Submitted:
15 March 2026
Posted:
16 March 2026
Read the latest preprint version here
Abstract
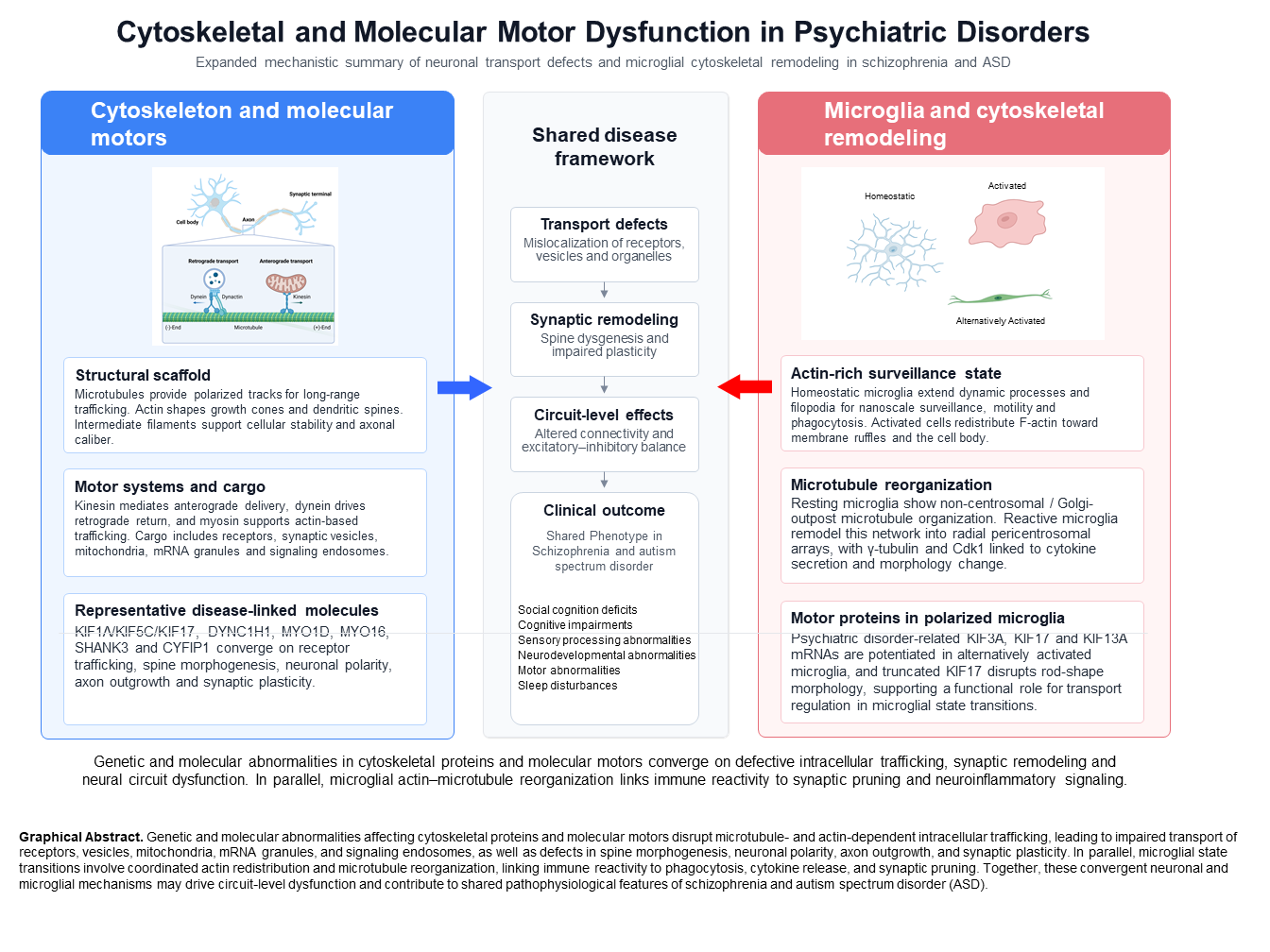
Keywords:
1. Introduction
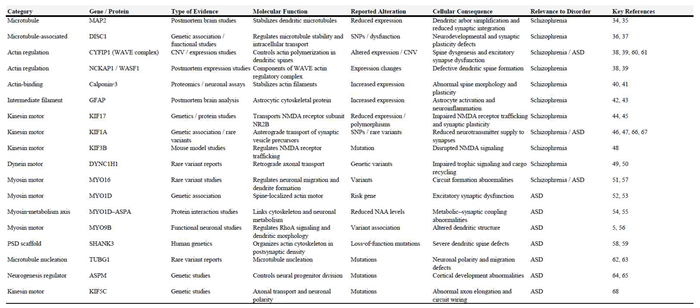 |
2. Cytoskeletal and Molecular Motor Abnormalities in Schizophrenia
3. Cytoskeletal and Molecular Motor Abnormalities in Autism Spectrum Disorder
4. Microglial Cytoskeleton, Molecular Motors, and Psychiatric Disorders
5. Conclusions and Future Directions
- Clarifying the relationships between glia-specific cytoskeletal and molecular motor gene variants and psychiatric disorders;
- Developing novel therapeutic strategies that directly target glial cytoskeletal and molecular motor pathways;
- Elucidating the roles of cytoskeletal and motor systems in neuron–glia interactions; and
- Defining how morphological changes in glial cells relate to functional alterations in the context of psychiatric illnesses.
