Submitted:
24 February 2026
Posted:
02 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
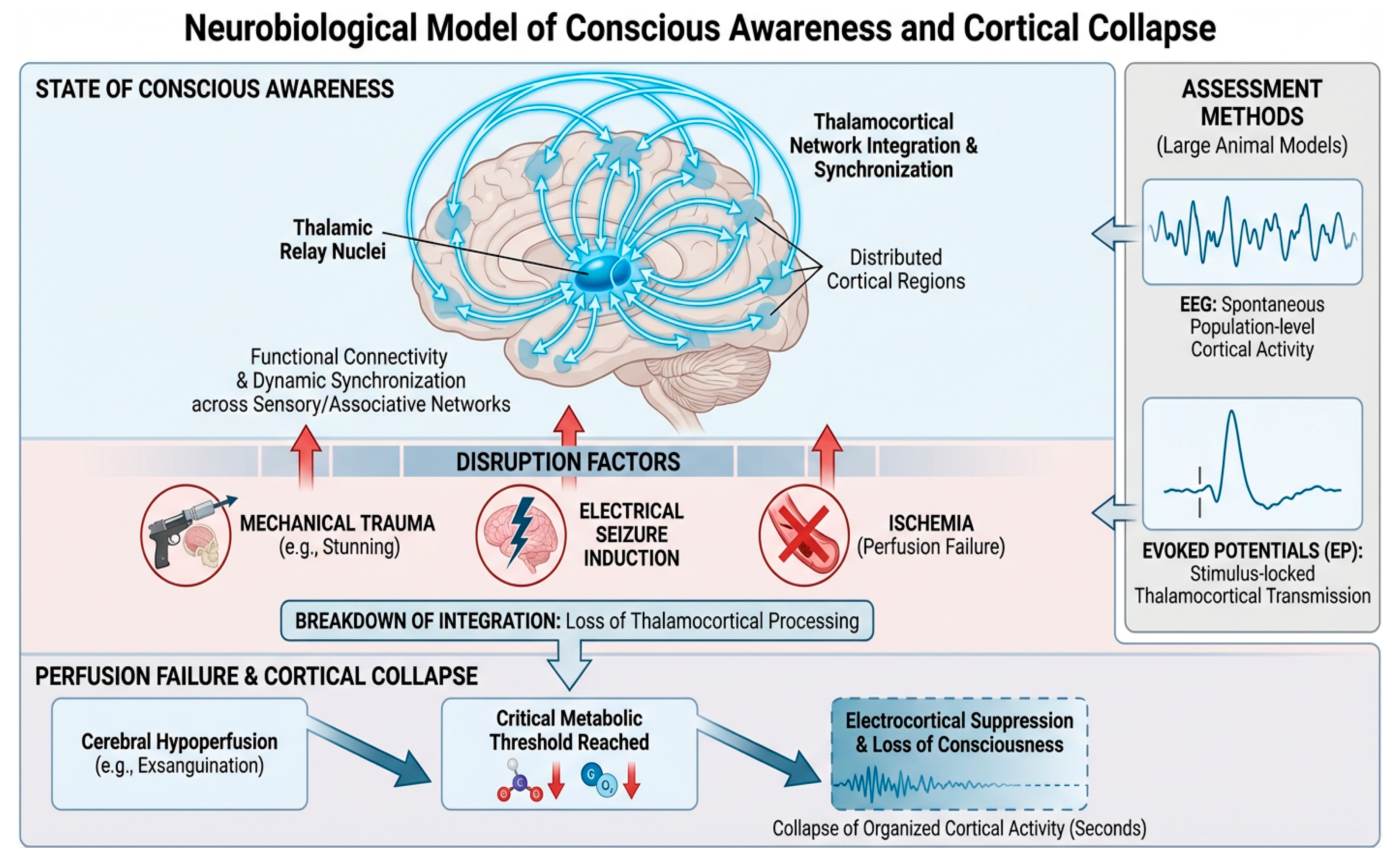
3. Neurobiological Foundations
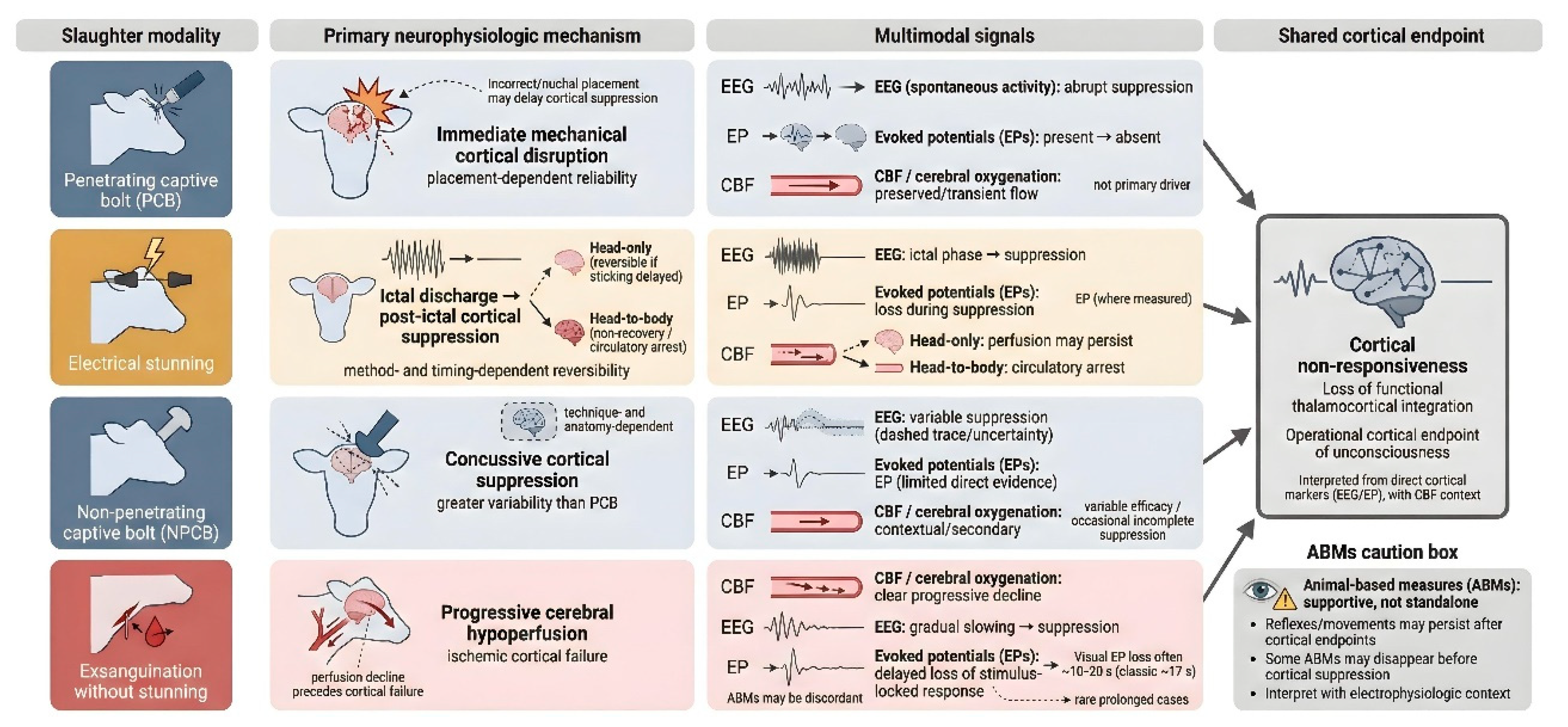
4. Electrophysiologic Evidence Across Slaughter Modalities
4.1. Penetrating Captive Bolt (PCB)
4.2. Electrical Stunning
4.3. Non-Penetrating Captive Bolt
4.4. Slaughter Without Stunning
5. Cerebral Blood Flow and Cortical Viability
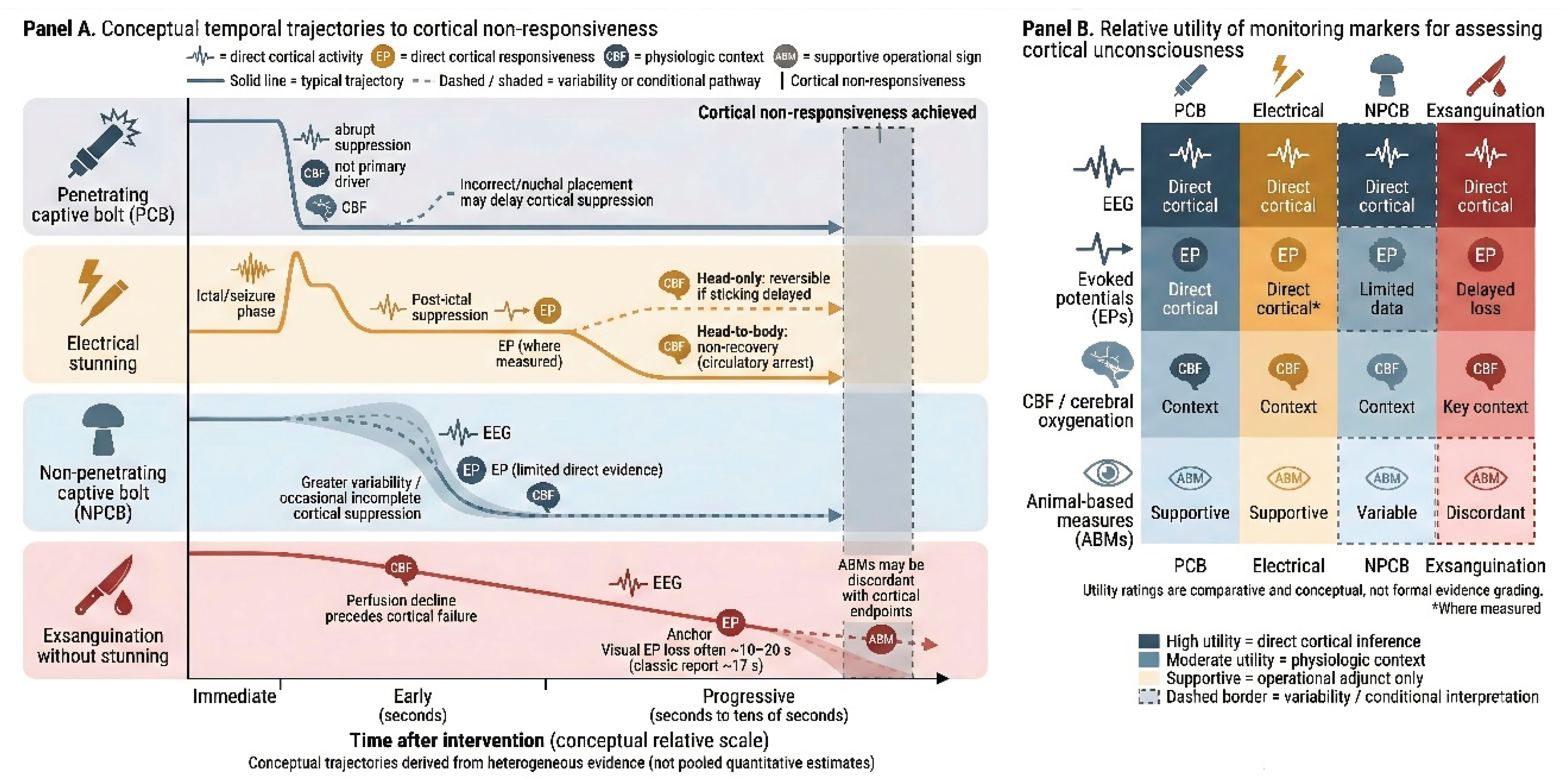
6. Comparative Mechanistic Interpretation
7. Discussion
7.1. Integrative Interpretation
7.2. Future Research Agenda
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ABMs | Animal-based Measures |
| CBF | Cerebral Blood Flow |
| ECoG | Electrocorticography |
| EEG | Electroencephalography |
| EFSA | European Food Safety Authority |
| EP | Evoked Potentials |
| EU | European Union |
| FFT | Fast Fourier Transform |
| HALF | High-amplitude low-frequency |
| LVFA | Low voltage fast activity |
| NIRS | Near-infrared spectroscopy |
| NPCB | Non-penetrating captive bolt |
| PCB | Penetrating captive bolt |
| Ptot | Total power |
| RMS | Root Mean Square |
| SANRA | Scale for the Assessment of Narrative Review Articles |
| SEF50 | Spectral Edge Frequency 50 |
| SEF95 | Spectral Edge Frequency 95 |
| VER | Visual evoked responses |
References
- Sgourdou, P. The Consciousness of Pain: A Thalamocortical Perspective. NeuroSci 2022, 3, 311–320. [Google Scholar] [CrossRef]
- Ong, W.-Y.; Stohler, C.S.; Herr, D.R. Role of the Prefrontal Cortex in Pain Processing. Mol. Neurobiol. 2018, 56, 1137–1166. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Larrea, L.; Bastuji, H. Pain and consciousness. Prog. Neuro-Psychopharmacology Biol. Psychiatry 2018, 87, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Daly, C.; Kallweit, E.; Ellendorf, F. Cortical function in cattle during slaughter: Conventional captive bolt stunning followed by exsanguination compared with shechita slaughter. Veter- Rec. 1988, 122, 325–329. [Google Scholar] [CrossRef] [PubMed]
