Submitted:
26 February 2026
Posted:
28 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
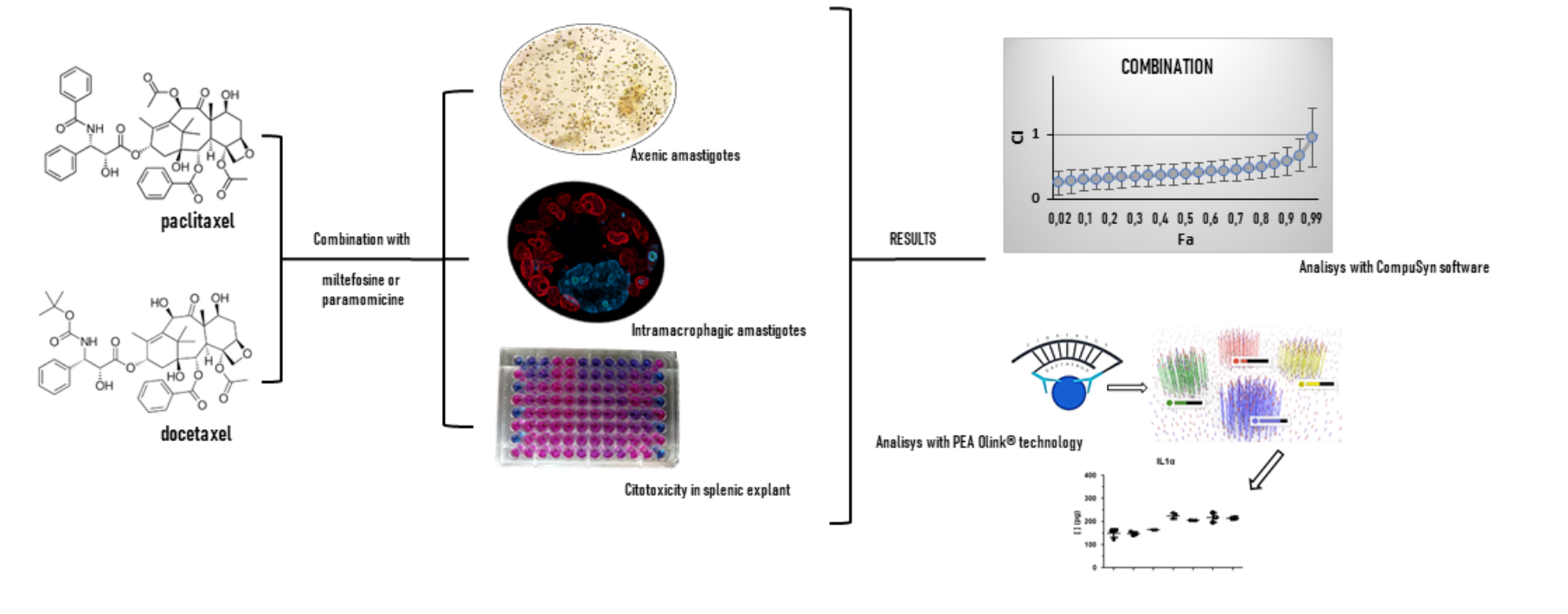
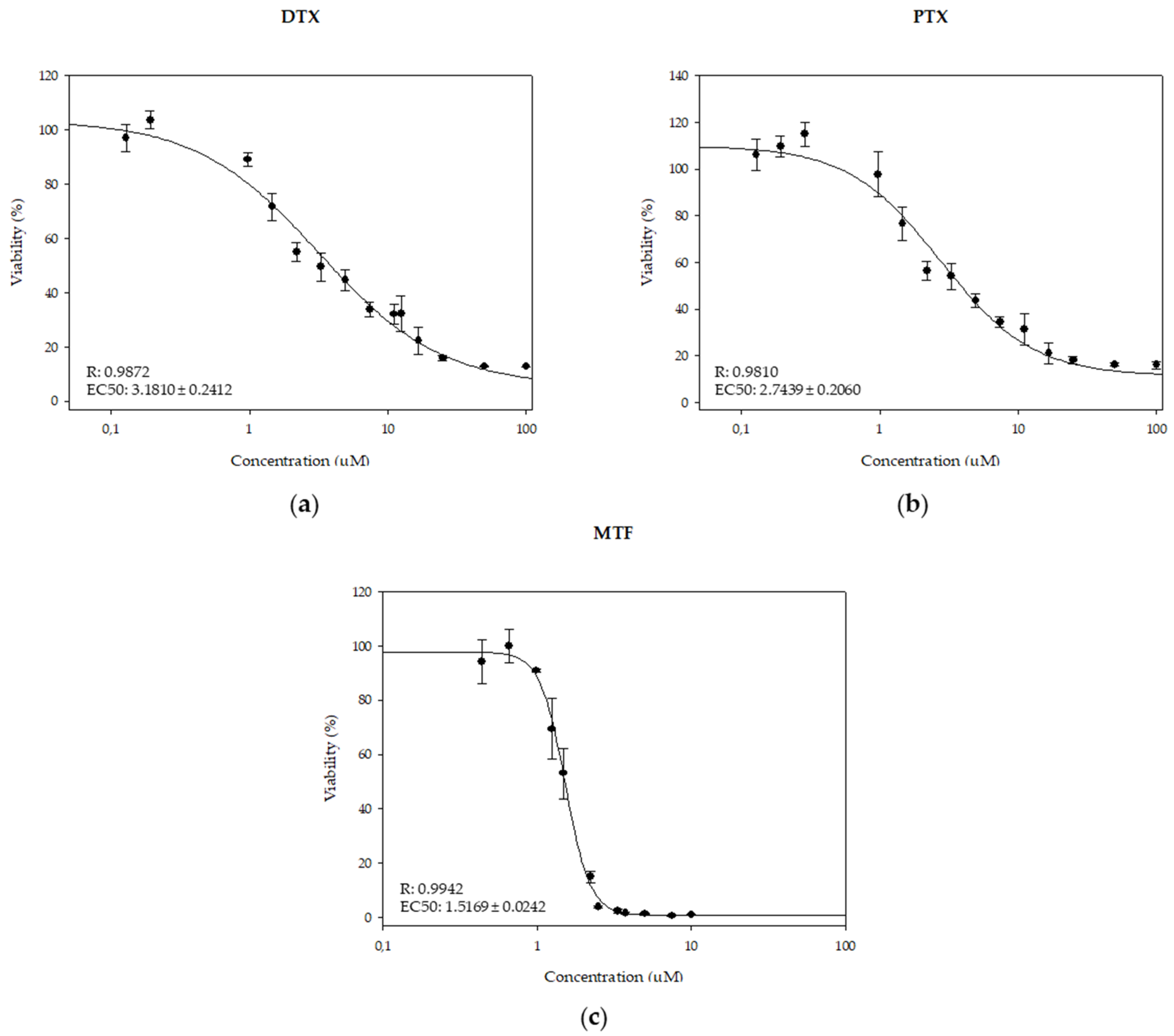
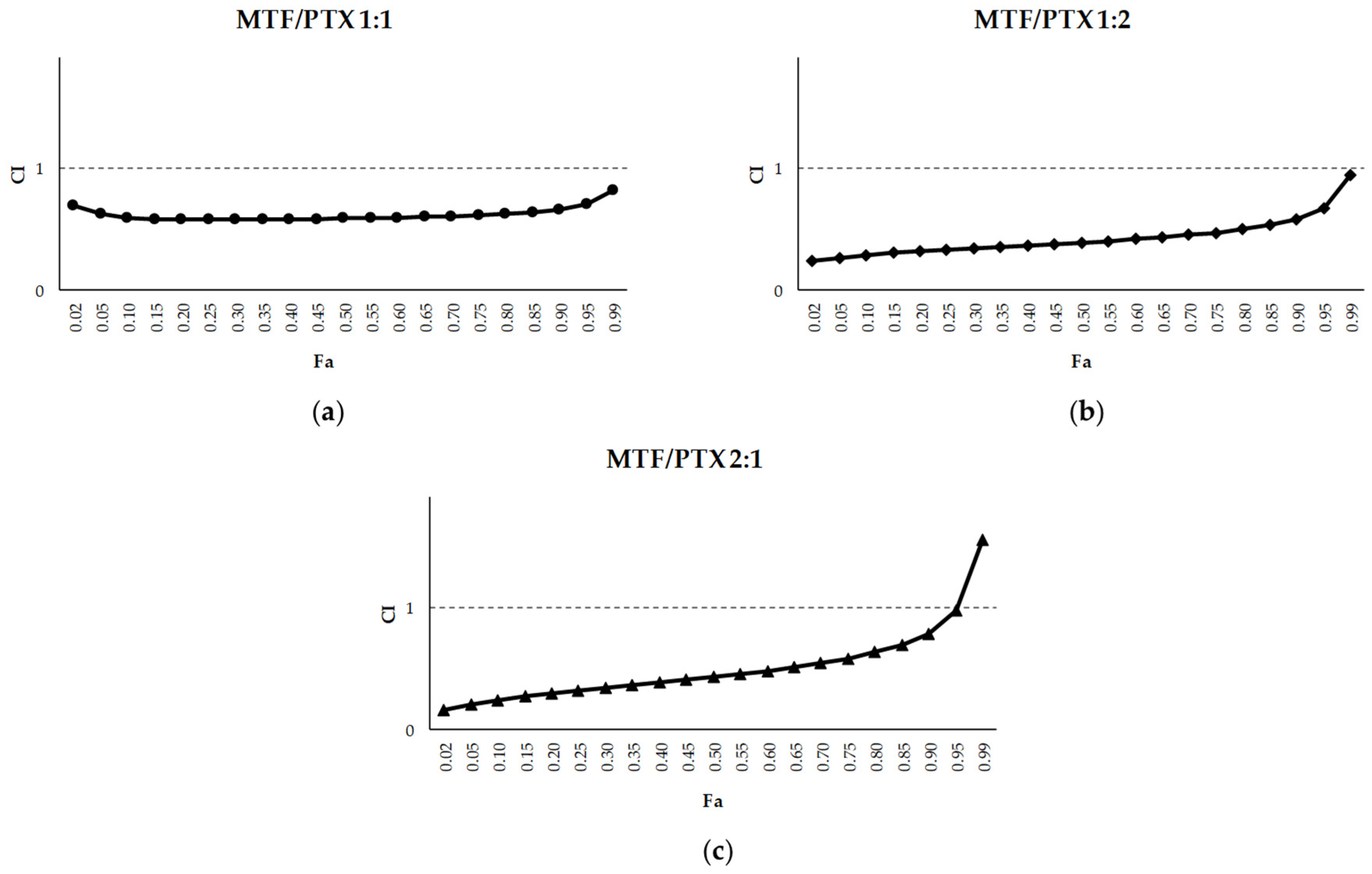
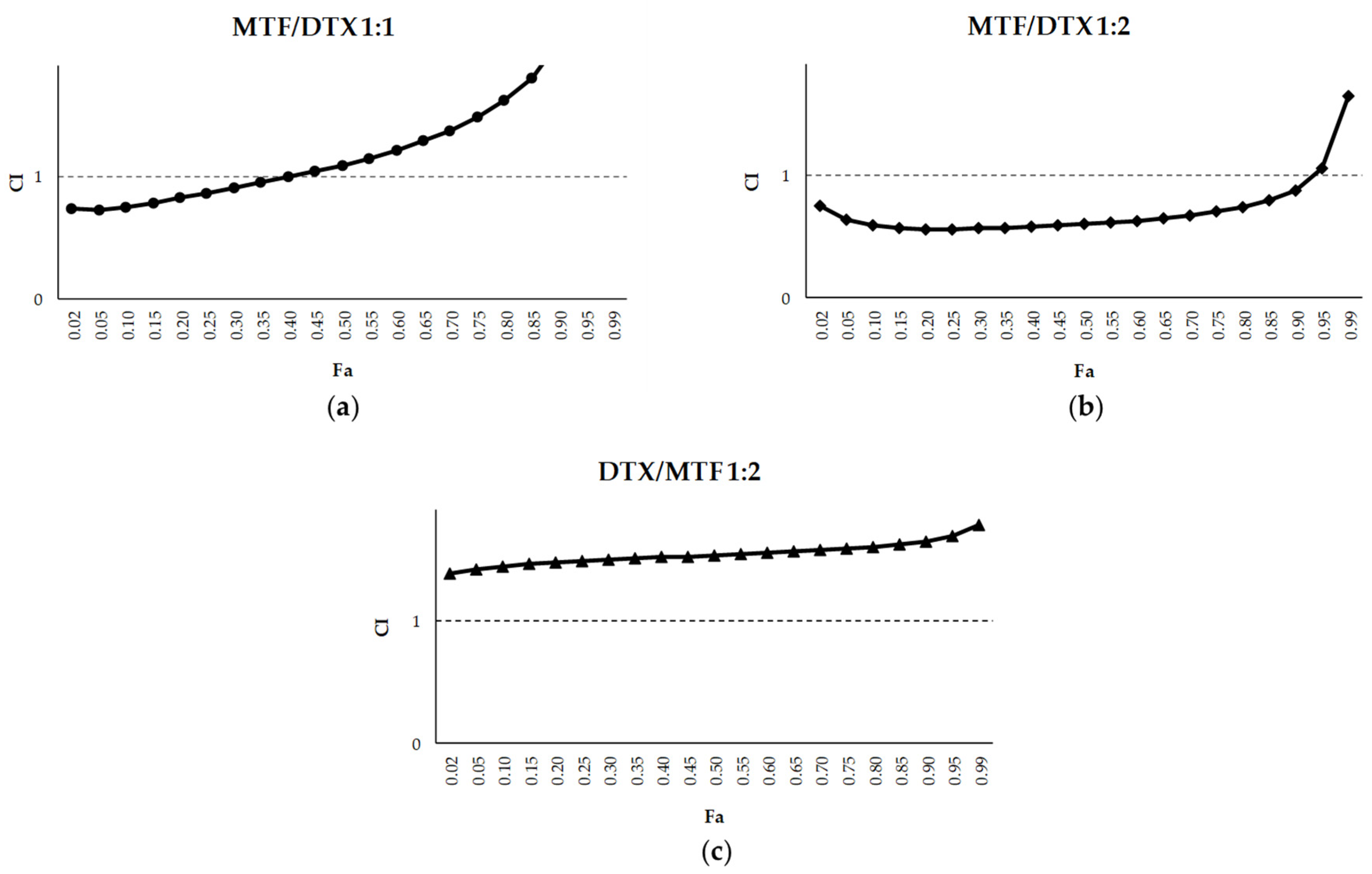
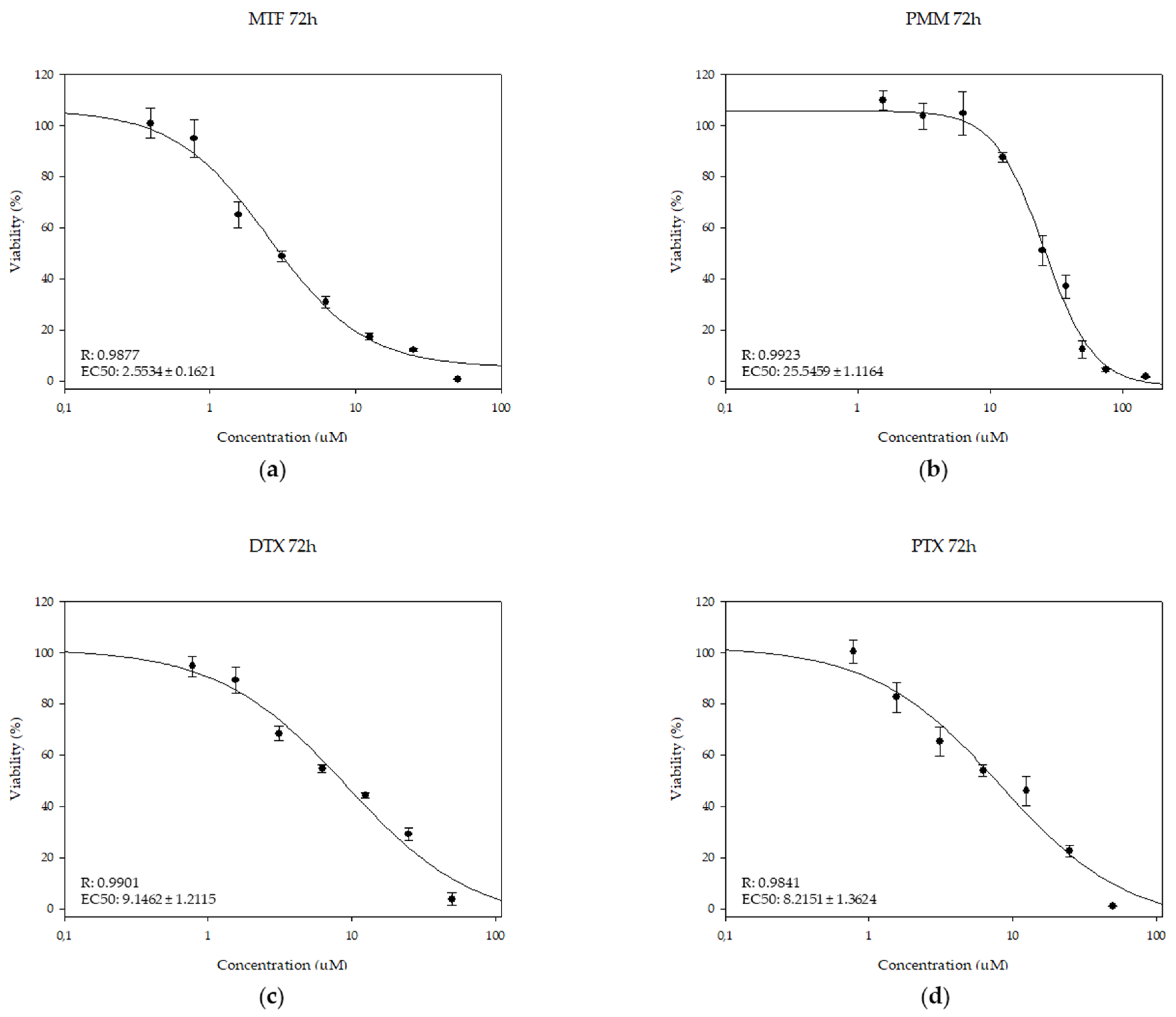
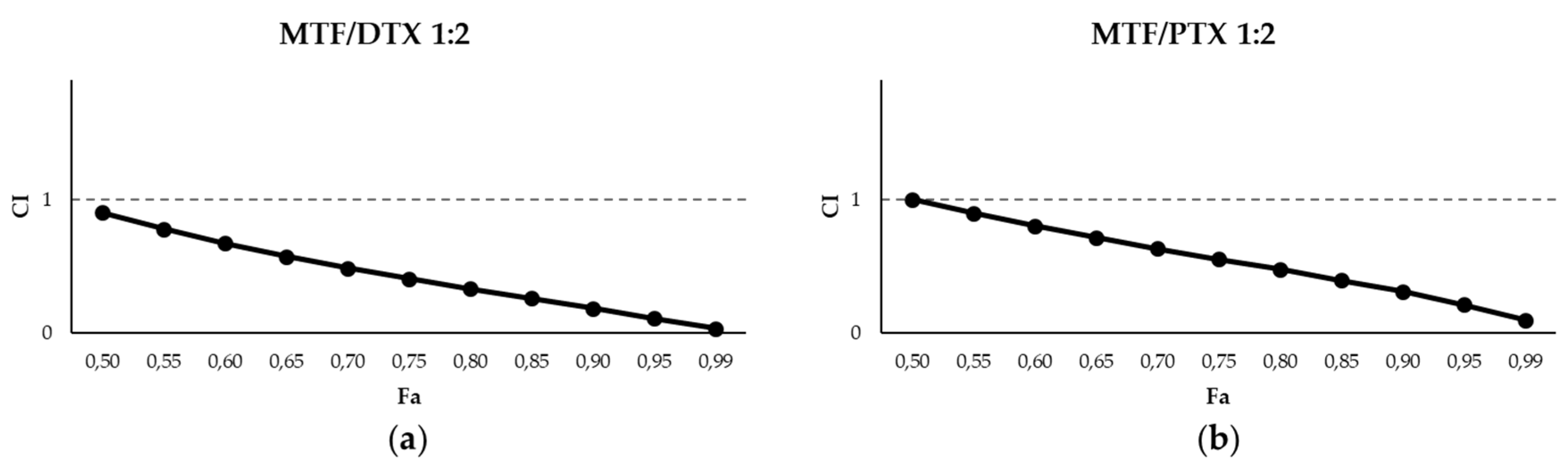
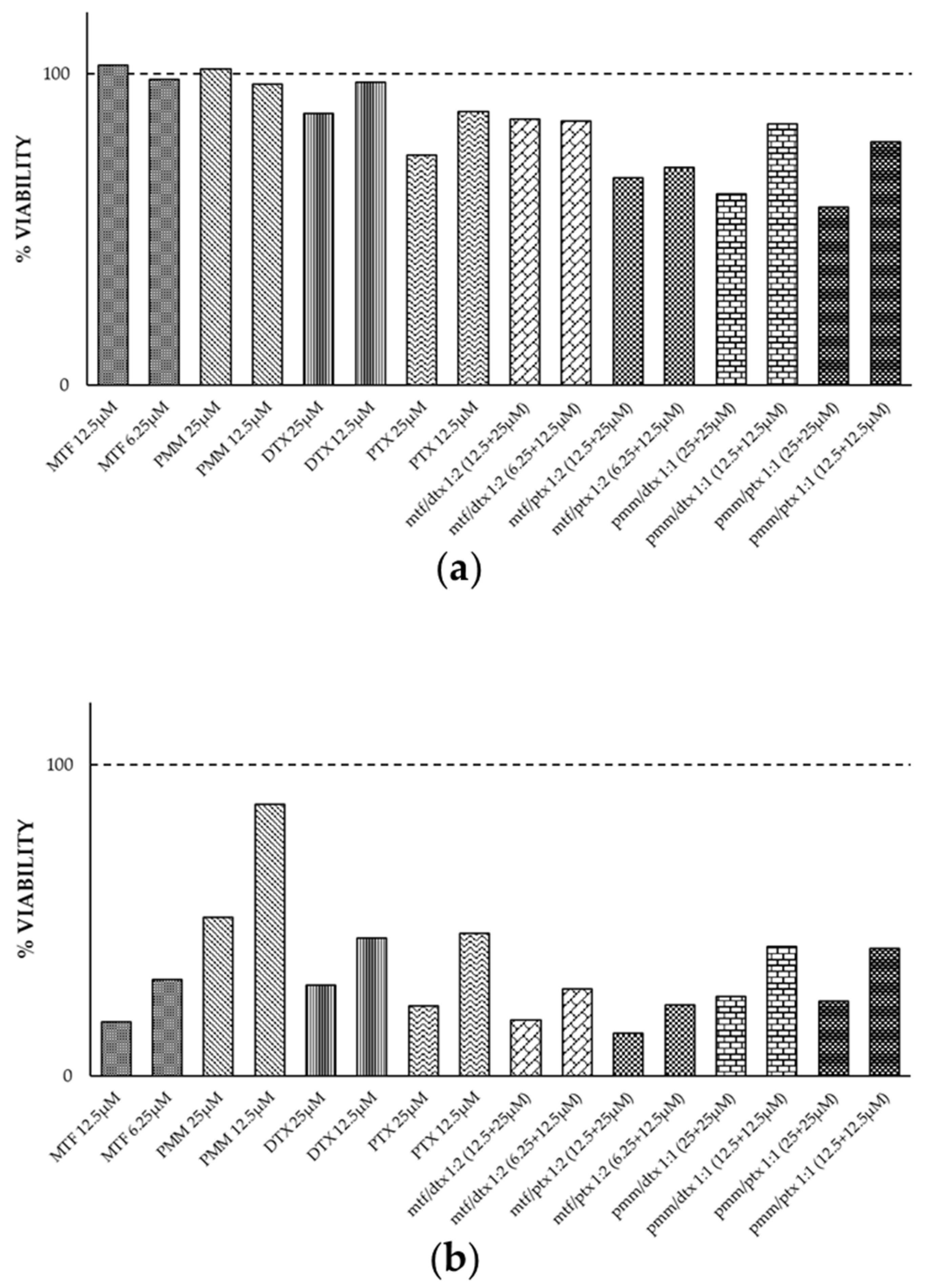
2.1. Leishmanicidal Effect of PTX and DTX in Combination with MTF
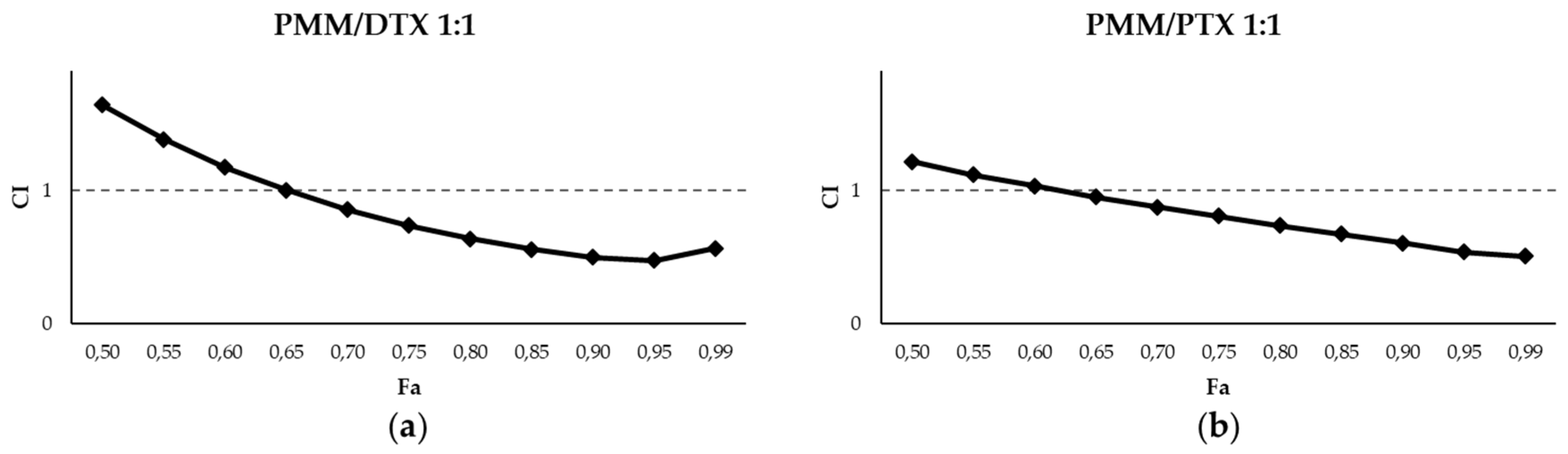
2.2. Leishmanicidal Effect of PTX and DTX in Combination with PMM
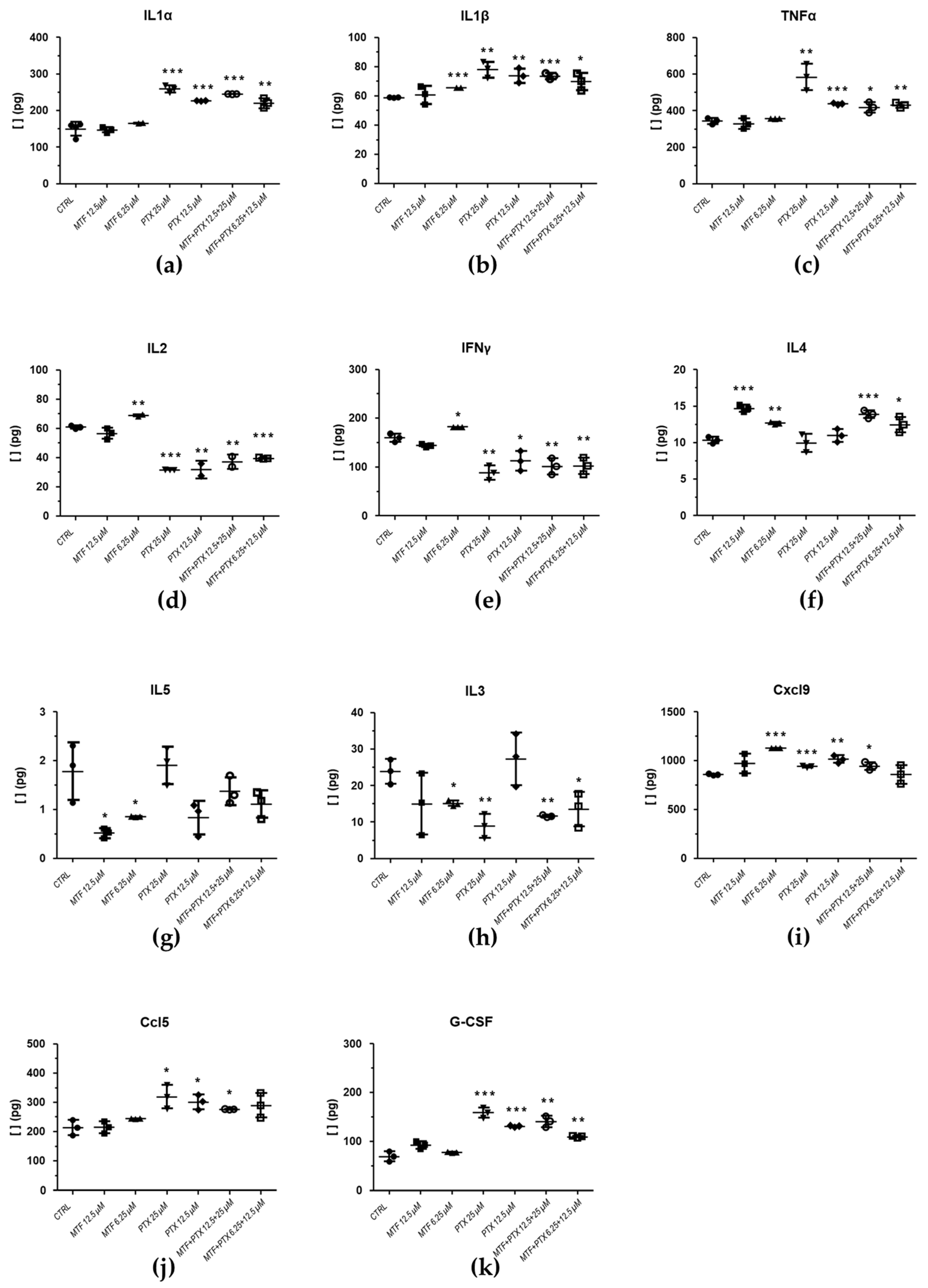
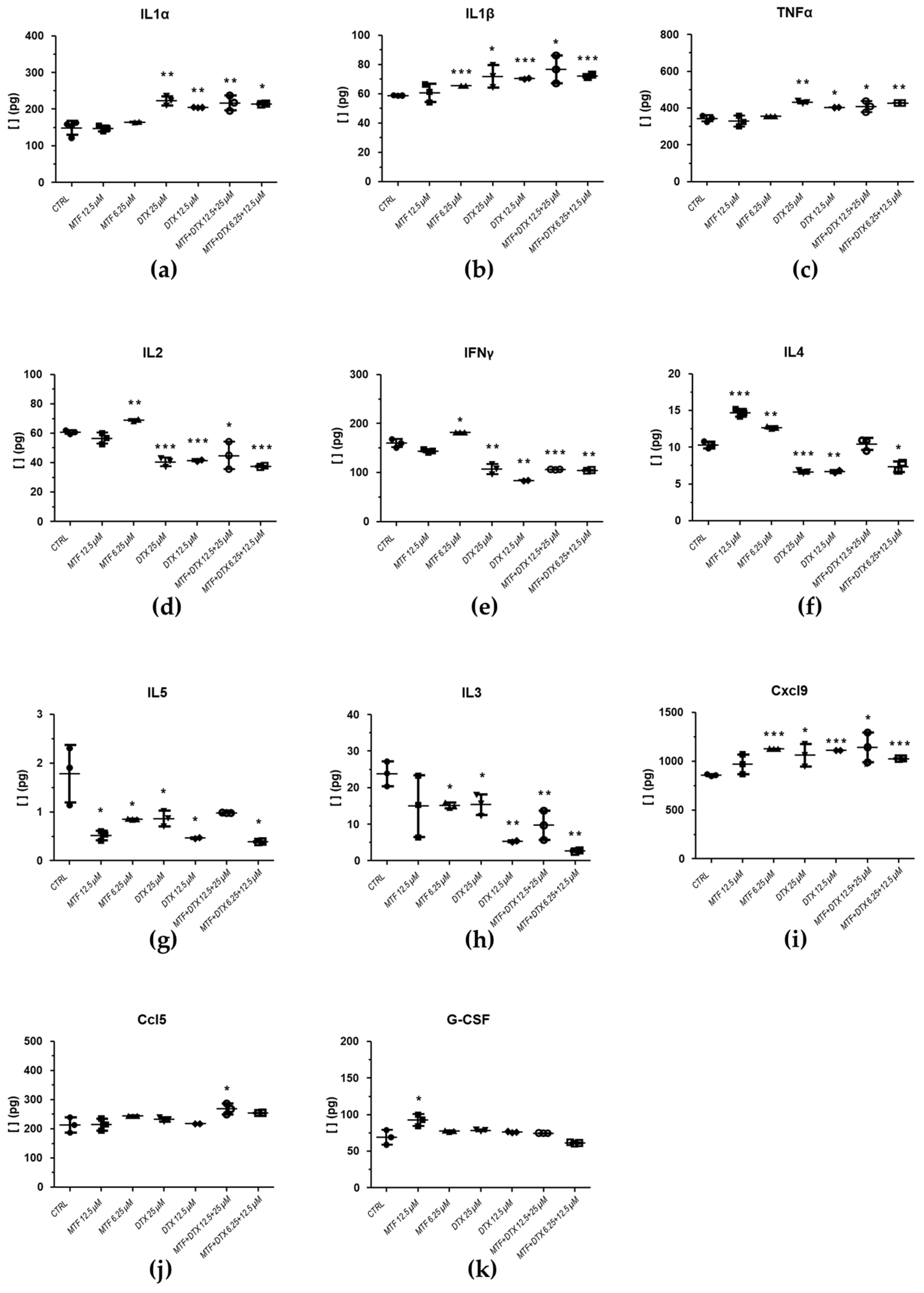
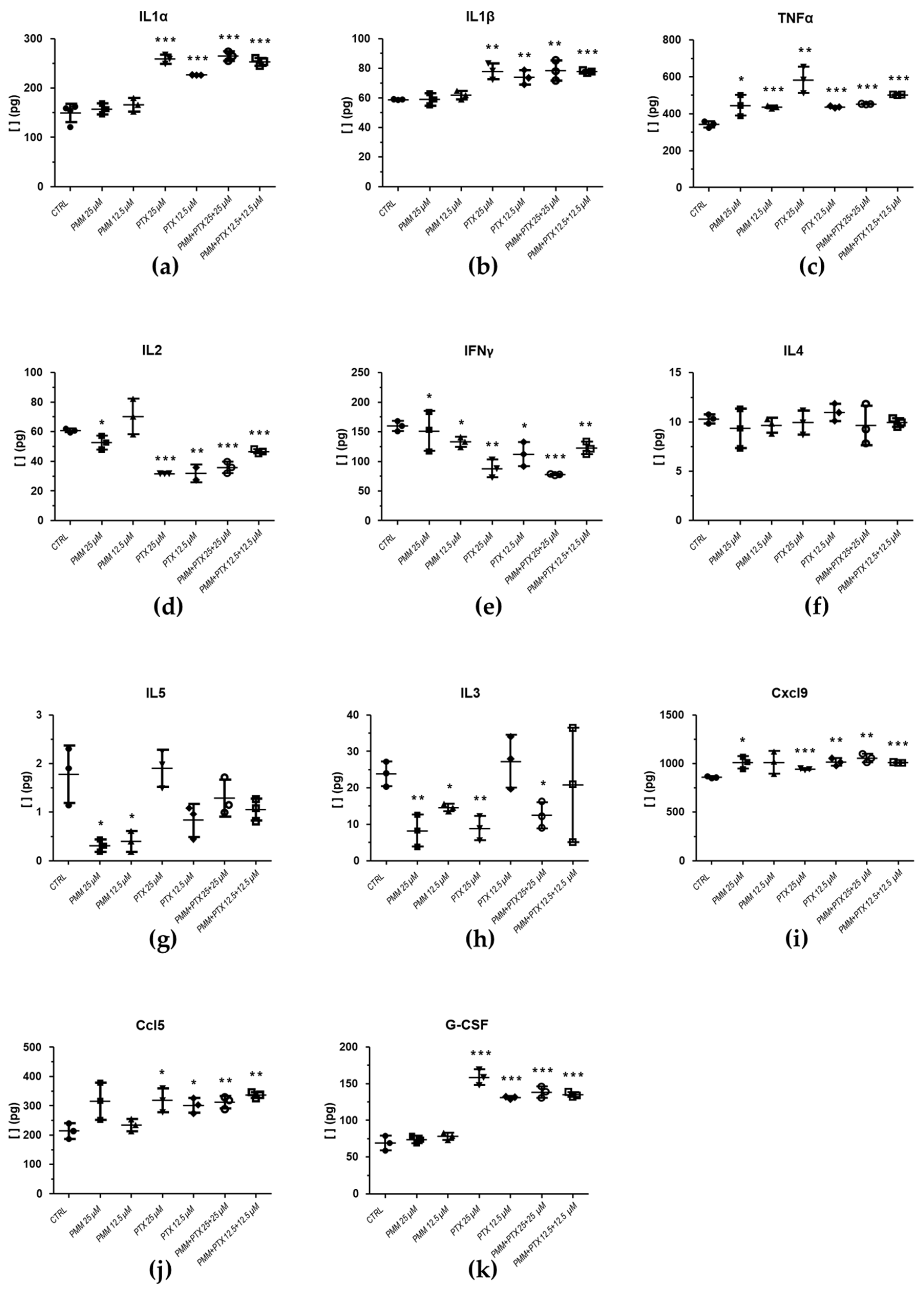
