Submitted:
01 March 2026
Posted:
03 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Green Nanotechnology Framework
3. Biogenic Sources of Nanoparticles
| Biogenic Source | Organism Type | NP Type | Typical Size (nm) | Key Phytochemicals / Active Compounds | Main Advantages | Representative Agricultural Application |
| Azadirachta indica (Neem) | Terrestrial plant | AgNPs | 10–35 | Terpenoids (azadirachtin), flavonoids, polyphenols | Synergistic antimicrobial + pesticidal coating; scalable; no sterile conditions required | Antifungal against Rhizoctonia solani and Sclerotinia sclerotiorum; nematicidal activity |
| Camellia sinensis (Green tea) | Terrestrial plant | AuNPs / AgNPs | 15–50 | Catechins (EGCG), polyphenols, ascorbic acid | Highly stable colloids; biocompatible coating; rapid synthesis | Antimicrobial applications; seed priming to improve germination |
| Curcuma longa (Turmeric) | Terrestrial plant | AuNPs / AgNPs | 20–60 | Curcumin, turmerone, phenolics | Anti-inflammatory capping; dual antifungal and antioxidant properties | Antimicrobial and antifungal applications in crop protection |
| Eucalyptus spp. | Terrestrial plant | ZnO-NPs | 20–50 | Terpenoids (cineole), tannins, flavonoids | UV-induced antimicrobial activity; growth-promoting Zn micronutrient | Disease suppression (powdery mildew); growth enhancement; abiotic stress mitigation |
| Aloe vera | Terrestrial plant | AgNPs / AuNPs / ZnO-NPs | 8–40 | Aloin, anthraquinones, polysaccharides | Broad-spectrum reducing capacity; gel matrix aids stabilisation | Antifungal and antibacterial activity; seed priming |
| Glycyrrhiza glabra (Licorice) | Terrestrial plant | AgNPs | 12–35 | Glycyrrhizin, flavonoids, coumarins | Strong nematicidal capping bioactivity; plant-health promoting compounds | Nematicidal activity against Meloidogyne incognita; induction of plant antioxidant defences |
| Tridax procumbens | Terrestrial plant | Cu / Fe / Zn NPs | 15–45 | Flavonoids, phenolics, alkaloids | Multi-metal synthesis capability; low-cost weed-derived feedstock | Nematicidal activity in cabbage; broad-spectrum soil pest management |
