1. Introduction
Neuroendocrine tumors (NETs) are heterogeneous, well-differentiated, generally slow-growing epithelial neoplasms that predominantly arise in the gastroenteropancreatic (GEP) and bronchopulmonary (BP) systems. BP NETs include typical carcinoids (mitotic index <2 per 2 mm² and absence of necrosis), atypical carcinoids (mitotic index of 2–10 per 2 mm² with possible necrosis), and high mitotic/Ki-67 variants (morphologically similar to atypical carcinoids but characterized by a mitotic index >10 per 2 mm² and/or Ki-67 >30%) [
1]. Overall, the incidence of NETs is increasing, and diagnosis frequently occurs at a metastatic stage, which is associated with a worse prognosis [
2]. NETs commonly express somatostatin receptors (SSTRs), a feature exploited for both diagnostic and therapeutic purposes. Surgery represents the standard first-line treatment for localized disease [
3], whereas therapeutic options for advanced NETs remain limited and include somatostatin analogues (SSAs) (Octreotide, Lanreotide), peptide receptor radionuclide therapy (PRRT with ^177Lu-DOTATATE), everolimus, and chemotherapy [
4,
5,
6,
7,
8]. Despite these options, validated molecular biomarkers and personalized therapeutic strategies are still lacking, particularly in BP-NETs. The biological complexity of NETs underscores the need to identify novel prognostic factors and therapeutic targets [
9,
10]. SSAs, such as octreotide LAR and lanreotide, are widely used in the management of NETs and exert their effects primarily through activation of somatostatin receptor subtype 2 (SSTR2), resulting in inhibition of proliferative and angiogenic signaling pathways.
There is growing interest in lipid metabolism and metabolic alterations, including metabolic syndrome, obesity, and dyslipidemia, which have already been implicated in the development and progression of several solid tumors [
11,
12]. Lipid metabolism supplies energy substrates and structural phospholipids essential for tumor growth; accordingly, dyslipidemia, characterized by elevated cholesterol and triglyceride levels, may promote angiogenesis, metastatic dissemination, and resistance to anticancer therapies [
13,
14]. Observational studies suggest that dyslipidemia is more prevalent in patients with NETs and has been associated with a worse prognosis [
15,
16]. In this context, increasing attention has been directed toward statins, cholesterol-lowering agents that inhibit 3-hydroxy-3-methylglutaryl–coenzyme A (HMG-CoA) reductase, thereby impairing cholesterol biosynthesis and protein prenylation. Beyond their lipid-lowering activity, statins exert pleiotropic effects, including inhibition of cell proliferation and migration, induction of apoptosis, and modulation of key oncogenic signaling pathways such as PI3K/AKT/mTOR and ERK. Accordingly, several studies have investigated their potential role as anticancer agents [
17,
18,
19,
20,
21].
A retrospective multicenter study including 393 patients with GEP- and BP- NETs reported that dyslipidemia tended to be associated with shorter progression-free survival (PFS); however, among dyslipidemic patients, statins use was associated with a significant improvement in PFS (108 months vs. 26 months in untreated patients), suggesting a possible antitumor role for statins in NETs [
22]. Consistent with these observations, recent studies have explored statins as potential adjuvant agents in antineoplastic therapy across different cancer models [
18]. Numerous preclinical studies have demonstrated the antiproliferative and pro-apoptotic effects of statins, which act on key cellular processes including the cell cycle, lipid metabolism, migration, and intracellular signaling in various tumors, such as breast, prostate, lung, liver, and adrenal NET cell lines [
23,
24,
25,
26,
27,
28]. In parallel, in vitro studies on NET models have investigated combination strategies involving somatostatin analogues (SSAs) and inhibitors of signaling pathways, including PI3K and mTOR [
29]. Pretreatment with these inhibitors enhanced the efficacy of SSAs, in part through reinduction of somatostatin receptor expression [
30]. Additionally, crosstalk between TGF-β signaling and the SST/SSTR pathway has been reported, affecting differentiation, receptor expression, and microRNA regulation, and suggesting a complex regulatory network [
31]. Taken together, these biological findings, together with emerging clinical evidence, support the hypothesis that modulation of lipid metabolism via statins may represent a promising strategy in NETs. Combining statins with SSAs, which are already part of standard therapy, could potentially enhance therapeutic efficacy and counteract resistance, thereby providing a rationale for future dedicated clinical trials.
The present study was designed as a translational proof-of-concept investigation integrating clinical and experimental evidence. Specifically, it aimed to assess the impact of dyslipidemia as well as statin therapy on progression-free survival (PFS) in patients with advanced BP-NETs treated with SSAs. A complementary in vitro study was conducted to provide preliminary insights on the biological interactions between SSAs and statins in BP-NET cell culture.
2. Materials and Methods
2.1. Clinical Study
Data from outpatients with well-differentiated (G1–G3) BP NETs referred to the ENETS center of excellence Sant’Andrea University Hospital of Rome, from January 2010 to November 2025, were retrospectively collected. Inclusion criteria were: 1) histological diagnosis of BP-NET (G1-G3); 2) age ≥ 18 years; 3) advanced disease treated with SSAs as first-line therapy; 4) follow-up time ≥ 12 months from the histological diagnosis; 5) available lipid profile at NET diagnosis.
For each patient, clinicopathological findings were collected, including demographic variables, NET-related characteristics (primary tumour histology, tumour grade, disease stage, anti-tumor therapy, and lipid profile status.
Dyslipidemia was defined by the presence of elevated serum LDL cholesterol and/or triglyceride levels on routine laboratory testing, or by a history of or ongoing treatment with lipid-lowering medications. No predefined time points for lipid measurements were established, with the exception of baseline assessment. The Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1 criteria were used to assess disease progression. Progression-free survival (PFS) was used as the primary outcome, whereas overall survival (OS) was not evaluated due to the limited number of death events expected. Data regarding lipid-lowering therapies, with main focus on statins, but also ezetimibe, bile acid sequestrants, fibrates, or supplements (omega-3 fatty acids, fermented red rice), and their dosages were also collected.
The study was approved by the Sapienza University Ethic Committee (Reference number 6648/2022) and conducted in accordance with the Declaration of Helsinki. All patients provided written informed consent to data collection.
2.2. In Vitro Study
Cell Cultures and Treatments
Lung NET cell line of typical carcinoid NCI-H727, which was purchased from the American Type Culture Collection (ATCC), was cultured in RPMI-1640 culture medium (Sigma Aldrich) supplemented with 10% heat-inactivated fetal bovine serum (FBS) (Invitrogen), 100 μg/ml streptomycin and 100 U/ml penicillin (Gibco) and incubated at 37°C, 5% CO2 in a sterile biosafety hood environment, and medium was changed every 48-h. Approximately 78.000 cells/cm2 were plated. After 24 hours, the cells reached approximately 70-80% confluence and were treated with atorvastatin, lanreotide and the combination treatment at different concentrations for 48 and 72 hours. atorvastatin Calcium (SML3030, Sigma Aldrich) is a hydrophobic substance, which is dissolved in organic solvents such as Dimethyl sulfoxide (DMSO). DMSO (Sigma Aldrich) has a lower final concentration (less than 0.1% in RPMI medium), thus we used it as an atorvastatin solvent. Atorvastatin was diluted with RPMI at the final concentration of 10μM. Lanreotide Acetate (SML0132, Sigma Aldrich) is a hydrophobic substance, which is dissolved in organic solvents such as Dimethyl sulfoxide (DMSO). DMSO (Sigma Aldrich) has a lower final concentration (less than 0.1% in RPMI medium), thus we used it as a lanreotide solvent. Lanreotide was diluted with RPMI at the final concentration of 5μM and 10μM. The combination treatment consisted of both atorvastatin 10μM and lanreotide 5/10 μM. atorvastatin was used at a concentration selected based on previously reported IC₅₀ values in cancer cell lines [
32]. Lanreotide was tested at two concentrations, chosen according to our internal preliminary experiments to assess potential dose-dependent effects. Cells were treated for 48 h or 72 with either single drugs or their combination, as indicated in each experiment.
2.3. Viability Assay
We utilized the MTT colorimetric protocol to assay the mitochondrial respiration rate of viable cells with and without treatment. The MTT(3-(4,5-Dimethylthiazol-2-yl)- 2,5-diphenyltetrazolium bromide) (Sigma Aldrich) was dissolved in PBS at a final concentration of 0.5 mg/ml. After two-day-incubation, we removed the medium, and 100 μl MTT reagents were added to each well and left for three hrs at 37°C in a CO2 incubator. After removing the MTT reagent, the colored Formazan product was dissolved in 150 μl DMSO and plates were agitated for 20 min at room temperature for fully dissolving the MTT product. We measured the optical density (OD) of products at a maximum absorbance wavelength of 560 nm with a micro-plate reader (GloMax, Promega). Results are presented as the log₂ fold change of the mean optical density (O.D.) relative to the control.
