Submitted:
01 March 2026
Posted:
02 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Ethical Approval
2.2. Study Design and Setting
2.3. Data Sources and Data Management
2.4. Case Definitions and Meat Inspection Procedures
2.5. Inclusion and Exclusion Criteria
2.6. Statistical Analysis
3. Results
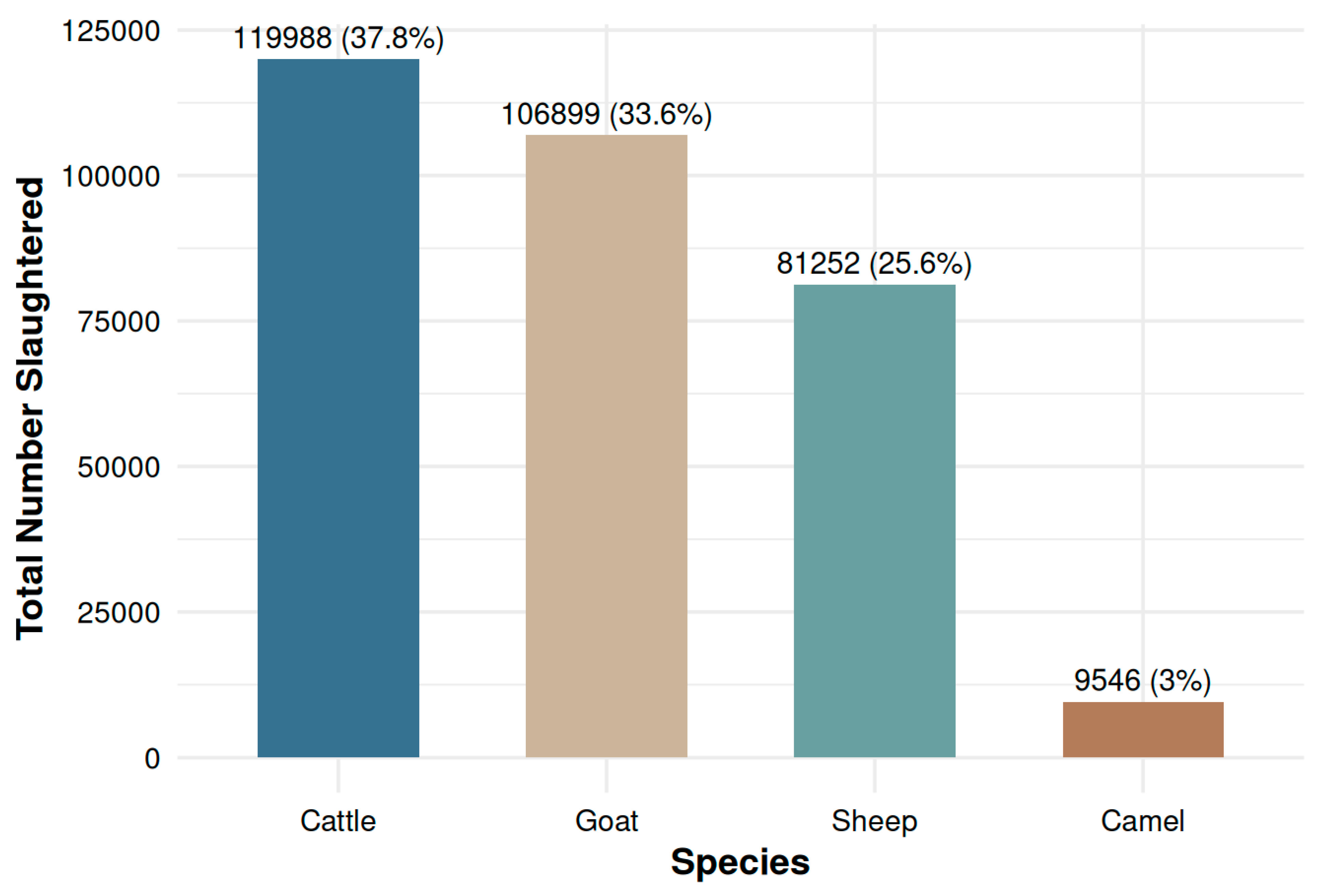
3.1. Slaughter Throughput and Overall Condemnation Burden
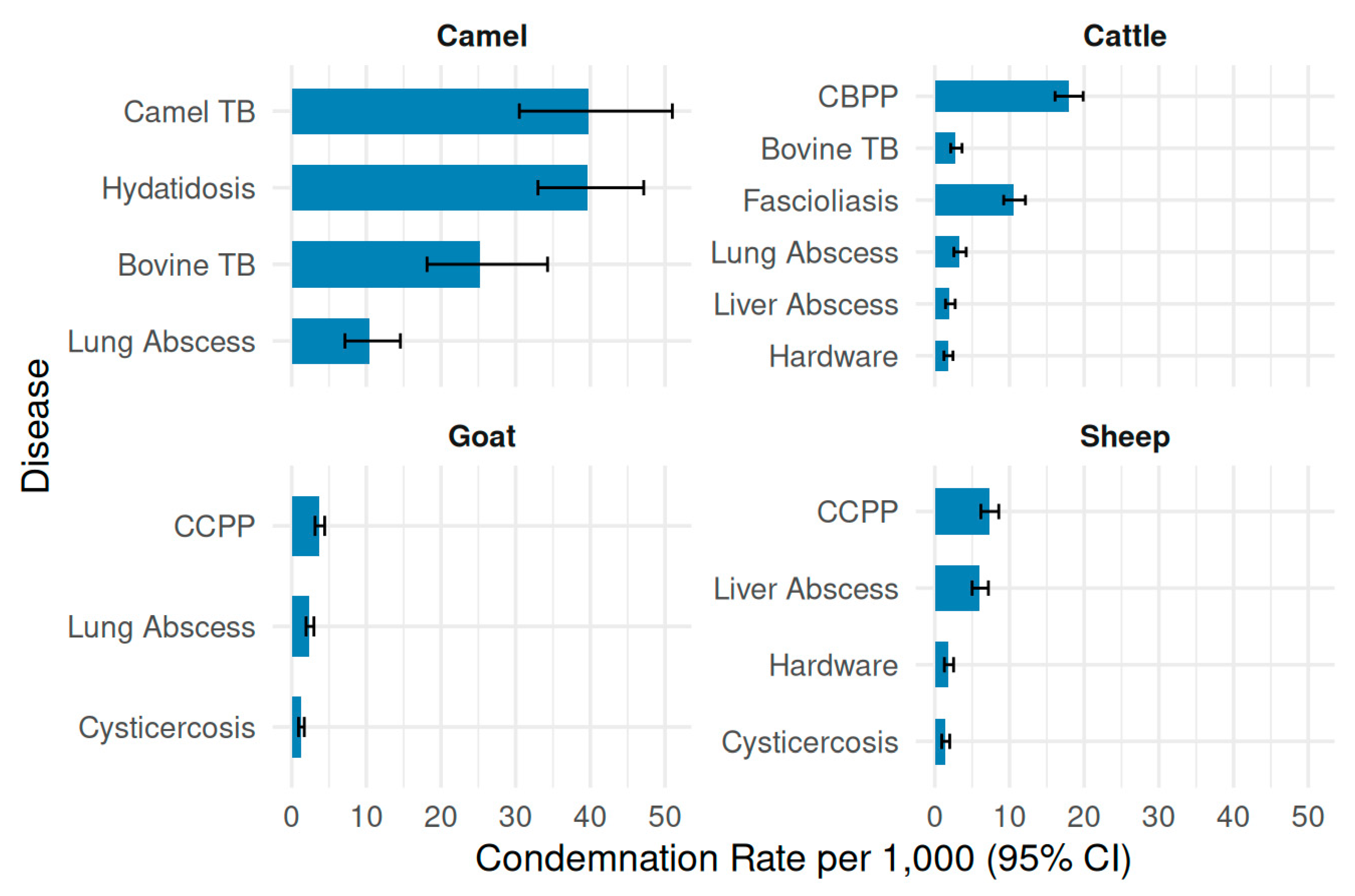
3.2. Distribution of Condemnation Cases by Recorded Cause
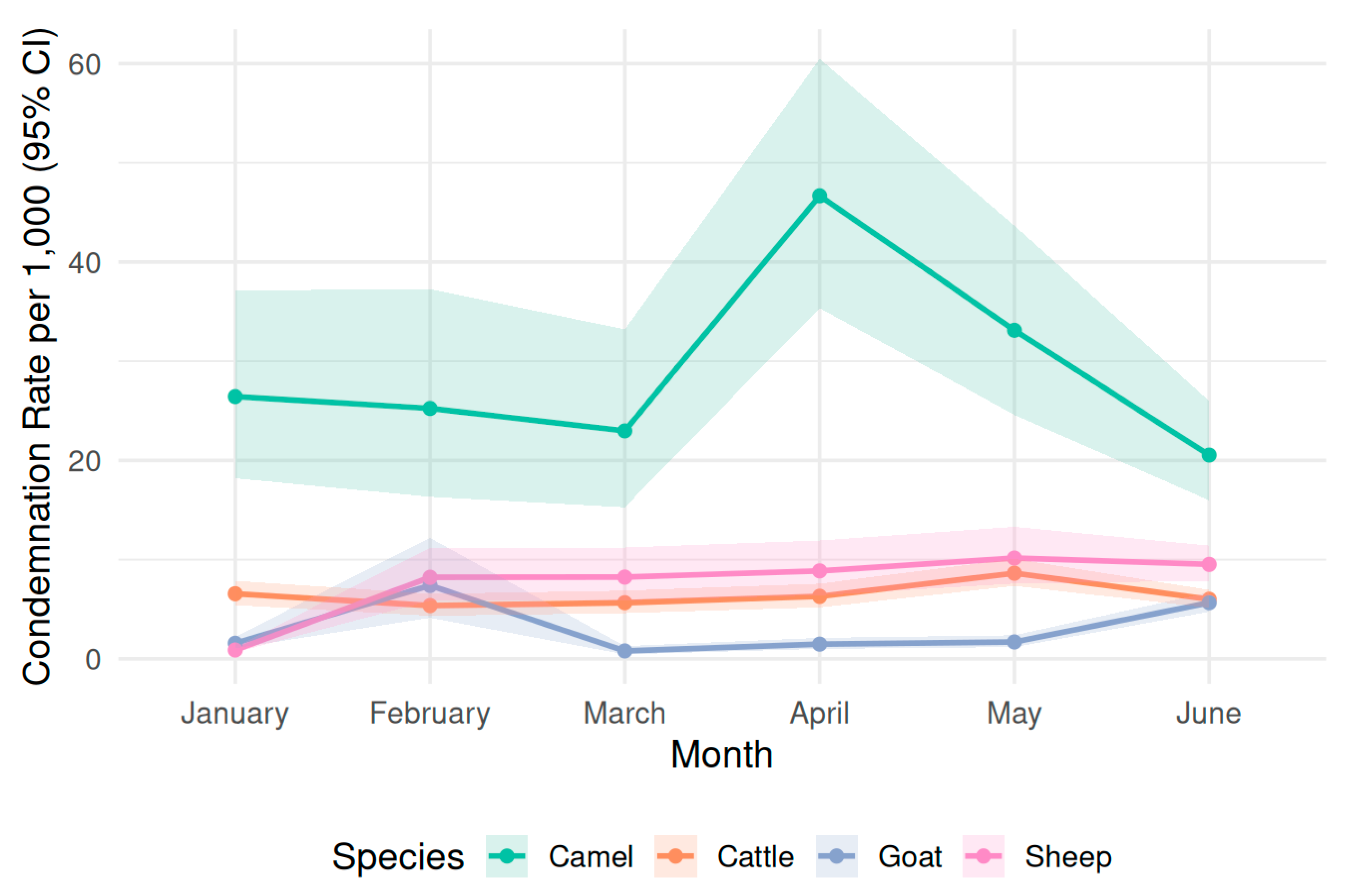
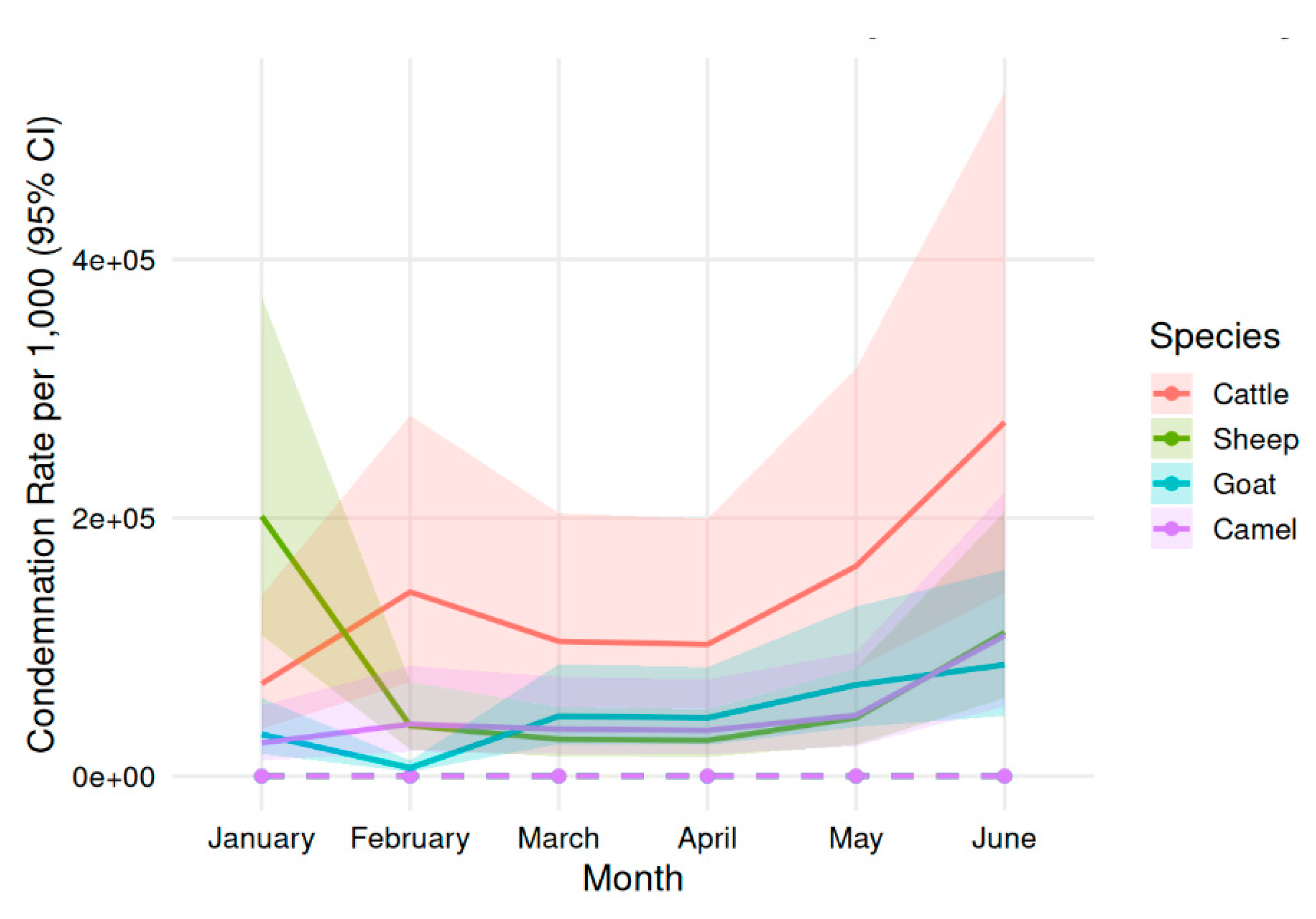
3.3. Temporal Trends in Species-Specific Condemnation Rates
3.4. Regression Analysis of Factors Associated with Condemnation
3.5. Negative Binomial Regression Analysis (Primary Model)
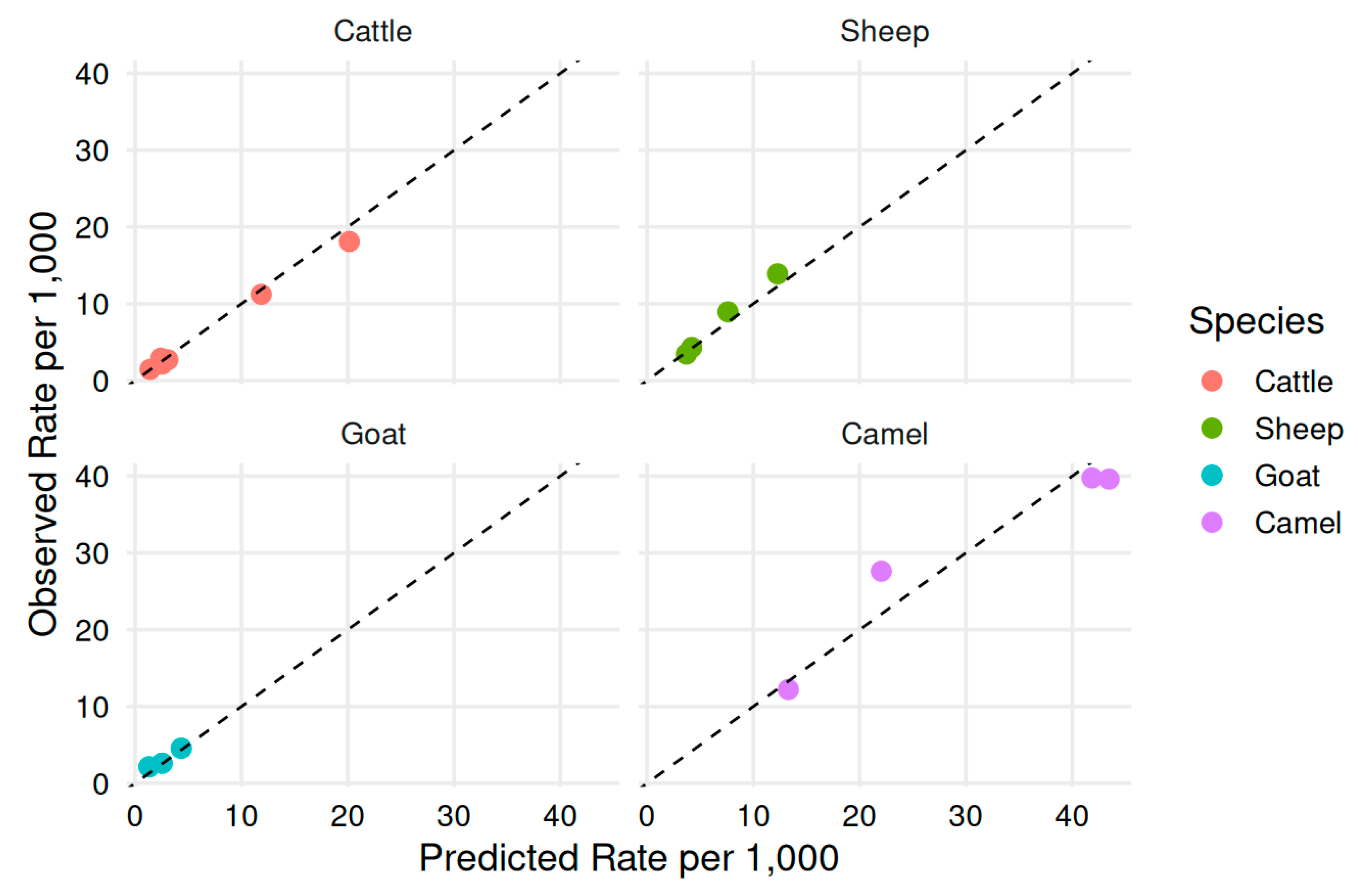
3.6. Model-Based Prediction of Condemnation Rates over Time
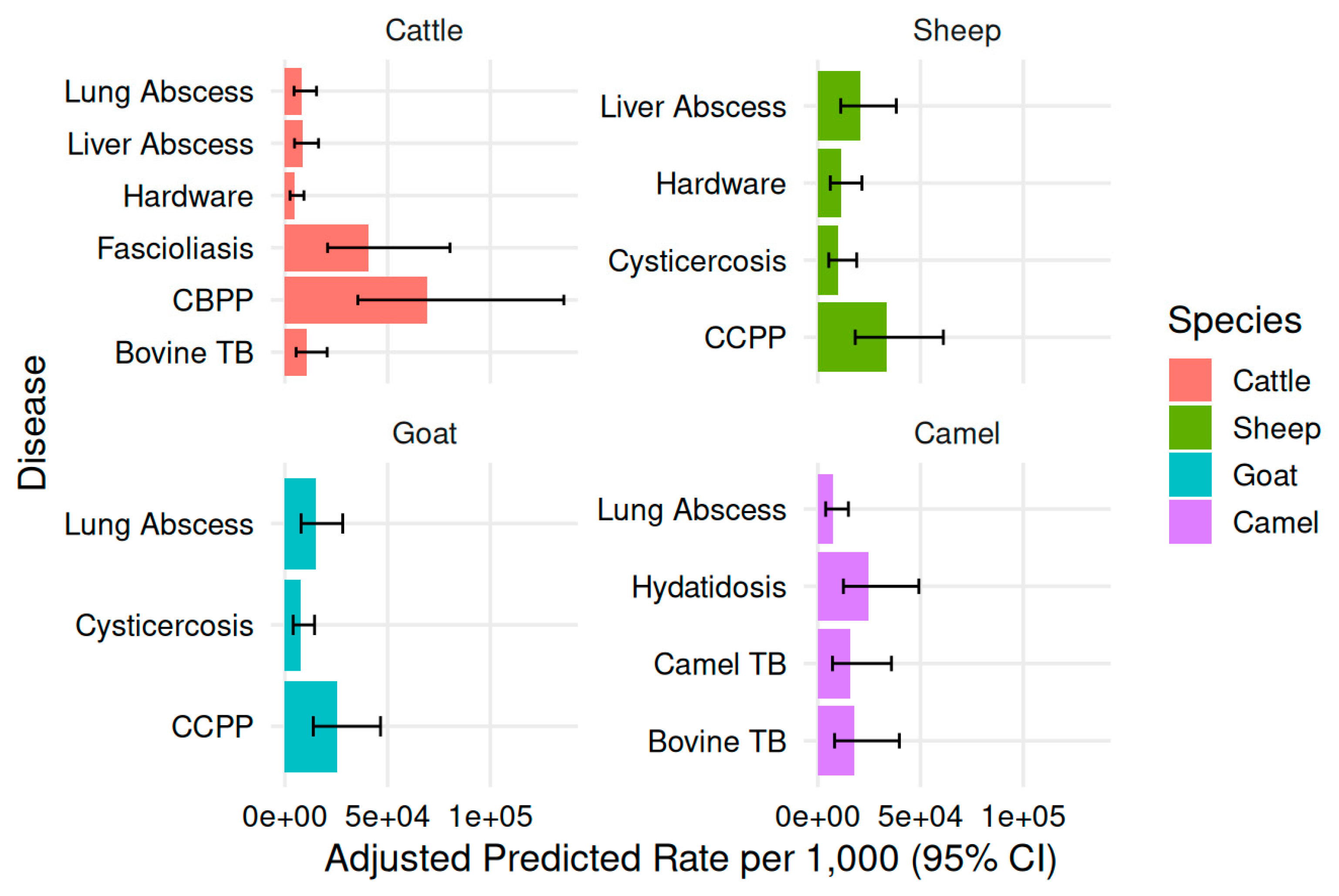
3.7. Model-Predicted Condemnation Rates by Species and Disease
| Species | Disease | Mean predicted rate per 1,000 | Lower 95% CI | Upper 95% CI |
|---|---|---|---|---|
| Cattle | CBPP | 69.47 | 35.54 | 135.78 |
| Cattle | Fascioliasis | 40.9 | 20.81 | 80.38 |
| Cattle | Bovine tuberculosis | 10.63 | 5.49 | 20.57 |
| Cattle | Liver abscess | 8.75 | 4.68 | 16.38 |
| Cattle | Lung abscess | 8.29 | 4.46 | 15.4 |
| Cattle | Hardware disease | 4.84 | 2.53 | 9.27 |
| Sheep | CCPP | 33.38 | 18.23 | 61.12 |
| Sheep | Liver abscess | 20.68 | 11.18 | 38.23 |
| Sheep | Hardware disease | 11.43 | 6.08 | 21.49 |
| Sheep | Cysticercosis | 10.06 | 5.35 | 18.92 |
| Goat | CCPP | 25.37 | 13.83 | 46.56 |
| Goat | Lung abscess | 14.89 | 7.87 | 28.15 |
| Goat | Cysticercosis | 7.65 | 4.05 | 14.45 |
| Camel | Hydatidosis | 24.76 | 12.46 | 49.19 |
| Camel | Bovine tuberculosis | 18.01 | 8.17 | 39.71 |
| Camel | Camel tuberculosis | 16 | 7.14 | 35.86 |
| Camel | Lung abscess | 7.57 | 3.85 | 14.89 |
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- García-Díez, J.; Saraiva, S.; Moura, D.; Grispoldi, L.; Cenci-Goga, B.T.; Saraiva, C. The importance of the slaughterhouse in surveilling animal and public health: A systematic review. Vet. Sci. 2023, 10, 167. [Google Scholar] [CrossRef]
- Nwankwo, I.; Nwanta, J.; Onunkwo, J. Abattoirs as meat safety and disease surveillance points in Nigeria: The case of Ikpa slaughterhouse, Nsukka, Nigeria. Sokoto J. Vet. Sci. 2023, 21, 47–50. [Google Scholar] [CrossRef]
- Nwanta, K.A.; Fair, J.M.; Bett, B.K.; Kerfua, S.D.; Fasina, F.O.; Bartlow, A.W. A scoping review of zoonotic parasites and pathogens associated with abattoirs in Eastern Africa and recommendations for abattoirs as disease surveillance sites. Front. Public Health 2023, 11, 1194964. [Google Scholar]
- Adebowale, O.; Ekundayo, O.; Olasoju, M.; Bankole, N.; Oladejo, O.; Awoseyi, A. Causes of organ condemnation in food animals slaughtered at a municipal abattoir in Oyo State, Nigeria. Savannah Vet. J. 2021, 1, 4. [Google Scholar]