| Salix alba (Willow bark) | Terrestrial plant | ZnO-NPs | 20–55 | Salicin, tannins, phenolic glycosides | Salicylate capping may induce plant systemic resistance; widely available bark waste | Nematicidal efficacy; potential systemic resistance induction in crops |
| Sargassum / Chlorella spp. | Macroalgae / Microalgae | AgNPs / ZnO-NPs | 10–40 | Polysaccharides, polyphenols, phycobiliproteins | Marine-derived; scalable in bioreactors; rich in reducing biomolecules | Antimicrobial and antifungal applications; emerging interest in biostimulant effects |
| Bacillus spp. | Bacteria | AgNPs | 5–20 | Enzymes (nitrate reductase), peptides, NADH | Very small, monodisperse NPs; potential for genetic enhancement | Antimicrobial applications; typically combined with plant-extract synthesis |
| Trichoderma / Aspergillus spp. | Filamentous fungi | AgNPs / AuNPs | 10–50 | Extracellular proteins, glucoamylase, cellulase | High NP yield; protein-rich capping enhances stability; can use agricultural substrates | Antifungal; compatible with biocontrol-based integrated pest management strategies |
| Citrus peel / Onion skin (agri-waste) | Agricultural by-product | AgNPs / AuNPs | 15–55 | Ascorbic acid, flavonoids (hesperidin, quercetin) | Upcycles farm waste; low cost; no dedicated crop cultivation required | Antimicrobial applications; potential use in postharvest disease control |
4. Mechanisms of Reduction and Stabilization
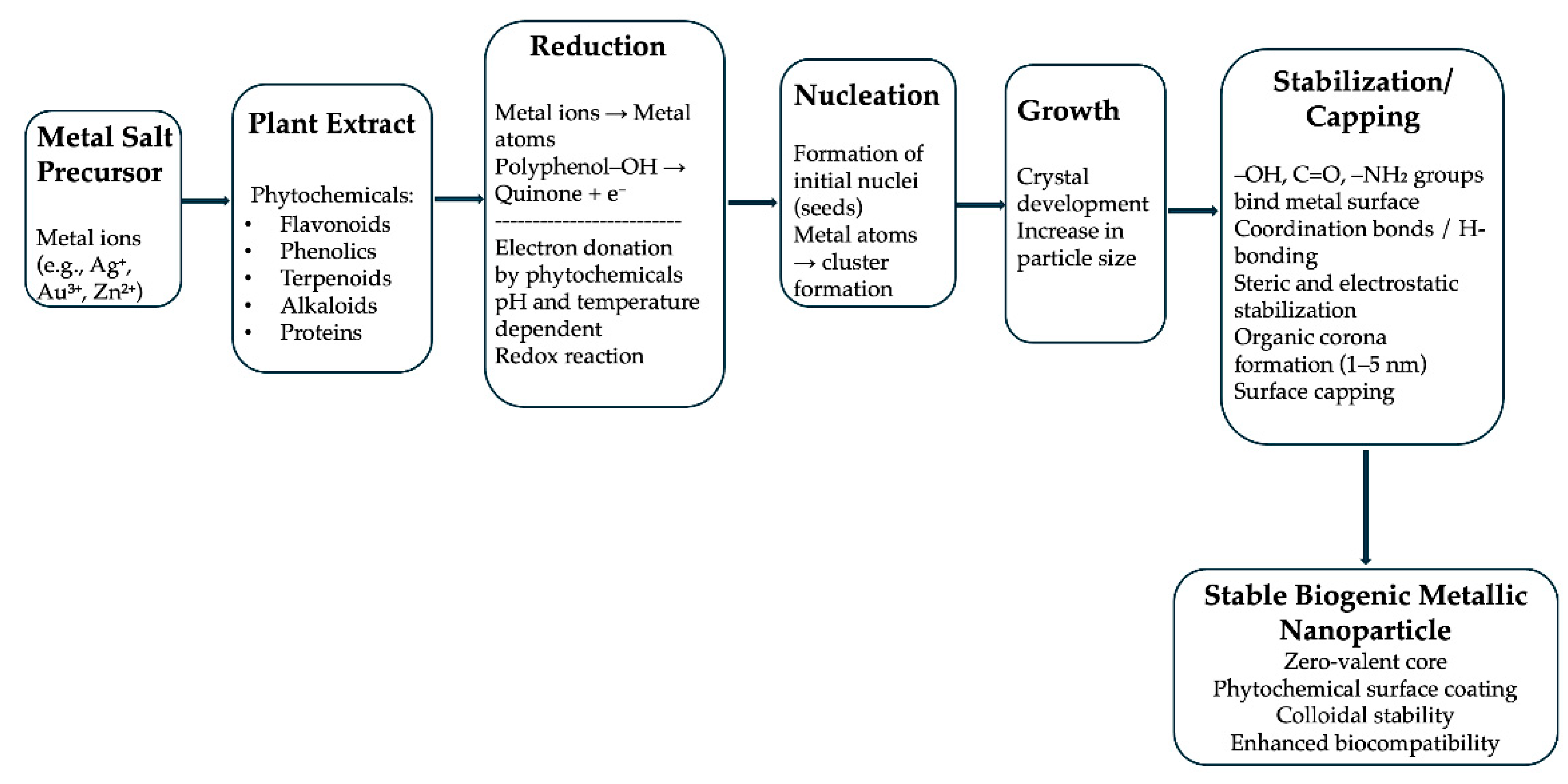
5. Characterization Methods
- UV-Visible Spectroscopy (UV-Vis): This is often the first and simplest tool. Metallic nanoparticles exhibit surface plasmon resonance (SPR) absorption bands in the UV-Vis range, which confirm their formation and give insight into size and concentration. For example, silver nanoparticles typically show an SPR peak around 400–450 nm (brownish colloid), whereas gold nanoparticles show a peak around 520–550 nm (reddish colloid) [16]. The position and width of the SPR band indicate particle size and dispersion, a red-shift or broadening might suggest larger or polydisperse particles, or aggregation. In green synthesis, UV-Vis can also be used kinetically to monitor the reaction progress by observing the growth of the nanoparticle SPR peak over time [17].
