1. Introduction
Global food systems face increasing pressure from population growth, environmental degradation, and rising demand for animal-source protein [
1,
2]. Innovative, sustainable protein sources are therefore sought to meet market demand [
3,
4]. In eastern and southern Africa, edible insects such as longhorn grasshoppers (
Ruspolia differens), winged termites (
Macrotermes spp.) and a variety of caterpillars remain an integral part of traditional food systems, local economies and cultural heritage especially in regions facing high poverty and malnutrition rates [
5,
6,
7]. Tanzania supports rich entomophagy traditions across multiple ecological zones, yet rigorous, countrywide documentation of indigenous harvesting, processing and preservation practices is missing. To the best of our knowledge, there has been no evidence-based georeferenced ethnographic study documented on edible insects, indigenous practices, processing and safety in Tanzania.
Processing and preservation are central for both food safety and marketability. Most edible insects consumed in Africa are still wild harvested, exposing them to seasonal limitations as well as microbial and environmental contaminants [
8,
9]. Gender role division within edible insect value chain exists across many Africa contexts where men dominate commercial harvesting and whole selling activities while women and children traditionally participate in collection, processing and preservation [
7,
10,
11]. Indigenous methods such as boiling, toasting, smoking, sun drying and frying are used across Tanzania, but their effectiveness in extending shelf life, preserving sensory attributes, and preventing microbial hazards has not been compared or documented. Similarly, technology adoption (e.g., solar drying, controlled smoking, HACCP based hygiene protocols, or value from adding into flours and pastes) is poorly characterized. Likewise, constraints faced by harvesters, gendered roles, suboptimal processing, knowledge transmission paths and local indicators of spoilage remain under-documented, limiting interventions that respect cultural values while meeting food-safety standards [
12]. As edible insects gain traction as a sustainable protein source worldwide, leveraging traditional practices alongside HACCP-aligned protocols offers a unique pathway to ensure microbial safety, enhance shelf life, and support commercialization in emerging markets.
Integrating indigenous knowledge systems with internationally recognized food safety standards is not only critical for local food security but also for shaping resilient, culturally inclusive global food systems. As edible insects gain traction as a sustainable protein source worldwide, leveraging traditional practices alongside HACCP-aligned protocols offers a unique pathway to ensure microbial safety, enhance shelf-life, and support commercialization in emerging markets [
13,
14]. This approach bridges cultural heritage with modern science, enabling scalable insect value chains that meet international trade requirements while preserving socio-cultural integrity (FAO, 2021).
This study addresses these gaps through a geo-referenced, evidence-based ethnography that integrates experimental microbial safety assessment with semi-structured interviews, participant observation, and enterprise surveys across nine regions of Tanzania. Using a mixed methods approach, the study aims to understand how edible insects are processed, preserved, and stored in Tanzania, what risks and benefits people associate with these practices, and whether these practices are microbiologically safe. By linking spatial, occurrence data with socio-cultural practices, post-harvest handling, and microbial safety outcomes, the study provides a critical evidence base for integrating traditional knowledge with food science innovations in emerging edible insect value chains.
2. Materials and Methods
2.1. Study Design and Sites
A mixed-methods, ethnographic study was conducted between July 2023 and March 2025 across nine regions representing seven major ecological and cultural zones namely, Coastal Zone, Arid, Semi-arid, Plateau, Northern Highlands Zone, Southern and Western Highlands Zone, Alluvial Zone in Tanzania. Sites selection was based on pre-surveyed insect consumption information and active harvesting traditions.
2.2. Sampling and Participants
A total of 149 participants were recruited using initial random community entry followed by purposive snowball sampling to identify individuals with experiential knowledge of edible insects. Initial participants were identified through local government leaders, village chairpersons, and market authorities in regions known for edible insect consumption. These facilitated introductions to knowledgeable individuals (e.g., elders, harvesters, traders, and processors).
Subsequently, participants referred the research team to other individuals recognized within the community for their expertise in harvesting, processing, or traditional knowledge of edible insects. This referral process was particularly appropriate because indigenous knowledge holders are not uniformly distributed within communities. Sampling continued until thematic saturation was reached; the point at which no substantially new information emerged during interviews and observations. The final sample comprised 131 adult consumers and 18 elders, harvesters, processors and traders across nine regions.
2.3. Data Collection
Data were collected through semi-structured interviews, key informant discussions, and direct formal and informal observations at harvesting, processing, and market sites in accordance with established study guidelines for ethnographic research [
16,
17].
2.3.1. Interviews and Key Informant Discussions
Interviews captured information on harvesting practices, processing and preservation methods, storage materials, raw consumption habits, spoilage experiences, sensory attributes, and knowledge transmission. Interviews were conducted in Swahili with translation support where other indigenous languages were used. Key informant discussions with seven (7) elders and eleven (11) experienced practitioners in each community provided historical and cultural context.
2.3.2. Observations
Formal and informal observations documented tools, workflows, hygiene practices, and environmental conditions surrounding processing and trade. Photographs, brief video clips, and detailed field notes were used to support triangulation across methods.
2.3.3. Geospatial Mapping and Sample Collection
Geospatial coordinates of harvesting, occurrence, and commercialization points were recorded using KOBO Collect software installed in handheld field tablets. ArcGIS® (version10.7) was used for mapping visualization, while QGIS was used to produce final distribution maps of edible insects’ species across regions.
A total of 18 edible insects samples representing 6 major taxonomic orders (Orthoptera, Lepidoptera, Hymenoptera, Coleoptera, Isoptera and Diptera) and five processing states – raw (freshly harvested), sun-dried, fried, deep-fried, and smoked products – were randomly collected from local markets during ethnographic fieldwork, coinciding with peak seasonal availability of the respective species. Samples were collected in triplicate for each insect type and processing form. Samples were aseptically obtained from markets within the study regions where edible insects were traded at the time of data collection, packed in sterile bags, transported on dry ice and stored at −80 °C prior to analysis. The researchers conducted no additional processing prior to the microbial analysis, which was carried out at the Natural Resources Institute Food microbiology laboratory, University of Greenwich, United Kingdom.
2.3.5. Microbial Safety Assessment of Selected Traditionally Processed Insects
Microbial safety of selected traditionally processed edible insects was evaluated using standard culture based methods in accordance with East African Standards for edible insects (EAS 1186:2023) and corresponding ISO microbiological procedures ISO 6887-1. Total viable counts (TVC), of Coagulase-Positive Staphylococci, and yeasts and molds, were enumerated to assess overall hygienic quality and potential safety risks. Samples (25g) were aseptically homogenized in Maximum Recovery Diluent (MRD; 1:5, w/v), serially diluted, and plated in triplicate. Total viable counts were determined using nutrient agar supplemented with cycloheximide to suppress fungal growth, while Staphylococcus spp. were enumerated on Baird Parker Agar supplemented with Egg Yolk Tellurite. Yeasts and molds were enumerated on Dichloran Rose Bengal Chloramphenicol (DRBC) Agar (Oxoid, UK). Plates were incubated under ISO recommended conditions, and microbial counts were expressed as log₁₀ colony forming units per gram (log₁₀ CFU/g).
Presumptive pathogenic isolates were subjected to biochemical confirmation tests for presence or absence of Escherichia coli, Salmonella spp., and Listeria monocytogenes using API™ 20E and API™ Listeria identification systems (BioMérieux, Marcy-l’Étoile, France).
2.4. Data Analysis
Survey data were analysed using R Version 4.5.1 and MS Excel. Descriptive statistics summarized processing frequencies, storage tools, spoilage indicators, and sensory perceptions. Cross-tabulations compared insect species with preferred processing or storage methods. Geospatial data were visualized in ArcGIS® (version10.7).
3. Results and Discussion
3.1. Demographic Characteristics of Respondents
We interviewed 149 respondents including 18 harvesters, processors traders across nine regions. Respondents were predominantly rural, with a median age of above 45 years, and relatively low education levels (>50% had above primary level formal schooling). Similar demographic profiles have been documented in South Africa and Kenya, where indigenous knowledge systems face erosion due to modernization and youth disengagement [
17,
18].
