1. Introduction
The success of aquaculture depends on maintaining optimal water quality. Key parameters include pH, temperature, salinity, turbidity, and dissolved oxygen (DO), all of which directly affect metabolism, growth rate, feed conversion efficiency, and the survival of cultured organisms [
1,
2]. Among these variables, dissolved oxygen is widely recognized as a critical indicator of water quality, as it directly affects respiratory processes and the physiological performance of crustaceans [
3]. Experimental and field studies have reported that DO concentrations below 5 mg/L induce significant physiological stress, reduce growth, and increase mortality in shrimp farming systems [
1,
4].
In aquaculture systems, dissolved oxygen dynamics are governed by the interaction of multiple factors, including biomass respiration, organic matter degradation, system hydrodynamics, mixing efficiency, and water temperature [
5,
6]. To mitigate hypoxic events, various aeration technologies have been developed, such as paddlewheel aerators, fine-bubble diffusers, venturi injectors, and mechanical surface aerators, each exhibiting specific operational characteristics and different oxygen transfer efficiencies [
1,
3,
7]. However, the overall effectiveness of these devices depends not only on their mechanical design but also on the implemented control strategy and the availability of reliable, continuous dissolved oxygen measurements.
Accurate and real-time measurement of dissolved oxygen is therefore a fundamental requirement for aquaculture automation. Conventional chemical methods, such as Winkler titration and colorimetric techniques, provide acceptable accuracy for spot analyses but are unsuitable for continuous monitoring and closed-loop control applications due to their discrete nature and lack of direct integration with automatic control systems [
8]. In contrast, electrochemical and optical dissolved oxygen sensors enable continuous digital data acquisition and integration into industrial automation and IoT-based architectures. Nevertheless, their practical performance strongly depends on signal conditioning, calibration procedures, temperature compensation, and the employed communication architecture [
9,
10].
In this context, laboratory-scale experimental platforms have become a key tool for research in smart aquaculture and aquatic process control [
11,
12,
13,
14]. These platforms allow the evaluation of different operational configurations representing real cultivation scenarios, such as variations in organic loading, thermal conditions, and turbidity levels [
15,
16]. Moreover, they facilitate systematic variable isolation, enabling controlled investigation of the individual effects of specific disturbances on dissolved oxygen dynamics, for instance by assessing aeration system performance under different temperature regimes or hydraulic mixing conditions [
17,
18]. Additionally, controlled experimental environments offer significant advantages, including reduced operational costs during the development phase, improved experimental reproducibility, and safe validation of advanced control algorithms prior to deployment in commercial production systems [
9,
19]. These characteristics align closely with current trends in smart aquaculture, which emphasize smaller-volume tanks, higher stocking densities, and extensive integration of monitoring and control technologies to ensure water quality while reducing energy and resource consumption [
15,
20].
Despite advances in automation and monitoring, a significant gap remains between large-scale production systems and experimental platforms used in research. In many commercial operations, aeration strategies continue to rely on empirical or time-based schemes without closed-loop feedback, leading to energy inefficiencies and suboptimal dissolved oxygen regulation [
1,
4]. Conversely, many laboratory studies employ low-cost hardware and non-industrial architectures, limiting robustness, scalability, and technological transferability of the obtained results [
21,
22]. This disconnect restricts systematic evaluation of advanced control strategies under controlled yet representative cultivation conditions.
Furthermore, conducting experimental studies using live organisms introduces inherent biological variability, ethical considerations, and risks to animal welfare, complicating reproducibility and rigorous validation of control algorithms [
23]. To address this limitation, several studies have proposed the use of chemical oxygen scavengers, such as sodium sulfite, to induce controlled deoxygenation conditions in laboratory aquatic systems [
23,
24]. However, the practical application of this approach is nontrivial, as the theoretical reaction stoichiometry does not fully represent the actual conditions of potable water used experimentally. In addition, reaction kinetics depend on factors such as mixing intensity, water motion, and temperature, while reagent purity and storage conditions significantly influence experimental reproducibility [
24].
Within this framework, the present work aims to design and construct a laboratory-scale experimental platform, oriented toward the modeling and control of dissolved oxygen dynamics in water. Beyond the platform development itself, the main contribution of this work lies in addressing a persistent gap in the existing literature, where clear, reproducible, and well-documented methodologies for the systematic study of aeration processes remain limited. The proposed framework integrates industrial-grade instrumentation, aeration systems, and a programmable logic controller, enabling real-time monitoring and implementation of control strategies under controlled experimental conditions. Carefully designed experimental protocols are conducted to achieve repeatable and reliable results, supported by consistent performance metrics. The proposed methodology and experimental dataset are intended to serve as a benchmark reference for future studies focused on the evaluation of alternative modeling approaches, aeration technologies, and control strategies in smart aquaculture and aquatic systems automation.
In summary, the main contributions of this work are:
The design and implementation of an industrial-grade DO control platform, integrating a programmable logic controller (PLC), an optical dissolved oxygen sensor, and a proportional flow valve driven by a current controlled actuator.
A reproducible experimental methodology for benchmarking control strategies under identical operating conditions, including repeated near-zero DO reset experiments, explicit signal conditioning, actuator saturation handling, and time domain performance metrics suitable for real world industrial deployment.
Quantitative comparison of On–Off and PID control strategies based on performance indicators and a statistical approach to establish the necessary number of experimental repetitions.
The remainder of this article is structured as follows.
Section 2 (Materials and Methods) describes the proposed experimental framework, including the design and construction of the laboratory-scale platform, the instrumentation employed, and the management methodology adopted. This section also details the control design and the experimental validation procedure.
Section 3 presents the experimental results and discussions obtained under controlled operating conditions. Finally, the conclusions are presented in
Section 4.
3. Results and Discussions
3.1. Implementation of the Experimental Platform and Management Methodology
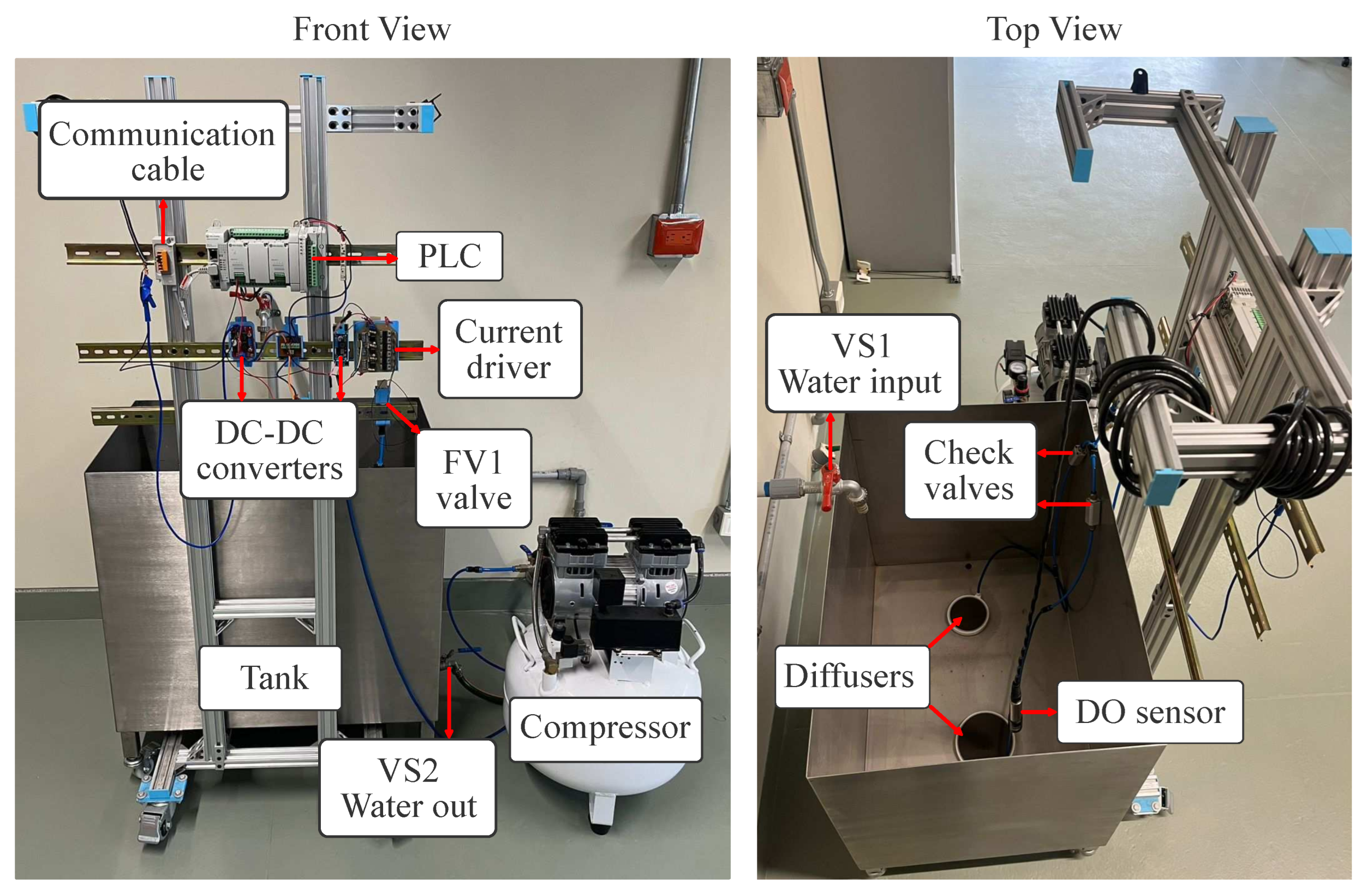
The experimental platform developed in this work was successfully implemented and operated as a laboratory-scale test bed for DO control experiments. The final system integrates the process tank, pneumatic aeration subsystem, sensing instrumentation, control hardware, and power distribution into a compact and functional structure, as shown in
Figure 6. The implemented platform is consistent with the proposed design and enabled systematic experimentation under controlled conditions.
