Submitted:
27 February 2026
Posted:
02 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Cell Culture
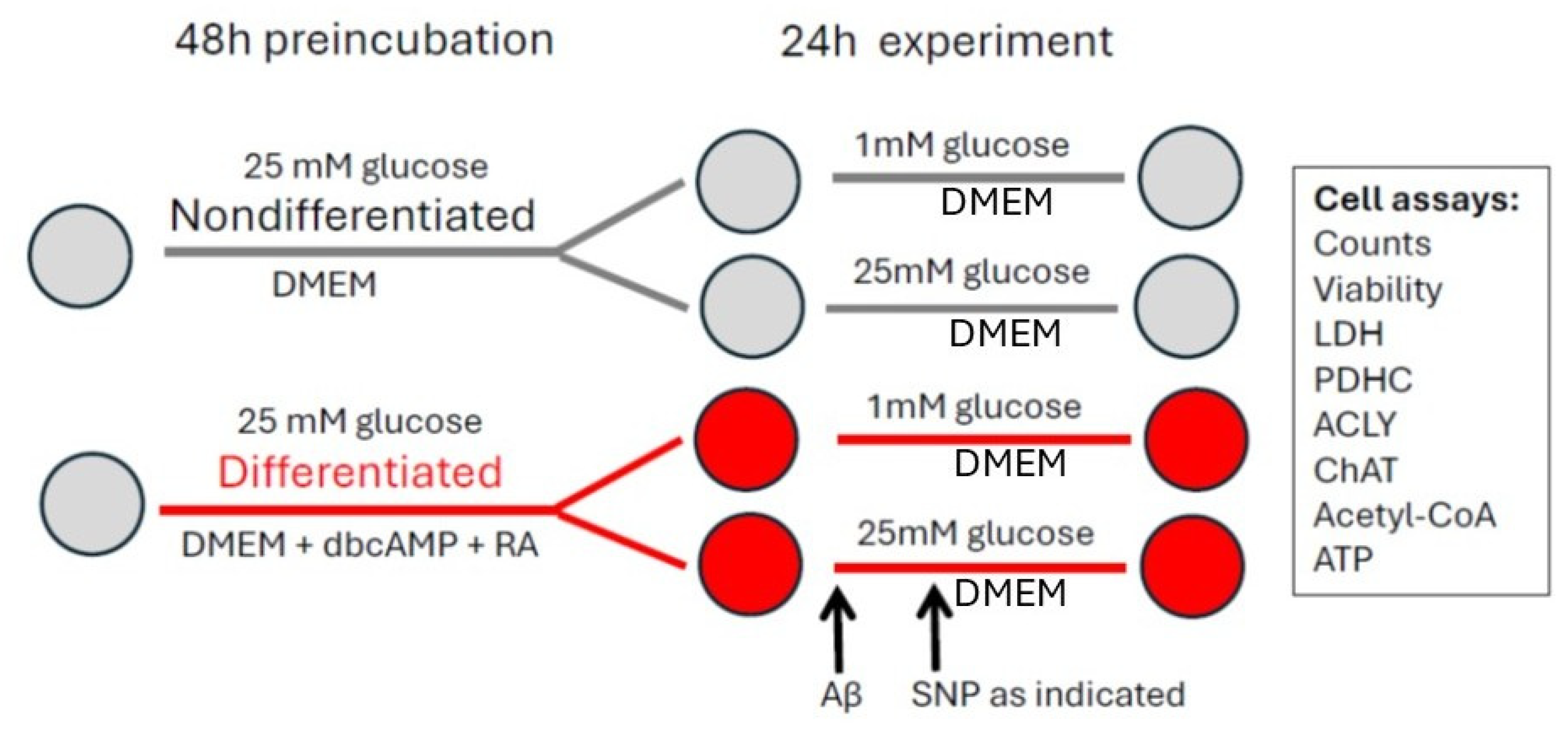
2.3. Viability and Cell Counts
2.4. Enzyme Assays
2.5. Protein Assay
2.6. Acetyl-CoA Assay
2.7. Statistics
3. Results
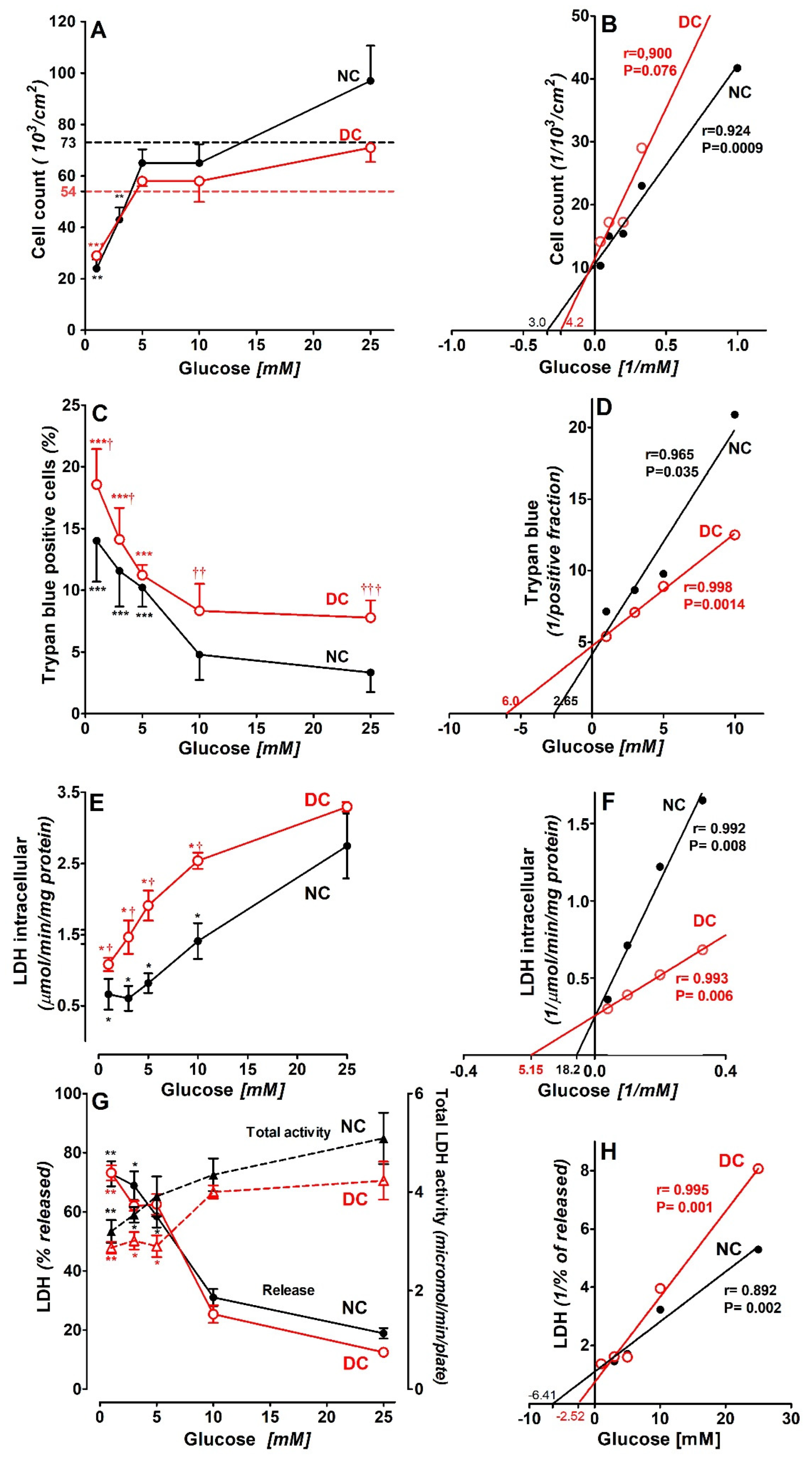
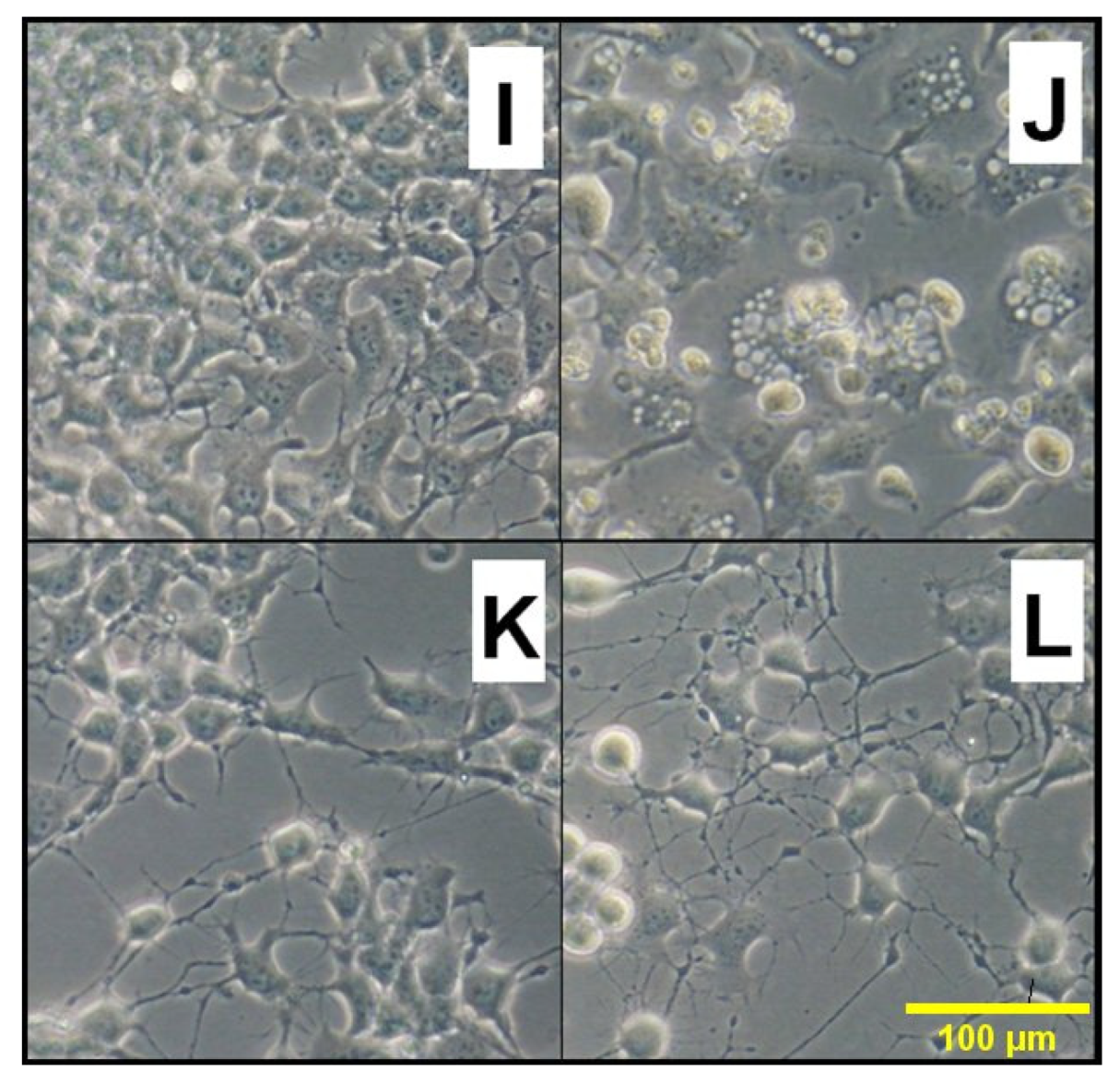
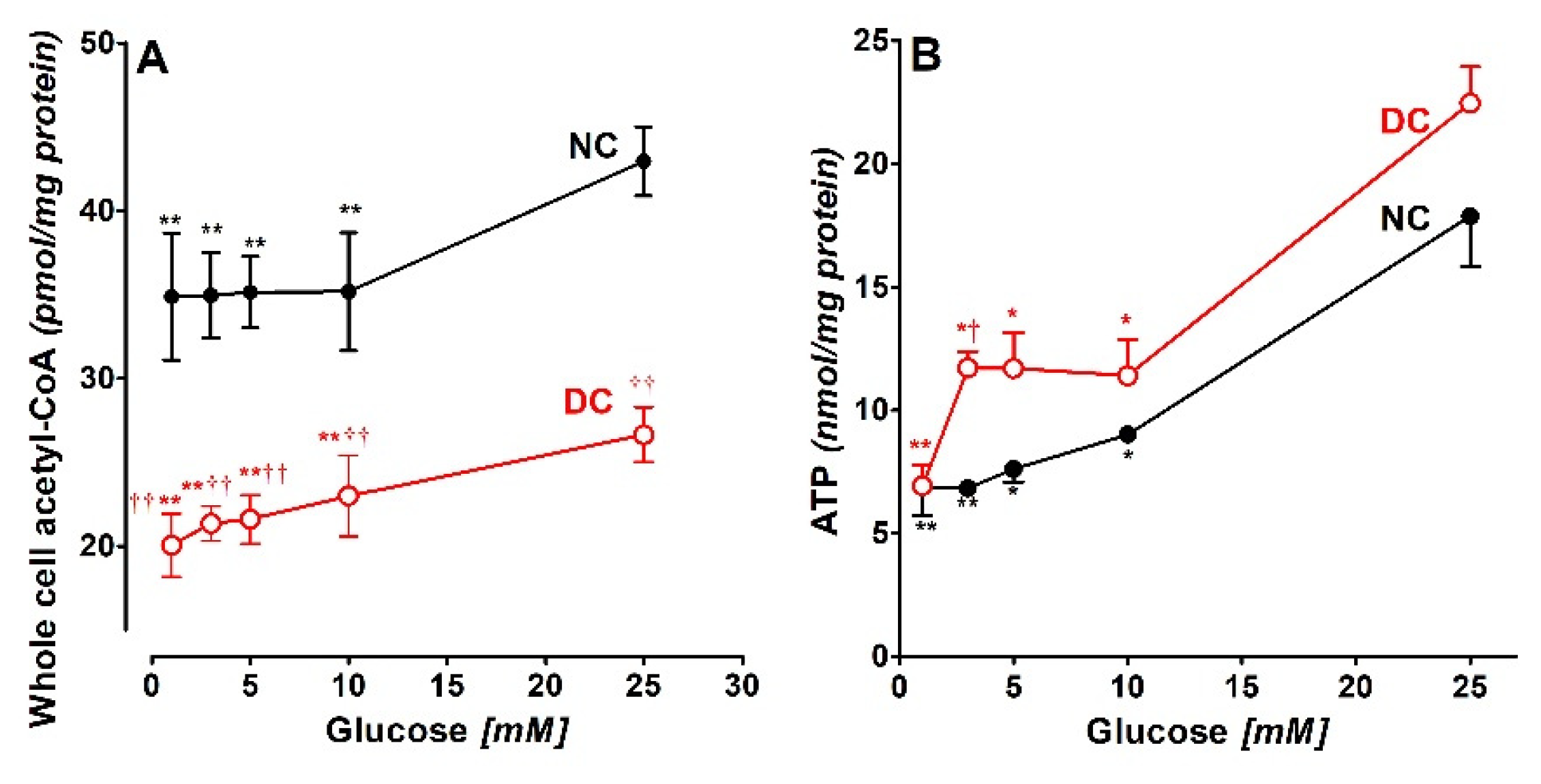
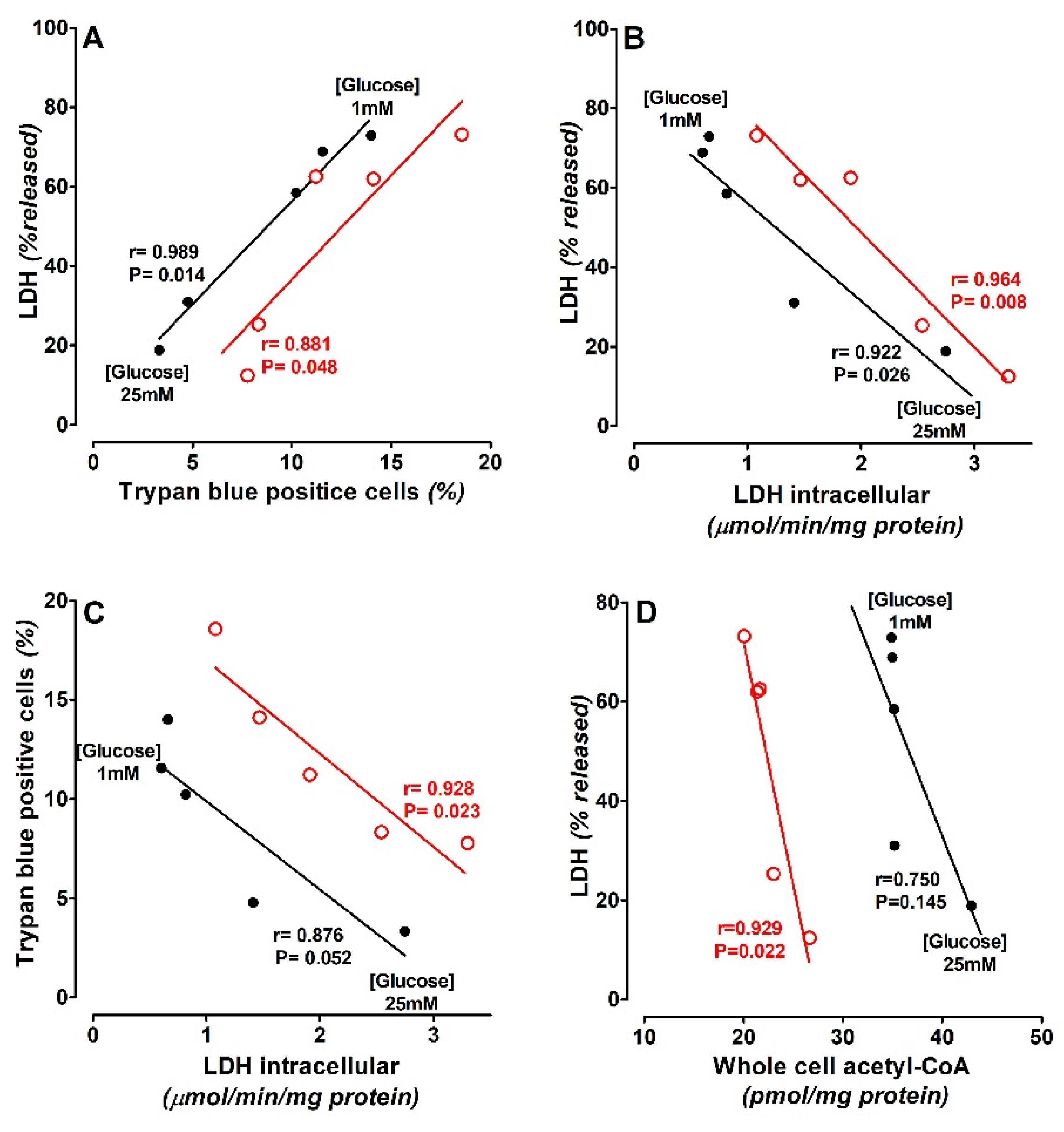
3.1. Effects of Glycaemia on SN56 Cells
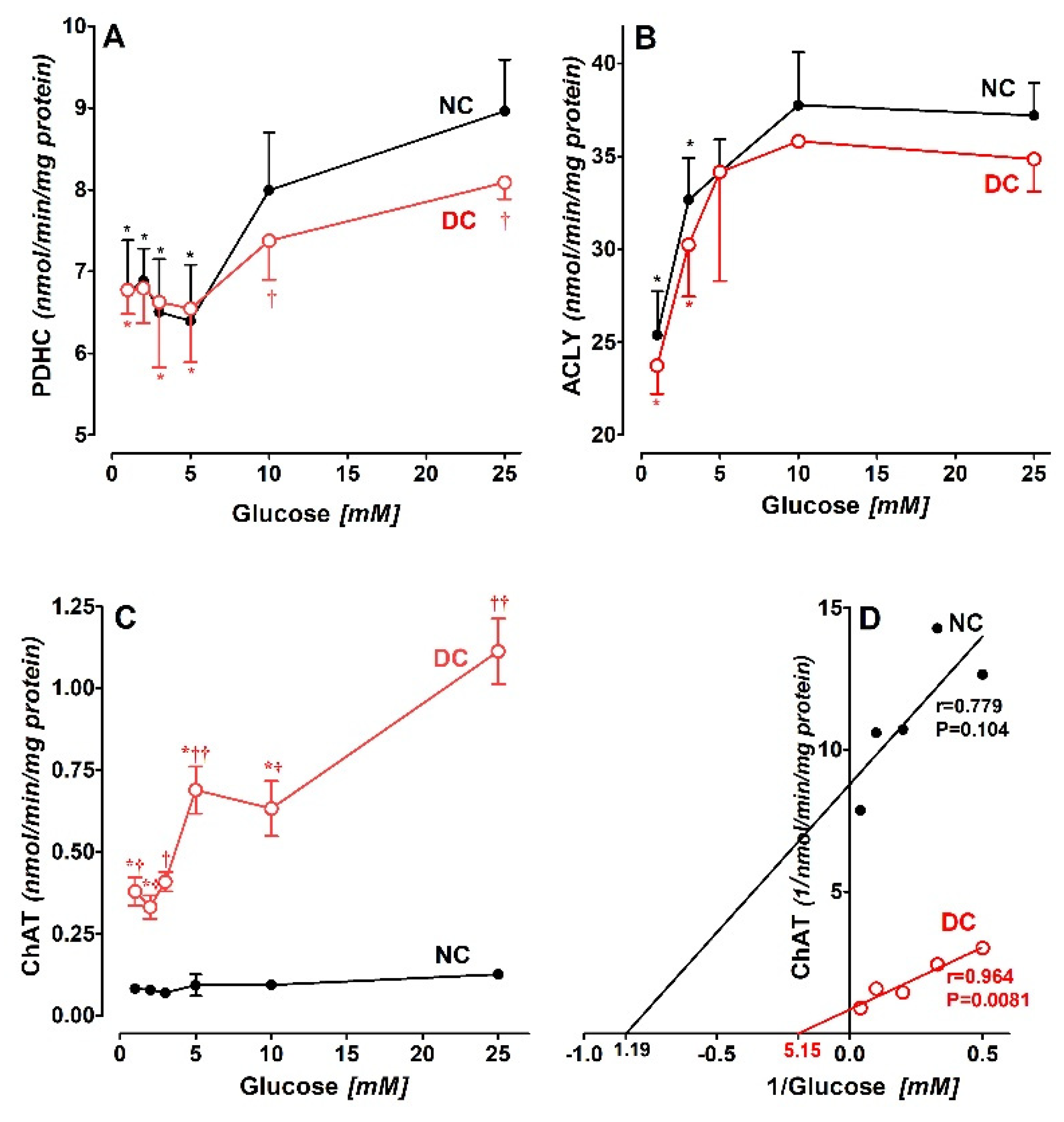
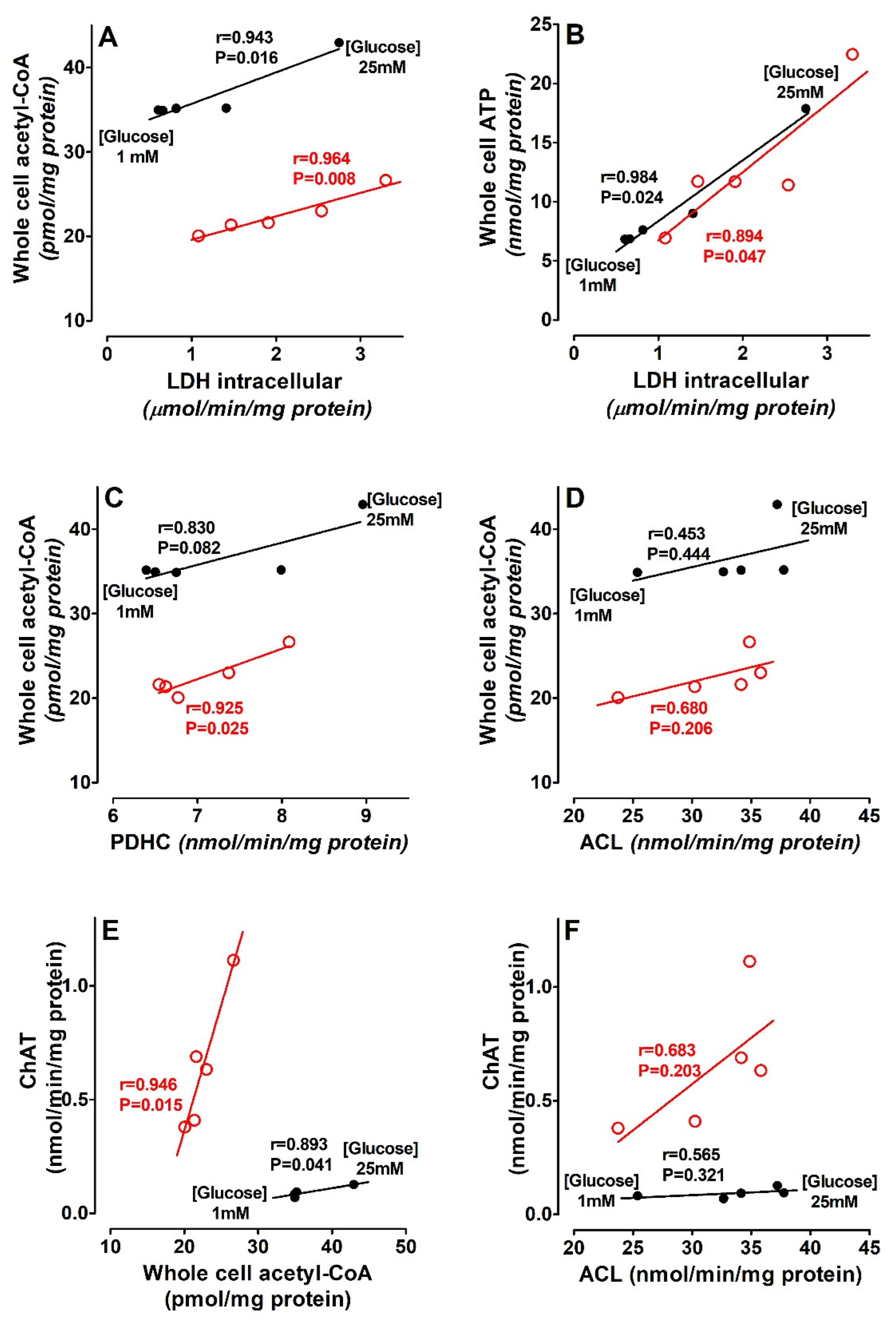
3.2. Glycaemia and Enzymes of Acetyl-CoA Metabolism in SN56 Cells
3.3. Glycemia and Energy Metabolite Levels in SN56 Cells
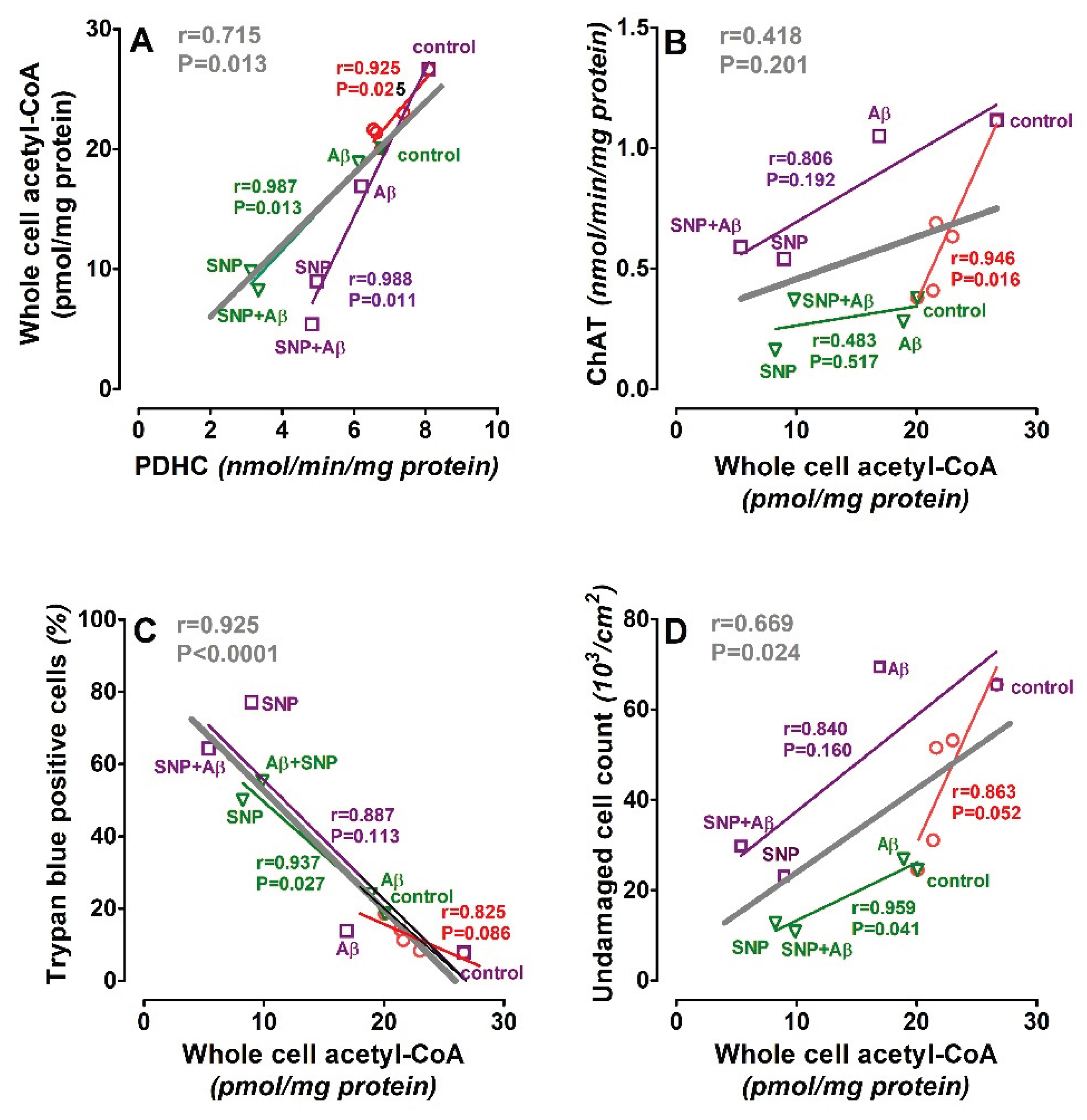
3.4. Glycemia-Dependent Effects of NO and Aβ on Viability and Parameters of Acetyl-CoA Metabolism
4. Discussion
Author Contributions
Resource Availability
Acknowledgments
Conflicts of Interest
Abbreviations
| Aβ | amyloid-β25-35 |
| Ach | acetylcholine |
| ACLY | ATP-citrate lyase |
| AD | Alzheimer’s disease |
| ChAT | choline acetyltransferase |
| dbcAMP | dibutyryl cyclic adenosine 5′ monophsphate |
| DC | differentiated SN56 cholinergic neuronal cells |
| DMEM | Dulbecco Modified Eagles Medium |
| EDTA | ethylenediaminetetraacetic acid |
| GLUT3 | glucose transporter 3 |
| HEPES |
 -2-hydroxyethylpiperazine-‘-2-etanosulfonic acid -2-hydroxyethylpiperazine-‘-2-etanosulfonic acid |
| [IC50] | half maximum inhibitory concentration |
| LDH | lactate dehydrogenase |
| NC | nondifferentiated SN56 cholinergic neuroblastoma cells- |
| PDHC | pyruvate dehydrogenase complex |
| RA | retinoic acid |
| SNP | sodium nitroprusside |
| TB+ | trypan blue retention assay |
References
- Deary, I.J. Effects of hypoglycaemia on cognitive function in: Hypoglycaemia and diabetes: clinical and physiological aspects; Edward Arnold: London, 1993; pp. 80–92. [Google Scholar]
- Warren, R.E.; Zammitt, N.N.; Deary, I.J.; Frier, B. The effects of acute hypoglycaemia on memory acquisition and recall and prospective memory in type 1 diabetes. Diabetologia 2007, 50, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Languren, G.; Montiel, T.; Amilpas, A.J.; Massieu, L. Neuronal damage and cognitive impairment associated with hypoglycemia: an integrated view. Neurochem. Int. 2013, 63, 331–43. [Google Scholar] [CrossRef]
- Won Suh, S.; Hamby, A.M.; Swanson, R.A. Hypoglycemia, brain energetics, and hypoglycemic neuronal death. Glia 2007, 55, 1280–6. [Google Scholar] [CrossRef]
- Boyle, P.J. Diabetes mellitus and macrovascular disease: mechanisms and mediators. Am. J. Med. 2007, 120 (Suppl 2), 12–7. [Google Scholar] [CrossRef]
- Rao, R.; Ennis, K.; Long, J.D.; Ugurbil, K.; Gruetter, R.; Tkac, I. Neurochemical changes in the developing rat hippocampus during prolonged hypoglycemia. J. Neurochem. 2010, 114, 728–38. [Google Scholar] [CrossRef]