Gendered roles were evident from harvesting to processing activities as women and children were more frequently involved in handpicking insects from farms, trees, and household adjacent environments, particularly caterpillars, grasshoppers, and termites, often for household consumption. In contrast, men were commonly engaged in labor intensive or equipment dependent harvesting methods, including digging anthills, climbing tall trees, nocturnal trapping, and large-scale collection intended for sale. Such gendered specialization is not only culturally embedded but also influences knowledge transmission and economic participation, necessitating the need for gender-sensitive interventions in emerging insect-based food systems [
10].
3.2. Edible Insects Harvesting Methods Diversity
Edible insect harvesting practices were primarily through wild harvesting and varied by insect type, habitat, and seasonal availability. Harvesting methods were insect and habitat specific ranging from handpicking locally made traps for bulk harvesting (
Table 1).
3.2.1. Direct Hand Picking and Indigenous Trapping Techniques
Table 1 presents edible insects harvesting methods from different habitats and the respective equipment used. Direct picking or hand collection from natural habitats was the most frequently reported method, particularly for grasshoppers (12.89%), caterpillars (9.52%), crickets (7.84%), and lake flies (5.04%). Trapping techniques were most prominent for swarm-forming and nocturnal species, notably winged termites (26.05%) and longhorn grasshoppers (10.64%). The reliance on wild harvesting observed strongly reflects the prevailing practice across Africa, although the specific methods used vary widely depending on the insect species, the local culture, and the particular country [
15,
19,
20,
21].
Anthill-dwelling insects, including termites and black ants, were collected using digging tools, bamboo/thorned sticks (Fig1A), smoke from burning green plants, and water light traps during swarming events. For insects such as termites, the observed practices included digging underground with tools, using smoke near anthills, and covering trenches with leaves and old clothes (Fig 1C). Beating the ground and singing to simulate rainfall to provoke termite emergence was also common and aligns with indigenous methods reported elsewhere in East Africa [
22,
23,
24].
Tree and crop associated insects, such as caterpillars, beetles, and grasshoppers, were predominantly harvested through handpicking, branch shaking, or trapping. Trapping particularly on tall host trees such as mango or cashew was done using a long stick with sticky tree paste (Fig2B) from Ficus sansibarica applied on its tip to hold the insect from flying out; the complete trap is locally known as “ulimbo” in swahili.
Figure 1.
Traditional Harvesting techniques, A) A thorned stick for termite harvesting; B) Sticky paste used in making trap; C) Trap for winged termites set on a mound.
Figure 1.
Traditional Harvesting techniques, A) A thorned stick for termite harvesting; B) Sticky paste used in making trap; C) Trap for winged termites set on a mound.
3.2.2. Bulk Trapping with Improved Techniques
At the lake zone, trapping Longhorn Grasshoppers and Winged Termites involved artificial light sources including high-voltage light bulbs placed along iron sheeting for a brighter spectrum (knowing the insects' attraction to light during mating flights or swarms) (
Table 1). The setup of this trap followed the description given by Mmari et al., (2017). This practice exemplifies the merging of indigenous techniques with modern tools to enable mass collection, often driven by the growing commercialization of edible insects. Lake flies, associated with aquatic environments, were harvested using water dampened sieves attached to bamboo sticks that were swirled around the lake flies ‘cloud’ or tree leaves, while others used mosquito nets. It was observed that lake flies that landed on the bitter leaf tree (
Vernonia amygdalina) locally known as
Nduluva tree leaves, were considered unfit for consumption. Other groups such as soil dwelling insects, including crickets and small beetles, were picked from the ground following probing burrows while longhorn beetles, called “madimu” locally, were harvested from dry, rotten logs of acacia trees using a hand axe. From a food safety perspective, harvesting methods involving soil disturbance, water contact, and smoke exposure, all of which present potential risks for microbial and physical contamination compared with controlled handpicking from vegetation [
25,
26].
While collection from the wild is the primary mode of acquisition, this approach presents sustainability concerns [
11]. In the Katavi region, the observed harvesting practice involved manually making specific incisions on acacia tree trunks to attract longhorn beetles to lay eggs. Under efforts to rescue the village forest, authorities had completely banned villagers’ entry to the forest, the violation of which could lead to 30 years' imprisonment. Although this practice has not stopped the villagers from sneaking into the forest search of their delicacy, it was reported to visibly slow down deforestation. This practice is also reported on palm weevils harvesting [
27,
28]. From a food safety perspective, harvesting methods involving soil disturbance, water contact, and smoke exposure present higher potential risks for microbial and physical contamination compared with controlled handpicking from vegetation [
25,
26]
.
3.3. Pre-Processing Storage
Prior to initial processing steps such as washing or thermal treatment, insects were held in storage containers, with the specific method reflecting the type of insect being held and the intended duration of storage (
Table 2). Plastic buckets were the most commonly used containers (63.1%) followed by breathable containers (28.9%) and traditional containers (18.1%). Plastic buckets were particularly used for termites, allowing an average freshness duration of 24–48 hours as reported by the harvesters. Buckets were often perforated or loosely covered to reduce heat accumulation, indicating indigenous knowledge of respiration and moisture control. The use of plastic reflects modernization and availability of plastic goods. While convenient, this may create anaerobic conditions promoting bacterial growth and contribute to plastic pollution, unlike traditional guards, clay pots and nests such as the
amaseme of Ukerewe villages, made of grasses and coconut leaves.
Sacks (28.9%), mesh cloth and containers with ventilation holes (25%) were commonly used for grasshoppers, especially the commercially harvested longhorn grasshoppers and were associated with shorter freshness windows (12–24 hours) during warmer seasons. We observed freshly harvested grasshopper sacks being transported via public transit to distant urban markets, including locations as far as Dar Es Salaam (1380 km away). This commercial demand highlights the critical link between temporary storage solutions and the safe marketability of these seasonal delicacies. The use of unhygienic woven sacks (recycled grain containers), has been directly linked to contamination by toxigenic molds (e.g.,
Aspergillus and
Penicillium strains) and the subsequent detection of aflatoxins in edible insects such as stink bugs [
29].
The survey results indicated that insects stored in plastic or safety bags exhibited the shortest freshness duration (<12 hours), attributed by respondents to rapid heating, moisture condensation, and odour development. In some tribes such as the Makonde, dead insects were considered unfit for consumption, "If one insect dies remove [it] immediately," reflecting understanding of cross-contamination. Traditional containers such as clay pots and nests, though infrequently used, were consistently associated with longer freshness retention (24-48 hours) by distant hunters, suggesting promising avenue for integrating and optimization of low-cost, breathable, biodegradable storage technologies to ensure quality and safety before the insects undergo processes such as drying or frying.
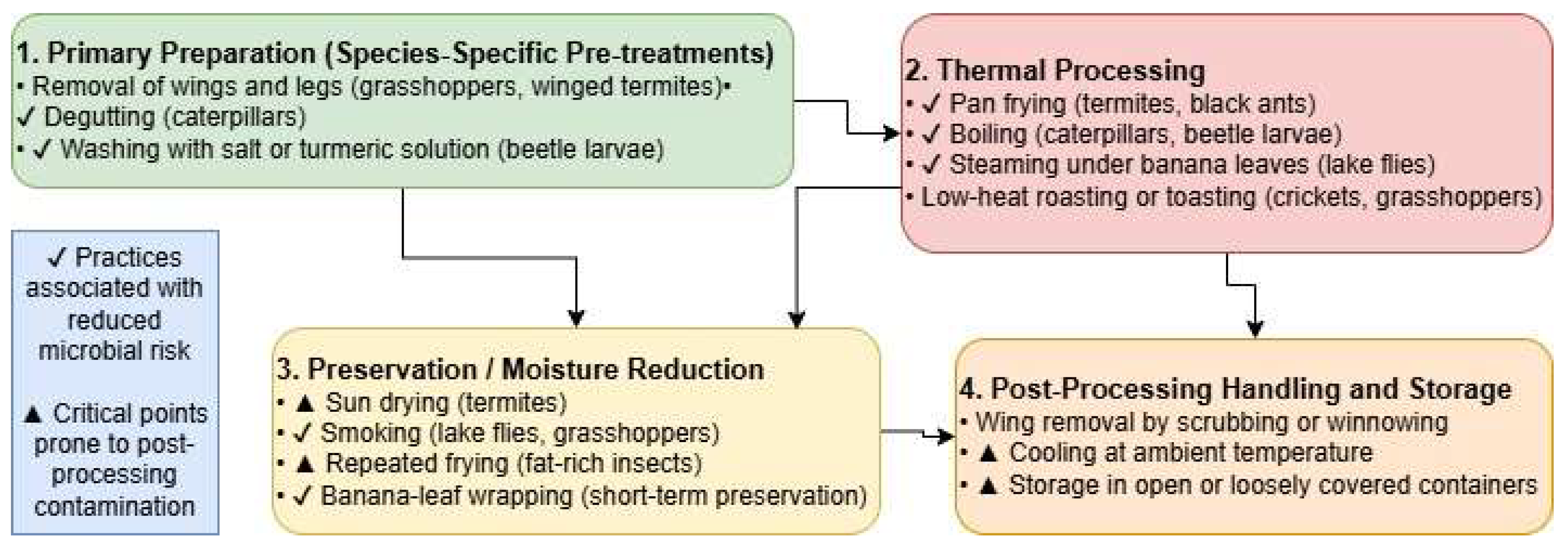
3.4. Processing and Preservation Practices
3.4.1. Sorting and Washing
Following harvesting, preliminary handling involved sorting to remove foreign materials and undesirable anatomical structures and washing by potable or salted water. This post-harvest sorting involved removal of soil, grasses, pieces of woods, wings, antennae, legs, and non-target insects. Species such as termites and grasshoppers required extensive sorting due to fragile appendages and high contamination risk from soil and debris.
