1. Introduction
Virus-induced diabetes mellitus has been documented in numerous human cases especially following infection with enteroviruses [
1]. Recent observations suggest that cats infected with SARS-CoV-2 may exhibit multiple forms of diabetes, including Type 1 Diabetes Mellitus (T1DM), as well as severe metabolic complications such as diabetic ketoacidosis (DKA) and hyperosmolar state [
2,
3]. Experimentally infected cats in the laboratory have demonstrated significant hyperglycemia and SARS-CoV-2 particles and antigens have been identified within pancreatic islet cells in humans [
4,
5]. Feline animal models of SARS-CoV-2 infection are valuable for elucidating the pathologic mechanisms leading to T1DM.
T1DM imposes substantial subclinical, economic, and social burdens. Historical precedents for viral etiology include a notable case of DKA in a child where Coxsackievirus B4 was isolated from the pancreas and the viral strain was proven to be diabetogenic in murine models [
6]. Similar associations where Koch’s postulates appear applicable to virus-induced diabetes in humans have since been reported for Coxsackievirus B5 [
7] and other enteroviruses [
8]. Since the onset of the COVID-19 pandemic, evidence has increasingly suggested a link between T1DM and SARS-CoV-2 infection [
4]. However, much of the current literature remains circumstantial or limited to case reports. Here, we provide some data for satisfying Koch’s postulates in animal models of SARS-CoV-2 infection.
2. Viral and Prion-Associated Diabetes Mellitus
Type 1 Diabetes Mellitus (T1DM) is mainly a disease of the youth which, if left untreated, culminates in fatal ketoacidosis as insulin production ceases. However, contemporary research suggests a more nuanced, slower progression toward complete insulin deficiency [
9]. Alternatively, authors propose an episodic decline, where successive bouts of beta-cell-damaging insults are interspersed with periods of partial recovery [
10,
11]. The prevalence of T1DM exhibits a distinct seasonality—peaking in winter and declining in summer—particularly in northern latitudes [
12]. This geographical and temporal variation appears linked to the circulation of specific enteroviruses, notably Coxsackievirus B1, B2, B4, and B5, as well as Echovirus 9 and 11, which some investigators term "diabetogenic viruses". Furthermore, spatial and familial clustering of T1DM has been documented in smaller populations [
13,
14].
Experimental murine models have demonstrated that inoculation with Coxsackievirus B4 leads to acute and persistent pancreatic infection [
15], a phenomenon also confirmed in the myocardium of infected mice [
16]. Similarly, human-isolated Reovirus types 1, 2, and 3 primarily target the gastrointestinal tract; however, when passaged in primary beta-cell cultures, they induce hypothyroidism and diabetes in neonatal mice [
17]. Ultrastructural analysis has revealed crystalline arrays of Reovirus type 1 within the cytoplasm of pancreatic islet α, β, and δ-cells [
18], illustrating the unexpected tropism these viruses exhibit toward various endocrine organs [
19].
In vitro studies using human pancreatic organ cultures have confirmed this tropism, observing both lytic and persistent enteroviral infections in insulin-producing and non-insulin-producing cells. In these lytic infections, the primary mechanism of cell death is characterized by initial pyknosis followed by delayed necrosis.
Prions represent a unique class of infectious agents devoid of nucleic acids, causing progressive neurodegenerative diseases with prolonged incubation periods. These diseases may arise spontaneously, through genetic inheritance, or via transmission through bodily fluids, environmental contact, or medical intervention [
20]. Pathologically, prion diseases are classified as protein misfolding disorders (PMDs), characterized by the systemic or localized accumulation of amyloid aggregates.
Felines are among the few species that develop a form of diabetes mellitus analogous to human Type 2 Diabetes Mellitus (T2DM). A hallmark of feline T2DM is the deposition of amyloid within the islets, derived from the beta-cell hormone amylin, or islet amyloid polypeptide (IAPP). Elevated IAPP concentrations in the islet cells and plasma of diabetic cats support the pathogenic role of this polypeptide [
21]. Similarly, IAPP accumulation is a frequent finding in human T2DM. Evidence suggests that IAPP aggregates can seed the misfolding of endogenous IAPP. For instance, the inoculation of IAPP aggregates into transgenic mice expressing human IAPP triggers accelerated amyloid deposition. This suggests that T2DM might be transmissible through ways reminiscent of prion propagation in neurological diseases [
22]. Notably, the nonapeptide SK9 (residues 55-63; SFYVYSRVK) from the SARS-CoV-2 envelope has demonstrated the capacity to induce IAPP-like structures and promote advanced aggregation [
23]. However, the specific role of IAPP during coronavirus infection remains underexplored both in human and feline diabetes.
In addition, beta-cell dysfunction and hyperglycemia have been observed in rodents infected with “scrapie” prions [
1,
24]. The cellular prion protein (PrP
C) is expressed in beta-cells and appears to be a critical regulator of glucose homeostasis. Specifically, PrP
C expression modulates pancreatic iron stores: the silencing of PrP
C leads to intracellular iron depletion and the subsequent upregulation of glucose transporter 2 (GLUT2) and insulin. Conversely, iron overload downregulates GLUT2 and insulin in a PrP
C-dependent manner. Consequently, glucose intolerance develops in iron-overloaded PrP
C{+/+} mice but not in PrP
C{-/-} mice, indicating that PrP
C-mediated modulation of intracellular iron significantly influences both insulin secretion and the sensitivity of target organs [
24,
25].
3. Murine Coronavirus Infection of Pancreas Cells
The association between murine coronaviruses and pancreatic pathology was first highlighted in 1967 by Hirano et al., who documented pancreatitis in mice infected with Mouse Hepatitis Virus-3 (MHV-3) [
26]. These pancreatic lesions exhibit unique features, and disease susceptibility is greatly strain-dependent: DBA/2 and BALB/c mice are susceptible, whereas CFW and CD-1 strains demonstrate resistance. Histologically, these lesions are characterized by coagulative necrosis of acinar cells, with mild inflammatory infiltration observed at the periphery of the endocrine tissue.