- Mohseni, S. Neurologic damage in hypoglycemia. Handbook of Clinical Neurology 2014, 126, 513–32. [Google Scholar]
- Niewiadomska, G.; Baksalerska-Pazera, M.; Riedel, G. The septo-hippocampal system, learning and recovery of function. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2009, 33, 791–805. [Google Scholar] [CrossRef] [PubMed]
- Moss, D.E.; Perez, R.G. The phospho-tau cascade, basal forebrain neurodegeneration, and dementia in Alzheimer’s disease: Anti-neurodegenerative benefits of acetylcholinesterase inhibitors. J. Alzheimers Dis. 2024, 102(3), 617–626. [Google Scholar] [CrossRef] [PubMed]
- Rinne, J.O.; Ma, Y.S.; Lee, M.S.; Collan, Y.; Röyttä, M. Loss of cholinergic neurons in the pedunculopontine nucleus in Parkinson’s disease in related to disability of the patients. Parkinsonism Relat. Disord. 2008, 14, 553–557. [Google Scholar] [CrossRef]
- Bielarczyk, H.; Jankowska, A.; Madziar, B.; Matecki, A.; Michno, A.; Szutowicz, A. Differential toxicity of nitric oxide, aluminium, and amyloid-β peptide in SN56 cholinergic cells from mouse septum. Neurochem. Int. 2003, 42, 323–331. [Google Scholar] [CrossRef]
- Blennow, K.; de Leon, M.J.; Zetterberg, H. Alzheimer’s disease. Lancet 2006, 368, 387–403. [Google Scholar] [CrossRef]
- Gul-Hinc, S.; Zyśk, M.; Jankowska-Kulawy, A.; Ronowska, A. Protection of cholinergic neurons against zinc toxicity by glial cells in thiamine-deficient media. Int. J. Mol. Sci. 2021, 22(24), 1–20. [Google Scholar] [CrossRef]
- Kipp, B.T.; Savage, L.M. Modulation of the p75NTR during adolescent alcohol exposure prevents cholinergic neuronal atrophy and associated acetylcholine activity and behavioral dysfunction. Int. J. Mol. Sci. 2024, 25(11), 57–92. [Google Scholar] [CrossRef] [PubMed]
- Bubber, P.; Haroutunian, V.; Fisch, G.; Blass, J.P.; Gibson, G.E. Mitochondrial abnormalities in Alzheimer brain: mechanistic implications. Ann. Neurol. 2005, 57(5), 695–703. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Liu, S.; Sun, Y.; Chen, Ch.; Hu, Z.; Li, Q.; Long, J.; Yan, Q.; Liang, J.; Lin, Y.; et al. Target modulation of glycolytic pathways as a new strategy for the treatment of neuroinflammatory diseases. Ageing Res. Rev. 2024, 101, 102472. [Google Scholar] [CrossRef]
- Moiseenok, A.G.; Kanunnikova, N.P. Brain CoA and acetyl CoA system in mechanism of neurodegeneration. Biochemistry 2023, 88(4), 466–480. [Google Scholar] [CrossRef]
- Szutowicz, A.; Bielarczyk, H.; Zyśk, M.; Dyś, A.; Ronowska, A.; Gul-Hinc, S.; Klimaszewska-Łata, J. Early and late pathomechanisms in Alzheimer’s disease: from zinc to amyloid-β neurotoxicity. Neurochem. Res. 2017, 42(3), 891–904. [Google Scholar] [CrossRef]