The process tank operated at a nominal working volume of 160 L in all experimental trials. Water handling components allowed reliable filling and draining between experiments, while the pneumatic subsystem operated without mechanical faults, providing consistent air delivery during closed-loop operation.
The sensing and control infrastructure functioned reliably throughout the experiments. The optical DO sensor provided continuous measurements, and stable RS-485 communication with the programmable logic controller (PLC) ensured uninterrupted data acquisition. Control execution within the PLC was performed in discrete time, with actuation commands continuously applied to the proportional valve.
The chemical oxygen depletion procedure based on the manual addition of 12 g of sodium sulfite () consistently reduced the DO concentration to low levels prior to control activation. Similar depletion behavior was observed across repeated trials, confirming the reproducibility of the selected dosing and mixing procedure.
Transitions between preparation, closed-loop operation, and reset phases were achieved without operational discontinuities. The reset procedure, involving tank draining and sensor cleaning, enabled recovery of comparable initial conditions between experiments. Dissolved oxygen measurements, control signals, and time stamps were logged continuously in all trials, supporting offline analysis. Basic signal conditioning reduced short-term measurement fluctuations while preserving the dominant process dynamics.
Overall, the obtained results confirm that the implemented platform and its associated management methodology provide a reliable and reproducible laboratory-scale environment for the experimental validation of dissolved oxygen control strategies.
3.2. Results of Dissolved Oxygen Control
Considering the platform above, this work uses the two mentioned controller methods to compare the results under the same conditions. The objective of each test is to track a desired DO level by regulating the valve aperture. The sensor measurements were filtered using an IIR filter with a forgetting factor of
. In this system, the increase in DO is determined by the valve, while sulfite determines the drop. Before control initialization, the DO level was reduced to a value close to zero by adding 12 g of sodium sulfite, exceeding the 9.3 g required by stoichiometry, due to the use of potable water instead of pure water, as described in
Section 2.2.
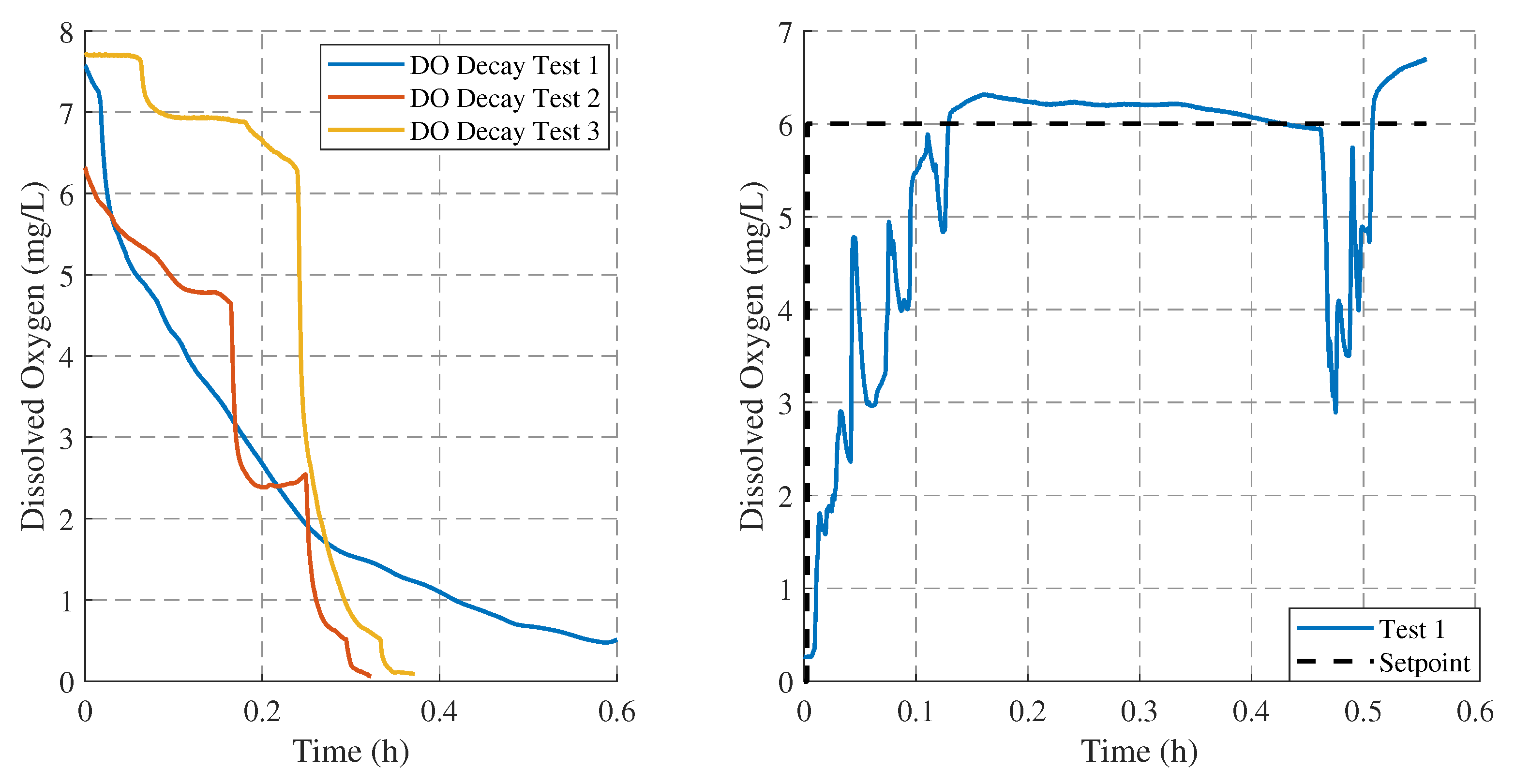
This reduction in DO is difficult to model because, in addition to the appropriate amount of sodium sulfite, it depends on water movement, which can accelerate the reaction through superficial aeration. Furthermore, the reduction in DO also depends on the initial DO value, which itself is influenced by the water flow rate when filling the tank.
Figure 7 (left) illustrates three examples of DO reduction from a high initial value. In Test 1, a typical decay is observed without water agitation. In Tests 2 and 3, the reduction occurs more rapidly due to agitation. However, in Test 2, a slight increase can be observed after 0.2 h, caused by aeration generated by water agitation. Altogether, these observations highlight the need to conduct experiments under the same initial DO condition, close to zero, prior to controlled aeration evaluated in
Section 3.2.1 and
Section 3.2.2, in order to reach repeatability under laboratory conditions.
In this empirical adjustment of the required amount of sodium sulfite, it is difficult to ascertain whether, when a DO value close to zero is measured, residual sodium sulfite remains in the water, ready to react once the water is aerated.
Figure 7(right) illustrates a typical aeration process under such excess conditions (about 20 g), where sodium sulfite reacts immediately upon air introduction, producing abrupt changes in the DO measurement from the outset, as both the increase in DO and the simultaneous consumption of sodium sulfite occur together.