- Ciui, S.; Morar, A.; Tîrziu, E.; Herman, V.; Ban-Cucerzan, A.; Popa, S.A.; Morar, D.; Imre, M.; Olariu-Jurca, A.; Imre, K. Causes of post-mortem carcass and organ condemnations and economic loss assessment in a cattle slaughterhouse. Animals 2023, 13, 3339. [Google Scholar] [CrossRef]
- Harley, S.; More, S.; Boyle, L.; Connell, N.O.; Hanlon, A. Good animal welfare makes economic sense: Potential of pig abattoir meat inspection as a welfare surveillance tool. Ir. Vet. J. 2012, 65, 11. [Google Scholar] [CrossRef]
- Jaja, I.F.; Mushonga, B.; Green, E.; Muchenje, V. Factors responsible for the post-slaughter loss of carcass and offal’s in abattoirs in South Africa. Acta Trop. 2018, 178, 303–310. [Google Scholar] [CrossRef] [PubMed]
- Sheferaw, D.; Abdu, K. Major causes of organ and carcass condemnation and associated financial losses in cattle slaughtered at Kombolcha ELFORA abattoir from 2008–2012, Ethiopia. Ethiop. Vet. J. 2017, 21, 54–66. [Google Scholar] [CrossRef]
- Tembo, W.; Nonga, H.E. A survey of the causes of cattle organs and/or carcass condemnation, financial losses and magnitude of foetal wastage at an abattoir in Dodoma, Tanzania. Onderstepoort J. Vet. Res. 2015, 82, 855. [Google Scholar] [CrossRef]
- Sa’idu, A.S.; Okolocha, E.C.; Dzikwi, A.A.; Gamawa, A.A.; Ibrahim, S.; Kwaga, J.K.; Usman, A.; Maigari, S.A. Public health implications and risk factors assessment of Mycobacterium bovis infections among abattoir personnel in Bauchi State, Nigeria. J. Vet. Med. 2015, 2015, 718193. [Google Scholar] [CrossRef]
- Odetokun, I.A.; Alhaji, N.B.; Aminu, J.; Lawan, M.K.; Abdulkareem, M.A.; Ghali-Mohammed, I. One Health risk challenges and preparedness regarding bovine tuberculosis at abattoirs in North-central Nigeria: Associated drivers and health belief. PLoS Negl. Trop. Dis. 2022, 16, e0010729. [Google Scholar] [CrossRef]
- Mbobo, S.; Byaruhanga, C.; Jaja, I.F. Knowledge, attitudes and practices of abattoir workers towards cystic echinococcosis in the Eastern Cape Province, South Africa. PAMJ-One Health 2025, 18, 17. [Google Scholar] [CrossRef]
- Bekederemo, B.O.; Onwumere-Idolor, O.S.; Mukoro, J.E.; Akpogheneoyibo-Owigho, O. Awareness, attitudes, and practices of abattoir workers towards bovine tuberculosis in Isoko north local government area of Delta State, Nigeria. Direct Res. J. Agric. Food Sci. 2025, 13, 24–29. [Google Scholar] [CrossRef]
- Anyanwu, U.; Dimov, A.; Zinsstag, J.; Tediosi, F.; Markosyan, T. Profitability and cost-effectiveness analysis of brucellosis control in Armenia: A One Health approach. CABI One Health 2024, 3. [Google Scholar] [CrossRef]
- Engdawork, A.; Negussie, H. Advances in animal disease surveillance and information systems and their role in disease control and prevention: Implications in Ethiopia. Vet. Med. Sci. 2025, 11, e70701. [Google Scholar] [CrossRef]