- Ronowska, A.; Szutowicz, A.; Bielarczyk, H.; Gul-Hinc, S.; Klimaszewska-Łata, J.; Dyś, A.; Zyśk, M.; Jankowska-Kulawy, A. The regulatory effects of acetyl-CoA distribution in the healthy and diseased brain. Front. Cell Neurosci. 2018, (10)12, 169. [Google Scholar] [CrossRef] [PubMed]
- Villasana-Salazar, B.; Hernández-Soto, R.; Guerrero-Gómez, M.E.; Ordaz, B.; Manrique-Maldonado, G.; Salgado-Puga, K.; Peña-Ortega, F. Chronic intermittent hypoxia transiently increases hippocampal network activity in the gamma frequency band and 4-Aminopyridine-induced hyperexcitability in vitro. Epilepsy Res. 2020, 166, 106–375. [Google Scholar] [CrossRef]
- Christensen, D.Z.; Bayer, A.; Wirts, O. Intracellular Aβ triggers neuron loss in the cholinergic system of the APP/PS1KI Mouse model of Alzheimer’s disease. Neurobiol. Aging 2010, 31, 1153–1163. [Google Scholar] [CrossRef]
- Szutowicz, A.; Tomaszewicz, M.; Jankowska, A.; Kisielevski, Y. Acetylcholine synthesis in nerve terminals of diabetic rats. Neuroreport 1994, 5, 2421–2424. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Zhang, J.; Zhang, M.; Zhang, Ch.; Liu, H.; Li, W.; Fan, Y. Impact of diabetes mellitus on the risk of Alzheimer’s disease: a mendelian randomization study. BMC Neurology 2024, 24, 448. [Google Scholar] [CrossRef] [PubMed]
- Szutowicz, A. Aluminium, NO and nerve growth factor neurotoxicity in cholinergic neurons. J. Neurosci. Res. 2001, 66, 1009–1018. [Google Scholar] [CrossRef]
- Aschner, M.; Skalny, A.V.; Lu, R.; Martins, A.C.; Tsatsakis, A.; Miroshnikov, S.A.; Santamaria, A.; Tinkov, A.T. Molecular mechanisms of zinc oxide nanoparticles neurotoxicity. Chem. Biol. Interact. 2024, 403, 11124. [Google Scholar] [CrossRef]
- Savolainen, K.M.; Loikkanen, J.; Eerikäinen, S.; Naarala, J. Glutamate-stimulated ROS production in neuronal cultures: interactions with lead and the cholinergic system. Neurotoxicology 1998, 19(4-5), 669–74. [Google Scholar]
- Jankowska-Kulawy, A.; Klimaszewska-Łata, J.; Gul-Hinc, S.; Ronowska, A.; Szutowicz, A. Metabolic and cellular compartments of acetyl-CoA in the healthy and diseased brain. Int. J. Mol. Sci. 2022, 23, 10073. [Google Scholar] [CrossRef] [PubMed]
- Blusztajn, J.K.; Venturini, A.; Jackson, D.A.; Lee, H.J.; Wainer, B.H. Acetylcholine synthesis and release is enhanced by dibutyryl cyclic AMP in a neuronal cell line derived from mouse septum. J. Neurosci. 1992, 12(3), 793–799. [Google Scholar] [CrossRef]
- Butler, M. Cell counting and viability measurements in: Animal Cell Biotechnology. In Methods in Biotechnology; Humana Press, 1999. [Google Scholar]
- Fonnum, F. Radiochemical micro assays for the determination of choline acetyltransferase and acetylcholinesterase activities. Biochem. J. 1969, 115(3), 465–472. [Google Scholar] [CrossRef]
- Szutowicz, A.; Stepien, M.; Piec, G. Determination of pyruvate dehydrogenase and acetyl-CoA synthetase activities using citrate synthase. Anal. Biochem. 1981, 115(1), 81–7. [Google Scholar] [CrossRef]