Removal of anatomical appendages such as wings and legs observed in Orthopterae such as locusts and grasshoppers is crucial for elimination of their rigid chitinous exoskeletons [
30]. Respondents explained that improper removal could result in stomach problems in the consumer, a perception that aligns with earlier reports linking chitinous appendages to gastrointestinal obstruction, in some cases leading to surgery [
31]. Sorting of longhorn grasshoppers commonly involved the use of wood ash, which was explained as increasing friction and facilitating removal of appendages from the fatty body surface.
Certain caterpillar species, including
makarata and
maswethi, were traditionally degutted prior to washing to expel intestinal contents and undigested plant material. Similar practices have been reported for mopane worms (
Gonimbrasia belina) Mutungi et al., (2019) and palm weevils [
30]. Overall, these sorting and washing practices represent an important indigenous risk mitigation step that aligns closely with modern food hygiene principles by reducing physical contaminants and initial microbial load. Consistently, the Tanzania Bureau of Standards requires removal of materials unfit for human consumption during insect processing [
32].
3.4.2. Thermal Processing
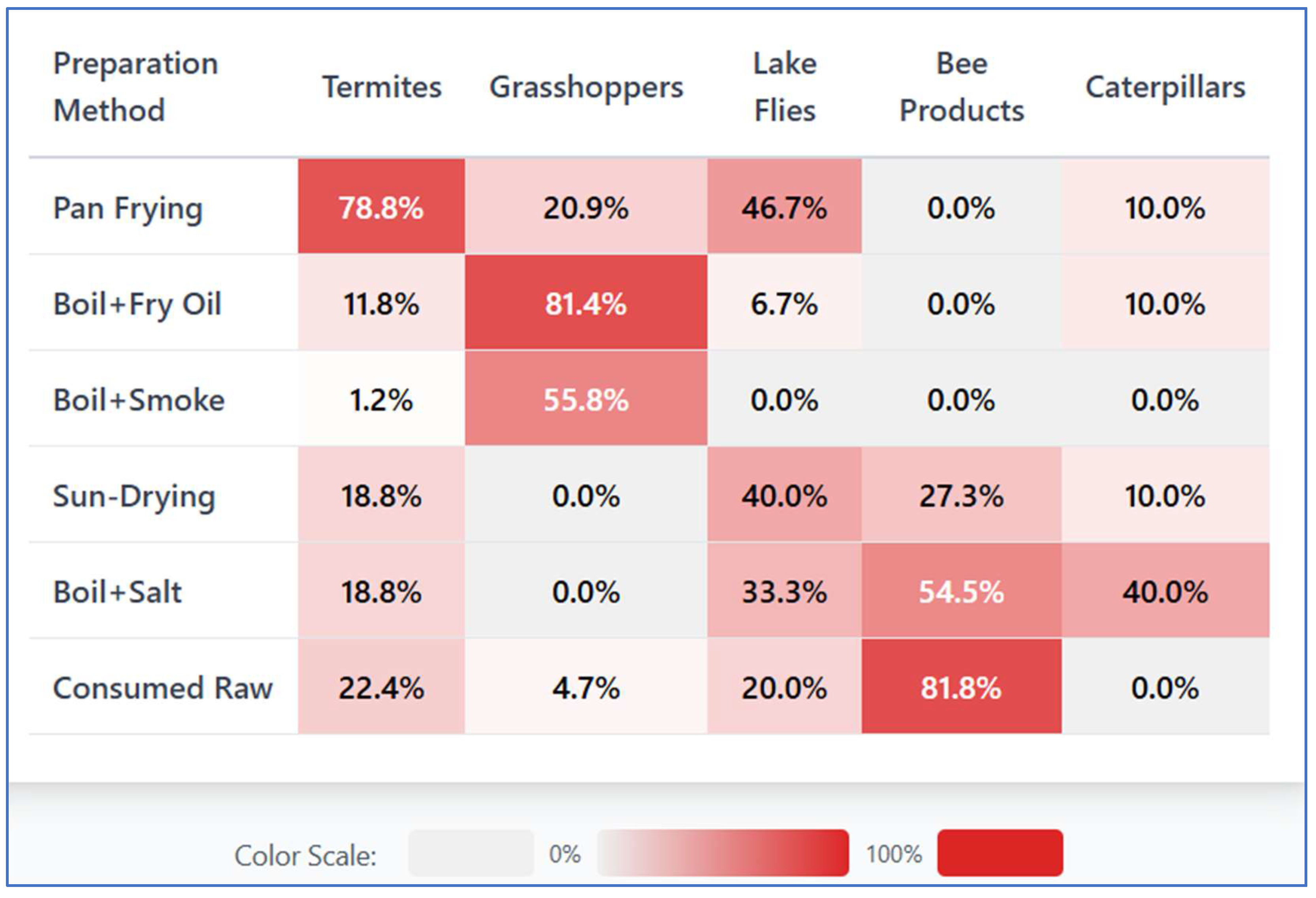
Traditional processing techniques were employed to enhance palatability, increase shelf life, and address food safety concerns associated with edible insects. Across the study sites, these practices demonstrated strong species-specific variation, shaped by insect morphology, perishability, cultural preferences, and intended use as shown in figure 2.
Figure 2.
A heat map showing different preparation methods for various edible insects.
Figure 2.
A heat map showing different preparation methods for various edible insects.
Heat-based processing formed the core of edible insect preparation. Across all respondents (n = 149), pan frying or dry toasting was the most frequently reported processing method (59.7%), predominantly applied to winged termites (n = 77). These were commonly fried in their own fat without added oil using rapid, low-moisture heating (5–15 min), a method perceived to enhance flavour while minimizing spoilage during peak emergence periods when harvest volumes are high but storage capacity is limited. Grasshoppers showed greater processing diversity, reflecting their larger body size and higher lipid content. Boiling with salt was a dominant first step especially for longhorn grasshoppers that were commonly boiled prior to frying in oil, toasting or smoke dried.