- Yang, L.; Fan, M.; Wang, Y. Dynamic modeling of prevention and control of brucellosis in China: A systematic review. Transbound. Emerg. Dis. 2025, 2025, 1393722. [Google Scholar] [CrossRef] [PubMed]
- Alhaji, N.B.; Yatswako, S.; Isola, T.O. A survey of organs/offal condemnations and foetal losses in slaughtered trade cattle at abattoirs in north-central Nigeria: Major causes and associated economic implications. Bull. Anim. Health Prod. Afr. 2017, 65, 81–93. [Google Scholar]
- Alhaji, N.B.; Babalobi, O.O. Economic impacts assessment of pleuropneumonia burden and control in pastoral cattle herds of north-central Nigeria. Bull. Anim. Health Prod. Afr. 2017, 65, 235–248. [Google Scholar]
- Cadmus, S.I.; Adesokan, H.K. Causes and implications of bovine organs/offal condemnations in some abattoirs in Western Nigeria. Trop. Anim. Health Prod. 2009, 41, 1455–1463. [Google Scholar] [CrossRef] [PubMed]
- Magaji, A.A.; Ibrahim, K.; Salihu, M.D.; Saulawa, M.A.; Mohammed, A.A.; Musawa, A.I. Prevalence of fascioliasis in cattle slaughtered in Sokoto metropolitan abattoir, Sokoto, Nigeria. Adv. Epidemiol. 2014, 2014, 247258. [Google Scholar] [CrossRef]
- Molla, D.; Nazir, S.; Mohammed, A.; Tintagu, T. Parasitic infections as major cause of abattoir condemnations in cattle slaughtered at an Ethiopian abattoir: 10-year retrospective study. J. Helminthol. 2019, 94, e31. [Google Scholar] [CrossRef] [PubMed]
- Abuseir, S. Major causes and associated economic losses of carcass and organ condemnation in cattle and sheep in the northern part of Palestine. World’s Vet. J. 2019, 9, 317–323. [Google Scholar] [CrossRef]
- Abunna, F.; Hordofa, D. Major causes of organ condemnation for cattle and its financial impact at Wolaita Soddo municipality abattoir, southern Ethiopia. Glob. Vet. 2013, 11, 730–734. [Google Scholar]
- Taha, A.; Saad, S.; Jubara, A.; Wani, C.; Phiri, A.M.; Simuunza, M.; Munyeme, M.; Hang’ombe, B.; Mumba, C. Financial losses arising from cattle organ and carcass condemnation at Lokoloko Abattoir in Wau, South Sudan. Adv. Prev. Med. 2023, 2023, 7975876. [Google Scholar] [CrossRef]
- Yatswako, S.; Alhaji, N.B. Survey of bovine fasciolosis burdens in trade cattle slaughtered at abattoirs in North-central Nigeria: The associated predisposing factors and economic implication. Parasite Epidemiol. Control 2017, 2, 30–39. [Google Scholar] [CrossRef]
- Zakariyau, U.; Sanyinna, Z.M.; Shafiu Samaila, A.M.; Ahmad, U.G.; Abdullahi, S.M.; Abdulrashid, B. Prevalence and trends of abattoir-detected diseases in slaughtered livestock between 2021–2024 at Sokoto metropolitan abattoir, Sokoto State, Nigeria. Direct Res. J. Public Health Environ. Technol. 2025, 10. [Google Scholar]
- Olusiyi, J.A.; Igila, T.T.; Jatutu, S.S.; Ebule, K. Seasonal trend of slaughter animals in the Sahelian zone of north eastern Nigeria: A case study of Borno State. Niger. J. Anim. Prod. 2021, 48, 1003–1006. [Google Scholar]