Acknowledgments
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fletcher, DA; Mullins, RD. Cell mechanics and the cytoskeleton. Nature 2010, 463(7280), 485–92. [Google Scholar] [CrossRef]
- Hirokawa, N; Noda, Y; Tanaka, Y; Niwa, S. Kinesin superfamily motor proteins and intracellular transport. Nat Rev Mol Cell Biol. 2009, 10(10), 682–96. [Google Scholar] [CrossRef]
- Franker, MAM; Hoogenraad, CC. Microtubule-based transport - basic mechanisms, traffic rules and role in neurological pathogenesis. J Cell Sci. 2013, 126 Pt 11, 2319–29. [Google Scholar] [CrossRef] [PubMed]
- Marchisella, F. Abnormalities of the microtubule system and impaired neurite outgrowth in psychiatric disorders. In Neural Plast; 2016. [Google Scholar]
- De Rubeis, S; He, X; Goldberg, AP; Poultney, CS; Samocha, K; et al.; The DDD Study Synaptic, transcriptional and chromatin genes disrupted in autism. Nature 2014, 515(7526), 209–15. [Google Scholar] [CrossRef]
- Moghaddam, B; Javitt, D. From revolution to evolution: the glutamate hypothesis of schizophrenia and its implication for treatment. Neuropsychopharmacology 2012, 37(1), 4–15. [Google Scholar] [CrossRef] [PubMed]
- Padmanabhan, A; Lynch, CJ; Schaer, M; Menon, V. The default mode network in autism. Biol Psychiatry Cogn Neurosci Neuroimaging 2017, 2(6), 476–86. [Google Scholar] [CrossRef] [PubMed]
- Estes, ML; McAllister, AK. Maternal immune activation: Implications for neuropsychiatric disorders. Science 2016, 353(6301), 772–7. [Google Scholar] [CrossRef]
- Müller, N. Neuroinflammation in schizophrenia and autism spectrum disorders: Recent advances. In Prog Neuropsychopharmacol Biol Psychiatry; 2018. [Google Scholar]
- Hirokawa, N; Takemura, R. Molecular motors and mechanisms of directional transport in neurons. Nat Rev Neurosci. 2005, 6(3), 201–14. [Google Scholar] [CrossRef]
- Miki, H; Setou, M; Kaneshiro, K; Hirokawa, N. All kinesin superfamily protein, KIF, genes in mouse and human. Proc Natl Acad Sci U S A 2001, 98(13), 7004–11. [Google Scholar] [CrossRef]
- Conde, C; Cáceres, A. Microtubule assembly, organization and dynamics in axons and dendrites. Nat Rev Neurosci 2009, 10(5), 319–32. [Google Scholar] [CrossRef]
- Takei, Y; Teng, J; Harada, A; Hirokawa, N. Defects in axonal elongation and neuronal migration in mice with disrupted tau and map1b genes. J Cell Biol. 2000, 150(5), 989–1000. [Google Scholar] [CrossRef]
- Kapitein, LC; Hoogenraad, CC. Building the neuronal microtubule cytoskeleton. Neuron 2015, 87(3), 492–506. [Google Scholar] [CrossRef] [PubMed]
- Dent, EW; Gupton, SL; Gertler, FB. The growth cone cytoskeleton in axon outgrowth and guidance. Cold Spring Harb Perspect Biol. 2011, 3(3), a001800–a001800. [Google Scholar] [CrossRef]
- Cingolani, LA; Goda, Y. Actin in action: the interplay between the actin cytoskeleton and synaptic efficacy. Nat Rev Neurosci 2008, 9(5), 344–56. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, K-I; Nagai, T; Miyawaki, A; Hayashi, Y. Rapid and persistent modulation of actin dynamics regulates postsynaptic reorganization underlying bidirectional plasticity. Nat Neurosci. 2004, 7(10), 1104–12. [Google Scholar] [CrossRef] [PubMed]
- Perrot, R; Eyer, J. Neuronal intermediate filaments and neurodegenerative disorders. Brain Res Bull. 2009, 80(4–5), 282–95. [Google Scholar] [CrossRef]
