Submitted:
02 March 2026
Posted:
02 March 2026
You are already at the latest version
Abstract
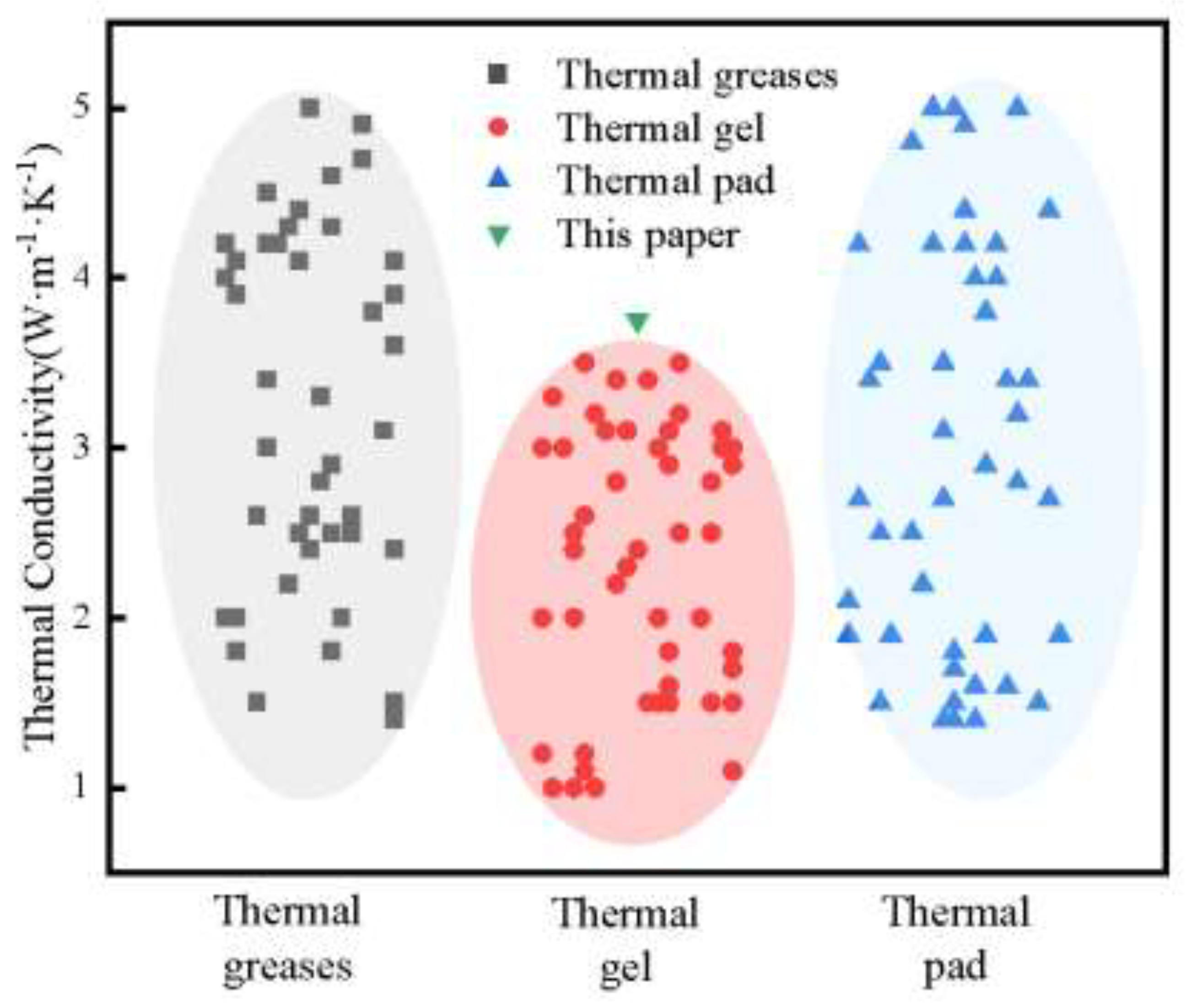
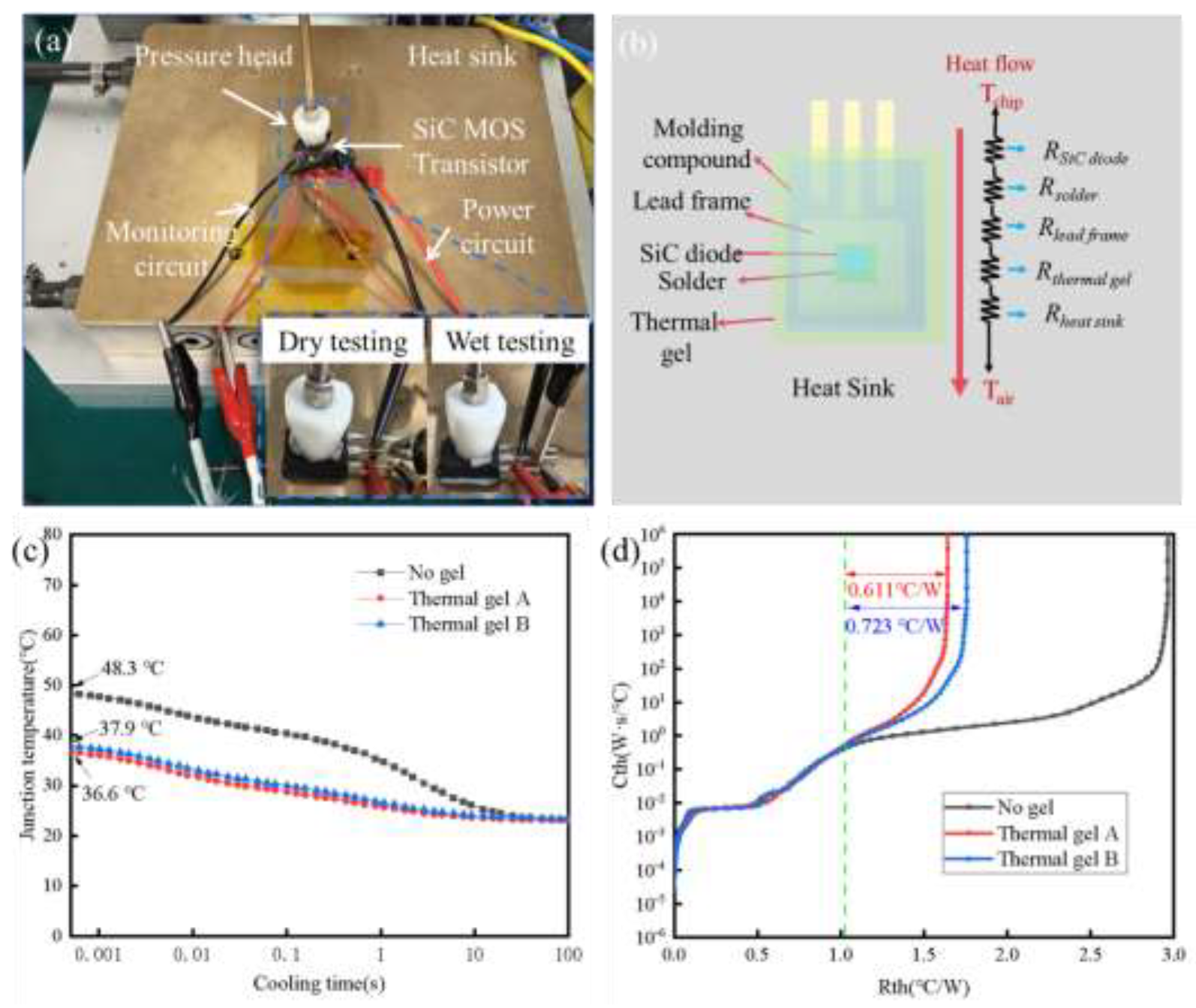
Thermal interface materials (TIMs) are essential for addressing heat dissipation challenges in high-performance electronic devices. Among various TIMs, thermal conductive gels exhibit significant potential in high heat flux applications due to their excellent flexibility and superior gap-filling capability. Current research primarily concentrates on the fabrication and performance characterization of novel thermal conductive gels, while comparatively little attention has been devoted to the optimization of processing