Submitted:
27 February 2026
Posted:
28 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Molecular and Cellular Mechanisms of Radiation Hormesis in the Central Nervous System
2.1. Modulation of Redox Homeostasis and Antioxidant Defense
2.2. Neuroimmunomodulation and Systemic Anti-inflammatory Response
2.3. Regulation of Cellular Signaling, Proliferation, and Synaptic Plasticity
2.4. Induction of Proteostasis and DNA Repair Mechanisms
2.5. Mitohormesis and Metabolic Remodeling
3. Neurobiological Effects of Low-Dose Ionizing Radiation in Experimental In Vivo Models of Neurodegenerative Diseases
3.1. Effects of LDIR in Experimental Models of Alzheimer’s Disease
3.2. Impact of LDIR on Pathogenetic Targets in in vivo Models of Parkinson’s Disease
3.3. Experimental Evidence and Prospects for Modulating Amyotrophic Lateral Sclerosis Pathogenesis via LDIR
3.4. Potential Applications of LDIR in Traumatic Injuries of the Nervous System
4. Clinical Translation Prospects of LDIR as a Systemic Modulator of Neurodegeneration
5. Concluding Remarks and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 2BS | Human embryonic lung diploid fibroblast cell line |
| A549 | Adenocarcinomic human alveolar basal epithelial cell line |
| AD | Alzheimer's disease |
| Akt (PKB) | Protein kinase B |
| ALS | Amyotrophic lateral sclerosis |
| ARE | Antioxidant response element |
| ATM | Ataxia-telangiectasia mutated (serine/threonine kinase) |
| Aβ | Amyloid-beta peptide |
| BDNF | Brain-derived neurotrophic factor |
| C9orf72 | Chromosome 9 open reading frame 72 protein |
| CAT | Catalase |
| CCR2 | C-C chemokine receptor type 2 |
| CCS | Copper chaperone for superoxide dismutase |
| cGAS | Сyclic GMP-AMP synthase |
| CNS | Central nervous system |
| CSCs | Cancer stem cells |
| CT | Computed tomography |
| DNA | Deoxyribonucleic acid |
| DSB | DNA double-strand break |
| DSBR | Double-strand break repair |
| EGb761 | Standardized extract of Ginkgo biloba |
| EMT | Epithelial-mesenchymal transition |
| ERK | Extracellular signal-regulated kinases |
| FIS1 | Mitochondrial fission protein 1 |
| FUS/TLS | Fused in Sarcoma/ Translocated in Liposarcoma protein |
| GAP-43 | Growth-associated protein 43 |
| G-CSF | Granulocyte colony-stimulating factor |
| GFAP | Glial fibrillary acidic protein |
| GM-CSF | Granulocyte-macrophage colony-stimulating factor |
| GPx | Glutathione peroxidase |
| GSH | Glutathione |
| GST | Glutathione S-transferase |
| Gy | Gray |
| HDIR | High-dose ionising radiation |
| HDMEC | Human dermal microvascular endothelial cells |
| HNSCC | Head and Neck Squamous Cell Carcinoma |
| HO-1 | Heme oxygenase-1 |
| HSP27/HSP70 | Heat shock protein 27 / Heat shock protein 70 |
| IL-1β, IL-6, IL-10, IL-2β | Interleukin-1 beta / 6 / 10 / 2 beta |
| JAK1 | Janus kinase 1 |
| JNK | c-Jun N-terminal kinases |
| Keap1 | Kelch-like ECH-associated protein 1 |
| LDIR | Low-dose ionising radiation |
| LET | Linear energy transfer |
| LRRK2 | Leucine-rich repeat kinase 2 |
| MAPK | Mitogen-activated protein kinase |
| MCF-7 | Breast cancer side population cells |
| MCP-1 | Monocyte chemoattractant protein-1 (also known as CCL2) |
| MDA | Malondialdehyde |
| MFN1 | Mitofusin-1 |
| MIP-1α | Macrophage inflammatory protein-1α |
| MMR | Mismatch repair |
| MnSOD | Manganese superoxide dismutase (also known as SOD2) |
| MPTP | 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine |
| mROS | Mitochondrial reactive oxygen species |
| mtDNA | Mitochondrial DNA |
| NDDs | Neurodegenerative diseases |
| NF-κB | Nuclear factor kappa-light-chain-enhancer of activated B cells |
| NGF | Nerve growth factor |
| NHF | Normal human fibroblasts |
| Notch1 | Neurogenic locus notch homolog protein 1 |
| NPY | Neuropeptide Y |
| NQO1 – NAD(P)H | Quinone oxidoreductase 1 – NAD(P)H |
| Nrf2 | Nuclear factor erythroid 2-related factor 2 |
| NSCLC | Non-small cell lung cancer |
| p53 | Tumor protein p53 |
| PARK2 | Parkin RBR E3 ubiquitin protein ligase (PRKN, PARKIN) |
| PARP1 | Poly [ADP-ribose] polymerase 1 |
| PCP4 | Purkinje cell protein 4 (also known as PEP-19) |
| PD | Parkinson's disease |
| PET | Positron emission tomography |
| PI3K | Phosphoinositide 3-kinase |
| PIAS | Protein inhibitor of activated STAT |
| Prdx2 | Peroxiredoxin-2 |
| pTDP43 | Phosphorylated TAR DNA-binding protein 43 |
| ROS | Reactive oxygen species |
