Submitted:
28 February 2026
Posted:
03 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
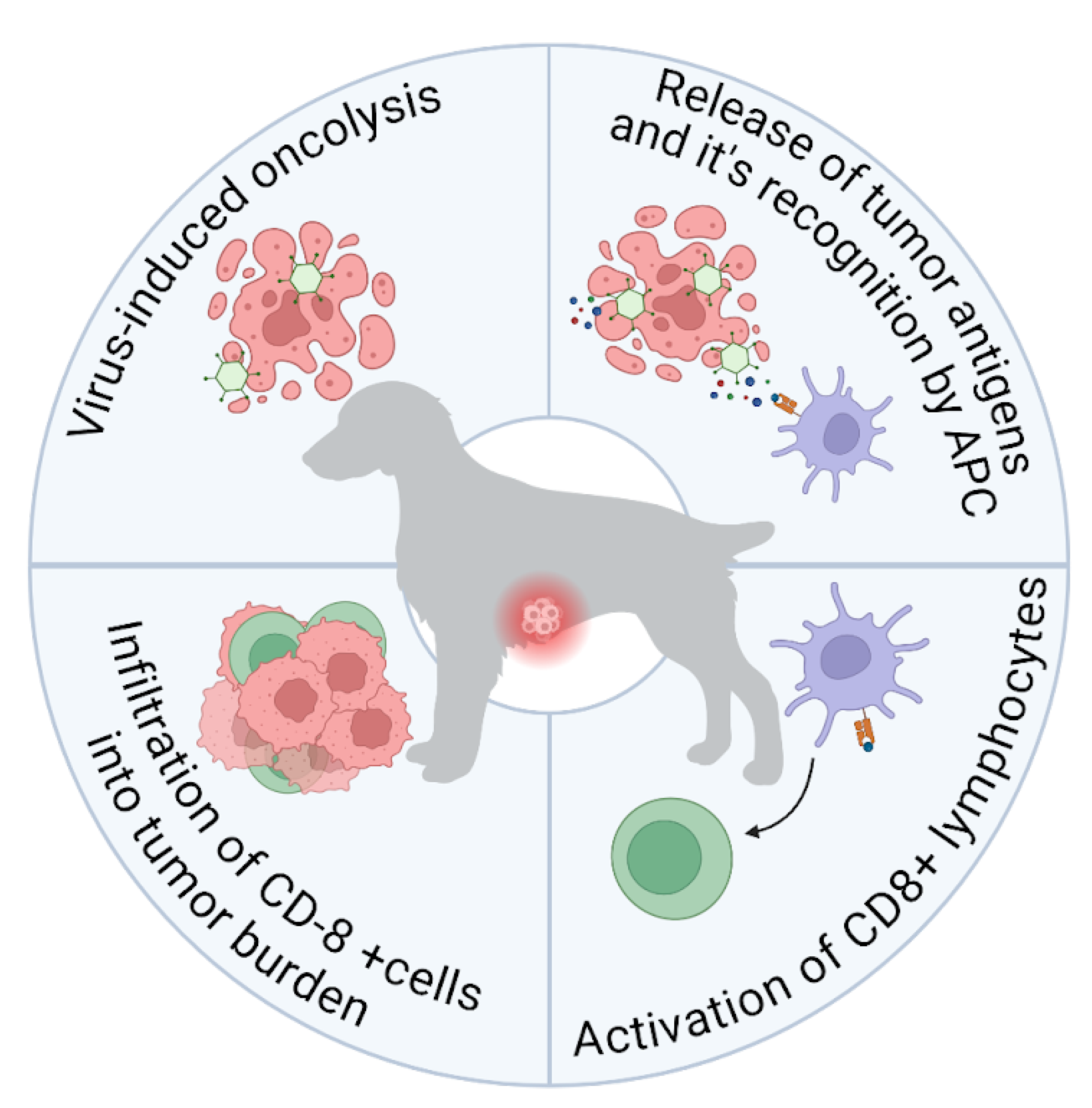
2. General Mechanisms of Virus-Mediated Oncolysis
3. Clinical Applications of OVs in Dogs
3.1. Myxoma Virus
3.2. Vaccinia virus
3.3. Reoviruses
3.4. Vesicular Stomatitis Virus
3.5. Adenoviruses
3.6. Sendai Virus
3.7. Newcastle Disease Virus
| Oncolytic Virus | Tumor Types Studied | Administration Route | Observation Period | Clinical Outcome | Adverse Events |
|---|---|---|---|---|---|
| Myxoma virus (MYXV) [21] | Soft tissue sarcoma | Intratumoral | 28-30 days after treatment | PD | Grade 1/2 |
| Reovirus (Reolysin) [46] | Various types of sarcomas and carcinomas, mastocytoma, Hodgkin's lymphoma | IV/Intratumoral | Individual | PR | Grade 1/2 |
| VSV-IFNβ-NIS [53,54,56] | Osteosarcoma, lymphomas | Intravenous | 28 days after treatment | SD | Grade 1/2 (dose-dependent) |
| Adenovirus (ICOCAV15) [72] | Carcinomas/adenocarcinomas | Intratumoral | 1 year after the treatment | PR, SD | Grade 1/2 |
| Adenovirus (ICOCAV17) [71,73] | Various types of sarcomas and gliomas, mastocytomas, schwannoma and melanoma | IV | Individual | CR, PR, SD | Grade 1/2 |
| Gliomas | |||||
| Vaccinia virus (TG6002) [29,32] | Solid tumors | Intratumoral | 38 days after the treatment | PR, SD | 3.3% grade 4 events |
| Sendai virus [81] | Mast cell tumors | Intratumoral | Until the death of the patients | SD | Grade 1/2 |
| NDV [85,86] | Breast cancer | IV | 6 days | PR, SD | Grade 1/2 |
| Cranial tumors | Intratumoral | Until the death of the patients |
4. Discussion
5. Conclusions and Perspectives
Funding
Conflicts of Interest
References
- Canine Cancer Statistics. American Veterinary Medical Association, Available at avma@org.
