1. Introduction
Due to the depletion of high-grade gold deposits, low-grade gold-bearing ores with a high content of clay minerals are increasingly involved in mineral processing. However, the processing of such ores is associated with numerous challenges caused by their fine-grained, layered structure and the anisotropic surface charge of clay minerals. In study [
1], a lignosulfonate-based biopolymer (DP-1777) was applied to mitigate the adverse effects of clay minerals on the flotation of gold-bearing ores with high clay content. It was established that DP-1777 improved both gold grade and recovery by reducing mechanical entrainment and pulp viscosity.
The flotation recovery of finely disseminated gold associated with sulfides and quartz does not exceed 60–80%, and in some cases is limited to 30–40%, despite extensive studies aimed at enhancing gold extraction efficiency and developing new beneficiation technologies [
2]. The improvement of flotation efficiency for refractory gold-bearing ore and technogenic raw materials can be achieved through the implementation of novel technological flowsheets and flotation regimes that ensure minimal gold losses, particularly in fine size fractions [
3].
In studies [
4,
5], the feasibility of beneficiating gold-bearing tailings without regrinding using gravity and flotation methods was investigated. It was demonstrated that combined reagent schemes provide higher gold recovery compared to the application of butyl xanthate alone. The proposed gravity–flotation beneficiation scheme enables the production of gravity and flotation concentrates containing approximately 60 g/t gold with a total metal recovery of about 63.6%.
A laboratory study [
6] investigated the influence of flotation reagents and clay minerals on the recovery of electrum from ore processed at Dundee Precious Metals Krumovgrad. It was found that removing a significant portion of fine particles (<10 μm) by desliming increased the gold grade in the concentrate by nine times. However, this improvement was accompanied by a slight decrease in recovery. Furthermore, a new set of collectors was tested to improve gold recovery and reduce the dosage of potassium amyl xanthate by replacing the existing promoter (AERO
®238) with a new reagent called AEROMAXGOLD™900.
In study [
7], amphiphilic polyethylene glycol polymer was used as a polymeric surfactant for selective and rapid flotation extraction of Au(III), demonstrating superior selectivity and adsorption properties. High separation efficiency and rapid flotation kinetics of gold were reported.
Studies [
8,
9] examined the effect of triethylenetetramine and staged addition of isobutyl xanthate in copper–gold flotation with pyrrhotite depression. Stepwise addition of the collector promotes the formation of a more stable xanthate/dixanthogen layer and increases gold recovery. In study [
10], the influence of particle size on flotation efficiency during copper, gold, and lead separation using potassium dichromate and sodium diethyldithiocarbamate was investigated. The most suitable fraction for gold enrichment in the copper concentrate was −58 + 20 μm, while the coarse fraction −100 + 74 μm proved ineffective for selective gold recovery.
A large-scale industrial case study [
11] integrated mineral processing techniques from laboratory to industrial scale to optimize flotation characteristics of Au–Te ore using Aerophine as a collector. The combination of Aerophine and xanthate significantly improved the recovery of gold, silver, and tellurium in industrial operations.
Gold flotation faces challenges because gold-bearing ores are often refractory and finely disseminated. Low recovery and poor flotation kinetics of fine particles are mainly attributed to the low frequency of particle–bubble collisions, while increased entrainment of fine gangue particles reduces concentrate grade. In study [
12], two pilot-scale Imhoflot™ G-14 cells with tangential feed into a 1.4 m diameter separator were investigated. Bubble size measurements indicated that the pneumatic Imhoflot™ system generates extremely fine bubbles, increasing the collision frequency between mineral particles and air bubbles.