| SOCS | Suppressor of cytokine signaling |
| SOD | Superoxide dismutase |
| SP cells | Side population cells |
| STAT1/STAT3 | Signal transducer and activator of transcription 1 / 3 |
| STING | Stimulator of interferon genes |
| STMN2 | Stathmin-2 (also known as SCG10) |
| Sv | Sievert |
| TGF- β | Transforming growth factor-beta |
| TNF-α | Tumor necrosis factor-alpha |
| TREM2 | Triggering receptor expressed on myeloid cells 2 |
| VEGF | Vascular endothelial growth factor |
References
- Vaiserman, A.; Cuttler, J.M.; Socol, Y. Low-dose ionizing radiation as a hormetin: experimental observations and therapeutic perspective for age-related disorders. Biogerontology 2021, 22, 145–164. [Google Scholar] [CrossRef]
- Paithankar, J.G.; Gupta, S.C.; Sharma, A. Therapeutic potential of low dose ionizing radiation against cancer, dementia, and diabetes: evidences from epidemiological, clinical, and preclinical studies. Mol Biol Rep 2023, 50, 2823–2834. [Google Scholar] [CrossRef] [PubMed]
- Luckey, T.D. Physiological benefits from low levels of ionizing radiation. Health Phys 1982, 43, 771–789. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Li, W.; Jiang, H.; Liang, X.; Zhao, Y.; Yu, D.; Zhou, L.; Wang, G.; Tian, H.; Han, F.; et al. Low-dose radiation may be a novel approach to enhance the effectiveness of cancer therapeutics. Int J Cancer 2016, 139, 2157–2168. [Google Scholar] [CrossRef] [PubMed]
- Au, N.P.B.; Wu, T.; Kumar, G.; Jin, Y.; Li, Y.Y.T.; Chan, S.L.; Lai, J.H.C.; Chan, K.W.Y.; Yu, K.N.; Wang, X.; et al. Low-dose ionizing radiation promotes motor recovery and brain rewiring by resolving inflammatory response after brain injury and stroke. Brain Behav Immun 2024, 115, 43–63. [Google Scholar] [CrossRef]
- Macklis, R.M. Radithor and the era of mild radium therapy. JAMA 1990, 264, 614–618. [Google Scholar] [CrossRef]
- Wolff, S. Is Radiation All Bad? The Search for Adaptation. Radiation research 1992, 131. [Google Scholar] [CrossRef]
- Desouky, O.; Ding, N.; Zhou, G. Targeted and non-targeted effects of ionizing radiation. Journal of Radiation Research and Applied Sciences 2015, 8, 247–254. [Google Scholar] [CrossRef]
- Tang, F.R.; Loke, W.K.; Khoo, B.C. Low-dose or low-dose-rate ionizing radiation–induced bioeffects in animal models. Journal of radiation research 2017, 58, 165–182. [Google Scholar] [CrossRef]
- Tubiana, M.; Arengo, A.; Averbeck, D.; Masse, R. Low-Dose Risk Assessment. Radiation research 2007, 167, 742–744. [Google Scholar] [CrossRef]
- Kokhan, V.S.; Dobynde, M.I. The Effects of Galactic Cosmic Rays on the Central Nervous System: From Negative to Unexpectedly Positive Effects That Astronauts May Encounter. Biology (Basel) 2023, 12. [Google Scholar] [CrossRef]
- Liu, B.; Hinshaw, R.G.; Le, K.X.; Park, M.A.; Wang, S.; Belanger, A.P.; Dubey, S.; Frost, J.L.; Shi, Q.; Holton, P.; et al. Space-like (56)Fe irradiation manifests mild, early sex-specific behavioral and neuropathological changes in wildtype and Alzheimer's-like transgenic mice. Scientific reports 2019, 9, 12118. [Google Scholar] [CrossRef] [PubMed]
- Arunkumar, N.; Gokul, M.; Narayanam, H.; Ananya, A.K. Low-dose radiation research insights in experimental animals: A gateway to therapeutic implications. Vet World 2024, 17, 2253–2258. [Google Scholar] [CrossRef] [PubMed]
- Coelho, C.M.; Pereira, L.; Teubig, P.; Santos, P.; Mendes, F.; Vinals, S.; Galaviz, D.; Herrera, F. Radiation as a Tool against Neurodegeneration-A Potential Treatment for Amyloidosis in the Central Nervous System. Int J Mol Sci 2022, 23. [Google Scholar] [CrossRef] [PubMed]
- Wan, Y.; Liu, J.; Mai, Y.; Hong, Y.; Jia, Z.; Tian, G.; Liu, Y.; Liang, H.; Liu, J. Current advances and future trends of hormesis in disease. NPJ Aging 2024, 10, 26. [Google Scholar] [CrossRef]
- Le, D.D.; Jang, Y.S.; Truong, V.; Yu, S.; Dinh, T.; Lee, M. Bioactivities of Quinic Acids from Vitex rotundifolia Obtained by Supercritical Fluid Extraction. Antioxidants (Basel) 2024, 13. [Google Scholar] [CrossRef]
- Hosztafi, S.; Galambos, A.R.; Koteles, I.; Karadi, D.A.; Furst, S.; Al-Khrasani, M. Opioid-Based Haptens: Development of Immunotherapy. Int J Mol Sci 2024, 25. [Google Scholar] [CrossRef]
- Haiying Wang, W.Z.; Cao, Yi. Radiation-induced cellular senescence and adaptive response: mechanistic interplay and implications 2025, Volume 6.