- Bager, F.; Braggins, T.; Devine, C.; Graafhuis, A.; Mellor, D.; Tavener, A.; Upsdell, M. Onset of insensibility at slaughter in calves: Effects of electroplectic seizure and exsanguination on spontaneous electrocortical activity and indices of cerebral metabolism. Res. Veter- Sci. 1992, 52, 162–173. [Google Scholar] [CrossRef]
- Gregory, N.; Wotton, S. Time to loss of brain responsiveness following exsanguination in calves. Res. Veter- Sci. 1984, 37, 141–143. [Google Scholar] [CrossRef]
- Verhoeven, M.T.W.; Gerritzen, M.A.; Hellebrekers, L.J.; Kemp, B. Validation of indicators used to assess unconsciousness in veal calves at slaughter. Animal 2016, 10, 1457–1465. [Google Scholar] [CrossRef]
- Baethge, C.; Goldbeck-Wood, S.; Mertens, S. SANRA—a scale for the quality assessment of narrative review articles. Res. Integr. Peer Rev. 2019, 26, 5. [Google Scholar] [CrossRef]
- Müller, E.J.; Munn, B.R.; Redinbaugh, M.J.; Lizier, J.; Breakspear, M.; Saalmann, Y.B.; Shine, J.M. The non-specific matrix thalamus facilitates the cortical information processing modes relevant for conscious awareness. Cell Rep. 2023, 42, 112844. [Google Scholar] [CrossRef]
- Whyte, C.J.; Redinbaugh, M.J.; Shine, J.M.; Saalmann, Y.B. Thalamic contributions to the state and contents of consciousness. Neuron 2024, 112, 1611–1625. [Google Scholar] [CrossRef]
- Cacciatore, M.; Magnani, F.G.; Barbadoro, F.; Ippoliti, C.; Stanziano, M.; Clementi, L.; Nigri, A.; Nanetti, L.; Marino, S.; La Porta, F.; et al. Thalamus and consciousness: A systematic review on thalamic nuclei associated with consciousness. Front. Neurol. 2025, 16, 1509668. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Abubakar, A.A.; Sazili, A.Q.; Kaka, U.; Goh, Y.-M. Application of Electroencephalography in Preslaughter Management: A Review. Animals 2022, 12, 2857. [Google Scholar] [CrossRef] [PubMed]
- Zulkifli, I.; Goh, Y.M.; Norbaiyah, B.; Sazili, A.Q.; Lotfi, M.; Soleimani, A.F.; Small, A.H. Changes in blood parameters and electroencephalogram of cattle as affected by different stunning and slaughter methods in cattle. Anim. Prod. Sci. 2013, 54, 187–193. [Google Scholar] [CrossRef]