- Aylate, A.; Shah, S.N.; Aleme, H.; Gizaw, T.T. Bovine tuberculosis: Prevalence and diagnostic efficacy of routine meat inspection procedure in Woldiya municipality abattoir north Wollo zone, Ethiopia. Trop. Anim. Health Prod. 2013, 45, 855–864. [Google Scholar] [CrossRef]
- Asare, D.A.; Sanogo, P.; Bannor, J.O.; Ware, G.J.; Dokrugu, B.A.; Tongban, M.; Emikpe, B.; Mahamadu, A.; Kikimoto, B.B.; Akita, C.; Jones, S. Retrospective analysis and the impact of improved meat inspection on organ condemnation at Kumasi Abattoir. PAMJ One Health 2024, 14. [Google Scholar] [CrossRef]
- Hilbe, J.M. Negative Binomial Regression; Cambridge University Press: Cambridge, UK, 2011. [Google Scholar]
- Musunuru, A.; Proffitt, D.; Ewing, R.; Greene, W.H. Poisson and negative binomial regression analysis. In Advanced Quantitative Research Methods for Urban Planners; Routledge: London, UK, 2020; pp. 74–94. [Google Scholar]
- Lawan, M.K.; Bello, M.; Kwaga, J.K.; Raji, M.A. Evaluation of physical facilities and processing operations of major abattoirs in North-western states of Nigeria. Sokoto J. Vet. Sci. 2013, 11, 56–61. [Google Scholar] [CrossRef]
- Shittu, A.; Zaharadeen, M.M.; Fasina, F.O.; Umaru, M.A.; Ahmed, A. Classification of slaughtered animals and estimation of body condition scores during rainy season in Sokoto abattoir. Sokoto J. Vet. Sci. 2014, 12, 31–40. [Google Scholar] [CrossRef]
- Schnöller, A. Pautas para los procedimientos de inspección en animales y carnes en un matadero [Guidelines for animal and meat inspection procedures in the slaughterhouse]. Rev. Sci. Tech. 2006, 25, 849–860. [Google Scholar] [CrossRef] [PubMed]
- Herenda, D.C.; Chambers, P.G. Manual on Meat Inspection for Developing Countries; Food & Agriculture Organization: Rome, Italy, 1994; Vol. 119. [Google Scholar]
- Ferri, M.; Blagojevic, B.; Maurer, P.; Hengl, B.; Guldimann, C.; Mojsova, S.; Sakaridis, I.; Antunovic, B.; Gomes-Neves, E.; Zdolec, N.; Vieira-Pinto, M. Risk-based meat safety assurance system—An introduction to key concepts for future training of official veterinarians. Food Control 2023, 146, 109552. [Google Scholar] [CrossRef]
- von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P.; STROBE Initiative. Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. BMJ 2007, 335, 806–808. [Google Scholar] [CrossRef]
- Sa'idu, A.S.; Mohammed, S.; Ashafa, M.; Gashua, M.M.; Mahre, M.B.; Maigado, A.I. Retrospective study of bovine tuberculosis in Gombe Township Abattoir, Northeastern Nigeria. Int. J. Vet. Sci. Med. 2017, 5, 65–69. [Google Scholar] [CrossRef]
- Chafe, U.M.; Musa, A.; Dogara, B. Studies of some health aspects of traditional camel management in Northwestern Nigeria. Livest. Res. Rural Dev. 2008, 20, 45. Available online: http://www.lrrd.org/lrrd20/2/chaf20031.htm (accessed on 14 February 2026).