- Tapio, S.; Jacob, V. Radioadaptive response revisited. Radiat Environ Biophys 2007, 46, 1–12. [Google Scholar] [CrossRef]
- Villavicencio Tejo, F.; Quintanilla, R.A. Contribution of the Nrf2 Pathway on Oxidative Damage and Mitochondrial Failure in Parkinson and Alzheimer's Disease. Antioxidants (Basel) 2021, 10. [Google Scholar] [CrossRef]
- Kaur, K.; Narang, R.K.; Singh, S. Role of Nrf2 in Oxidative Stress, Neuroinflammation and Autophagy in Alzheimer's Disease: Regulation of Nrf2 by Different Signaling Pathways. Curr Mol Med 2025, 25, 372–387. [Google Scholar] [CrossRef] [PubMed]
- Bellezza, I.; Giambanco, I.; Minelli, A.; Donato, R. Nrf2-Keap1 signaling in oxidative and reductive stress. Biochim Biophys Acta Mol Cell Res 2018, 1865, 721–733. [Google Scholar] [CrossRef] [PubMed]
- Qiu, D.; Song, S.; Wang, Y.; Bian, Y.; Wu, M.; Wu, H.; Shi, Y.; Duan, H. NAD(P)H: quinone oxidoreductase 1 attenuates oxidative stress and apoptosis by regulating Sirt1 in diabetic nephropathy. J Transl Med 2022, 20, 44. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Luo, C.; Li, Z.; Huang, W.; Zheng, S.; Liu, C.; Shi, X.; Ma, Y.; Ni, Q.; Tan, W.; et al. Astaxanthin activates the Nrf2/Keap1/HO-1 pathway to inhibit oxidative stress and ferroptosis, reducing triphenyl phosphate (TPhP)-induced neurodevelopmental toxicity. Ecotoxicol Environ Saf 2024, 271, 115960. [Google Scholar] [CrossRef]
- Jiang, X.; Yu, M.; Wang, W.K.; Zhu, L.Y.; Wang, X.; Jin, H.C.; Feng, L.F. The regulation and function of Nrf2 signaling in ferroptosis-activated cancer therapy. Acta Pharmacol Sin 2024, 45, 2229–2240. [Google Scholar] [CrossRef]
- Shan, C.; Wang, Y.; Wang, Y. The Crosstalk between Autophagy and Nrf2 Signaling in Cancer: from Biology to Clinical Applications. Int J Biol Sci 2024, 20, 6181–6206. [Google Scholar] [CrossRef]
- Eldridge, A.; Fan, M.; Woloschak, G.; Grdina, D.J.; Chromy, B.A.; Li, J.J. Manganese superoxide dismutase interacts with a large scale of cellular and mitochondrial proteins in low-dose radiation-induced adaptive radioprotection. Free Radic Biol Med 2012, 53, 1838–1847. [Google Scholar] [CrossRef]
- Yamaoka, K.; Edamatsu, R.; Itoh, T.; Mori, A. Effects of low-dose X-ray irradiation on biomembrane in brain cortex of aged rats. Free Radic Biol Med 1994, 16, 529–534. [Google Scholar] [CrossRef]
- Lu, W. Sulforaphane regulates AngII-induced podocyte oxidative stress injury through the Nrf2-Keap1/ho-1/ROS pathway. Ren Fail 2024, 46, 2416937. [Google Scholar] [CrossRef]
- Khan, N.M.; Sandur, S.K.; Checker, R.; Sharma, D.; Poduval, T.B.; Sainis, K.B. Pro-oxidants ameliorate radiation-induced apoptosis through activation of the calcium-ERK1/2-Nrf2 pathway. Free Radic Biol Med 2011, 51, 115–128. [Google Scholar] [CrossRef]
- Mathew, S.T.; Bergstrom, P.; Hammarsten, O. Repeated Nrf2 stimulation using sulforaphane protects fibroblasts from ionizing radiation. Toxicol Appl Pharmacol 2014, 276, 188–194. [Google Scholar] [CrossRef] [PubMed]
- Chen, N.; Wu, L.; Yuan, H.; Wang, J. ROS/Autophagy/Nrf2 Pathway Mediated Low-Dose Radiation Induced Radio-Resistance in Human Lung Adenocarcinoma A549 Cell. Int J Biol Sci 2015, 11, 833–844. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Mao, A.; Yan, J.; Sun, C.; Di, C.; Zhou, X.; Li, H.; Guo, R.; Zhang, H. Downregulation of Nrf2 promotes radiation-induced apoptosis through Nrf2 mediated Notch signaling in non-small cell lung cancer cells. Int J Oncol 2016, 48, 765–773. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Liang, M.; Jiang, J.; He, R.; Wang, M.; Guo, X.; Shen, M.; Qin, R. Combined inhibition of autophagy and Nrf2 signaling augments bortezomib-induced apoptosis by increasing ROS production and ER stress in pancreatic cancer cells. Int J Biol Sci 2018, 14, 1291–1305. [Google Scholar] [CrossRef]
- Wu, H.; Wu, J.; Jiang, J.; Qian, Z.; Yang, S.; Sun, Y.; Cui, H.; Li, S.; Zhang, P.; Zhou, Z. Compound 7 regulates microglia polarization and attenuates radiation-induced myelopathy via the Nrf2 signaling pathway in vivo and in vitro studies. Mol Med 2024, 30, 198. [Google Scholar] [CrossRef]