- Lendahl, U; Zimmerman, LB; McKay, RD. CNS stem cells express a new class of intermediate filament protein. Cell 1990, 60(4), 585–95. [Google Scholar] [CrossRef]
- Hirokawa, N; Niwa, S; Tanaka, Y. Molecular motors in neurons: transport mechanisms and roles in brain function, development, and disease. Neuron 2010, 68(4), 610–38. [Google Scholar] [CrossRef]
- Mandal, A; Drerup, CM. Axonal transport and mitochondrial function in neurons. Front Cell Neurosci 2019, 13, 373. [Google Scholar] [CrossRef]
- Kanai, Y; Dohmae, N; Hirokawa, N. Kinesin transports RNA: isolation and characterization of an RNA-transporting granule. Neuron 2004, 43(4), 513–25. [Google Scholar] [CrossRef]
- Cosker, KE; Segal, RA. Neuronal signaling through endocytosis. Cold Spring Harb Perspect Biol. 2014, 6(2), a020669–a020669. [Google Scholar] [CrossRef]
- Roberts, AJ; Kon, T; Knight, PJ; Sutoh, K; Burgess, SA. Functions and mechanics of dynein motor proteins. Nat Rev Mol Cell Biol. 2013, 14(11), 713–26. [Google Scholar] [CrossRef]
- Caviston, JP; Holzbaur, ELF. Microtubule motors at the intersection of trafficking and transport. Trends Cell Biol. 2006, 16(10), 530–7. [Google Scholar] [CrossRef]
- Vallee, RB; Tsai, J-W. The cellular roles of the lissencephaly gene LIS1, and what they tell us about brain development. Genes Dev. 2006, 20(11), 1384–93. [Google Scholar] [CrossRef]
- Heerssen, HM; Pazyra, MF; Segal, RA. Dynein motors transport activated Trks to promote survival of target-dependent neurons. Nat Neurosci. 2004, 7(6), 596–604. [Google Scholar] [CrossRef]
- Tojima, T; Itofusa, R; Kamiguchi, H. Steering neuronal growth cones by shifting the imbalance between exocytosis and endocytosis. J Neurosci. 2014, 34(21), 7165–78. [Google Scholar] [CrossRef]
- Hartman, MA; Spudich, JA. The myosin superfamily at a glance. J Cell Sci. 2012, 125 Pt 7, 1627–32. [Google Scholar] [CrossRef] [PubMed]
- Rex, CS; Gavin, CF; Rubio, MD; Kramar, EA; Chen, LY; Jia, Y; et al. Myosin IIb regulates actin dynamics during synaptic plasticity and memory formation. Neuron 2010, 67(4), 603–17. [Google Scholar] [CrossRef] [PubMed]
- Kneussel, M; Wagner, W. Myosin motors at neuronal synapses: drivers of membrane transport and actin dynamics. Nat Rev Neurosci. 2013, 14(4), 233–47. [Google Scholar] [CrossRef]
- Wang, Z; Edwards, JG; Riley, N; Provance, DW, Jr.; Karcher, R; Li, X-D; et al. Myosin Vb mobilizes recycling endosomes and AMPA receptors for postsynaptic plasticity. Cell 2008, 135(3), 535–48. [Google Scholar] [CrossRef] [PubMed]
- Huber, KM; Gallagher, SM; Warren, ST; Bear, MF. Altered synaptic plasticity in a mouse model of fragile X mental retardation. Proc Natl Acad Sci U S A 2002, 99(11), 7746–50. [Google Scholar] [CrossRef]
- Jones, LB; Johnson, N; Byne, W. Alterations in MAP2 immunocytochemistry in areas 9 and 32 of schizophrenic prefrontal cortex. Psychiatry Res Neuroimaging 2002, 114(3), 137–48. [Google Scholar] [CrossRef] [PubMed]
- DeGiosio, R; Kelly, RM; DeDionisio, AM; Newman, JT; Fish, KN; Sampson, AR; et al. MAP2 immunoreactivity deficit is conserved across the cerebral cortex within individuals with schizophrenia. NPJ Schizophr 2019, 5(1), 13. [Google Scholar] [CrossRef] [PubMed]