2.4. Proliferation Assay
Cell proliferation was assessed using the EdU incorporation assay and Ki-67 immunofluorescence (IF). EdU, a thymidine nucleoside analog, is incorporated into newly synthesized DNA during cell proliferation and is detected as green nuclear staining. Cells were cultured and treated as previously described for 48 hours and incubated with 10 μM EdU for the last 24 hours. Cells were fixed with 4% paraformaldehyde, and EdU incorporation was detected using the Click-iT™ Plus EdU Imaging Kit (Invitrogen, #C10337) according to the manufacturer’s instructions. Ki-67 immunofluorescence staining was subsequently performed by blocking cells with 3% BSA in PBS for 30 min, followed by incubation with the anti-Ki-67 antibody (DAKO, M7240; 1:100 dilution) overnight at 4 °C and then with the appropriate fluorescent secondary antibody (Alexa Fluor™ 594, Invitrogen, #A-11005). Nuclei were counterstained with 1X Hoechst33342 solution for 10 min, followed by three washes. Images were acquired using Zeiss Axiovert 200M with a 20X air objective and analyzed with ImageJ software.
2.5. Cytotoxicity Assay
NCI-H727 cells were cultured in ibidi chambers at 7,8 x10^4 cells /cm2 and treated as previously described for 48 hours. After treatment, the cells were twice washed with PBS and stained with 2μM Calcein AM and 4μM EthD-1 (LIVE/DEAD viability/Cytotoxicity kit, Invitrogen ## L3224), diluted in PBS for 20 min at 37 °C. The fluorescence was examined according to the manufacturer’s instructions.
2.6.Western Blot
Total protein extracts from cells were obtained in RIPA buffer (150 mM NaCl, 50 μM Tris-HCl pH 8.0, 1% NP40, 0.5% Sodium deoxycholate, 0.1% SDS) containing fresh proteases and phosphatases inhibitors cocktail and 0.005 M Na3VO4 and 0.05 M NaF. Cell lysates were centrifuged at 16.200 xg for 30’ and the supernatants were separated by SDS-PAGE and blotted into the nitrocellulose membrane. Membranes were blocked with 5% BSA and incubated with primary and secondary antibodies at the appropriate dilutions. Primary Abs were as follows: Cleaved Caspase-3 (Asp175) Antibody #9661, Invitrogen; Phospho-Histone H2A.X (Ser139) (20E3) #9718, Invitrogen; Vinculin #sc-73614 Santa Cruz. Immunoreactive bands were visualized using WesternBright ECL HRP substrate.
2.7. Seahorse Metabolic Assay
Bioenergetic changes following treatment of the NCI-H727 cell line were evaluated using the Seahorse XF 96 Cell Culture Microplate analyzer (Agilent Technologies, Santa Clara, CA, USA), which allows real-time analysis of oxygen consumption rates (OCR) and extracellular acidification rates (ECAR) in living cells. The cells were plated at a density of 3.5 x 104 per well. Following 48 hours of treatment, the wells were washed with XF BASE culture medium (Seahorse Biosciences) supplemented with 2 mM L-glutamine, 11 mM glucose, and 1.2 mM pyruvate for Atp Rate Assay tests, adjusted to pH 7.35, and then incubated for 30 minutes at 37 °C in a CO2-free incubator before reading. The XF Atp Rate kits were purchased from Seahorse Biosciences and used according to the manufacturer's instructions. OCR and ECAR were measured after sequential injection of oligomycin (2.5 μM) and a mixture of antimycin A (14 μM) and rotenone (14 μM) (all reagents from Merck KGaA, Germany). All values were normalized with respect to viability. The data were analyzed using dedicated software (XF Wave version 2.6.4, Agilent Technologies, CA, USA).
2.8. Statistical Analysis
Regarding the clinical study, descriptive statistics were summarized using the median for continuous variables and frequencies for categorical variables. Patients were classified according to the presence or absence of dyslipidemia and further stratified based on ongoing pharmacological treatment with statins. Comparisons between groups (dyslipidemic vs. non-dyslipidemic and statin-treated vs. untreated patients) were performed using the χ² test or Fisher’s exact test, as appropriate, for categorical variables. Clinical outcome was assessed in terms of PFS, defined as the interval from initiation of SSA therapy to disease progression, evaluated according to routine clinical practice at the time of diagnosis, or to the last follow-up visit, death from any cause, or loss to follow-up. Survival analyses were conducted using the Kaplan–Meier method, and median survival estimates were reported. Regarding the in vitro study, statistical significance was calculated using Student's t-test, Mann-Whitney's U test, or Wilcoxon's test in the calculation of means and standard deviations from three or more independent analyses. In vitro results were expressed as means ± standard deviation (SD) of biological replicates. The significance of differences between means was calculated using unpaired T-test. Results were considered significant at p < 0.05. Graphs and statistical analyses were performed using Microsoft Excel (Microsoft, United States), GraphPad Prism 6 (GraphPad Software, United States), Image J (NIH, United States) and IBM SPSS Statistics (IBM, United States).
3. Results
3.1. Clinical Investigation
3.1.1. Patients’ Characteristics
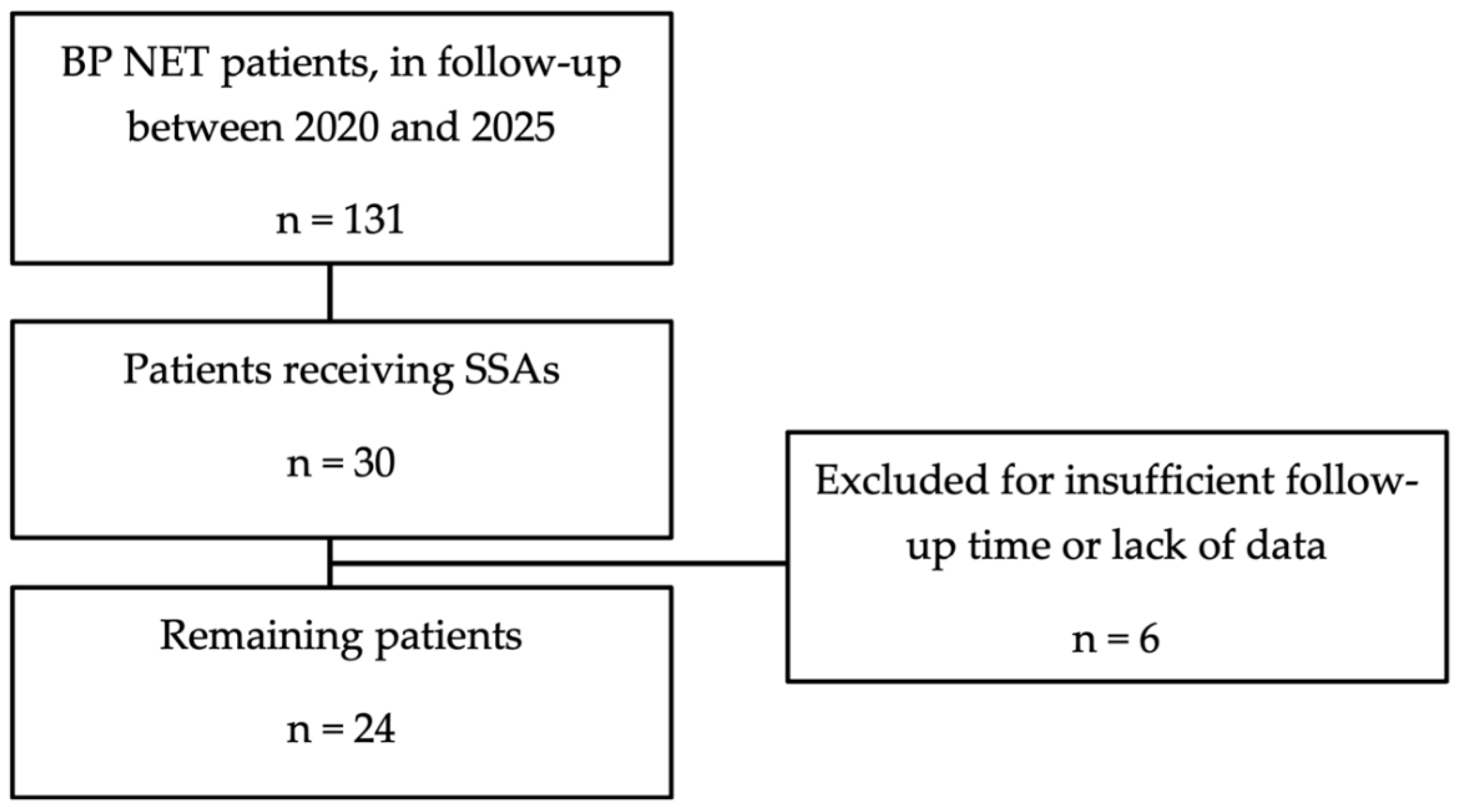
We analysed data from a total of 131 patients; among these, 30 patients (22.9%) were treated with somatostatin analogues (SSAs) (
Figure 1). Six patients were subsequently excluded due to insufficient data or too short follow-up time.