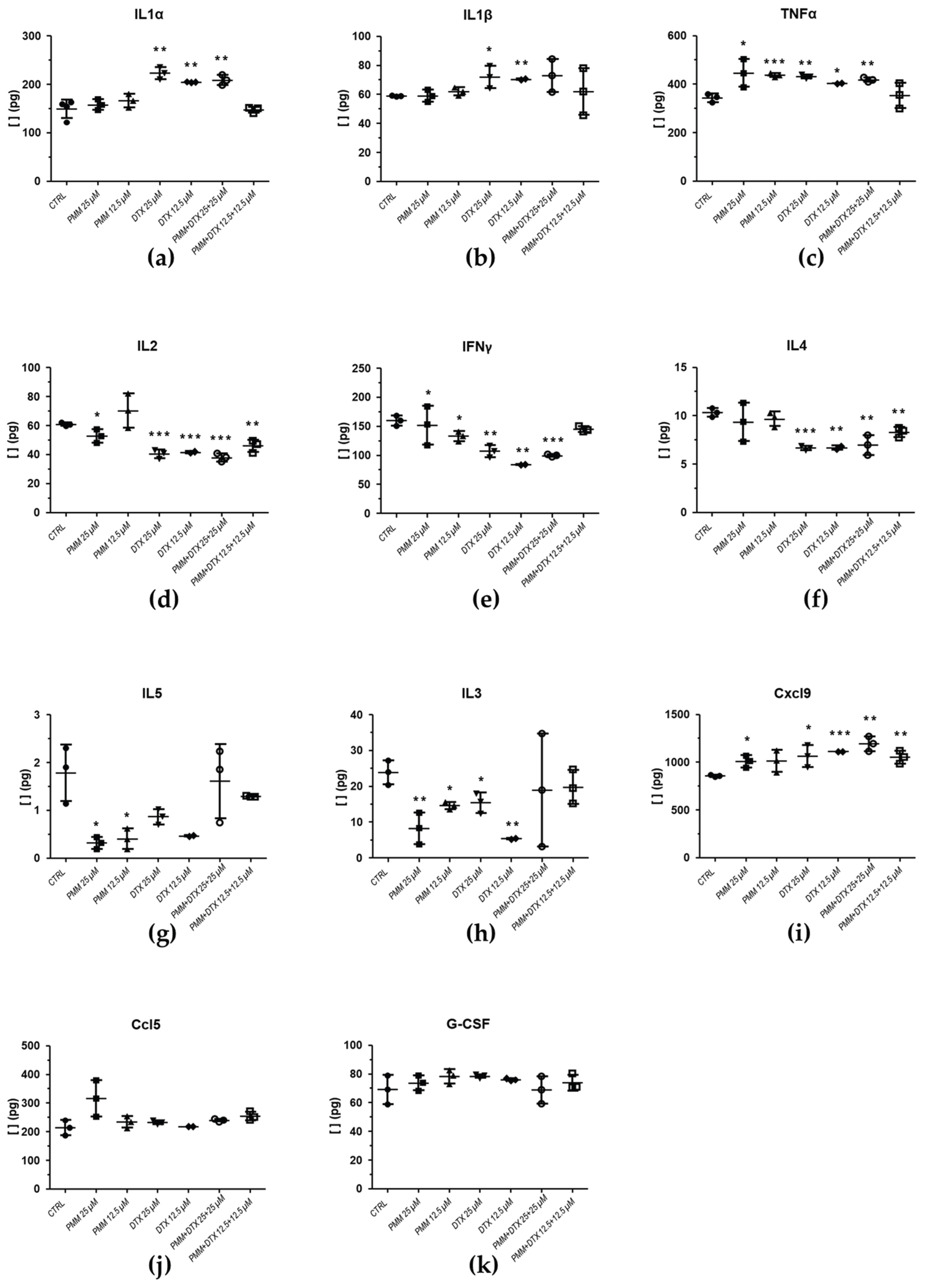
2.3. Cytokine Evaluation
2.3.1. Effect of PTX and DTX in Combination with MTF
2.3.2. Effect of PTX and DTX in Combination with PMM
3. Discussion
4. Materials and Methods
4.1. Drugs
4.2. Experimental Animals and Ethical Statement
4.3. Parasites
4.4. Experimental Infections and Set Up of Primary Cultures
4.5. Axenic and Intramacrophagic Amastigotes Viability Assays
4.6. Cell Cytotoxicity
4.7. Cytokine Analysis
|
Interleukin superfamily Cytokines Pro-interleukin-16 (Il16) O54824 C-C motif chemokine 22 (Ccl22) O88430 Interleukin-1 alpha (Il1a) P01582 Granulocyte-macrophage colony-stimulating factor (Csf2) P01587 Interleukin-4 (Il4) P07750 