Submitted:
19 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
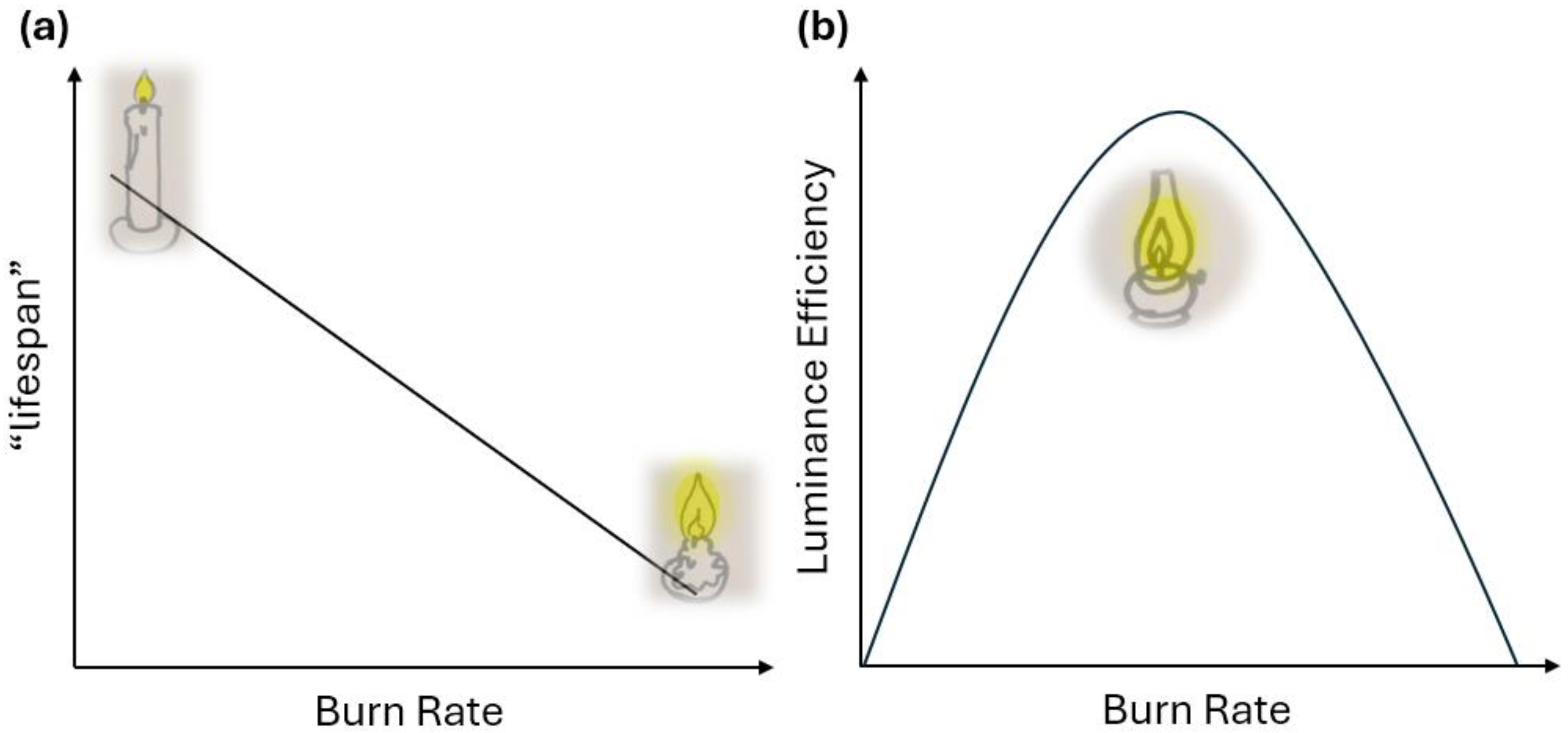
2. The Model
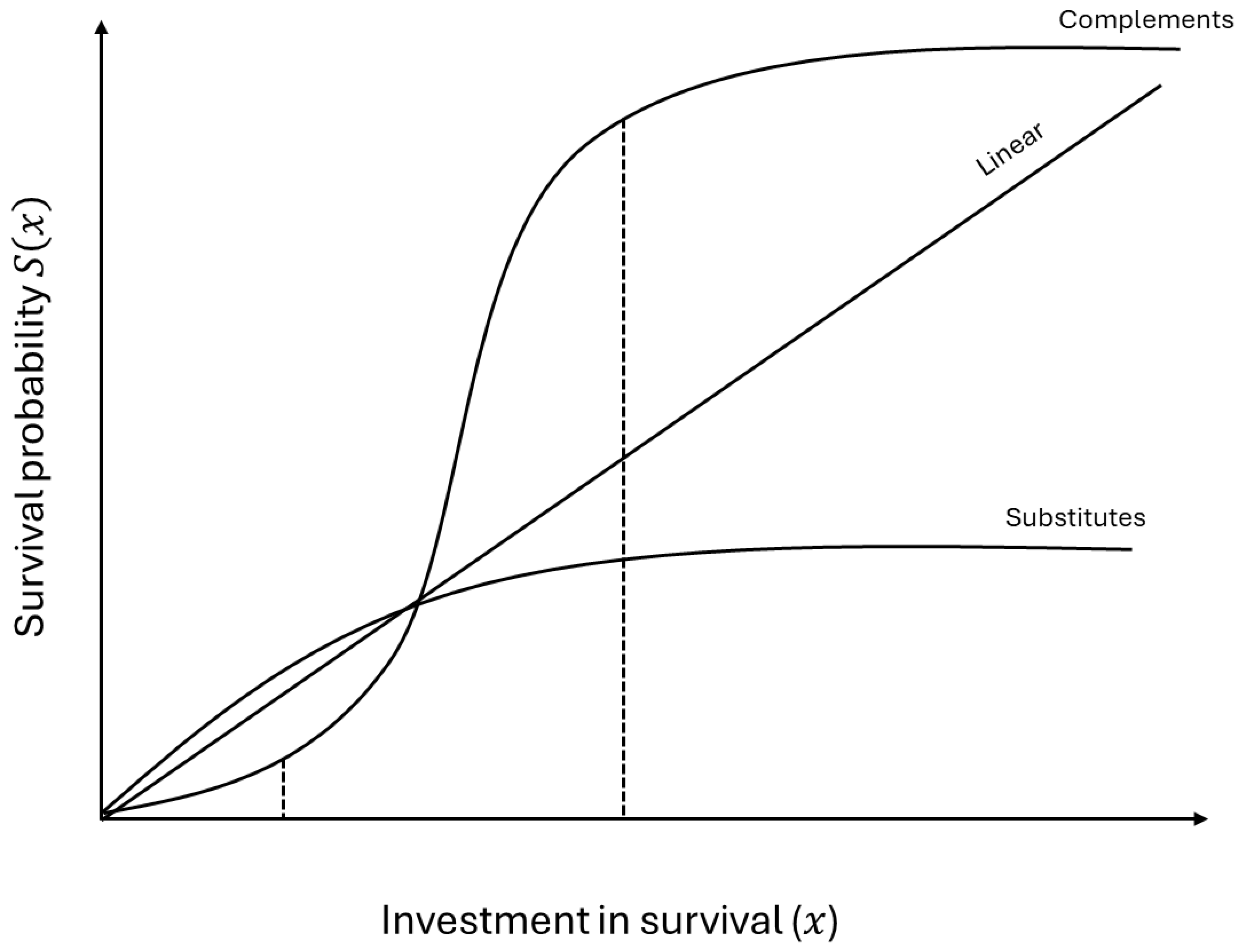
2.1. The Basic Becker-Hamilton Model