- Zandvliet, M.; Teske, E. Mechanisms of Drug Resistance in Veterinary Oncology- A Review with an Emphasis on Canine Lymphoma. Vet Sci 2015, 2((3)), 150–184. [Google Scholar] [CrossRef]
- Stephens, T. The Use of Chemotherapy to Prolong the Life of Dogs Suffering from Cancer: The Ethical Dilemma. Animals (Basel) 2019, 9((7)). [Google Scholar] [CrossRef] [PubMed]
- Klingemann, H. Immunotherapy for Dogs: Still Running Behind Humans. Frontiers in immunology 2021, 12, 665784. [Google Scholar] [CrossRef]
- Sinkovics, J. G.; Horvath, J. C. Natural and genetically engineered viral agents for oncolysis and gene therapy of human cancers. Arch Immunol Ther Exp 2008, 56((1)), 23. [Google Scholar] [CrossRef]
- Frampton, J. E. Teserpaturev/G47Δ: First Approval. BioDrugs 2022, 36((5)), 667–672. [Google Scholar] [CrossRef]
- Liang, M. Oncorine, the World First Oncolytic Virus Medicine and its Update in China. Curr Cancer Drug Targets 2018, 18((2)), 171–176. [Google Scholar] [CrossRef]
- Alberts, P.; Tilgase, A.; Rasa, A.; Bandere, K.; Venskus, D. The advent of oncolytic virotherapy in oncology: The Rigvir® story. Eur J Pharmacol 2018, 837, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Ferrucci, P. F.; Pala, L.; Conforti, F.; Cocorocchio, E. Talimogene Laherparepvec (T-VEC): An Intralesional Cancer Immunotherapy for Advanced Melanoma. Cancers 2021, 13((6)). [Google Scholar] [CrossRef] [PubMed]
- Andtbacka, R. H. I.; Curti, B.; Daniels, G. A.; Hallmeyer, S.; Whitman, E. D.; Lutzky, J.; Spitler, L. E.; Zhou, K.; Bommareddy, P. K.; Grose, M.; Wang, M.; Wu, C.; Kaufman, H. L. Clinical Responses of Oncolytic Coxsackievirus A21 (V937) in Patients With Unresectable Melanoma. J Clin Oncol 2021, 39((34)), 3829–3838. [Google Scholar] [CrossRef] [PubMed]
- Lin, D.; Shen, Y.; Liang, T. Oncolytic virotherapy: basic principles, recent advances and future directions. Signal Transduction and Targeted Therapy 2023, 8((1)), 156. [Google Scholar] [CrossRef]
- Matveeva, O. V.; Chumakov, P. M. Defects in interferon pathways as potential biomarkers of sensitivity to oncolytic viruses. Rev Med Virol 2018, e2008. [Google Scholar] [CrossRef]
- Kooti, W.; Esmaeili Gouvarchin Ghaleh, H.; Farzanehpour, M.; Dorostkar, R.; Jalali Kondori, B.; Bolandian, M. Oncolytic Viruses and Cancer, Do You Know the Main Mechanism? Frontiers in oncology 2021, 11, 761015. [Google Scholar] [CrossRef]
- Bai, Y.; Hui, P.; Du, X.; Su, X. Updates to the antitumor mechanism of oncolytic virus. Thoracic cancer 2019. [Google Scholar] [CrossRef] [PubMed]
- Vitiello, G. A. F.; Ferreira, W. A. S.; Cordeiro de Lima, V. C.; Medina, T. D. S. Antiviral Responses in Cancer: Boosting Antitumor Immunity Through Activation of Interferon Pathway in the Tumor Microenvironment. Frontiers in immunology 2021, 12, 782852. [Google Scholar] [CrossRef] [PubMed]
- Spiesschaert, B.; McFadden, G.; Hermans, K.; Nauwynck, H.; Van de Walle, G. R. The current status and future directions of myxoma virus, a master in immune evasion. Vet Res 2011, 42((1)), 76. [Google Scholar] [CrossRef] [PubMed]
- Enow, J. A.; Lopes, A. M.; Abrantes, J.; Esteves, P. J.; Rahman, M. M. The Role of Myxoma Virus Immune Modulators and Host Range Factors in Pathogenesis and Species Leaping. Viruses 2025, 17((8)). [Google Scholar] [CrossRef]
- Schin, A. M.; Diesterbeck, U. S.; Moss, B. Insights into the Organization of the Poxvirus Multicomponent Entry-Fusion Complex from Proximity Analyses in Living Infected Cells. J Virol 2021, 95((16)), e0085221. [Google Scholar] [CrossRef]
- Shaban, M. Myxoma Virus Oncolytic Activity in a Murine Melanoma Model: An in Vitro to in Vivo Study 2006, 262.