- Kim, S.; Chung, H.; Ngoc Mai, H.; Nam, Y.; Shin, S.J.; Park, Y.H.; Chung, M.J.; Lee, J.K.; Rhee, H.Y.; Jahng, G.H.; et al. Low-Dose Ionizing Radiation Modulates Microglia Phenotypes in the Models of Alzheimer's Disease. International journal of molecular sciences 2020, 21. [Google Scholar] [CrossRef]
- Seong, K.M.; Kim, C.S.; Lee, B.S.; Nam, S.Y.; Yang, K.H.; Kim, J.Y.; Park, J.J.; Min, K.J.; Jin, Y.W. Low-dose radiation induces Drosophila innate immunity through Toll pathway activation. J Radiat Res 2012, 53, 242–249. [Google Scholar] [CrossRef]
- Feinendegen, L.E.; Pollycove, M.; Neumann, R.D. Feinendegen. Low-dose cancer risk modeling must recognize up-regulation of protection. Dose Response 2009, 8, 227–252. [Google Scholar] [CrossRef]
- Schaue, D.; Kachikwu, E.L.; McBride, W.H. Cytokines in radiobiological responses: a review. Radiat Res 2012, 178, 505–523. [Google Scholar] [CrossRef]
- Kiang, J.G.; Smith, J.T.; Hegge, S.R.; Ossetrova, N.I. Circulating Cytokine/Chemokine Concentrations Respond to Ionizing Radiation Doses but not Radiation Dose Rates: Granulocyte-Colony Stimulating Factor and Interleukin-18. Radiat Res 2018, 189, 634–643. [Google Scholar] [CrossRef]
- Chen, H.; Liao, S.B.; Cheung, M.P.; Chow, P.H.; Cheung, A.L.; W.S., O. Effects of sperm DNA damage on the levels of RAD51 and p53 proteins in zygotes and 2-cell embryos sired by golden hamsters without the major accessory sex glands. Free Radic Biol Med 2012, 53, 885–892. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.K.; Wang, J.Y.; Chen, C.F.; Chao, K.Y.; Chang, M.C.; Chen, W.M.; Hung, S.C. Early Passage Mesenchymal Stem Cells Display Decreased Radiosensitivity and Increased DNA Repair Activity. Stem Cells Transl Med 2017, 6, 1504–1514. [Google Scholar] [CrossRef] [PubMed]
- Kaushik, N.; Kim, M.J.; Kim, R.K.; Kumar Kaushik, N.; Seong, K.M.; Nam, S.Y.; Lee, S.J. Low-dose radiation decreases tumor progression via the inhibition of the JAK1/STAT3 signaling axis in breast cancer cell lines. Sci Rep 2017, 7, 43361. [Google Scholar] [CrossRef] [PubMed]
- Averbeck, D. Low-Dose Non-Targeted Effects and Mitochondrial Control. Int J Mol Sci 2023, 24. [Google Scholar] [CrossRef]
- Tang, F.R.; Loke, W.K. Molecular mechanisms of low dose ionizing radiation-induced hormesis, adaptive responses, radioresistance, bystander effects, and genomic instability. Int J Radiat Biol 2015, 91, 13–27. [Google Scholar] [CrossRef]
- Liang, X.; Gu, J.; Yu, D.; Wang, G.; Zhou, L.; Zhang, X.; Zhao, Y.; Chen, X.; Zheng, S.; Liu, Q.; et al. Low-Dose Radiation Induces Cell Proliferation in Human Embryonic Lung Fibroblasts but not in Lung Cancer Cells: Importance of ERK1/2 and AKT Signaling Pathways. Dose Response 2016, 14, 1559325815622174. [Google Scholar] [CrossRef]
- Khan, M.G.M.; Wang, Y. Advances in the Current Understanding of How Low-Dose Radiation Affects the Cell Cycle. Cells 2022, 11. [Google Scholar] [CrossRef]
- Zhang, J.; Dai, K.; An, R.; Wang, C.; Zhou, X.; Tian, Z.; Liao, Z. Single Low-Dose Ionizing Radiation Transiently Enhances Rat RIN-m5F Cell Function via the ROS/p38 MAPK Pathway Without Inducing Cell Damage. Antioxidants (Basel) 2025, 14. [Google Scholar] [CrossRef]
- Wei, L.C.; Ding, Y.X.; Liu, Y.H.; Duan, L.; Bai, Y.; Shi, M.; Chen, L.W. Low-dose radiation stimulates Wnt/beta-catenin signaling, neural stem cell proliferation and neurogenesis of the mouse hippocampus in vitro and in vivo. Curr Alzheimer Res 2012, 9, 278–289. [Google Scholar] [CrossRef]
- Betlazar, C.; Middleton, R.J.; Banati, R.B.; Liu, G.J. The impact of high and low dose ionising radiation on the central nervous system. Redox Biol 2016, 9, 144–156. [Google Scholar] [CrossRef]