2.2. Multiplicative Damage and Survival Capital
2.3. Tradeoffs Between Survival Capital and Reproduction
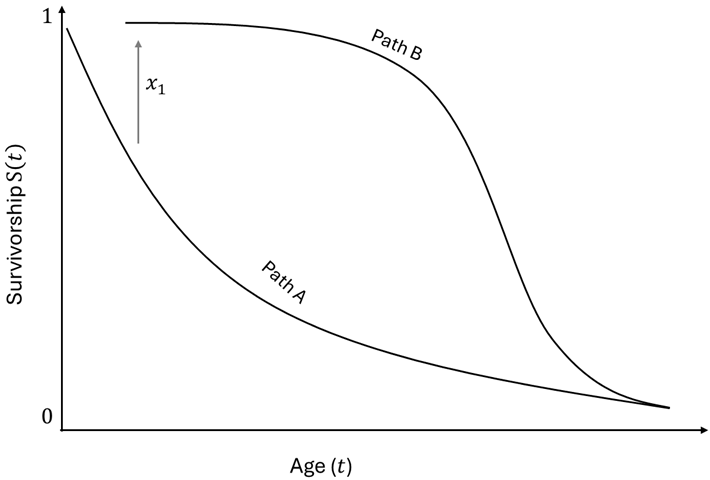
2.4. Multiple Stage Tradeoff Model and Response to Resource Effects