- Kellish, P.; Shabashvili, D.; Rahman, M. M.; Nawab, A.; Guijarro, M. V.; Zhang, M.; Cao, C.; Moussatche, N.; Boyle, T.; Antonia, S.; Reinhard, M.; Hartzell, C.; Jantz, M.; Mehta, H. J.; McFadden, G.; Kaye, F. J.; Zajac-Kaye, M. Oncolytic virotherapy for small-cell lung cancer induces immune infiltration and prolongs survival. J Clin Invest 2019, 129((6)), 2279–2292. [Google Scholar] [CrossRef]
- MacNeill, A. L.; Weishaar, K. M.; Seguin, B.; Powers, B. E. Safety of an Oncolytic Myxoma Virus in Dogs with Soft Tissue Sarcoma. Viruses 2018, 10((8)). [Google Scholar] [CrossRef]
- Messud-Petit, F.; Gelfi, J.; Delverdier, M.; Amardeilh, M. F.; Py, R.; Sutter, G.; Bertagnoli, S. Serp2, an inhibitor of the interleukin-1beta-converting enzyme, is critical in the pathobiology of myxoma virus. J Virol 1998, 72((10)), 7830–9. [Google Scholar] [CrossRef] [PubMed]
- Ashton, L. V.; Weishaar, K. M.; Séguin, B.; MacNeill, A. L. Oclacitinib and Myxoma Virus Therapy in Dogs with High-Grade Soft Tissue Sarcoma. Biomedicines 2023, 11, 9. [Google Scholar] [CrossRef]
- Jacobs, B. L.; Langland, J. O.; Kibler, K. V.; Denzler, K. L.; White, S. D.; Holechek, S. A.; Wong, S.; Huynh, T.; Baskin, C. R. Vaccinia virus vaccines: past, present and future. Antiviral Res 2009, 84((1)), 1–13. [Google Scholar] [CrossRef]
- Cloquell, A.; Mateo, I.; Gambera, S.; Pumarola, M.; Alemany, R.; García-Castro, J.; Perisé-Barrios, A. J. Systemic cellular viroimmunotherapy for canine high-grade gliomas. Journal for immunotherapy of cancer 2022, 10, 12. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Zhang, M.; Ye, Q.; Liu, Y.; Qian, W. Preclinical and clinical trials of oncolytic vaccinia virus in cancer immunotherapy: a comprehensive review. Cancer Biol Med 2023, 20((9)), 646–661. [Google Scholar] [CrossRef]
- Stawowczyk, M.; Ye, Y.; Chen, N. G. Vaccinia Virus-A Swiss Army Knife Against Cancer. Cancers 2025, 17, 14. [Google Scholar] [CrossRef] [PubMed]
- MacLeod, D. T.; Nakatsuji, T.; Wang, Z.; di Nardo, A.; Gallo, R. L. Vaccinia virus binds to the scavenger receptor MARCO on the surface of keratinocytes. J Invest Dermatol 2015, 135((1)), 142–150. [Google Scholar] [CrossRef]
- Béguin, J.; Laloy, E.; Cochin, S.; Gantzer, M.; Farine, I.; Pichon, C.; Moreau, B.; Foloppe, J.; Balloul, J. M.; Machon, C.; Guitton, J.; Tierny, D.; Klonjkowski, B.; Quéméneur, E.; Maurey, C.; Erbs, P. Oncolytic virotherapy with intratumoral injection of vaccinia virus TG6002 and 5-fluorocytosine administration in dogs with malignant tumors. Molecular therapy oncolytics 2023, 30, 103–116. [Google Scholar] [CrossRef]
- Béguin, J.; Foloppe, J.; Maurey, C.; Laloy, E.; Hortelano, J.; Nourtier, V.; Pichon, C.; Cochin, S.; Cordier, P.; Huet, H.; Quemeneur, E.; Klonjkowski, B.; Erbs, P. Preclinical Evaluation of the Oncolytic Vaccinia Virus TG6002 by Translational Research on Canine Breast Cancer. Molecular therapy oncolytics 2020, 19, 57–66. [Google Scholar] [CrossRef]
- Foloppe, J.; Kempf, J.; Futin, N.; Kintz, J.; Cordier, P.; Pichon, C.; Findeli, A.; Vorburger, F.; Quemeneur, E.; Erbs, P. The Enhanced Tumor Specificity of TG6002, an Armed Oncolytic Vaccinia Virus Deleted in Two Genes Involved in Nucleotide Metabolism. Molecular therapy oncolytics 2019, 14, 1–14. [Google Scholar] [CrossRef]
- Béguin, J.; Nourtier, V.; Gantzer, M.; Cochin, S.; Foloppe, J.; Balloul, J. M.; Laloy, E.; Tierny, D.; Klonjkowski, B.; Quemeneur, E.; Maurey, C.; Erbs, P. Safety studies and viral shedding of intramuscular administration of oncolytic vaccinia virus TG6002 in healthy beagle dogs. BMC veterinary research 2020, 16((1)), 307. [Google Scholar] [CrossRef]