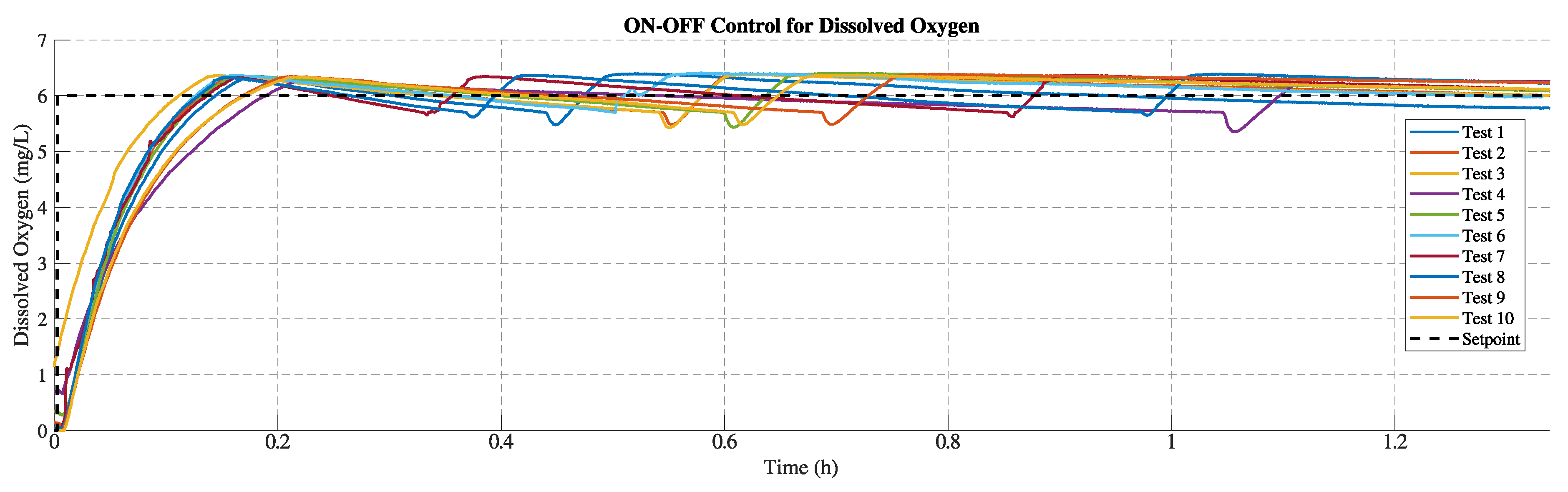
For both control approaches, this study performed 10 tests for each to evaluate the consistency of their responses.
3.2.1. Results of the On-Off Control Strategy
The On-Off control employs 5% hysteresis with respect to
mg/L (i.e.
mg/L). In this case, the 10 tests last approximately 1.3 hours to track the DO setpoint. Sodium sulfite provides a repeatable oxygen demand disturbance that reduces DO in the bench system. This controller provided a fair response in tracking the reference throughout the test. In
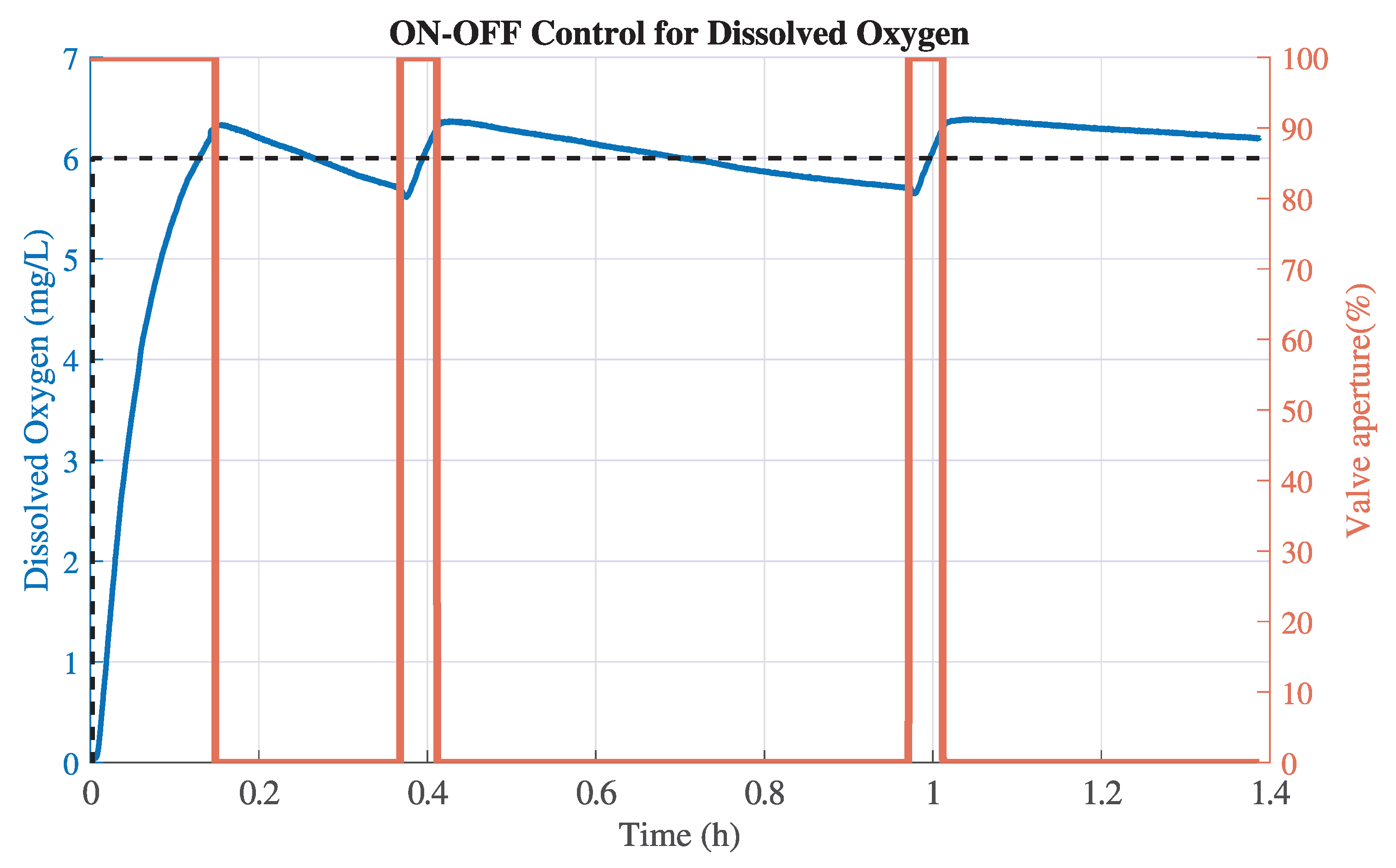
Figure 8 and
Figure 9, note that the peaks reached in each test are produced due to the delay effect of the aerator. The DO level continues to rise despite the control valve being completely closed. Conversely, the minimum peak corresponds to the drop in DO caused by the sulfite reaction when the valve is opened to regulate the DO level.
Table 2 summarizes the performance obtained in each test using the proposed controller. In terms of overshoot, the average is
(mg/L), with a maximum of
(mg/L) in test 5 and a minimum of
(mg/L) in test 4. The settling time has an average of 451.4 seconds (0.125 hours) with a minimum of 339 and a maximum of 595 at tests 3 and 4, respectively. On the other hand, the average rising time is 355.4 seconds (0.099 hours), with a maximum of 523.5 at test 4 and a minimum of 291 at test 3. Finally, the maximum mean error was observed in test 5, while the minimum occurred in test 10. These On-Off control performance results can be adjusted depending on the value of the hysteresis band
H[
31].