While the exact mechanisms underlying these lesions remain to be fully elucidated [
27], it is well-established that murine coronaviruses naturally infect the brain, liver, and pancreas. Despite this, detailed analyses of coronavirus-induced pancreatitis have remained scarce since the initial 1967 report. However, given their multi-organ tropism, murine coronaviruses remain strong candidates for modeling virus-induced diabetes. Further investigations using coronavirus strains originally isolated from wild rodents is likely necessary to better define their role in this endocrine dysfunction.
4. SARS-CoV-2 Infection in Cats and Diabetes Induction
The first natural case of SARS-CoV-2 infection in a domestic cat was reported in 2021 [
28]. Serological surveys conducted during the COVID-19 pandemic revealed a higher prevalence of antiviral antibodies in cats and dogs residing in households with COVID-19 patients [
29]. However, clinical manifestations, such as overt respiratory disease, are rarely reported in these companion animals [
30]. In a French study, respiratory swabs and serological testing identified four natural cases of feline SARS-CoV-2 infection: three were asymptomatic, while one exhibited a coryza-like syndrome [
31]. Genome sequencing identified these isolates as the α-variant [
32].
Subsequent reports documented the α-variant in pets across Texas, USA, Italy, and Spain [
33,
34,
35,
36]. While the δ-variant was initially less frequently reported in Western companion animals, a natural infection in a Japanese cat demonstrated significant virulence, manifesting as severe respiratory distress, sneezing, nasal discharge, and coughing [
37]. These findings suggest that the δ-variant may possess enhanced pathogenicity in felines.
Experimental transmission studies, largely conducted at the BSL-4 facility of the Harbin Veterinary Research Institute in China, have provided deeper insights [
36]. Utilizing both human prototype (Wuhan) strains and environmental isolates, researchers successfully infected cats and dogs intranasally. In felines, the infectious virus was recovered from the nasal cavity, throat, salivary glands, bronchi, and lungs. While viral RNA was detected in the intestines, infectious virions were not recovered from the digestive tract. In contrast, dogs showed evidence of viral RNA in the upper respiratory tract, but no infectious particles were detected, suggesting limited replication [