- Dynamic Light Scattering (DLS) and Zeta Potential: DLS provides the hydrodynamic diameter of nanoparticles in colloid, which is useful for estimating the size distribution in solution (including the capping layer and any solvation shell). Zeta potential measurement reveals the surface charge, which is an indicator of colloidal stability. Green-synthesized NPs often carry a negative zeta potential due to deprotonated carboxylate or phenolic groups from capping biomolecules [24]. A zeta potential magnitude above ±25 mV usually implies good electrostatic stability. These measurements help ensure that the biogenic NPs will remain well-dispersed when applied (e.g., in foliar sprays) [24].
- Fourier-Transform Infrared Spectroscopy (FTIR): FTIR is extensively used to identify the functional groups of phytochemicals attached to the nanoparticle surface [17]. By comparing the FTIR spectrum of the pure plant extract with that of the dried nanoparticle powder, one can observe shifts or attenuations in certain peaks. For instance, a decrease in the O–H stretching intensity and appearance of C=O peaks on the nanoparticle sample might indicate that polyphenols have oxidized and bound to the NP surface as quinones or carboxylates [17]. FTIR evidence of N–H or C–N vibrations on NPs would suggest proteins or amino acids capping the particles. Thus, FTIR confirms that the nanoparticles are indeed bio-capped and provides clues about which biomolecules are involved (e.g., peaks around 1600 cm^-1 for aromatic C=C suggests polyphenols; amide I and II bands ~1650 and 1540 cm^-1 suggest proteins) [17].
- X-Ray Diffraction (XRD): XRD is used to determine the crystal structure and phase of metallic nanoparticles [5]. Green-synthesized metallic NPs (like Ag, Au, Cu) typically show distinct Bragg reflections corresponding to their face-centered cubic (fcc) lattices. For example, AgNPs will exhibit XRD peaks at 2θ ≈ 38°, 44°, 64°, 77° corresponding to the (111), (200), (220), (311) planes of fcc silver [16]. The presence of these peaks confirms that the product is crystalline metal and not some unreduced compound. The average crystalline size can also be estimated from peak broadening using the Scherrer equation, often yielding size estimates in the 5–50 nm range consistent with microscopy. XRD patterns sometimes contain additional features due to the organic matrix (which generally appears as a broad amorphous background). But overall, XRD verifies that metallic nanoparticles with the expected crystal structure have been synthesized.
- Electron Microscopy (TEM and SEM): Transmission electron microscopy (TEM) is the gold standard to directly visualize nanoparticles, measure their size and shape, and assess dispersion/agglomeration at the nanoscale. Green-synthesized nanoparticles are commonly imaged by TEM, which often reveals roughly spherical particles with size distributions that can be measured (TEM can pick up the inorganic core but not the organic shell unless staining is used). For example, a TEM image might show AgNPs of ~10 nm average diameter synthesized with curry leaf extract, along with a thin halo around each particle indicating the organic corona [17]. High-resolution TEM (HRTEM) can resolve lattice fringes, confirming crystallinity and allowing identification of crystal facets. Selected area electron diffraction (SAED) patterns further corroborate the crystalline nature as rings matching the metal’s known d-spacings [16]. Scanning electron microscopy (SEM) can also be used, especially for larger nanoparticles or to examine surface morphology of nanoparticle-coated plant materials (e.g., a leaf after nano-treatment). However, SEM typically provides lower resolution for sub-20 nm particles compared to TEM. Energy-dispersive X-ray spectroscopy (EDS) attached to SEM/TEM can confirm the elemental composition (peaks for the metal and possibly signals for elements like O, N from the capping). For instance, EDS of biosynthesized AuNPs will show a strong Au peak and weaker signals for C and O from the capping layer [16].
