Submitted:
28 February 2026
Posted:
28 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Experimental Site and Materials
2.2. Experimental Design
2.3. Sample Collection and Processing
2.4. Measurement Indicators and Methods
2.4.1. Soil Physicochemical Property Measurements
2.4.2. Soil Enzyme Activity Measurements
2.4.3. Microbial Community Analysis
2.5. Data Analysis
3. Results
3.1. Spatiotemporal Effects of Spent Substrate Application on Soil Physicochemical Properties
| Trt | Layer | Period | OM (%) | TN (mg/kg) | TP (mg/kg) | AN (mg/kg) | AP (mg/kg) | AK (mg/kg) | pH | EC (S/cm) |
|---|---|---|---|---|---|---|---|---|---|---|
| T1 | S | I | 1.74±0.08a | 478.9±85.4a | 38.2±8.7a | 51.3±1.4a | 51.8±4.6a | 117.2±11.7a | 8.22±0.11a | 16.55±2.33a |
| T1 | S | M | 0.65±0.03g | 666.8±11.1f | 53.6±6.8g | 50.3±4.6a | 15.8±0.5d | 328.3±7.8f | 5.50±0.17g | 43.43±8.40e |
| T1 | S | F | 0.39±0.06m | 317.8±16.6k | 531.2±11.4k | 29.3±0.6g | 46.2±1.9g | 189.1±4.6h | 6.56±0.02j | 324.67±19.06j |