Among the 24 patients considered for the study, 14 (58.3%) were dyslipidemic and 10 (41.7%) had normal lipid profile. Eleven of the 14 dyslipidemic patients were receiving statin therapy: 1 patient was treated with simvastatin, 6 with atorvastatin, and 4 with rosuvastatin. In addition, 2 dyslipidemic patients were receiving ezetimibe, 2 were taking dietary supplements, and 1 was receiving both; these treatments were not mutually exclusive with statin therapy. One dyslipidemic patient was not receiving any lipid-lowering treatment.
Women represented 70.8% of the population, while men were 29.2%. Three patients were affected by clinical or biochemical carcinoid syndrome.
The disease was loco-regional in 35% of cases, whereas distant metastases were present in 65%; no significant difference in disease extent was observed between the two subgroups of dyslipidemic and non-dyslipidemic patients (p = 0.377). The majority of patients showed low to intermediate proliferative activity, with Ki-67 index ≤ 3% in 7/21 patients (mean value 1.71%, 95% CI: 1.02–2.41) and between 4% and 20% in 13/21 patients (mean value 9.62%, 95% CI: 6.43–12.80). Only one patient showed a Ki-67 index ≥ 21%. Data on Ki-67 index were not available in 3 patients (
Table 1).
3.1.2. Clinical Outcome
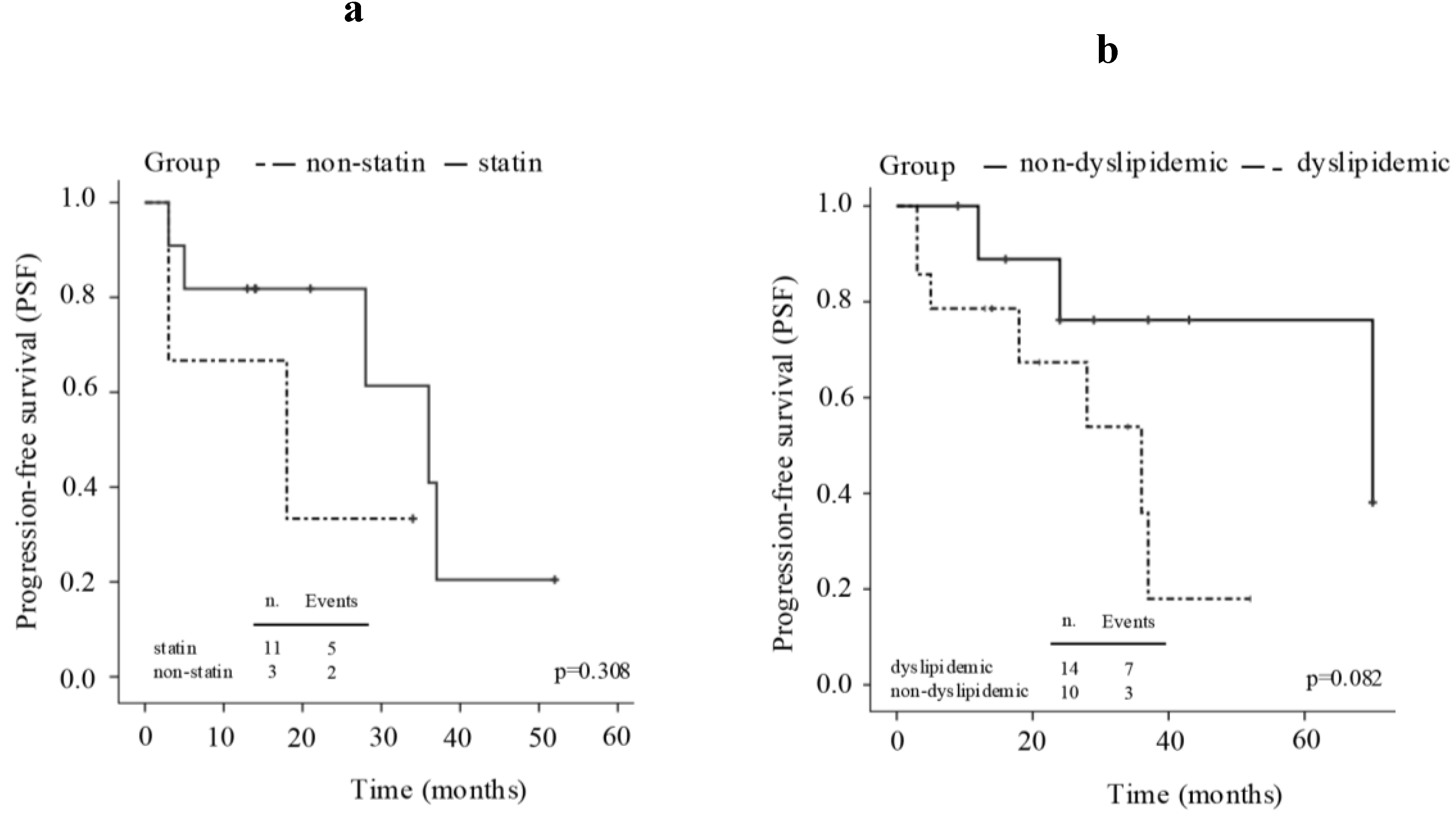
Among the 24 patients treated with SSAs, the median PFS (mPFS) was 22.5 months (95% CI: 18.32-34.01).
The mPFS showed a trend toward longer duration in patients without dyslipidemia compared with those with dyslipidemia (70 months, 95% CI: 3.8-136.1 vs 36 months, 95% CI: 15.4-56.5, p=0.08) (
Figure 2).
A similar trend was observed between patients receiving statin therapy and those without statins (mPFS 36 months, 95% CI 19.7 - 52.3, vs 18 months, 95% CI 0.0 - 42.0, p=0.30), (
Figure 2).
3.2. In Vitro Investigations
3.2.1. Clinical Outcome Combined Lanreotide-Atorvastatin Treatment Reduces Cell Viability
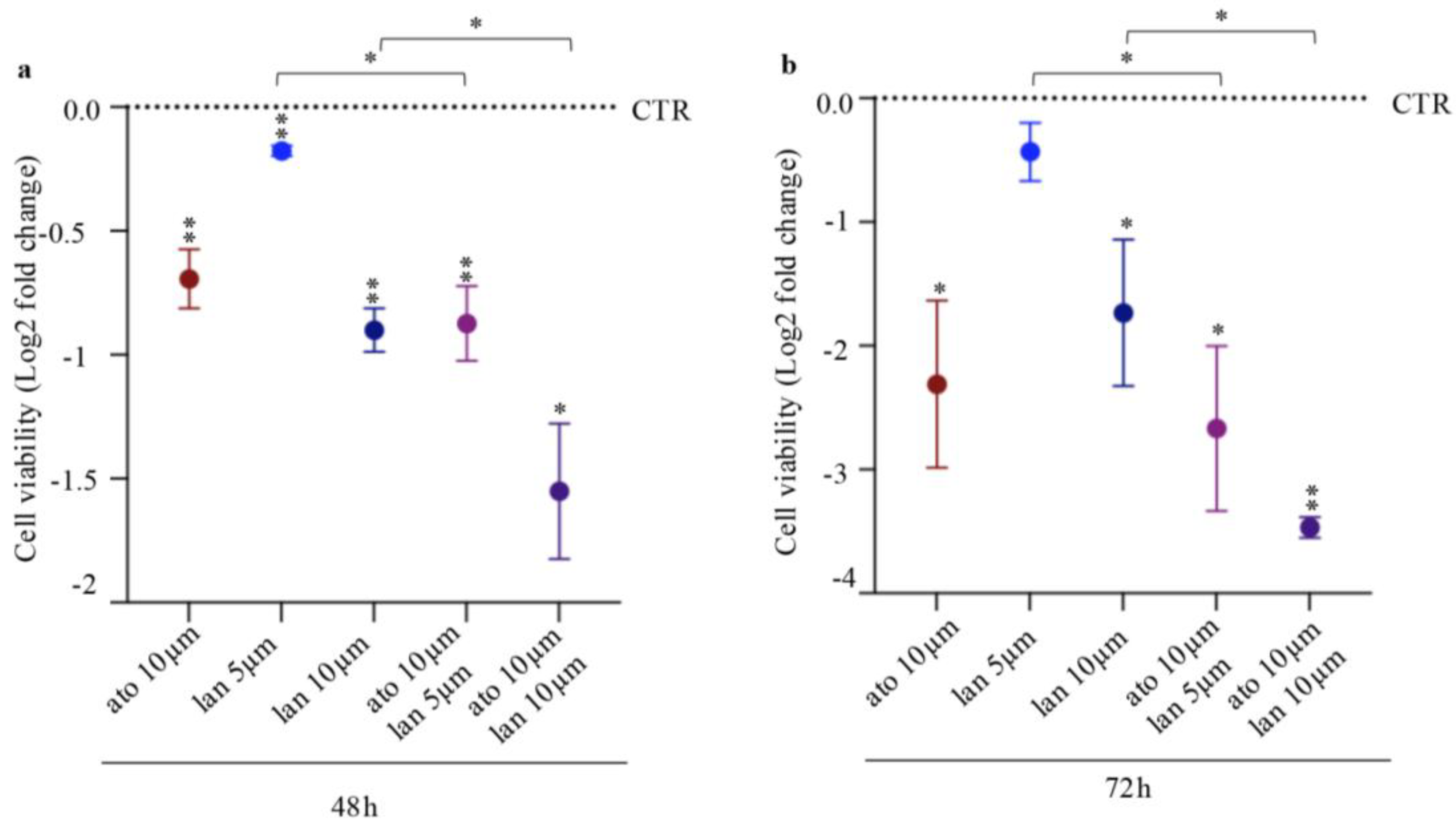
The effects of combined atorvastatin and lanreotide treatment (hereafter referred to as ato and lan) on cell viability were assessed using the MTT assay. NCI-H727 cells were treated with ato (0 or 10 µM), lan (0, 5, or 10 µM), or their combination. After 48 and 72 hours, all treatments, both single agents and combinations, significantly reduced cell viability compared with the control (Fig. 3a, b, Suppl.