- Thermal and Surface Analysis: Techniques like thermogravimetric analysis (TGA) can quantify the amount of organic capping by showing weight loss at certain temperature ranges (e.g., a 10% weight loss at 200–500°C might correspond to burning off plant organics on the NP) [17]. Similarly, BET surface area analysis can be used if needed to understand surface area of dried nanopowders, though this is less common for colloidal use. X-ray photoelectron spectroscopy (XPS) is another advanced tool to analyze the surface chemistry and oxidation states; for green NPs, XPS can confirm the metal is in the 0-oxidation state and that surface elements like O, N from organics are present [16].
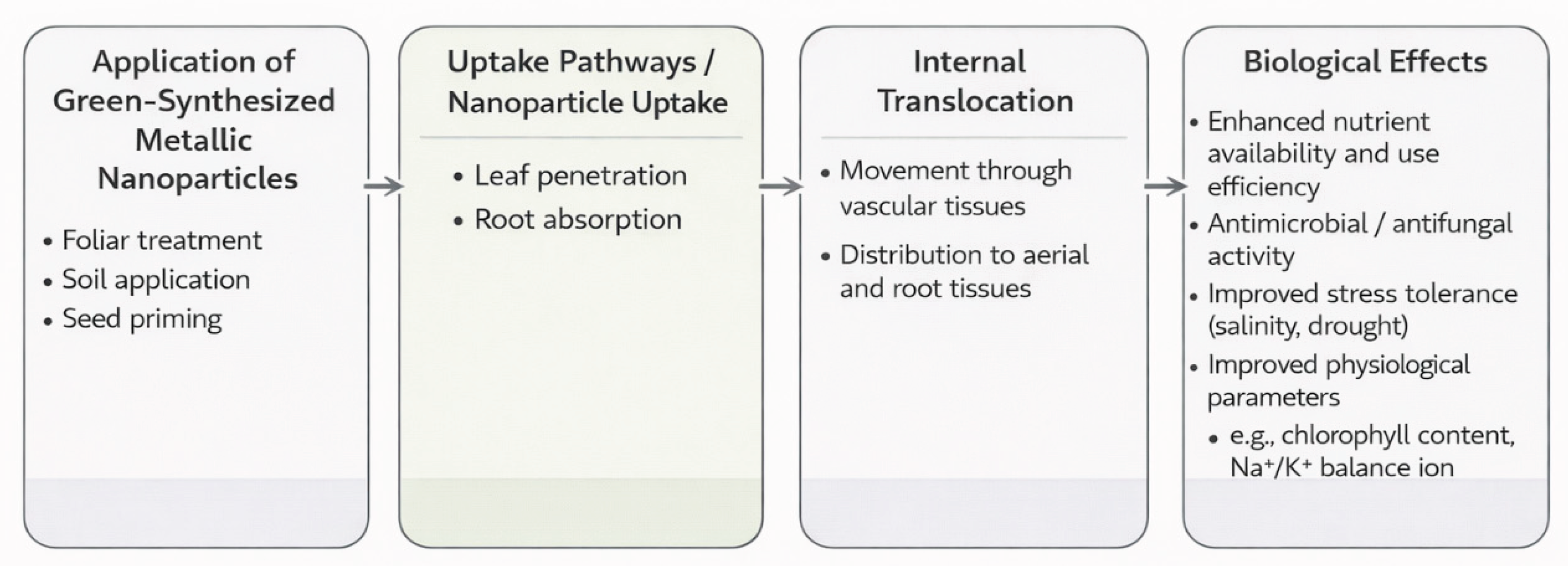
6. Applications in Agriculture
6.1. Nanofertilizers and Growth Enhancement
6.2. Nanopesticides and Disease Management
6.3. Enhancing Stress Tolerance and Productivity
| Crop | NP Type (Biogenic Source) | Dose / Concentration | Application Method | Study Scale | Main Outcomes | Application Category |
| Wheat | Nano-NPK formulations (plant-extract-assisted) | 50% of standard dose | Soil application | Field / greenhouse | Equivalent or higher yield vs. full conventional dose; reduced fertiliser input by ~50% | Nanofertiliser |
| Wheat | Nano-NPK + nano-silica (biogenic) | Low-moderate | Soil + foliar | Semi-arid field | More grains per spike; higher grain weight under drought; prolonged photosynthetic activity | Nanofertiliser / Stress mitigation |
| Rice | Nano-fertilisers (biogenic NPK) | Reduced vs. conventional | Soil application | Greenhouse / field | Yield increase of 13–40% reported across studies | Nanofertiliser |
| Rice | AgNPs (yeast extract) | Low dose (seed priming solution) | Seed priming | Laboratory / greenhouse | Improved germination percentage and early seedling vigour vs. untreated controls | Nanofertiliser |
| Maize | AgNPs / AuNPs (plant extract) | Very low (µg/mL range) | Seed priming | Laboratory | Enhanced seedling vigour; increased amylase activity; faster germination | Nanofertiliser |
| Soybean | Fe₃O₄-NPs (tea polyphenol-coated) | Single soil application | Soil drench | Pot study | Season-long Fe supply; improved chlorophyll and yield vs. bulk Fe requiring multiple applications | Nanofertiliser |
| Soybean | Fe₃O₄-NPs (green-synthesized) | Low dose | Soil application | Pot study | More extensive root system; higher nodule number; increased N fixation and yield | Nanofertiliser / Growth stimulation |
| Tomato | CuNPs (plant extract) | Lower Cu than Cu(OH)2 sprays | Foliar spray | Greenhouse | Control of fungal blight comparable to conventional Cu spray at reduced Cu dose; lower phytotoxicity | Nanopesticide |
| Citrus | CuNPs (plant extract) | Reduced vs. conventional | Foliar spray | Greenhouse | Effective control of bacterial spot at lower copper concentration than traditional salts | Nanopesticide |
| Cucurbits | ZnO-NPs (gum arabic + leaf extract) | Moderate | Foliar spray | Greenhouse | Reduced Erysiphe (powdery mildew) infection via oxidative damage to fungal spores | Nanopesticide |