Macrophage colony-stimulating factor 1 (Csf1) P07141 Interleukin-6 (Il6) P08505 Granulocyte colony-stimulating factor (Csf3) P09920 Interleukin-1 beta (Il1b) P10749 C-C motif chemokine 2 (Ccl2) P10148 Interleukin-9 (Il9) P15247 C-C motif chemokine 4 (Ccl4) P14097 Interferon lambda-2 (Ifnl2) Q4VK74 C-C motif chemokine 5 (Ccl5) P30882 Interleukin-31 (Il31) Q6EAL8 Eotaxin (Ccl11) P48298 Interleukin-33 (Il33) Q8BVZ5 C-C motif chemokine 12 (Ccl12) Q62401 Interleukin-27 subunit alpha (Il27) Q8K3I6 C-C motif chemokine 17 (Ccl17) Q9WUZ6 Interleukin-21 (Il21) Q9ES17 Interleukin-3 (Il3) P01586 Other Interleukin-2 (Il2) P04351 Fibroblast growth factor 21 (Fgf21) Q9JJN1 Interleukin-5 (Il5) P04401 Cytotoxic T-lymphocyte protein 4 (Ctla4) P09793 Interleukin-7 (Il7) P10168 Programmed cell death 1 ligand 1 (Cd274) Q9EP73 Interleukin-10 (Il10) P18893 Tumor necrosis factor (Tnf) P06804 Interleukin-12 (Il12a, Il12b) P43431, P43432 Interferon alpha-2 (Ifna2) P01573 Interleukin-17A (Il17a) Q62386 Interferon gamma (Ifng) P01580 Interleukin-17F (Il17f) Q7TNI7 Hepatocyte growth factor (Hgf) Q08048 Interleukin-22 (Il22) Q9JJY9 Programmed cell death 1 ligand 2 (Pdcd1lg2) Q9WUL5 Chemokines C-X-C motif chemokine 2 (Cxcl2) P10889 Growth-regulated alpha protein (Cxcl1) P12850 C-X-C motif chemokine 9 (Cxcl9) P18340 Stromal cell-derived factor 1 (Cxcl12) P40224 C-X-C motif chemokine 11 (Cxcl11) Q9JHH5 |