| T1 | D | I | 0.52±0.08b | 276.6±22.8b | 31.4±3.1b | 15.1±4.2b | 10.4±1.1b | 61.5±4.8b | 6.05±0.19b | 19.75±2.29b |
| T1 | D | M | 0.58±0.02h | 544.9±16.9g | 36.8±1.6e | 21.5±1.3f | 11.8±0.7b | 88.9±3.4g | 6.00±0.08b | 52.10±13.79f |
| T1 | D | F | 0.26±0.05n | 239.0±16.7l | 383.2±3.5l | 24.3±2.2i | 50.1±3.1h | 106.3±7.8j | 6.64±0.04k | 438.33±126.87k |
| T2 | S | I | 2.00±0.10c | 571.1±86.7c | 43.1±4.3c | 63.1±3.9c | 50.7±7.0a | 108.7±11.5c | 7.67±0.09c | 19.70±2.50b |
| T2 | S | M | 0.86±0.03i | 1165.2±50.1h | 49.9±1.8h | 51.2±1.8a | 14.2±0.8e | 192.5±9.6h | 5.46±0.14g | 41.70±8.72e |
| T2 | S | F | 0.49±0.07o | 404.8±22.9m | 520.6±8.0m | 42.6±2.1j | 48.4±1.3i | 219.5±4.9k | 6.58±0.03j | 331.33±26.08j |
| T2 | D | I | 0.99±0.22d | 437.8±59.4d | 35.1±1.2d | 26.8±6.0d | 9.9±0.9b | 67.1±2.3d | 5.86±0.06d | 16.87±2.53c |
| T2 | D | M | 0.68±0.04j | 869.4±24.0i | 40.4±1.7i | 29.6±0.9g | 10.8±0.4b | 98.8±3.5i | 5.65±0.15h | 33.03±4.27g |
| T2 | D | F | 0.35±0.02p | 336.7±10.5n | 400.6±15.9n | 31.8±2.2k | 48.5±2.1i | 114.2±3.7l | 6.59±0.04j | 332.00±26.54j |