- Eledge, M. R.; Zita, M. D.; Boehme, K. W. Reovirus: Friend and Foe. Current clinical microbiology reports 2019, 6((3)), 132–138. [Google Scholar] [CrossRef]
- Lawson, K. A.; Mostafa, A. A.; Shi, Z. Q.; Spurrell, J.; Chen, W.; Kawakami, J.; Gratton, K.; Thakur, S.; Morris, D. G. Repurposing Sunitinib with Oncolytic Reovirus as a Novel Immunotherapeutic Strategy for Renal Cell Carcinoma. Clin Cancer Res 2016, 22((23)), 5839–5850. [Google Scholar] [CrossRef]
- Hwang, C. C.; Mochizuki, M.; Maeda, K.; Okuda, M.; Mizuno, T. Seroepidemiology of reovirus in healthy dogs in six prefectures in Japan. The Journal of veterinary medical science / the Japanese Society of Veterinary Science 2014, 76((3)), 471–5. [Google Scholar] [CrossRef]
- Barton, E. S.; Forrest, J. C.; Connolly, J. L.; Chappell, J. D.; Liu, Y.; Schnell, F. J.; Nusrat, A.; Parkos, C. A.; Dermody, T. S. Junction adhesion molecule is a receptor for reovirus. Cell 2001, 104((3)), 441–51. [Google Scholar] [CrossRef] [PubMed]
- Campbell, J. A.; Schelling, P.; Wetzel, J. D.; Johnson, E. M.; Forrest, J. C.; Wilson, G. A.; Aurrand-Lions, M.; Imhof, B. A.; Stehle, T.; Dermody, T. S. Junctional adhesion molecule a serves as a receptor for prototype and field-isolate strains of mammalian reovirus. J Virol 2005, 79((13)), 7967–78. [Google Scholar] [CrossRef] [PubMed]
- Mandell, K. J.; Parkos, C. A. The JAM family of proteins. Adv Drug Deliv Rev 2005, 57((6)), 857–67. [Google Scholar] [CrossRef]
- Bazzoni, G.; Tonetti, P.; Manzi, L.; Cera, M. R.; Balconi, G.; Dejana, E. Expression of junctional adhesion molecule-A prevents spontaneous and random motility. Journal of cell science 2005, 118 (Pt 3), 623–32. [Google Scholar] [CrossRef]
- Barton, E. S.; Connolly, J. L.; Forrest, J. C.; Chappell, J. D.; Dermody, T. S. Utilization of sialic acid as a coreceptor enhances reovirus attachment by multistep adhesion strengthening. J Biol Chem 2001, 276((3)), 2200–11. [Google Scholar] [CrossRef]
- Thompson, B. Oncolytics Biotech(®) Inc.: REOLYSIN(®) for melanoma therapy. Melanoma management 2015, 2((2)), 105–107. [Google Scholar] [CrossRef] [PubMed]
- Hwang, C. C.; Umeki, S.; Kubo, M.; Hayashi, T.; Shimoda, H.; Mochizuki, M.; Maeda, K.; Baba, K.; Hiraoka, H.; Coffey, M.; Okuda, M.; Mizuno, T. Oncolytic reovirus in canine mast cell tumor. PLoS One 2013, 8((9)), e73555. [Google Scholar] [CrossRef]
- Igase, M.; Hwang, C. C.; Kambayashi, S.; Kubo, M.; Coffey, M.; Miyama, T. S.; Baba, K.; Okuda, M.; Noguchi, S.; Mizuno, T. Oncolytic reovirus synergizes with chemotherapeutic agents to promote cell death in canine mammary gland tumor. Canadian journal of veterinary research = Revue canadienne de recherche veterinaire 2016, 80((1)), 21–31. [Google Scholar] [PubMed]
- Hwang, C. C.; Umeki, S.; Igase, M.; Coffey, M.; Noguchi, S.; Okuda, M.; Mizuno, T. The effects of oncolytic reovirus in canine lymphoma cell lines. Vet Comp Oncol 2016, 14 Suppl 1, 61–73. [Google Scholar] [CrossRef]
- Igase, M.; Hwang, C. C.; Coffey, M.; Okuda, M.; Noguchi, S.; Mizuno, T. The oncolytic effects of reovirus in canine solid tumor cell lines. The Journal of veterinary medical science / the Japanese Society of Veterinary Science 2015. [Google Scholar] [CrossRef] [PubMed]
- Hwang, C. C.; Igase, M.; Sakurai, M.; Haraguchi, T.; Tani, K.; Itamoto, K.; Shimokawa, T.; Nakaichi, M.; Nemoto, Y.; Noguchi, S.; Coffey, M.; Okuda, M.; Mizuno, T. Oncolytic reovirus therapy: Pilot study in dogs with spontaneously occurring tumours. Vet Comp Oncol 2018, 16((2)), 229–238. [Google Scholar] [CrossRef]