- Sampadi, B.; Vermeulen, S.; Misovic, B.; Boei, J.J.; Batth, T.S.; Chang, J.G.; Paulsen, M.T.; Magnuson, B.; Schimmel, J.; Kool, H.; et al. Divergent Molecular and Cellular Responses to Low and High-Dose Ionizing Radiation. Cells 2022, 11. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Yang, J.; Qi, Z.; Wu, H.; Wang, B.; Zou, F.; Mei, H.; Liu, J.; Wang, W.; Liu, Q. Heat shock proteins: Biological functions, pathological roles, and therapeutic opportunities. MedComm (2020) 2022, 3, e161. [Google Scholar] [CrossRef] [PubMed]
- Muschter, D.; Geyer, F.; Bauer, R.; Ettl, T.; Schreml, S.; Haubner, F. A comparison of cell survival and heat shock protein expression after radiation in normal dermal fibroblasts, microvascular endothelial cells, and different head and neck squamous carcinoma cell lines. Clin Oral Investig 2018, 22, 2251–2262. [Google Scholar] [CrossRef] [PubMed]
- Matchuk, O.N.; Zamulaeva, I.A. High Level of Radiation-Induced Heat Shock Protein with a Molecular Weight of 27 and 70 kDa is the Hallmark of Radioresistant SP Cells of MCF-7 Breast Cancer Culture. Radiats Biol Radioecol 2016, 56, 382–388. [Google Scholar]
- Lagisz, M.; Hector, K.L.; Nakagawa, S. Life extension after heat shock exposure: assessing meta-analytic evidence for hormesis. Ageing Res Rev 2013, 12, 653–660. [Google Scholar] [CrossRef]
- Mladenov, E.; Fan, X.; Dueva, R.; Soni, A.; Iliakis, G. Radiation-dose-dependent functional synergisms between ATM, ATR and DNA-PKcs in checkpoint control and resection in G(2)-phase. Sci Rep 2019, 9, 8255. [Google Scholar] [CrossRef]
- Laconi, E.; Cheri, S.; Fanti, M.; Marongiu, F. Aging and Cancer: The Waning of Community Bonds. Cells 2021, 10. [Google Scholar] [CrossRef]
- Li, W.H.; Wang, F.; Song, G.Y.; Yu, Q.H.; Du, R.P.; Xu, P. PARP-1: a critical regulator in radioprotection and radiotherapy-mechanisms, challenges, and therapeutic opportunities. Front Pharmacol 2023, 14, 1198948. [Google Scholar] [CrossRef]
- Khodarev, N.N.; Beckett, M.; Labay, E.; Darga, T.; Roizman, B.; Weichselbaum, R.R. STAT1 is overexpressed in tumors selected for radioresistance and confers protection from radiation in transduced sensitive cells. Proc Natl Acad Sci U S A 2004, 101, 1714–1719. [Google Scholar] [CrossRef]
- Machi, A., Jr.; Moreira Perez, M.; Luciano da Veiga, G.; Cristiano Pereira, E.; Adami, F.; Alves, B.; Fonseca, F. Expression of DNA repair genes in association with ionizing radiation. Acta Biomed 2022, 93. [Google Scholar] [CrossRef]
- Yang, H.M. Mitochondrial Dysfunction in Neurodegenerative Diseases. Cells 2025, 14. [Google Scholar] [CrossRef] [PubMed]
- Kostyuk, S.V.; Proskurnina, E.V.; Konkova, M.S.; Abramova, M.S.; Kalianov, A.A.; Ershova, E.S.; Izhevskaya, V.L.; Kutsev, S.I.; Veiko, N.N. Effect of Low-Dose Ionizing Radiation on the Expression of Mitochondria-Related Genes in Human Mesenchymal Stem Cells. Int J Mol Sci 2021, 23. [Google Scholar] [CrossRef] [PubMed]
- Ristow, M.; Schmeisser, K. Ristow. Mitohormesis: Promoting Health and Lifespan by Increased Levels of Reactive Oxygen Species (ROS). Dose Response 2014, 12, 288–341. [Google Scholar] [CrossRef] [PubMed]
- Yuyun, X.; Jinjun, Q.; Minfang, X.; Jing, Q.; Juan, X.; Rui, M.; Li, Z.; Jing, G. Gao. Effects of Low Concentrations of Rotenone upon Mitohormesis in SH-SY5Y Cells. Dose Response 2013, 11, 270–280. [Google Scholar] [CrossRef]
- Sosin, D.V.; Baranovskii, D.S.; Nechaev, D.N.; Sosina, M.A.; Shaposhnikov, A.V.; Trusov, G.A.; Titova, A.G.; Krasnikov, B.F.; Lomov, A.N.; Makarov, V.V.; et al. Population Studies and Molecular Mechanisms of Human Radioadaptive Capabilities: Is It Time to Rethink Radiation Safety Standards? Int J Mol Sci 2024, 25. [Google Scholar] [CrossRef]
- Chen, Y.; Jiang, Y.; Yang, Y.; Huang, X.; Sun, C. SIRT1 Protects Dopaminergic Neurons in Parkinson's Disease Models via PGC-1alpha-Mediated Mitochondrial Biogenesis. Neurotox Res 2021, 39, 1393–1404. [Google Scholar] [CrossRef]
- Zhou, Y.; Wang, S.; Li, Y.; Yu, S.; Zhao, Y. SIRT1/PGC-1alpha Signaling Promotes Mitochondrial Functional Recovery and Reduces Apoptosis after Intracerebral Hemorrhage in Rats. Front Mol Neurosci 2017, 10, 443. [Google Scholar] [CrossRef]