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AMB | amphotericin B |
| CI | combination index |
| CL | cutaneous leishmaniasis |
| Dm | median-effect dose producing 50% inhibition |
| DRI | dose reduction index |
| DTX | docetaxel |
| fa | fraction affected |
| L-AMB | liposomal encapsulated amphotericin B |
| m | coefficient signifying the shape of the dose-response curve |
| MCL | mucocutaneous leishmaniasis |
| MTF | miltefosine |
| PKDL | post-kala-azar dermal leishmaniasis |
| PMM | paromomycin |
| PTX | paclitaxel |
| r | correlation coefficient |
| VL | visceral leishmaniasis |
References
- Pareyn, M; Alves, F; Burza, S; et al. Leishmaniasis. Nat Rev Dis Primers 2025, 11, 81. [Google Scholar] [CrossRef]
- Paz, S. The potential of climatic suitability indicator for Leishmania transmission modelling in Europe: insights and suggested directions. Lancet Reg Health Eur. 2024, 43, 100995. [Google Scholar] [CrossRef]
- Surveillance, prevention and control of leishmaniases in the European Union and its neighbouring countries. Available online: https://www.ecdc.europa.eu/sites/default/files/documents/leishmaniasis-surveillance-eu.pdf (accessed on 24 february 2026).
- Leishmaniasis. Available online: https://www.who.int/news-room/fact-sheets/detail/leishmaniasis (accessed on 24 february 2026).