| T3 | S | I | 2.18±1.10e | 495.8±54.9e | 36.8±1.6e | 50.9±12.8a | 52.4±3.8c | 91.0±27.2e | 7.45±0.18e | 18.71±5.17b |
| T3 | S | M | 0.79±0.03k | 891.4±27.2j | 62.0±2.9j | 37.3±0.5h | 15.7±0.7d | 195.5±5.4h | 5.49±0.09g | 36.73±2.48h |
| T3 | S | F | 0.40±0.03q | 390.9±14.3o | 749.6±54.3o | 41.9±1.9j | 46.9±2.2g | 180.4±8.6m | 6.58±0.01j | 350.67±12.50l |
| T3 | D | I | 0.66±0.13f | 281.9±98.8b | 29.8±0.6f | 19.9±1.0e | 11.2±2.0b | 67.2±1.5d | 5.56±0.11f | 23.96±5.40d |
| T3 | D | M | 0.61±0.02l | 665.5±20.8f | 49.5±0.7h | 23.8±0.4i | 12.8±0.4f | 108.5±2.3j | 5.33±0.07i | 28.73±0.64i |
| T3 | D | F | 0.24±0.01r | 278.9±12.0l | 401.9±9.9n | 28.0±1.6l | 45.2±1.2j | 114.7±7.1l | 6.62±0.02k | 270.33±26.21m |
3.2. Spatiotemporal Effects of Spent Substrate Application on Soil Enzyme Activities
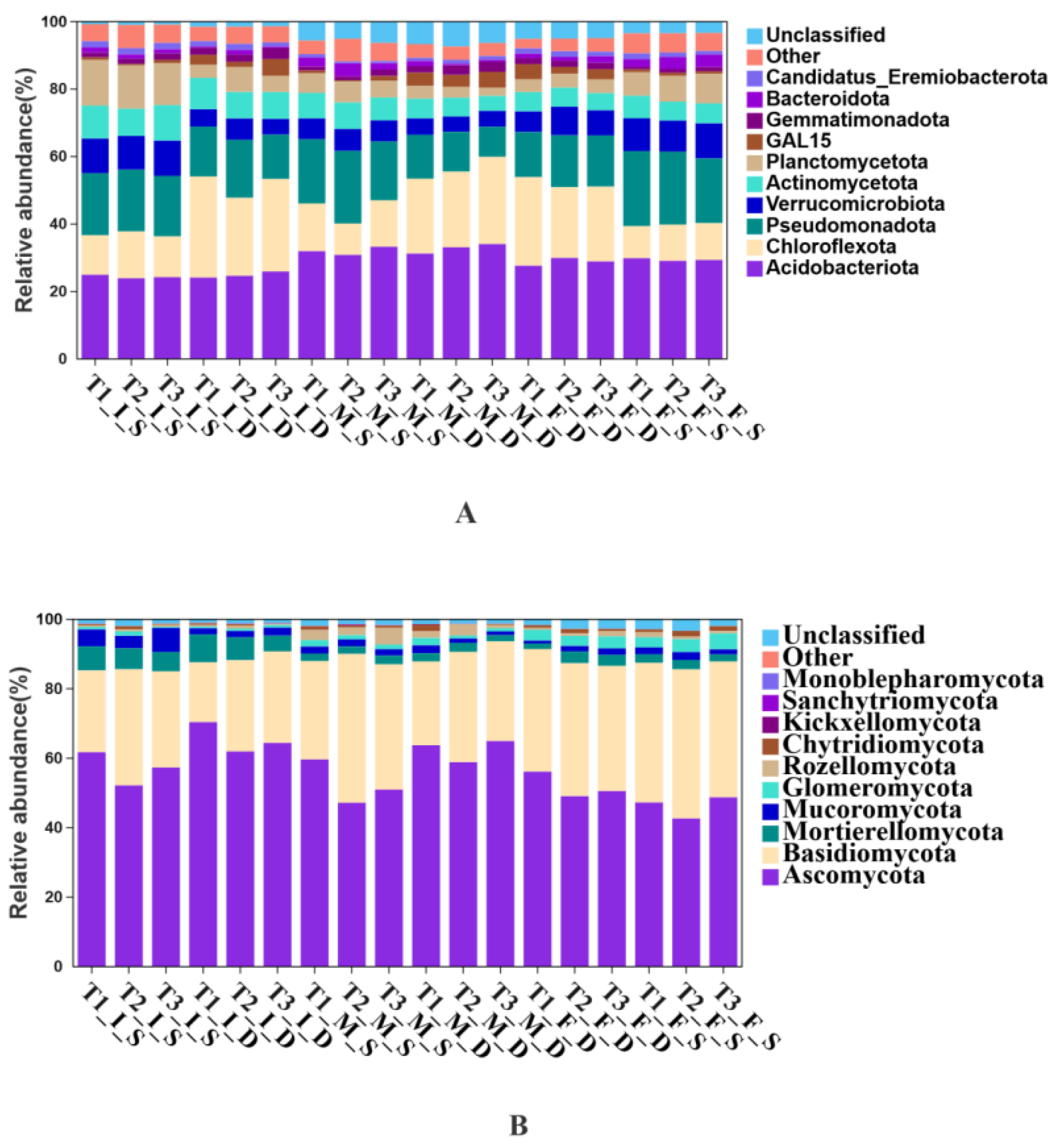
3.3. Effects of Spent Substrate Application on Soil Microbial Community Composition
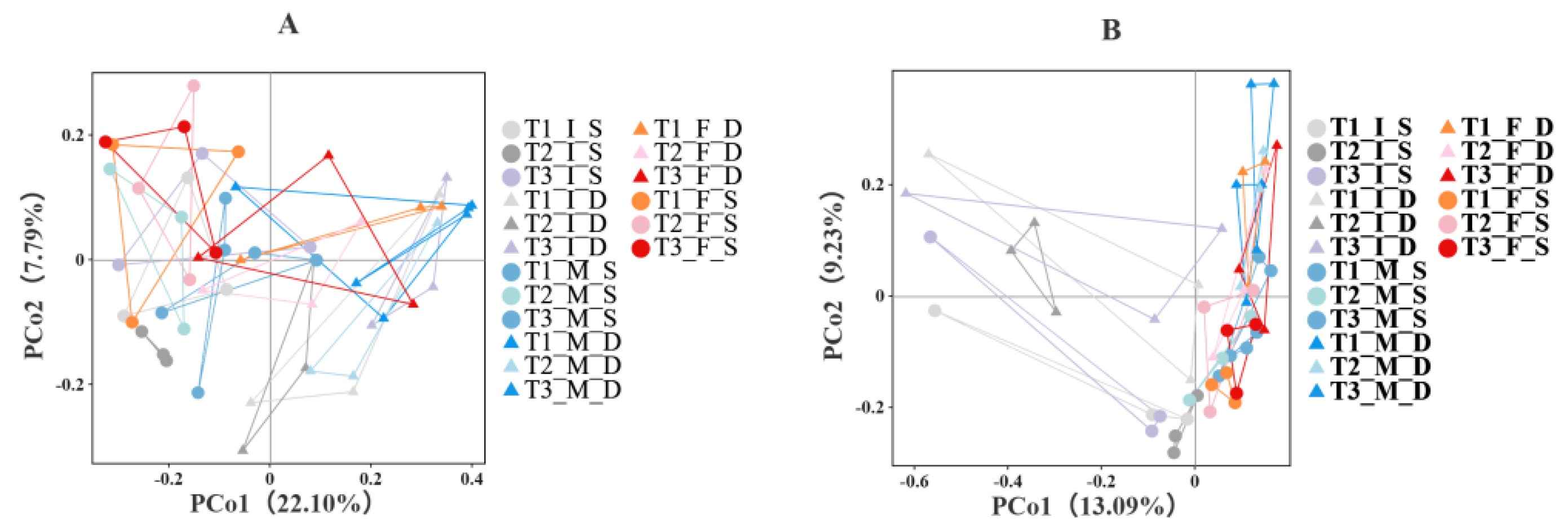
3.4. Effects of Spent Substrate Application on Soil Microbial Community Structure
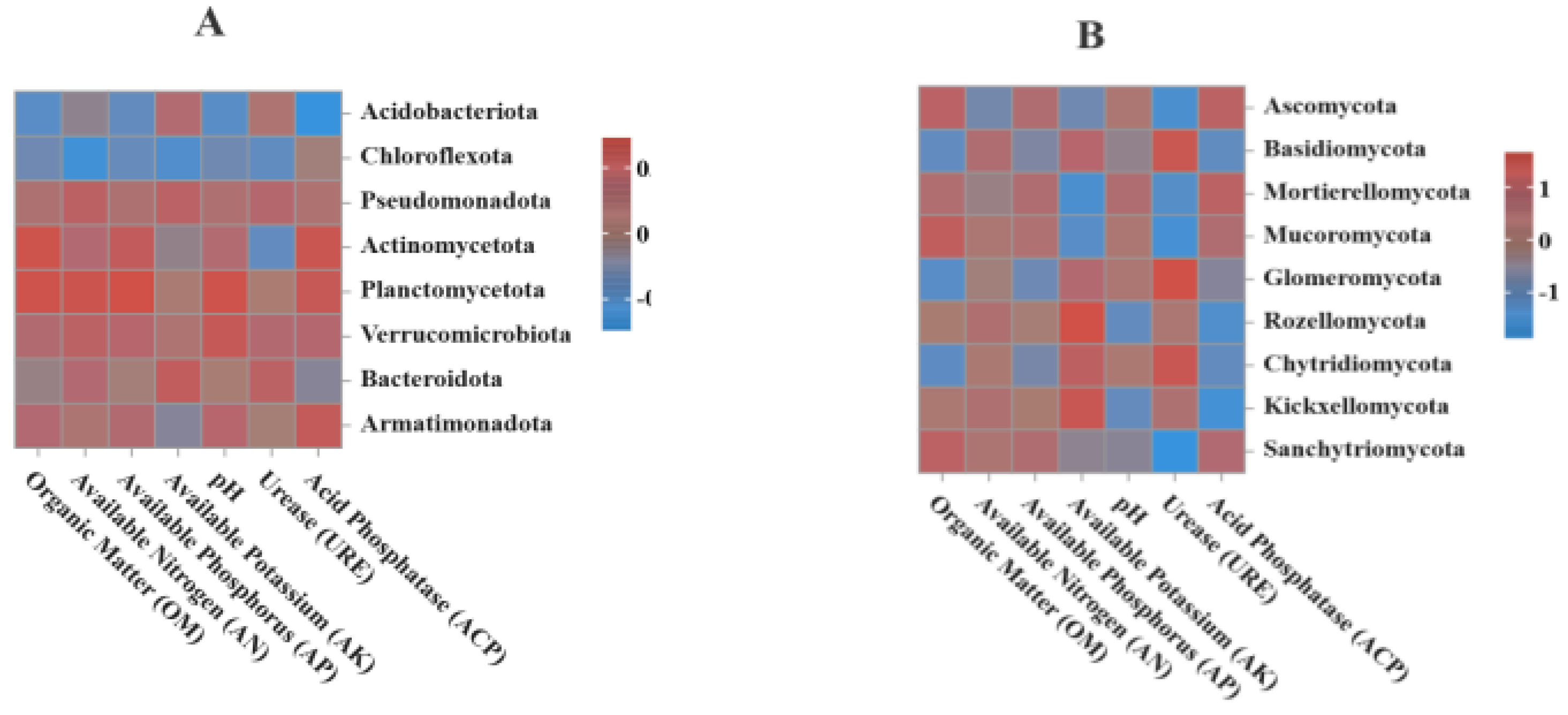
3.5. Correlation Analysis Between Soil Microbial Communities and Environmental Factors
4. Discussion
4.1. Synergistic and Differential Responses of Soil Multifunctional Enzyme Activities
4.2. Functional Directional Succession of Microbial Communities
4.3. Environmental Factor-Driven "Functional-Community" Association Network
4.4. Practical Implications and Prospects
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| SMS | Spent mushroom substrate |
| TP | Total phosphorus |
| AP | Available phosphorus |
| AK | Available potassium |
| TN | Total nitrogen |
| SOM | Soil organic matter |
| EC | Electrical conductivity |
| UA | Urease activity |
| SA | Sucrase activity |
| ACP | Acid phosphatase |
| CAT | Catalase |
| APR | Acid protease |
| PCoA | Principal coordinate analysis |
| PERMANOVA | Permutational multivariate analysis of variance |
| LEfSe | Linear discriminant analysis effect size |
| ANOVA | Analysis of variance |
References
- FAO. (2025). Global mushroom production statistics. Food and Agriculture Organization.