- Okolugbo, B.C.; Luka, S.A.; Ndams, I.S. Hydatidosis of camels and cattle slaughtered in Sokoto State, Northern Nigeria. Food Sci. Qual. Manag. 2013, 21. [Google Scholar]
- Lorusso, V.; Wijnveld, M.; Latrofa, M.S.; Fajinmi, A.; Majekodunmi, A.O.; Dogo, A.G.; Igweh, A.C.; Otranto, D.; Jongejan, F.; Welburn, S.C.; Picozzi, K. Canine and ovine tick-borne pathogens in camels, Nigeria. Vet. Parasitol. 2016, 228, 90–92. [Google Scholar] [CrossRef]
- Abdullahi, M.; Mohammed, A.K.; Okubanjo, O.O. Prevalence of haemoparasites and their effects on haematological values of infected one-humped camel (Camelus dromedarius) in the semi-arid region of Sokoto State, Nigeria. Niger. J. Anim. Prod. 2022, 49, 355–359. [Google Scholar]
- Anvari, D.; Pourmalek, N.; Rezaei, S.; Fotovati, A.; Hosseini, S.A.; Daryani, A.; Spotin, A.; Sarvi, S.; Hosseini, M.; Narouei, M.R.; Kalkali, M.; Pendar, F.; Gholami, S. The global status and genetic characterization of hydatidosis in camels (Camelus dromedarius): A systematic literature review with meta-analysis based on published papers. Parasitology 2021, 148, 259–273. [Google Scholar] [CrossRef]
- Gareh, A.; Saleh, A.A.; Moustafa, S.M.; Tahoun, A.; Baty, R.S.; Khalifa, R.M.A.; Dyab, A.K.; Yones, D.A.; Arafa, M.I.; Abdelaziz, A.R.; El-Gohary, F.A.; Elmahallawy, E.K. Epidemiological, morphometric, and molecular investigation of cystic echinococcosis in camel and cattle from Upper Egypt: Current status and zoonotic implications. Front. Vet. Sci. 2021, 8, 750640. [Google Scholar] [CrossRef]
- Bekele Atoma, T.; Szonyi, B.; Haile, A.F.; Fries, R.; Baumann, M.P.O.; Randolph, D.G. Assessment of health problems of sheep and goats based on ante-mortem and post-mortem inspection at Addis Ababa Abattoir, Ethiopia. Front. Vet. Sci. 2024, 11, 1406801. [Google Scholar] [CrossRef] [PubMed]
- Njoga, E.O.; Ezenduka, E.V.; Ogbodo, C.G.; Ogbonna, C.U.; Jaja, I.F.; Ofomatah, A.C.; Okpala, C.O.R. Detection, distribution and health risk assessment of toxic heavy metals/metalloids, arsenic, cadmium, and lead in goat carcasses processed for human consumption in South-Eastern Nigeria. Foods 2021, 10, 798. [Google Scholar] [CrossRef]
- Nwanta, J.A.; Onunkwo, J.I.; Ezenduka, V.E.; Phil-Eze, P.O.; Egege, S.C. Abattoir operations and waste management in Nigeria: A review of challenges and prospects. Sokoto J. Vet. Sci. 2008, 7. [Google Scholar]
- Shima, K.; Mosugu, I.; Apaa, T. Assessment of livestock slaughtered for food and meat inspection issues in selected abattoirs in Benue State, Nigeria. Cogent Food Agric. 2015, 1, 1106386. [Google Scholar] [CrossRef]
- Al-Mustapha, A.I.; Adetunji, V.; Ogundijo, O.A.; Odetokun, I.A.; Oyafajo, L.; Abali, H.W.; Oyewo, M.; Abubakar, A.T.; Muhammad, S.O.; Adetunji, D.A.; Odukoya, A.; Haruna, A.; Bamidele, F.; Elelu, N.; Fasina, F.O. Animal disease burden in Nigeria, 2006–2023. Transbound. Emerg. Dis. 2025, 2025, 1694850. [Google Scholar] [CrossRef]
- Markus, I.F.; Adamu, J.; Allam, L.; Kwanashie, C.N.; Raji, M.A.; Mohammed, B. Epidemiological and pathological screening of suspected cases of contagious bovine pleuropneumonia in Yola Modern Abattoir, Adamawa State, Nigeria. Niger. Vet. J. 2021, 42, 292–300. [Google Scholar] [CrossRef]
- Masiga, W.N.; Domenech, J.; Windsor, R.S. Manifestation and epidemiology of contagious bovine pleuropneumonia in Africa. Rev. Sci. Tech. 1996, 15, 1283–1308. [Google Scholar] [CrossRef]
- Ahmad, K.H.; Mamman, P.H.; Adamu, J.; Bello, M.; Olorunshola, I.D.; Umar, B.N.; Dalis, J.S.; Salawudeen, M.T.; Sada, A. Seroprevalence of contagious bovine pleuropneumonia in cattle slaughtered from Sokoto and Zamfara States, North-western Nigeria. Sahel J. Vet. Sci. 2024, 21, 20–26. [Google Scholar] [CrossRef]
- Alhaji, N.B.; Ankeli, P.I.; Ikpa, L.T.; Babalobi, O.O. Contagious bovine pleuropneumonia: Challenges and prospects regarding diagnosis and control strategies in Africa. Vet. Med. (Auckl.) 2020, 11, 71–85. [Google Scholar] [CrossRef]
- Khoramian, H.; Arbabi, M.; Osqoi, M.M.; Delavari, M.; Hooshyar, H.; Asgari, M. Prevalence of ruminants fascioliasis and their economic effects in Kashan, center of Iran. Asian Pac. J. Trop. Biomed. 2014, 4, 918–922. [Google Scholar] [CrossRef]
- Liba, J.W.; Atsanda, N.N.; Francis, M.I. Economic loss from liver condemnation due to fasciolosis in slaughtered ruminants in Maiduguri abattoir, Borno State, Nigeria. J. Adv. Vet. Anim. Res. 2017, 4, 65–70. [Google Scholar] [CrossRef]
- Nyirenda, S.S.; Sakala, M.; Moonde, L.; Kayesa, E.; Fandamu, P.; Banda, F.; Sinkala, Y. Prevalence of bovine fascioliasis and economic impact associated with liver condemnation in abattoirs in Mongu district of Zambia. BMC Vet. Res. 2019, 15, 33. [Google Scholar] [CrossRef]
- Arias-Pacheco, C.; Lucas, J.R.; Rodríguez, A.; Córdoba, D.; Lux-Hoppe, E.G. Economic impact of the liver condemnation of cattle infected with Fasciola hepatica in the Peruvian Andes. Trop. Anim. Health Prod. 2020, 52, 1927–1932. [Google Scholar] [CrossRef] [PubMed]
- Girma, A.; Teshome, K.; Abdu, I.; Genet, A.; Tamir, D. Prevalence and associated economic losses of bovine fasciolosis from postmortem inspection in municipal abattoirs in Ethiopia: A systematic review and meta-analysis. Vet. Anim. Sci. 2024, 24, 100360. [Google Scholar] [CrossRef]
- Opio, L.G.; Abdelfattah, E.M.; Terry, J.; Odongo, S.; Okello, E. Prevalence of fascioliasis and associated economic losses in cattle slaughtered at Lira Municipality Abattoir in Northern Uganda. Animals 2021, 11, 681. [Google Scholar] [CrossRef] [PubMed]
- Salihu, M.D.; Musawa, A.I.; Garba, B.; Yakubu, Y.; Bello, M.B.; Magaji, A.A.; Junaidu, A.U.; Jibril, A.H.; Ballah, F.M.; Achi, C.R. Molecular characterization and species differentiation of Fasciola parasite isolated from cattle slaughtered at Sokoto Modern Abattoir, Nigeria. Niger. J. Parasitol. 2022, 43. [Google Scholar] [CrossRef]
- Mamo, G.; Bayleyegn, G.; Sisay Tessema, T.; Legesse, M.; Medhin, G.; Bjune, G.; Abebe, F.; Ameni, G. Pathology of camel tuberculosis and molecular characterization of its causative agents in pastoral regions of Ethiopia. PLoS ONE 2011, 6, e15862. [Google Scholar] [CrossRef]