Reuse of frying oil was observed among 22.1% respondents, mainly for economical reasons. Sunflower oil was the most commonly used (14.4%), reflecting availability and affordability. Of those who re-used oil, 78.3% employed multiple re-uses "until finished" and (39.1%) stored the re-used oil in plastic bottles. This practice could potentially pose food safety risks from high peroxide values (oxidation), free fatty acids (hydrolysis), acrylamide formation and microbial contamination from improper storage [
33,
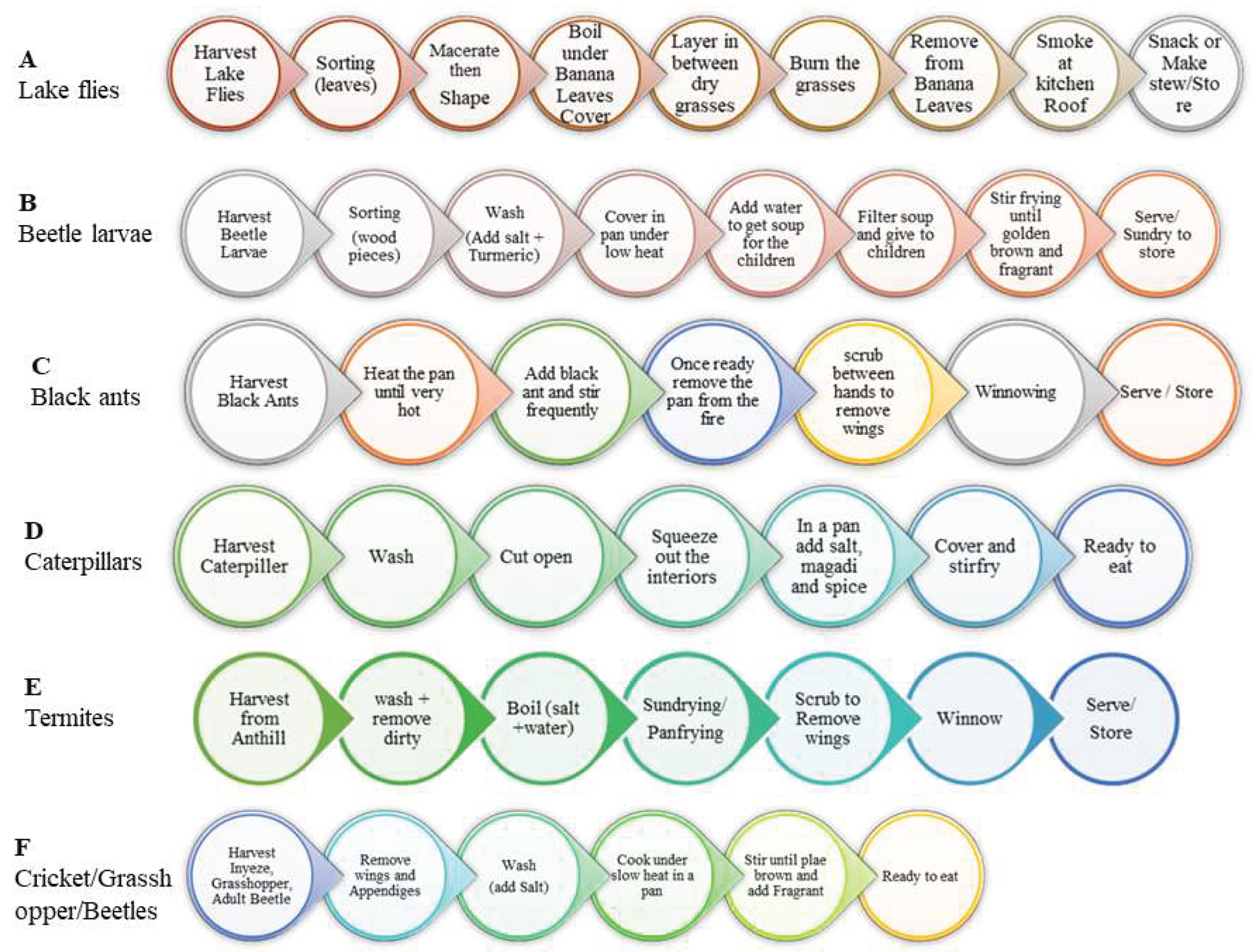
34].
Figure 3 presents traditional processing and preservation pathways of individual edible insects, indicating potential entry points for microbial hazard and elimination. Smoking was a common preservation method that extended processing time to 2-5 days. It was typically conducted on traditional drying structures under the firewood kitchen roof. Fuel sources included dry grasses and wood from eucalyptus, coffee, and cassava trees, with respondents associating smoke exposure with improved flavour and enhanced preservation, particularly for insects intended for storage or transport. Respondents displayed deep ethnobotanical knowledge, avoiding trees such as
Muhumula, Mujumba, and Mgenga because
"they are bitter/ sour thus affecting the taste”.
OR
Figure 3.
Traditional processing and preservation pathways of various edible insects.
Figure 3.
Traditional processing and preservation pathways of various edible insects.
Similar preservative and sensory roles of smoking have been reported in other studies [
25]. The polycyclic aromatic hydrocarbons (PAHs) accumulation risks associated with smoke produced by the pyrolysis of wood have been reported in some studies, though specific precise dangerous concentration are not clear. [
29,
35,
36]. After maceration (figure 3), lake flies were commonly wrapped in banana leaves and briefly burned on an open fire before smoking, while caterpillars were boiled with
magadi prior to toasting, indicating species-specific adaptations to texture and perceived safety. This traditional hurdle-like food safety approach suggests an implied understanding of shelf-life extension beyond immediate consumption as agreed in other studies [
37].
Sun drying emerged as the dominant preservation strategy for soil-dwelling termites reflecting both ecological accessibility and cultural norms (figure 3). Drying lasted from one day to one week. Insects were spread on mats, trays, tents, cow skins, or ‘clean’ rocks raising concerns about contamination from dust, insects, and domestic animals. While respondents perceived sun drying as effective for extending usability, emphasis on hygiene was noted from phrases like: "By keeping to the safe place," "You clean the tray every time," and "We clean the place first."
We observed minimal processing for crickets and locusts, likely reflecting their limited seasonal availability and lower cultural prominence in the surveyed regions. These were grilled on open fire after removal of appendages then consumed on spot (figure 3).
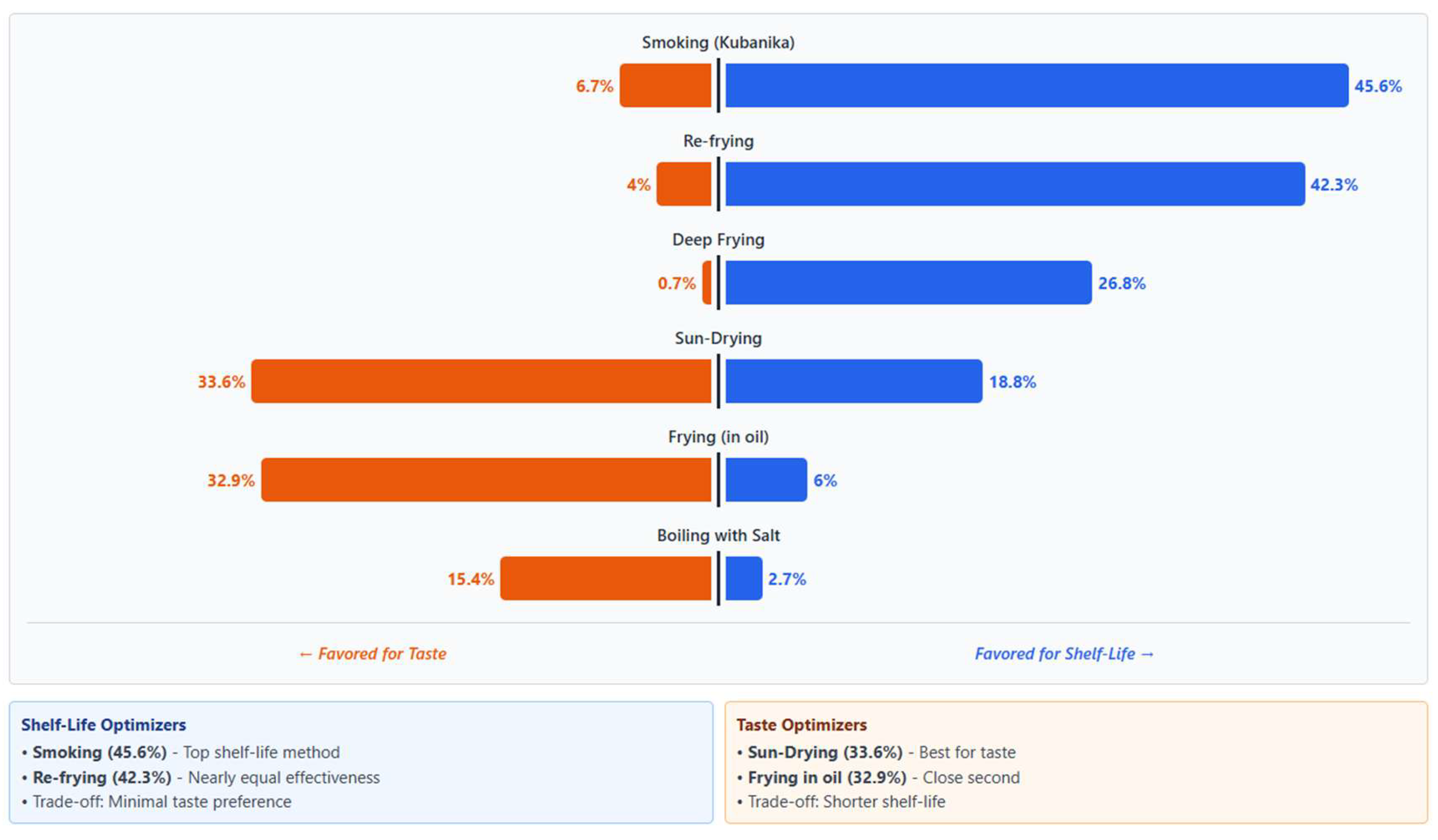
3.4.5. Perceived Preservation Trade-Offs: Sensory Quality Versus Shelf Life
Across insect species, respondents clearly distinguished methods based on goals and consistently balanced sensory quality against shelflife extension when selecting preservation methods.