3.2.2. Results of the PID Control Strategy
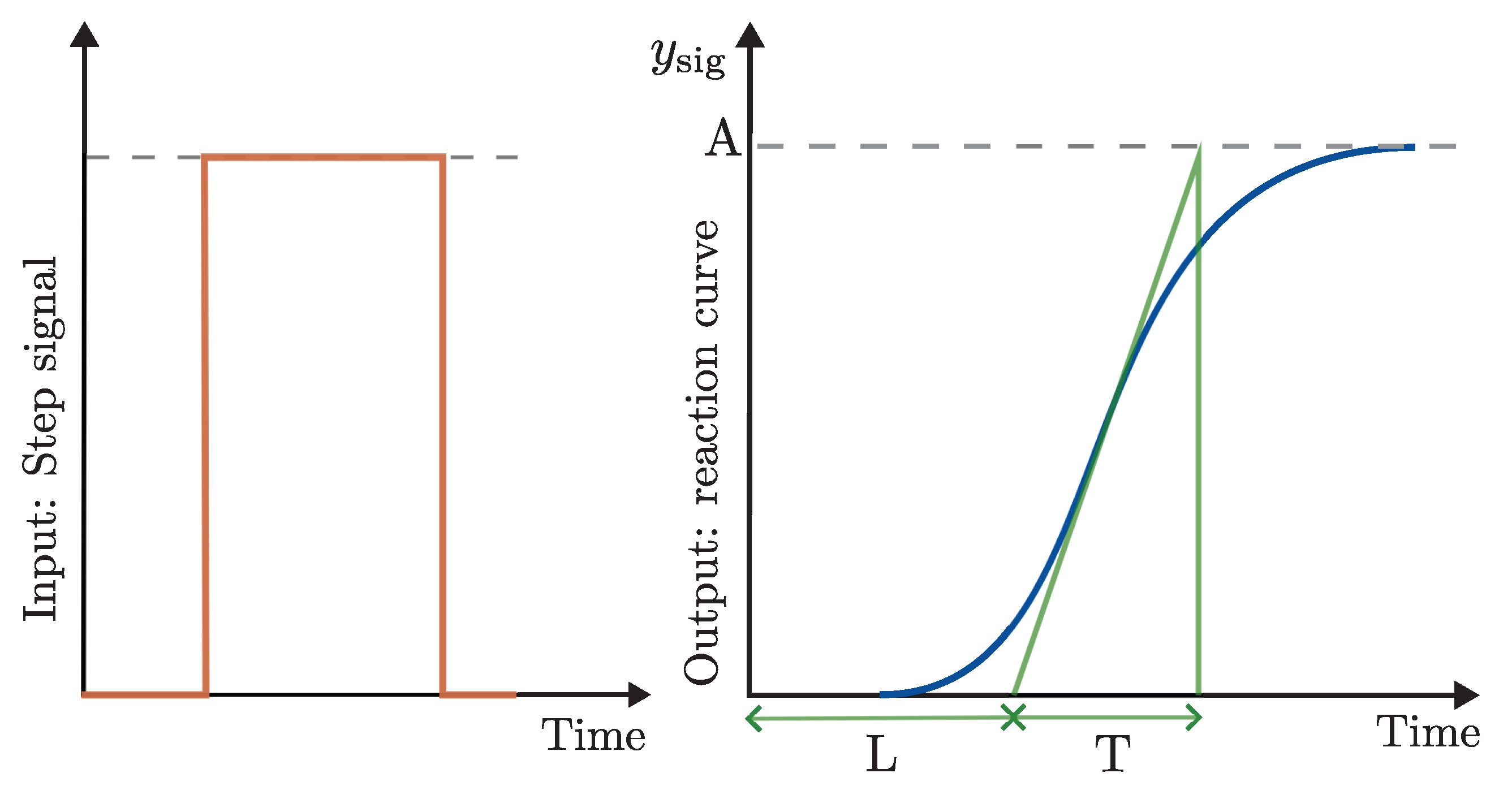
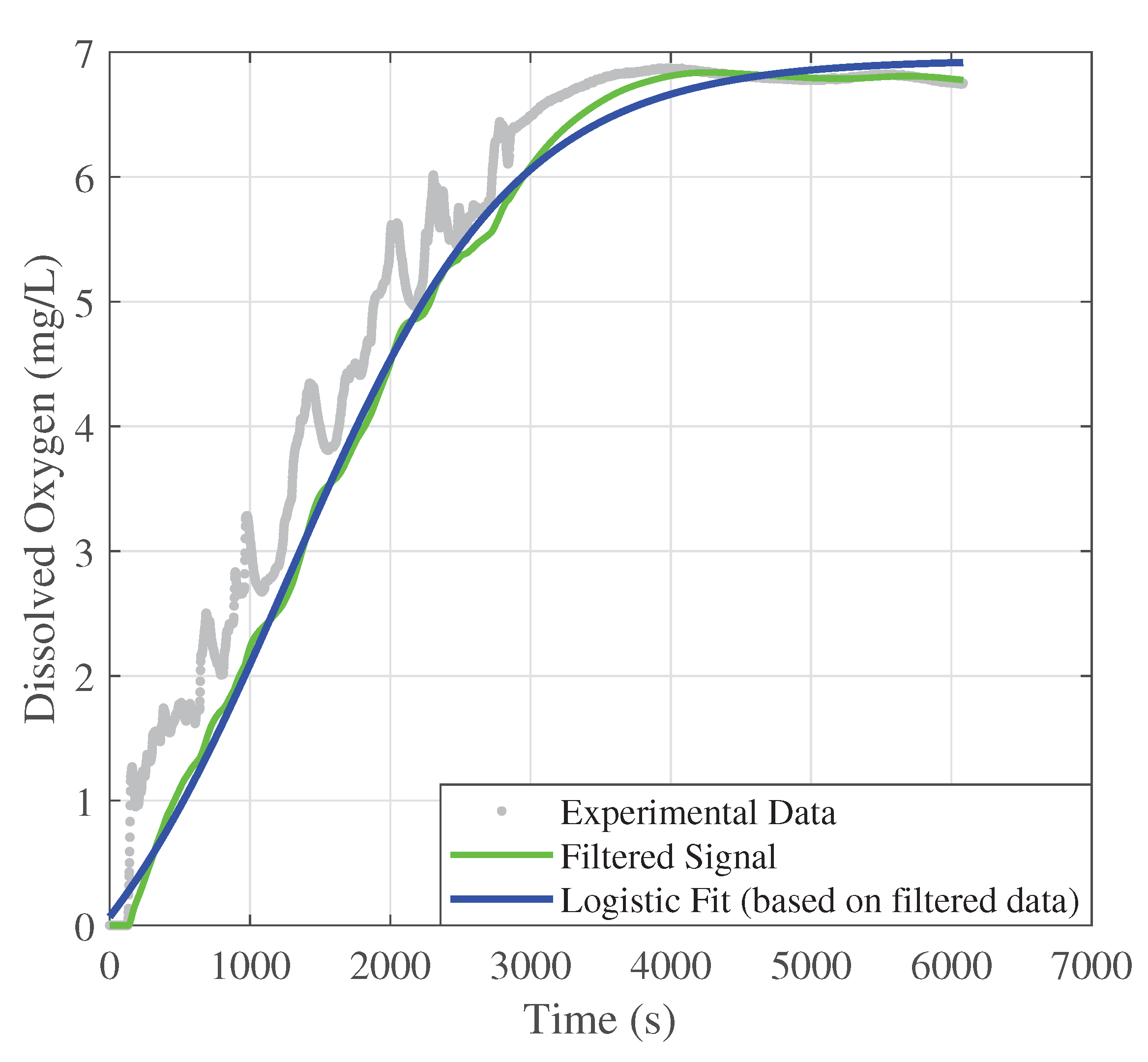
The second controller proposed by this work is the PID control algorithm. In this case, the test bench uses all the same conditions considered in the previous controller. The Ziegler-Nichols method is applied in the reaction curve, shown in
Figure 10, to obtain the proportional
, integral
, and derivative gains
, with
and
.
Figure 10 illustrates the system’s reaction curve, as presented in
Section 2.3.3. The gray dots represent the response to a step input until steady state, while the green line corresponds to the filtered data obtained using the discrete filter described in Equation (
3). The adjusted sigmoidal reaction curve is shown as the blue line, derived from Equation (
4).
