1. Introduction
Pneumococcal pneumonia is among the leading causes of severe respiratory infections worldwide and may advance to septic disease even if treated promptly [
1,
2]. Elderly patients and those with comorbidities or concurrent viral respiratory infections are especially vulnerable [
3]. Viral coinfections can interfere with mucociliary clearance, disrupt the epithelial barrier and create an environment suitable for bacterial superinfection [
4,
5,
6,
7,
8,
9].
Although traditionally characterized as agents of self-limiting upper respiratory tract infections, the
rhinovirus/enterovirus (RV/EV) complex—taxonomically situated within the genus Enterovirus and family
Picornaviridae — is increasingly implicated in severe pulmonary morbidity. Emerging clinical data suggests that these pathogens frequently invade the lower respiratory tract, predisposing patients to complicated pneumonia and secondary bacterial coinfections. In high-income clinical settings, the
RV/EV complex has been identified as a contributing factor of acute respiratory distress syndrome (ARDS) and multiorgan dysfunction cases [
10,
11,
12].
Additionally, it can cause viral meningitis, encephalitis, acute flaccid paralysis, myocarditis and stress cardiomyopathy, which are associated with a higher mortality [
11,
13].
In contrast, low- and middle-income countries (LMICs) lack standardized registries to document the epidemiology, natural progression, and specific risk factors associated with RV/EV infections. The data that is published is mostly derived from clinical cases or small cohort studies. In these resource-limited regions, the clinical burden of acute respiratory infections (ARIs) is significantly exacerbated by systemic barriers to healthcare, the prevalence of comorbidities, and deep-seated socioeconomic disparities, which collectively drive disproportionately high rates of morbidity and mortality.
The case presented here describes severe right-sided lobar pneumonia with confirmed Streptococcus pneumoniae bacteremia, combined with a RV/EV co-infection. The coexistence of these pathogens and the occurrence of respiratory, liver and incipient renal organ dysfunction underscore the complex clinical nature of this case.
2. Case Description
A 64-year-old male patient reported having progressive dyspnea, myalgia and a predominantly dry cough that had been present for four days. Due to worsening symptoms and a lack of response to symptomatic treatment, which the patient had initiated himself once symptoms appeared, he was consulted by his general practitioner (GP) prior to admission.
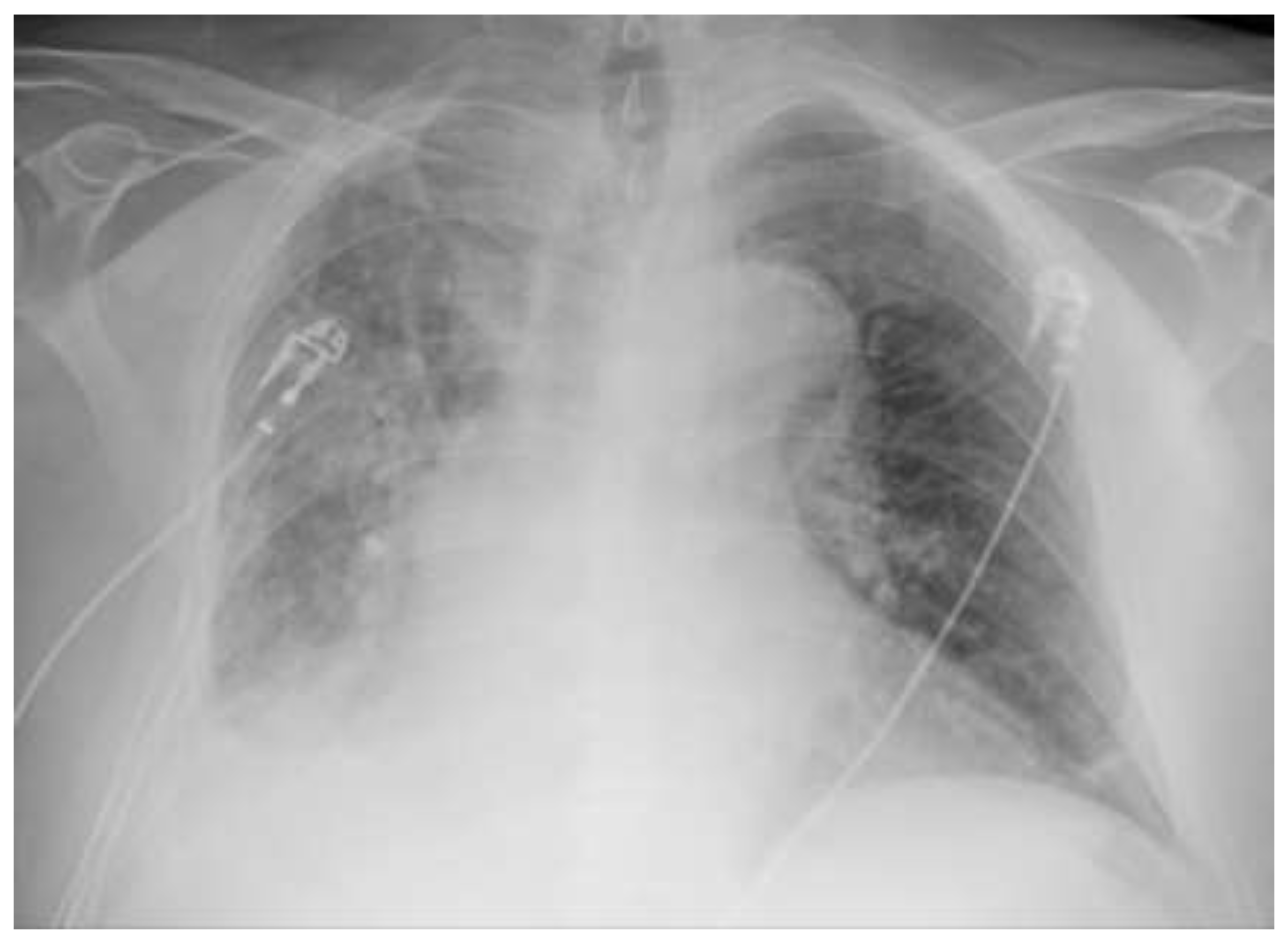
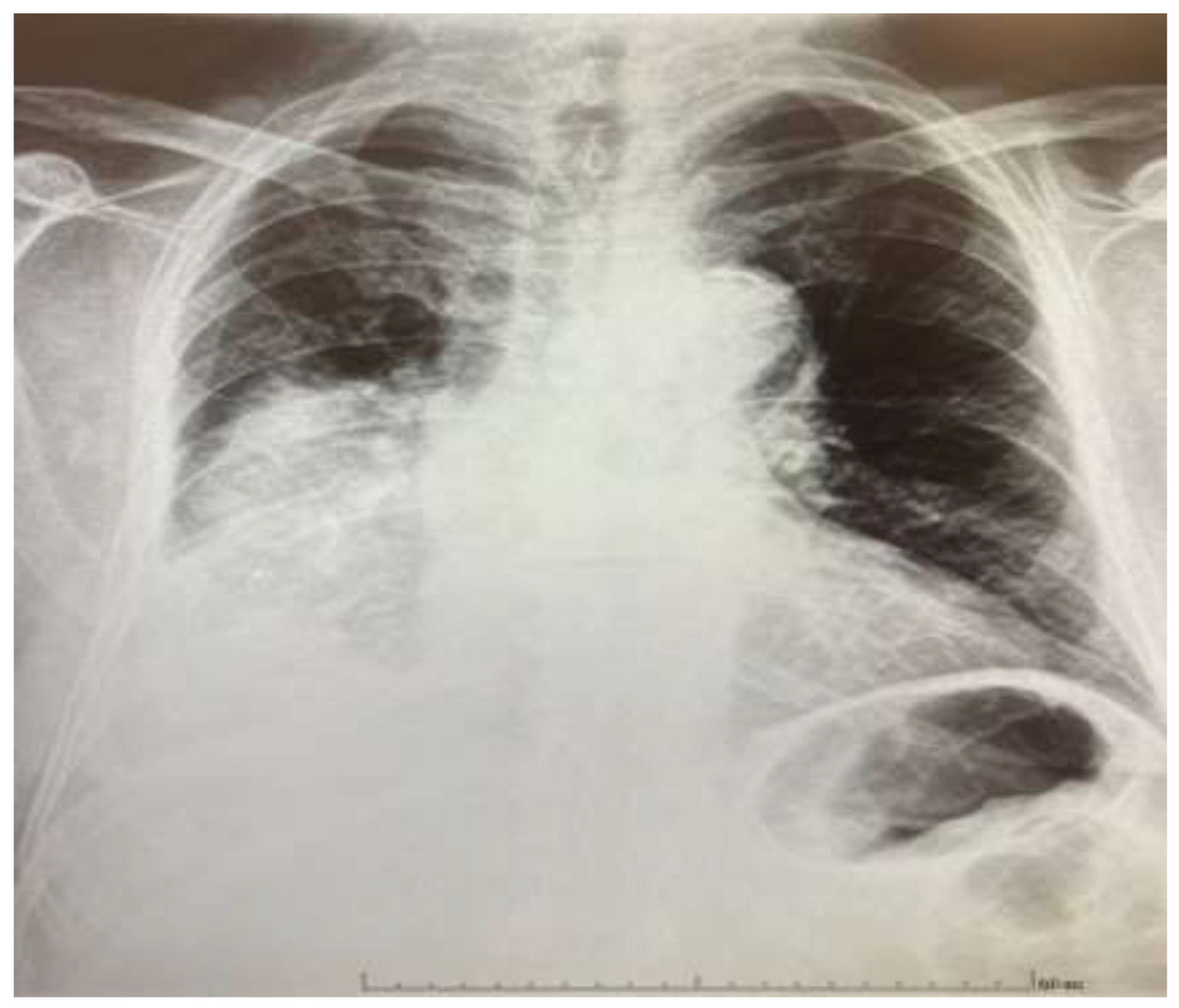
Ambulatory chest radiography (
Figure 1), revealed findings consistent with pneumonia. Concurrently, laboratory analysis demonstrated a profound elevation in inflammatory markers, with a C-reactive protein (CRP) level of 595 mg/L (<5 mg/L). Given the severity of these findings, the patient was immediately referred for hospitalization.