- Shittu, A.; Clifton-Hadley, R.S.; Ely, E.R.; Upton, P.U.; Downs, S.H. Factors associated with bovine tuberculosis confirmation rates in suspect lesions found in cattle at routine slaughter in Great Britain, 2003–2008. Prev. Vet. Med. 2013, 110, 395–404. [Google Scholar] [CrossRef]
- Jibril, Y.; Mamo, G.; Hanur, I.; Zewude, A.; Ameni, G. Prevalence of camel tuberculosis and associated risk factors in camels slaughtered at Akaki Abattoir, Ethiopia. Ethiop. Vet. J. 2016, 20, 23–38. [Google Scholar] [CrossRef]
- Ghebremariam, M.K.; Michel, A.L.; Vernooij, J.C.M.; Nielen, M.; Rutten, V.P.M.G. Prevalence of bovine tuberculosis in cattle, goats, and camels of traditional livestock raising communities in Eritrea. BMC Vet. Res. 2018, 14, 73. [Google Scholar] [CrossRef]
- Jajere, S.M.; Atsanda, N.N.; Bitrus, A.A.; Hamisu, T.M.; Goni, M.D. A retrospective study of bovine tuberculosis at the municipal abattoir of Bauchi State, Northeastern Nigeria. Vet. World 2018, 11, 598–605. [Google Scholar] [CrossRef]
- Danladi, J.; Kwaghe, A.V.; Olasoju, T.; Ibrahim, H.I.; Buba, M.I.; Dakogi, A.Y.; Vakuru, C.T. Prevalence, trends, and magnitude of bovine tuberculosis in slaughtered cattle across States in Nigeria, 2020–2022: A retrospective study. PAMJ One Health 2024, 15. [Google Scholar] [CrossRef]
- Vicenzi, J.M.; Cerva, C.; Karam, F.S.C.; de Moraes, L.B.; Rodrigues, R.O.; Oliveira, P.L.; Bertagnolli, A.C.; Mayer, F.Q. Accuracy of inspection surveillance in detecting bovine tuberculosis during slaughter in Rio Grande do Sul. Vet. Res. Commun. 2025, 49, 215. [Google Scholar] [CrossRef]
- Woldemariyam, F.T.; Markos, T.; Shegu, D.; Abdi, K.D.; Paeshuyse, J. Evaluation of postmortem inspection procedures to diagnose bovine tuberculosis at Debre Birhan municipal abattoir. Animals 2021, 11, 2620. [Google Scholar] [CrossRef] [PubMed]
- Boko, C.K.; Zoclanclounon, A.R.; Adoligbe, C.M.; Dedehouanou, H.; M'Po, M.; Mantip, S.; Farougou, S. Molecular diagnosis of bovine tuberculosis on postmortem carcasses during routine meat inspection in Benin: GeneXpert® testing to improve diagnostic scheme. Vet. World 2022, 15, 2506–2510. [Google Scholar] [CrossRef] [PubMed]
- Harandi, M.F.; Hobbs, R.P.; Adams, P.J.; Mobedi, I.; Morgan-Ryan, U.M.; Thompson, R.C.A. Molecular and morphological characterization of Echinococcus granulosus of human and animal origin in Iran. Parasitology 2002, 125, 367–373. [Google Scholar] [CrossRef]
- Ahmadi, N.A. Hydatidosis in camels (Camelus dromedarius) and their potential role in the epidemiology of Echinococcus granulosus in Iran. J. Helminthol. 2005, 79, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Mirzaei, M.; Rezaei, H.; Nematollahi, A.; Ashrafihelan, J. Survey of hydatidosis infection in slaughtered camel (Camelus dromedarius) in Tabriz area, Northwest Iran. J. Parasit. Dis. 2016, 40, 444–447. [Google Scholar] [CrossRef]
- Ebrahimipour, M.; Sadjjadi, S.M.; Yousofi Darani, H.; Najjari, M. Molecular studies on cystic echinococcosis of camel (Camelus dromedarius) and report of Echinococcus ortleppi in Iran. Iran. J. Parasitol. 2017, 12, 323–331. [Google Scholar]
- Garba, Y.; Haruna, K.A.; Bello, N.U. Slaughter practices, figures and incidence of post-mortem condemnation as affected by seasons in Karu Mini Abattoir Abuja, Nigeria. Niger. J. Anim. Prod. 2022, 49, 351–355. [Google Scholar]
- Alhaji, N.B.; Odetokun, I.A.; Shittu, A.; Onyango, J.; Chafe, U.M.; Abubakar, M.S.; Muraina, I.A.; Fasina, F.O.; Lee, H.S. Time-series analysis of ruminant foetal wastage at a slaughterhouse in North Central Nigeria between 2001 and 2012. Onderstepoort J. Vet. Res. 2015, 82, 1010. [Google Scholar] [CrossRef] [PubMed]
- Athiyarath, S.; Paul, M.; Krishnaswamy, S. A comparative study and analysis of time series forecasting techniques. SN Comput. Sci. 2020, 1, 175. [Google Scholar] [CrossRef]
- Ola-Fadunsin, S.D.; Uwabujo, P.I.; Halleed, I.N.; Richards, B. Prevalence and financial loss estimation of parasitic diseases detected in slaughtered cattle in Kwara State, North-central Nigeria. J. Parasit. Dis. 2020, 44, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Muhammed, B.A.; Salisu, U.S.; Idris, M.; Auwalu, Y.; Garasin, M.U.; Mamuda, M.A.; Garga, M.A.; Bakomi, O.W.; Garba, I.L. Retrospective study on condemned carcass and organ at Katsina Central Abattoir for a period of 2013 to 2019. UMYU Scientifica 2023, 2, 62–67. [Google Scholar] [CrossRef]
- Agbajelola, V.I.; Agbajelola, B.S. A systematic review and meta-analysis on the prevalence of bovine fascioliasis in Nigeria. Acta Parasitol. 2025, 70, 57. [Google Scholar] [CrossRef]
- Amuda, T.O.; Li, L.; Wang, L.Q.; Dai, G.D.; Wu, Y.D.; Morenikeji, O.A.; Pu, G.T.; Afayibo, D.J.A.; Wu, K.K.; Jia, W.Z.; Luo, X.N.; Yan, H.B. Cystic echinococcosis and Taenia spp. infections in Nigeria: A systematic review and meta-analysis of prevalence and geographical distribution. Acta Trop. 2025, 271, 107826. [Google Scholar] [CrossRef]
- Chukwu, E.; Yunusa, H.; Onyekaihu, A.L.; Aremu, S.; Oraka, O.; Simon, K. Concurrent hepatic and pulmonary fasciolosis in a White Fulani cow: A case report highlighting food safety and economic implications. Sahel J. Life Sci. FUDMA 2025, 3, 320–323. [Google Scholar] [CrossRef]
- Omotosho, O.O.; Emikpe, B.O.; Lasisi, O.T.; Oladunjoye, O.V. Pig slaughtering in Southwestern Nigeria: Peculiarities, animal welfare concerns and public health implications. Afr. J. Infect. Dis. 2016, 10, 146–155. [Google Scholar] [CrossRef]
- Ibrahim, S.; Kaltungo, B.Y.; Uwale, H.B.; Baba, A.Y.; Saidu, S.N.; Mohammed, F.U.; Dahiru, H.M. Role of slaughter facilities management in zoonoses and safety of meat produced for human consumption in Nigeria: A review. Bull. Natl. Res. Cent. 2021, 45, 137. [Google Scholar] [CrossRef]
- Njoga, E.O.; Ilo, S.U.; Nwobi, O.C.; Onwumere-Idolor, O.S.; Ajibo, F.E.; Okoli, C.E.; Jaja, I.F.; Oguttu, J.W. Pre-slaughter, slaughter and post-slaughter practices of slaughterhouse workers in Southeast Nigeria: Animal welfare, meat quality, food safety and public health implications. PLoS ONE 2023, 18, e0282418. [Google Scholar] [CrossRef] [PubMed]