- Aubert, A.; Costalata, R.; Agnèsaubertab. A Model of the Coupling between Brain Electrical Activity, Metabolism, and Hemodynamics: Application to the Interpretation of Functional Neuroimaging. NeuroImage 2002, 17, 1162–1181. [Google Scholar] [CrossRef]
- Van Lieshout, J.J.; Wieling, W.; Karemaker, J.M.; Secher, N.H. Syncope, cerebral perfusion, and oxygenation. J. Appl. Physiol. 2003, 94, 833–848. [Google Scholar] [CrossRef]
- Lambooy, E.; Spanjaard, W. Effect of the shooting position on the stunning of calves by captive bolt. Veter- Rec. 1981, 109, 359–361. [Google Scholar] [CrossRef]
- Gibson, T.J.; Oliveira, S.E.O.; Costa, F.A.D.; Gregory, N.G. Electroencephalographic assessment of pneumatically powered penetrating and non-penetrating captive-bolt stunning of bulls. Meat Sci. 2019, 151, 54–59. [Google Scholar] [CrossRef]
- Devine, C.; Tavener, A.; Graafhuis, A.; Gilbert, K. Electroencephalographic studies of calves associated with electrical stunning, throat cutting and carcass electro-immobilisation. New Zealand Veter- J. 1987, 35, 107–112. [Google Scholar] [CrossRef]
- Devine, C.; Tavener, A.; Gilbert, K.; Day, A. Electroencephalographic studies of adult cattle associated with electrical stunning, throat cutting and carcass electro-immobilization. New Zealand Veter- J. 1986, 34, 210–213. [Google Scholar] [CrossRef]
- Beausoleil, N.; Farouk, M.; Webster, J.; Johnson, C.; Dowling, S.; Sazili, A.; Cameron, C. Comparison of recovery of sheep, goats, and calves from reversible electrical head-only and head-to-body stunning for halal meat production. New Zealand Veter- J. 2024, 72, 288–299. [Google Scholar] [CrossRef]
- Musk, G.C.; Johnson, C.B. Commentary: A Comparison of the Methods of the Pre-Slaughter Stunning of Cattle in Australia—Mechanical, Electrical, and Diathermic Syncope. Animals 2024, 14, 3141. [Google Scholar] [CrossRef] [PubMed]