parameters. Furthermore, existing characterization methods often fail to accurately replicate real-world operating conditions, resulting in discrepancies between laboratory measurements and actual performance. An orthogonal experimental design was adopted to systematically elucidate the influence of filler ratio, wetting time, and silicone oil viscosity on the bonding strength of thermal conductive gels. The filler ratio exerts the most significant influence, followed by silicone oil viscosity and wetting time. Subsequently, the thermal conductivity and thermal resistance of both commercial thermal conductive gels and the as-prepared gels were characterized using the steady-state heat flow method and the double-interface method, respectively. The prepared thermal conductive gel exhibits a thermal conductivity of 3.75 W·m−1·K−1 and a service thermal resistance of 0.611 ℃·W−1, outperforming commercial counterparts and demonstrating promising application potential. This study provides a practical reference for the development and engineering application of high thermal conductivity, low thermal resistance thermal conductive gels.
Keywords:
1. Introduction
2. Materials and Methods
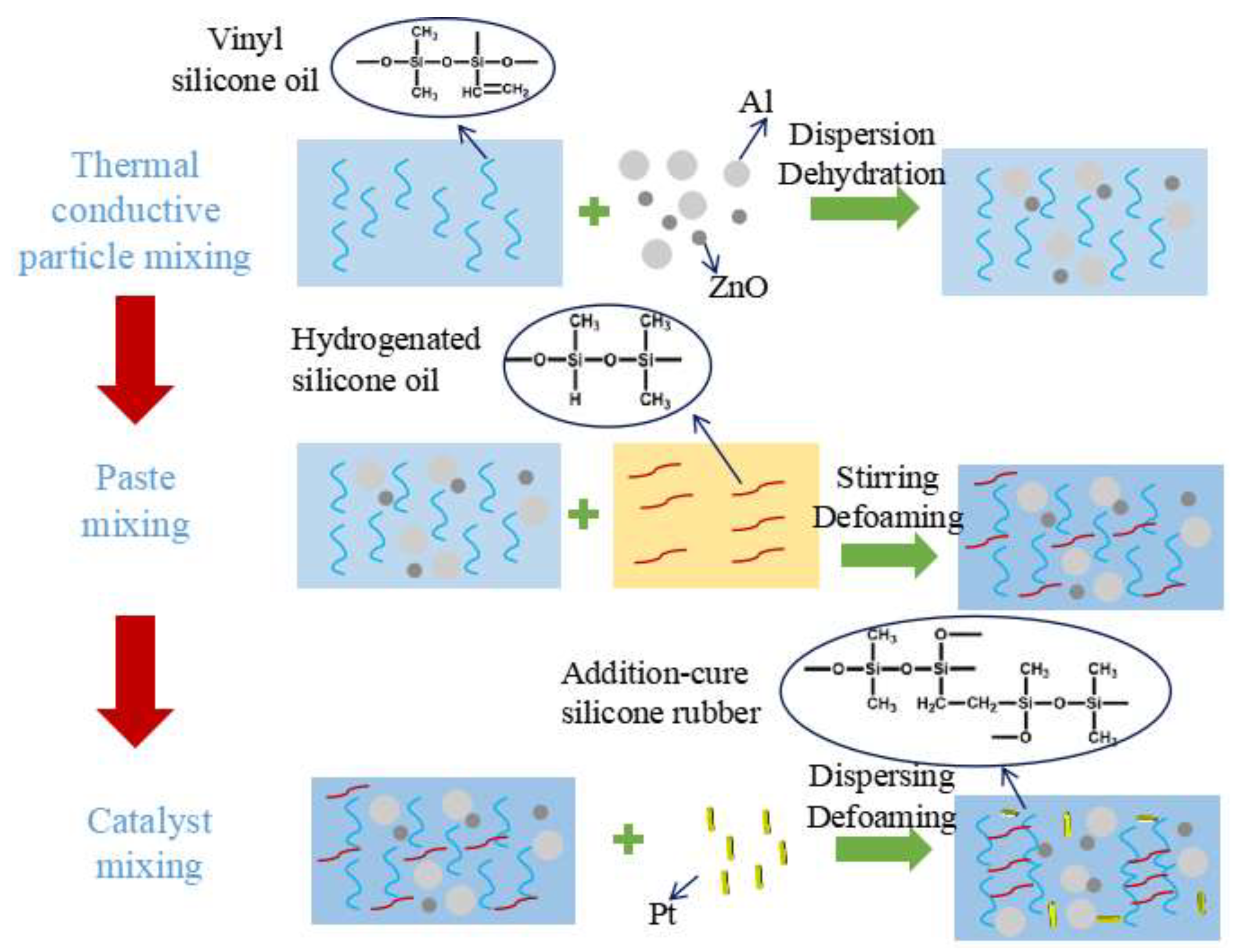
3. Results and Discussion
3.1. Process Optimization and Preparation of Thermal Conductive Gel
3.2. Characterization of Intrinsic Thermal Conductivity of Thermal Conductive Gels
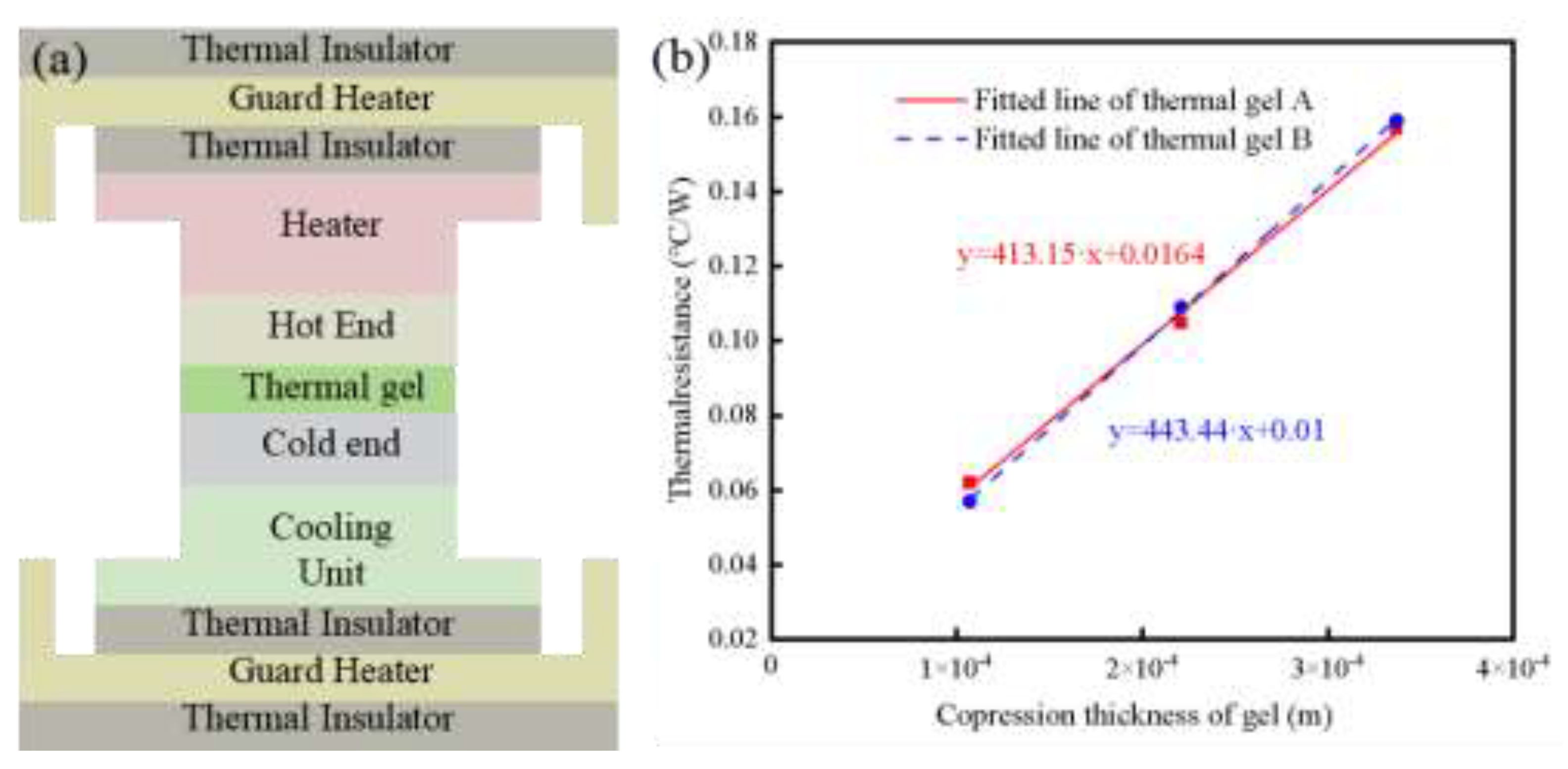
