethanol with water, ethyl acetate, and methyl acetate at T=(293.15, 298.15, and 303.15)K. The Journal of Chemical Thermodynamics 2007, 39, 1578–1588. [Google Scholar] [CrossRef]
- Klinov, A.; Anashkin, I. Diffusion in Binary Aqueous Solutions of Alcohols by Molecular Simulation. Processes 2019, 7, 947. [Google Scholar] [CrossRef]
- Roy, S.; Bachchhav, S.D.; Mukhopadhyay, M. Analysis of the Mechanism of Cholesterol Particle Formation by Liquid Antisolvent Crystallization. Industrial & Engineering Chemistry Research 2021, 60, 7975–7986. [Google Scholar] [CrossRef]
- Ramkrishna, D. Population Balances: Theory and Applications to Particulate Systems in Engineering; Academic Press, 2000. [Google Scholar]
- Mersmann, A. Crystallization Technology Handbook; Taylor & Francis, 2001. [Google Scholar]
- Hulburt, H.M.; Katz, S. Some problems in particle technology: A statistical mechanical formulation. Chemical Engineering Science 1964, 19, 555–574. [Google Scholar] [CrossRef]
- Shin, S.; Devos, C.; Udepurkar, A.P.; Inguva, P.K.; Myerson, A.S.; Braatz, R.D. Mechanistic modeling of lipid nanoparticle (LNP) precipitation via population balance equations (PBEs). Chemical Engineering Journal 2025, 523, 167786. [Google Scholar] [CrossRef]
- Iggland, M.; Mazzotti, M. Population Balance Modeling with Size-Dependent Solubility: Ostwald Ripening. Crystal Growth & Design 2012, 12, 1489–1500. [Google Scholar] [CrossRef]
- Shiea, M.; Querio, A.; Buffo, A.; Boccardo, G.; Marchisio, D. CFD-PBE modelling of continuous Ni-Mn-Co hydroxide co-precipitation for Li-ion batteries. Chemical Engineering Research and Design 2022, 177, 461–472. [Google Scholar] [CrossRef]
- Saad, W.S.; Prud’homme, R.K. Principles of nanoparticle formation by flash nanoprecipitation. Nano Today 2016, 11, 212–227. [Google Scholar] [CrossRef]
- Carugo, D.; Bottaro, E.; Owen, J.; Stride, E.; Nastruzzi, C. Liposome production by microfluidics: potential and limiting factors. Scientific Reports 2016, 6, 25876. [Google Scholar] [CrossRef] [PubMed]
- Hood, R.R.; DeVoe, D.L. High-Throughput Continuous Flow Production of Nanoscale Liposomes by Microfluidic Vertical Flow Focusing. Small 2015, 11, 5790–5799. [Google Scholar] [CrossRef]
- Jahn, A.; Stavis, S.M.; Hong, J.S.; Vreeland, W.N.; DeVoe, D.L.; Gaitan, M. Microfluidic Mixing and the Formation of Nanoscale Lipid Vesicles. ACS Nano 2010, 4, 2077–2087. [Google Scholar] [CrossRef]
- Wagner, A.; Vorauer-Uhl, K. Liposome Technology for Industrial Purposes. Journal of Drug Delivery 2011, 2011, 591325. [Google Scholar] [CrossRef]
- Kakran, M.; Sahoo, N.G.; Tan, I.L.; Li, L. Preparation of nanoparticles of poorly water-soluble antioxidant curcumin by antisolvent precipitation methods. Journal of Nanoparticle Research 2012, 14, 757. [Google Scholar] [CrossRef]
- Johnson, B.K.; Prud’homme, R.K. Mechanism for Rapid Self-Assembly of Block Copolymer Nanoparticles. Physical Review Letters 2003, 91, 118302. [Google Scholar] [CrossRef] [PubMed]

| Reagent | Concentration [M] | |
|---|---|---|
| Inner tube | 0.1 | |
| Outer tube | 0.035 | |
| 0.007 | ||
| 0.5 | ||
| 0.25 |
| Parameter | Symbol | Value | Unit |
|---|---|---|---|
| Outer tube length | 20.5 | cm | |
| Outer tube internal diameter | 3.0 | mm | |
| Needle internal diameter (23G) | 337 | μm | |
| Needle external diameter (23G) | 641.4 | μm |
| FVR | Regime | [ms] | ||
|---|---|---|---|---|
| 5 | 1503 | Turbulent | 45.91 | 0.09 |
| 7.5 | 361 | Laminar | >170.53 | 0.38 |
| 7.5 | 820 | Laminar | >167.17 | 0.37 |
| 7.5 | 1082 | Transitional | 121.00 | 0.23 |
| 7.5 | 1202 | Turbulent | 75.00 | 0.16 |
| 7.5 | 1503 | Turbulent | 33.93 | 0.04 |
| 7.5 | 2801 | Turbulent | 12.85 | 0.03 |
| 10 | 481 | Transitional | 218.33 | 0.51 |
| 10 | 842 | Turbulent | 96.24 | 0.05 |
| 10 | 962 | Turbulent | 62.38 | 0.05 |
| 10 | 1082 | Turbulent | 52.67 | 0.04 |
| Inner flow rate [ml/min] | Outer flow rate [ml/min] | FVR | [ms] | |
|---|---|---|---|---|
| 4,5 | 45 | 240 | 7.5 | >1200 |
| 19 | 190 | 1032 | 7.5 | 34 |
| 36 | 360 | 1920 | 7.5 | 13 |
| System | Flow rate [mL/min] |
[m-3s-1] |
[nm L g-1 s-1] |
[nm] |
Z-average [nm] |
|---|---|---|---|---|---|
| Curcumin NPs | 36/360† | 141 | 141 | ||
| Curcumin NPs | 19/190 | - | - | 144 | 148 |
| Liposomes | 36/360† | 139 | 139 | ||
| Liposomes | 19/190 | - | - | 149 | 167 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