| Rice | AgNPs (Ocimum sanctum extract) | Moderate spray concentration | Foliar spray | Greenhouse / field | Significant reduction of brown planthopper population | Nanopesticide |
| Various (soil) | AgNPs (Glycyrrhiza glabra) | Soil drench concentration | Soil drench | Laboratory / greenhouse | Complete inhibition of Meloidogyne incognita egg hatching; enhanced antioxidant activity in host roots | Nanopesticide (nematicidal) |
| Cabbage | Cu / Fe / Zn NPs (Tridax procumbens) | Soil drench | Soil application | Laboratory / greenhouse | Reduced root-knot nematode infection; high nematode mortality rates | Nanopesticide (nematicidal) |
| Pea | ZnO-NPs (plant extract) | 20 ppm (optimal dose) | Foliar / soil | Greenhouse | Higher chlorophyll content; better growth; lower Na accumulation under salt stress | Stress mitigation (salinity) |
| Cotton | ZnO-NPs (biogenic) | 20 ppm | Foliar spray | Greenhouse | Improved Na+/K+ ratio; modulation of stress-responsive genes; maintained K in leaves under salinity | Stress mitigation (salinity) |
| Wheat / Cotton | Si-NPs (green-synthesized) | Moderate | Foliar spray | Greenhouse | Improved Na+/K+ ratio; enhanced proline accumulation; better growth under salt stress | Stress mitigation (salinity) |
| Cucumber / Tomato | SiO2-NPs (rice husk ash, biogenic) | Foliar spray dose | Foliar spray | Greenhouse | Increased plant height; higher chlorophyll; improved yield under drought by reducing water loss | Nanofertiliser / Stress mitigation (drought) |
7. Challenges and Future Perspectives
8. Conclusions
References
- Rizzo, DM; Lichtveld, M; Mazet, JA; Togami, E; Miller, SA. Plant health and its effects on food safety and security in a One Health framework: four case studies. One Health Outlook 2021, 3(1), 6. [Google Scholar] [CrossRef] [PubMed]
- Rajput, VD; Minkina, T; Kumari, A; Harish; Singh, VK; Verma, KK; et al. Coping with the challenges of abiotic stress in plants: new dimensions in the field application of nanoparticles. Plants 2021, 10(6), 1221. [Google Scholar] [CrossRef]
- Usman, M; Farooq, M; Wakeel, A; Nawaz, A; Cheema, SA; ur Rehman, H; et al. Nanotechnology in agriculture: current status, challenges and future opportunities. Science of the Total Environment 2020, 721, 137778. [Google Scholar] [CrossRef]
- Ying, S; Guan, Z; Ofoegbu, PC; Clubb, P; Rico, C; He, F; et al. Green synthesis of nanoparticles: current developments and limitations. Environmental Technology & Innovation 2022, 26, 102336. [Google Scholar] [CrossRef]
- Beig, B; Niazi, MBK; Sher, F; Jahan, Z; Malik, US; Khan, MD; et al. Nanotechnology-based controlled release of sustainable fertilizers: a review. Environmental Chemistry Letters 2022, 20(4), 2709–2726. [Google Scholar] [CrossRef]
- Cummings, CL; Kuzma, J; Kokotovich, A; Glas, D; Grieger, K. Barriers to responsible innovation of nanotechnology applications in food and agriculture: a study of US experts and developers. NanoImpact 2021, 23, 100326. [Google Scholar] [CrossRef]
- Adeel, M; Shakoor, N; Shafiq, M; Pavlicek, A; Part, F; Zafiu, C; et al. A critical review of the environmental impacts of manufactured nano-objects on earthworm species. Environmental Pollution 2021, 290, 118041. [Google Scholar] [CrossRef]