- Igase, M.; Shibutani, S.; Kurogouchi, Y.; Fujiki, N.; Hwang, C. C.; Coffey, M.; Noguchi, S.; Nemoto, Y.; Mizuno, T. Combination Therapy with Reovirus and ATM Inhibitor Enhances Cell Death and Virus Replication in Canine Melanoma. Molecular therapy oncolytics 2019, 15, 49–59. [Google Scholar] [CrossRef]
- Ahmed, M. M.; Okesanya, O. J.; Ukoaka, B. M.; Ibrahim, A. M.; Lucero-Prisno, D. E., 3rd. Vesicular Stomatitis Virus: Insights into Pathogenesis, Immune Evasion, and Technological Innovations in Oncolytic and Vaccine Development. Viruses 2024, 16, 12. [Google Scholar] [CrossRef]
- Johnson, K. M.; Vogel, J. E.; Peralta, P. H. Clinical and serological response to laboratory-acquired human infection by Indiana type vesicular stomatitis virus (VSV). Am J Trop Med Hyg 1966, 15((2)), 244–246. [Google Scholar] [CrossRef]
- Hastie, E.; Cataldi, M.; Marriott, I.; Grdzelishvili, V. Z. Understanding and altering cell tropism of vesicular stomatitis virus. Virus Res 2013, 176((1-2)), 16–32. [Google Scholar] [CrossRef] [PubMed]
- Felt, S. A.; Grdzelishvili, V. Z. Recent advances in vesicular stomatitis virus-based oncolytic virotherapy: a 5-year update. J Gen Virol 2017. [Google Scholar] [CrossRef]
- Bennani, N. N.; Cook, J.; Geyer, S. M.; Ginos, B. F.; Zhang, L.; Munoz, J.; Broski, S.; Russell, S.; Peng, K.-W.; Lacy, M. Phase I Trial of Systemic Administration of Vesicular Stomatitis Virus Genetically Engineered to Express NIS and Human Interferon, in Patients with Relapsed or Refractory Multiple Myeloma, Lymphomas, or Histiocytic/Dendritic Cell Neoplasms. Blood 2023, 142, 5004. [Google Scholar] [CrossRef]
- LeBlanc, A. K.; Naik, S.; Galyon, G. D.; Jenks, N.; Steele, M.; Peng, K. W.; Federspiel, M. J.; Donnell, R.; Russell, S. J. Safety studies on intravenous administration of oncolytic recombinant vesicular stomatitis virus in purpose-bred beagle dogs. Human gene therapy. Clinical development 2013, 24((4)), 174–81. [Google Scholar] [CrossRef]
- Naik, S.; Galyon, G. D.; Jenks, N. J.; Steele, M. B.; Miller, A. C.; Allstadt, S. D.; Suksanpaisan, L.; Peng, K. W.; Federspiel, M. J.; Russell, S. J.; LeBlanc, A. K. Comparative Oncology Evaluation of Intravenous Recombinant Oncolytic Vesicular Stomatitis Virus Therapy in Spontaneous Canine Cancer. Molecular cancer therapeutics 2018, 17((1)), 316–326. [Google Scholar] [CrossRef]
- LeBlanc, A. K.; Atherton, M.; Bentley, R. T.; Boudreau, C. E.; Burton, J. H.; Curran, K. M.; Dow, S.; Giuffrida, M. A.; Kellihan, H. B.; Mason, N. J.; Oblak, M.; Selmic, L. E.; Selting, K. A.; Singh, A.; Tjostheim, S.; Vail, D. M.; Weishaar, K. M.; Berger, E. P.; Rossmeisl, J. H.; Mazcko, C. Veterinary Cooperative Oncology Group-Common Terminology Criteria for Adverse Events (VCOG-CTCAE v2) following investigational therapy in dogs and cats. Vet Comp Oncol 2021, 19((2)), 311–352. [Google Scholar] [CrossRef] [PubMed]
- Makielski, K. M.; Sarver, A. L.; Henson, M. S.; Stuebner, K. M.; Borgatti, A.; Suksanpaisan, L.; Preusser, C.; Tabaran, A.-F.; Cornax, I.; O'Sullivan, M. G.; Chehadeh, A.; Groschen, D.; Bergsrud, K.; Pracht, S.; Winter, A.; Mills, L. J.; Schwabenlander, M. D.; Wolfe, M.; Farrar, M. A.; Cutter, G. R.; Koopmeiners, J. S.; Russell, S. J.; Modiano, J. F.; Naik, S. Neoadjuvant systemic oncolytic vesicular stomatitis virus is safe and may enhance long-term survivorship in dogs with naturally occurring osteosarcoma. Molecular therapy oncolytics 2023, 31, 100736. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Liu, Z.; Li, L.; Wu, J.; Zhang, H.; Zhang, H.; Lei, T.; Xu, B. Oncolytic Adenovirus: Prospects for Cancer Immunotherapy. Front Microbiol 2021, 12, 707290. [Google Scholar] [CrossRef] [PubMed]