Figure 3 shows a clear inverse relationship demonstrating that smoking and re-frying maximize storage duration but compromise sensory quality, while sun-drying and oil-frying preserve taste at the expense of shelf-life.
Figure 4.
Diverging bar chart showing indigenous knowledge of preservation trade-offs.
Figure 4.
Diverging bar chart showing indigenous knowledge of preservation trade-offs.
Rapid frying and toasting were preferred for immediate consumption due to superior taste and aroma, whereas boiling, smoking, and sun drying were favoured for products intended for storage. For longer preservation, frying in oil was often criticized for making insects "sore on the throat", "bitter," or "sour." Sun-drying was also noted by some to cause "off flavour and aftertaste" if not done properly.
These trade-offs highlight the practical decision making embedded within indigenous processing systems and identify clear entry points for HACCP-aligned optimization, including improved drying infrastructure, standardized boiling times, and hygienic storage containers, while preserving culturally embedded knowledge [
38].
3.5. Raw Consumption Patterns and Safety Perceptions
Raw consumption of edible insects was reported by 30.9% of respondents (figure 4), indicating that while traditionally discouraged and not dominant in many tribes, the practice remains culturally relevant. Raw consumption was strongly insect specific, with termites accounting for 70.5% followed by honeycombs/bee broods (25%) and lake flies (11.4%) reflecting perceptions of softness, sweetness, and cleanliness. Chi-square analysis confirmed a significant association between insect type and raw consumption (χ² = 35.42,
p = 0.008; Cramer's V = 0.32), indicating that to some extent species choice dictates whether an insect is eaten raw (figure 4). The traditional practice of consuming insects uncooked aligns with historical patterns seen across Africa, Asia, and Latin America [
39]
.
Motivations for consuming insects raw were primarily sensory (69.6%);
"They are delicious," perceived nutrition (29.5%);
"They have more nutrients," and habitual (15.9%);
"Just grew up consuming them raw" (figure 5C). These perceptions can be justifiable given that thermal processing, such as boiling or blanching, is known to result in the leaching and loss of water soluble nutrients [
40,
41,
42]. Despite these motivations, the majority of respondents expressed safety concerns, most notably fear of illness (71.1%), unpleasant taste (58.4%), bacterial contamination (28.9%) and “yucky factor” (figure 5D). The acknowledged safety concerns are scientifically sound as raw insects inherently harbor a high microbial load, including pathogenic bacteria, yeasts, molds, and spore-forming bacteria [
10,
43].
3.6. Perceived Spoilage Indicators, Causes, and Management of Processed Insects
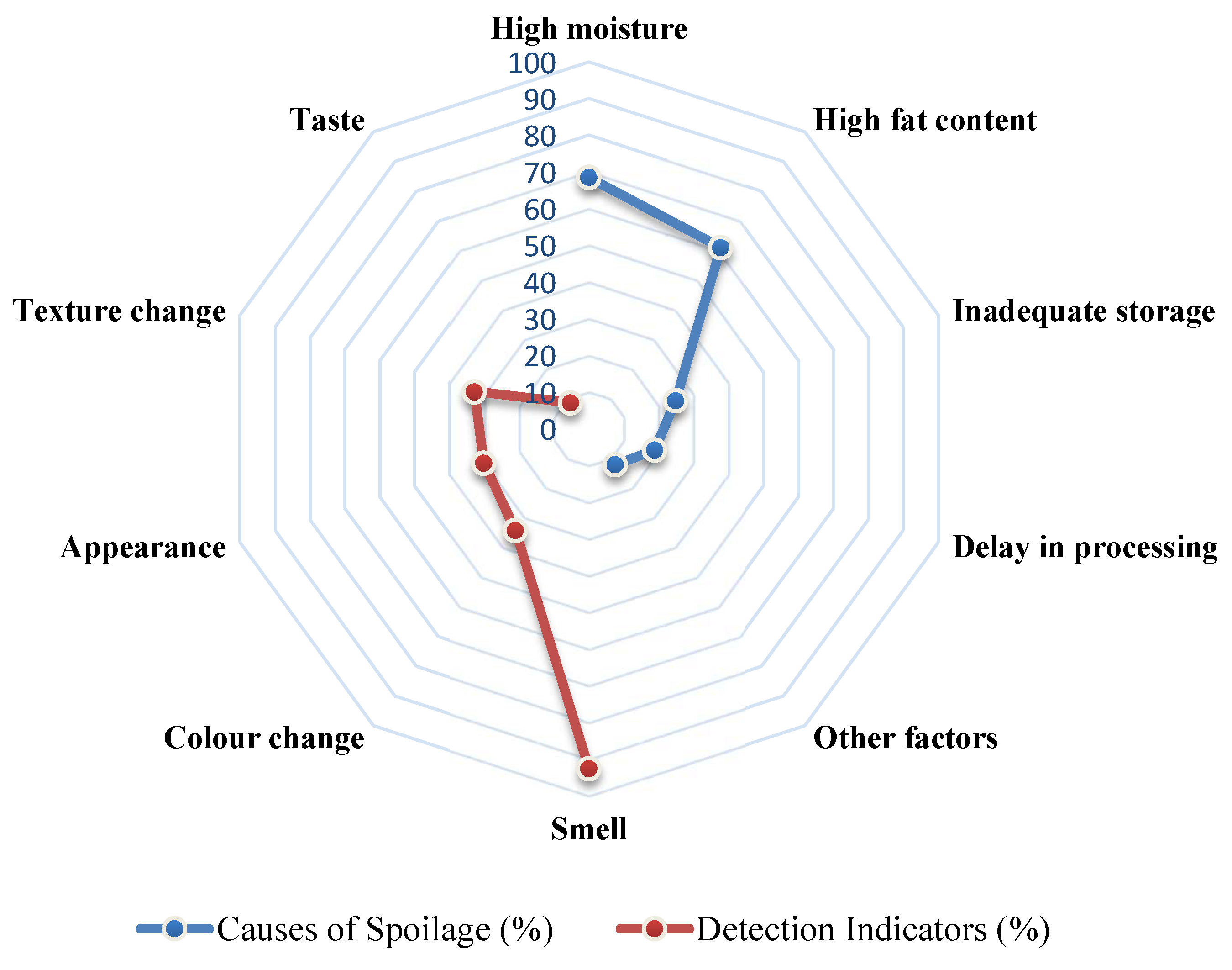
Spoilage management of processed edible insects relies on multi-sensory assessment, leveraging traditional knowledge systems. Smell was the most frequently used spoilage indicator (92.6%);
"Producing offensive/rotten smell," followed by changes in colour (34.2%), texture change (32.9%), and appearance (30.2%) (
Figure 5). Although this sensory based monitoring is often triggered when spoilage is well advanced, these indicators were coherent across insect types and processing methods, underlining the role of experiential knowledge in food safety assessment [
10,
29].