Table 1). Notably, combination treatments resulted in a significantly greater reduction in cell viability than lan alone. In particular, the combined treatment with ato and lan exerted a markedly enhanced inhibitory effect at 72 hours (Fig. 3b, Suppl.
Table 1). Overall, these findings indicate that the combination of atorvastatin and lanreotide reduces cell viability in a dose- and time-dependent manner. To further investigate the mechanisms underlying this effect, subsequent analyses focused on cell proliferation, cell death, and cellular metabolic activity.
Figure 3.
Combined lanreotide-atorvastatin treatment reduces cell viability. Dot plot showing cell viability values expressed as log2 fold change relative to the control following the indicated treatments at 48 h (a) and 72 h (b). The horizontal segmented line (= 0) indicates the control (DMSO). Data represent the mean (± SD) of three independent biological replicates (** 0.001 < p < 0.01; * 0.01 < p < 0.05).
Figure 3.
Combined lanreotide-atorvastatin treatment reduces cell viability. Dot plot showing cell viability values expressed as log2 fold change relative to the control following the indicated treatments at 48 h (a) and 72 h (b). The horizontal segmented line (= 0) indicates the control (DMSO). Data represent the mean (± SD) of three independent biological replicates (** 0.001 < p < 0.01; * 0.01 < p < 0.05).
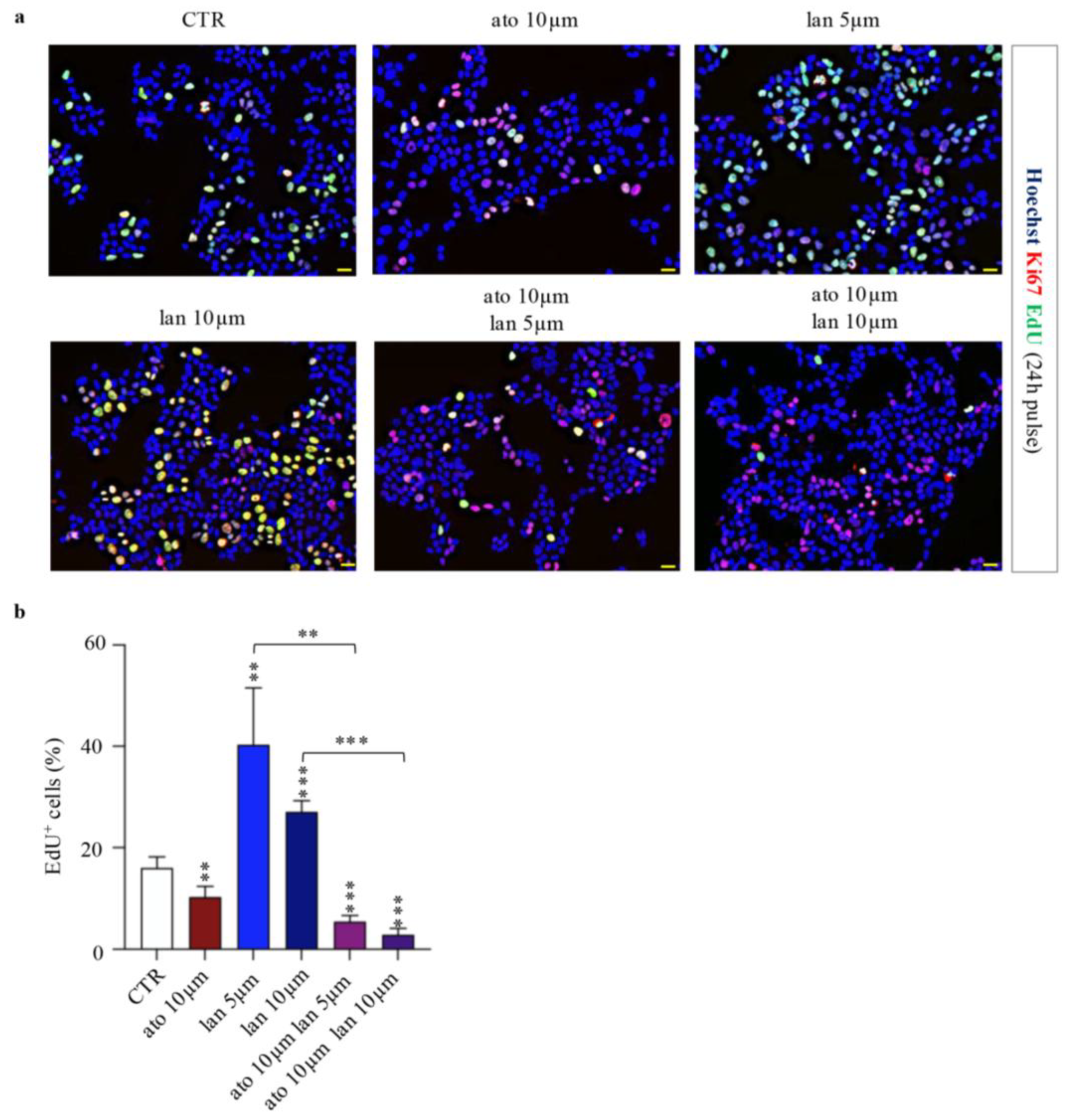
3.2.2. Reduced Proliferation in combined Lanreotide-Atorvastatin Treated Cells
Cell proliferation was evaluated using Ki-67 immunofluorescence (IF) and EdU incorporation assay. After 48 hours, Ki-67 expression in cells treated with both ato and lan showed no significant changes (Fig. 5a; Supplementary Fig. 1). In contrast, EdU incorporation during the final 24 hours revealed a significant decrease in the proportion of cells progressing through S-phase in both combination treatments compared with lan alone (with reductions of 35% for ato + lan 5 µM and 24% for ato + lan 10 µM) and the control (Fig. 4a, b). Interestingly, treatment with lan alone (5 and 10 µM) resulted in increased EdU incorporation, suggesting a potential compensatory proliferative response of tumor cells (Fig. 4a, b).
Figure 4.
Reduced proliferation in combined lanreotide-atorvastatin treated cells. a) Representative IF images of Ki-67 (red) and EdU (green) in NCI-H727 cells treated for 48 h with ato and lan and their combination at the indicated doses. EdU was pulsed during the last 24 h. Nuclei are stained with Hoechst (blue). Scale bar: 20 µm. b) Bar plot showing the percentage of EdU-positive cells treated as in (a). Data are presented as mean ± SD (*** p < 0.001; ** 0.001 < p < 0.01).
Figure 4.