- Ramamoorthy, S.; Garg, S.; Mishra, B.; Radotra, B. D.; Saikia, U. N. Coxsackievirus and Adenovirus Receptor (CAR) Expression in Autopsy Tissues: Organ-Specific Patterns and Clinical Significance. Cureus 2023, 15((4)), e37138. [Google Scholar] [CrossRef]
- Zeimet, A. G.; Müller-Holzner, E.; Schuler, A.; Hartung, G.; Berger, J.; Hermann, M.; Widschwendter, M.; Bergelson, J. M.; Marth, C. Determination of molecules regulating gene delivery using adenoviral vectors in ovarian carcinomas. Gene Ther 2002, 9((16)), 1093–100. [Google Scholar] [CrossRef]
- Hasenburg, A.; Fischer, D. C.; Tong, X. W.; Rojas-Martinez, A.; Kaufman, R. H.; Ramzy, I.; Kohlberger, P.; Orlowska-Volk, M.; Aguilar-Cordova, E.; Kieback, D. G. Adenovirus-mediated thymidine kinase gene therapy for recurrent ovarian cancer: expression of coxsackie-adenovirus receptor and integrins alphavbeta3 and alphavbeta5. J Soc Gynecol Investig 2002, 9((3)), 174–80. [Google Scholar] [CrossRef] [PubMed]
- Martin, T. A.; Watkins, G.; Jiang, W. G. The Coxsackie-adenovirus receptor has elevated expression in human breast cancer. Clinical and experimental medicine 2005, 5((3)), 122–8. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, S.; Bao, Y.; Ni, C.; Guan, N.; Zhao, J.; Salford, L. G.; Widegren, B.; Fan, X. Coxsackievirus and adenovirus receptor expression in non-malignant lung tissues and clinical lung cancers. J Mol Histol 2006, 37((3-4)), 153–60. [Google Scholar] [CrossRef]
- Qin, M.; Escuadro, B.; Dohadwala, M.; Sharma, S.; Batra, R. K. A novel role for the coxsackie adenovirus receptor in mediating tumor formation by lung cancer cells. Cancer Res 2004, 64((18)), 6377–80. [Google Scholar] [CrossRef] [PubMed]
- Pandha, H. S.; Stockwin, L. H.; Eaton, J.; Clarke, I. A.; Dalgleish, A. G.; Todryk, S. M.; Blair, G. E. Coxsackie B and adenovirus receptor, integrin and major histocompatibility complex class I expression in human prostate cancer cell lines: implications for gene therapy strategies. Prostate Cancer Prostatic Dis 2003, 6((1)), 6–11. [Google Scholar] [CrossRef] [PubMed]
- Uotani, K.; Tazawa, H.; Hasei, J.; Fujiwara, T.; Yoshida, A.; Yamakawa, Y.; Omori, T.; Sugiu, K.; Komatsubara, T.; Kondo, H.; Morita, T.; Kiyono, M.; Yokoo, S.; Hata, T.; Kunisada, T.; Takeda, K.; Urata, Y.; Fujiwara, T.; Ozaki, T. Fluorescence-guided assessment of bone and soft-tissue sarcomas for predicting the efficacy of telomerase-specific oncolytic adenovirus. PLoS One 2024, 19((2)), e0298292. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Tai, P. W. L.; Gao, G. Adeno-associated virus vector as a platform for gene therapy delivery. Nat Rev Drug Discov 2019, 18((5)), 358–378. [Google Scholar] [CrossRef]
- Soudais, C.; Boutin, S.; Hong, S. S.; Chillon, M.; Danos, O.; Bergelson, J. M.; Boulanger, P.; Kremer, E. J. Canine adenovirus type 2 attachment and internalization: coxsackievirus-adenovirus receptor, alternative receptors, and an RGD-independent pathway. J Virol 2000, 74((22)), 10639–49. [Google Scholar] [CrossRef]
- Zhu, Y.; Xu, J.; Lian, S.; Zhang, R.; Hou, J.; Wang, M.; Yan, X. Difference Analysis Between Canine Adenovirus Types 1 And 2. Front Cell Infect Microbiol 2022, 12, 854876. [Google Scholar] [CrossRef]
- Bass, E. P.; Gill, M. A.; Beckenhauer, W. H. Evaluation of a canine adenovirus type 2 strain as a replacement for infectious canine hepatitis vaccine. J Am Vet Med Assoc 1980, 177((3)), 234–242. [Google Scholar] [CrossRef]