- Bradford, M. A rapid sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Szutowicz, A.; Bielarczyk, H. Elimination of CoASH interferences form acetyl-CoA assay by maleic anhydride. Anal. Biochem. 1987, 164, 292–6. [Google Scholar] [CrossRef]
- Ding, L.; Zhexu, D.; Manjin, G.; Yanmei, H.; Kui, X. Metabolic enhancement of glycolysis and mitochondrial respiration are essential for neuronal differentiation. Cell Reprogram 2020, 22(6), 291–299. [Google Scholar]
- Simon, M.; Benary, U.; Baum, K.; Schramm, A.; Wolf, J. A computational model elucidates the effects of oncogene-induced expression alterations on the energy metabolism of neuroblastoma. Sci. Rep. 2025, 15, 32708. [Google Scholar] [CrossRef] [PubMed]
- Okechukwu, N.G.; Zaccone, C.; Barbera, L.; Nobili, A.; D’Amelio, M. Excitation–inhibition imbalance as a common thread linking early Alzheimer’s disease with temporal lobe epilepsy. Exp. Neurol. 2026, 397, 115581. [Google Scholar] [CrossRef]
- Koepsell, H. Glucose transporters in brain in health and disease. Pflugers Arch. 2020, 472(9), 1299–1343. [Google Scholar] [CrossRef]
- Rehni, A.K.; Dave, K.R. Impact of hypoglycemia on brain metabolism during diabetes. Mol. Neurobiol. 2018, 55(12), 9075–9088. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, S.; Moreira, P.I. Insulin-induced recurrent hypoglycemia up-regulates glucose metabolism in the brain cortex of chemically induced diabetic rats. Int. J. Mol. Sci. 2021, 22, 13470. [Google Scholar] [CrossRef]
- Ramakrishnan, R.K.; Srivastava, A.; Reeja, R.; Abusnana, S.; Mussa, B.M. Investigating the role of an immediate early gene FOS as a potential regulator of autophagic response to hypoglycemia in embryonic hypothalamic neurons. Clin. Transl. Sci. 2024, 17(3), e13749. [Google Scholar] [CrossRef]
- Rosenfeld, E.; Thornton, P.S.; Feingold, K.R.; Anawalt, B.; Blackman, M.R.; Boyce, A.; Chrousos, G.; Corpas, E.; de Herder, W.W.; Dhatariya, K.; et al. Hypoglycemia in neonates, infants and children in Endotext [Internet], 2023; MDText.com, Inc.: South Dartmouth (MA), 2000. [Google Scholar]
- Pattanaik, S.; Prusty, S.K.; Sahu, P.K. Exploring DPP IV inhibitors for Alzheimer’s disease: bridging diabetes and neurodegeneration. Brain Res. 2024, 149342. [Google Scholar] [CrossRef]
- Zott, B.; Konnerth, A. Impairments of glutamatergic synaptic transmission in Alzheimer’s disease. Semin. Cell Dev. Biol. 2023, 139, 24–34. [Google Scholar] [CrossRef]
- Suh, S.W.; Hamby, A.M.; Swanson, R.A. Hypoglycemia, brain energetics, and hypoglycemic neuronal death. Glia 2007, 55(12), 1280–86. [Google Scholar] [CrossRef]
- Auer, R.N. Hypoglycemic brain damage. Metab. Brain Dis. 2004, 19(3-4), 169–75. [Google Scholar] [CrossRef]
- Rohde, B.H.; Chiou, G.C. Effects of glucose on neuroblastoma in vitro and in vivo. J. Pharm. Sci. 1987, 76(5), 366–70. [Google Scholar] [CrossRef]