- Gibson, T.; Johnson, C.; Murrell, J.; Mitchinson, S.; Stafford, K.; Mellor, D. Electroencephalographic responses to concussive non-penetrative captive-bolt stunning in halothane-anaesthetised calves. New Zealand Veter- J. 2009, 57, 90–95. [Google Scholar] [CrossRef] [PubMed]
- Imlan, J.C.; Kaka, U.; Goh, Y.-M.; Idrus, Z.; Awad, E.A.; Abubakar, A.A.; Ahmad, T.; Nizamuddin, H.N.Q.; Sazili, A.Q. Effects of Slaughter Knife Sharpness on Blood Biochemical and Electroencephalogram Changes in Cattle. Animals 2020, 10, 579. [Google Scholar] [CrossRef] [PubMed]
- Imlan, J.C.; Kaka, U.; Goh, Y.-M.; Idrus, Z.; Awad, E.A.; Abubakar, A.A.; Ahmad, T.; Nizamuddin, H.N.Q.; Sazili, A.Q. Effects of Slaughter Positions on Catecholamine, Blood Biochemical and Electroencephalogram Changes in Cattle Restrained Using a Modified Mark IV Box. Animals 2021, 11, 1979. [Google Scholar] [CrossRef]
- Small, A.; Jenson, I.; Phillips, A.; McLean, D.; Kalinowski, T.; Ralph, J. Cattle recover completely from unconsciousness induced by controlled application of 150–180 kJ of 915 MHz microwave energy to the forehead. Veter- Anim. Sci. 2025, 29, 100466. [Google Scholar] [CrossRef]
- Sherman, D.L.; Thakor, N.V. EEG Signal Processing: Theory and Applications. In Neural Engineering; He, B., Ed.; Springer: Cham, Switzerland, 2020; pp. 97–129. [Google Scholar] [CrossRef]
- Chung, C.K.E.; Poon, C.C.M.; Irwin, M.G. Peri-operative neurological monitoring with electroencephalography and cerebral oximetry: A narrative review. Anaesthesia 2022, 77, 113–122. [Google Scholar] [CrossRef]
- Nischal, S.A.; Patel, S.; Yuen, J.; Mortimer, A.; Patel, N.K. Cerebral blood flow and modern approaches for clinical assessment & monitoring: A view to the future. Br. J. Neurosurg. 2025, 1–15. [Google Scholar] [CrossRef]
- Gallucci, A.; Varoli, E.; Del Mauro, L.; Hassan, G.; Rovida, M.; Comanducci, A.; Casarotto, S.; Re, V.L.; Lauro, L.J.R. Multimodal approaches supporting the diagnosis, prognosis and investigation of neural correlates of disorders of consciousness: A systematic review. Eur. J. Neurosci. 2023, 59, 874–933. [Google Scholar] [CrossRef]