Relevant medical history included arterial hypertension, managed with an angiotensin II receptor blocker and a calcium channel antagonist. Social history was notable for smoking, with approximately 40 pack years. Occupational history revealed employment as a welder. Immunization history was notable for the absence of pneumococcal, influenza, and COVID-19 vaccinations.
Upon admission to the hospital (on day 1), the patient was alert and oriented in terms of time and place but in a significantly reduced general condition. Vital signs were notable for pyrexia (38.6 °C), tachycardia at 110 beats/min (bpm), and tachypnoea (30 breaths/min). His blood pressure was stable at 120/80 mmHg.
Clinically, auscultation revealed bronchial breath sounds with discrete rales over large areas of the right hemithorax, as well as dullness to percussion and an increased vocal fremitus, aligning with the suspicion of extensive consolidation.
Arterial blood gas analysis confirmed severe hypoxemia, persisting under O₂ administration (10 L/min), which led to an increase in O₂ administration (15 L/min) and a subsequent follow-up BGA (
Table 1).
Laboratory diagnostics initiated immediately on day 1 confirmed the clinical suspicion of severe systemic inflammation with leukocytosis of 18.21×10⁹/L (4.0 – 10.0 × 10⁹/L) and a continued excessively elevated CRP of 401 mg/L (< 5 mg/L). In addition, there were signs of multiple organ dysfunction characterized by elevated bilirubin and renal dysfunction, as well as elevated coagulation parameters, including D-dimer and fibrinogen (
Table 2).
Due to the fulfilment of the Sepsis–3 criteria (proven infection plus organ dysfunction: hepatic and renal involvement yielding a SOFA score ≥2)[
14,
15,
16,
17,
18] and the risk of further rapid deterioration, the patient was transferred to the Intensive Care Unit (ICU) on the second day for close monitoring.
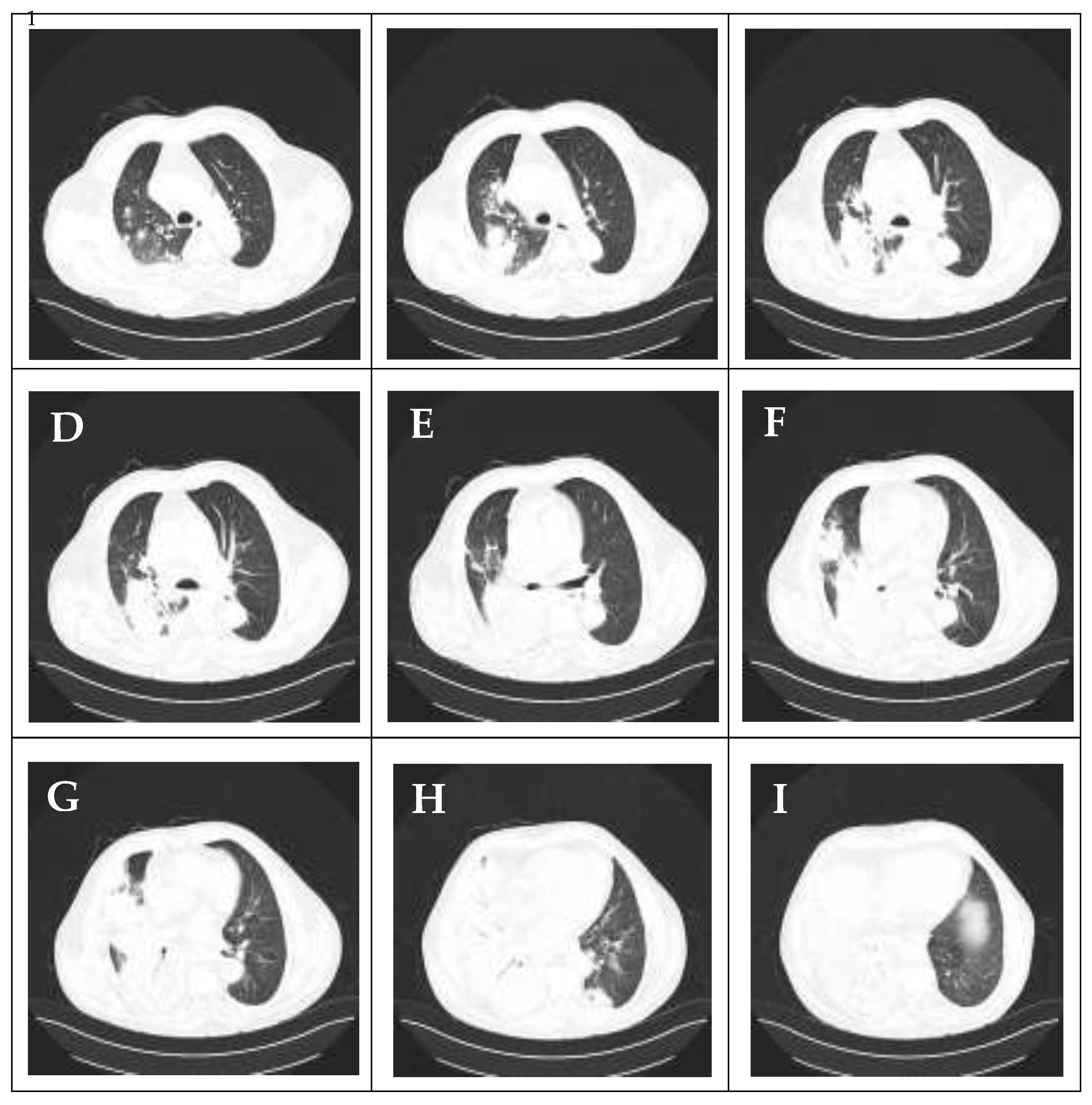
Computed tomography (CT) of the thorax, performed on admission, confirmed the diagnosis of right lower lobe pneumonia with involvement of small areas in the right upper lobe. Furthermore, the CT revealed an air bronchogram, a “tree-in-bud” sign, a small area of inflammation in the left lower lobe, and a small right-sided pleural effusion (less than 20 mm) not indicated for drainage (
Figure 2).