3.3. Thermal Resistance Test of Thermal Conductive Gel in Service
3.4. Analysis of Heat Transfer Mechanism of Thermal Conductive Gel
4. Conclusions
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Feng, C.; Yang, L.; Yang, J.; et al. Recent advances in polymer-based thermal interface materials for thermal management: A mini-review. Compos. Commun. 2020, 22, 100528. [Google Scholar] [CrossRef]
- Rahru, R.; Faiz, M.; Halim, A.; et al. A review of thermal interface material fabrication method toward enhancing heat dissipation. Energy Res. 2021, 45, 3548–3568. [Google Scholar]
- Dhumal, A.R.; Kulkarni, A.P.; Ambhore, N.H. A comprehensive review on thermal management of electronic devices. J. Eng. Appl. Sci. 2023, 70, 140. [Google Scholar] [CrossRef]
- Chakarvarti, S.K.; Gehlawat, D.; Manocha, A. Thermal interface materials for thermal management of microelectronic devices: A review. J. Therm. Anal. Calorim. 2025, 150, 8847–8860. [Google Scholar] [CrossRef]
- Cheng, Z.; Han, Y.; Zhang, X. Investigation of the thermal conductance of MEMS contact switches. Micromachines 2025, 16, 120. [Google Scholar] [CrossRef]
- Susani, L.B.; Coda, F.; Santomauro, M. Reliability of thermal interface materials in combined mechanical and thermal stress. In Proceedings of the 2023 22nd IEEE Intersociety Conference on Thermal and Thermomechanical Phenomena in Electronic Systems (ITherm), 2023; pp. 1–7.