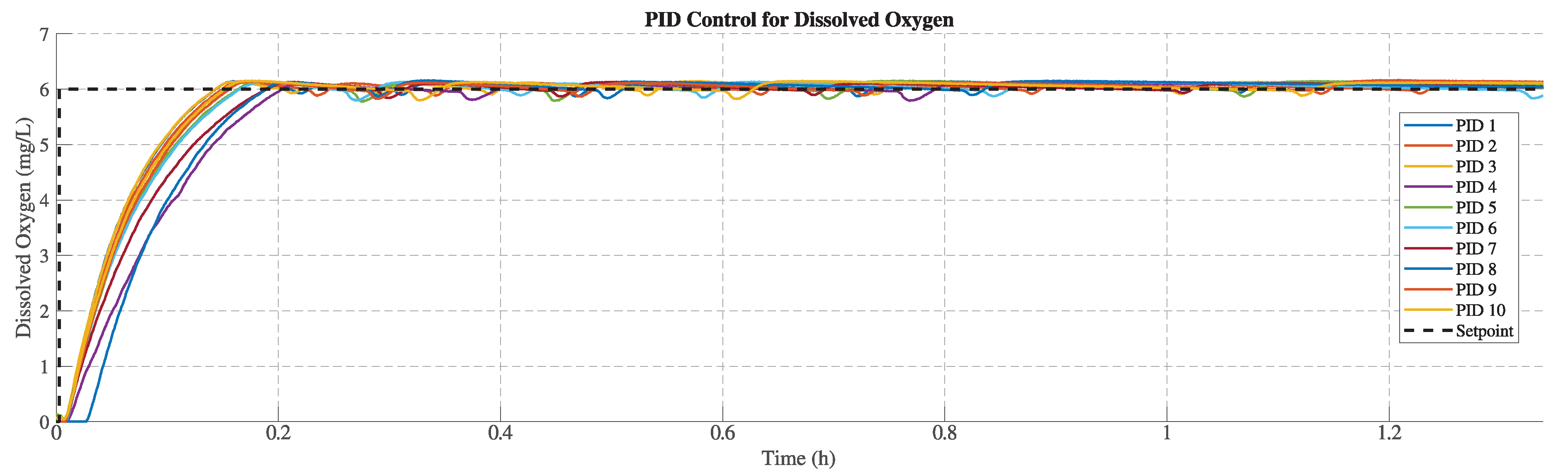
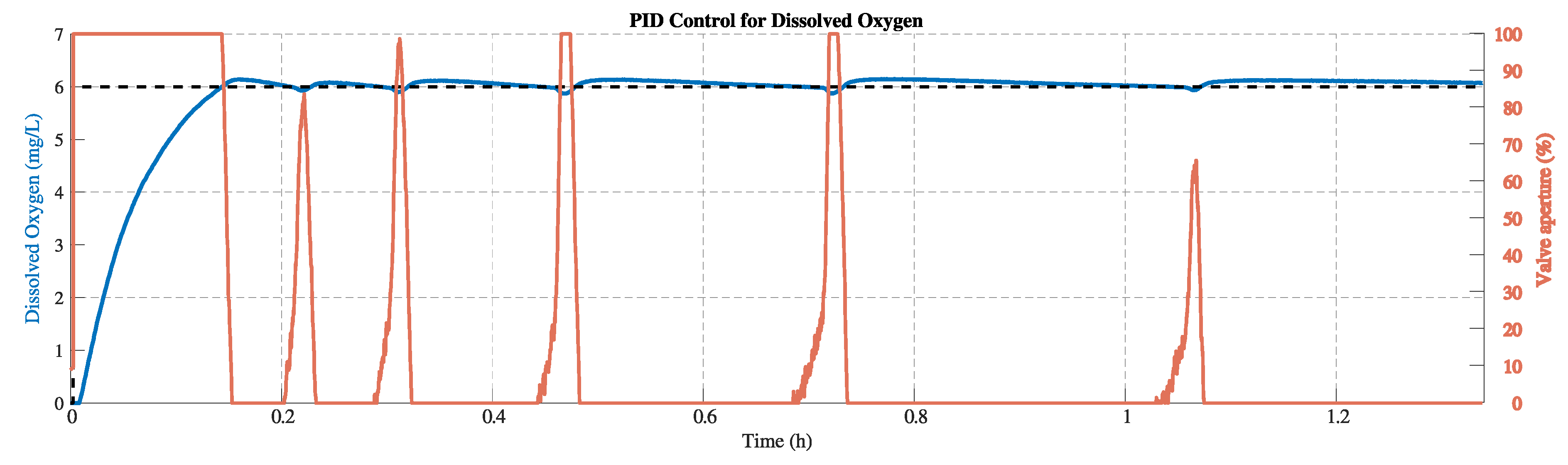
Figure 11 and
Figure 12 illustrate the responses obtained by implementing the PID controller in the system. The setpoint remains at 6 mg/L, considering the same amount of sulfite in the tank. Each test shows a minimum peak generated due to the reaction of the sulfite with water when the aerator is activated. Compared with the On-Off, the responses do not exhibit oscillations, and the behavior is smoother.
Table 3 presents the performance metrics of the PID implementation. The maximum overshoot is
(mg/L) at test 5; meanwhile, the minimum is
(mg/L) at test 4, with an average of
(mg/L). The average rising time is 394.3 seconds (0.11 hours), with a maximum of 505 seconds recorded in test 4 and a minimum of 340.5 seconds recorded in test 1. In terms of settling time, the average is 521.5 seconds, with a maximum of 651 (test 4) and a minimum of 446.5 (test 3), respectively. Finally, the maximum mean error was observed in test 4, while the minimum was observed in test 9.
The Z–N method was used as a standardized baseline; further fine-tuning was not pursued to keep methodology comparable and replicable. Therefore, these PID control results can be improved with more advanced tuning alternatives and other, more robust control strategies [
31,
36].
During the control tests, the PID controller shows lower steady-state error and variability under the tested conditions compared to the On–Off controller, see
Table 4, particularly in the steady state. In the transient state, however, the settling time of both controllers is similar, since the PID controller saturates at 100% owing to the large error, see
Figure 12. It is difficult to draw definitive conclusions in this regime, partly because it is further influenced by the residual sodium sulfite. In practical applications, however, it is the steady state that is most relevant.
3.3. Numerical Evaluation of the Required Number of Repetitions
The confidence interval based criterion was applied to the On-Off and PID controllers in order to determine the number of repetitions required for each case to obtain a reliable estimate of the steady-state mean error. The analysis follows the procedure described in
Section 2.4. The required number of independent experimental repetitions was determined using a two-sided
confidence interval.
For each controller, the minimum N (number of Test) was selected such that the confidence interval semi-width satisfies the practical accuracy criterion where mg/L is the reference value. This condition ensures that the estimation uncertainty remains within of the reference.
For the On-Off controller, the minimum number of repetitions satisfying this requirement was
, whereas for the PID controller only
repetitions were required.
Table 4 summarizes the corresponding steady-state error mean
, the standard error
, the confidence semi-width
, and the relative error with respect to the reference.
It can be observed that, under the selected tuning and experimental conditions, the PID controller exhibits lower dispersion and therefore requires fewer repetitions to achieve the same statistical confidence level. However, this comparison should be interpreted as referential. Each controller may improve its performance and variability characteristics through further tuning adjustments. Consequently, the purpose of this analysis is not to establish a strict superiority of one control strategy over the other, but rather to illustrate a systematic and statistically grounded procedure for determining the sufficient number of experimental repetitions in each case.