Microbiologically, multiplex PCR for 22 respiratory viruses performed on day 2, detected a co-infection with rhinovirus/enterovirus and failed to detect the following targets: Adenovirus, Bocavirus, Coronavirus 229E, Coronavirus HKU1, Coronavirus NL63, Coronavirus OC43, Human Metapneumovirus A/B, Influenza A subtype H1N1/2009, Influenza A subtype H1, Influenza subtype H3, Parainfluenza Virus 1, Parainfluenza Virus 2, Parainfluenza Virus 3, Parainfluenza Virus 4, Mycoplasma pneumoniae, and Bordetella pertussis.
Due to the severity of the disease, empirical antibiotic therapy with Bacteripime (4 g/day) and Levofloxacin (500 mg/day) was administered. However, on the second day, after additional results, Bacteripime was replaced by Meropenem (4 g/day). Blood cultures taken on days 1 and 2 later revealed Streptococcus pneumoniae, confirming the diagnosis of bacterial infection with bacteremia, while all sputum and urine cultures were negative.
During treatment in the ICU, the patient’s respiratory function initially deteriorated progressively, accompanied by the development of yellowish purulent sputum. The further course was prolonged and fluctuating. While kidney function and bilirubin rapidly normalized under the initiated therapy, the inflammatory response presented a mixed picture.
On day 6, leukocytes rose significantly again to 23.5 × 10⁹/L (4.0–10.0 × 10⁹/L), whereas CRP had already fallen to 87 mg/l (< 5 mg/L) (
Table 2).
Figure 2.
CT images of the chest obtained on admission demonstrate lobar pneumonia predominantly affecting the right lung. The scan reveals: (A–C) multiple inflammatory foci in the right upper/middle lobe; (D–I) dense consolidation with air bronchograms in the right lower lobe, alongside with smaller foci of inflammation in the left lower lobe; and a small associated right-sided pleural effusion.
Figure 2.
CT images of the chest obtained on admission demonstrate lobar pneumonia predominantly affecting the right lung. The scan reveals: (A–C) multiple inflammatory foci in the right upper/middle lobe; (D–I) dense consolidation with air bronchograms in the right lower lobe, alongside with smaller foci of inflammation in the left lower lobe; and a small associated right-sided pleural effusion.
The patient’s respiratory failure remained critical during this phase and required the administration of high-dose oxygen supplement at up to 15 L/min via a Venturi mask to ensure adequate oxygenation. Microbiological testing on day 7 detected Micrococcus luteus, which was interpreted as contamination by the hospital laboratory. However, the radiological follow-up (chest radiograph on day 8) showed a significant improvement (
Figure 3).
Following this critical phase, the patient showed significant clinical improvement. By day 10, CRP fell significantly to 53 mg/L (< 5 mg/L). A sputum analysis on day 10 detected Candida lusitaniae, which was interpreted as secondary colonization, and Fluconazole was added. With the infection under control, respiratory function improved noticeably.
A follow-up BGA documented a significant improvement in gas exchange with reduced oxygen supply. Also, the chest radiograph (Figure 4), performed before discharge, showed almost a full resolution of the consolidation. The patient was finally discharged on day 20 in a good general condition.
Figure 2.
Follow-up chest X-ray (Day 8 post treatment initiation). Anteroposterior chest radiograph showing favorable dynamics, with partial resorption of the consolidation.
Figure 2.
Follow-up chest X-ray (Day 8 post treatment initiation). Anteroposterior chest radiograph showing favorable dynamics, with partial resorption of the consolidation.