Reduced proliferation in combined lanreotide-atorvastatin treated cells. a) Representative IF images of Ki-67 (red) and EdU (green) in NCI-H727 cells treated for 48 h with ato and lan and their combination at the indicated doses. EdU was pulsed during the last 24 h. Nuclei are stained with Hoechst (blue). Scale bar: 20 µm. b) Bar plot showing the percentage of EdU-positive cells treated as in (a). Data are presented as mean ± SD (*** p < 0.001; ** 0.001 < p < 0.01).
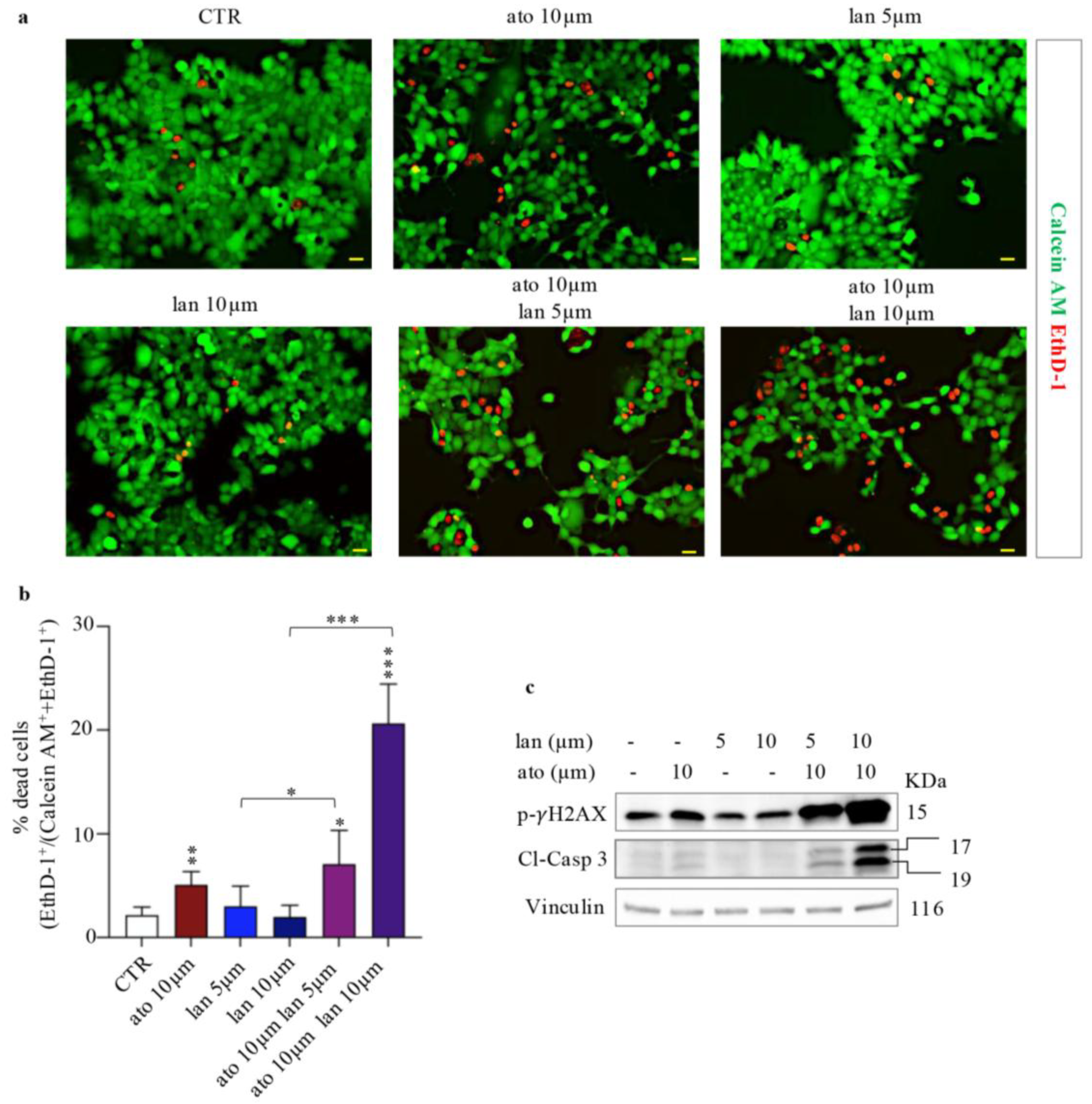
3.2.3. Enhanced Cell Death in Combined Lanreotide-Atorvastatin Treated Cells
To evaluate in vitro toxicity, NCI-H727 cells were treated as described above for 48 hours, and cell viability was assessed using the LIVE/DEAD assay. The results demonstrated a significant dose-dependent increase in the percentage of dead cells, with combination treatments exhibiting a more pronounced cytotoxic effect compared with individual treatments, with a twofold increase for ato + lan at 5 µM and a tenfold increase for ato + lan at 10 µM (Fig. 5a, b).
Furthermore, the potential effect of the combined treatment on cell death was assessed by Western blot. As shown in
Figure 6c, cleaved Caspase-3 levels were increased in cells treated with the drug combination compared to single treatments and control, suggesting enhanced apoptosis. Concomitantly, p-γH2AX levels were elevated, indicative of DNA damage, likely associated with treatment-induced cellular stress. These results suggest that the combined treatment with atorvastatin and lanreotide, particularly at the higher concentration of lanreotide, induces both DNA damage and activation of the apoptotic pathway (Fig. 5c).
Figure 5.
Enhanced cell death in combined lanreotide-atorvastatin treated cells. a) Fluorescence imaging of Calcein AM (green = live cells) and EthD-1 (red = dead cells) in NCI-H727 cells treated for 48 h with ato, lan and their combination at the indicated doses. Scale bar: 20 μm. b) Bar plot showing the percentage of dead cells (EthD-1-positive cells / total cells) from treatments as in (a). Data are presented as mean ± SD (*** p < 0.001; ** 0.001 < p < 0.01; * 0.01 < p < 0.05). c) Western blot analysis of the indicated proteins in NCI-H727 cells treated as in (a). Vinculin was used as a loading control.
Figure 5.
Enhanced cell death in combined lanreotide-atorvastatin treated cells. a) Fluorescence imaging of Calcein AM (green = live cells) and EthD-1 (red = dead cells) in NCI-H727 cells treated for 48 h with ato, lan and their combination at the indicated doses. Scale bar: 20 μm. b) Bar plot showing the percentage of dead cells (EthD-1-positive cells / total cells) from treatments as in (a). Data are presented as mean ± SD (*** p < 0.001; ** 0.001 < p < 0.01; * 0.01 < p < 0.05). c) Western blot analysis of the indicated proteins in NCI-H727 cells treated as in (a). Vinculin was used as a loading control.