- Meyer, R.E. Physiologic Measures of Animal Stress during Transitional States of Consciousness. Animals 2015, 5, 702–716. [Google Scholar] [CrossRef]
- Hascalovici, J.R.; Schipper, H.M.; Regenstein, J.M.; Rosen, S.D.; Zivotofsky, A.; St-Jean, G.; Freilich, S.; Morrison, T.J.; Rabkin, S.W.; Haut, S. Rapid loss of consciousness in cattle following nonstun slaughter: Evidence from a systematic review. Am. J. Veter- Res. 2025, 86, 1–10. [Google Scholar] [CrossRef]
| Domains | Study (Year) | Animal | Group | EEG/ ECoG | EEG Criterion for Unconsciousness | Key Findings |
|---|---|---|---|---|---|---|
| Penetrating Captive Bolt (PCB) | Lambooy (1981) [16] | Veal calves | Frontal vs occipital vs nape/neck placement | EEG (implanted electrodes) | Delta/theta tending to isoelectric line; consciousness judged from EEG | Frontal & occipital: 'almost certainly immediate' unconsciousness; nape: mean 21±6 s to unconsciousness; calves conscious until then |
| Daly et al (1987) [4] | Cattle | Bolt velocity comparison: 41 vs 47 vs 55 vs 58 m/s | ECoG (spontaneous cortical activity; epidural/dural electrodes) + VER (visual evoked responses) | - Not an EEG-only criterion; - Use loss of VER as primary indicator of insensibility; “severe reduction” of spontaneous ECoG defined as <10% of pre-shot baseline; VER considered “present” if >10% baseline |
- Higher bolt velocity (55–58 m/s) ↓ likelihood of VER presence in the first ~8 s and ↑ time to VER recovery; however VER could persist immediately after stun at all velocities. Spontaneous ECoG suppression (<10%) did not differ significantly by velocity. - 55 m/s as minimum recommended velocity |
|
| Electrical stunning | Devine et al (1986) [19] | Adult cattle | Immobilization (n=10) vs no immobilization (n=2) | Implanted EEG (frontal bone screw electrodes; transhemispheric bipolar); recorder disconnected during stun/ immobilization (no EEG during current) | Uses “window of sensibility” based on LVFA amplitude ~10–40 µV - insensibility inferred when EEG falls <10 µV (toward isoelectric) and LVFA absent/ outside window |