36].
To assess the impact of SARS-CoV-2 on glucose metabolism, blood glucose levels were monitored in cats following viral inoculation [
38]. At 7 days post-infection (dpi), nearly all inoculated cats exhibited significant hyperglycemia, whereas control groups remained normoglycemic. Although gross pathological examination did not show distinct morphological changes in the pancreas, IHC analysis revealed the expression of the SARS-CoV-2 nucleocapsid protein (NP) within pancreatic islet cells and ductal epithelial cells. Given that ductal epithelial cells differentiate into islet cells during development, this suggests a direct pathway for viral invasion of the endocrine pancreas.
Detailed IHC triple-staining by Zhang et al. for viral NP, glucagon, and insulin revealed a high frequency of NP-glucagon double-positive cells, whereas NP-insulin double-positive cells were rare [
38]. These results were corroborated using S-protein-specific antibodies. Additionally, triple-staining for NP, glucagon, and angiotensin-converting enzyme 2 (ACE2) demonstrated that these cells were triple-positive, suggesting that α-cells are a primary target for SARS-CoV-2 entry via the ACE2 receptor. This tropism mirrors observations in mouse encephalomyocarditis virus (EMCV) models, where viral infection is concentrated at the islet periphery, the region where glucagon-producing alpha-cells are predominantly located (data not shown).
Finally, SARS-CoV-2 appears to induce apoptosis within feline islets. Transferase dUTP nick end labeling (TUNEL) assays confirmed apoptotic signaling in both ductal epithelial and islet cells [
38]. While further research is required to fully characterize these islet abnormalities, these findings likely represent the early phases of pancreatic islet destruction and explain the observed elevations in blood glucose levels following SARS-CoV-2 infection.
5. Possible Antivirals for Coronavirus-Induced Diabetes
For treatment of coronavirus-related manifestations, including virus-induced diabetes, this study focuses on remdesivir, molnupiravir, ritonavir and ensitrelvir, as these agents have been approved by the Japanese government for clinical use in humans. In addition, remdesivir and molnupiravir are being used off-label by Japanese veterinarians to treat distinct feline coronavirus infections.
5.1. Remdesivir
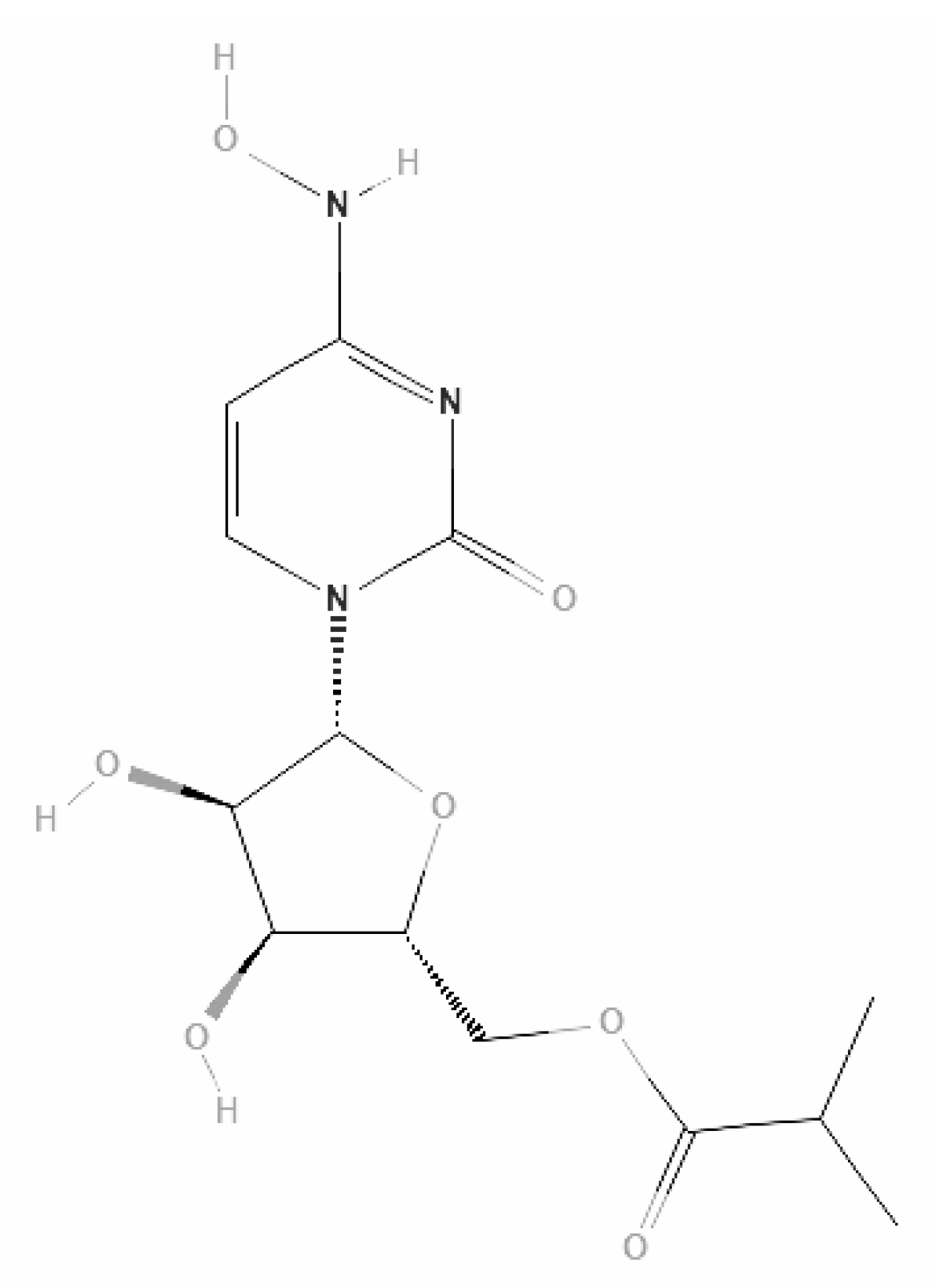