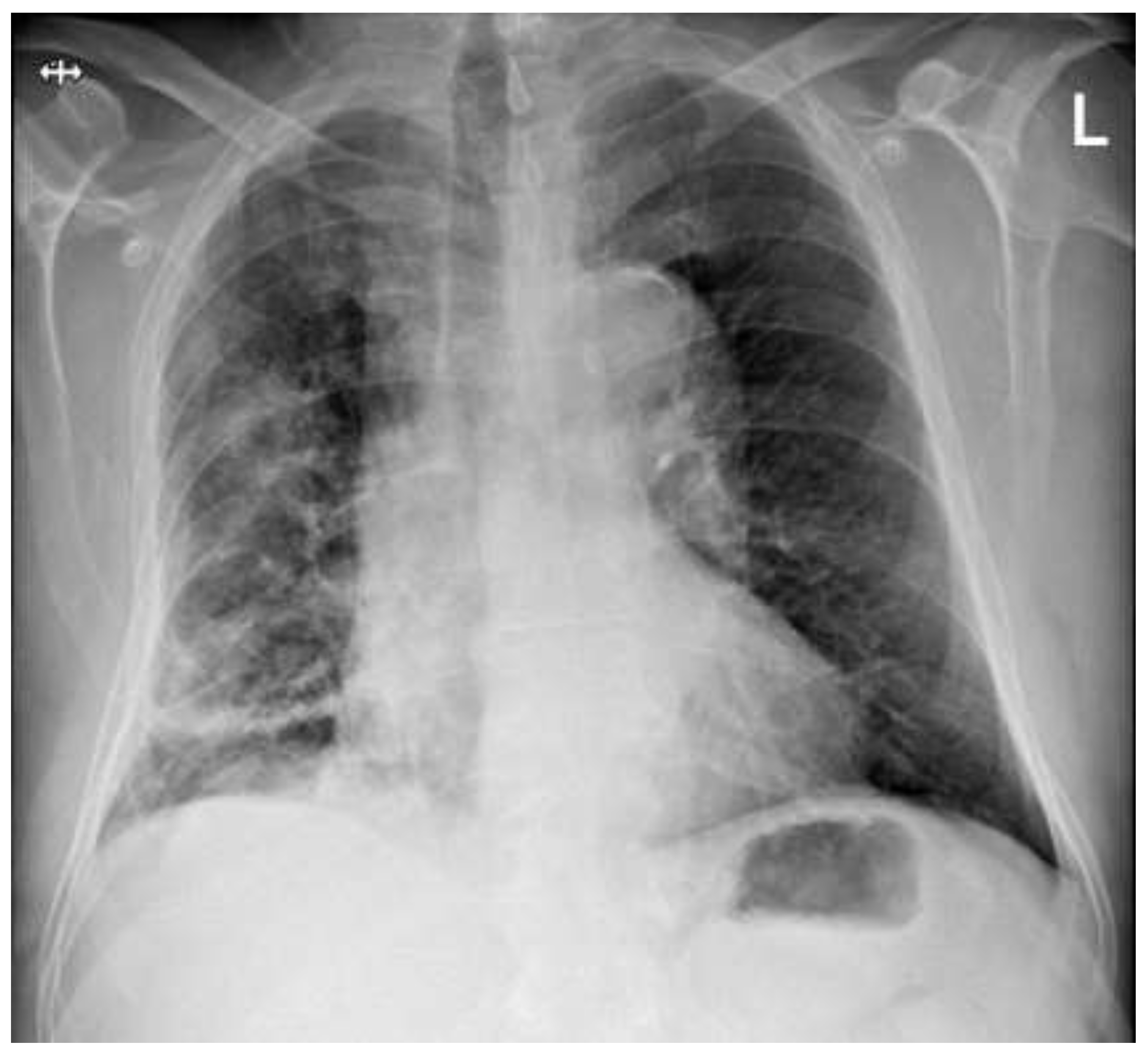
Figure 3.
Follow-up chest X-ray (Day 15 post treatment initiation). The chest radiograph shows substantial improvement compared to
Figure 1 and
Figure 2. There is significant aeration of the right lung with the resolution of the pleural effusion, though residual opacities persist in the right lower zone.
Figure 3.
Follow-up chest X-ray (Day 15 post treatment initiation). The chest radiograph shows substantial improvement compared to
Figure 1 and
Figure 2. There is significant aeration of the right lung with the resolution of the pleural effusion, though residual opacities persist in the right lower zone.
Table 2.
A selection of the patient’s lab results over the course of his hospitalization.
Table 2.
A selection of the patient’s lab results over the course of his hospitalization.