- Tokarz, D.A. Red deads, ghosts, and the afterlife: a guide to neuronal cell death. Toxicol. Pathol 2025, 53(8), 651–656. [Google Scholar] [CrossRef]
- Sahin, K.; Tuzcu, M.; Orhan, C.; Ali, S.; Sahin, N.; Gencoglu, H.; Ozkan, Y.; Hayirli, A.; Gozel, N.; Komorowski, J. Chromium modulates expressions of neuronal plasticity markers and glial fibrillaryacidic proteins in hypoglycemia-induced brain injury. Life Sci. 2013, 93, 1029–1048. [Google Scholar] [CrossRef] [PubMed]
- Sherin, A.; Peeyush, K.T.; Naijil, P.G.; Nandhu, M.S.; Jayanarayanan, S.; Jes, P.; Paulose, Ch.S. The effects of abnormalities of glucose homeostasis on the expression and binding of muscarinic receptors in brain cortex of rats. Eur. J. Pharmacol. 2011, 90, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Gao, R.; Huang, Q.; Huang, C.; Wang, L.; Lin, L.; He, G.; Wu, K.; Liu, X.; Liu, X.; et al. Lactate supplementation after hypoglycemia alleviates cognitive dysfunction induced by recurrent non-severe hypoglycemia in diabetic mice. Exp. Neurol. 2025, 383, 115037. [Google Scholar] [CrossRef]
- Li, D.; Ding, Z.; Gui, M.; Hou, Y.; Xie, K. Metabolic enhancement of glycolysis and mitochondrial respiration are essential for neuronal differentiation. Cell Reprogram 2020, 22(6), 291–299. [Google Scholar] [CrossRef] [PubMed]
- Szutowicz, A.; Jankowska, A.; Blusztajn, J.K.; Tomaszewicz, M. Acetylcholine and acetyl-CoA metabolism in differentiating SN56 septal cell line. J. Neurosci. Res. 1999, 57(1), 131–6. [Google Scholar] [CrossRef]
- Szutowicz, A.; Tomaszewicz, M.; Bielarczyk, H. Disturbances of acetyl-CoA, energy and acetylcholine metabolism in some encephalopathies. Acta Neurobiol. Exp. 1996, 56(1), 323–39. [Google Scholar] [CrossRef]
- Fontanella, R.A.; Ghosh, P.; Pesapane, A.; Taktaz, F.; Puocci, A.; Franzese, M.; Feliciano, M.F.; Tortorella, G.; Scisciola, L.; Sommella, E.; et al. Tirzepatide prevents neurodegeneration through multiple molecular pathways. J. Transl. Med. 2024, 22, 114. [Google Scholar] [CrossRef]
- Dienel, G.A. Lack of appropriate stechiometry: strong evidence against an energetically important astrocyte-neuron lactate shuttle in brain. J. Neurosci. Res. 2017, 95(11), 2103–125. [Google Scholar] [CrossRef]
- Siesjo, B.K. Mechanism of ischemic brain damage. In Problems in critical care; Shackford, S. R., Peral, A., Eds.; JB Lippincott: Philadelphia, 1987; pp. 611–27. [Google Scholar]
- Sapkota, S.; Briski, K.P. Sex-dimorphic effects of hypoglycemia on metabolic sensor mRNA expression in ventromedial hypothalamic nucleus-dorsomedial division (VMNdm) growth hormone-releasing hormone neurons. ACS Chem. Neurosci. 2024, 15(11), 2350–2358. [Google Scholar] [CrossRef] [PubMed]
