Submitted:
17 February 2026
Posted:
28 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
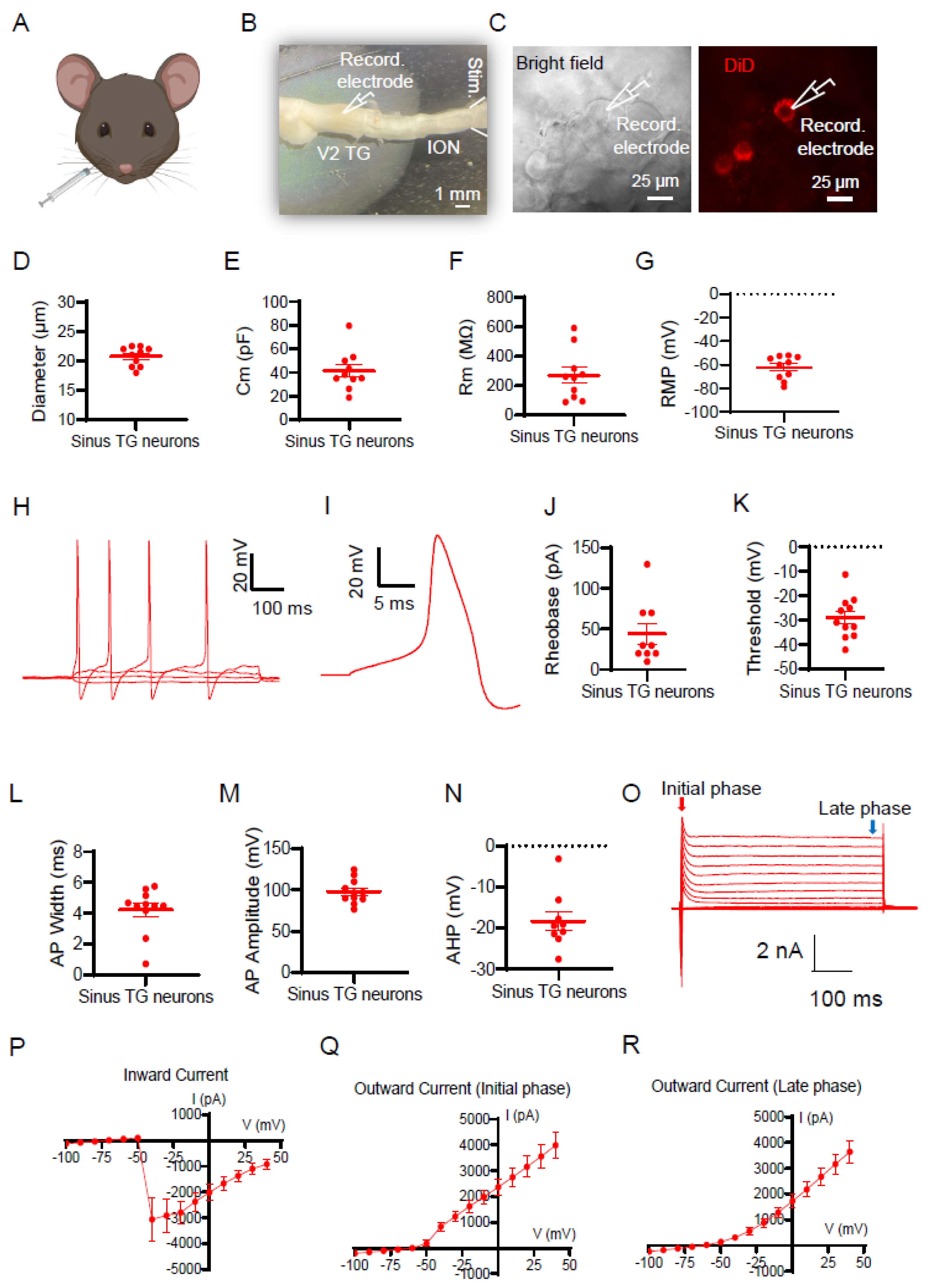
2.1. Animals
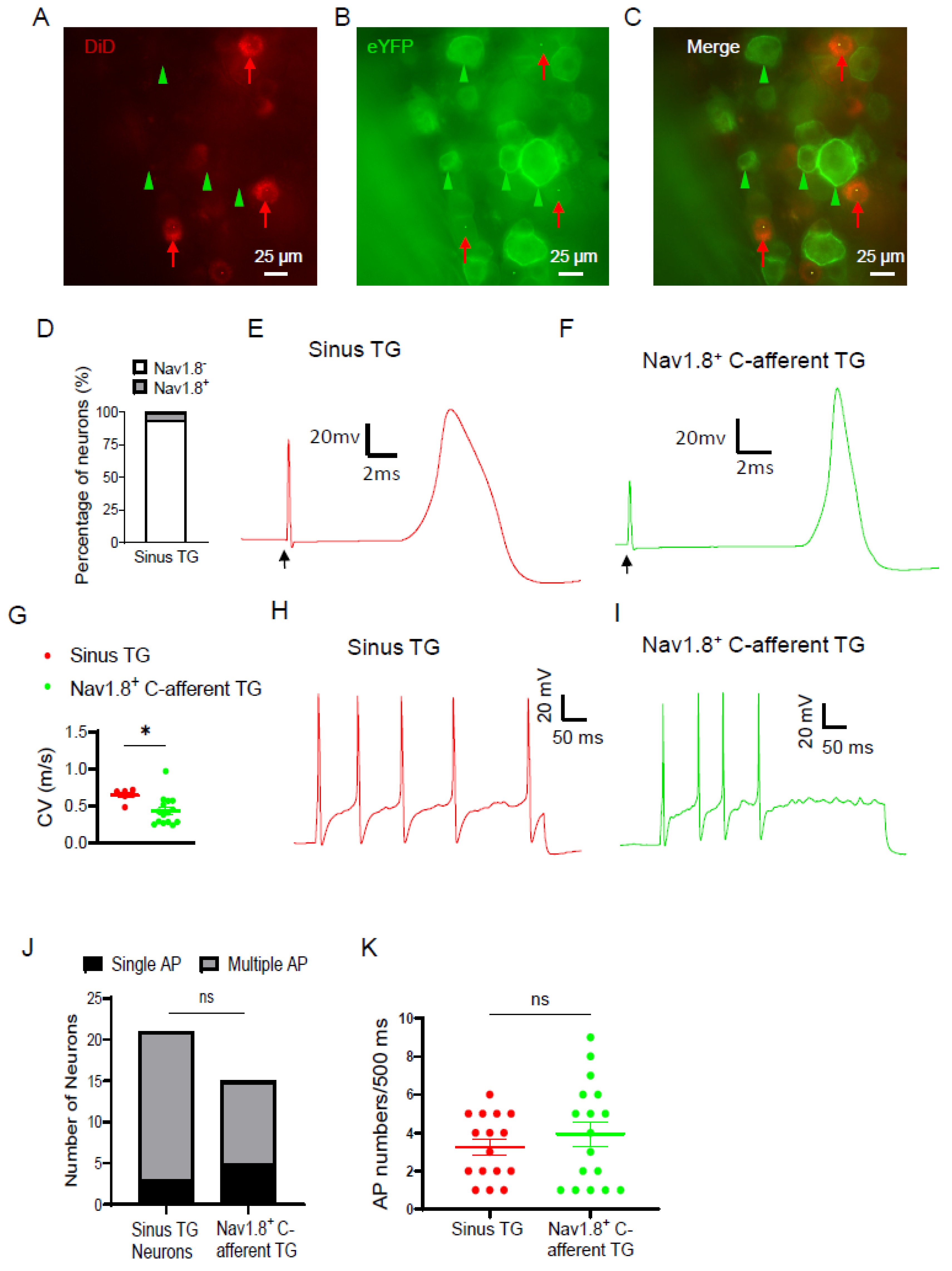
2.2. Retrograde Labeling of Maxillary Sinus Neurons with Fluorescent Dye DiD
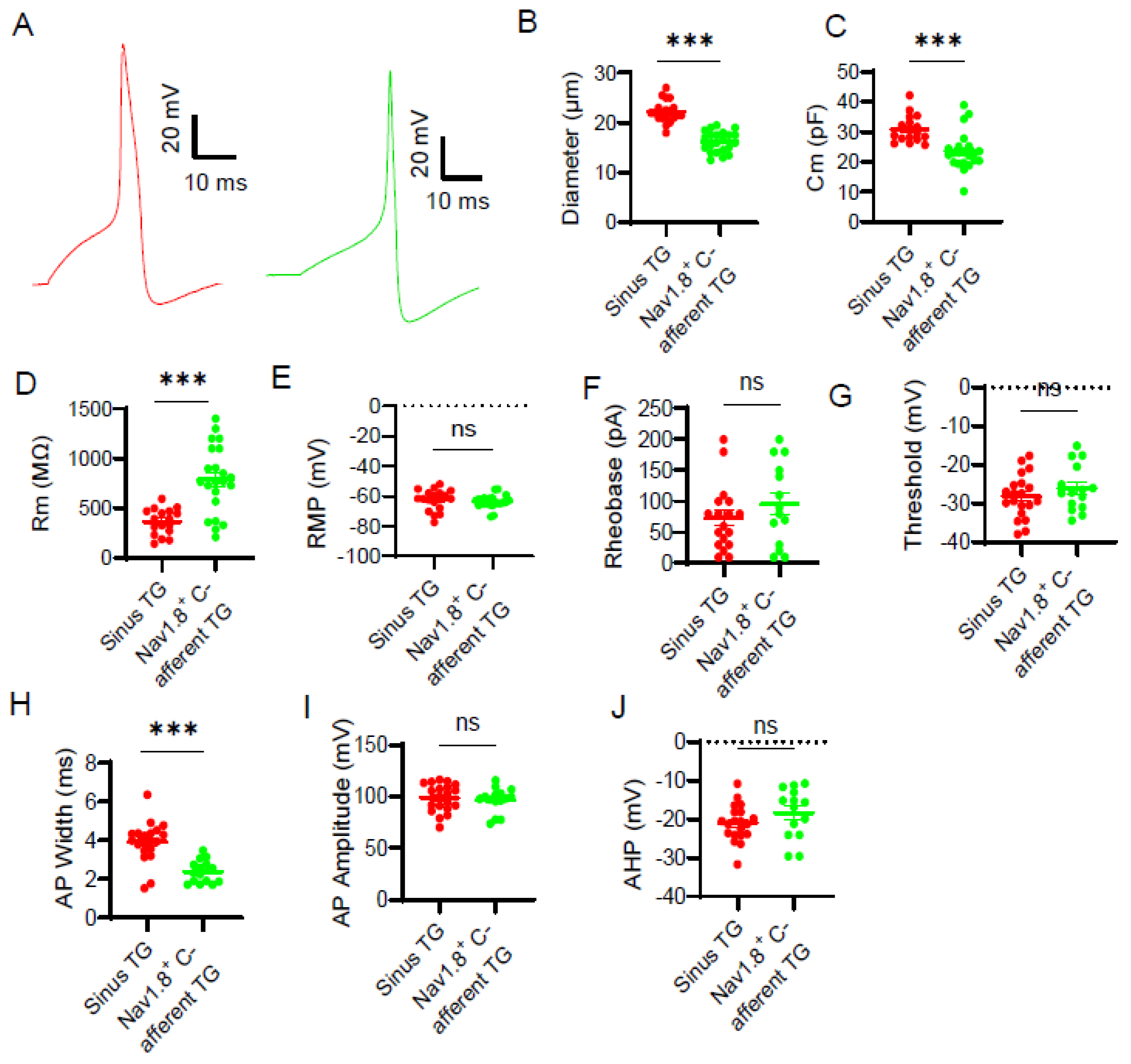
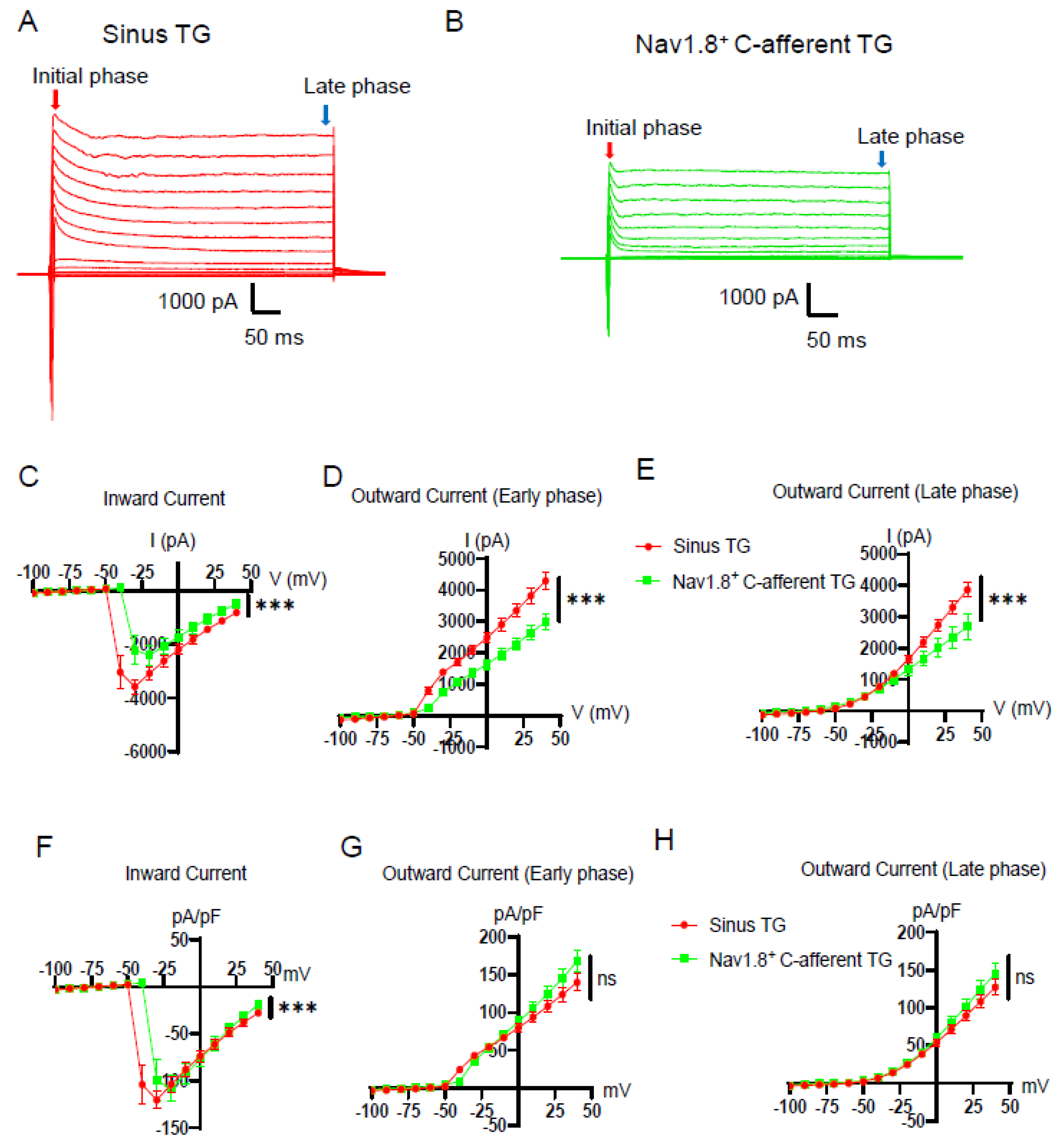
2.3. Ex Vivo Trigeminal Ganglion Preparations and Patch-Clamp Recordings
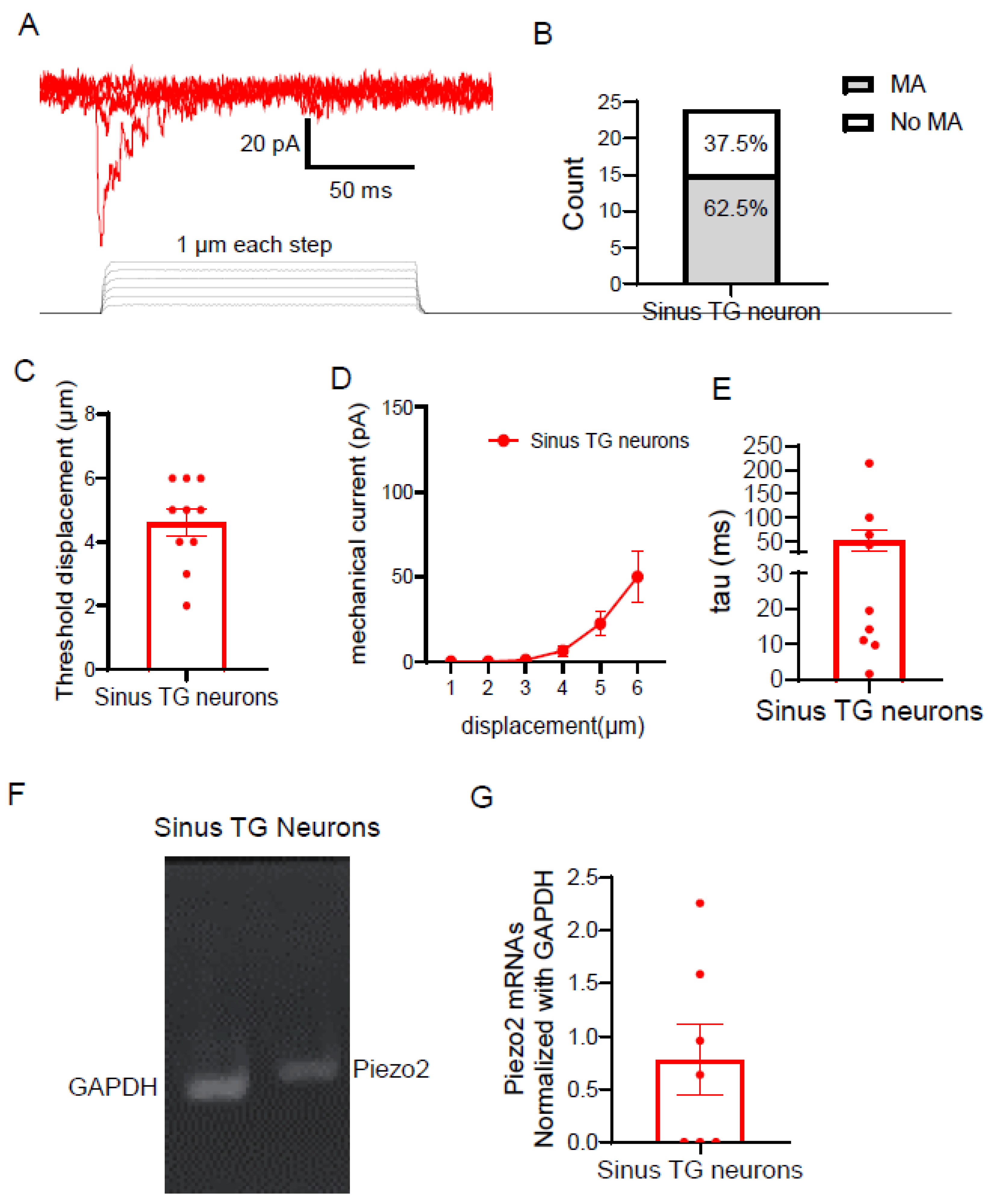
2.4. Mechanical Stimulation
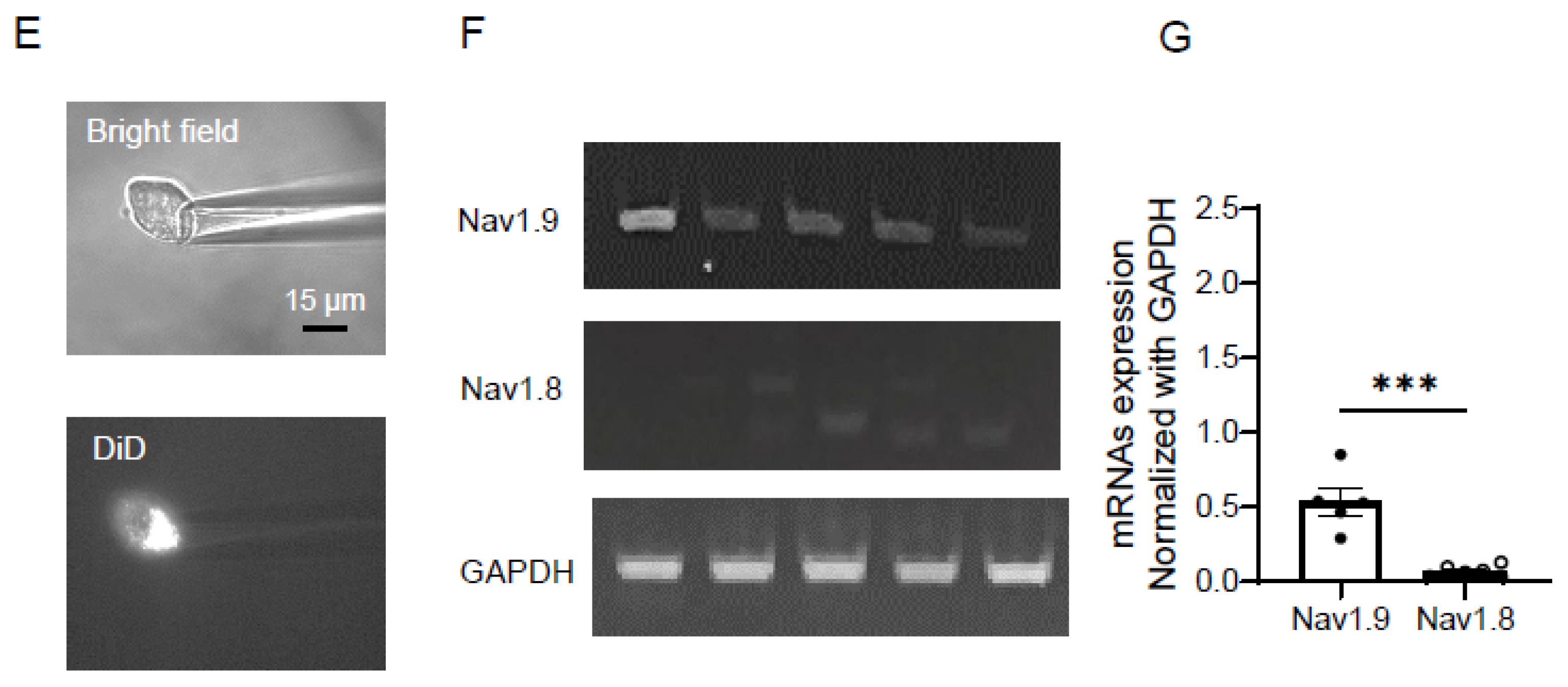
2.5. Dissociation of Individual Sinus TG Neurons and Single-Cell RT-PCR
2.6. Data Analysis