| Parameter |
Ref. Range |
Day 1 |
Day 4 |
Day 6 |
Day 8 |
Day 10 |
Day 18 |
| Leukocytes x10⁹/L |
4.0 – 10.0 |
18.21 |
– |
23.5 |
– |
13.57 |
11.8 |
| Neutrophils |
40 – 70 |
91.4 |
– |
72.7 |
– |
73 |
72.1 |
| C-reactive protein mg/L |
<5 |
401 |
277 |
87 |
– |
53 |
12.6 |
| Bilirubin, direct µmol/L |
0 – 5 |
29.6 |
10.7 |
– |
– |
– |
– |
| Bilirubin, total mol/L |
<21 |
34 |
15 |
– |
– |
– |
– |
| D-Dimer mg/L |
<0.5 |
2.52 |
– |
– |
– |
– |
– |
| Fibrinogen g/L |
1.8 – 3.5 |
12.7 |
– |
– |
– |
3.5 |
– |
| Urea mmol/L |
2.5 –7.5 |
14.1 |
13.2 |
– |
8.6 |
7.5 |
– |
| Creatinine µmol/L |
62 – 106 |
114 |
77 |
– |
67 |
– |
– |
| Anti C.pneumoniae (ELISA’U) |
(<0.8)
(<16) |
IgM: 0.1
IgG: 72 |
– |
– |
– |
– |
– |
| Anti M.pneumoniae (ELISA’U) |
(<0.8)
(<16) |
IgM: 0.2
IgG: 44 |
– |
– |
– |
– |
– |
| Legionella antigen, urine |
Negative |
Negative |
– |
– |
– |
– |
– |
3. Treatment and Outcome
The patient received broad-spectrum antibiotics with Bacteripime for two days followed by Meropenem and Levofloxacin for fourteen days, Methylprednisolone 40 mg (from 4th day) with a tapered dose for 7 days; Fluconazole for ten days; fluids; antihypertensives; anticoagulation prophylaxis with Nadroparin; oxygen supplement up to 15 L/min; and symptomatic therapy. At present, there is no specific treatment for the RV/EV infection, and it was entirely supportive.
Clinical evolution was favorable, with resolution of fever, cough, full normalization of renal and liver markers, and with almost full resolution of infiltrates and the pleural effusion at discharge.
At the 15- and 30-day follow-up, physical examination was unremarkable. Laboratory tests and BGAs showed normal values. However, the patient still felt exhausted, easily fatigued, and not ready to return to his workplace.
4. Discussion
EV/RV infection is notoriously known for causing a relatively mild URTI in the majority of cases. However, it can also rarely be a cause of ARDS in immunocompetent adults [
10,
12], cardiomyopathy [
11], meningitis, encephalitis, even paralysis [
10,
11,
12,
13]. In some cases, patients can develop multiorgan dysfunction, even death, in the most complicated cases [
10].
In the adult population, epidemiological data from influenza-like illness cohorts reveal frequent viral-bacterial pairs, with
RV and
S. pneumoniae co-detection occurring in approximately 4% of cases, correlating with increased risk of lower respiratory complications. Likewise, in intensive care units, concomitant viral-bacterial pneumonia involving
RV/EV and
S. pneumoniae is associated with prolonged mechanical ventilation and higher mortality rates compared to infections caused by a single pathogen. Additionally,
adenovirus/RV and
EV/RV were the two most incident concomitant viral pairs (1.43% and 0.83%), while
RV and
S. pneumoniae was one of the top 3 viral-bacterial pairs [
19].
Another study revealed that human RV represents the second leading cause of severe acute respiratory infection-related hospitalizations (23.7%), trailing only behind influenza. Predominantly manifesting as pneumonia (68.8%), these cases frequently require critical care intervention (31.2%) and are associated with a substantial in-hospital mortality rate of 12.5% [
20,
21,
22,
23,
24].
Surveys indicate that severe acute respiratory infections due to RV often seen in children, can appear and progress similarly in elderly patients, especially in those with multiple overlying risk factors.
Our case demonstrated rapid progression of invasive pneumococcal disease (IPD) with PCR confirmed
RV/EV coinfection in an individual with specific occupational risk exposure and non-specific other risk factors (i.e. smoking and inadequate immunization status). The patient’s work-related exposure as a welder is a crucial element in this case. Epidemiological investigations show significantly increased risk for welders developing severe pneumococcal pneumonia [
25].
On the one hand, an inhalation of metallic vapors (primarily iron particles) induces oxidative stress and impairs pulmonary macrophage function, thereby compromising local immune defense and facilitating bacterial invasion. The fact that the patient didn’t have the appropriate vaccination protection highlights the necessity for a better applicability of occupational health prevention recommendations (pneumococcal vaccination) in such at-risk demographics [
25].
On the other hand, the infection with
RV/EV induces injury to the respiratory epithelium and impairs mucociliary clearance, allowing for adherence to and invasion of
Streptococcus pneumoniae [
3,
4,
5,
6,
7,
8,
9].
Clinically, our case was associated with rapid onset of sepsis, according to the Sepsis-3 definition. This was manifested by mild renal and hepatic dysfunction, yielding a SOFA Score ≥ 2. Additionally, acute respiratory failure with severe ARDS (PaO2/FiO2=185), coagulopathy and increased inflammatory markers were notable. The patient’s initial SOFA score of 5 served as an early predictor of a severe clinical course, associated with a predicted mortality of 20.2% [
14,
15,
16,
17,
18].
The clinical course also demonstrated that biochemical recovery (normalization of kidney function and bilirubin) and the inflammatory response (leukocytes, CRP) may be separated in time. The gradual increase in the WBC on day 6 (with CRP already falling) was quite interesting. Although this initially aroused suspicion of a nosocomial superinfection or failure of the antibiotic therapy, microbiological diagnostics (exclusion of pathogenic germs and detection of M. luteus as a contaminant) revealed that this was probably a rather delayed inflammatory reaction or lysis effect rather than a further fulminant infection. Another logical explanation may be due to the application of corticosteroids (less than 48 hours). The decision to continue antibiotic therapy based on the overall clinical picture and the drop in CRP proved to be correct.