- Olson, P.; Klingeborn, B.; Hedhammar, A. Serum antibody response to canine parvovirus, canine adenovirus-1, and canine distemper virus in dogs with known status of immunization: study of dogs in Sweden. Am J Vet Res 1988, 49((9)), 1460–1466. [Google Scholar] [CrossRef]
- Laborda, E.; Puig-Saus, C.; Rodriguez-García, A.; Moreno, R.; Cascalló, M.; Pastor, J.; Alemany, R. A pRb-responsive, RGD-modified, and hyaluronidase-armed canine oncolytic adenovirus for application in veterinary oncology. Mol Ther 2014, 22((5)), 986–98. [Google Scholar] [CrossRef]
- Delgado-Bonet, P.; Tomeo-Martín, B. D.; Delgado-Bonet, B.; Sardón-Ruiz, D.; Torrado-Carvajal, A.; Mateo, I.; Perisé-Barrios, A. J. Intracranial Virotherapy for a Canine Hemangioma. Int J Mol Sci 2022, 23, 19. [Google Scholar] [CrossRef]
- Cejalvo, T.; Perisé-Barrios, A. J.; Del Portillo, I.; Laborda, E.; Rodriguez-Milla, M. A.; Cubillo, I.; Vázquez, F.; Sardón, D.; Ramirez, M.; Alemany, R.; Del Castillo, N.; García-Castro, J. Remission of Spontaneous Canine Tumors after Systemic Cellular Viroimmunotherapy. Cancer Res 2018, 78((17)), 4891–4901. [Google Scholar] [CrossRef] [PubMed]
- Inoue, H.; Iga, M.; Nabeta, H.; Yokoo, T.; Suehiro, Y.; Okano, S.; Inoue, M.; Kinoh, H.; Katagiri, T.; Takayama, K.; Yonemitsu, Y.; Hasegawa, M.; Nakamura, Y.; Nakanishi, Y.; Tani, K. Non-transmissible Sendai virus encoding granulocyte macrophage colony-stimulating factor is a novel and potent vector system for producing autologous tumor vaccines. Cancer Sci 2008, 99((11)), 2315–26. [Google Scholar] [CrossRef]
- Matveeva, O. V.; Guo, Z. S.; Shabalina, S. A.; Chumakov, P. M. Oncolysis by paramyxoviruses: multiple mechanisms contribute to therapeutic efficiency. Molecular therapy oncolytics 2015, 2, 15011. [Google Scholar] [CrossRef]
- Matveeva, O. V.; Shabalina, S. A. Prospects for Using Expression Patterns of Paramyxovirus Receptors as Biomarkers for Oncolytic Virotherapy. Cancers 2020, 12, 12. [Google Scholar] [CrossRef]
- Zimmermann, M.; Armeanu-Ebinger, S.; Bossow, S.; Lampe, J.; Smirnow, I.; Schenk, A.; Lange, S.; Weiss, T. S.; Neubert, W.; Lauer, U. M.; Bitzer, M. Attenuated and protease-profile modified sendai virus vectors as a new tool for virotherapy of solid tumors. PLoS One 2014, 9((3)), e90508. [Google Scholar] [CrossRef]
- Martin, C. E.; List, K. Cell surface-anchored serine proteases in cancer progression and metastasis. Cancer metastasis reviews 2019, 38((3)), 357–387. [Google Scholar] [CrossRef]
- Fujita, K.; Nakai, Y.; Kawashima, A.; Ujike, T.; Nagahara, A.; Nakajima, T.; Inoue, T.; Lee, C. M.; Uemura, M.; Miyagawa, Y.; Kaneda, Y.; Nonomura, N. Phase I/II clinical trial to assess safety and efficacy of intratumoral and subcutaneous injection of HVJ-E in castration-resistant prostate cancer patients. Cancer Gene Ther 2017, 24((7)), 277–281. [Google Scholar] [CrossRef]
- Sakura, K.; Kuroyama, M.; Shintani, Y.; Funaki, S.; Atagi, S.; Kadota, Y.; Kuribayashi, K.; Kijima, T.; Nakano, T.; Nakajima, T.; Sasai, M.; Okumura, M.; Kaneda, Y. Dose-escalation, tolerability, and efficacy of intratumoral and subcutaneous injection of hemagglutinating virus of Japan envelope (HVJ-E) against chemotherapy-resistant malignant pleural mesothelioma: a clinical trial. Cancer immunology, immunotherapy: CII 2024, 73((12)), 243. [Google Scholar] [CrossRef] [PubMed]
- Ilyinskaya, G. V.; Mukhina, E. V.; Soboleva, A. V.; Matveeva, O. V.; Chumakov, P. M. Oncolytic Sendai Virus Therapy of Canine Mast Cell Tumors (A Pilot Study). Front Vet Sci 2018, 5, 116. [Google Scholar] [CrossRef] [PubMed]
- Ganar, K.; Das, M.; Sinha, S.; Kumar, S. Newcastle disease virus: current status and our understanding. Virus Res 2014, 184, 71–81. [Google Scholar] [CrossRef]