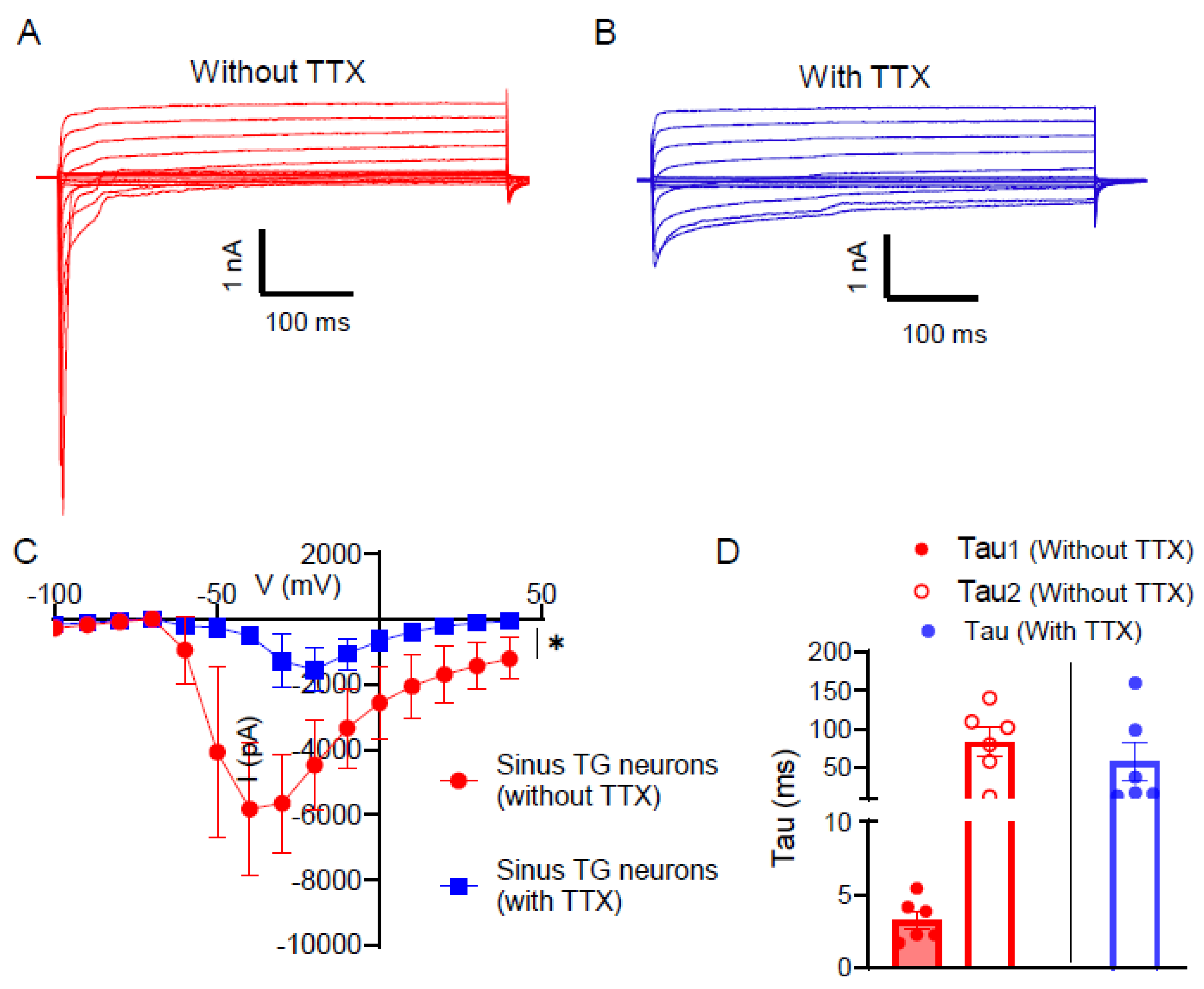
3. Results
4. Discussion
Author Contributions
Acknowledgments
Declaration of Conflicting Interests
References
- Lafci Fahrioglu, S., N. VanKampen, and C. Andaloro, Anatomy, Head and Neck, Sinus Function and Development, in StatPearls. 2025: Treasure Island (FL).
- Whyte, A. and R. Boeddinghaus, The maxillary sinus: physiology, development and imaging anatomy. Dentomaxillofac Radiol, 2019. 48(8): p. 20190205. [CrossRef]
- De Corso, E., et al., Facial pain: sinus or not? Acta Otorhinolaryngol Ital, 2018. 38(6): p. 485-496. [CrossRef]