We observed a significant dissociation between inflammatory markers, with leukocytes increasing despite declining CRP and clinical stabilization. This report emphasizes that the biochemical response may be non-linear and potentially misleading; therefore, decisions regarding antibiotic management should be guided by the patient’s overall clinical development rather than isolated laboratory parameters. Ultimately, rapid detection of sepsis combined with immediate initiation of therapeutic intervention resulted in a very good recovery without unnecessary escalation of therapy, while also avoiding potentially severe lifelong complications.
The case exemplifies the dangerous combination of viral-bacterial co-infection. This interaction underscores the need for multiplex diagnostic approaches to guide appropriate therapies, potentially mitigating the progression to severe complications.
5. Conclusions
This case demonstrates the successful treatment of life-threatening invasive pneumococcal disease with concomitant RV/EV infection. Probably, invasive pneumococcal disease was attributable to the lack of immunization. Pneumococcal immunization remains a cornerstone of prevention, significantly reducing invasive disease rates even in the high-risk context of concomitant viral infections. While individual risk factors such as occupational exposure contributed to the rapid onset of sepsis, the most important lesson to take away is the importance of clinical monitoring during the recovery phase. In addition, the case also demonstrates the extreme importance of early assessment of patients’ risk factors, occupational history, and immunization status to correctly analyze their risk for future complications during the course of disease.
Author Contributions
Conceptualization: G.K., L.N. and P.P.; methodology: G.K., L.N. and P.P.; software: G.K., L.N. R.P.C., and P.P; resources: G.K., L.N. and P.P.; data curation: G.K., L.N. and P.P; writing—original draft preparation: G.K., L.N., P.P. and R.P.C; writing—review and editing: G.K., L.N., P.P., R.P.C.; visualization: G.K., L.N. and P.P; supervision P.P.; project administration: P.P., P.Y., D.P., V.D., T.D., A.A., P.G., N.M., F.M., M.T., A.B., N.I., V.T., Y.D. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Acknowledgments
During the preparation of this manuscript/study, the author(s) used [tool name, version information] for the purposes of [description of use]. The authors have reviewed and edited the output and take full responsibility for the content of this publication.”.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| ARDS |
Acute Respiratory Distress Syndrome |
| BGA |
Blood Gas Analysis |
| CAP |
community-acquired pneumonia |
| CT |
Computed Tomography |
| GP |
general practitioner |
| ICU |
Intensive Care Unit |
| LMICs |
low- and middle-income countries |
| PCR |
Polymerase Chain Reaction |
| RV/EV |
rhinovirus/enterovirus complex |
| SOFA |
the Sequential Organ Failure Assessment |
References
- European Centre for Disease Prevention and Control Invasive Pneumococcal Disease - Annual Epidemiological Report for 2022; European Centre for Disease Prevention and Control: Stockholm, 2025; p. 12.
- Centers for Disease Control and Prevention Active Bacterial Core Surveillance Report, Emerging Infections Program Network, Streptococcus Pneumoniae, 2023; Centers for Disease Control and Prevention, 2023;
- Bush, L.M.; Vazquez-Pertejo, M.T. Pneumococcal Infections - Infectious Diseases Available online: https://www.msdmanuals.com/professional/infectious-diseases/gram-positive-cocci/pneumococcal-infections (accessed on 8 December 2025).
- Greenberg, S.B. Respiratory Consequences of Rhinovirus Infection. Arch. Intern. Med. 2003, 163, 278–284. [CrossRef]
- Fica, A.; Dabanch, J.; Andrade, W.; Bustos, P.; Carvajal, I.; Ceroni, C.; Triantafilo, V.; Castro, M.; Fasce, R. Clinical Relevance of Rhinovirus Infections among Adult Hospitalized Patients. Braz. J. Infect. Dis. 2015, 19, 118–124. [CrossRef]
- Wang, G. Enteroviruses and Rhinoviruses. In Molecular Medical Microbiology; Elsevier, 2024; pp. 2451–2475 ISBN 978-0-12-818619-0.
- Messacar, K. Overview of Enterovirus Infections - Infectious Diseases Available online: https://www.msdmanuals.com/professional/infectious-diseases/enteroviruses/overview-of-enterovirus-infections (accessed on 8 December 2025).