- Majoor, A; Michel, G; Marty, P; Boyer, L; Pomares, C. Leishmaniases: Strategies in treatment development. Leishmanioses: stratégies de développement de traitements. Parasite. 2025, 32, 18. [Google Scholar] [CrossRef]
- Zhang, H; Yan, R; Liu, Y; et al. Progress in antileishmanial drugs: Mechanisms, challenges, and prospects. PLoS Negl Trop Dis. 2025, 19, e0012735. [Google Scholar] [CrossRef]
- Hefnawy, A; Berg, M; Dujardin, JC; De Muylder, G. Exploiting Knowledge on Leishmania Drug Resistance to Support the Quest for New Drugs. Trends Parasitol. 2017, 33, 162–174. [Google Scholar] [CrossRef]
- Hefnawy, A; Cantizani, J; Peña, I; et al. Importance of secondary screening with clinical isolates for anti-leishmania drug discovery. Sci Rep. 2018, 8, 11765. [Google Scholar] [CrossRef] [PubMed]
- Khanra, S; Sarraf, NR; Das, AK; Roy, S; Manna, M. Miltefosine Resistant Field Isolate From Indian Kala-Azar Patient Shows Similar Phenotype in Experimental Infection. Sci Rep. 2017, 7, 10330. [Google Scholar] [CrossRef] [PubMed]
- Carrasco-Martin, M; Martí-Carreras, J; Gómez-Ponce, M; et al. Insights on genomic profiles of drug resistance and virulence in a cohort of Leishmania infantum isolates from the Mediterranean area. Parasit Vectors. 2025, 19, 28. [Google Scholar] [CrossRef]
- Mougneau, E; Bihl, F; Glaichenhaus, N. Cell biology and immunology of Leishmania. Immunol Rev. 2011, 240, 286–296. [Google Scholar] [CrossRef] [PubMed]
- Mirzaei, A; Maleki, M; Masoumi, E; Maspi, N. A historical review of the role of cytokines involved in leishmaniasis. Cytokine 2021, 145, 155297. [Google Scholar] [CrossRef]
- Serafim, TD; Coutinho-Abreu, IV; Dey, R; et al. Leishmaniasis: the act of transmission. Trends Parasitol. 2021, 37, 976–987. [Google Scholar] [CrossRef] [PubMed]
- Alves, NO; Oshiro, JA; Silva, YC; et al. VL-HIV co-infection with Leishmania containing skin lesions resembling para-kala-azar dermal leishmaniasis. PLoS Negl Trop Dis. 2024, 18, e0012438. [Google Scholar] [CrossRef] [PubMed]
- Ibarra-Meneses, AV; Moreno, J; Carrillo, E. New Strategies and Biomarkers for the Control of Visceral Leishmaniasis. Trends Parasitol. 2020, 36, 29–38. [Google Scholar] [CrossRef]
- Iqbal Yatoo, M; Hamid, Z; Rather, I; et al. Immunotherapies and immunomodulatory approaches in clinical trials - a mini review. Hum Vaccin Immunother. 2021, 17, 1897–1909. [Google Scholar] [CrossRef]
- Singh, VK; Tiwari, R; Rajneesh; et al. Advancing Treatment for Leishmaniasis: From Overcoming Challenges to Embracing Therapeutic Innovations. ACS Infect Dis. 2025, 11, 47–68. [Google Scholar] [CrossRef]
- Blackwell, JM; Fakiola, M; Castellucci, LC. Human genetics of leishmania infections. Hum Genet. 2020, 139, 813–819. [Google Scholar] [CrossRef]
- Oliveira, EG; Lanza, JS; Cojean, S; et al. Re-sensitization of antimony-resistant Leishmania by highly potent SbV-porphyrin through the involvement of ERG6-coding gene. Biomed Pharmacother. 2025, 187, 118059. [Google Scholar] [CrossRef]
- Uliana, SRB; Trinconi, CT; Coelho, AC. Chemotherapy of leishmaniasis: present challenges. Parasitology. 2018, 145, 464–480. [Google Scholar] [CrossRef]
- van Griensven, J; Dorlo, TP; Diro, E; Costa, C; Burza, S. The status of combination therapy for visceral leishmaniasis: an updated review. Lancet Infect Dis. 2024, 24, e36–e46. [Google Scholar] [CrossRef]
- Pushpakom, S; Iorio, F; Eyers, PA; et al. Drug repurposing: progress, challenges and recommendations. Nat Rev Drug Discov. 2019, 18, 41–58. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Rubio, C; Larrea, E; Peña Guerrero, J; et al. Leishmanicidal Activity of Isoselenocyanate Derivatives. Antimicrob Agents Chemother. 2019, 63, e00904-18. [Google Scholar] [CrossRef]
- El-Dirany, R; Fernández-Rubio, C; Peña-Guerrero, J; et al. Repurposing the Antibacterial Agents Peptide 19-4LF and Peptide 19-2.5 for Treatment of Cutaneous Leishmaniasis. Pharmaceutics. 2022, 14, 2528. [Google Scholar] [CrossRef]
- Melcon-Fernandez, E; Galli, G; García-Estrada, C; Balaña-Fouce, R; Reguera, RM; Pérez-Pertejo, Y. Miltefosine and Nifuratel Combination: A Promising Therapy for the Treatment of Leishmania donovani Visceral Leishmaniasis. Int J Mol Sci. 2023, 24, 1635. [Google Scholar] [CrossRef]
- Melcón-Fernández, E; Galli, G; Balaña-Fouce, R; et al. In Vitro and Ex Vivo Synergistic Effect of Pyrvinium Pamoate Combined with Miltefosine and Paromomycin against Leishmania. Trop Med Infect Dis. 2024, 9, 30. [Google Scholar] [CrossRef]
- Domínguez-Asenjo, B; Gutiérrez-Corbo, C; Álvarez-Bardón, M; Pérez-Pertejo, Y; Balaña-Fouce, R; Reguera, RM. Ex Vivo Phenotypic Screening of Two Small Repurposing Drug Collections Identifies Nifuratel as a Potential New Treatment against Visceral and Cutaneous Leishmaniasis. ACS Infect Dis. 2021, 7, 2390–2401. [Google Scholar] [CrossRef]
- Doherty, TM; Sher, A; Vogel, SN. Paclitaxel (Taxol)-induced killing of Leishmania major in murine macrophages. Infect Immun. 1998, 66, 4553–4556. [Google Scholar] [CrossRef] [PubMed]
- Chou, TC; Talalay, P. Quantitative analysis of dose-effect relationships: the combined effects of multiple drugs or enzyme inhibitors. Adv Enzyme Regul. 1984, 22, 27–55. [Google Scholar] [CrossRef] [PubMed]
- Chou, TC. Theoretical basis, experimental design, and computerized simulation of synergism and antagonism in drug combination studies. Pharmacol Rev. 2006, 58, 621–681. [Google Scholar] [CrossRef]
- Almeida, FS; Vanderley, SER; Comberlang, FC; et al. Leishmaniasis: Immune Cells Crosstalk in Macrophage Polarization. Trop Med Infect Dis. 2023, 8, 276. [Google Scholar] [CrossRef] [PubMed]
- Bhor, R; Rafati, S; Pai, K. Cytokine saga in visceral leishmaniasis. Cytokine. 2021, 147, 155322. [Google Scholar] [CrossRef]
- Samant, M; Sahu, U; Pandey, SC; Khare, P. Role of Cytokines in Experimental and Human Visceral Leishmaniasis. Front Cell Infect Microbiol. 2021, 11, 624009. [Google Scholar] [CrossRef] [PubMed]
- Ramer-Tait, AE; Petersen, CA; Jones, DE. IL-2 limits IL-12 enhanced lymphocyte proliferation during Leishmania amazonensis infection. Cell Immunol. 2011, 270, 32–39. [Google Scholar] [CrossRef]
- Upadhyay, S; Kumar, S; Singh, VK; et al. Chemokines Signature and T Cell Dynamics in Leishmaniasis: Molecular insight and therapeutic application. Expert Rev Mol Med. 2024, 27, 1–55. [Google Scholar] [CrossRef]