- Helliwell, T., Inflammatory diseases of the nasal cavities and paranasal sinuses. Diagn Histopathol (Oxf), 2010. 16(6): p. 255-264. [CrossRef]
- Dykewicz, M.S. and D.L. Hamilos, Rhinitis and sinusitis. J Allergy Clin Immunol, 2010. 125(2 Suppl 2): p. S103-15.
- Misirovs, R. and S. Mohamad, Reverse Squeeze Maxillary Sinus Barotrauma. Ear Nose Throat J, 2020. 99(1): p. NP9-NP10. [CrossRef]
- Lee, S. and A.P. Lane, Chronic rhinosinusitis as a multifactorial inflammatory disorder. Curr Infect Dis Rep, 2011. 13(2): p. 159-68. [CrossRef]
- Eross, E., D. Dodick, and M. Eross, The Sinus, Allergy and Migraine Study (SAMS). Headache, 2007. 47(2): p. 213-24. [CrossRef]
- Kari, E. and J.M. DelGaudio, Treatment of sinus headache as migraine: the diagnostic utility of triptans. Laryngoscope, 2008. 118(12): p. 2235-9. [CrossRef]
- Agius, A.M., N.S. Jones, and R. Muscat, Prospective three-year follow up of a cohort study of 240 patients with chronic facial pain. J Laryngol Otol, 2014. 128(6): p. 518-26. [CrossRef]
- Lal, D., A. Rounds, and D.W. Dodick, Comprehensive management of patients presenting to the otolaryngologist for sinus pressure, pain, or headache. Laryngoscope, 2015. 125(2): p. 303-10. [CrossRef]
- Ali, A.H.A., et al., Incidental detection of paranasal sinuses abnormalities on CT imaging of the head in Saudi adult population. PLoS One, 2022. 17(9): p. e0270764. [CrossRef]