Remdesivir is widely employed in veterinary medicine for the treatment of FIP, a previously fatal disease caused by a feline coronavirus [
39,
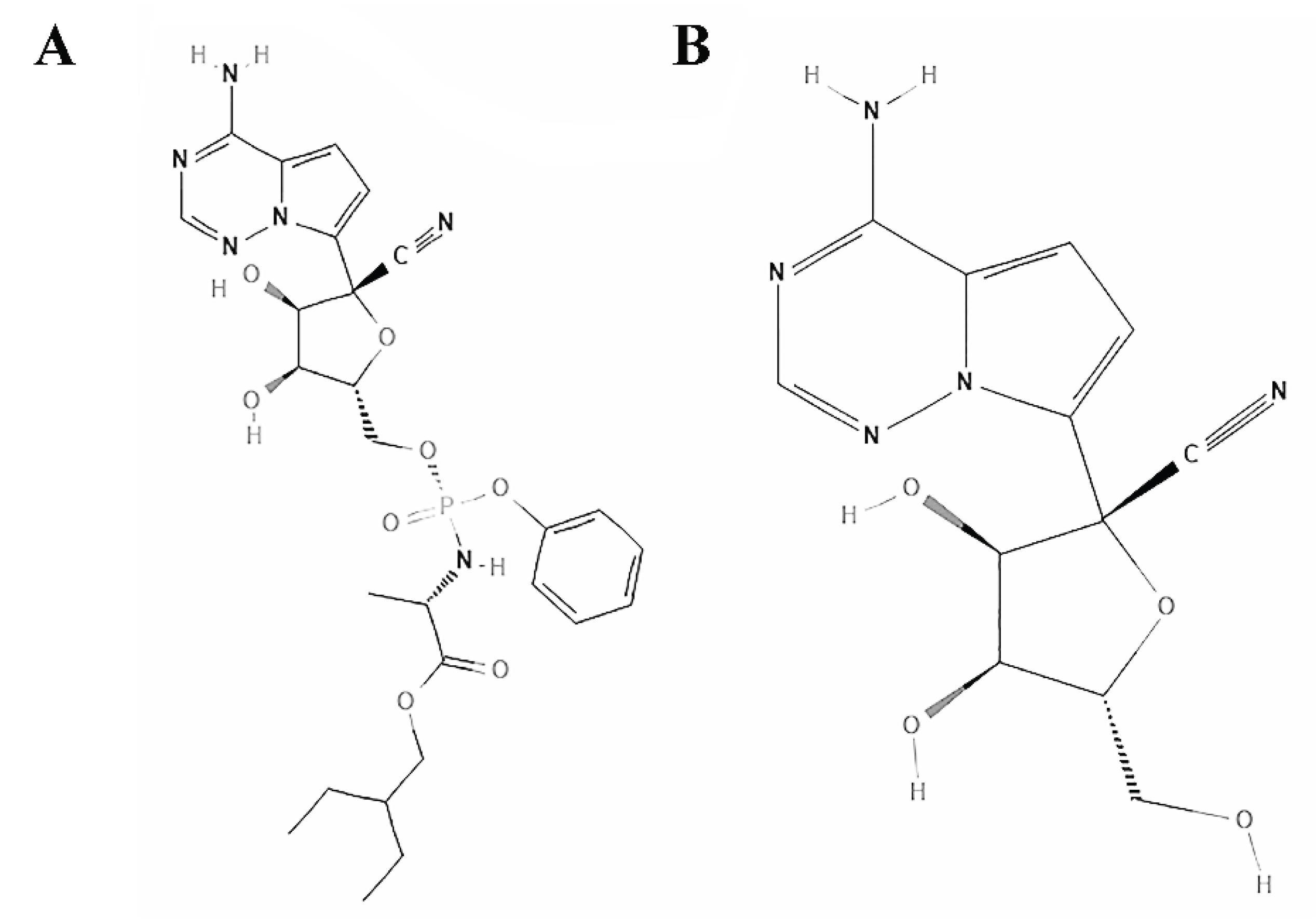
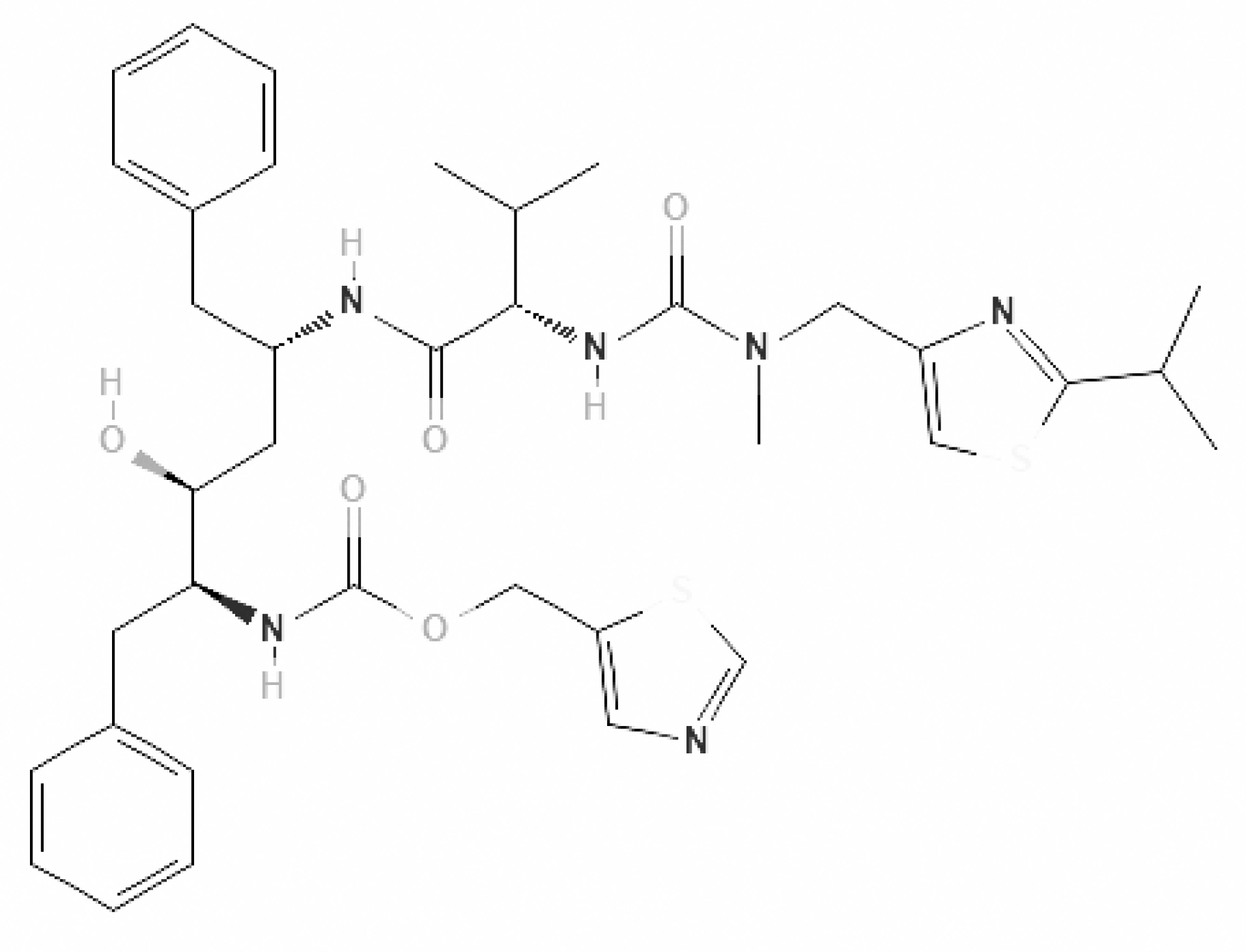
40]. Originally developed to combat the Ebola virus, remdesivir is a prodrug of the parent adenosine analog GS-441524 that is FDA-approved for COVID-19. Both compounds are converted by cellular enzymes into bioactive nucleoside triphosphate (NTP) (
Figure 1).
The parent nucleoside, GS-441524, has demonstrated robust antiviral activity against a spectrum of coronaviruses, including SARS-CoV, MERS-CoV, and feline infectious peritonitis virus (FIPV) [
40]. As a nucleoside analog, remdesivir functions as an inhibitor of the viral RNA-dependent RNA polymerase (RdRp). It competes with natural ATP for incorporation into the nascent RNA strand, leading to premature chain termination or delayed template processing, thereby halting viral genome replication [
39].
Given the genetic heterogeneity among coronavirus strains, the broad-spectrum efficacy of RdRp inhibitors is highly relevant. This genetic diversity is driven by the large viral genome and the unique proofreading capabilities of the viral replication machinery. However, since the RdRp remains a highly conserved enzyme across the Coronaviridae family, remdesivir remains a good candidate for mitigating the systemic effects of SARS-CoV-2, including its impact on pancreatic function and glucose metabolism.
5.2. Molnupiravir
Molnupiravir (EIDD-2801) is a potent orally bioavailable antiviral that has garnered significant attention for its broad-spectrum efficacy against human coronaviruses [
41]. Beyond its applications for SARS-CoV-2, it has demonstrated effectiveness against multiple RNA viruses, including Middle East respiratory syndrome coronavirus (MERS-CoV), seasonal influenza, and Venezuelan equine encephalitis virus (VEEV). The mechanism of action involves the inhibition of the viral RdRp. Unlike remdesivir, which primarily acts via chain termination, molnupiravir induces a "viral error catastrophe" or lethal mutagenesis. Once metabolized into its active form, β-D-N
4-hydroxycytidine triphosphate, it is incorporated into viral RNA. During replication cycles, the polymerase misinterprets the modified nucleotide(s), leading to an accumulation of transition mutations that ultimately render the viral genome non-functional (
Figure 2).