- Koretz, B.; Ahern, K.B.; Wang, N.; Greenberg, D.A. Pre- and post-synaptic modulatorsof excitatory neurotransmission: comparative effects on hypoxia/hypoglycemia in cortical cultures. Brain Res. 1994, 18(643(1-2)), 334–7. [Google Scholar] [CrossRef]
- Nath, R.; Probert, A., Jr.; McGinnis, K.M.; Wang, K.K. Evidence for activation of caspase-3-like protease in excitotoxin- and hypoxia/hypoglycemia-injured neurons. J. Neurochem. 1998, 71(1), 186–95. [Google Scholar] [CrossRef]
- Wang, J.; Green, P.S.; Simpkins, W. Estradiol protects against ATP depletion, mitochondria membrane potential decline and generation of reactive oxygen species induced by 3-nitropropionic acid in SK-N-SH human neuroblastoma cells. J. Neurochem. 2001, 77, 804–811. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Lachance, B.B.; Mattson, M.P.; Jia, X. Glucose metabolic crosstalk and regulation in brain function and diseases. Prog. Neurobiol. 2021, 204, 102089. [Google Scholar] [CrossRef] [PubMed]
- de Bittencourt Pasquali, M.A.; de Ramos, V.M.; D′Oliveira Albanus, R.; Kunzler, A.; de Souza, L.H.T.; Dalmolin, R.J.S.; Gelain, D.P.; Ribeiro, L.; Carro, L.; Moreira, J.C.F. Gene expression profile of NF-κB, Nrf2, glycolytic, and p53 pathways during the SH-SY5Y neuronal differentiation mediated by retinoic acid. Mol. Neurobiol. 2016, 53, 423–435. [Google Scholar] [CrossRef]
- Klepinin, A.; Chekulayev, V.; Timohhina, N.; Shevchuk, I.; Tepp, K.; Kaldma, A.; Koit, A.; Saks, V.; Kaambre, T. Comparative analysis of some aspects of mitochondrial metabolism in differentiated and undifferentiated neuroblastoma cells. J. Bioenerg. Biomembr. 2014, 46(1), 17–31. [Google Scholar] [CrossRef]
- Andres, D.; Keyser, B.M.; Petrali, J.; Benton, B.; Hubbard, K.S.; McNutt, P.M.; Ray, R. Morphological and functional differentiation in BE(2)-M17 human neuroblastoma cells by treatment with trans-retinoic acid. BMC Neurosci. 2013, 14, 49. [Google Scholar] [CrossRef]
- Odelstad, L.; Phlman, S.; Nilsson, K.; Larsson, E.; Läckgren, G.; Johansson, K.E.; Hjertén, S.; Grotte, G. Neuron-specific enolase in relation to differentiation in human neuroblastoma. Brain Res. 1981, 224(1), 69–82. [Google Scholar] [CrossRef]
- Påhlman, S.; Ruusala, A.I.; Abrahamsson, L.; Odelstad, L.; Nilsson, K. Kinetics and concentration effects of TPA-induced differentiation of cultured human neuroblastoma cells. Cell Differ. 1983, 12(3), 165–70. [Google Scholar] [CrossRef]
- Marín-Hernández, A.; López-Ramírez, S.Y.; Del Mazo-Monsalvo, I.; Gallardo-Pérez, J.C.; Rodríguez-Enríquez, S.; Moreno-Sánchez, R.; Saavedra, E. Modeling cancer glycolysis under hypoglycemia, and the role played by the differential expression of glycolytic isoforms. FEBS J. 2014, 281(15), 3325–45. [Google Scholar] [CrossRef]