- Yu, L.; Yang, X.; Li, X.; Qin, L.; Xu, W.; Cui, H.; Jia, Z.; He, Q.; Wang, Z. Pink1/PARK2/mROS-Dependent Mitophagy Initiates the Sensitization of Cancer Cells to Radiation. Oxid Med Cell Longev 2021, 2021, 5595652. [Google Scholar] [CrossRef]
- Kurtman, C; Üçöz, Ö.M.; Karakoyun Çelik, M; Sokur, Ö; Kemal Özbilgin, I.M. Mitophagy in the A549 lung cancer cell line, radiation-induced damage, and the effect of ATM and PARKIN on the mitochondria. Int J Radiat Res 2022, 20, 9–13. [Google Scholar] [CrossRef]
- Bajinskis, A.; Lindegren, H.; Johansson, L.; Harms-Ringdahl, M.; Forsby, A. Low-dose/dose-rate gamma radiation depresses neural differentiation and alters protein expression profiles in neuroblastoma SH-SY5Y cells and C17.2 neural stem cells. Radiat Res 2011, 175, 185–192. [Google Scholar] [CrossRef]
- Kipnis, J.; Avidan, H.; Markovich, Y.; Mizrahi, T.; Hauben, E.; Prigozhina, T.B.; Slavin, S.; Schwartz, M. Low-dose gamma-irradiation promotes survival of injured neurons in the central nervous system via homeostasis-driven proliferation of T cells. Eur J Neurosci 2004, 19, 1191–1198. [Google Scholar] [CrossRef]
- Iacono, D.; Hatch, K.; Murphy, E.K.; Post, J.; Cole, R.N.; Perl, D.P.; Day, R.M. Proteomic changes in the hippocampus of large mammals after total-body low dose radiation. PLoS One 2024, 19, e0296903. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Singla, N.; Chadha, V.D.; Dhawan, D.K. A concept of radiation hormesis: stimulation of antioxidant machinery in rats by low dose ionizing radiation. Hell J Nucl Med 2019, 22, 43–48. [Google Scholar] [PubMed]
- Kojima, S.; Matsuki, O.; Nomura, T.; Yamaoka, K.; Takahashi, M.; Niki, E. Elevation of antioxidant potency in the brain of mice by low-dose gamma-ray irradiation and its effect on 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-induced brain damage. Free Radic Biol Med 1999, 26, 388–395. [Google Scholar] [CrossRef] [PubMed]
- Tharmalingam, S.; Sreetharan, S.; Kulesza, A.V.; Boreham, D.R.; Tai, T.C. Low-Dose Ionizing Radiation Exposure, Oxidative Stress and Epigenetic Programing of Health and Disease. Radiat Res 2017, 188, 525–538. [Google Scholar] [CrossRef]
- Caratero, A.; Courtade, M.; Bonnet, L.; Planel, H.; Caratero, C. Effect of a continuous gamma irradiation at a very low dose on the life span of mice. Gerontology 1998, 44, 272–276. [Google Scholar] [CrossRef]
- Dimberg, Y.; Vazquez, M.; Soderstrom, S.; Ebendal, T. Effects of X-irradiation on nerve growth factor in the developing mouse brain. Toxicol Lett 1997, 90, 35–43. [Google Scholar] [CrossRef]
- Lin, S.; Zhan, Y.; Wang, R.; Pei, J. Decoding neuroinflammation in Alzheimer's disease: a multi-omics and AI-driven perspective for precision medicine. Front Immunol 2025, 16, 1616899. [Google Scholar] [CrossRef]
- Marples, B.; McGee, M.; Callan, S.; Bowen, S.E.; Thibodeau, B.J.; Michael, D.B.; Wilson, G.D.; Maddens, M.E.; Fontanesi, J.; Martinez, A.A. Cranial irradiation significantly reduces beta amyloid plaques in the brain and improves cognition in a murine model of Alzheimer's Disease (AD). Radiother Oncol 2016, 118, 579–580. [Google Scholar] [CrossRef]
- Ceyzeriat, K.; Tournier, B.B.; Millet, P.; Frisoni, G.B.; Garibotto, V.; Zilli, T. Low-Dose Radiation Therapy: A New Treatment Strategy for Alzheimer's Disease? J Alzheimers Dis 2020, 74, 411–419. [Google Scholar] [CrossRef]
- Kokhan, V.S.; Levashova, A.I.; Nesterov, M.S.; Pikalov, V.A.; Chicheva, M.M. Combined Ionizing Radiation Caused Cognition and Non-Cognition Behavior Benefits and Modulated Microglial Activity in Wild-Type and Alzheimer's-like Transgenic Mice. Biology (Basel) 2025, 14. [Google Scholar] [CrossRef]
- Ricciardi, N.R.; Modarresi, F.; Lohse, I.; Andrade, N.S.; Newman, I.R.; Brown, J.M.; Borja, C.; Marples, B.; Wahlestedt, C.R.; Volmar, C.H. Investigating the Synergistic Potential of Low-Dose HDAC3 Inhibition and Radiotherapy in Alzheimer's Disease Models. Mol Neurobiol 2023, 60, 4811–4827. [Google Scholar] [CrossRef] [PubMed]