Molnupiravir has received regulatory approval for treatment of COVID-19 in several jurisdictions, including the United Kingdom and Japan. Clinical data suggests a favorable safety profile. While generally well-tolerated, its use is typically monitored to mitigate potential mutagenic risks, particularly in certain patient populations [
41]. Comparative studies have indicated that molnupiravir may exhibit a potency significantly higher - estimated at 2 to 10 times - than that of remdesivir in certain
in vitro models and viral inhibition assays.
The veterinary relevance of this agent is significant, as SARS-CoV-2 can manifest in domestic pets with symptoms mirroring human pathology, including hyperinflammation and the risk of multi-organ failure. This clinical presentation is similar in the systemic inflammatory response seen in FIP [
42]. Consequently, molnupiravir is increasingly being investigated and utilized in feline medicine, leveraging data from COVID-19 clinical trials to treat cats infected with both FIP virus and SARS-CoV-2.
5.3. Ritonavir
Ritonavir, an aspartic protease inhibitor originally developed for the treatment of Human Immunodeficiency Virus (HIV) and Hepatitis C virus (HCV), has been evaluated for its potential
in vitro activity against SARS-CoV and MERS-CoV [
43] (
Figure 3). While it targets the viral 3C-like protease (3CLpro), clinical data has shown that ritonavir monotherapy offers negligible therapeutic benefit against COVID-19.
The primary pharmacological utility of ritonavir in COVID-19 treatment is its role as a pharmacokinetic enhancer or booster. By inhibiting the cytochrome P450 3A4 (CYP3A4) enzyme, ritonavir slows the metabolism of co-administered antiviral agents, thereby increasing their plasma concentrations and extending their half-life. This synergy is most notably observed when used in combination with ribavirin, interferon β-1b, or as part of the nirmatrelvir/ritonavir (Paxlovid) regimen.
Large-scale clinical evidence has led to a shift in how ritonavir is utilized. Specifically, the Randomized Evaluation of COVID-19 Therapy (RECOVERY) trial—a landmark national clinical program in the UK involving 1,596 patients—discontinued the lopinavir/ritonavir treatment arm after observing no significant clinical benefit in hospitalized patients [
43]. Despite this, research continues globally, including initiatives sponsored by the Japanese government, to explore more effective combination therapies.
In veterinary medicine, specifically regarding feline coronaviruses, no clinical trials have yet been conducted to evaluate ritonavir’s efficacy or safety in cats. Given the drug’s complex metabolic profile and significant potential for drug-drug interactions via CYP450 inhibition, its application in veterinary species still requires investigations before considering it a viable treatment for FIP or feline SARS-CoV-2 infections.
5.4. Ensitrelvir
Ensitrelvir is a potent, non-covalent, non-peptide inhibitor of the SARS-CoV-2 main protease 3CLpro which has emerged as a primary therapeutic target due to its essential role in viral polyprotein processing [
44] (
Figure 4). In biochemical assays, ensitrelvir demonstrated high affinity for the wild-type (WT) SARS-CoV-2 protease, with a half-maximal inhibitory concentration IC
50 = 0.049mM. This potency is comparable to that of nirmatrelvir IC
50 = 0.044mM, indicating that ensitrelvir is an effective inhibitor of the viral 3CLpro
in vitro.
While ensitrelvir is currently authorized only for human use, its clinical development is expanding globally. It has moved into Phase 3 clinical trials in the United States and other regions to evaluate its efficacy across broader populations [
44]. In the context of veterinary medicine, like other novel protease inhibitors, ensitrelvir has not yet been evaluated for its safety or efficacy in feline populations, though the conservation of the main protease enzyme (M
pro) target across coronaviruses appears to recommend investigations for feline-specific viral pathogens.
Recent investigations into viral structural biology have revealed that certain viruses, including respiratory syncytial virus (RSV) and herpes simplex virus type 1 (HSV-1), can recruit host-derived proteins to form a specialized layer known as a "protein corona" [
45]. These viruses effectively bind amyloidogenic peptides within their corona, facilitating surface-assisted nucleation and catalyzing the formation of amyloid fibrils (
Figure 5). Metwally reported that diabetes was already known to be a key risk factor for developing severe forms of COVID-19 and people with the condition are more likely to die from the infection. He suggested that the virus might also trigger some form of diabetes [
46].
Clinically, the intersection of COVID-19 and glucose metabolism is becoming increasingly evident. Epidemiological data indicate that SARS-CoV-2 infection can precipitate new-onset diabetes. This manifests as acute hyperglycemia in patients without prior history of the disease and as DKA in patients with pre-existing diabetes [
47]. The SARS-CoV-2 spike protein interacts with the renin-angiotensin-aldosterone system (RAAS), entering host cells by binding to ACE2 receptors. These receptors are expressed across multiple tissues, including the exocrine and endocrine pancreas. Emerging research suggests that viral entry into pancreatic β-cells, coupled with the subsequent upregulation of angiotensin II, may be the primary driver of this diabetogenic effect [
47]. Given the rising incidence of post-COVID-19 metabolic complications, the integrated management of glycemic control is essential during viral recovery and in the long-COVID syndrome.
The expression profile of ACE2 in the human pancreas remains a critical area of study. While earlier reports suggested a widespread expression in pancreas [
48], higher resolution analyses confirm that ACE2 is specifically expressed in both α- and β-cells [
49]. This tropism parallels the affinity for the endocrine pancreas of other diabetogenic viruses such as enteroviruses [
50,
51,
52,
53], Coxsackievirus B [
54] and rotaviruses [
55], that have been implicated in the pathogenesis of T1DM. Consequently, longitudinal monitoring of post-COVID-19 patients is necessary to evaluate the virus long-term contribution to the progression toward multiple forms of diabetes.
Drawing from previous experience with Coxsackievirus B4-related DKA [
6], we propose a four-pronged approach to investigate the role of SARS-CoV-2 in diabetes:
Development of robust animal models specifically designed to study the diabetogenic potential of SARS-CoV-2.
Selection of highly susceptible mouse strains or transgenic models that express human ACE2 under specific promoters.
Isolation and cloning of potentially diabetogenic viral strains via plaque assay to identify genomic variants endowed with β-cell tropism.