- Quintarelli, V; Ben Hassine, M; Radicetti, E; Stazi, SR; Bratti, A; Allevato, E; et al. Advances in nanotechnology for sustainable agriculture: a review of climate change mitigation. Sustainability 2024, 16(21), 9280. [Google Scholar] [CrossRef]
- Zeng, S.; Shakoor, N.; Rui, Y. Nanotechnology and Agricultural Sustainability: A Review. Nanomaterials 2025, 15(23), 1755. [Google Scholar] [CrossRef]
- Atanda, S. A.; Shaibu, R. O.; Agunbiade, F. O. Nanoparticles in agriculture: balancing food security and environmental sustainability. Discover Agriculture 2025, 3(1), 26. [Google Scholar] [CrossRef]
- Hong, J; Wang, C; Wagner, DC; Gardea-Torresdey, JL; He, F; Rico, CM. Foliar application of nanoparticles: mechanisms of absorption, transfer, and multiple impacts. Environmental Science: Nano 2021, 8(5), 1196–1210. [Google Scholar] [CrossRef]
- Ali, A; Aasim, M; Çelik, K; Nadeem, MA; Baloch, FS. Frontiers in bacterial-based green synthesized nanoparticles (NPs): a sustainable strategy for combating infectious plant pathogens. Biocatalysis and Agricultural Biotechnology 2024, 60, 103293. [Google Scholar] [CrossRef]
- Khot, L. R.; Sankaran, S.; Maja, J. M.; Ehsani, R.; Schuster, E. W. Applications of nanomaterials in agricultural production and crop protection: a review. Crop protection 2012, 35, 64–70. [Google Scholar]
- Raliya, R; Saharan, V; Dimkpa, C; Biswas, P. Nanofertilizer for precision and sustainable agriculture: current state and future perspectives. Journal of Agricultural and Food Chemistry 2018, 66(26), 6487–6503. [Google Scholar]
- Iravani, S. Green synthesis of metal nanoparticles using plants. Green chemistry 2011, 13(10), 2638–2650. [Google Scholar]
- Patil, S; Chandrasekaran, R. Biogenic nanoparticles: a comprehensive perspective in synthesis, characterization, application and its challenges. Journal of Genetic Engineering and Biotechnology 2020, 18(1), 67. [Google Scholar] [CrossRef]
- Jadoun, S; Arif, R; Jangid, NK; Meena, RK. Green synthesis of nanoparticles using plant extracts: a review. Environmental Chemistry Letters 2021, 19(1), 355–374. [Google Scholar] [CrossRef]
- Hano, C; Abbasi, BH. Plant-based green synthesis of nanoparticles: production, characterization and applications. Biomolecules 2021, 12(1), 31. [Google Scholar] [CrossRef]
- Martínez-Cisterna, D; Rubilar, O; Tortella, G; Chen, L; Chacón-Fuentes, M; Lizama, M; et al. Silver nanoparticles as a potent nanopesticide: toxic effects and action mechanisms on pest insects of agricultural importance—a review. Molecules 2024, 29(23), 5520. [Google Scholar] [CrossRef]
- Dikshit, P. K.; Kumar, J.; Das, A. K.; Sadhu, S.; Sharma, S.; Singh, S.; et al. Green synthesis of metallic nanoparticles: Applications and limitations. Catalysts 2021, 11(8), 902. [Google Scholar] [CrossRef]
- Rajput, V; Minkina, T; Mazarji, M; Shende, S; Sushkova, S; Mandzhieva, S; et al. Accumulation of nanoparticles in the soil-plant systems and their effects on human health. Annals of Agricultural Sciences 2020, 65(2), 137–143. [Google Scholar] [CrossRef]