- Edward, I.G.; Akpabio, U. Preliminary investigation of food safety practices and environmental hygiene in Uyo Central Abattoir, Uyo, Nigeria. J. Sustain. Vet. Allied Sci. 2024, 6. [Google Scholar]
- Ekpunobi, N.F.; Adesanoye, S.; Orababa, O.; Adinnu, C.; Okorie, C.; Akinsuyi, S. Public health perspective of public abattoirs in Nigeria, challenges and solutions. GSC Biol. Pharm. Sci. 2024, 26, 115–127. [Google Scholar] [CrossRef]
- Nigussie, A.G.; Velde, F.V.; Sarba, E.J.; Kumsa, B.; Gabriel, S. African abattoirs: A scoping review of practices, factors influencing implementation of good practices, and recommended solutions for improvement. BMC Vet. Res. 2025, 21, 415. [Google Scholar] [CrossRef]
- Aliyu, M.M.; Obi, T.U.; Egwu, G.O. Prevalence of contagious bovine pleuropneumonia (CBPP) in northern Nigeria. Prev. Vet. Med. 2000, 47, 263–269. [Google Scholar] [CrossRef]
- Tambuwal, F.M.; Egwu, G.O.; Shittu, A.; Sharubutu, G.H.; Umaru, M.A.; Umar, H.U. Vaccination coverage and prevalence of contagious bovine pleuropneumonia (1999–2008) in two transboundary states of North-Western Nigeria. Niger. Vet. J. 2011, 32, 3. [Google Scholar]
- Billy, I.L.; Balami, A.G.; Sackey, A.K.; Tekdek, L.B.; Sa’Idu, S.N.; Okaiyeto, S.O. Sero-prevalence of contagious bovine pleuropneumonia in three senatorial district of Kaduna State, Nigeria using latex agglutination test. World’s Vet. J. 2017, 7, 65–73. [Google Scholar] [CrossRef]
- Ameh, A.O.; Kabir, J.; Jatau, I.D.; Ashar, S.J.; Kaoje, A.B.; Halilu, L.L.; Igah, O.E.; Isa, U.; Maikai, B.V. High prevalence of human and cattle fascioliasis in Zaria and environs: An emerging zoonotic problem. medRxiv 2025. [Google Scholar] [CrossRef]
- Yuguda, A.U.; Iliyasu, M.Y.; Mudi, H.; Muhammad, A.; Panda, S.M.; Samaila, A.B. Prevalence and intensity of fascioliasis in cattle slaughtered at Central Abattoir Gombe, Gombe State, Nigeria. South Asian J. Parasitol. 2024, 7, 321–332. [Google Scholar]
- Bolajoko, M.B. Biosecurity and economic impact of major diseases of livestock among rural farmers in Plateau State, Nigeria: A pilot study. J. Vet. Biomed. Sci. 2023, 5, 74–86. [Google Scholar]
- Chen, W.; Qian, L.; Shi, J.; Franklin, M. Comparing performance between log-binomial and robust Poisson regression models for estimating risk ratios under model misspecification. BMC Med. Res. Methodol. 2018, 18, 63. [Google Scholar] [CrossRef]
- Gardner, W.; Mulvey, E.P.; Shaw, E.C. Regression analyses of counts and rates: Poisson, overdispersed Poisson, and negative binomial models. Psychol. Bull. 1995, 118, 392–404. [Google Scholar] [CrossRef] [PubMed]
- Riley, R.D.; Collins, G.S.; Kirton, L.; Snell, K.I.; Ensor, J.; Whittle, R.; Dhiman, P.; van Smeden, M.; Liu, X.; Alderman, J.; Nirantharakumar, K.; Manson-Whitton, J.; Westwood, A.J.; Cazier, J.B.; Moons, K.G.M.; Martin, G.P.; Sperrin, M.; Denniston, A.K.; Harrell, F.E., Jr.; Archer, L. Uncertainty of risk estimates from clinical prediction models: Rationale, challenges, and approaches. BMJ 2025, 388, e080749. [Google Scholar] [CrossRef] [PubMed]
- Rostron, P.D.; Fearn, T.; Ramsey, M.H. Confidence intervals for robust estimates of measurement uncertainty. Accredit. Qual. Assur. 2020, 25, 107–119. [Google Scholar] [CrossRef]
- Biffa, D.; Bogale, A.; Skjerve, E. Diagnostic efficiency of abattoir meat inspection service in Ethiopia to detect carcasses infected with Mycobacterium bovis: Implications for public health. BMC Public Health 2010, 10, 462. [Google Scholar] [CrossRef]
- Stärk, K.D.; Alonso, S.; Dadios, N.; Dupuy, C.; Ellerbroek, L.; Georgiev, M.; Hardstaff, J.; Huneau-Salaün, A.; Laugier, C.; Mateus, A.; Nigsch, A. Strengths and weaknesses of meat inspection as a contribution to animal health and welfare surveillance. Food Control 2014, 39, 154–162. [Google Scholar] [CrossRef]
- Horst, A.; Gertz, M.; Krieter, J. Challenges and opportunities of using meat inspection data to improve pig health traits by breeding: A review. Livest. Sci. 2019, 221, 155–162. [Google Scholar] [CrossRef]
- Buzdugan, S.N.; Alarcon, P.; Huntington, B.; Rushton, J.; Blake, D.P.; Guitian, J. Enhancing the value of meat inspection records for broiler health and welfare surveillance: Longitudinal detection of relational patterns. BMC Vet. Res. 2021, 17, 278. [Google Scholar] [CrossRef]
- Comin, A.; Jonasson, A.; Rockström, U.; Kautto, A.H.; Keeling, L.; Nyman, A.K.; Lindberg, A.; Frössling, J. Can we use meat inspection data for animal health and welfare surveillance? Front. Vet. Sci. 2023, 10, 1129891. [Google Scholar] [CrossRef]
- Ambaw, M.; Gelalcha, B.D.; Bayissa, B.; Worku, A.; Yohannis, A.; Zewude, A.; Ameni, G. Pathology of bovine tuberculosis in three breeds of dairy cattle and spoligotyping of the causative mycobacteria in Ethiopia. Front. Vet. Sci. 2021, 8, 715598. [Google Scholar] [CrossRef]
- Maric, D.; Vetter-Lang, S.; Klinger, J.; Böhm, N.; Schwaiger, K.; Käsbohrer, A. Improvement in the usability of meat inspection findings for swine herd health management. Animals 2025, 15, 688. [Google Scholar] [CrossRef] [PubMed]
- Claxton, P.D.; Eamens, G.J.; Mylrea, P.J. Laboratory diagnosis of bovine tuberculosis. Aust. Vet. J. 1979, 55, 514–520. [Google Scholar] [CrossRef]
- Bland, J.M.; Altman, D.G. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1986, 1, 307–310. [Google Scholar] [CrossRef]
- Mansournia, M.A.; Waters, R.; Nazemipour, M.; Bland, M.; Altman, D.G. Bland–Altman methods for comparing methods of measurement and response to criticisms. Glob. Epidemiol. 2020, 3, 100045. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Animal Health and Welfare (AHAW); More, S.; Bøtner, A.; Butterworth, A.; Calistri, P.; Depner, K.; Edwards, S.; Garin-Bastuji, B.; Good, M.; Gortázar Schmidt, C.; Michel, V.; Miranda, M.A.; Nielsen, S.S.; Raj, M.; Sihvonen, L.; Spoolder, H.; Stegeman, J.A.; Thulke, H.H.; Velarde, A.; Willeberg, P.; Winckler, C.; Baldinelli, F.; Broglia, A.; Candiani, D.; Beltrán-Beck, B.; Kohnle, L.; Bicout, D. Assessment of listing and categorisation of animal diseases within the framework of the Animal Health Law (Regulation (EU) No 2016/429): Contagious bovine pleuropneumonia. EFSA J. 2017, 15, e04995. [Google Scholar] [CrossRef]