- Sheer, A., Sardar, M. F., Younas, F., et al. (2024). Trends and social aspects in the management and conversion of agricultural residues into valuable resources: A comprehensive approach to counter environmental degradation, food security, and climate change. Bioresource Technology, 394, 130258. [CrossRef]
- Tang, Q., Liu, W., Huang, H., et al. (2024). Responses of Crop Yield, Soil Fertility, and Heavy Metals to Spent Mushroom Residues Application. Plants, 13(5), 663. [CrossRef]
- Zhao, F., Shi, S., Meng, R., et al. (2023). Soil Habitats Are Affected by Fungal Waste Recycling on Farmland in Agro-Pastoral Ecotone in Northern China. Agronomy, 13(9), 2432. [CrossRef]
- Idowu, O. O., Akintola, O. A., Emmanuel, O. C., et al. (2023). Utilization of spent mushroom wastes as an alternative to use of inorganic fertilizer. International Journal of Vegetable Science, 29(6), 516-526. [CrossRef]
- Zhang, B., Su, S., Chen, F., et al. (2015). Effects of mulch with mushroom residue on soil enzyme activities and soil physical-chemical property in Corylus orchard. Nonwood Forest Research.
- Yuan, X., Qin, H., Wang, Y., et al. (2026). Coupled Effects of Tree Species and Understory Morel on Modulating Soil Microbial Communities and Nutrient Dynamics. Microorganisms, 14(1), 99. PMID: 41194727. [CrossRef]
- Sunil, C., & Xu, B. (2022). Mycochemical profile and health-promoting effects of morel mushroom Morchella esculenta (L.) – A review. Food Research International, 159, 111571. [CrossRef]
- Wu, H., Chen, J., Li, J., et al. (2021). Recent Advances on Bioactive Ingredients of Morchella esculenta. Applied Biochemistry and Biotechnology, 193(12), 4197-4213. [CrossRef]
- Li, J., Wu, H., Liu, Y., et al. (2021). The chemical structure and immunomodulatory activity of an exopolysaccharide produced by Morchella esculenta under submerged fermentation. Food & Function, 12(19), 9327-9338. [CrossRef]
- Gao, C., Liu, P., Li, W., et al. (2025). Preparation of Morchella esculenta protein and its preventive effect on nonalcoholic fatty liver disease in mice. Food & Function, 16(3), 1086-1099. [CrossRef]
- Wang, C. Y. (2020). A review on the potential reuse of functional polysaccharides extracted from the by-products of mushroom processing. Food and Bioprocess Technology, 13(2), 217-228. [CrossRef]
- Törős, G., El-Ramady, H., Abdalla, N., et al. (2025). Valorization of Mushroom Residues for Functional Food Packaging. International Journal of Molecular Sciences, 26(22), 10870. [CrossRef]
- Domínguez-Núñez, J. A., & Oliet, J. A. (2022). Management of mushroom resources in Spanish forests: a review. Forestry: An International Journal of Forest Research, 96(2), 135-154. [CrossRef]
- Fierer, N. (2017). Embracing the unknown: disentangling the complexities of the soil microbiome. Nature Reviews Microbiology, 15(10), 579-590. [CrossRef]
- Bose, T., Vivas, M., Slippers, B., et al. (2023). Retention of post-harvest residues enhances soil fungal biodiversity in Eucalyptus plantations. Forest Ecology and Management, 532, 120806. [CrossRef]
- Kielak, A. M., Barreto, C. C., Kowalchuk, G. A., et al. (2016). The ecology of Acidobacteria: moving beyond genes and genomes. Frontiers in Microbiology, 7, 744. [CrossRef]
- Tan, X., Cao, J., Liu, J., et al. (2024). Characteristics of three organic fertilizers and their influence on the mobility of cadmium and arsenic in a soil-rice (Oryza sativa L.) system. Environmental Science and Pollution Research, 31(37), 49469-49480. [CrossRef]
- Mawarda, P. C., Le Roux, X., Dirk van Elsas, J., et al. (2020). Deliberate introduction of invisible invaders: A critical appraisal of the impact of microbial inoculants on soil microbial communities. Soil Biology and Biochemistry, 148, 107874. [CrossRef]