- Brandon, NJ; Sawa, A. Linking neurodevelopmental and synaptic theories of mental illness through DISC1. Nat Rev Neurosci. 2011, 12(12), 707–22. [Google Scholar] [CrossRef]
- Lipska, BK; Peters, T; Hyde, TM; Halim, N; Horowitz, C; Mitkus, S; et al. Expression of DISC1 binding partners is reduced in schizophrenia and associated with DISC1 SNPs. Hum Mol Genet. 2006, 15(8), 1245–58. [Google Scholar] [CrossRef]
- Kähler, AK; Djurovic, S; Rimol, LM; Brown, AA; Athanasiu, L; Jönsson, EG; et al. Candidate gene analysis of the human natural killer-1 carbohydrate pathway and perineuronal nets in schizophrenia: B3GAT2 is associated with disease risk and cortical surface area. Biol Psychiatry 2011, 69(1), 90–6. [Google Scholar] [CrossRef]
- Soderling, SH; Guire, ES; Kaech, S; White, J; Zhang, F; Schutz, K; et al. A WAVE-1 and WRP signaling complex regulates spine density, synaptic plasticity, and memory. J Neurosci. 2007, 27(2), 355–65. [Google Scholar] [CrossRef]
- Föcking, M; Dicker, P; English, JA; Schubert, KO; Dunn, MJ; Cotter, DR. Common proteomic changes in the hippocampus in schizophrenia and bipolar disorder and particular evidence for involvement of cornu ammonis regions 2 and 3. Arch Gen Psychiatry 2011, 68(5), 477–88. [Google Scholar] [CrossRef] [PubMed]
- Rami, G; Caillard, O; Medina, I; Pellegrino, C; Fattoum, A; Ben-Ari, Y; et al. Change in the shape and density of dendritic spines caused by overexpression of acidic calponin in cultured hippocampal neurons. Hippocampus 2006, 16(2), 183–97. [Google Scholar] [CrossRef]
- Toro, CT; Hallak, JEC; Dunham, JS; Deakin, JFW. Glial fibrillary acidic protein and glutamine synthetase in subregions of prefrontal cortex in schizophrenia and mood disorder. Neurosci Lett. 2006, 404(3), 276–81. [Google Scholar] [CrossRef]
- Sofroniew, MV; Vinters, HV. Astrocytes: biology and pathology. Acta Neuropathol 2010, 119(1), 7–35. [Google Scholar] [CrossRef]
- Ratta-Apha, W; Mouri, K; Boku, S; Ishiguro, H; Okazaki, S; Otsuka, I; et al. A decrease in protein level and a missense polymorphism of KIF17 are associated with schizophrenia. Psychiatry Res. 2015, 230(2), 424–9. [Google Scholar] [CrossRef]
- Yin, X; Feng, X; Takei, Y; Hirokawa, N. Regulation of NMDA receptor transport: a KIF17-cargo binding/releasing underlies synaptic plasticity and memory in vivo. J Neurosci. 2012, 32(16), 5486–99. [Google Scholar] [CrossRef]
- Rivero, O; Sich, S; Popp, S; Schmitt, A; Franke, B; Lesch, K-P. Impact of the ADHD-susceptibility gene CDH13 on development and function of brain networks. Eur Neuropsychopharmacol 2013, 23(6), 492–507. [Google Scholar]
- Okada, Y; Yamazaki, H; Sekine-Aizawa, Y; Hirokawa, N. The neuron-specific kinesin superfamily protein KIF1A is a unique monomeric motor for anterograde axonal transport of synaptic vesicle precursors. Cell. 1995, 81(5), 769–80. [Google Scholar] [CrossRef] [PubMed]
- Alsabban, AH; Morikawa, M; Tanaka, Y; Takei, Y; Hirokawa, N. Kinesin Kif3b mutation reduces NMDAR subunit NR2A trafficking and causes schizophrenia-like phenotypes in mice. EMBO J 2020, 39(1), e101090. [Google Scholar] [CrossRef] [PubMed]
- Vissers, LELM; de Ligt, J; Gilissen, C; Janssen, I; Steehouwer, M; de Vries, P; et al. A de novo paradigm for mental retardation. Nat Genet. 2010, 42(12), 1109–12. [Google Scholar] [CrossRef]