- Tayeb, S.; Zakay-Rones, Z.; Panet, A. Therapeutic potential of oncolytic Newcastle disease virus: a critical review. Oncolytic virotherapy 2015, 4, 49–62. [Google Scholar]
- Schirrmacher, V. Fifty Years of Clinical Application of Newcastle Disease Virus: Time to Celebrate! Biomedicines 2016, 4((3)), 16. [Google Scholar] [CrossRef]
- Sánchez, D.; Cesarman-Maus, G.; Romero, L.; Sánchez-Verin, R.; Vail, D.; Guadarrama, M.; Pelayo, R.; Sarmiento-Silva, R. E.; Lizano, M. The NDV-MLS as an Immunotherapeutic Strategy for Breast Cancer: Proof of Concept in Female Companion Dogs with Spontaneous Mammary Cancer. Viruses 2024, 16((3)). [Google Scholar] [CrossRef] [PubMed]
- Rossmeisl, J. H.; King, J. N.; Robertson, J. L.; Weger-Lucarelli, J.; Elankumaran, S. Phase I/II Trial of Urokinase Plasminogen Activator-Targeted Oncolytic Newcastle Disease Virus for Canine Intracranial Tumors. Cancers 2024, 16((3)). [Google Scholar] [CrossRef]
- Nguyen, S. M.; Thamm, D. H.; Vail, D. M.; London, C. A. Response evaluation criteria for solid tumours in dogs (v1.0): a Veterinary Cooperative Oncology Group (VCOG) consensus document. Vet Comp Oncol 2015, 13((3)), 176–83. [Google Scholar] [CrossRef] [PubMed]
- Oh, J. H.; Cho, J. Y. Comparative oncology: overcoming human cancer through companion animal studies. Experimental & molecular medicine 2023, 55((4)), 725–734. [Google Scholar]
- Panda, A.; Huang, Z.; Elankumaran, S.; Rockemann, D. D.; Samal, S. K. Role of fusion protein cleavage site in the virulence of Newcastle disease virus. Microbial pathogenesis 2004, 36((1)), 1–10. [Google Scholar] [CrossRef]
- Schiffman, J. D.; Breen, M. Comparative oncology: what dogs and other species can teach us about humans with cancer. Philos Trans R Soc Lond B Biol Sci 2015, 370, 1673. [Google Scholar] [CrossRef]
- Rowell, J. L.; McCarthy, D. O.; Alvarez, C. E. Dog models of naturally occurring cancer. Trends in molecular medicine 2011, 17((7)), 380–8. [Google Scholar] [CrossRef]
- Sarkisova, V. A.; Dalina, A. A.; Neymysheva, D. O.; Zenov, M. A.; Ilyinskaya, G. V.; Chumakov, P. M. Cell Carriers for Oncolytic Virus Delivery: Prospects for Systemic Administration. Cancers 2025, 17, 14. [Google Scholar] [CrossRef]
- Park, J. S.; Lee, M. E.; Kim, J.; Oh, K.; Lee, N.; Jung, M.; Jang, W. S.; Ham, W. S. PD-1 inhibitor plus oncolytic vaccinia virus is a safe and effective treatment option for metastatic renal cell carcinoma. Cancer Cell Int 2024, 24((1)), 50. [Google Scholar] [CrossRef]
- Tran, T. Q.; Grein, J.; Selman, M.; Annamalai, L.; Yearley, J. H.; Blumenschein, W. M.; Sadekova, S.; Chackerian, A. A.; Phan, U.; Wong, J. C. Oncolytic virus V937 in combination with PD-1 blockade therapy to target immunologically quiescent liver and colorectal cancer. Mol Ther Oncol 2024, 32((2)), 200807. [Google Scholar] [CrossRef] [PubMed]
- Chen, C. Y.; Hutzen, B.; Wedekind, M. F.; Cripe, T. P. Oncolytic virus and PD-1/PD-L1 blockade combination therapy. Oncolytic virotherapy 2018, 7, 65–77. [Google Scholar] [CrossRef] [PubMed]
- Jhala, D.; Nathani, N.; Joshi, M.; Patel, A.; Joshi, C. G. Oncolytic Activity of Canine Distemper Virus in Human Ductal Breast Carcinoma Cells. Oncology research and treatment 2024, 47((1-2)), 10–17. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Wang, J.; Chen, G.; Zhang, X.; Lin, D.; Zhou, Y.; Yu, Y.; Liu, W.; Zhang, D. Oncolytic activity of canine distemper virus in canine mammary tubular adenocarcinoma cells. Vet Comp Oncol 2019, 17((2)), 174–183. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