- Wilson, G.D.; Wilson, T.G.; Hanna, A.; Fontanesi, G.; Kulchycki, J.; Buelow, K.; Pruetz, B.L.; Michael, D.B.; Chinnaiyan, P.; Maddens, M.E.; et al. Low Dose Brain Irradiation Reduces Amyloid-beta and Tau in 3xTg-AD Mice. J Alzheimers Dis 2020, 75, 15–21. [Google Scholar] [CrossRef]
- Kokhan, V.S.; Ageldinov, R.A.; Anokhin, P.K.; Shamakina, I.Y. Combined Ionizing Radiation Exposure Improves Behavioral Symptoms and Modulates Brain Innate Immune System Activity in the Tau P301S Mice Line. Biochemistry (Mosc) 2025, 90, 400–412. [Google Scholar] [CrossRef] [PubMed]
- Jiang, B.; Zhang, Y.; Zhao, J.; She, C.; Zhou, X.; Dong, Q.; Wang, P. Effects of Localized X-Ray Irradiation on Peripheral Nerve Regeneration in Transected Sciatic Nerve in Rats. Radiat Res 2017, 188, 455–462. [Google Scholar] [CrossRef] [PubMed]
- Cuttler, J.M.; Moore, E.R.; Hosfeld, V.D.; Nadolski, D.L. Treatment of Alzheimer Disease With CT Scans: A Case Report. Dose Response 2016, 14, 1559325816640073. [Google Scholar] [CrossRef]
- Cuttler, J.M.; Moore, E.R.; Hosfeld, V.D.; Nadolski, D.L. Update on a Patient With Alzheimer Disease Treated With CT Scans. Dose Response 2017, 15, 1559325817693167. [Google Scholar] [CrossRef]
- Park, M.; Ha, J.; Lee, Y.; Choi, H.S.; Kim, B.S.; Jeong, Y.K. Low-moderate dose whole-brain gamma-ray irradiation modulates the expressions of glial fibrillary acidic protein and intercellular adhesion molecule-1 in the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced Parkinson's disease mouse model. Neurobiol Aging 2023, 132, 175–184. [Google Scholar] [CrossRef]
- Iacono, D.; Murphy, E.K.; Stimpson, C.D.; Perl, D.P.; Day, R.M. Low-dose radiation decreases Lrrk2 levels in the striatum of large mammalian brains: New venues to treat Parkinson's disease? Parkinsonism Relat Disord 2024, 124, 107024. [Google Scholar] [CrossRef]
- Murphy, E.K.; Perl, D.P.; Day, R.M.; Iacono, D. Evaluating Parkinson's disease biomarkers in substantia nigra following sublethal gamma-radiation exposure in a large animal model. NPJ Parkinsons Dis 2025, 11, 286. [Google Scholar] [CrossRef]
- El-Ghazaly, M.A.; Sadik, N.A.; Rashed, E.R.; Abd-El-Fattah, A.A. Neuroprotective effect of EGb761(R) and low-dose whole-body gamma-irradiation in a rat model of Parkinson's disease. Toxicol Ind Health 2015, 31, 1128–1143. [Google Scholar] [CrossRef] [PubMed]
- Feldman, E.L.; Goutman, S.A.; Petri, S.; Mazzini, L.; Savelieff, M.G.; Shaw, P.J.; Sobue, G. Amyotrophic lateral sclerosis. Lancet 2022, 400, 1363–1380. [Google Scholar] [CrossRef] [PubMed]
- Iacono, D.; Murphy, E.K.; Perl, D.P.; Day, R.M. gamma-Radiation induces region-specific subcellular alterations of amyotrophic lateral sclerosis and frontotemporal dementia markers in swine brain. Sci Rep 2026, 16, 5627. [Google Scholar] [CrossRef] [PubMed]
- Harriman, M.; Morrison, M.; Hay, J.; Revonta, M.; Eisen, A.; Lentle, B. Use of radiotherapy for control of sialorrhea in patients with amyotrophic lateral sclerosis. J Otolaryngol 2001, 30, 242–245. [Google Scholar] [CrossRef]
- Otani, A.; Kojima, H.; Guo, C.; Oishi, A.; Yoshimura, N. Low-dose-rate, low-dose irradiation delays neurodegeneration in a model of retinitis pigmentosa. Am J Pathol 2012, 180, 328–336. [Google Scholar] [CrossRef]
- Gocmen, S.; Sirin, S.; Oysul, K.; Ulas, U.H.; Oztas, E. The effects of low-dose radiation in the treatment of sciatic nerve injury in rats. Turk Neurosurg 2012, 22, 167–173. [Google Scholar] [CrossRef]
- Avgerinos, K.I.; Manolopoulos, A.; Ferrucci, L.; Kapogiannis, D. Critical assessment of anti-amyloid-beta monoclonal antibodies effects in Alzheimer's disease: a systematic review and meta-analysis highlighting target engagement and clinical meaningfulness. Scientific reports 2024, 14, 25741. [Google Scholar] [CrossRef]