3.4. Limitations and Future Work
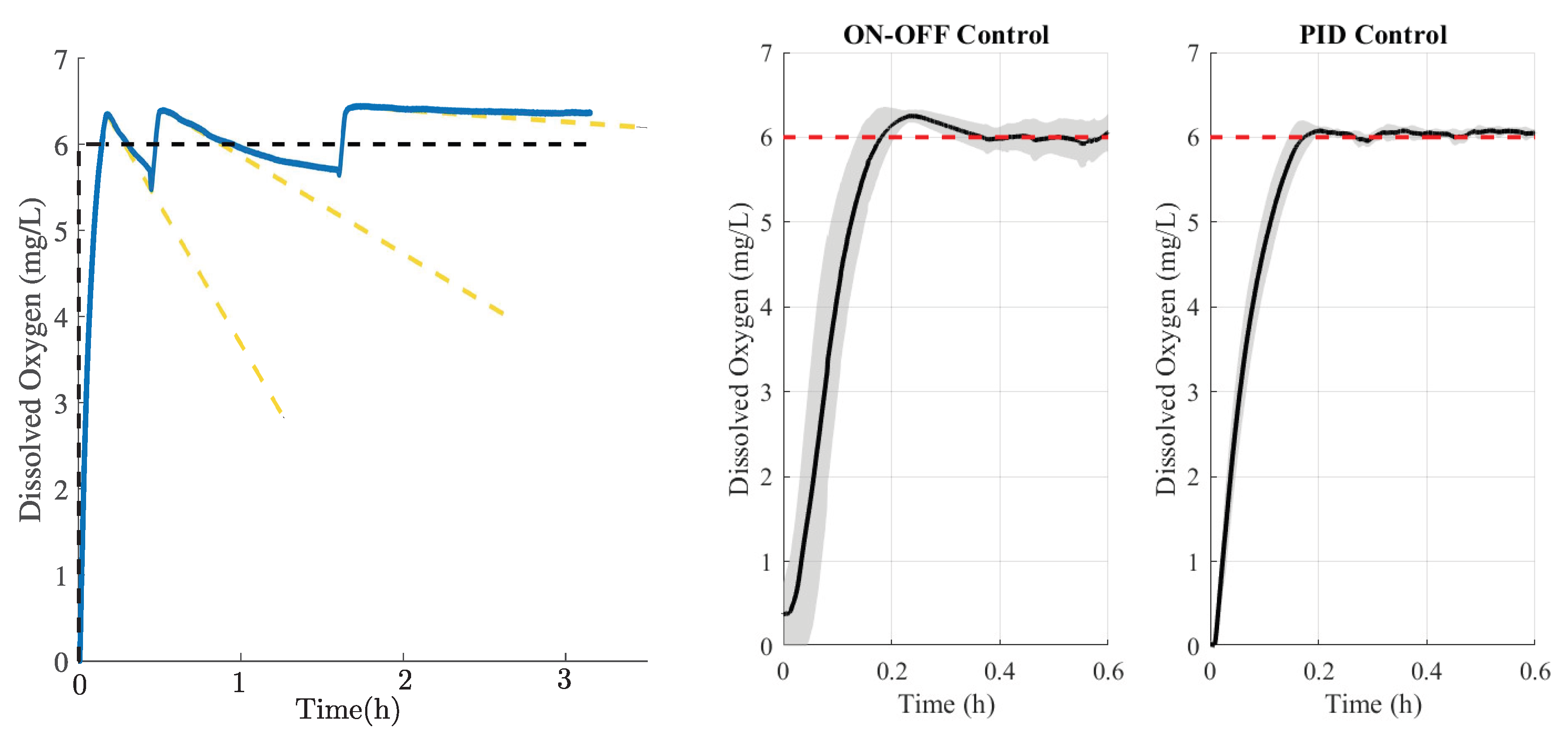
An interesting observation regarding DO regulation under On-Off control is that each oscillation exhibits a downward slope proportional to the residual sodium sulfite, as illustrated by the yellow dashed line in
Figure 13 (left) following the initialization stage. Consequently, during the third oscillation, the variation in DO level becomes nearly constant, indicating that the sodium sulfite has been almost completely consumed. In other words, the disturbance generated in the aeration system due to the residual sodium sulfite is temporary, lasting on average three cycles in the steady state under On-Off control, although it is also expected to occur under PID control. Future work could focus on developing a methodology that not only reduces DO to its minimum value but also eliminates residual sodium sulfite prior to aeration control, thereby reducing the error variance observed in
Figure 13 (middle and right).
Furthermore, in this study, we assumed that the amount of air supplied by the diffusers is proportional to the valve aperture. However, this relationship is not strictly linear in real applications. According to the technical specifications of both the air compressor and the diffusers, the dependence between these parameters is nearly linear across most operating ranges, but becomes nonlinear at the extremes. As future work, we suggest modeling this nonlinear behavior at the boundaries, using some data based modeling method [
30,
37,
38]. Moreover, data acquisition was carried out at a sampling frequency of 2 Hz (
s), which corresponds to the maximum rate permitted by the sensor. This rate can subsequently be reduced to lower the computational cost, where the minimum sampling rate value must satisfy the Nyquist criterion.
Finally, the results presented in this work encourage us to further complicate the problem by introducing disturbances into the system, such as varying the pond temperature or including living species in the tank. In addition, the aeration mechanism could be replaced with one that generates significant mass movement [
11], so that new and more advanced control strategies can be studied on the platform proposed in this paper.
Author Contributions
Conceptualization, J.M. and E.J.; methodology, J.M., S.P., A.G., and E.J.; software, J.M.; validation, J.M.; formal analysis, J.M. and E.J.; investigation, J.M. and E.J.; resources, J.M.; data curation, J.M.; writing original draft preparation, J.M., S.P., A.G., and E.J.; writing, review and editing, J.M., S.P., A.G., and E.J.; visualization, J.M., S.P., A.G., and E.J.; supervision, E.J.; project administration, E.J.; funding acquisition, E.J. All authors have read and agreed to the published version of the manuscript.
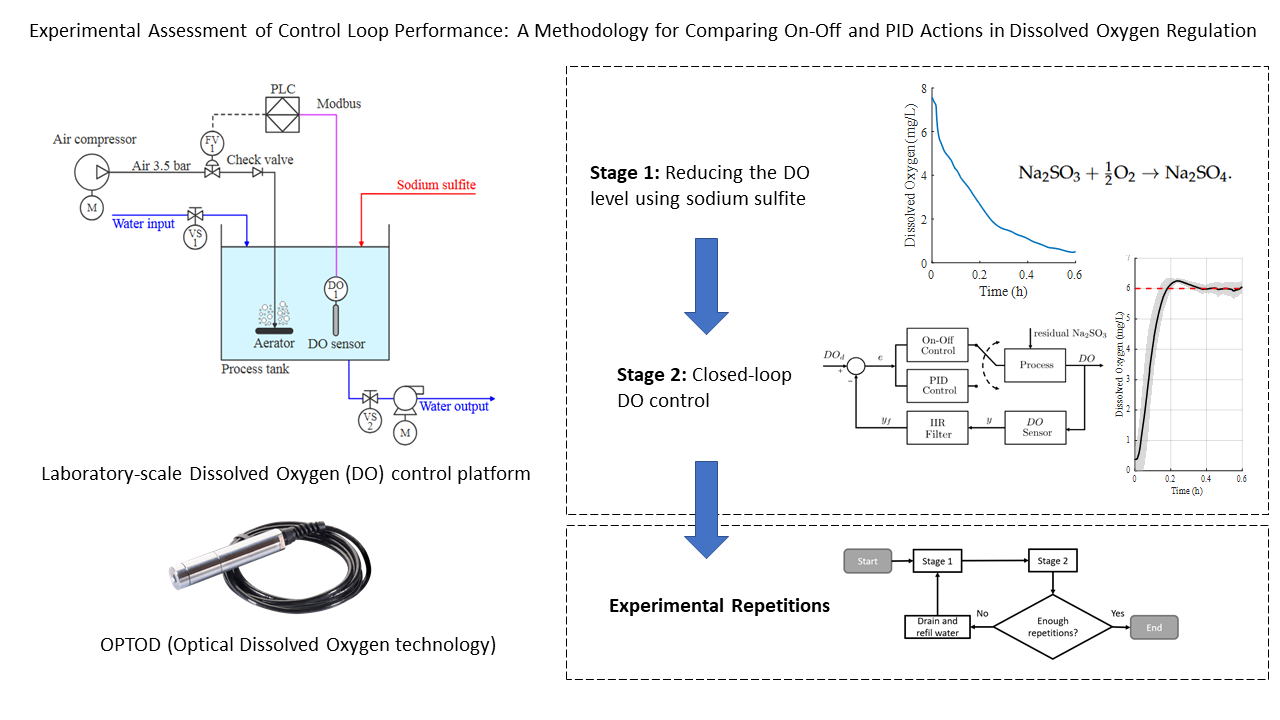
Figure 1.
Functional architecture and integrated air, water, and signal flows of the experimental DO control platform.
Figure 1.
Functional architecture and integrated air, water, and signal flows of the experimental DO control platform.