- Zyśk, M.; Bielarczyk, H.; Gul-Hinc, S.; Dyś, A.; Gapys, B.; Ronowska, A.; Sakowicz-Burkiewicz, M.; Szutowicz, A.; Kleinschmidt, M. Phenotype-dependent interactions between N-acetyl-L-aspartate and acetyl-CoA in septal SN56 cholinergic cells exposed to an excess of zinc. J. Alzheimer’s Dis. 2017, 56(3), 1145–1158. [Google Scholar] [CrossRef]
- Szutowicz, A.; Jankowska, A.; Blusztajn, J.K.; Tomaszewicz, M. Acetylcholine and acetyl-CoA metabolism in differentiating SN56 septal cell line. J. Neurosci. Res. 1999, 57, 131–136. [Google Scholar] [CrossRef]
- Jankowska, A.; Madziar, B.; Tomaszewicz, M.; Szutowicz, A. Acute and chronic effects of aluminum on acetyl-CoA and acetylcholine metabolism in differentiated and nondifferentiated SN56 cholinergic cells. J. Neurosci. Res. 2000, 62(4), 615–22. [Google Scholar] [CrossRef] [PubMed]
- Auld, D.S.; Mennicken, F.; Day, J.C.; Quirion, R. Neurotrophins differentially enhance acetylcholine release, acetylcholine content and choline acetyltransferase activity in basal forebrain neurons. J. Neurochem. 2001, 77, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Cheney, D.L.; LeFevere, H.F.; Racagni, G. Choline acetyltransferase activity and mass fragmentographic measurement of acetylcholine in specific nuclei and tracts of rat brain. Neuropharmacology 1975, 14(11), 801–9. [Google Scholar] [CrossRef]
- Vizi, S.E.; Palkovits, M. Acetylcholine content in different regions of the rat brain. Brain Res. Bull. 1978, 3(2), 93–96. [Google Scholar] [CrossRef]
- Bignami, F.; Bevilacqua, S.; Biagioni, S.; De Jaco, A.; Casamenti, F.; Felsani, A.; Augusti-Tocco, G. Cellular acetylcholine content and neuronal differentiation. J. Neurochem. 1997, 69(4), 1374–81. [Google Scholar] [CrossRef] [PubMed]
- Bielarczyk, H.; Tomaszewicz, M.; Madziar, B.; Cwikowska, J.; Pawełczyk, T.; Szutowicz, A. Relationships between cholinergic phenotype and acetyl-CoA level in hybrid murine neuroblastoma cells of septal origin. J. Neurosci. Res. 2003, 73(5), 717–21. [Google Scholar] [CrossRef]
- Kagawa, Y.; Umaru, B.A.; Shima, H.; Ito, R.; Zama, R.; Islam, A.; Kanno, S.I.; Yasui, A.; Sato, S.; Jozaki, K. FABP7 regulates acetyl-CoA metabolism through the interaction with ACLY in the nucleus of astrocytes. Mol. Neurobiol. 2020, 57, 4891–4910. [Google Scholar] [CrossRef] [PubMed]
- Barrett, G.L.; Naim, T.; Trieu, J.; Huang, M. In vivo knockdown of basal forebrain p75 neurotrophin receptor stimulates cholinę acetyltransferase activity in the mature hippocampus. J. Neurosci. Res. 2016, 94, 389–400. [Google Scholar] [CrossRef] [PubMed]
- Szutowicz, A.; Bielarczyk, H.; Jankowska-Kulawy, A.; Pawełczyk, T.; Ronowska, A. Acetyl-CoA the key factor for survival or death of cholinergic neurons in course of neurodegenerative diseases. Neurochem. Res. 2013, 38(8), 1523–42. [Google Scholar] [CrossRef] [PubMed]
- Gallego-Rudolf, J.; Wiesman, A.I.; Binette, A.P.; Villeneuve, S.; Baillet, S. Synergistic association of Aβ and tau pathology with cortical neurophysiology and cognitive decline in asymptomatic older adults. Nat. Neurosci. 2024, 27(11), 2130–7. [Google Scholar] [CrossRef]