- Schiavo, G; Greensmith, L; Hafezparast, M; Fisher, EMC. Cytoplasmic dynein heavy chain: the servant of many masters. Trends Neurosci. 2013, 36(11), 641–51. [Google Scholar] [CrossRef]
- Yue, W; Yu, X; Zhang, D. Progress in genome-wide association studies of schizophrenia in Han Chinese populations. NPJ Schizophr 2017, 3(1), 24. [Google Scholar] [CrossRef] [PubMed]
- Stone, JL; Merriman, B; Cantor, RM; Geschwind, DH; Nelson, SF. High density SNP association study of a major autism linkage region on chromosome 17. Hum Mol Genet. 2007, 16(6), 704–15. [Google Scholar] [CrossRef]
- Koshida, R; Tome, S; Takei, Y. Myosin Id localizes in dendritic spines through the tail homology 1 domain. Exp Cell Res. 2018, 367(1), 65–72. [Google Scholar] [CrossRef]
- Benesh, AE; Fleming, JT; Chiang, C; Carter, BD; Tyska, MJ. Expression and localization of myosin-1d in the developing nervous system. Brain Res. 2012, 1440, 9–22. [Google Scholar] [CrossRef] [PubMed]
- Moffett, JR; Ross, B; Arun, P; Madhavarao, CN; Namboodiri, AMA. N-Acetylaspartate in the CNS: from neurodiagnostics to neurobiology. Prog Neurobiol 2007, 81(2), 89–131. [Google Scholar] [CrossRef]
- Long, H; Zhu, X; Yang, P; Gao, Q; Chen, Y; Ma, L. Myo9b and RICS modulate dendritic morphology of cortical neurons. Cereb Cortex 2013, 23(1), 71–9. [Google Scholar] [CrossRef]
- Wang, K; Zhang, H; Ma, D; Bucan, M; Glessner, JT; Abrahams, BS; et al. Common genetic variants on 5p14.1 associate with autism spectrum disorders. Nature 2009, 459(7246), 528–33. [Google Scholar] [CrossRef]
- Durand, CM; Betancur, C; Boeckers, TM; Bockmann, J; Chaste, P; Fauchereau, F; et al. Mutations in the gene encoding the synaptic scaffolding protein SHANK3 are associated with autism spectrum disorders. Nat Genet. 2007, 39(1), 25–7. [Google Scholar] [CrossRef] [PubMed]
- Naisbitt, S; Kim, E; Tu, JC; Xiao, B; Sala, C; Valtschanoff, J; et al. Shank, a novel family of postsynaptic density proteins that binds to the NMDA receptor/PSD-95/GKAP complex and cortactin. Neuron 1999, 23(3), 569–82. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, Y; Martin, CL; Vazquez-Lopez, A; Spence, SJ; Alvarez-Retuerto, AI; Sigman, M; et al. Genome-wide expression profiling of lymphoblastoid cell lines distinguishes different forms of autism and reveals shared pathways. Hum Mol Genet. 2007, 16(14), 1682–98. [Google Scholar] [CrossRef]
- De Rubeis, S; Pasciuto, E; Li, KW; Fernández, E; Di Marino, D; Buzzi, A; et al. CYFIP1 coordinates mRNA translation and cytoskeleton remodeling to ensure proper dendritic spine formation. Neuron 2013, 79(6), 1169–82. [Google Scholar] [CrossRef]
- Poirier, K; Saillour, Y; Bahi-Buisson, N; Jaglin, XH; Fallet-Bianco, C; Nabbout, R; et al. Mutations in the neuronal ß-tubulin subunit TUBB3 result in malformation of cortical development and neuronal migration defects. Hum Mol Genet. 2010, 19(22), 4462–73. [Google Scholar] [CrossRef]
- Tovey, CA; Conduit, PT. Microtubule nucleation by γ-tubulin complexes and beyond. Essays Biochem. 2018, 62(6), 765–80. [Google Scholar] [CrossRef]
- Bond, J; Roberts, E; Mochida, GH; Hampshire, DJ; Scott, S; Askham, JM; et al. ASPM is a major determinant of cerebral cortical size. Nat Genet. 2002, 32(2), 316–20. [Google Scholar] [CrossRef]