- Wei, Z., Liu, L., Lei, Y., et al. (2024). Establishment of Pinus massoniana–Lactarius hatsudake Symbiosis. Forests, 15(4), 578. [CrossRef]
- Leonardi, M., Furtado, A. N. M., Comandini, O., et al. (2020). Halimium as an ectomycorrhizal symbiont: new records and an appreciation of known fungal diversity. Mycological Progress, 19(12), 1495-1509. [CrossRef]
- Phanpadith, P., Yu, Z., Yu, D., et al. (2019). Promotion of maize growth by a yellow morel, Morchella crassipes. Symbiosis, 80(1), 33-41. [CrossRef]
- Kang, X., Liu, T., Li, J., Martin, F.M., Tan, H., et al. (2025). Biochar amendment improves Morchella sextelata yield by enhancing soil NO3--N availability and increasing fungal diversity. Microbiological Research, 302, 128325. PMID: 40913927. [CrossRef]
- Virk, A.L., Shakoor, A., Ahmad, N., Du, H., Chang, S.X., Cai, Y. (2025). Organic amendments restore soil biological properties under pesticides application. Pesticide Biochemistry and Physiology, 210, 106394. PMID: 40262872. [CrossRef]
- Wu, Y., Qi, L., et al. (2026). Soil calcium ion enrichment associated with altered microbial functional gene networks in bamboo forest expansion. Applied Soil Ecology, 218, 106685. [CrossRef]
- Li, S., et al. (2025). Comparative analysis of soil properties before and after Morchella sextelata cultivation across various soil types. Frontiers in Microbiology, 16, 1700246. PMC12602436. [CrossRef]
- Nuralykyzy, B., Nie, J., Mei, H., Zhang, Y., Rogers, K.M., Li, C., Yuan, Y. (2025). Synergies between Carbon Sequestration, Nitrogen Utilization, and Mushroom Quality: A Comprehensive Review. Journal of Agricultural and Food Chemistry, 73(23), 14144-14157. PMID: 40406890. [CrossRef]
- Quisth, I., Ulfsparre, I., Müller, B., Passoth, V., Solberg, S.Ø., Martín, C., Romero-Soto, L.A., Pent, M., Mousavi, H. (2026). Spent mushroom substrate (SMS) as a sustainable soil amendment and biofertilizer: A review. Agricultural and Food Science, 34(4), 323-338. DOI: 10.23986/afsci.163895. [CrossRef]
- Yang, Z.Y., Zhao, J.C., Ni, H.J. (2025). Silkworm excrement organic fertilizer substitution compound fertilizer improves bamboo shoot yield by altering soil properties and bacterial communities of Moso bamboo forests. Frontiers in Plant Science, 16, 1550946. [CrossRef]
- Huang, Z., et al. (2025). On- and off-year management-induced changes in microbial communities cause microbial necromass carbon variation in subtropical Moso bamboo forests. Applied Soil Ecology, 204, 105722. [CrossRef]
- Richardson, A. E., & Simpson, R. J. (2011). Soil microorganisms mediating phosphorus availability. Plant Physiology, 156(3), 989-996. [CrossRef]
- Negassa, W., & Leinweber, P. (2009). How does the Hedley sequential phosphorus fractionation reflect impacts of land use and management on soil phosphorus: a review. Journal of Plant Nutrition and Soil Science, 172(3), 305-325. [CrossRef]
- Blagodatskaya, E., & Kuzyakov, Y. (2008). Mechanisms of real and apparent priming effects and their dependence on soil microbial biomass and community structure: critical review. Biology and Fertility of Soils, 45(2), 115-131. [CrossRef]
- Burns, R. G., DeForest, J. L., Marxsen, J., et al. (2013). Soil enzymes in a changing environment: Current knowledge and future directions. Soil Biology and Biochemistry, 58, 216-234. [CrossRef]
- Sinsabaugh, R. L. (2010). Phenol oxidase, peroxidase and organic matter dynamics of soil. Soil Biology and Biochemistry, 42(3), 391-404. [CrossRef]
- Nannipieri, P., Giagnoni, L., Landi, L., et al. (2011). Role of phosphatase enzymes in soil. In E. Bünemann, A. Oberson, & E. Frossard (Eds.), Phosphorus in Action: Biological Processes in Soil Phosphorus Cycling (pp. 215-243). Springer Berlin Heidelberg.