- Liu, Y., et al., Clinical Manifestations and Management Challenges in Symptomatic Sinonasal Disorders With Normal CT Scans. Ear Nose Throat J, 2025: p. 1455613251356347. [CrossRef]
- Cady, R.K. and C.P. Schreiber, Sinus headache or migraine? Considerations in making a differential diagnosis. Neurology, 2002. 58(9 Suppl 6): p. S10-4. [CrossRef]
- Al-Hashel, J.Y., et al., Migraine misdiagnosis as a sinusitis, a delay that can last for many years. J Headache Pain, 2013. 14(1): p. 97. [CrossRef]
- Schreiber, C.P., et al., Prevalence of migraine in patients with a history of self-reported or physician-diagnosed "sinus" headache. Arch Intern Med, 2004. 164(16): p. 1769-72. [CrossRef]
- Kim, J.R., et al., Healthcare resource use and costs associated with the misdiagnosis of migraine. Headache, 2025. 65(1): p. 35-44. [CrossRef]
- Kim, H.K., et al., The Trigeminal Sensory System and Orofacial Pain. Int J Mol Sci, 2024. 25(20). [CrossRef]
- Pena, E., et al., Neuronal body size correlates with the number of nucleoli and Cajal bodies, and with the organization of the splicing machinery in rat trigeminal ganglion neurons. J Comp Neurol, 2001. 430(2): p. 250-63.