EEG amplitude <10 μV at end of immobilization; in non-immobilized animals reached <10 μV at 57 s and 63 s after stun initiation |
| Bager et al. (1992) [5] | Calves | Head-only electrical stun + slaughter vs no-stun slaughter | ECoG+ FFT analysis of power bands 8–30 Hz (sensibility-associated) and 2–8 Hz (insensibility/ anoxia-associated) | Loss of sensibility inferred from electrocortical activity analysis (FFT; HALF etc.) | Stunned calves: permanently insensible at time of stun. Non-stunned: 6 calves lost sensibility within 10 s; one calf with carotid clot had cortical activity >52 s but sensibility not regained (FFT) |
|
| Non-penetrating captive bolt (NPCB) | Gibson et al. (2009) [22] | Calves | Single group | EEG + FFT to compute total power (Ptot); visual classification: active / transitional / high-amplitude low-frequency / isoelectric | Visual inspection + changes in total power (Ptot) | Changes sufficient to produce insensibility within 0–14 s in conscious animals (inference); biphasic Ptot response in 5 animals |
| Verhoeven et al. (2016) [7] | Veal calves | (1) Inverted captive bolt + neck cut (n=25) (2) Upright no-stun (n=7) (3) Inverted no-stun (n=25); (4) Upright no-stun then captive bolt at ~34±8 s (n=25) |
Non-invasive EEG electrodes | Visual EEG staging: baseline → transitional → unconscious when low-freq/high-amp dominates → minimal brain activity/flat EEG defined as <10% baseline amplitude | After captive bolt, absence of threat/withdrawal/corneal/eyelid reflex indicated unconsciousness per EEG; after non-stun, threat/withdrawal lost before EEG-defined unconsciousness; eyelid/corneal were conservative, persisting until ~76±50 and 85±45 s after EEG loss of consciousness | |
| Gibson et al. (2019) [17] | Bulls | PCB (n=20) vs NPCB (n=11) | EEG; visual classification + spectral analysis | Visual EEG states: Normal, Transitional (suppressed; <½ baseline amp), HALF (high-amp low-freq), Isoelectric (amp <1/8 = 12.25% baseline, little/no low-freq). | PCB: 20/20 inconsistent with consciousness; NPCB: 82% complete unconsciousness; 2 bulls showed normal EEG activity periods indicating incomplete concussion | |