When asked to identify the primary causes of spoilage, respondents frequently cited high moisture content (68.5%) and high fat content (61.1%) followed by delay in processing and improper storage causing insect death;
"When one dies [it] spoils others." (
Figure 6). These perceived causes align with known food science principles, particularly in sun-dried and fried insects where residual moisture and lipid oxidation accelerate deterioration [
33,
40]. Despite recognizing improper storage as a factor, statistical analysis indicated no significant association between the specific storage method used and the reported spoilage time (
χ2=15.47,
p=0.051). This suggests that environmental conditions and handling practices drove variation rather than the container type alone. This observation emphasizes that training in basic hygiene practices and sanitation is more immediately impactful than changing storage containers.
Once spoilage was detected, discarding insects was the most common response (55.0%), followed by repurposing to animal feed (40.3%). While a few concerning practices were noted:
"Mixed with fresh and sell" and
"Recook and eat," mixing spoiled insects with fresh ones was infrequently reported (8.1%), indicating strong cultural norms against consuming visibly deteriorated products. Though minimizing economic loss, repurposing spoiled insects as animal feed (depending on the quality of the spoiled material) can transfer toxigenic molds such as aflatoxins from insects stored in unhygienic containers, which poses a serious threat if transferred to livestock [
44].
Overall, these findings demonstrate that while traditional processing and storage practices support widespread edible insect consumption, moisture control, fat stabilization, and hygienic handling remain critical vulnerability points. The results provide empirical justification for introducing context-appropriate improvements, such as controlled drying, improved ventilation during storage, and basic hygiene training, without undermining established indigenous practices.
3.7. Microbial Safety of Indigenous Processing and Preservation Methods
Microbial analysis of market-ready indigenously processed edible insects revealed considerable variation in microbial loads across insect species and processing methods. These differences reflect variations in moisture content, thermal intensity, and post-processing handling practices.
Table 3 shows the microbial safety results of the selected edible insects interpreted against national and regional food safety benchmarks, including EAS 1186:2023 and relevant European Union guidance (EU 2017/625). Samples exceeding 5.0 log₁₀ CFU/g for total plate count or 3.0 log₁₀ CFU/g for yeasts and molds were classified as non-compliant, indicating inadequate post-processing hygiene, incomplete moisture reduction, or suboptimal storage conditions [
45,
46].
3.7.1. Total Viable Counts (TVC)
Total viable counts ranged from 1.56 to 6.89 log₁₀ CFU/g across all samples. The lowest counts were consistently observed in deep-fried and smoked insect products, including deep-fried Ruspolia differens (1.56 log₁₀ CFU/g) and smoked lake flies (1.91 log₁₀ CFU/g). These processing methods combine high thermal input with effective moisture reduction, limiting microbial survival and re-growth.
In contrast, toasted winged termites (
Macrotermes spp.) and boiled/pan-cooked beetle larvae (
Thithoes confinis) recorded higher TVC values exceeding the EAS 1186:2023 maximum limit of 1 × 10⁵ CFU/g (5.0 log₁₀ CFU/g). These higher counts are likely attributable to post-processing contamination, prolonged cooling at ambient temperatures, and residual moisture retained during cooking and storage in non-airtight containers. Observations from the processing flow diagrams (
Figure 3) indicate multiple open-handling steps that may facilitate recontamination. Similar patterns have been reported for traditionally processed insect products sold in informal markets, where open-air handling and storage promote microbial proliferation [
25,
27,
33].
3.7.2. Coagulase-Positive Staphylococci (Presumptive) Count
Presumptive coagulase-positive staphylococci enumerated using Baird–Parker agar supplemented with egg yolk tellurite revealed that, with the exception of deep-fried R. differens, all samples had detectable levels of coagulase-positive staphylococci, with counts ranging from 1.59 to 6.28 log₁₀ CFU/g. Sun-dried grasshoppers, toasted termites, and cooked beetle larvae exceeded the EAS 1186:2023 requirement for absence of coagulase-positive staphylococci.
Smoked products consistently recorded lower counts (<2 log₁₀ CFU/g), highlighting the effectiveness of combined heat treatment and moisture reduction in controlling staphylococcal contamination. The presence of coagulase-positive staphylococci in minimally dried and manually handled products suggests post-processing contamination from handlers, utensils, or environmental surfaces, rather than survival through cooking. This observation is consistent with previous studies identifying handling hygiene as a critical control point in traditional insect value chains [
40,
42,
45].
3.7.3. Yeast and Mold
Yeasts and molds were detected only in toasted winged termites (2.99 log₁₀ CFU/g) and cooked beetle larvae (2.61 log₁₀ CFU/g), with all values remaining within the EAS limit of ≤3.0 log₁₀ CFU/g. Although fungal counts were generally low, their presence indicates potential risks associated with incomplete drying, high residual moisture, and prolonged exposure during traditional processing, particularly under humid environmental conditions. These findings align with ethnographic observations of sun drying and toasting conducted on open mats or ground surfaces, often without protective covers, which may facilitate fungal contamination [
29]. For example, study on the mopane caterpillar (
Imbrasia belina) showed that sun-drying after boiling risks mold growth and aflatoxin contamination, leading to quality deterioration [
47].
3.7.4. Escherichia coli, Salmonella spp. and Listeria Spp
All insect samples tested negative for
Escherichia coli,
Salmonella spp., and
Listeria monocytogenes, fully complying with [
32] pathogen requirements. Biochemical confirmation using API™ 20E verified that all suspect isolates were non-pathogenic background flora. API™ Listeria testing identified
Listeria welshimeri,
Listeria grayi, and
Listeria ivanovii. The detection of these non-monocytogenes
Listeria species suggests environmental contamination during post-processing handling, consistent with the post-processing exposure stages identified in
Figure 3, particularly ambient cooling and open-container storage, The absence of these key foodborne pathogens indicates that indigenous processing practices, particularly thermal treatments such as frying, smoking, boiling, and toasting are effective at eliminating major microbial hazards, even in informal market settings with limited formal hygiene controls. These findings align with previous studies demonstrating the strength of traditional insect processing methods in microbial elimination when adequate heat treatment is applied [
10,
29,
33]..
4. Food Safety taboos and Indigenous Knowledge Transmission
Processing practices were primarily transmitted orally and experientially through storytelling, observation, and informal evening “fire chats,” locally referred to as
kikoomi among the Wakerewe tribe. No written records of these practices were identified during fieldwork, and younger generations were observed to have limited awareness of these traditions, reflecting a broader trend of erosion of indigenous knowledge systems [
48]. Survey data further indicated that very few (n=5) respondents involved in harvesting and processing had received formal training in food hygiene from a local institute, emphasizing the central role of inherited knowledge in shaping food safety and preservation behaviors [
27].
The survey reveals specific science-informed taboos and practices regarding food safety and risk mitigation strategies (
Table 4). The use of
magadi (alkaline salts) during insect cooking was widely reported and culturally justified to prevent abdominal discomfort or insects “becoming alive” in the stomach. While framed symbolically, this practice aligns with functional food safety outcomes, as alkaline treatment and boiling can reduce microbial loads and undesirable compounds [
10]. Similarly, the consumption of raw insects was broadly prohibited due to beliefs that it could cause intestinal damage, stomach pain, or speech impairment as insects bite the tongue. The strong association of raw consumption with stomach pain and diarrhea and the attribution of spoilage to bacteria demonstrate a realistic understanding of microbial food safety risks, even without formal training. These taboos reinforce the importance of thermal processing and combined methods (e.g., boiling + smoking + sun-drying) as a form of inherent “hurdle technology” reducing water activity and adding antimicrobial compounds from smoke, creating multiple barriers to microbial growth. This is consistent with empirical evidence that combined methods reduces microbial risks and gastrointestinal illness [
17].
Cultural protocols also governed waste handling and preservation practices. Among
senene (
Ruspolia differens) harvesters, wings and appendages were discarded at least 10 feet from households to avoid ghost attraction, contributing to household hygiene. In Katavi, hunters were prohibited from carrying salt into forests, a violation believed to chase away the ghost responsible for hunting fortune. This restriction promoted smoking as a preservation strategy, allowing insects to be stored safely for several days during extended hunting periods [
27].