- Hameed, S., Na(v)1.7 and Na(v)1.8: Role in the pathophysiology of pain. Mol Pain, 2019. 15: p. 1744806919858801. [CrossRef]
- Luiz, A.P., et al., The role of Nav1.9 channel in the development of neuropathic orofacial pain associated with trigeminal neuralgia. Mol Pain, 2015. 11: p. 72. [CrossRef]
- Aust, R., B. Falck, and H. Svanholm, Studies of the gas exchange and pressure in the maxillary sinuses in normal and infected humans. Rhinology, 1979. 17(4): p. 245-51.
- Li, Q., et al., Characterizing the respiratory-induced mechanical stimulation at the maxillary sinus floor following sinus augmentation by computational fluid dynamics. Front Bioeng Biotechnol, 2022. 10: p. 885130. [CrossRef]
- Ikeda, R., et al., Merkel cells transduce and encode tactile stimuli to drive Abeta-afferent impulses. Cell, 2014. 157(3): p. 664-75. [CrossRef]
- Woo, S.H., et al., Piezo2 is required for Merkel-cell mechanotransduction. Nature, 2014. 509(7502): p. 622-6. [CrossRef]
- Szczot, M., et al., PIEZO2 mediates injury-induced tactile pain in mice and humans. Science Translational Medicine, 2018. 10(462). [CrossRef]
- Yamada, A., et al., Properties of Nav1.8(ChR2)-positive and Nav1.8(ChR2)-negative afferent mechanoreceptors in the hindpaw glabrous skin of mice. Mol Brain, 2023. 16(1): p. 27. [CrossRef]
- Gelbard, A., et al., An orthotopic murine model of sinonasal malignancy. Clin Cancer Res, 2008. 14(22): p. 7348-57. [CrossRef]
- Okutsu, Y., et al., Electrophysiological properties of maxillary trigeminal Abeta-afferent neurons of rats. Mol Pain, 2021. 17: p. 17448069211021271. [CrossRef]
- Ranade, S.S., et al., Piezo2 is the major transducer of mechanical forces for touch sensation in mice. Nature, 2014. 516(7529): p. 121-5. [CrossRef]
- Odem, M.A., et al., Isolated nociceptors reveal multiple specializations for generating irregular ongoing activity associated with ongoing pain. Pain, 2018. 159(11): p. 2347-2362. [CrossRef]
- Viatchenko-Karpinski, V., J. Ling, and J.G. Gu, Down-regulation of Kv4.3 channels and a-type K(+) currents in V2 trigeminal ganglion neurons of rats following oxaliplatin treatment. Mol Pain, 2018. 14: p. 1744806917750995. [CrossRef]
- Gee, M.D., et al., The relationship between axonal spike shape and functional modality in cutaneous C-fibres in the pig and rat. Neuroscience, 1999. 90(2): p. 509-18. [CrossRef]
- Fang, X., et al., Electrophysiological differences between nociceptive and non-nociceptive dorsal root ganglion neurones in the rat in vivo. J Physiol, 2005. 565(Pt 3): p. 927-43. [CrossRef]
- Korner, J. and A. Lampert, Functional subgroups of rat and human sensory neurons: a systematic review of electrophysiological properties. Pflugers Arch, 2022. 474(4): p. 367-385. [CrossRef]
- Ritter, D.M., et al., Modulation of Kv3.4 channel N-type inactivation by protein kinase C shapes the action potential in dorsal root ganglion neurons. J Physiol, 2012. 590(1): p. 145-61. [CrossRef]
- Ho, C. and M.E. O'Leary, Single-cell analysis of sodium channel expression in dorsal root ganglion neurons. Mol Cell Neurosci, 2011. 46(1): p. 159-66. [CrossRef]
- Bonnet, C., et al., Maladaptive activation of Nav1.9 channels by nitric oxide causes triptan-induced medication overuse headache. Nat Commun, 2019. 10(1): p. 4253. [CrossRef]
- Carvalho, T., et al., Perivascular Innervation in the Nasal Mucosa and Clinical Findings in Patients with Allergic Rhinitis and Idiopathic Rhinitis. Int Arch Otorhinolaryngol, 2023. 27(4): p. e723-e732. [CrossRef]
- Maggi, C.A., Tachykinins and calcitonin gene-related peptide (CGRP) as co-transmitters released from peripheral endings of sensory nerves. Prog Neurobiol, 1995. 45(1): p. 1-98. [CrossRef]
- Athnaiel, O., et al., The Role of Sex Hormones in Pain-Related Conditions. Int J Mol Sci, 2023. 24(3). [CrossRef]
- Greaves, E., et al., Elevated peritoneal expression and estrogen regulation of nociceptive ion channels in endometriosis. J Clin Endocrinol Metab, 2014. 99(9): p. E1738-43. [CrossRef]