Utilization of high-resolution immunofluorescence and immunohistochemistry to definitively localize viral replication within pancreatic islets.
Since genetic predisposition is a cornerstone of susceptibility to T1DM, studies in monozygotic twins suggest that environmental triggers (especially microorganisms) are the most likely etiologic agents. Common intestinal and respiratory viral infections may act as catalysts in T1DM by inducing pro-inflammatory cytokine cascades and facilitating the aberrant activation of T-cells [
56]. Understanding whether SARS-CoV-2 may act as a trigger will contribute to prevention and treatment attempts.
Figure 5.
IAPP is frequently observed in the diabetics. SARS-CoV-2 SK9 show increase of the production in the islets for the mature forms [
23]. That will develop progress of diabetes. SARS-CoV-2 interacts to ACE2 and helps invasion to β-cells. Feline type 1 diabetes is induced with some unknown process [
38]. Secondary or bystander effect of T cells will enhance the disease process inside the pancreatic islets [
56].
Figure 5.
IAPP is frequently observed in the diabetics. SARS-CoV-2 SK9 show increase of the production in the islets for the mature forms [
23]. That will develop progress of diabetes. SARS-CoV-2 interacts to ACE2 and helps invasion to β-cells. Feline type 1 diabetes is induced with some unknown process [
38]. Secondary or bystander effect of T cells will enhance the disease process inside the pancreatic islets [
56].
7. Conclusions
Cats are one of the few species that develop a form of diabetes mellitus analogous to T2DM. The characteristic finding in cats with T2DM is deposition within islets of amyloid derived from the β-cell hormone amylin (islet amyloid polypeptide, IAPP). Increased IAPP concentration has been documented in islet cells and plasma of diabetic cats, supporting a pathogenic role for this polypeptide. Interestingly, a part of the nonapeptide of SARS-CoV-2 envelop SK9 (residue 55–63; SFYVYSRVK) showed the ability to induce an IAPP-like structure with an advanced increase in the number of mature fibrils. The role of IAPP during coronavirus infection has not been well studied in human type 2 diabetes or in cats. SARS-CoV-2 virions interact with the receptor ACE2 and help the invasion of islet β-cells. The mechanisms through which SARS-CoV-2 may induce islet damage and diabetes mellitus remain under discussion and warrant further study.
Author Contributions
Conceptualization, T.O., A.S. and S.S.; writing – original draft, T.O.; writing – review & editing, T.O., A.S., S.S., A.T. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
Conflicts of Interest
The authors declare no competing interests.
Abbreviations
| ACE2 |
Angiotensin-converting enzyme 2 |
| DE |
Definitive endothelium |
| DKA |
Diabetic ketoacidosis |
| EMC |
Encephalomyocarditis |
| FIP |
Feline infectious peritonitis |
| GLUT |
Glucose transporter |
| IAPP |
Islet amyloid polypeptide |
| IC₅₀ |
Half-maximal inhibitory concentration |
| IHC |
Immunohistochemical |
| Mpro
|
Main protease enzyme |
| NP |
Nucleocapsid protein |
| PMDs |
Protein misfolding disorders |
| RAAS |
Renin-Angiotensin-Aldosterone System |
| RdRp |
RNA-dependent RNA-polymerase |
| T1DM |
Type 1 diabetes mellitus |
| T2DM |
Type 2 diabetes mellitus |
| TUNEL |
Terminal deoxynucleotidyl transferase dUTP nick end labeling |
| WT |
Wild type |
References
- Toniolo, A.; Cassani, G.; Puggioni, A.; Rossi, A.; Colombo, A.; Onodera, T.; Ferrannini, E. The diabetes pandemic and associated infections: suggestions for clinical microbiology. Rev. Med. Virol., Diabetes Infect. 2019, 30, 1–17.
- Dietter, D.; Bjork, J.; Palmkvist, E.; Carlsson, A. Tends in type 1 diabetes incidence between 2007 and 2023 and their association with SARS-CoV-2 infection in a population-based match cohort study among individuals under 30 years old in Sweden. Diabetologia 2025, 68, 2732–2742.
- Velasquez Garcia, H.A.; Wong, S.; Jeong, D. et al. Long-term risk of incident of type 2 diabetes following SARS-CoV-2 infection: a population-based study in British Columbia, Canada. Diabetes Metab. Res. Rev. 2026, 42, e70136.
- Debuysschere, C.; Nekoha, M.P.; Alidjinou, E.K.; Hober, D. The relationship between SARS-CoV-2 and type 1 diabetes mellitus. Nat. Rev. Endocrinol. 2024, 10, 588–599.
- Steenblock, C.; Richter, S.; Berger, i. et al. Viral infiltration of pancreatic islets in patients with COVID-19. Nat. Commun. 2021, 12, 3534.
- Yoon, J.W.; Austin, M.; Onodera, T.; Notkins, A.L Virus-induced diabetes mellitus, isolation of a virus from the pancreas of a child with diabetic ketoacidosis. New Eng. J. Med. 1979, 300, 1173–1179.
- Champsaur, H.; Dussaix, E.; Samolyk, D.; Fabre. N.; Back, C.; Assan, R. Diabetes and coxsackie B5 infection. Lancet 1980, 1, 251.
- Szopa, T.M.; Titchener, P.A.; Portwood, N.D.; Taylor, K.W. Diabetes due to viruses – some recent developments. Diabetologia 1993, 36, 687–695.
- Nishimura, A.; Matsumura, K.; Kikuno, S.; Nagasawa, K.; Okubo, M.; Mori, Y.; Kobayashi, T. Slowly progressive type 1 diabetes mellitus: Current knowledge and future perspectives. Diabetes Metab. Syndr. Obes. 2019, 12, 2461–2477.
- Toniolo, A.; Onodera, T.; Yoon, J.W.; Notkins, A.L. Induction of diabetes by cumulative environmental insults from virus and chemicals. Nature 1980, 288, 383–385.