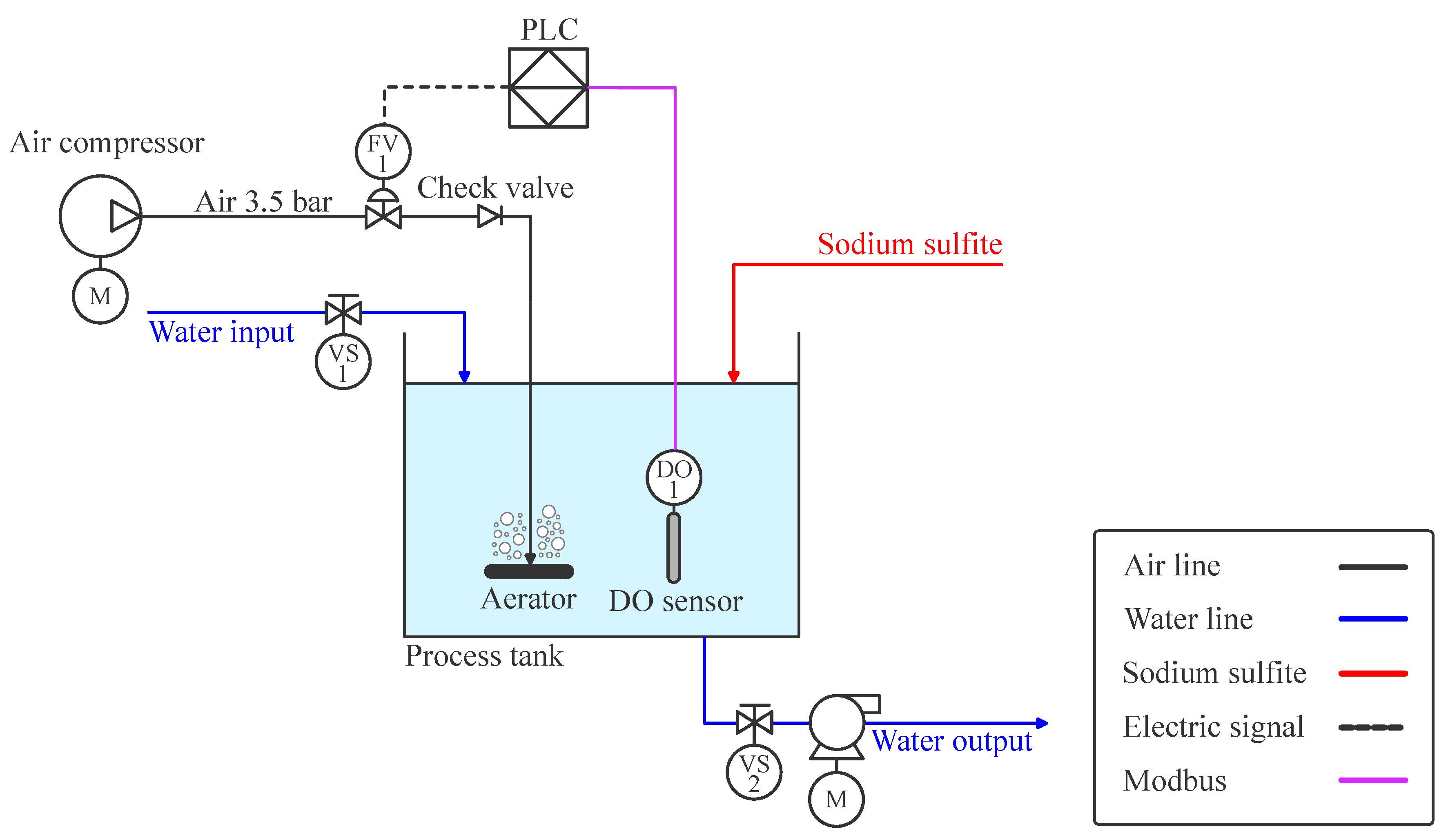
Figure 2.
Control and instrumentation architecture of the experimental DO control platform.
Figure 2.
Control and instrumentation architecture of the experimental DO control platform.
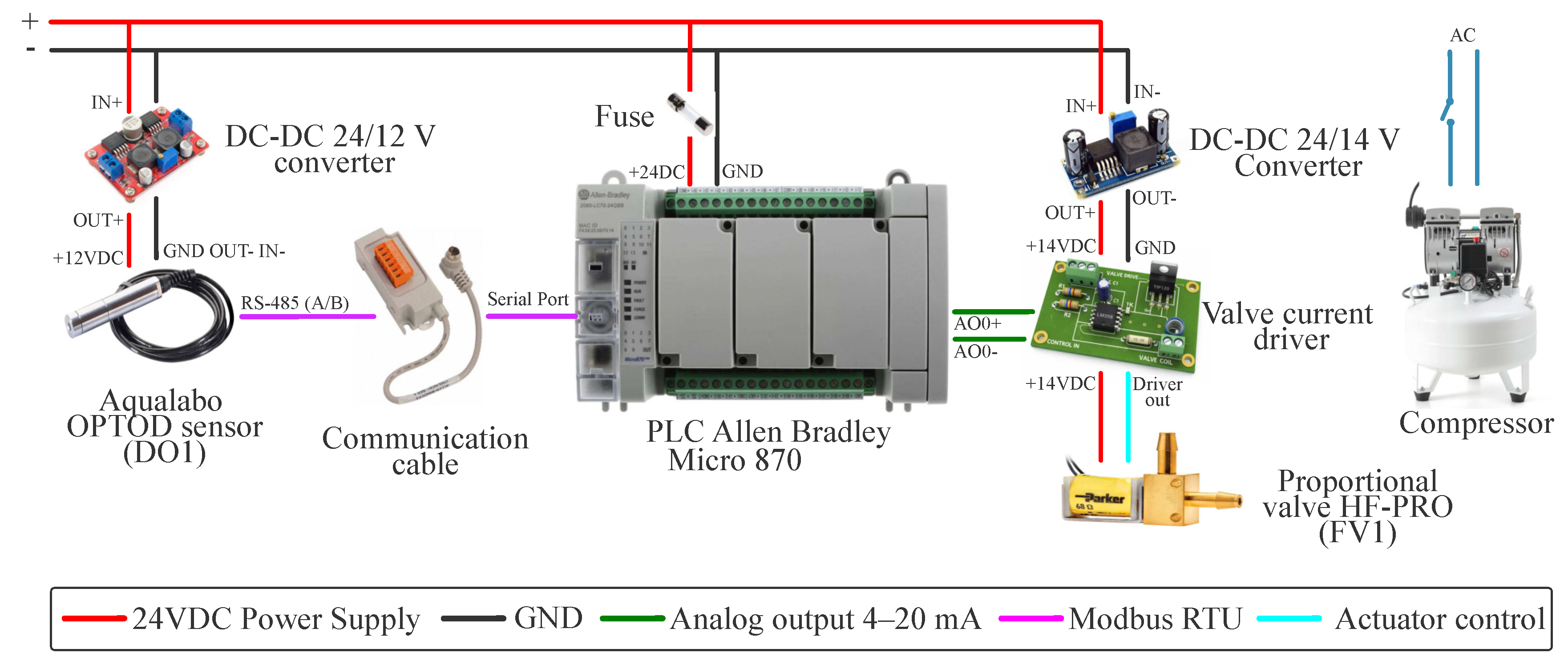
Figure 3.
Operational workflow for experimental execution on the DO control platform.
Figure 3.
Operational workflow for experimental execution on the DO control platform.
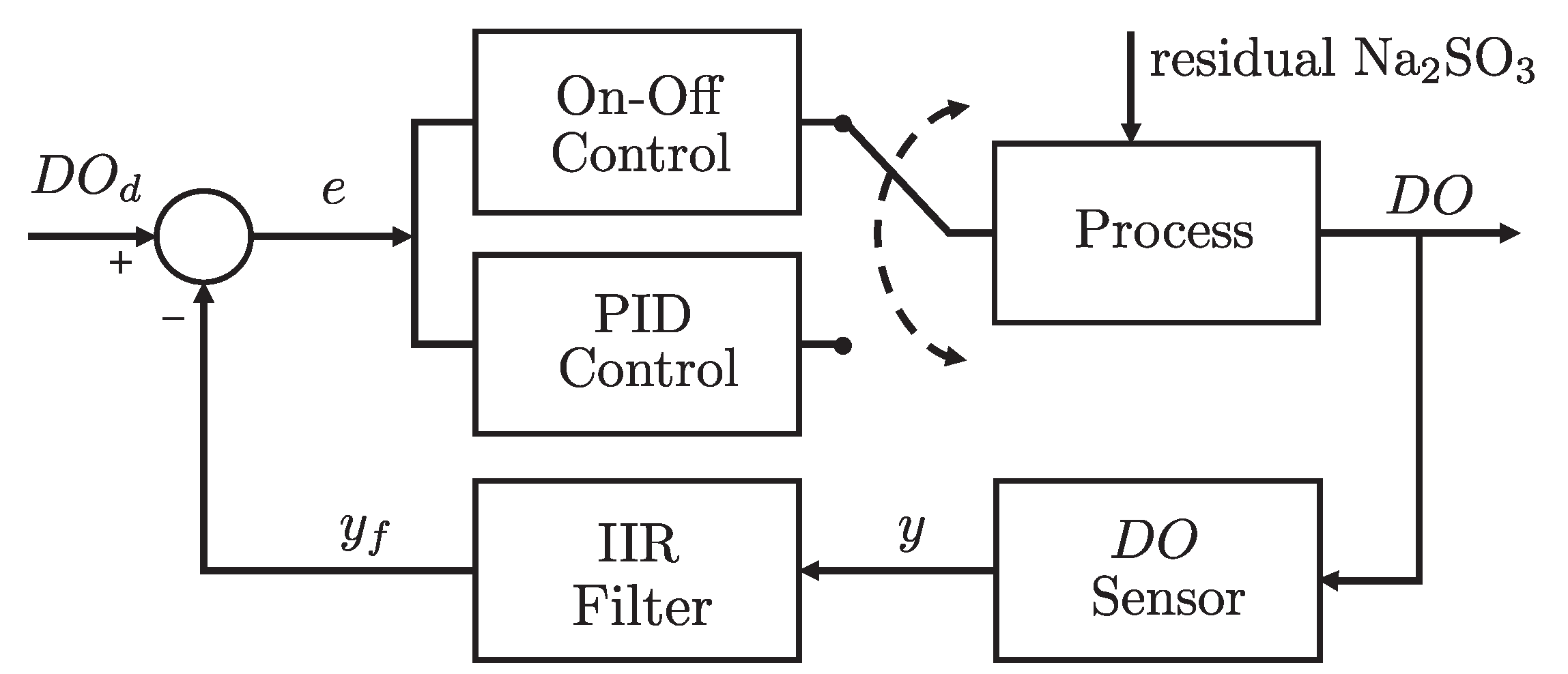
Figure 4.