- Li, SN; Wang, R; Ho, SH. Algae-mediated biosystems for metallic nanoparticle production: from synthetic mechanisms to aquatic environmental applications. Journal of Hazardous Materials 2021, 420, 126625. [Google Scholar] [CrossRef]
- Khan, AU; Khan, M; Khan, AA; Parveen, A; Ansari, S; Alam, M. Effect of phyto-assisted synthesis of magnesium oxide nanoparticles (MgO-NPs) on bacteria and the root-knot nematode. Bioinorganic Chemistry and Applications 2022, 2022(1), 3973841. [Google Scholar]
- Shandila, P; Mahatmanto, T; Hsu, JL. Metal-based nanoparticles as nanopesticides: opportunities and challenges for sustainable crop protection. Processes 2025, 13(5), 1278. [Google Scholar] [CrossRef]
- Al Banna, LS; Salem, NM; Jaleel, GA; Awwad, AM. Green synthesis of sulfur nanoparticles using Rosmarinus officinalis leaves extract and nematicidal activity against Meloidogyne javanica. Chemistry International 2020, 6(3), 137–143. [Google Scholar]
- Sherin, S; Rose Rizvi, NF; Parveen, M; Jaseem, KP; Favas, A; Mubeena, ES. Innovative nano-solution: bio-synthesized nickel oxide nanoparticles (NPs) protect carrot roots from root-knot nematode Meloidogyne incognita infestation. International Journal of Environment, Agriculture and Biotechnology 2024, 9, 28–40. [Google Scholar] [CrossRef]
- Ahmad, S; Ahmad, MA; Umar, F; Munir, I; Iqbal, N; Ahmad, N; et al. Green nano-synthesis: Salix alba bark-derived zinc oxide nanoparticle and their nematicidal efficacy against root-knot nematode Meloidogyne incognita. Advancements in Life Sciences 2024, 10(4), 675–681. [Google Scholar] [CrossRef]
- Kim, DY; Kim, M; Sung, JS; Koduru, JR; Nile, SH; Syed, A; et al. Extracellular synthesis of silver nanoparticles using yeast extracts: antibacterial and seed priming applications. Applied Microbiology and Biotechnology 2024, 108(1), 150. [Google Scholar] [CrossRef]
- Basavegowda, N; Baek, KH. Multimetallic nanoparticles as alternative antimicrobial agents: challenges and perspectives. Molecules 2021, 26(4), 912. [Google Scholar] [CrossRef]
- Khan, M; Khan, AU; Bogdanchikova, N; Garibo, D. Antibacterial and antifungal studies of biosynthesized silver nanoparticles against plant parasitic nematode Meloidogyne incognita and plant pathogens Ralstonia solanacearum and Fusarium oxysporum. Molecules 2021, 26(9), 2462. [Google Scholar] [CrossRef] [PubMed]
- Rani, K; Devi, N; Banakar, P; Kharb, P; Kaushik, P. Nematicidal potential of green silver nanoparticles synthesized using aqueous root extract of Glycyrrhiza glabra. Nanomaterials 2022, 12(17), 2966. [Google Scholar] [CrossRef]
- Soliman, MM; Abdallah, AM; Hafez, E; Kadous, E; Kassem, FA. Nematicidal activity of chemical and green biosynthesis of copper nanoparticles against root-knot nematode Meloidogyne incognita. Alexandria Science Exchange Journal 2022, 43(4), 583–591. [Google Scholar] [CrossRef]
- Sarip, NA; Aminudin, NI; Danial, WH. Green synthesis of metal nanoparticles using Garcinia extracts: a review. Environmental Chemistry Letters 2022, 20(1), 469–493. [Google Scholar] [CrossRef]