- Murthy, S.E., et al., The mechanosensitive ion channel Piezo2 mediates sensitivity to mechanical pain in mice. Sci Transl Med, 2018. 10(462). [CrossRef]
- Yam, M.F., et al., General Pathways of Pain Sensation and the Major Neurotransmitters Involved in Pain Regulation. Int J Mol Sci, 2018. 19(8). [CrossRef]
- Cong, J., H. Lv, and Y. Xu, The role of nociceptive neurons in allergic rhinitis. Front Immunol, 2024. 15: p. 1430760. [CrossRef]
- Shusterman, D., Trigeminal Function in Sino-Nasal Health and Disease. Biomedicines, 2023. 11(7). [CrossRef]
- Huang, D., et al., Expression of the transient receptor potential channels TRPV1, TRPA1 and TRPM8 in mouse trigeminal primary afferent neurons innervating the dura. Mol Pain, 2012. 8: p. 66. [CrossRef]
| Category | Nasal skin [44] | Nasal mucosa [45,46] | Paranasal sinus mucosa (Findings of this study) | Dura [47] |
|---|---|---|---|---|
| Nerve types [44] |
Aβ fibers - abundant Heavily myelinated (fast touch/pressure) Aδ fibers - prominent Lightly myelinated (intermediate speed transmission of pain and temperature) C fibers - Unmyelinated – slow, dull pain, itch, and thermal sensations |
Aβ fibers: sparse and minimal function Aδ fibers: rapid pain/itch present but less dominant C fibers (polymodal): slow, burning pain - dominant |
Aβ fibers - none present Aδ fibers - few C fibers - dominant provides sensation of pressure |
Aβ fibers - sparse and minimal function Aδ fibers - present C fibers - dominant provides sensation of sharp pain |
| Nociceptors | A broad variety of encapsulated and unencapsulated sensory receptors (e.g., Meissner’s, Pacinian corpuscles for touch/pressure) Piezo2 - touch sensors |
Free endings mechanosensitive ion channels active nociceptors - present silent nociceptors - dominant Solitary chemosensory cells |
Free endings mechanosensitive ion channels active nociceptors - present silent nociceptors - dominant |
Free endings mechanosensitive ion channels active nociceptors - present silent nociceptors - dominant |
| Sodium ion channels | Transmit sensations of touch, temperature, and pain Nav1.7 - tetrodotoxin-sensitive (TTX-S), initiate action potential Nav1.8 - tetrodotoxin-resistant (TTX-R), enhance propagation – major role in pain sensation Nav1.9 - (TTX-R), sets firing threshold |
detect chemical irritants, temperature, and mechanical stimuli Nav1.7 - sets firing threshold, involved in inflammation and cough - high Nav1.8 - regulates airway defense mechanisms and airway hyperreactivity - lower Nav1.9 - regulating sensitivity - more prominent |
Nav1.7 - not present Nav1.8 - low concentration Nav1.9 - high concentration promotes hypersensitivity + persistent pain |
Nav1.7 - high expression Nav1.8 - variable Nav1.9 - prominent |
| Mechanosensitive ion channels |
Piezo2 - touch ASICs - present P2X3 - present |
Piezo2 - responds to tissue distortion, edema ASICs: acidic sensing - prominent P2X3: inflammation-related, on olfactory nerves - relevant, specialized | MA ion channels - high concentration Piezo2 (and likely TRPV4, ASICs, or other mechanoreceptors) respond to stretch, edema - senses pressure | MA ion channels - high concentration Piezo2 (and likely TRPV4, ASICs) responds to stretch/pulsation |
| TRP (Transient Receptor Potential) cation channels |
TRPV3, TRPV4 - prominent role in temperature sensation - high concentration TRPV1 - promotes or suppresses inflammation TRPA1 - mediates pain, itch, and cold sensation TRPM8 - little function -low concentration |
TRPV1 - responds to heat, capsaicin, irritants, inflammatory sensitization – more prominent TRPA1 - chemical irritants, oxidative stress – respond to allergens TRPM8 - cooling sensations (menthol sensitivity) |
TRPV1: responds to inflammatory heat signaling TRPA1: responds to oxidative/inflammatory stress - highly expressed TRPM8 - sparse |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