- van der Hide, V.; McArdle, S.; Nelson, M.S. et al. Integrated histopathology of the human pancreas throughout stages of type 1 diabetes progression. Nat. Commun. 2026, online ahead of print.
- Vazeou, G.A.; Kordonouri, O.; Witsch, M.; et al. Seasonality at the clinical onset of type 1 diabetes- lessons from the SWEET database. Pediatr. Diabetes 2016, 17, suppl 32–37.
- Hanafusa, T.; Imagawa, A. Fluminant type 1 diabetes: a novel clinical entity requiring special attentions by all medical practitioners. Nat. Clin. Pract. Endoclinol. Metab. 2007, 3, 36–45.
- Salvatoni, A.; Baj, A.; Bianchi, G.; Federico, G.; Colombo, M.; Toniolo, A. Intrafamilial spread of enterovirus infections at the clinical onset of type 1 diabetes. Pediatr. Diabetes 2013, 14, 407–416.
- Toniolo, A.; Onodera, T.; Jordan, G.; Yoon, J.W.; Notkins, A.L. Glucose abnormalities produced in mice by the six members of Coxsackie B virus group. Diabetes 1982, 31, 496–499.
- Bopegamage, S.; Precechteva, J.; Marosova, L.; Stipalova, D.; Sojka, M.; Borsanyiova, M.; Gomolcak, P.; Berkakova, K.; Galama, J.M.D. Outcome of challenge with Coxsackievirus B4 in young mice after maternal infection with the same virus during gestation. FEMS Immunol. Med. Microbiol. 2012, 64, 184–190.
- Onodera, T.; Jenson, A.B.; Yoon, J.W.; Notkins, A.L. Virus-induced diabetes mellitus: reovirus infection of pancreatic beta cells in mice. Science 1978, 201, 529–531.
- Onodera, T.; Toniolo, A.; Ray, U.R.; Jenson, A.B.; Knazek, R.A.; Notkins, A.L. Virus-induced diabetes mellitus. XX. Polyendocrinopathy and autoimmunity. J. Exp. Med. 1981, 153, 1457–1473.
- Srinivasappa, J; Garzelli, C.; Onodera, T.; Ray. U.R.; Notkins, A.L. Endocrinol. 1988, 122, 563–566.
- Condello, C.; Westaway, D.; Prusiner, S.B. Expanding the prion paradigm to include Alzheimer and Parkinson Diseases. JAMA Neurol. 2024, 81, 1023–1024.
- Jutz, T.A.; Rand, J.S. Pathogenesis of feline diabetes mellitus. The veterinary clinics of North America. Small Anim. Pract. 1995, 25, 527–552.
- Mukherjee, A.; Soto, C. Prion-like protein aggregates and type 2 diabetes. Cold Spring Harb. Perspect. Med. 2017, 7, a024315.
- Bilog, M.; Cerosimo, J.; Vigil, I.; Desamero, R.Z.B.; Profit, A.A. Effect of a SARS-CoV-2 protein fragment on the amyloidogenic property of human islet amyloid polypeptide. ACS Chemi. Neurosci. 2024, 15, 4431–4440.
- Onodera, T.; Sakudo, A.; Tsubone, H.; Itohara, S. Review of studies that have used knockout mice to assess normal function of prion protein under immunological or pathophysiological stress. Microbiol. Immunol. 2014, 58, 361–374.
- Ashok, A.; Singh, N. Prion protein modulates glucose homeostasis by altering intracellular iron. Sci. Rep. 2018, 8, 6556.
- Hirano, T. Studies on the pancreatitis in a mouse hepatitis virus (MHV-3) infection. Nagoya J. Med. Sci. 1967, 30, 319–325.
- Craighead, J.E. The role of viruses in the pathogenesis of pancreatic disease and diabetes mellitus. Prog. Med. Virol. 1975, 19, 161–214.
- Pagani, G.; Lai, A.; Berga, A.; Rizzo, A.; Stranieri, A.; Giordano, A.; Paltrinieri, S.; Lelli, D.; Decaro, N.; Rusconi, S. et al. Human-to-cat SARS-CoV-2 transmission; Case report and full-genome sequencing from an infected pet and its owner in Northern Italy, Pathogens 2021, 10, 252.
- USDA APHIS. Cases of SARS-CoV-2 in animals in the United States. Available on line: https//www.aphis.usda.gov/aphis/dashboards/tableau/sars-dashboard (accessed on 27 July 2021).
- Homepage-FEDIAF, Available online: https://fediaf.org/ (accessed on 6 August 2021).
- Kraft, E.; Denolly, S.; Boson, B.; Angelloz-Pessey, S.; Levaltier, S.; Nesi, N.; Corber, S.; Leterrier, B.; Fritz, M.; Leroy, E.M.; et al. Report of one-year prospective surveillance of SARS-CoV-2 in dog and cats in France with various exposure risks: Confirmation of a low prevalence of shedding, detection and complete sequencing of alpha variant in a cat. Viruses 2021, 13, 1759.
- Tracking SARS-CoV-2 variants. Available online: https://www.who.int/emergencies/emergency-health-kits/trauma-emergency-surgery-kit-wto-tesk-2019/tracking-sars-cov-2-variants 8, accessed on 3 August 2021).
- Hamer, S.A.; Chai, R.R.; Zecca, I.B.; Auchland, L.D.; Boundy, C.M.; Davila, E.; Busselman, R.E.; Tang, W.; Pauvolid-Correa, A.; Kilan, M.L.; et al. SARS-CoV-2B.1.1.7 variant of concern detected in a pet dog and cat after exposure to a person with COVID-19, USA. Transbound. Emerg. Dis. 2022, 69, 1656–1658.
- Zoccola, F.; Beltramo, C.; Magris, G.; Peletto, S.; Acutis, P.; Bozzetta, E.; Radovic, S.; Zappulla, F; Porzio, A.M.; Gennero, M.S. et al. First detection of an Italian human-to-cat SARS-CoV-2 alfa variant-linage B.1.1.7. One Health 2021, 13, 100295.