- Saha, B; Saini, A; Germond, R; Perrin, PJ; Harlan, DM; Davis, TA. Susceptibility or resistance to Leishmania infection is dictated by the macrophages evolved under the influence of IL-3 or GM-CSF. Eur J Immunol. 1999, 29, 2319–2329. [Google Scholar] [CrossRef]
- Martin, KR; Wong, HL; Witko-Sarsat, V; Wicks, IP. G-CSF - A double edge sword in neutrophil mediated immunity. Semin Immunol. 2021, 54, 101516. [Google Scholar] [CrossRef]
- Havens, CG; Bryant, N; Asher, L; et al. Cellular effects of leishmanial tubulin inhibitors on L. donovani. Mol Biochem Parasitol. 2000, 110, 223–236. [Google Scholar] [CrossRef]
- Christensen, SB. Drugs That Changed Society: Microtubule-Targeting Agents Belonging to Taxanoids, Macrolides and Non-Ribosomal Peptides. Molecules. 2022, 27, 5648. [Google Scholar] [CrossRef]
- Marupudi, NI; Han, JE; Li, KW; Renard, VM; Tyler, BM; Brem, H. Paclitaxel: a review of adverse toxicities and novel delivery strategies. Expert Opin Drug Saf. 2007, 6, 609–621. [Google Scholar] [CrossRef] [PubMed]
- Abdu, SA; Caiyun, H; Asaad, WA; et al. Comparative efficacy and safety of cabazitaxel versus other taxanes: a systematic review and meta-analysis. Syst Rev. 2025, 14, 210. [Google Scholar] [CrossRef] [PubMed]
- Mills, CD. M1 and M2 Macrophages: Oracles of Health and Disease. Crit Rev Immunol. 2012, 32, 463–488. [Google Scholar] [CrossRef]
- Dayakar, A; Chandrasekaran, S; Kuchipudi, SV; Kalangi, SK. Cytokines: Key Determinants of Resistance or Disease Progression in Visceral Leishmaniasis: Opportunities for Novel Diagnostics and Immunotherapy. Front Immunol. 2019, 10, 670. [Google Scholar] [CrossRef]
- Vacas, A; Fernández-Rubio, C; Larrea, E; Peña-Guerrero, J; Nguewa, PA. LmjF.22.0810 from Leishmania major Modulates the Th2-Type Immune Response and Is Involved in Leishmaniasis Outcome. Biomedicines. 2020, 8, 452. [Google Scholar] [CrossRef] [PubMed]
- Costa-da-Silva, AC; Nascimento, DO; Ferreira, JRM; et al. Immune Responses in Leishmaniasis: An Overview. Trop Med Infect Dis. 2022, 7, 54. [Google Scholar] [CrossRef]
- Ayala, A; Llanes, A; Lleonart, R; Restrepo, CM. Advances in Leishmania Vaccines: Current Development and Future Prospects. Pathogens. 2024, 13, 812. [Google Scholar] [CrossRef]
- Murray, HW; Luster, AD; Zheng, H; Ma, X. Gamma Interferon-Regulated Chemokines in Leishmania donovani Infection in the Liver. Infect Immun. 2016, 85, e00824-16. [Google Scholar] [CrossRef] [PubMed]
- de Araújo, FF; Costa-Silva, MF; Pereira, AAS; et al. Chemokines in Leishmaniasis: Map of cell movements highlights the landscape of infection and pathogenesis. Cytokine. 2021, 147, 155339. [Google Scholar] [CrossRef]
- Santiago, HC; Oliveira, CF; Santiago, L; et al. Involvement of the chemokine RANTES (CCL5) in resistance to experimental infection with Leishmania major. Infect Immun. 2004, 72, 4918–4923. [Google Scholar] [CrossRef]
- Rêgo, FD; Fradico, JRB; Teixeira-Carvalho, A; Gontijo, CMF. Molecular variants of Leishmania (Viannia) braziliensis trigger distinct patterns of cytokines and chemokines expression in golden hamster. Mol Immunol. 2019, 106, 36–45. [Google Scholar] [CrossRef]
- Menezes-Souza, D; Guerra-Sá, R; Carneiro, CM; et al. Higher expression of CCL2, CCL4, CCL5, CCL21, and CXCL8 chemokines in the skin associated with parasite density in canine visceral leishmaniasis. PLoS Negl Trop Dis. 2012, 6, e1566. [Google Scholar] [CrossRef] [PubMed]
- Bodas, M; Jain, N; Awasthi, A; et al. Inhibition of IL-2 induced IL-10 production as a principle of phase-specific immunotherapy. J Immunol. 2006, 177, 4636–4643. [Google Scholar] [CrossRef]
- Pérez-Cabezas, B; Cecílio, P; Gaspar, TB; Gärtner, F; Vasconcellos, R; Cordeiro-da-Silva, A. Understanding Resistance vs. Susceptibility in Visceral Leishmaniasis Using Mouse Models of Leishmania infantum Infection. Front Cell Infect Microbiol. 2019, 9, 30. [Google Scholar] [CrossRef] [PubMed]
- Mulè, A; Crosato, V; Kuhns, DB; et al. Visceral Leishmaniasis in Immunocompetent Hosts in Brescia: A Case Series and Analysis of Cytokine Cascade. Microorganisms. 2024, 12, 394. [Google Scholar] [CrossRef]
- Rutella, S; Bonanno, G; Pierelli, L; et al. Granulocyte colony-stimulating factor promotes the generation of regulatory DC through induction of IL-10 and IFN-alpha. Eur J Immunol. 2004, 34, 1291–1302. [Google Scholar] [CrossRef]
- Suprien, C; Guimarães, LH; de Carvalho, LP; Machado, PRL. Pentavalent Antimony Associated with G-CSF in the Treatment of Cutaneous Leishmaniasis Caused by Leishmania (Viannia) braziliensis. Pathogens. 2024, 13, 301. [Google Scholar] [CrossRef]
- Murray, HW; Cervia, JS; Hariprashad, J; Taylor, AP; Stoeckle, MY; Hockman, H. Effect of granulocyte-macrophage colony-stimulating factor in experimental visceral leishmaniasis. J Clin Invest. 1995, 95, 1183–1192. [Google Scholar] [CrossRef] [PubMed]
- Zaks-Zilberman, M; Zaks, TZ; Vogel, SN. Induction of proinflammatory and chemokine genes by lipopolysaccharide and paclitaxel (Taxol) in murine and human breast cancer cell lines. Cytokine. 2001, 15, 156–165. [Google Scholar] [CrossRef] [PubMed]
- Calvo-Álvarez, E; Stamatakis, K; Punzón, C; et al. Infrared fluorescent imaging as a potent tool for in vitro, ex vivo and in vivo models of visceral leishmaniasis. PLoS Negl Trop Dis. 2015, 9, e0003666. [Google Scholar] [CrossRef]
| DRI Values at following effect levels | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| 25% | 50% | 75% | 95% | ||||||
| Drug | r | MTF | Taxane | MTF | Taxane | MTF | Taxane | MTF | Taxane |
| MTF+DTX 1:1 | 0.84 | 1.74 | 3.48 | 1.16 | 4.44 | 0.77 | 5.66 | 0.39 | 8.50 |
| MTF+DTX 1:2 | 0.98 | 3.54 | 3.55 | 2.54 | 4.87 | 1.82 | 6.67 | 1.04 | 11.33 |
| MTF+DTX 2:1 | 0.74 | 0.91 | 3.64 | 0.75 | 5.73 | 0.61 | 9.02 | 0.44 | 19.35 |
| MTF+PTX 1:1 | 0.91 | 2.29 | 7.04 | 2.04 | 10.20 | 1.82 | 14.79 | 1.50 | 27.59 |
| MTF+PTX 1:2 | 0.92 | 4.14 | 11.10 | 3.22 | 12.33 | 2.50 | 13.69 | 1.64 | 16.33 |
| MTF+PTX 2:1 | 0.92 | 3.82 | 23.47 | 1.92 | 19.16 | 0.96 | 15.64 | 0.30 | 11.12 |
| DRI Values at following effect levels | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| 25% | 50% | 75% | 95% | ||||||
| Drug | r | MTF | Taxane | MTF | Taxane | MTF | Taxane | MTF | Taxane |
| MTF/DTX 1:2 | 0.999 | 0.58 | 1.24 | 1.46 | 4.54 | 3.04 | 12.67 | 10.41 | 71.17 |
| MTF/PTX 1:2 | 0.97 | 0.53 | 3.34 | 1.23 | 5.19 | 2.38 | 7.35 | 7.23 | 13.20 |
| PMM/DTX 1:1 | 0.99 | 5.30 | 0.16 | 4.13 | 0.71 | 3.38 | 2.27 | 2.42 | 15.87 |
| PMM/PTX 1:1 | 0.96 | 5.01 | 0.55 | 4.32 | 1.01 | 3.73 | 1.85 | 2.91 | 5.10 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