Overall, indigenous taboos and knowledge transmission systems function as adaptive food safety mechanisms embedded within cultural practice [
18,
49]. Although often explained through symbolic narratives, many of these practices align closely with established food science principles related to microbial control, spoilage reduction, and hygiene [
18,
49,
50]. Recognizing and integrating such indigenous knowledge into formal food safety frameworks presents an opportunity to strengthen culturally appropriate and scalable insect value chains without eroding traditional practices [
48].
4. Conclusions
This study provides an integrated ethnographic and microbiological assessment of indigenous edible insect processing and preservation practices in Tanzania. The findings demonstrate strong method insect specificity in traditional processing, reflecting relationship to insect morphology, perishability and intended use. Combination of treatments such as frying, boiling, smoking, and sun drying were widely applied and generally effective in ensuring pathogen safety, as evidenced by the absence of Salmonella spp., Escherichia coli, Staphylococcus aureus, and Listeria monocytogenes in analysed samples. However, variability in total viable counts and yeast and mold levels highlights persistent challenges in microbial load control and spoilage prevention, particularly for sun dried and stored products. These challenges are closely linked to drying efficiency, post-processing handling, and storage conditions, reinforcing the relevance of critical control points emphasized in the IPIFF, Tanzania Bureau of Standards (TBS) and East African edible insect standards. Overall, the results confirm that indigenous processing systems offer foundation for safe insect food production, while also revealing key areas requiring technical refinement to support storage stability and commercialization.
5. Recommendations
The method of insect specificity embedded within indigenous processing practices provides a valuable entry point for HACCP aligned optimization without undermining cultural integrity. Priority interventions should focus on documentation, improving drying infrastructure, promoting hygienic cooling and handling, and encouraging the use of food grade storage and packaging materials suited to local contexts. Low cost innovations such as raised drying platforms, protective covers, improved smoking structures, and standardized thermal processing times could substantially reduce microbial and spoilage risks. Traditional additives such as alkaline agents (e.g., magadi and ash), widely used to enhance palatability and perceived safety are worth investigating as potential functional additives in scaling insect processing.
6. Limitations and Future Research
6.1. Study Limitations
Key limitations include reliance on self-reported practices that may introduce recall or reporting bias for seasonal availability. Microbial analyses were culture-based, conducted on insect species found on the market during the sampling period, which may not capture the full diversity of practices and extended storage across all ecological zones and seasons. Molecular detection methods were beyond the scope of this work and may be explored in future studies to complement food safety assessment.
6.2. Future Research
Future research should integrate microbiological outcomes with physicochemical parameters, including moisture content, water activity, and pH, to generate predictive shelf-life models for traditionally processed insects. The findings will be essential for supporting commercialization and regional trade under harmonized standards.
Author Contributions
Conceptualization; M.M., R.S. and J.K. Methodology; M.M., R.S. and J.K. Validation; All authors; Data collection; M.M. Drafting paper; M.M. Review & Editing of the manuscript; All authors.
Funding
This work was supported by Partnership for Skills in Applied Sciences, Engineering and Technology—Regional Scholarship and Innovation Fund (PASET-RSIF) (Project Grant No. P165581) and developing the final draft support was given through the BA International Writing Workshop Programme Grant IWW24\100690, Enhancing academic writing processes and practice: strengthening academic writing skills in East Africa (Kenya, Uganda, Tanzania).
Institutional Review Board Statement
Ethical approval was granted by the Sokoine University of Agriculture Ethical Review Board (SUA/ADM/R.1/8/890). For the transportation of samples an international health certificate for samples and biologicals export permit was issued by the United Republic of Tanzania (DVS-TZ/14/2025/00099). Permission was obtained from community elders prior to fieldwork.
Data Availability Statement
Most generated data during this study are included in this manuscript. More information is available on request.
Acknowledgments
The authors would like to thank key informants for their indigenous knowledge contribution. Mary Tanyitiku of NRI, Food Microbiology lab for technical support. The authors have reviewed and edited the output and take full responsibility for the content of this publication.
Conflicts of Interest
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
- FAO. Edible insects. Future prospects for food and feed security. 2013. [Google Scholar]
- Omuse, ER; Tonnang, HEZ; Yusuf, AA; Machekano, H; Egonyu, JP; Kimathi, E; et al. The global atlas of edible insects: analysis of diversity and commonality contributing to food systems and sustainability. Scientific Reports 2024, 14, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Olivadese, M; Dindo, ML. Edible Insects: A Historical and Cultural Perspective on Entomophagy with a Focus on Western Societies. Insects 2023, 14, 1–16. [Google Scholar] [CrossRef] [PubMed]
- van Huis, A; Oonincx, DGAB. The environmental sustainability of insects as food and feed. A review. In Agronomy for Sustainable Development; 2017; p. 37. [Google Scholar] [CrossRef]
- Bodenheimer, FS. Insects as Human Food; 1951. [Google Scholar] [CrossRef]
- Matandirotya, NR; Filho, WL; Mahed, G; Maseko, B; Murandu, CV. Edible Insects Consumption in Africa towards Environmental Health and Sustainable Food Systems: A Bibliometric Study. International Journal of Environmental Research and Public Health 2022, 19. [Google Scholar] [CrossRef] [PubMed]
- Mmari, MW; Kinyuru, JN; Laswai, HS; Okoth, JK. Traditions, beliefs and indigenous technologies in connection with the edible longhorn grasshopper Ruspolia differens ( Serville 1838 ) in Tanzania. Journal of Ethnobiology and Ethnomedicine 2017, 1–11. [Google Scholar] [CrossRef]
- Halloran, A; Flore, R; Vantomme, P; Roos, N. Edible Insects in Sustainable Food Systems; 2018. [Google Scholar] [CrossRef]
- Musundire, R; Ngonyama, D; Chemura, A; Ngadze, RT; Jackson, J; Matanda, MJ; et al. Stewardship of Wild and Farmed Edible Insects as Food and Feed in Sub-Saharan Africa: A Perspective. Frontiers in Veterinary Science 2021, 1–9. [Google Scholar] [CrossRef]
- Akullo, JO; Egonyu, JP; Ng’ang’a, J; Kiiru, S; Mmari, M; Ndiritu, A; et al. Post-harvest handling, processing and value addition of edible insects as a sustainable food resource in Africa: A review. International Journal of Tropical Insect Science 2025, 45, 21–41. [Google Scholar] [CrossRef]
- Ishara, J; Cokola, MC; Buzera, A; Mmari, M; Bugeme, D; Niassy, S; et al. Edible insect biodiversity and anthropo-entomophagy practices in Kalehe and Idjwi territories, D.R. Congo. Journal of Ethnobiology and Ethnomedicine 2023, 19, 1–17. [Google Scholar] [CrossRef]
- Murefu, TR; Macheka, L; Musundire, R; Manditsera, FA. Safety of wild harvested and reared edible insects: A review. Food Control 2019, 101. [Google Scholar] [CrossRef]
- Kumar, P.; Chidozie Ogwu , M. Regulatory Frameworks and Challenges for Edible Insects: Pathways to Mainstream Adoption. In Edible Insects: Nutritional Benefits, Culinary Innovations and Sustainability; Springer Nature Switzerland: Cham, 2025; pp. 165–88. [Google Scholar] [CrossRef]