- Honig, L.S.; Sabbagh, M.N.; van Dyck, C.H.; Sperling, R.A.; Hersch, S.; Matta, A.; Giorgi, L.; Gee, M.; Kanekiyo, M.; Li, D.; et al. Updated safety results from phase 3 lecanemab study in early Alzheimer's disease. Alzheimers Res Ther 2024, 16, 105. [Google Scholar] [CrossRef]
- Li, T.; Lu, L.; Pember, E.; Li, X.; Zhang, B.; Zhu, Z. New Insights into Neuroinflammation Involved in Pathogenic Mechanism of Alzheimer's Disease and Its Potential for Therapeutic Intervention. Cells 2022, 11. [Google Scholar] [CrossRef]
- Kim, BH; Park, K.K.; Kim, WY; Hong, A; Kim, EH; Cho, JY; Rhee, SJ; Chung, HY. WK. Low-Dose Radiation Therapy in Alzheimer’s disease: An Interim Neurocognitive Analysis from a Multicenter Phase II Clinical Trial. Alzheimers Dement. 2025, 20, e084076. [Google Scholar] [CrossRef]
- Teipel, S.J.; Tang, Y.; Khachaturian, A. Sex differences in treatment effects of lecanemab and donanemab: A Bayesian reanalysis of CLARITY-AD and TRAILBLAZER-ALZ2. Alzheimers Dement (N Y) 2025, 11, e70155. [Google Scholar] [CrossRef]
- Andrews, D.; Ducharme, S.; Chertkow, H.; Sormani, M.P.; Collins, D.L.; Alzheimer's Disease Neuroimaging, I. The higher benefit of lecanemab in males compared to females in CLARITY AD is probably due to a real sex effect. Alzheimers Dement 2025, 21, e14467. [Google Scholar] [CrossRef]
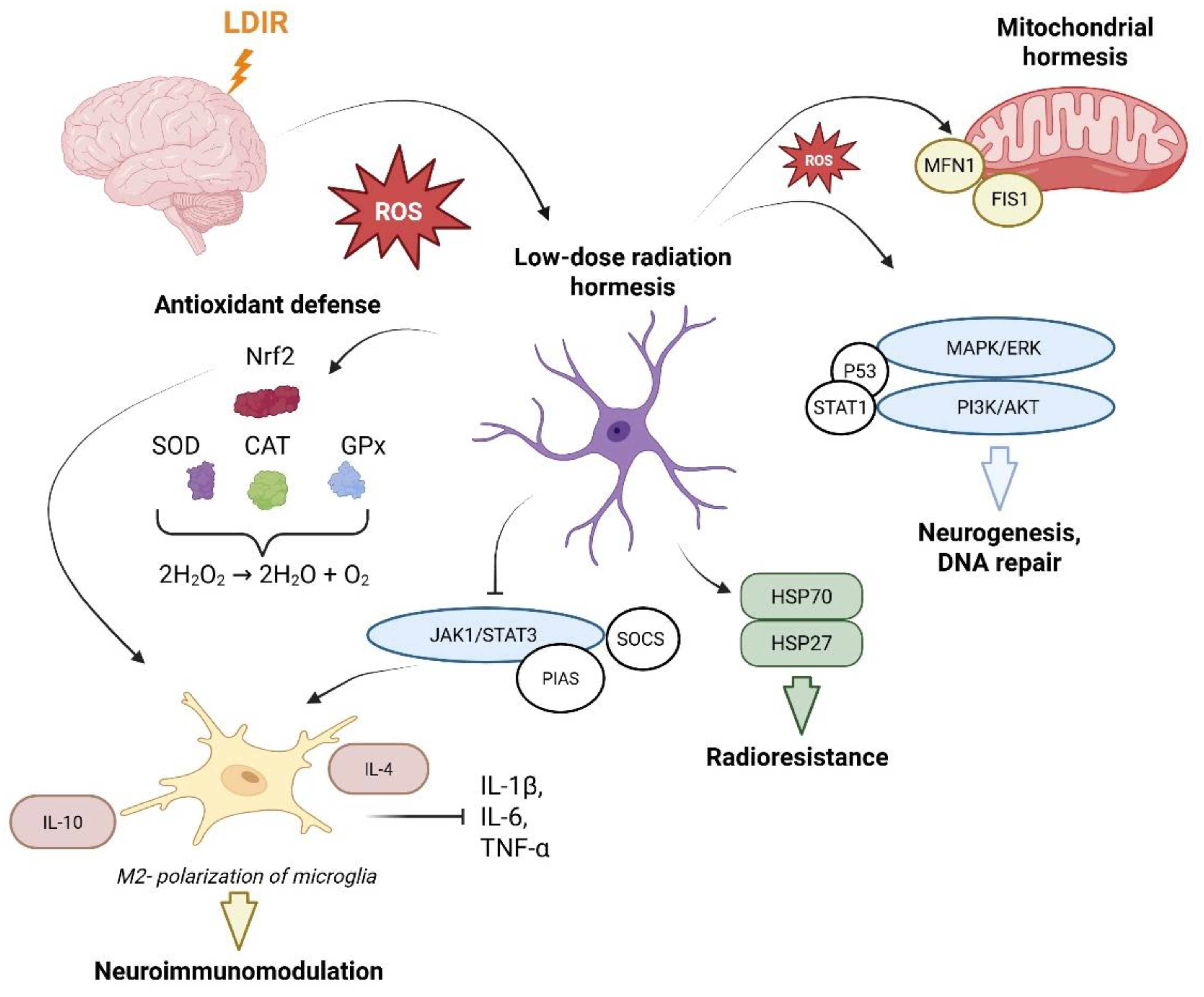
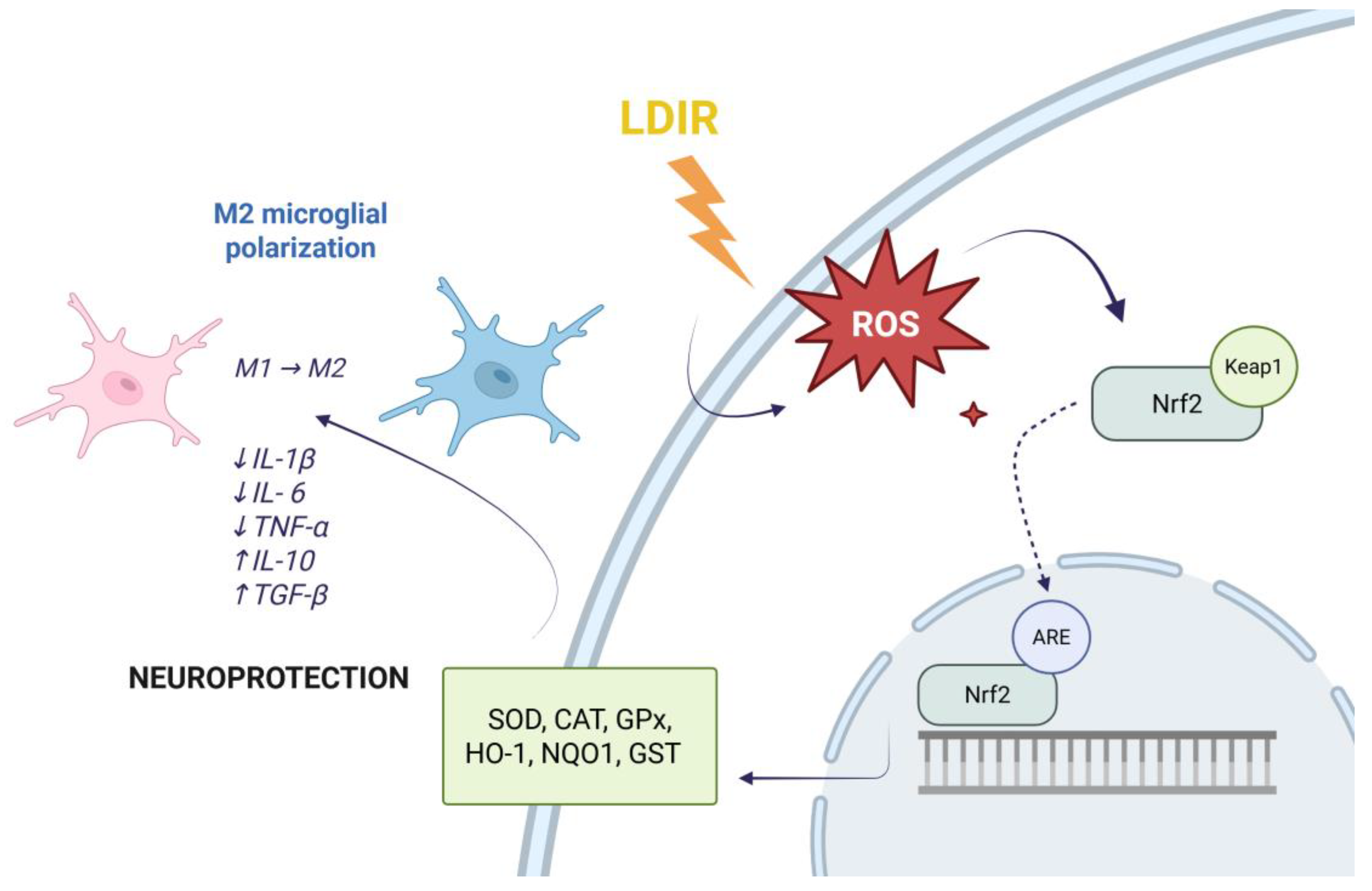
| Protective and Adaptive Mechanisms | Key Markers and Signaling Pathways | Biological Effects | References | |||
|---|---|---|---|---|---|---|
| Antioxidant Defense | Nrf2/ARE pathway, SOD, MnSOD, CAT, GPx, HO-1, NQO1, GST |
ROS neutralization, reduction of oxidative stress, neuronal protection against apoptosis |
[27,28,30,32,33] | |||
| Neuroimmunomodulation | Microglial polarization (M1 → M2), ↓IL-1β, ↓IL-6, ↓TNF-α, ↑IL-10, ↑TGF-β, ↑T-cell response |
Mitigation of chronic neuroinflammation, establishment of a regenerative CNS microenvironment | [35,36,39,40,41,42,71] | |||
| Proliferation and Neuroplasticity | MAPK/ERK pathway, PI3K/Akt, p53, p38/MAPK/PDX-1 pathway | Stimulation of hippocampal neurogenesis, improvement of cognitive functions and synaptic transmission | [45,46,47,48,49,50,51] | |||
| Proteostasis and DNA Repair | HSP70, HSP27, ATM kinase, PARP1, STAT1, ATM/ERK/NF-κB, ↑hMSH2 and ↑hMSH6, NPY, PCP4 | Correct protein folding, prevention of protein aggregation and proteopathy, restoration of genomic integrity | [53,54,56,59,60,72] | |||
| Mitohormesis | SIRT1/PGC-1α pathway, FIS1/MFN1, Pink1/PARK2 pathway* | Restoration of neuronal bioenergetics, clearance of damaged mitochondria (mitophagy) | [62,66,67,68,69] |
| Pathology | Subject (model) | Irradiation regimen | Key biological effects | Refs. |
|---|---|---|---|---|
| Alzheimer’s disease | Mice (5XFAD, 3xTg-AD, APPswePSEN1) | γ-rays 0,5–2 Gy (fractionated); γ-rays 0,24 Gy + 12C nuclei 0,18 Gy (combined) | Reduction of amyloid burden (↓Aβ); M1→M2 microglia polarization (TREM2); increased neuronal viability; ↑VEGF, ↑GAP-43; improvement of cognitive functions | [81,82,84,85] |
| Parkinson’s disease | Mice (MPTP); rats (reserpine); minipigs |
γ-rays 0.5–0.6 Gy (single dose); γ-rays 0.25 Gy x 6 weeks (cumulative dose 1,5 Gy) | Activation of CAT and GSH; ↓LRRK2 in the striatum; suppression of neuroinflammation (↓GFAP, ↓CD54); synergy with Ginkgo biloba – restoration of dopamine, GSH, MDA levels, and iron ions in the striatum | [74,88,89,91] |
| Amyotrophic lateral sclerosis | Minipigs | γ-rays 1,79 Gy (single dose) | Modulation of expression and distribution of FUS/TLS, C9orf72, STMN2, and pTDP43 proteins | [93] |
| Retinitis pigmentosa | rd10 mice | γ-rays 0,65 Gy (repeated) | Delayed photoreceptor cell death; activation of the antioxidant enzyme Prdx2 | [95] |
| Sciatic nerve injury | Female Wistar rats | γ-rays 7 Gy (single dose) | Stimulation of regeneration; suppression of scarring | [96] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