- Barroso-Arevalo, S.; Rivera, B.; Dominguez, L.; Sanchez-Vizcaino, J. First detection of SARS-CoV-2B.1.1.7 variant of concern in an asymptomatic dog in Spain. Viruses 2021, 13, 1379.
- Shi, J.; Wen, Z.; Zhong, G.; Yang, H.; Wang, C.; Huang, B., Liu, R.; He, X.; Shuai, L.; Sun, Z. et al. Susceptibility of ferrets, cats, dogs and other domesticated animals to SARS-coronavirus-2. Science 2020, 368, 1016–1020.
- Yamada, K.; Kuroda, Y.; Yamamoto, T.; Nishio, H.; Yamada, C.; Kobayashi, M.; Morishima, Y.; Maeda, K. A domestic cat with respiratory symptoms caused by severe acute respiratory syndrome coronavirus 2 infection. J. Jpn. Vet. Med. Assoc. 2022, 75, e62–e68.
- Zhang, Y.; Gao, J.; Huang, K.; Zhao, Y.; Hui, X.; Wang, T.; Hu, C.; Sun, X.; Yang, Y.; Wu, C.; Chen, X.; Zou, Z.; Zhao, L. Z.; Jin, M. SARS-CoV-2 infection causes hyperglycemia in cats. J. Inf. Dis. 2022, 226, 1568–1576.
- Vuong, W.; Khan, M.B.; Fischer, C.; arutyunova, E.; Lamer, T.; Shields, J.; Saffan, H.A.; McKay, R.T.; van Belkum, M. J.; Joyce, MA.; Young, H.S.; Tyrrell D.T.; Vederas, J.C.; Lemieux, M.J. Feline coronavirus drug inhibits the main protease of SARS-CoV-2 and block virus replication. Nat. Comm. 2020, 11, 4282.
- Amirian, E.S.; Levy, J.K. Current knowledge about the antivirals remdesivir (GS-5734) and GS-441524 as therapeutic options for coronavirus. One Health 2020, 9, 100128.
- Ender, F.; Saymer, S.; Abacioglu, N.; Sehirli, A.O. Coud molnupiravir has an ameliorative effect in pets with COVID-19? Lett. Appl. NanoBioSci. 2023, 12, 120.
- Sase, O.; Iwami, T.; Sasaki, T.; Sano, T. GS-441524 and molnupiravir are similarly effective for the treatment of cats with feline infectious peritonitis. Front. Vet. Sci. 2024, 11, 14422408.
- Hu, B.; Guo, H.; Zhou, P.; Shi, Z.-L. Characteristics of SARS-CoV-2 and COVID-19. Nat. Rev. Microbiol. 2021, 19, 141–154.
- Lin, M.; Zeng, X.; Duan, Y.; Yang, Z.; Ma, Y.; Yang, H.; Yang, X.; Liu, X. Molecular mechanism of ensitrelvir inhibiting SARS-CoV-2 main protease and its variants. Commun. Biol. 2023, 6, 694.
- Ezzat, K.; Perrnemalm, M.; Palsson, S.; Roberts, T.C.; Jarver, P.; Dondalska, A.; Bestas, B.; Sobkowiak, M.J.; Levanen, B.; Skold, M. et al. The viral protein corona directs pathogenesis and amyloid aggregation. Nat. Commun. 2019, 10, 2331.
- Metwally, A.A.; Mehta, P.; Johnson, B.S.; Nagarjuna, A.; Snyder, M.P. Covid-19-induced new-onset diabetes: trends and technologies. Diabetes 2021, 70, 2733–2744.
- Dahl-Jogansen, K. Virus as the cause of type 1 diabetes. Trend. Mol. Med. 2025, 30, 1020–1027.
- Yang, J. K.; Lin, S.S.; Ji, X.L.; et al. Binding of SARS coronavirus to its receptor damages islets and causes acute diabetes. Acta Diabetol. 2010, 47, 193–199.
- Yang, L.; Han, Y.; Nilsson-Payant, B.E.; Gupta, V.; Wang, P.; Duan, X.; Tang, X.; Zhu, Z.; Jaffre, F.: Zhang, T.; Kim, T.W.; Harschnitz, O.; Redmond, D. et al. A human pluripotent stem cell-based platform to study SARS-CoV-2 tropism and model virus infection in human cell and organoids. Cell Stem Cell 2020, 27, 125–136.
- Rodriguez-Calvo, T. Enterovirus infection and type 1 diabetes: unraveling the crime scene. Clin. Exp. Immunol. 2018, 195, 15-24.
- Poma, A.M.; Genoni, A.; Broccolo, F.; Pugliese, A.; Basolo, F.; Toniolo, A. Immune transcriptome of cells infected with enterovirus strains obtained from cases of type 1 diabetes. Microorganisms 2020, 8, 1031.
- Krogvold, L.; Genoni, A.; Puggioni, A.; Campani, D.; Richardson, S.J.; Flaxman, C.S.; Edwin, B.; Buanes, T.; Dahl-Jorgensen, K.; Toniolo, A. Live enteroviruses, but not other viruses, detected in human pancreas at the onset of type 1 diabetes in the DiViD study. Diabetologia 2022, 65, 2108–2120.
- Federico, G.; Genoni, A.; Puggioni, A.; Saba, A.; Gallo, D.; Randazzo, E.; Salvatoni, A, Toniolo, A. Vitamin D status, enterovirus infection, and type 1 diabetes in Italian children/adolescents. Pediatr. Diabetes 2018, 19, 923–929.
- Carre, A.; Vecchio, F.; Flodstrom-Tullberg, M.; You, S.; Mallone, R. Coxsackievirus and type 1 diabetes: diabetogenic mechanisms and implication for prevention. Endocr. Rev. 2023, 44, 737–751.
- Rogers, M.A.M. Decline in type 1 diabetes in young children after rotavirus vaccination: data from 8 countries. AJPM Focus 2025, 4, 100409.
- Rodriguez-Calvo, T.; Sabouri, S.; Anquetil, F.; von Herrath, M.G. The viral paradigm in type 1 diabetes: who are the main suspects? Autoimm. Rev. 2016, 15, 964–969.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).