- Angelova, P.R.; Abramov, A.Y. Interplay of mitochondrial calcium signalling and reactive oxygen species production in the brain. Biochem. Soc. Trans. 2024, 52(4), 1939–1946. [Google Scholar] [CrossRef]
- Granzotto, A.; Canzoniero, L.M.T.; Sensi, S.L. A neurotoxic ménage-à-trois: glutamate, calcium, and zinc in the excitotoxic cascade. Front. Mol. Neurosci. 2020, 600089. [Google Scholar] [CrossRef]
- Kho, A.R.; Choi, B.Y.; Lee, S.H.; Hong, D.K.; Jeong, J.H.; Kang, B.S.; Kang, D.H.; Park, K.H.; Park, J.B.; Suh, S.W. The effects of sodium dichloroacetate on mitochondrial dysfunction and neuronal death following hypoglycemia-induced injury. Cells 2019, 8(5), 405. [Google Scholar] [CrossRef]
- Paulsen, M.E.; Rao, R.B. Cerebral effects of neonatal dysglycemia. Clin. Perinatol. 2022, 49(2), 405–426. [Google Scholar] [CrossRef]
|
Additions mM |
Nondifferentiated SN56 | Differentiated SN56 | ||
| 1mM Glucose | 25 mM Glucose | 1mM Glucose | 25 mM Glucose | |
| A | Whole cell count (103/cm2) | |||
| Control Aβ 0.001 SNP 0.4 Aβ 0.001+SNP 0.4 |
24.0±1.1*** 23.7±2.2*** 13.2±2.2***‡ 11.2±1.9***‡‡ |
97.0±7.9 92.3±6.4 44.0±4.6‡‡ 30.8±3.6‡‡‡ |
29.0±0.7*** 36.4±5.4*** 15.9±4.8**‡ 15.8±3.5**‡‡ |
71.0±5.6 85.0±7.9 39.1±5.8‡‡ 38.8±5.9‡‡ |
| B | Trypan blue positive cells (%) | |||
| Control Aβ 0.001 SNP 0.4 Aβ 0.001+SNP 0.4 |
14.0±3.3* 21.9 ± 2.8** 97.0±12.5***‡‡‡ 100.0±3.7***‡‡‡ |
3.3±1.6 6.5±1.3 36.9±1.1‡‡‡ 36.8±0.3‡‡‡ |
18.6±2.9††† 16.9±3.4 35.3±4.8†††*** 57.3±1.8†††* |
7.8±1.4††† 13.8±3.4†‡ 77.1±2.3†††‡‡‡ 64.3±0.5†††‡‡‡ |
| C | Viable cell count (103/cm2) | |||
| Control Aβ 0.001 SNP 0.4 Aβ 0.001+SNP 0.4 |
20.2±0.9*** 18.5±1.7*** 0.4±0.1***‡‡‡ 0***‡‡‡ |
94.7±7.6 86.3±6.0 27.8±2.9‡‡‡ 19.5±2.3‡‡‡ |
23.6±0.6*** 30.2±0.9***† 10.3±3.1‡ 6.7±1.5‡‡ |
65.5±5.2††† 73.3±6.8† 9.0±1.3†††‡‡‡ 13.9±2.1†‡‡‡ |
| D | PDHC specific activity(nmol/min./mg protein) | |||
| Control Aβ 0.001 SNP 0.4 Aβ 0.001+SNP 0.4 |
6.8±0.6* 6.4±0.52 3.4±0.2‡‡‡ 5.4±0.4‡‡‡ |
9.0±0.6 8.1±0.7 6.5±0.2‡‡ 4.0±0.4‡‡‡ |
6.8±0.3* 5.8 ±0.3 4.4±0.2‡‡ 4.7±0.5‡‡ |
8.1±0.2 7.3±0.7 6.2±0.4‡ 5.7±0.4‡ |
| E | Acetyl-CoA content (pmol/mg protein) | |||
| Control Aβ 0.001 SNP 0.4 Aβ 0.001+SNP 0.4 |
34.9±3.8** 34.5±5.4 22.6±5.5‡ 30.8±2.5* |
42.9±2.1 27.9±1.7‡ 17.6±2.7‡‡‡ 21.2±2.7‡‡ |
20.1±1.9**††† 15.6±2.4†† 9.8±5.1‡ 9.0±0.7†††‡‡‡ |
26.7±1.6††† 20.9±1.7† 10.1±0.1†‡‡‡ 6.2±0.9†††‡‡‡ |
| F | ChAT specific activity (nmol/min./mg protein) | |||
| Control Aβ 0.001 SNP 0.4 Aβ 0.001+SNP 0.4 |
0.08±0.01 0.08 ±0.01 0.06±0.01 0.05±0.01 |
0.13±0.01 0.11±0.02 0.08±0.01 0.04±0.02‡ |
0.38±0.04***††† 0.26±0.01***†† 0.22±0.02**††‡‡ 0.21±0.03**††‡ |
1.11±0.04††† 0.99±0.10††† 0.72±0.11†††‡‡‡ 0.78±0.11†††‡ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