- Lisboa, HM; Nascimento, A; Arruda, A; Sarinho, A; Lima, J; Batista, L; et al. Unlocking the Potential of Insect-Based Proteins: Sustainable Solutions for Global Food Security and Nutrition. Foods 2024, 13, 1846. [Google Scholar] [CrossRef]
- FAO. Looking at edible insects from a food safety perspective. 2021. [Google Scholar] [CrossRef]
- Kwame Harrison A. Ethnography. 1st edition. Oxford University PressNew York. 2018. [CrossRef]
- Raheem, D; Carrascosa, C; Oluwole, OB; Nieuwland, M; Saraiva, A; Millán, R; et al. Traditional consumption of and rearing edible insects in Africa, Asia and Europe. Critical Reviews in Food Science and Nutrition 2019, 59, 2169–88. [Google Scholar] [CrossRef] [PubMed]
- Chidozie Ogwu, M. Entomophagy Through a Cultural Lens: Traditions, Barriers, and Opportunities. Edible Insects: Nutritional Benefits, Culinary Innovations and Sustainability 2025, 325–44. [Google Scholar] [CrossRef]
- Netshifhefhe, SR; Kunjeku, EC; Duncan, FD. Human uses and indigenous knowledge of edible termites in Vhembe District, Limpopo Province, South Africa. South African Journal of Science 2018, 114, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Selaledi, L; Hassan, Z; Manyelo, TG; Mabelebele, M. Insects’ production, consumption, policy, and sustainability: What have we learned from the indigenous knowledge systems? Insects 2021, 12. [Google Scholar] [CrossRef]
- Sileshi, GW; Nyeko, P; Nkunika, POY; Sekematte, BM; Akinnifesi, FK; Ajayi, OC. Integrating ethno-ecological and scientific knowledge of termites for sustainable termite management and human welfare in Africa. Ecology and Society 2009, 14. [Google Scholar] [CrossRef]
- van Huis, A. Cultural significance of termites in sub-Saharan Africa. Journal of Ethnobiology and Ethnomedicine 2017, 13, 8. [Google Scholar] [CrossRef]
- Kinyuru, JN; Konyole, SO; Roos, N; Onyango, CA; Owino, VO; Owuor, BO; et al. Author ’ s personal copy Nutrient composition of four species of winged termites consumed in western Kenya. Journal of Food Composition and Analysis 2013, 30, 120–4. [Google Scholar] [CrossRef]
- Ayieko, MA; Obonyo, GO; Odhiambo, JA; Ogweno, PL; Achacha, J; Anyango, J. Constructing and Using a Light Trap Harvester : Rural Technology for Mass Collection of Agoro Termites (Macrotermes subhylanus ). 2011, 3, 105–9. [Google Scholar]
- Melgar-Lalanne, G; Hernández-Álvarez, A; Salinas-Castro, A. Edible Insects Processing: Traditional and Innovative Technologies. Comprehensive Reviews in Food Science and Food Safety 2019, 18, 1166–91. [Google Scholar] [CrossRef]
- Ng’ang’a, J; Fombong, F; Kiiru, S; Kipkoech, C; Kinyuru, J. Food safety concerns in edible grasshoppers: a review of microbiological and heavy metal hazards. International Journal of Tropical Insect Science 2021, 41, 2103–11. [Google Scholar] [CrossRef]
- Niassy, S; Fiaboe, KKM; Affognon, HD; Akutse, KS; Tanga, MC; Ekesi, S. African indigenous knowledge on edible insects to guide research and policy. Journal of Insects as Food and Feed. 2016, 2, 161–70. [Google Scholar] [CrossRef]
- Van Itterbeeck, J; van Huis, A. Environmental manipulation for edible insect procurement: a historical perspective. Journal of Ethnobiology and Ethnomedicine 2012, 8, 3. [Google Scholar] [CrossRef] [PubMed]
- Thanasakran, S; Abu Hasan, H; Chong, AS-C; Baidurah, S. A Review of Pre-treatments, Drying Methods, and Processing of High-protein Insect Products. Pertanika Journal of Tropical Agricultural Science 2025, 48. [Google Scholar] [CrossRef]
- Ishara, J; Ayagirwe, R; Karume, K; Mushagalusa, GN; Bugeme, D; Niassy, S; et al. Inventory reveals wide biodiversity of edible insects in the Eastern Democratic Republic of Congo. Scientific Reports | 2022, 12, 1576. [Google Scholar] [CrossRef]
- Bouvier, G. Quelques questions d’entomologie vétérinaire et lutte contre certains arthropodes en Afrique tropicale. Acta Trop 1945, 2, 42–59. [Google Scholar]
- EAS1186, EA. East African Standard. East African Standards 2023, 3 EAS 1186, 2023:9. [Google Scholar]
- FAO; Food and Agriculture Organization of the United Nations (FAO). Looking at edible insects from a food safety perspective; Challenges and opportunities for the sector: Rome, 2021. [Google Scholar]
- FASFC. Food safety aspects of insects intended for human consumption ( Sci Com dossier 2014 / 04 ; SHC dossier No 9160 ). 2014; pp. 1–22.
-
Insects as food and food ingredients: technological improvements, sustainability, and safety aspects; Garcia-Vaquero, M, García, CÁ, Eds.; Academic Press: Lodno San Diego, CA Cambridge, MA Kidlington, Oxford, 2024. [Google Scholar]
- Sun, Y; Wu, S; Gong, G. Trends of research on polycyclic aromatic hydrocarbons in food: A 20-year perspective from 1997 to 2017. Trends in Food Science & Technology 2019, 83, 86–98. [Google Scholar] [CrossRef]
- Škvorová, P; Kulma, M; Kouřimská, L; Švejstil, R. EFFECT OF KILLING AND TECHNOLOGICAL PROCESSING ON MICROBIOLOGICAL QUALITY OF EDIBLE INSECTS. Journal of microbiology, biotechnology and food sciences 2024, e11018. [Google Scholar] [CrossRef]
- IPIFF. IPIFF Guide on Good Hygiene Practices for European Union producers of insects as food and feed. 2018. [Google Scholar]
- van Huis, A. Prospects of insects as food and feed. Organic Agriculture 2021, 11, 301–8. [Google Scholar] [CrossRef]
- Mutungi, C; Irungu, FG; Nduko, J; Mutua, F; Affognon, H; Nakimbugwe, D; et al. Postharvest processes of edible insects in Africa: A review of processing methods, and the implications for nutrition, safety and new products development. Critical Reviews in Food Science and Nutrition 2019, 59, 276–98. [Google Scholar] [CrossRef] [PubMed]
- Kinyuru, JN; Kenji, GM; Njoroge, SM; Ayieko, M. Effect of Processing Methods on the In Vitro Protein Digestibility and Vitamin Content of Edible Winged Termite (Macrotermes subhylanus) and Grasshopper (Ruspolia differens). Food and Bioprocess Technology 2010, 3, 778–82. [Google Scholar] [CrossRef]
- Nyangena, DN; Mutungi, C; Imathiu, S; Kinyuru, J; Affognon, H; Ekesi, S; et al. Effects of Traditional Processing Techniques on the Nutritional and Microbiological Quality of Four Edible Insect Species Used for Food and Feed in East Africa. Foods 2020, 9, 574. [Google Scholar] [CrossRef] [PubMed]
- Ng’ang’a, J; Imathiu, S; Fombong, F; Ayieko, M; Vanden, J; John, B. Microbial quality of edible grasshoppers Ruspolia differens ( Orthoptera : Tettigoniidae ): From wild harvesting to fork in the Kagera Region, Tanzania; May 2018. [Google Scholar] [CrossRef]
- Baiano, A. Edible insects: An overview on nutritional characteristics, safety, farming, production technologies, regulatory framework, and socio-economic and ethical implications. Trends in Food Science and Technology 2020, 35–50. [Google Scholar] [CrossRef]
- Belluco, S; Losasso, C; Maggioletti, M; Alonzi, C; Ricci, A; Paoletti, MG. Edible insects: A food security solution or a food safety concern? Animal Frontiers 2015, 5, 25–30. [Google Scholar] [CrossRef]
- EFSA. Risk profile related to production and consumption of insects as food and feed. EFSA Journal 2015, 13. [Google Scholar] [CrossRef]
- Simpanya, MF; Allotey, J; Mpuchane, SF. A Mycological Investigation of Phane, an Edible Caterpillar of an Emperor Moth, Imbrasia belina. Journal of Food Protection 2000, 63, 137–40. [Google Scholar] [CrossRef]
- Zabentungwa T. Hlongwane RS and TCM. Indigenous Knowledge about Consumption of Edible Insects in South Africa. Insects, 2021; 14, 17131. [CrossRef]
- Abbasi, E. A review of cultural aspects and barriers to the consumption of edible insects. Health Science Monitor 2024, 3, 179–94. [Google Scholar] [CrossRef]
- Chidozie Ogwu, M; Chibueze Izah, S. Edible Insects: Cultural Heritage and a Global Solution for Food Security. In Edible Insects: Nutritional Benefits, Culinary Innovations and Sustainability; Springer Nature Switzerland: Cham, 2025; pp. 1–20. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).