- Onu, J.E. Fasciolosis and bovine liver condemnation in Sokoto metropolitan abattoir. J. Appl. Anim. Res. 2001, 20, 251–254. [Google Scholar] [CrossRef]
- McManus, D.P.; Zhang, W.; Li, J.; Bartley, P.B. Echinococcosis. Lancet 2003, 362, 1295–1304. [Google Scholar] [CrossRef]
- Heath, D.; Yang, W.; Li, T.; Xiao, Y.; Chen, X.; Huang, Y.; Yang, Y.; Wang, Q.; Qiu, J. Control of hydatidosis. Parasitol. Int. 2006, 55, S247–S252. [Google Scholar] [CrossRef]
- Craig, P.S.; McManus, D.P.; Lightowlers, M.W.; Chabalgoity, J.A.; Garcia, H.H.; Gavidia, C.M.; Gilman, R.H.; Gonzalez, A.E.; Lorca, M.; Naquira, C.; Nieto, A. Prevention and control of cystic echinococcosis. Lancet Infect. Dis. 2007, 7, 385–394. [Google Scholar] [CrossRef] [PubMed]
- Borhani, M.; Fathi, S.; Harandi, M.F.; Casulli, A.; Ding, J.; Liu, M.; Zhang, W.; Wen, H. Echinococcus granulosus sensu lato control measures: A specific focus on vaccines for both definitive and intermediate hosts. Parasit. Vectors 2024, 17, 533. [Google Scholar] [CrossRef]
- Correia-Gomes, C.; Smith, R.P.; Eze, J.I.; Henry, M.K.; Gunn, G.J.; Williamson, S.; Tongue, S.C. Pig abattoir inspection data: Can it be used for surveillance purposes? PLoS ONE 2016, 11, e0161990. [Google Scholar] [CrossRef]
- Gadisa, B.; Yusuf, Y.; Kurtu, M.Y. Evaluation of physical facilities, operation, and management practice in selective public abattoirs in Eastern Oromia, Ethiopia. Int. J. Agric. Sci. Food Technol. 2019, 5, 043–049. [Google Scholar] [CrossRef]
- George, J.; Häsler, B.; Komba, E.; Sindato, C.; Rweyemamu, M.; Mlangwa, J. Towards an integrated animal health surveillance system in Tanzania: Making better use of existing and potential data sources for early warning surveillance. BMC Vet. Res. 2021, 17, 109. [Google Scholar] [CrossRef] [PubMed]
- Vandenbroucke, J.P.; von Elm, E.; Altman, D.G.; Gøtzsche, P.C.; Mulrow, C.D.; Pocock, S.J.; Poole, C.; Schlesselman, J.J.; Egger, M.; STROBE Initiative. Strengthening the Reporting of Observational Studies in Epidemiology (STROBE): Explanation and elaboration. PLoS Med. 2007, 4, e297. [Google Scholar] [CrossRef] [PubMed]
- Cuschieri, S. The STROBE guidelines. Saudi J. Anaesth. 2019, 13, S31–S34. [Google Scholar] [CrossRef]
| Species of Food animal | Number of animal slaughtered | Percentage Slaughtered |
Number of condemned cases | Rate per 1,000 | Lower-Upper At 95% CI |
|---|---|---|---|---|---|
| Cattle | 119,988 | 37.8 | 767 | 6.39 | 5.95-6.86 |
| Goat | 106,899 | 33.6 | 264 | 2.47 | 2.18-2.79 |
| Sheep | 81,252 | 25.6 | 335 | 4.12 | 3.69-4.59 |
| Camel | 9,546 | 3.0 | 262 | 27.4 | 24.2-31.0 |
| Total | 317,685 | 100 | 1,628 | 5.12 | 4.88-5.38 |
| Species | Recorded cause of condemnation | Number of cases |
|---|---|---|
| Camel | Hydatidosis | 126 |
| Camel | Camel tuberculosis | 62 |
| Camel | Bovine tuberculosis | 41 |
| Camel | Lung abscess | 33 |
| Cattle | Contagious bovine pleuropneumonia (CBPP) | 358 |
| Cattle | Fascioliasis | 212 |
| Cattle | Lung abscess | 66 |
| Cattle | Bovine tuberculosis | 56 |
| Cattle | Liver abscess | 40 |
| Cattle | Hardware disease | 35 |
| Goat | Contagious caprine pleuropneumonia (CCPP) | 133 |
| Goat | Lung abscess | 86 |
| Goat | Cysticercosis | 45 |
| Sheep | Contagious caprine pleuropneumonia (CCPP) | 148 |
| Sheep | Liver abscess | 122 |
| Sheep | Hardware disease | 37 |
| Sheep | Cysticercosis | 28 |
| Total | 1,628 |
| Month | Camel_Rate (95% CI) | Cattle_Rate (95% CI) | Goat_Rate (95% CI) | Sheep_Rate (95% CI) |
|---|---|---|---|---|
| January | 26.4 (18.2–37.1) | 6.56 (5.42–7.87) | 2.46 (1.80–3.28) | 4.17 (3.20–5.36) |
| February | 25.2 (16.3–37.2) | 5.37 (4.35–6.56) | 2.23 (1.59–3.05) | 3.75 (2.80–4.92) |
| March | 23.0 (15.3–33.3) | 5.66 (4.61–6.89) | 2.39 (1.72–3.23) | 4.16 (3.14–5.41) |
| April | 46.7 (35.4–60.4) | 6.30 (5.19–7.60) | 2.51 (1.82–3.36) | 4.67 (3.59–5.98) |
| May | 33.1 (24.6–43.6) | 6.07 (5.20–7.06) | 2.54 (1.85–3.40) | 3.90 (2.92–5.11) |
| June | 20.5 (16.0–25.8) | 8.63 (7.41–10.03) | 2.54 (1.88–3.37) | 4.09 (3.06–5.35) |
| Variable domain | Predictor | aIRR | 95% CI | p-value |
|---|---|---|---|---|
| Temporal effects (Month) | Month (linear) | 1.67 | 1.13 – 2.48 | 0.008 |
| Month (quadratic) | 0.98 | 0.66 – 1.45 | 0.896 | |
| Month (cubic) | 1.3 | 0.87 – 1.94 | 0.181 | |
| Month (4th order) | 0.67 | 0.45 – 0.99 | 0.04 | |
| Month (5th order) | 1 | 0.67 – 1.49 | 0.987 | |
| Species effects | Cattle | 0.18 | 0.09 – 0.36 | <0.001 |
| Goat | 0.19 | 0.08 – 0.43 | <0.001 | |
| Sheep | 0.54 | 0.23 – 1.23 | 0.138 | |
| Disease-specific effects | Camel tuberculosis | 2.88 | 1.01 – 8.49 | 0.053 |
| CBPP | 6.54 | 2.88 – 15.20 | <0.001 | |
| CCPP | 1.33 | 0.56 – 3.12 | 0.508 | |
| Cysticercosis | 0.4 | 0.16 – 0.98 | 0.038 | |
| Fascioliasis | 3.85 | 1.67 – 9.05 | 0.001 | |
| Hardware disease | 0.46 | 0.20 – 1.00 | 0.047 | |
| Hydatidosis | 2.55 | 0.98 – 6.64 | 0.055 | |
| Liver abscess | 0.82 | 0.37 – 1.79 | 0.617 | |
| Lung abscess | 0.78 | 0.39 – 1.53 | 0.482 | |
| Model intercept | Intercept | 0.016 | 0.008 – 0.035 | <0.001 |
| Species | Disease | Observed rate/1,000 | Predicted rate/1,000 | Difference (Predicted − Observed) |
|---|---|---|---|---|
| Cattle | CBPP | 18.1 | 20.1 | 2.0 |
| Cattle | Fascioliasis | 11.2 | 11.9 | 0.7 |
| Cattle | Bovine tuberculosis | 2.7 | 3.08 | 0.38 |
| Cattle | Lung abscess | 2.91 | 2.4 | −0.51 |
| Cattle | Liver abscess | 2.15 | 2.54 | 0.39 |
| Cattle | Hardware disease | 1.45 | 1.4 | −0.05 |
| Sheep | CCPP | 13.9 | 12.3 | −1.60 |
| Sheep | Liver abscess | 8.95 | 7.6 | −1.35 |
| Sheep | Hardware disease | 4.31 | 4.2 | −0.11 |
| Sheep | Cysticercosis | 3.45 | 3.7 | 0.25 |
| Goat | CCPP | 4.56 | 4.33 | −0.23 |
| Goat | Lung abscess | 2.62 | 2.54 | −0.08 |
| Goat | Cysticercosis | 2.14 | 1.31 | −0.83 |
| Camel | Hydatidosis | 39.6 | 43.5 | 3.9 |
| Camel | Camel tuberculosis | 39.8 | 41.9 | 2.1 |
| Camel | Bovine tuberculosis | 27.6 | 22 | −5.60 |
| Camel | Lung abscess | 12.2 | 13.3 | 1.1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