Block diagram of the feedback aeration control system.
Figure 4.
Block diagram of the feedback aeration control system.
Figure 5.
Step input signal (left) and corresponding reaction curve with Z-N parameters (right).
Figure 5.
Step input signal (left) and corresponding reaction curve with Z-N parameters (right).
Figure 6.
Implemented experimental platform for dissolved oxygen control experiments: front and top views.
Figure 6.
Implemented experimental platform for dissolved oxygen control experiments: front and top views.
Figure 7.
Example of three typical DO decay curves, starting from the high values characteristic of freshly collected drinking water and decreasing to a minimum due to reaction with sodium sulfite (left), and typical DO evolution when an excessive amount of sodium sulfite (about 20 g) is added (right).
Figure 7.
Example of three typical DO decay curves, starting from the high values characteristic of freshly collected drinking water and decreasing to a minimum due to reaction with sodium sulfite (left), and typical DO evolution when an excessive amount of sodium sulfite (about 20 g) is added (right).
Figure 8.
Dissolved oxygen responses obtained with On–Off control over 10 repetitions at a setpoint of 6 mg/L.
Figure 8.
Dissolved oxygen responses obtained with On–Off control over 10 repetitions at a setpoint of 6 mg/L.
Figure 9.
Corresponding valve aperture (orange line) for Test 1 under On–Off control (blue line).
Figure 9.
Corresponding valve aperture (orange line) for Test 1 under On–Off control (blue line).
Figure 10.
Measured, filtered, and sigmoidal-adjusted reaction curves of the DO signals (gray, green, and blue lines, respectively).
Figure 10.
Measured, filtered, and sigmoidal-adjusted reaction curves of the DO signals (gray, green, and blue lines, respectively).
Figure 11.
Dissolved oxygen responses obtained with PID control over 10 repetitions at a setpoint of 6 mg/L.
Figure 11.
Dissolved oxygen responses obtained with PID control over 10 repetitions at a setpoint of 6 mg/L.
Figure 12.
Corresponding valve aperture (orange line) for Test 1 under PID control (blue line).
Figure 12.
Corresponding valve aperture (orange line) for Test 1 under PID control (blue line).
Figure 13.
Long-term effect of aeration decrease: On–Off control (left), DO control with variance shadow applied to On–Off (middle), and PID (right).
Figure 13.
Long-term effect of aeration decrease: On–Off control (left), DO control with variance shadow applied to On–Off (middle), and PID (right).
Table 1.
Main instrumentation and hardware specifications of the experimental DO control platform.
Table 1.
Main instrumentation and hardware specifications of the experimental DO control platform.
| Element |
Key specifications |
Function in the system |
| Process tank |
Rigid tank, 160 L working volume; laboratory-scale geometry; single-phase liquid operation. |
Physical process where dissolved oxygen dynamics are regulated. |
| Aerators |
Submerged air diffusers; porous stone type; characteristic pore size in the sub-millimeter range; installed symmetrically at the tank bottom; nominal airflow ≈1–3 L/min per aerator. |
Promote gas-liquid oxygen transfer and spatially uniform aeration. |
| Air compressor |
Oil-free air compressor; Set to a pressure of 3.5 bar; air tank capacity ≈40 L; nominal flow capacity ≈160 L/min. |
Provides pressurized air to the aeration subsystem. |
| Flow control valve (FV1) |
Proportional valve; 2-way normally closed; maximum flow up to ≈60 SLPM; operating pressure up to 3.5 bar; current-driven actuation (0–175 mA). |
Final control element regulating aeration rate. |
| Valve driver |
Analog current driver; PLC-compatible input; valve supply voltage 12–14 VDC; maximum output current ≈200 mA. |
Conditions the PLC output to drive the proportional valve. |
| DO sensor |
Optical dissolved oxygen probe; measurement range 0–20 mg/L; accuracy ±0.1 mg/L; Modbus RS-485 communication; integrated temperature compensation. |
Provides real-time DO measurements for feedback control. |
| Controller |
Programmable Logic Controller (PLC); discrete-time execution; analog output (4–20 mA); serial communication via RS-485 (Modbus). |
Executes the closed-loop DO control algorithm. |
| DC power conversion |
DC-DC converters; 24 VDC main supply stepped down to 12 VDC and 14 VDC; current capacity up to 3 A. |
Supplies regulated power to valve driver and instrumentation. |
Table 2.
Test results and average performance of the On-Off Control
Table 2.
Test results and average performance of the On-Off Control
| Test |
Overshoot () |
Rising time (s) |
Settling time (s) |
(mg/L) |
| 1 |
44.4 |
346.5 |
454.0 |
36.0 |
| 2 |
46.2 |
313.5 |
412.5 |
25.2 |
| 3 |
37.8 |
291.0 |
339.0 |
9.6 |
| 4 |
34.2 |
523.5 |
595.0 |
21.0 |
| 5 |
93.6 |
326.5 |
425.5 |
51.6 |
| 6 |
71.4 |
303.0 |
400.5 |
28.8 |
| 7 |
37.2 |
339.5 |
426.0 |
10.8 |
| 8 |
38.4 |
301.5 |
402.0 |
22.2 |
| 9 |
38.4 |
406.5 |
529.5 |
9.6 |
| 10 |
36.6 |
402.0 |
530.0 |
2.4 |
| Average |
47.4 |
355.4 |
451.4 |
21.6 |
Table 3.
Test results and average performance of the PID Control
Table 3.
Test results and average performance of the PID Control
| Test |
Overshoot ( mg/L) |
Rising time (s) |
Settling time (s) |
(mg/L) |
| 1 |
15.0 |
340.5 |
449.0 |
4.2 |
| 2 |
13.8 |
355.5 |
468.5 |
6.6 |
| 3 |
13.8 |
338.5 |
446.5 |
6.0 |
| 4 |
12.0 |
505.0 |
651.0 |
10.8 |
| 5 |
42.6 |
396.5 |
514.0 |
6.6 |
| 6 |
15.6 |
399.0 |
519.0 |
6.0 |
| 7 |
12.6 |
459.5 |
590.0 |
9.0 |
| 8 |
15.6 |
412.5 |
606.0 |
10.2 |
| 9 |
16.2 |
372.5 |
493.0 |
3.6 |
| 10 |
20.4 |
363.5 |
477.5 |
6.0 |
| Average |
17.4 |
394.3 |
521.5 |
7.2 |
Table 4.
Summary of required repetitions and statistical indicators for each controller (95% confidence interval).
Table 4.
Summary of required repetitions and statistical indicators for each controller (95% confidence interval).
| Controller |
|
|
|
|
Rel. Error (%) |
Rel. (%) |
| On-Off |
8 |
0.2565 |
0.0482 |
0.1140 |
4.28 |
|
| PID |
3 |
0.0560 |
0.0072 |
0.0310 |
0.93 |
|