- Asif, M.; Husain, S.; Rehman, S.; et al. Experimental studies on selected thermal interface materials. Therm. Sci. 2024, 28, 2857–2866. [Google Scholar] [CrossRef]
- Pedram, M.; Nazarian, S. Thermal modeling, analysis, and management in VLSI circuits: Principles and methods. Proc. IEEE 2006, 94, 1487–1501. [Google Scholar] [CrossRef]
- Singh, P.K.; Singh, A.K.; Singh, V.K. Impact of the pump-out-effect on the thermal long-term behaviour of power electronic modules. Microelectron. Reliab. 2024, 155, 115362. [Google Scholar]
- Lewis, J.S.; Perrier, T.; Barani, Z.; Kargar, F.; Balandin, A.A. Thermal interface materials with graphene fillers: Review of the state of the art and outlook for future applications. Nanotechnol. 2021, 32, 142003. [Google Scholar] [CrossRef]
- Yang, W.; Kim, J.; Lee, H. Improving the thermal conductivity of an epoxy composite with chemically boron nitride-grafted carbon fiber. Compos. B Eng. 2024, 268, 111089. [Google Scholar] [CrossRef]
- Sun, Y.; Ma, Q.; Liang, T.; et al. Optimization of effective thermal conductivity of thermal interface materials based on the genetic algorithm-driven random thermal network model. ACS Appl. Mater. Interfaces 2021, 13, 45050–45058. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Wang, Y.; Liu, Z. Application of power battery under thermal conductive silica gel plate in new energy vehicles. Sci. Rep. 2024, 14, 312. [Google Scholar] [CrossRef]
- Burger, N.; Laachachi, A.; Ferriol, M.; et al. Review of thermal conductivity in composites: Mechanisms, parameters and theory. Prog. Polym. Sci. 2016, 61, 1–28. [Google Scholar] [CrossRef]
- Yang, X.; Zhang, X.; Zhang, T.; et al. Modeling and optimization of thermal interface materials featuring horizontally oriented fillers by flow-field driven strategy. Compos. Part A 2025, 198, 109157–109169. [Google Scholar] [CrossRef]
- Leung, S.N. Thermally conductive polymer composites and nanocomposites: Processing structure property relationships. Compos. Part B 2018, 150, 78–92. [Google Scholar] [CrossRef]
- Zhang, C.; Liu, J.; Sun, R.; et al. Effects of in situ modification of aluminum fillers on the rheological properties and thermal resistance of gel thermal interface materials. IEEE Trans. Compon. Packag. Manuf. Technol. 2022, 12, 1302–1310. [Google Scholar] [CrossRef]
- Tu, Y.; Liu, B.; Yao, G.; et al. A review of advanced thermal interface materials with oriented structures for electronic devices. Electronics 2024, 13, 4287–4314. [Google Scholar] [CrossRef]
- Wang, Z.; Li, J.; Zhang, X. Significantly enhancing the through-plane thermal conductivity of epoxy dielectrics by constructing aramid nanofiber/boron nitride three-dimensional interconnected framework. AIP Adv. 2024, 14, 075332. [Google Scholar]
- Kim, Y.; Choi, H.; Park, S. Fabrication and characterization of Al2O3-siloxane composite thermal pads for thermal interface materials. Polymers 2024, 16, 543. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, Y.; Wu, Z. Promoting thermal conductivity of alumina-based composite materials by systematically incorporating modified graphene oxide. J. Compos. Sci. 2024, 8, 189. [Google Scholar]
- Shinde, S.; Sampath, S. A critical analysis of the tensile adhesion test for thermally sprayed coatings. J. Therm. Spray Technol. 2022, 31, 2247–2279. [Google Scholar] [CrossRef]
- Wang, Y.; Li, X.; Liu, Z.; et al. A novel modeling and analysis of mechanical properties of single-component thermal conductive silica gel. Sci. Rep. 2025, 15, 15163. [Google Scholar] [CrossRef]
- Uddin, M.A.; Alam, M.O.; Chan, Y.C.; Chan, H.P. Adhesion strength and contact resistance of flip chip on flex packages––effect of curing degree of anisotropic conductive film. Microelectron. Reliab. 2004, 44, 505–514. [Google Scholar] [CrossRef]
- Yap, T.F.; et al. Understanding silicone elastomer curing and adhesion for stronger soft devices. Sci. Adv. 2025, 11, 2681. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Singh, S.; Kumar, R. Optimizing thermal conductivity in 3D-printed composites via Taguchi parameter analysis. Int. J. Adv. Manuf. Technol. 2025, 130, 2451–2463. [Google Scholar]
- Sazali, S.B.; Hassan, H.B.; Yusof, N.; Husaini, Y.; Aziz, A.B.A.; Yaakub, T.N.B.T. Optimization of design parameters using Taguchi method for thermal stress analysis in a 3D IC. In Proceedings of the 2024 IEEE 14th Symposium on Computer Applications & Industrial Electronics (ISCAIE), 2024; pp. 1–4.