- Thiruvenkataswamy, S; Paulpandi, S; Narayanasamy, M. Biosynthesis, characterization, and nematicidal efficacy of silver nanoparticles synthesized using Solanum nigrum fruit against root-knot nematode Meloidogyne incognita. Nano Science and Nano Technology: An Indian Journal 2022, 16(1), 140. [Google Scholar]
- Nasr, A; Yousef, AF; Hegazy, MG; Abdel-Mageed, MA; Elshazly, EH; Gad, M; et al. Biosynthesized silver nanoparticles mitigate charcoal rot and root-knot nematode disease complex in faba bean. Physiological and Molecular Plant Pathology 2025, 136, 102610. [Google Scholar] [CrossRef]
- Fabiyi, O; Lateef, A; Gueguim-Kana, EB; Beukes, LS; Matyumza, N; Bello, T; et al. Characterization and nematicidal potential of copper, iron and zinc nanoparticles synthesized from Tridax procumbens L. extract on Meloidogyne incognita-infected cabbage plants. European Journal of Plant Pathology 2024, 168(4), 683–695. [Google Scholar]
- Danish, M; Altaf, M; Robab, MI; Shahid, M; Manoharadas, S; Hussain, SA; et al. Green synthesized silver nanoparticles mitigate biotic stress induced by Meloidogyne incognita in Trachyspermum ammi (L.) by improving growth, biochemical, and antioxidant enzyme activities. ACS Omega 2021, 6(17), 11389–11403. [Google Scholar] [CrossRef]
- El-Saadony, M. T.; Saad, A. M.; Soliman, S. M.; Salem, H. M.; Desoky, E. S. M.; Babalghith, A. O.; et al. Role of nanoparticles in enhancing crop tolerance to abiotic stress: A comprehensive review. Frontiers in Plant Science 2022, 13, 946717. [Google Scholar] [CrossRef] [PubMed]
- Yadav, A; Yadav, K; Abd-Elsalam, KA. Exploring the potential of nanofertilizers for a sustainable agriculture. Plant Nano Biology 2023, 5, 100044. [Google Scholar] [CrossRef]
- Djanaguiraman, M; Anbazhagan, V; Dhankher, OP; Prasad, PV. Uptake, translocation, toxicity, and impact of nanoparticles on plant physiological processes. Plants 2024, 13(22), 3137. [Google Scholar] [CrossRef]
- Qian, J.; Shan, R.; Shi, Y.; Li, H.; Xue, L.; Song, Y.; et al. Zinc oxide nanoparticles alleviate salt stress in cotton (Gossypium hirsutum L.) by adjusting Na+/K+ ratio and antioxidative ability. Life 2024, 14(5), 595. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Yin, L.; Deng, X.; Wang, S.; Tanaka, K.; Zhang, S. Aquaporin-mediated increase in root hydraulic conductance is involved in silicon-induced improved root water uptake under osmotic stress in Sorghum bicolor L. Journal of Experimental Botany 2014, 65(17), 4747–4756. [Google Scholar] [CrossRef] [PubMed]
- EFSA Scientific Committee. Guidance on risk assessment of nanomaterials to be applied in the food and feed chain: human and animal health. EFSA Journal. 2021, 19(8), e06768. [Google Scholar]
- Kookana, RS; Boxall, ABA; Reeves, PT; Ashauer, R; Beulke, S; Chaudhry, Q; et al. Nanopesticides: Guiding Principles for Regulatory Evaluation of Environmental Risks. Journal of Agricultural and Food Chemistry 2014, 62(18), 4227–4240. [Google Scholar] [CrossRef] [PubMed]
- Kumari, S; Sharma, N; Sehrawat, R; Yadav, M; Singh, G; Rathore, SS; et al. Regulation and safety measures for nanotechnology-based agri-products. Frontiers in Genome Editing 2023, 5, 1200987. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