- Cecchini, A.; Othman, A.; Kaur, K.; Richardson, A.; Cecchini, A. Enterovirus-Human-Rhinovirus Infection Leading to Acute Respiratory Distress Syndrome: A Case Report. Cureus 2022. [CrossRef]
- Avgoustou, E.; Spyridaki, A.; Pothitos, G.; Papadopoulos, A.; Kois, S.; Vassilara, F. Enterovirus-Rhinovirus-Induced Acute Respiratory Distress Syndrome in Adults: A Case Report and Short Literature Review. Case Rep. Infect. Dis. 2023, 2023, 1–6. [CrossRef]
- Smith, M.E.; Wilson, P.T. Human Rhinovirus/Enterovirus in Pediatric Acute Respiratory Distress Syndrome. J. Pediatr. Intensive Care 2020, 09, 081–086. [CrossRef]
- Sharfi, M.; Elshakh, H.; Jee Ah Rhee, C.; Genin, D.I.; Basu, A. FROM COLD TO CRISIS: A RARE CASE OF ENTEROVIRUS-HUMAN RHINOVIRUS-INDUCED ARDS AND STRESS-INDUCED CARDIOMYOPATHY. CHEST 2023, 164, A2850–A2851. [CrossRef]
- Hamid, K.; Ali, M.; Devasahayam, J. Acute Respiratory Distress Syndrome Secondary to Enterovirus-Human-Rhinovirus Infection in an Adult. Cureus 2022. [CrossRef]
- Tapparel, C.; Siegrist, F.; Petty, T.J.; Kaiser, L. Picornavirus and Enterovirus Diversity with Associated Human Diseases. Infect. Genet. Evol. 2013, 14, 282–293. [CrossRef]
- Singer, M.; Deutschman, C.S.; Seymour, C.W.; Shankar-Hari, M.; Annane, D.; Bauer, M.; Bellomo, R.; Bernard, G.R.; Chiche, J.-D.; Coopersmith, C.M.; et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016, 315, 801. [CrossRef]
- Seymour, C.W.; Liu, V.X.; Iwashyna, T.J.; Brunkhorst, F.M.; Rea, T.D.; Scherag, A.; Rubenfeld, G.; Kahn, J.M.; Shankar-Hari, M.; Singer, M.; et al. Assessment of Clinical Criteria for Sepsis: For the Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016, 315, 762. [CrossRef]
- Shankar-Hari, M.; Phillips, G.S.; Levy, M.L.; Seymour, C.W.; Liu, V.X.; Deutschman, C.S.; Angus, D.C.; Rubenfeld, G.D.; Singer, M.; for the Sepsis Definitions Task Force Developing a New Definition and Assessing New Clinical Criteria for Septic Shock: For the Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016, 315, 775. [CrossRef]
- Abraham, E. New Definitions for Sepsis and Septic Shock: Continuing Evolution but With Much Still to Be Done. JAMA 2016, 315, 757. [CrossRef]
- Jacob, J.A. New Sepsis Diagnostic Guidelines Shift Focus to Organ Dysfunction. JAMA 2016, 315, 739. [CrossRef]
- Ferrari, A.; Schiavetti, I.; Ogliastro, M.; Minet, C.; Sibilio, R.; Giberti, I.; Costa, E.; Massaro, E.; Lai, P.L.; Mosca, S.; et al. Co-Detection of Respiratory Pathogens among ILI Patients: Characterization of Samples Collected during the 2018/19 and 2019/20 Pre-Pandemic Seasons. BMC Infect. Dis. 2024, 24, 881. [CrossRef]
- Mcmillan, J.A.; Weiner, L.B.; Higgins, A.M.; Macknight, K. Rhinovirus Infection Associated with Serious Illness among Pediatric Patients: Pediatr. Infect. Dis. J. 1993, 12, 321–325. [CrossRef]
- Fernandez-Sarmiento, J.; Corrales, S.C.; Obando, E.; Amin, J.; Bastidas Goyes, A.; Barrera Lopez, P.A.; Bernal Ortiz, N. Factors Associated with Severe Acute Respiratory Infections Due to Rhinovirus/Enterovirus Complex in Children and Their Comparison with Those of Respiratory Syncytial Virus. Arch. Pediatr. Infect. Dis. 2021, 10. [CrossRef]
- Covaci, S.; Filimon, C.; Craiu, M. Exploring the Clinical Characteristics and Outcomes of Rhinovirus Infection in Hospitalized Children Compared with Other Respiratory Viruses. Children 2024, 11, 1303. [CrossRef]
- Ruohtula, T.; Kondrashova, A.; Lehtonen, J.; Oikarinen, S.; Hämäläinen, A.-M.; Niemelä, O.; Peet, A.; Tillmann, V.; Nieminen, J.K.; Ilonen, J.; et al. Immunomodulatory Effects of Rhinovirus and Enterovirus Infections During the First Year of Life. Front. Immunol. 2021, 11, 567046. [CrossRef]
- Zlateva, K.T.; de Vries, J.J.C.; Coenjaerts, F.E.J.; van Loon, A.M.; Verheij, T.; Little, P.; Butler, C.C.; Goossens, H.; Ieven, M.; Claas, E.C.J. Prolonged Shedding of Rhinovirus and Re-Infection in Adults with Respiratory Tract Illness. Eur. Respir. J. 2014, 44, 169–177. [CrossRef]
- Riccò, M.; Ferraro, P.; Zaffina, S.; Camisa, V.; Marchesi, F.; Gori, D. Vaccinating Welders against Pneumococcus: Evidence from a Systematic Review and Meta-Analysis. Vaccines 2023, 11, 1495. [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).