| Slaughter Without Stunning | Gregory & Wotton (1984) [6] | Calves | Exsanguination after cutting carotids/jugulars | ECoG (implanted electrodes) + flash visually evoked cortical responses (VER/EP) | Loss of visually evoked cortical response to flash used as indicator of loss of brain responsiveness | Time from sticking to loss of brain responsiveness: 17 ± 4 s (range 11.5–23 s). “Flat” ECoG occurred later on average (23 ± 11 s), supporting that evoked response loss is a more direct marker than waiting for isoelectric ECoG. |
| Imlan et al. (2020) [23] | Cattle | Sharp knife (n=10) vs commercial sharp knife (n=10) | EEG + FFT spectral analysis | Not an unconsciousness criterion study. - - F50 and Ptot (and band powers) as indices of pain/stress after neck cut, not as thresholds for loss of consciousness | Higher EEG median frequency and total power in commercial sharp knife group post-slaughter, interpreted as higher pain/stress | |
| Imlan et al. (2021) [24] | Cattle | Lateral recumbency (LP) n=10 vs Upright position (UP) n=10 | EEG + FFT spectral analysis | Not unconsciousness; EEG changes interpreted as pain/stress | Differences in catecholamines; EEG responses measured; details require full text | |
| Small et al. (2025) [25] | Cattle | EEG recorded in 6; one handled commercially without EEG delay | Non-invasive EEG; offline analysis: RMS power, FFT Ptot (0–30 Hz), SEF50, SEF95, and band power | No fixed numeric EEG threshold; “insensibility duration” operationally based on absence of eye reflexes and awareness, with EEG used to document suppression and frequency shifts (↓SEF50/95, ↓Ptot) during the unconscious period | - Behavioural loss of consciousness within 10 s; EEG-based insensibility duration ~63 s or more; - EEG showed suppression and shift to lower frequencies (↓SEF50/95; ↓Ptot) for ~60–90 s or longer, with gradual recovery - Full behavioural recovery observed |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