- Jia, X. The optimization of extrusion process parameters utilizing the Taguchi method. Int. J. Front. Eng. Technol. 2024, 6, 109–114. [Google Scholar] [CrossRef]
- Zhang, C.; Ren, L.; Zeng, X.; et al. Effects of rheological properties on the thermal resistance of gel thermal interface materials. In Proceedings of the 23rd International Conference on Electronic Packaging Technology (ICEPT), 2022.
- Prasher, R.S.; Matayabas, J.C.; et al. Thermal contact resistance of cured gel polymeric thermal interface material. IEEE Trans. Compon. Packag. Technol. 2004, 27, 702–709. [Google Scholar] [CrossRef]
- Tong, X. Advanced Materials for Thermal Management of Electronic Packaging; Springer, 2011. [Google Scholar]
- Yang, J.; Tang, L.; Bao, R.; et al. Largely enhanced thermal conductivity of poly(ethylene glycol)/boron nitride composite phase change materials for solar-thermal-electric energy conversion and storage with very low content of graphene nanoplatelets. Chem. Eng. J. 2017, 315, 481–490. [Google Scholar] [CrossRef]
- Zhang, B.; Lu, X.; Feng, H.; Wang, Y.; Liu, Y.; Mei, Y.-H. Self-oxygenation silver paste for SiC power modules with high thermal dissipation reliability. J. Alloys Compd. 2025, 1038, 182720. [Google Scholar] [CrossRef]
- Alavi, O.; Abdollah, M.; Hooshmand Viki, A. Assessment of thermal network models for estimating IGBT junction temperature of a buck converter. In Proceedings of the 2017 8th Power Electronics, Drive Systems & Technologies Conference (PEDSTC), 2017; pp. 102–107.