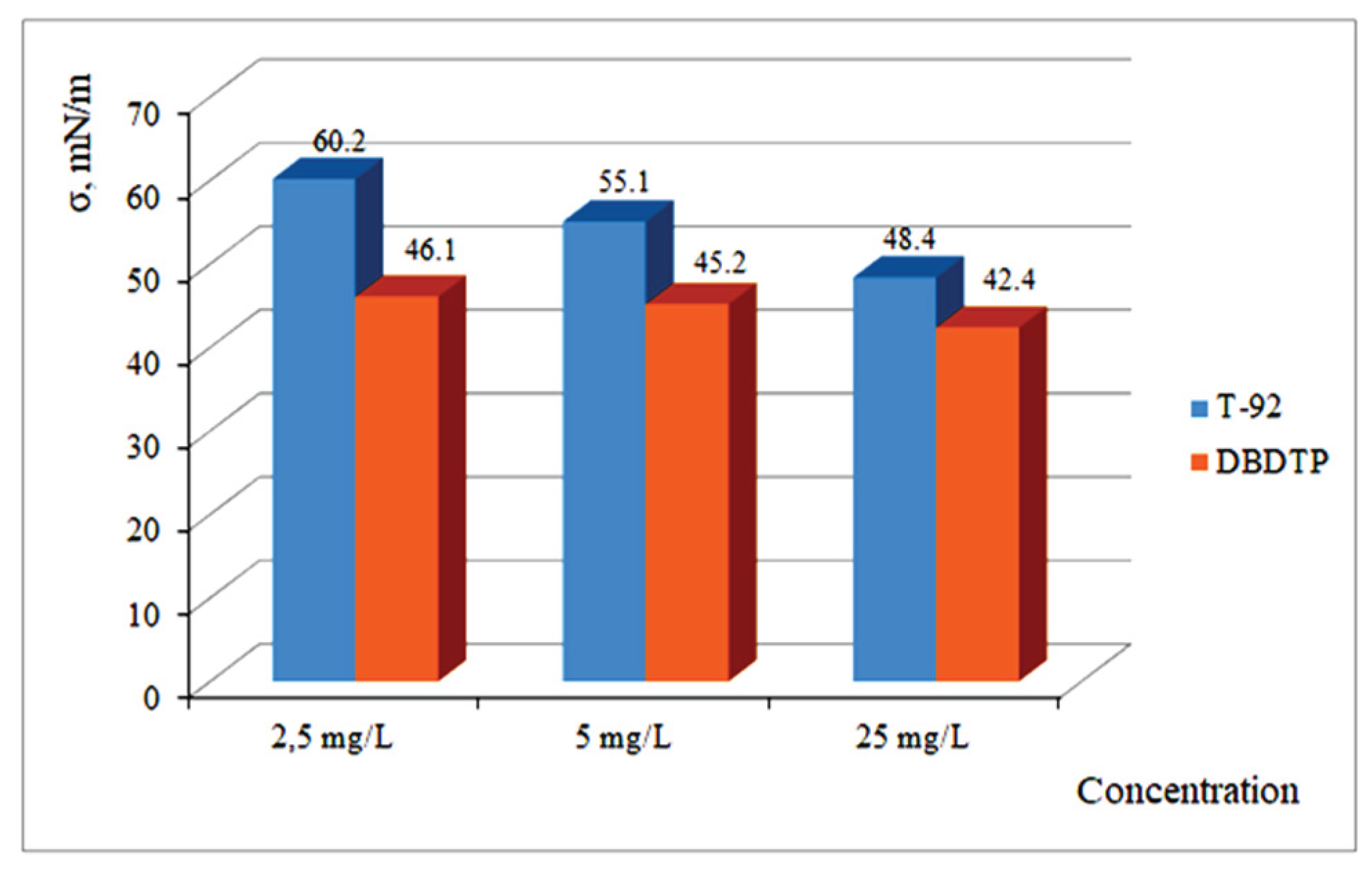
One of the most promising approaches for recovering finely disseminated sulfide-associated gold is the use of flotation reagents in microemulsion form [
13,
14,
15,
16,
17]. Previous studies have demonstrated that the application of microemulsified composite flotation reagents enhances the recovery of gold and copper from technogenic raw materials by 3–5%.
The challenges associated with gold flotation, the presence of finely dispersed particles, and the necessity for fine grinding served as the motivation for the present study. The aim of this work is to investigate the effect of microemulsified flotation reagents on the flotation of gold-bearing ore and technogenic materials, focusing on improving the recovery of fine size fractions smaller than 30 μm. Additionally, this study seeks to identify methods for minimizing gold losses in beneficiation tailings, which is of considerable significance for processing precious metal ores. The application of microemulsions enables more effective separation of gold-bearing sulfides (e.g., pyrite) from gangue minerals, enhancing selectivity. The research objectives included the investigation of flotation kinetics, selection, and optimization of the main flotation process parameters.