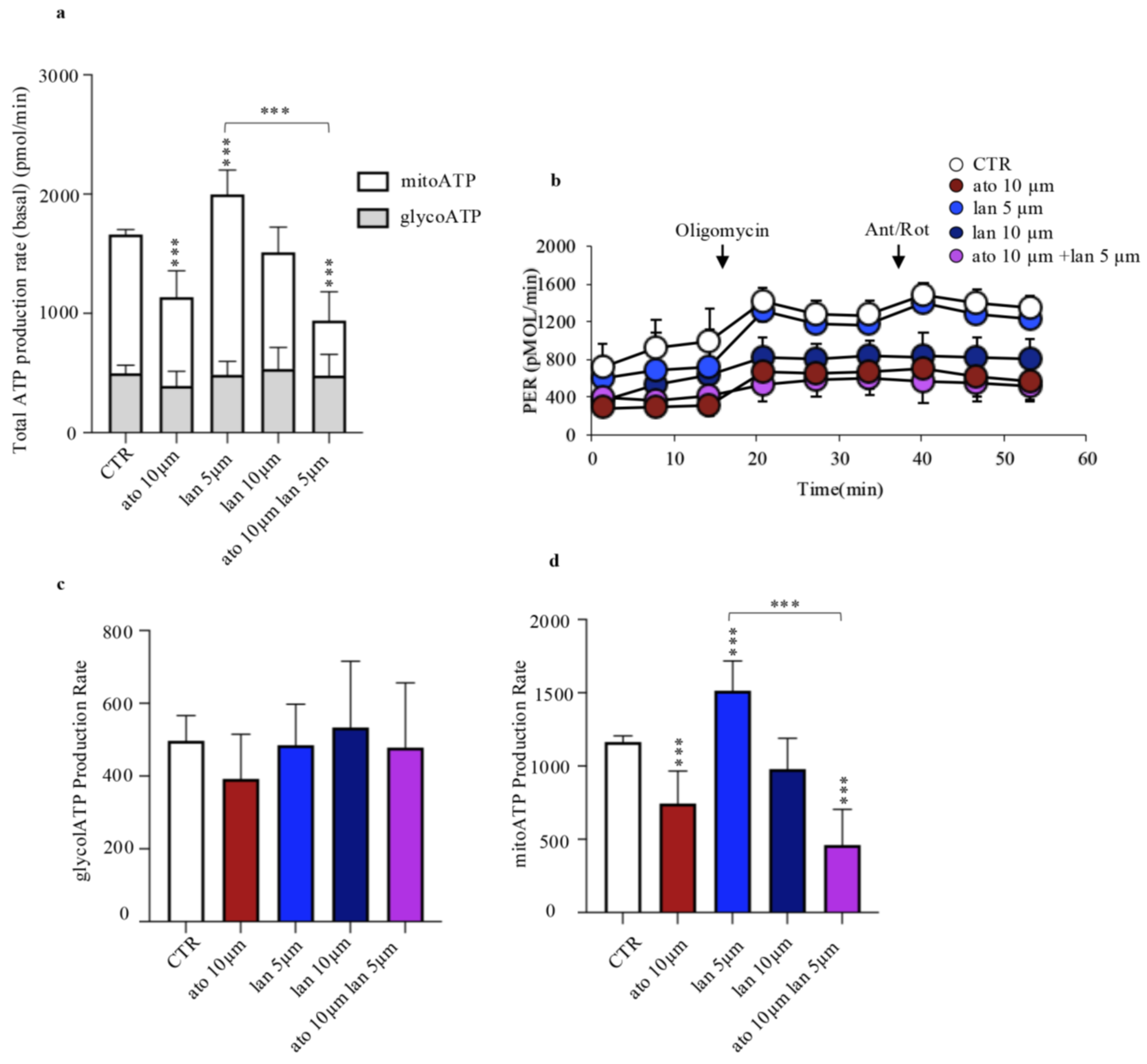
3.2.4. Impact of Lanreotide-Atorvastatin Treatment on Mitochondrial and Glycolytic ATP Production
The NCI-H727 cell line was treated for 48 hours with ato (10 µM), lan (5 µM and 10 µM) and their respective combinations. After incubation, an ATP rate measurement test was performed to quantify ATP production per unit time, distinguishing between the contributions of oxidative phosphorylation and glycolysis. Treatment with ato (10 µM) resulted in a statistically significant reduction in total ATP production compared to the control (mean total ATP value 1660.8 ± 99.9 pMOl/min in the control vs. 1137.2 ± 315.1 pMOl/min min with 10 µM ato) (fig.7a), mainly due to a decrease in mitochondrial respiration (mitoATP levels decreased from 1163.6 ± 41.5 pMOL/min in the control to 744.9 ± 220.5 pMOL/min with 10 µM atorvastatin) (fig.6d).
In contrast, exposure to 5 µM lan significantly enhanced total ATP levels compared to the control (total ATP 1998.5 ± 250.7 pMOl/min) caused by an increase of mitochondrial respiration (fig.6a). The combination of ato (10 µM) and lan (5 µM) had a similar effect to that of ato alone, resulting in a statistically significant reduction in total ATP production (total ATP 938.2 ± 341.1 pMOL/min), compared to the control and to lan (5 µM), determined by the mitochondrial contribution (mitoATP levels decreased from 1163.5 ± 41.5 pMOL/min in the control, 1513.72 ± 203.3 pMOL/min in lan (5 µM) to 461.6 ± 241.8 pMOL/min in the combination) (Fig. 6d). Lanreotide (10 µM) did not affect ATP production, and its combination with ato (10 µM) prevented reliable analysis due to extensive cell death.
Figure 6.
Evaluation of ATP production in cells treated with lanreotide and atorvastatin alone and in combination. a) Quantification of total ATP production, expressed as the sum of mitochondrial and glycolytic contributions. b) Representative trend of the Seahorse ATP Rate Assay experiment, as displayed during instrumental acquisition. c) Mitochondrial contribution to ATP production d) Glycolytic contribution to ATP production. Data are expressed as [mean ± SD] and normalized to viability.
Figure 6.
Evaluation of ATP production in cells treated with lanreotide and atorvastatin alone and in combination. a) Quantification of total ATP production, expressed as the sum of mitochondrial and glycolytic contributions. b) Representative trend of the Seahorse ATP Rate Assay experiment, as displayed during instrumental acquisition. c) Mitochondrial contribution to ATP production d) Glycolytic contribution to ATP production. Data are expressed as [mean ± SD] and normalized to viability.
4. Discussion
NETs represent a heterogeneous group of malignancies rapidly increasing in incidence. Although the majority of NETs arise from the gastroenteropancreatic tract, the incidence of bronchopulmonary NETs has also been steadily increasing [
2]. The contribution of potential risk factors to NET development, including familial predisposition, smoking, and metabolic disorders, remains a matter of ongoing debate [
33].
The treatment of localized NETs is primarily surgical, whereas systemic therapies are usually required in more advanced disease stages. In this context, SSAs play a central role, representing the most widely used therapeutic option for gastroenteropancreatic NETs and, more recently, for the control of tumor growth in BP-NETs expressing somatostatin receptors [
34].
Systematic reviews have shown that SSAs not only control hormone-related symptoms in NET patients but also exert antiproliferative activity, slowing tumor growth and stabilizing disease in a subset of patients. In a phase II clinical study, lanreotide demonstrated tumor size stabilization and reduction of biochemical markers in patients with carcinoid tumors, supporting its antitumor potential beyond symptom control [
35,
36]. Mechanistically, SSAs are known to bind to somatostatin receptors (particularly SSTR2 and SSTR5) and inhibit downstream proliferative pathways such as cyclic AMP, MAPK, and insulin-like growth factor signaling, thereby reducing cell proliferation and promoting apoptosis [
35].
Emerging clinical evidence also supports a potential role for statins in NET progression. A recent observational cohort study of patients with gastroenteropancreatic and BP-NETs found that statin use was associated with improved PFS compared to non-users, suggesting an antiproliferative effect of statins in NET patients [
22]. These findings are consistent with the growing recognition of dyslipidemia as a relevant factor in oncology, maybe comparable in clinical impact to diabetes mellitus [
37,
38].
In line with literature, in our cohort of BP-NETs treated with SSAs, dyslipidemia appears to be associated with a less favorable disease course. Dyslipidemic patients showed a shorter PFS, a higher proportion of progression events, and a greater prevalence of advanced-stage disease compared to non-dyslipidemic patients, despite comparable baseline clinicopathological characteristics, including sex distribution and histological subtypes.
Although these differences did not reach statistical significance, a clear trend toward worse outcomes was observed, suggesting that dyslipidemia may represent a marker of increased tumor aggressiveness rather than a simple metabolic comorbidity. Notably, consistently with our in vitro findings, statin use was associated with a prolongation of mPFS, even if without reaching statistical significance.
While combinations of SSAs with targeted therapies such as mTOR inhibitors and antiangiogenic agents have been explored in clinical settings with encouraging disease control and acceptable safety profiles, studies focusing on SSA-statin combinations are lacking [
19].
Although clinical evidence suggests that SSAs are an effective treatment for NETs, preclinical studies show highly variable results [
39,
40,
41]. Indeed, in our study, we observed a reduction of cell viability in vitro, accompanied by a controversial effect on cell proliferation, as reported in the literature, as well as on cell death and metabolism [
40]. We hypothesise that the observed in vitro single-agent effect may reflect a compensatory survival response of tumor cells. Such adaptive responses have been described in previous studies, indicating that tumor cells can activate compensatory mechanisms when exposed to conditions of strong cellular stress [
42]. But we also demonstrate that concomitant treatment with atorvastatin leads to a greater effect on cell viability in vitro associated with reduced proliferation, increased apoptosis/DNA damage and impaired ATP production.
These data indicate that dual targeting of SSTR signaling and cholesterol biosynthesis pathways may enhance antitumor efficacy in NET models. Statins, including atorvastatin, have been increasingly investigated for their antitumor properties across diverse cancer types. Beyond lipid lowering, statins inhibit HMG-CoA reductase, thereby reducing the production of mevalonate intermediates required for prenylation of small GTPases such as Ras and Rho, which play central roles in cell proliferation and survival [
43]. Statins have been shown to reduce proliferation, induce apoptosis, and inhibit angiogenesis in various tumor models, including lung cancer, suggesting potential applicability in NETs where similar proliferative mechanisms operate [
23,
24,
25,
26,
27,
28]. These molecular effects are consistent with our observation of decreased viability and increased apoptosis upon combined treatment.