- Lan, X.; Zhao, N. Development of a steady state electrothermal cosimulation model of SiC power modules. Int. J. Heat Mass Transf. 2024, 266, 125460. [Google Scholar] [CrossRef]
- Chung, D.D.L. Materials for thermal conduction. Appl. Therm. Eng. 2001, 21, 1593–1605. [Google Scholar] [CrossRef]
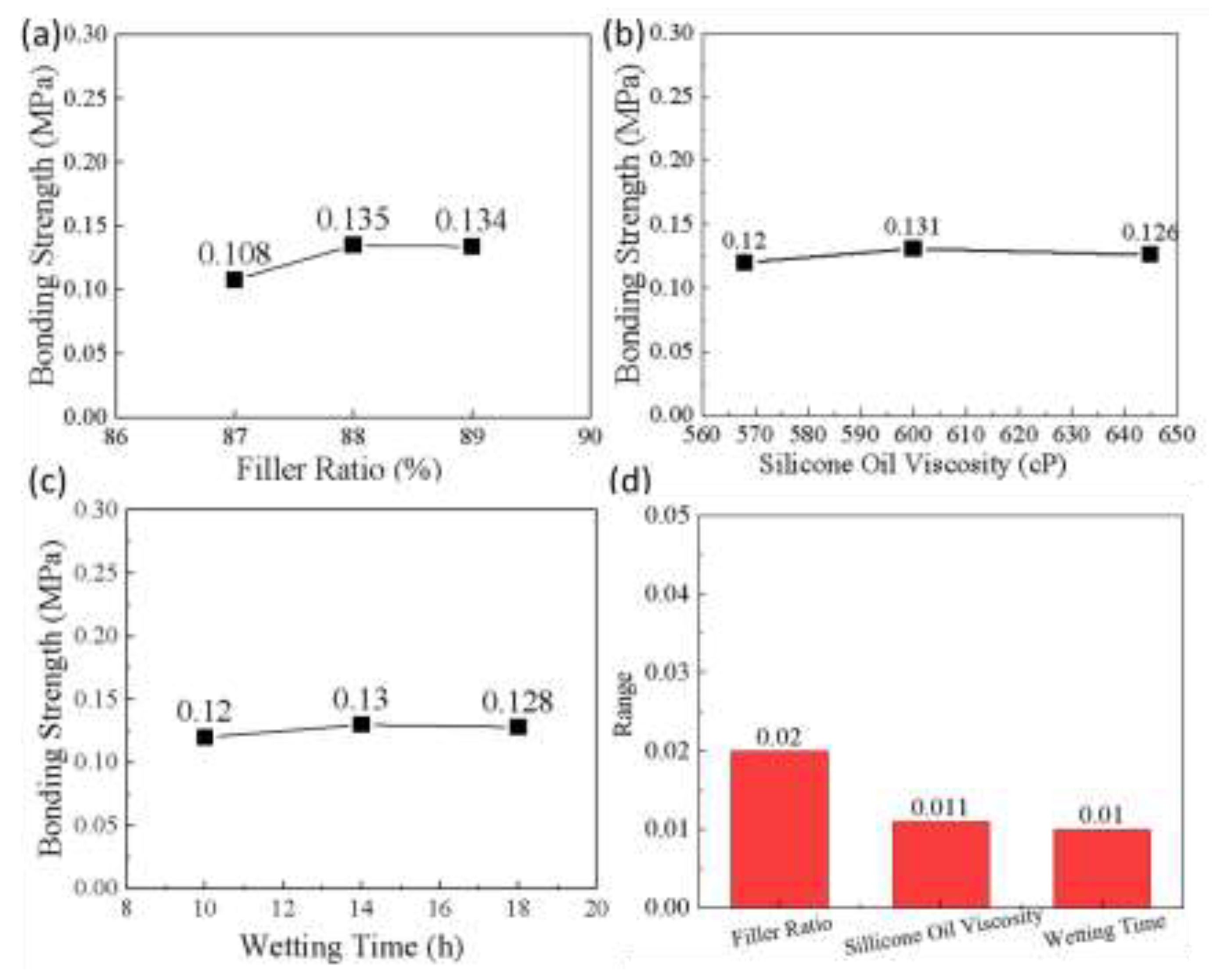

| Serial number | Filler ratio (%) | Silicone oil viscosity (cP) | Wetting time (h) |
bonding strength (MPa) |
|---|---|---|---|---|
| 1 | 88 | 600 | 14 | 0.147 |
| 2 | 88 | 645 | 18 | 0.135 |
| 3 | 88 | 568 | 10 | 0.124 |
| 4 | 89 | 600 | 18 | 0.142 |
| 5 | 89 | 645 | 10 | 0.130 |
| 6 | 89 | 568 | 14 | 0.129 |
| 7 | 87 | 600 | 10 | 0.105 |
| 8 | 87 | 645 | 14 | 0.113 |
| 9 | 87 | 568 | 18 | 0.107 |
| Gel Type | Heat transfer area (mm) | Compression thickness (mm) |
Hot end temperature (℃) |
Cold end temperature (℃) |
Input power (W) |
thermal resistance (℃·W−1) |
|---|---|---|---|---|---|---|
| Thermal conductive gel A | 6.452 | 0.337 | 50 | 45.45 | 28.46 | 0.157 |
| 6.452 | 0.221 | 50 | 46.59 | 30.31 | 0.105 | |
| 6.452 | 0.107 | 50 | 47.89 | 33.82 | 0.062 | |
| Thermal conductive gel B | 6.452 | 0.337 | 50 | 45.82 | 28.61 | 0.159 |
| 6.452 | 0.221 | 50 | 47.10 | 31.22 | 0.109 | |
| 6.452 | 0.107 | 50 | 48.21 | 34.36 | 0.057 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