- Fish, JL; Kosodo, Y; Enard, W; Pääbo, S; Huttner, WB. Aspm specifically maintains symmetric proliferative divisions of neuroepithelial cells. Proc Natl Acad Sci U S A 2006, 103(27), 10438–43. [Google Scholar] [CrossRef]
- Hamdan, FF; Gauthier, J; Araki, Y; Lin, D-T; Yoshizawa, Y; Higashi, K; et al. Excess of DE Novo deleterious mutations in genes associated with glutamatergic systems in nonsyndromic intellectual disability. Am J Hum Genet. 2011, 88(4), 516. [Google Scholar] [CrossRef]
- Yonekawa, Y; Harada, A; Okada, Y; Funakoshi, T; Kanai, Y; Takei, Y; et al. Defect in synaptic vesicle precursor transport and neuronal cell death in KIF1A motor protein-deficient mice. J Cell Biol. 1998, 141(2), 431–41. [Google Scholar] [CrossRef] [PubMed]
- Poirier, K; Lebrun, N; Broix, L; Tian, G; Saillour, Y; Boscheron, C; et al. Mutations in TUBG1, DYNC1H1, KIF5C and KIF2A cause malformations of cortical development and microcephaly. Nat Genet. 2013, 45(6), 639–47. [Google Scholar] [CrossRef]
- Rosito, M; Sanchini, C; Gosti, G; Moreno, M; De Panfilis, S; Giubettini, M; et al. Microglia reactivity entails microtubule remodeling from acentrosomal to centrosomal arrays. Cell Rep. 2023, 42(2), 112104. [Google Scholar] [CrossRef]
- Adrian, M; Weber, M; Tsai, M-C; Glock, C; Kahn, OI; Phu, L; et al. Polarized microtubule remodeling transforms the morphology of reactive microglia and drives cytokine release. Nat Commun. 2023, 14(1), 6322. [Google Scholar] [CrossRef]
- Iwata, S; Morikawa, M; Takei, Y; Hirokawa, N. An activity-dependent local transport regulation via degradation and synthesis of KIF17 underlying cognitive flexibility. Sci Adv 2020, 6(51), eabc8355. [Google Scholar] [CrossRef] [PubMed]
- Trubetskoy, V; Pardiñas, AF; Qi, T; Panagiotaropoulou, G; Awasthi, S; Bigdeli, TB; et al. Mapping genomic loci implicates genes and synaptic biology in schizophrenia. Nature 2022, 604, 502–508. [Google Scholar] [CrossRef]
- Satterstrom, FK; Kosmicki, JA; Wang, J; Breen, MS; De Rubeis, S; An, JY; et al. Large-Scale Exome Sequencing Study Implicates Both Developmental and Functional Changes in the Neurobiology of Autism. Cell. 2020, 180(3), 568–584.e23. [Google Scholar] [CrossRef] [PubMed]
- Park, GH; Noh, H; Shao, Z; Ni, P; Qin, Y; Liu, D; et al. Activated microglia cause metabolic disruptions in developmental cortical interneurons that persist in interneurons from individuals with schizophrenia. Nature Neuroscience 2020, 23, 1352–1364. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, S-M; Heider, J; Wüst, R; Fallgatter, AJ; Volkmer, H. Microglia-neuron interactions in schizophrenia. Frontiers in Cellular Neuroscience 2024, 18, 1345349. [Google Scholar] [CrossRef]
- Teter, OM; McQuade, A; Hagan, V; Liang, W; Dräger, NM; Sattler, SM; et al. CRISPRi-based screen of autism spectrum disorder risk genes in microglia uncovers roles of ADNP in microglia endocytosis and synaptic pruning. Molecular Psychiatry 2025, 30, 4176–4193. [Google Scholar] [CrossRef]
- Iwata, S; Hyugaji, M; Soga, Y; Morikawa, M; Sasaki, T; Takei, Y. Gene expression of psychiatric disorder-related kinesin superfamily proteins (Kifs) is potentiated in alternatively activated primary cultured microglia. BMC research notes 2025, 18(44), 1–8. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