Metabolic studies have shown that some neuroendocrine cells, including BP ones, are particularly susceptible to drugs that affect their primary energy pathways. For example, Safari M et al. demonstrated how some neuroendocrine cell lines, relying on mitochondrial respiration, were sensitive to the combination of NAMPT and HDAC inhibitors [
44]. Consistent with these findings, in our study, we demonstrated that BP neuroendocrine cells could be particularly sensitive to drugs that interfere with mitochondrial respiration. Indeed, atorvastatin, both alone and especially in combination with lanreotide, significantly impaired mitochondrial ATP production, that associated with an increase in cell death, supported the hypothesis of induced oxidative stress. Limitations of the current study are: first, the in vitro findings were based on a single cell line model, and the lack of formal drug interaction analysis (e.g., combination index) limits conclusions about the nature (additive vs synergistic) of the interaction. Second, considering the in vivo investigation, the retrospective design of the study, the limited sample size, and the relatively small number of events substantially limit the statistical power of the analysis and preclude definitive conclusions. In addition, the restriction of the study population to patients with a disease burden requiring SSA treatment, while reducing clinical heterogeneity, further reduced the number of evaluable cases. While preclinical and observational clinical data suggest potential benefits of statins in NETs, prospective clinical trials are needed to validate the translational potential of combining statins with SSAs.
5. Conclusions
In summary, this study indicates, for the first time, that atorvastatin could potentiate the antitumor effects of lanreotide, providing a compelling preclinical and clinical foundation for further exploration of this combination. Given the established clinical use of both classes of drugs and emerging evidence of their anti-tumour properties, future work should investigate the biological mechanism in more depth in additional NET models and in well-designed clinical trials.
Supplementary Materials
Table S1. Table showing the mean ± SD of cell viability expressed as log2 fold change relative to the control following the indicated treatments. Figure S1: Bar plot showing the percentage of Ki67-positive cells treated for 48 h with ato and lan and their combination at the indicated doses. Data are presented as mean ± SD (*** p < 0.001; ** 0.001 < p < 0.01).
Author Contributions
Conceptualization, A.F., C.M., F.F. and G.P.; methodology, A.F., C.M., F.F., F.R., G.P., R.M. and V.Z.; software C.M., F.F., F.R., G.P., and S.C.; formal analysis, F.F., F.R. and S.C.; investigation, A.Y., C.M., E.P., F.F., G.P., F.R. and S.C.; resources, A.F., F.B., F.P. and M.R.R.; data curation, C.M., F.F., F.R., G.P., R.M. and V.Z.; writing-original draft preparation, C.M., F.F., F.R., G.P., and S.C.; writing-review and editing, A.F., F.B., M.R.R., R.M. and V.Z.; supervision, A.F., F.B., F.P. and M.R.R.; project administration, A.F, C.M. and G.P.; funding acquisition, A.F. and G.P. All authors have read and agreed to the published version of the manuscript.
Funding
This work was funded by Progetto UBUNTU- Codice T3-AN-01-CUP F83C22001410001-PIANO SVILUPPO E COESIONE DEL MINISTERO DELLA SALUTE (FSC 2014-2020).
Institutional Review Board Statement
This study was approved by the Sapienza University Ethic Committee (Reference number 6648/2022) and conducted in accordance with the Declaration of Helsinki.
Informed Consent Statement
All patients provided written informed consent to data collection.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors on request.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| NETs |
Neuroendocrine tumors |
| GEP |
Gastroenteropancreatic |
| BP |
Bronchopulmonary |
| SSTRs |
Linear dichroism |
| PRRT |
Peptide Receptor Radionuclide Therapy |
| PFS |
Progression Free Survival |
| SSAs |
Somatostatin Analogs |
| ATCC |
American Type Culture Collection |
| OCR |
Oxygen Consumption Rates |
| ECAR |
Extracellular Acidification Rates |
| ATO |
atorvastatin |
| LAN |
lanreotide |
References
- Rindi, G; Mete, O; Uccella, S; Basturk, O; La Rosa, S; Brosens, LAA; et al. Overview of the 2022 WHO Classification of Neuroendocrine Neoplasms. Endocr Pathol 2022, 33(1), 115–54. [Google Scholar] [CrossRef]
- Dasari, A; Wallace, K; Halperin, DM; Maxwell, J; Kunz, P; Singh, S; et al. Epidemiology of Neuroendocrine Neoplasms in the US. JAMA Netw Open 2025, 8(6), e2515798. [Google Scholar]
- Davini, F; Gonfiotti, A; Comin, C; Caldarella, A; Mannini, F; Janni, A. Typical and atypical carcinoid tumours: 20-year experience with 89 patients. J Cardiovasc Surg (Torino) 2009, 50(6), 807–11. [Google Scholar]
- Faggiano, A. Long-acting somatostatin analogs and well differentiated neuroendocrine tumors: a 20-year-old story. J Endocrinol Invest. 2023, 47(1), 35–46. [Google Scholar]
- Rinke, A; Müller, H-H; Schade-Brittinger, C; Klose, K-J; Barth, P; Wied, M; et al. Placebo-Controlled, Double-Blind, Prospective, Randomized Study on the Effect of Octreotide LAR in the Control of Tumor Growth in Patients with Metastatic Neuroendocrine Midgut Tumors: A Report From the PROMID Study Group. Journal of Clinical Oncology 2009, 27(28), 4656–63. [Google Scholar] [CrossRef]
- Cives, M; Strosberg, JR. Gastroenteropancreatic Neuroendocrine Tumors. CA Cancer J Clin. 2018, 68(6), 471–87. [Google Scholar]
- Fazio, N; Buzzoni, R; Delle Fave, G; Tesselaar, ME; Wolin, E; Van Cutsem, E; et al. Everolimus in advanced, progressive, well-differentiated, non-functional neuroendocrine tumors: RADIANT -4 lung subgroup analysis. Cancer Sci. 2018, 109(1), 174–81. [Google Scholar] [CrossRef] [PubMed]
- Faggiano, A; Malandrino, P; Modica, R; Agrimi, D; Aversano, M; Bassi, V; et al. Efficacy and Safety of Everolimus in Extrapancreatic Neuroendocrine Tumor: A Comprehensive Review of Literature. Oncologist 2016, 21(7), 875–86. [Google Scholar]
- Abdalla, TSA; Klinkhammer-Schalke, M; Zeissig, SR; Tol, KK; Honselmann, KC; Braun, R; et al. Prognostic factors after resection of locally advanced non-functional pancreatic neuroendocrine neoplasm: an analysis from the German Cancer Registry Group of the Society of German Tumor Centers. J Cancer Res Clin Oncol. 2023, 149(11), 8535–43. [Google Scholar] [CrossRef] [PubMed]
- Marciello, F; Mercier, O; Ferolla, P; Scoazec, J-Y; Filosso, PL; Chapelier, A; et al. Natural History of Localized and Locally Advanced Atypical Lung Carcinoids after Complete Resection: A Joined French Italian Retrospective Multicenter Study. Neuroendocrinology 2018, 106(3), 264–73. [Google Scholar] [CrossRef] [PubMed]
- Vasseur, S; Guillaumond, F. Lipids in cancer: a global view of the contribution of lipid pathways to metastatic formation and treatment resistance. Oncogenesis 2022, 11(1), 46. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F. Dysregulated lipid metabolism in cancer. World J Biol Chem. 2012, 3(8), 167. [Google Scholar]
- Modica, R; La Salvia, A; Liccardi, A; Cozzolino, A; Di Sarno, A; Russo, F; et al. Dyslipidemia, lipid-lowering agents and neuroendocrine neoplasms: new horizons. Endocrine 2024, 85(2), 520–31. [Google Scholar] [CrossRef]
- Modica, R; La Salvia, A; Liccardi, A; Cannavale, G; Minotta, R; Benevento, E; et al. Lipid Metabolism and Homeostasis in Patients with Neuroendocrine Neoplasms: From Risk Factor to Potential Therapeutic Target. Metabolites 2022, 12(11), 1057. [Google Scholar] [CrossRef]