- Hartmann, M., Frey, B., Mayer, J., et al. (2015). Distinct soil microbial diversity under long-term organic and conventional farming. The ISME Journal, 9(5), 1177-1194. [CrossRef]
- Floudas, D., Binder, M., Riley, R., et al. (2012). The Paleozoic origin of enzymatic lignin decomposition reconstructed from 31 fungal genomes. Science, 336(6089), 1715-1719. [CrossRef]
- Eilers, K. G., Debenport, S., Anderson, S., et al. (2012). Digging deeper to find unique microbial communities: the strong effect of depth on the structure of bacterial and archaeal communities in soil. Soil Biology and Biochemistry, 50, 58-65. [CrossRef]
- Rousk, J., Bååth, E., Brookes, P. C., et al. (2010). Soil bacterial and fungal communities across a pH gradient in an arable soil. The ISME Journal, 4(10), 1340-1351. [CrossRef]
- Hao, H., Yue, Y., Chen, Q., et al. (2024). Effects of an efficient straw decomposition system mediated by Stropharia rugosoannulata on soil properties and microbial communities in forestland. Science of The Total Environment, 916, 170226. [CrossRef]
- Liu, Q., He, G., Wei, J., et al. (2021). Comparative transcriptome analysis of cells from different areas reveals ROS responsive mechanism at sclerotial initiation stage in Morchella importuna. Scientific Reports, 11(1). [CrossRef]
- Miina, J., Muttilainen, H., Vornanen, J., et al. (2023). Supply Chain Actors’ Perspectives Concerning the Cultivation of Specialty Wood-Decay Mushrooms in Finland. Forests, 14(1), 134. [CrossRef]
- Legendre, P., & Legendre, L. (2012). Numerical Ecology (3rd ed.). Elsevier.
| Trt | Layer | Period | UA (U/g) | SA (U/g) | ACP (nmol/d/g) | CAT (mol/d/g) | APR (U/g) |
|---|---|---|---|---|---|---|---|
| T1 | S | I | 13.9±1.6a | 127.8±21.4a | 503.8±33.0a | 45.3±6.8a | 188.5±36.7a |
| T1 | D | I | 6.9±2.9b | 48.2±18.0b | 421.9±20.0b | 50.4±15.2b | 211.5±35.1b |
| T2 | S | I | 23.9±9.8c | 155.2±26.9c | 482.4±40.5c | 49.2±8.4c | 158.7±28.7c |
| T2 | D | I | 15.2±7.4d | 48.4±8.1b | 406.6±25.4d | 72.7±11.9d | 178.4±25.4d |
| T3 | S | I | 19.4±9.2e | 140.0±31.8d | 559.0±104.1e | 39.3±3.7e | 171.3±87.3e |
| T3 | D | I | 8.2±4.7f | 46.4±23.8b | 502.7±101.3f | 59.3±23.8f | 162.8±39.0f |
| T1 | S | M | 39.3±1.0g | 21.5±1.6e | 260.6±12.4g | 26.0±1.3g | 198.0±10.2a |
| T1 | D | M | 35.5±1.3h | 14.5±1.2f | 241.5±3.5h | 51.2±1.8b | 175.0±5.6d |
| T2 | S | M | 42.1±0.6i | 23.9±0.3g | 253.6±5.1i | 43.3±4.6a | 157.9±10.6c |
| T2 | D | M | 39.3±0.7g | 16.8±0.4h | 236.2±5.8j | 58.2±1.3f | 139.5±3.5g |
| T3 | S | M | 43.6±0.8j | 17.6±0.5i | 264.6±9.9g | 42.2±2.4a | 159.3±4.8c |
| T3 | D | M | 41.8±0.8k | 12.7±0.4j | 250.5±2.1k | 60.5±1.9h | 130.7±1.9h |
| T1 | S | F | 212.9±6.4l | 546.7±3.1k | 354.2±6.2l | 26.99±8.66g | 89.8±6.0i |
| T1 | D | F | 63.2±4.2m | 317.6±6.2l | 354.2±6.2l | 37.08±9.00i | 90.9±5.9i |
| T2 | S | F | 245.2±18.9n | 780.1±21.6m | 361.7±19.0m | 22.27±4.55j | 86.8±8.3j |
| T2 | D | F | 77.3±6.4o | 478.7±29.3n | 361.7±19.0m | 42.12±5.13k | 86.8±8.3j |
| T3 | S | F | 221.1±11.4p | 641.8±38.8o | 348.0±20.2n | 23.24±6.04l | 79.9±6.9k |
| T3 | D | F | 80.4±7.1q | 488.5±38.1p | 348.0±20.2n | 45.58±5.48m | 79.9±6.9k |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