- Gallo, M; Muscogiuri, G; Pizza, G; Ruggeri, RM; Barrea, L; Faggiano, A; et al. The management of neuroendocrine tumours: A nutritional viewpoint. Crit Rev Food Sci Nutr. 2019, 59(7), 1046–57. [Google Scholar]
- Pyo, JH; Hong, SN; Min, B-H; Lee, JH; Chang, DK; Rhee, P-L; et al. Evaluation of the risk factors associated with rectal neuroendocrine tumors: a big data analytic study from a health screening center. J Gastroenterol. 2016, 51(12), 1112–21. [Google Scholar] [CrossRef]
- Duarte, JA; de Barros, ALB; Leite, EA. The potential use of simvastatin for cancer treatment: A review. Biomedicine & Pharmacotherapy 2021, 141, 111858. [Google Scholar] [CrossRef]
- Beckwitt, CH; Brufsky, A; Oltvai, ZN; Wells, A. Statin drugs to reduce breast cancer recurrence and mortality. Breast Cancer Research 2018, 20(1), 144. [Google Scholar] [CrossRef] [PubMed]
- Herrera-Martínez, AD; Pedraza-Arevalo, S; L-López, F; Gahete, MD; Gálvez-Moreno, MA; Castaño, JP; et al. Type 2 Diabetes in Neuroendocrine Tumors: Are Biguanides and Statins Part of the Solution? J Clin Endocrinol Metab. 2019, 104(1), 57–73. [Google Scholar] [CrossRef]
- Pusceddu, S; Vernieri, C; Di Maio, M; Marconcini, R; Spada, F; Massironi, S; et al. Metformin Use Is Associated With Longer Progression-Free Survival of Patients With Diabetes and Pancreatic Neuroendocrine Tumors Receiving Everolimus and/or Somatostatin Analogues. Gastroenterology 2018, 155(2), 479–489.e7. [Google Scholar] [CrossRef] [PubMed]
- Pusceddu, S; Vernieri, C; Prinzi, N; Torchio, M; Coppa, J; Antista, M; et al. The potential role of metformin in the treatment of patients with pancreatic neuroendocrine tumors: a review of preclinical to clinical evidence. Therap Adv Gastroenterol 2020, 13. [Google Scholar] [CrossRef]
- Faggiano, A; Russo, F; Zamponi, V; Sesti, F; Puliani, G; Modica, R; et al. Impact of dyslipidemia and lipid-lowering therapy with statins in patients with neuroendocrine tumors. J Neuroendocrinol. 2025, 37(2). [Google Scholar] [CrossRef]
- Koyuturk, M; Ersoz, M; Altiok, N. Simvastatin induces apoptosis in human breast cancer cells: p53 and estrogen receptor independent pathway requiring signalling through JNK. Cancer Lett. 2007, 250(2), 220–8. [Google Scholar] [CrossRef]
- Bjarnadottir, O; Romero, Q; Bendahl, P-O; Jirström, K; Rydén, L; Loman, N; et al. Targeting HMG-CoA reductase with statins in a window-of-opportunity breast cancer trial. Breast Cancer Res Treat 2013, 138(2), 499–508. [Google Scholar] [CrossRef] [PubMed]
- Nölting, S; Maurer, J; Spöttl, G; Aristizabal Prada, ET; Reuther, C; Young, K; et al. Additive Anti-Tumor Effects of Lovastatin and Everolimus In Vitro through Simultaneous Inhibition of Signaling Pathways. PLoS One 2015, 10(12), e0143830. [Google Scholar]
- Miyazawa, Y; Sekine, Y; Kato, H; Furuya, Y; Koike, H; Suzuki, K. Simvastatin Up-Regulates Annexin A10 That Can Inhibit the Proliferation, Migration, and Invasion in Androgen-Independent Human Prostate Cancer Cells. Prostate 2017, 77(4), 337–49. [Google Scholar]
- Vázquez-Borrego, MC; Fuentes-Fayos, AC; Herrera-Martínez, AD; Venegas-Moreno, E; L-López, F; Fanciulli, A; et al. Statins Directly Regulate Pituitary Cell Function and Exert Antitumor Effects in Pituitary Tumors. Neuroendocrinology 2020, 110(11–12), 1028–41. [Google Scholar] [CrossRef] [PubMed]
- Vernieri, C; Pusceddu, S; Fucà, G; Indelicato, P; Centonze, G; Castagnoli, L; et al. Impact of systemic and tumor lipid metabolism on everolimus efficacy in advanced pancreatic neuroendocrine tumors (pNETs). Int J Cancer 2019, 144(7), 1704–12. [Google Scholar] [CrossRef] [PubMed]
- Krug, S; Mordhorst, J-P; Moser, F; Theuerkorn, K; Ruffert, C; Egidi, M; et al. Correction: Interaction between somatostatin analogues and targeted therapies in neuroendocrine tumor cells. PLoS One 2020, 15(2), e0228905. [Google Scholar]
- von Hessert-Vaudoncourt, C; Lelek, S; Geisler, C; Hartung, T; Bröker, V; Briest, F; et al. Concomitant inhibition of PI3K/mTOR signaling pathways boosts antiproliferative effects of lanreotide in bronchopulmonary neuroendocrine tumor cells. Front Pharmacol 2024, 15. [Google Scholar] [CrossRef]
- Ungefroren, H; Künstner, A; Busch, H; Franzenburg, S; Luley, K; Viol, F; et al. Differential Effects of Somatostatin, Octreotide, and Lanreotide on Neuroendocrine Differentiation and Proliferation in Established and Primary NET Cell Lines: Possible Crosstalk with TGF-β Signaling. Int J Mol Sci. 2022, 23(24), 15868. [Google Scholar] [CrossRef]
- Abolghasemi, R; Ebrahimi-Barough, S; Bahrami, N; Ai, J. Atorvastatin Inhibits Viability and Migration of MCF7 Breast Cancer Cells. Asian Pac J Cancer Prev. 2022, 23(3), 867–75. [Google Scholar] [PubMed]
- Leoncini, E; Carioli, G; La Vecchia, C; Boccia, S; Rindi, G. Risk factors for neuroendocrine neoplasms: a systematic review and meta-analysis. Annals of Oncology 2016, 27(1), 68–81. [Google Scholar]
- Baudin, E; Caplin, M; Garcia-Carbonero, R; Fazio, N; Ferolla, P; Filosso, PL; et al. Lung and thymic carcinoids: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up☆. Annals of Oncology 2021, 32(4), 439–51. [Google Scholar] [CrossRef] [PubMed]
- Sidéris, L; Dubé, P; Rinke, A. Antitumor Effects of Somatostatin Analogs in Neuroendocrine Tumors. Oncologist 2012, 17(6), 747–55. [Google Scholar] [CrossRef] [PubMed]
- Michael, M; Garcia-Carbonero, R; Weber, MM; Lombard-Bohas, C; Toumpanakis, C; Hicks, RJ. The Antiproliferative Role of Lanreotide in Controlling Growth of Neuroendocrine Tumors: A Systematic Review. Oncologist 2017, 22(3), 272–85. [Google Scholar]
- Natalicchio, A; Marrano, N; Montagnani, M; Gallo, M; Faggiano, A; Zatelli, M; et al. Glycemic control and cancer outcomes in oncologic patients with diabetes: an Italian Association of Medical Oncology (AIOM), Italian Association of Medical Diabetologists (AMD), Italian Society of Diabetology (SID), Italian Society of Endocrinology (SIE), Italian Society of Pharmacology (SIF) multidisciplinary critical view. J Endocrinol Invest. 2024, 47(12), 2915–28. [Google Scholar]
- Ben-Shmuel, S; Rostoker, R; Scheinman, EJ; LeRoith, D. Metabolic Syndrome, Type 2 Diabetes, and Cancer: Epidemiology and Potential Mechanisms 2015, pp 355–72.
- Sciammarella, C; Luce, A; Riccardi, F; Mocerino, C; Modica, R; Berretta, M; et al. Lanreotide Induces Cytokine Modulation in Intestinal Neuroendocrine Tumors and Overcomes Resistance to Everolimus. Front Oncol. 2020, 10, 1047. [Google Scholar] [CrossRef]
- Fotouhi, O; Kjellin, H; Larsson, C; Hashemi, J; Barriuso, J; Juhlin, CC; et al. Proteomics Suggests a Role for APC-Survivin in Response to Somatostatin Analog Treatment of Neuroendocrine Tumors. J Clin Endocrinol Metab. 2016, 101(10), 3616–27. [Google Scholar]
- Vitale, G; Lupoli, G; Guarrasi, R; Colao, A; Dicitore, A; Gaudenzi, G; et al. Interleukin-2 and Lanreotide in the Treatment of Medullary Thyroid Cancer: In Vitro and In Vivo Studies. J Clin Endocrinol Metab. 2013, 98(10), E1567–74. [Google Scholar] [CrossRef]
- Friedman, R. Drug resistance in cancer: molecular evolution and compensatory proliferation. Oncotarget 2016, 7(11), 11746–55. [Google Scholar] [CrossRef] [PubMed]
- Amin, F; Fathi, F; Reiner, Ž; Banach, M; Sahebkar, A. The role of statins in lung cancer. Archives of Medical Science 2021. [Google Scholar] [CrossRef] [PubMed]
- Safari, M; Scotto, L; Litman, T; Petrukhin, LA; Zhu, H; Shen, M; et al. Novel Therapeutic Strategies Exploiting the Unique Properties of Neuroendocrine Neoplasms. Cancers (Basel) 2023, 15(20), 4960. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).