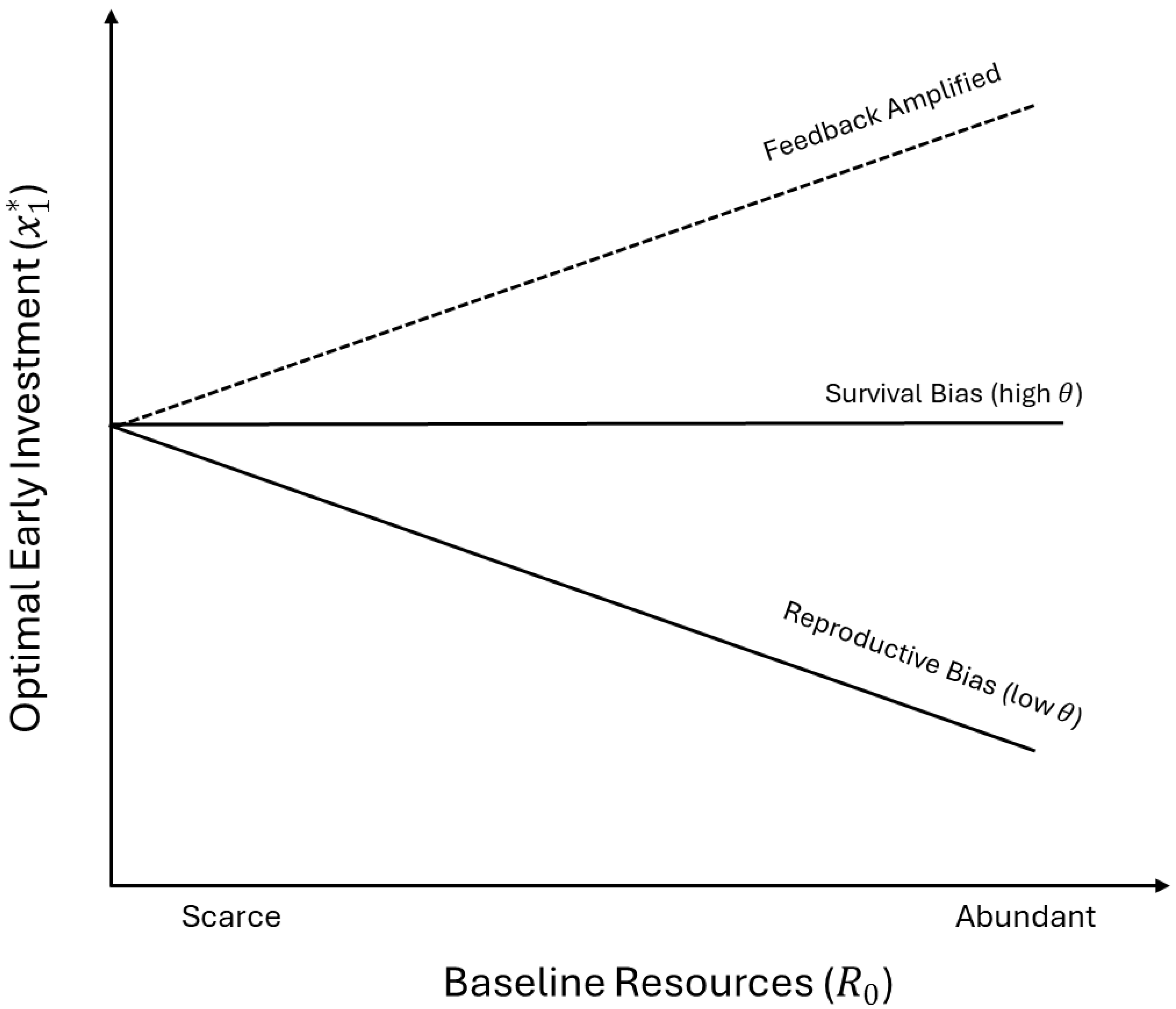
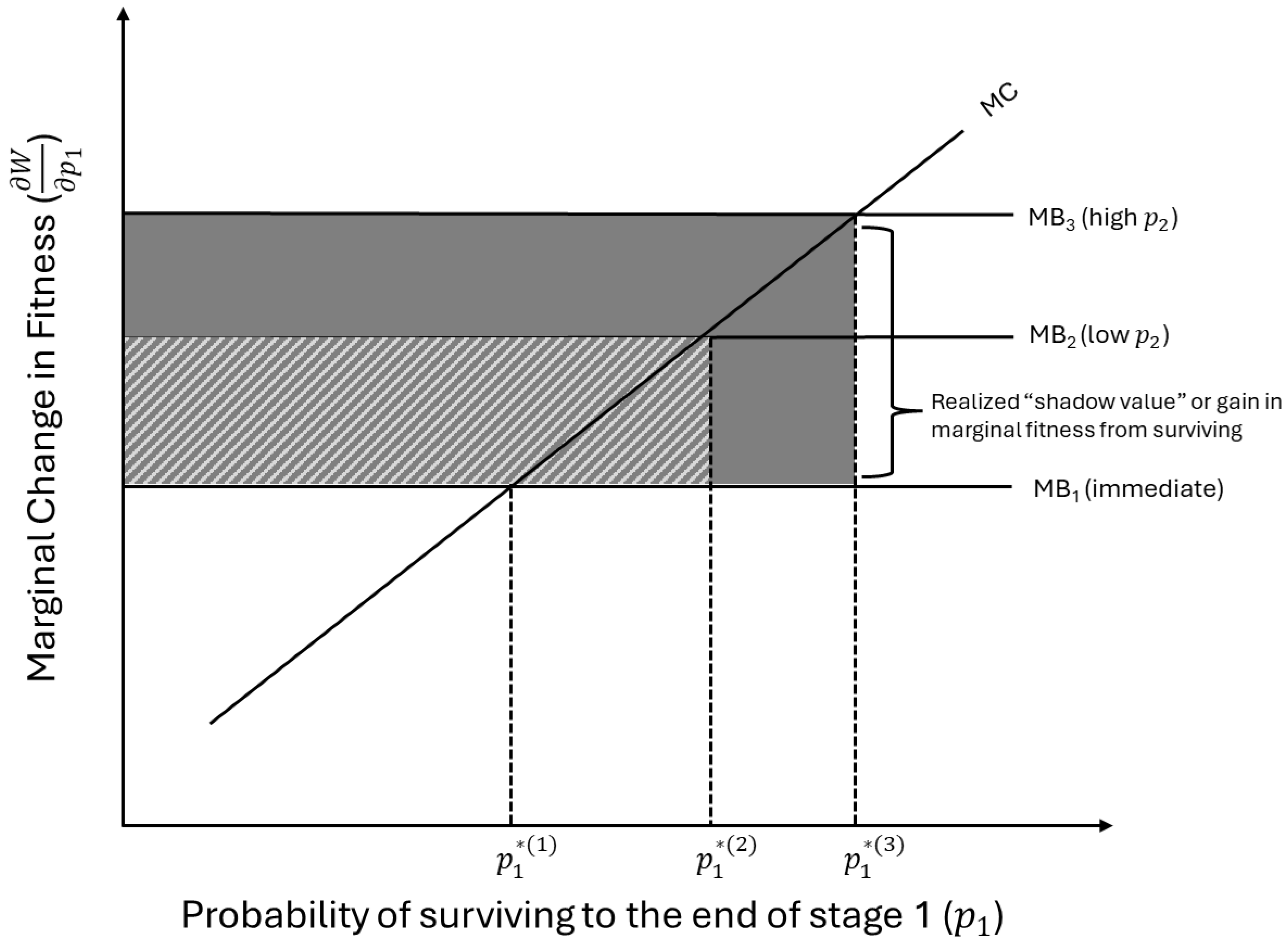
2.5. Endogenizing Stages and Resources
2.6. Endogenizing Stages and Resources
3. Discussion
3.1. Other Theories
3.1.1. Disposable Soma and Antagonistic Pleiotropy
3.1.2. Mutation Accumulation and Constraints
3.1.3. Life History Strategies and Senescence
3.1.4. Exercise and Other Elixirs
3.2. Evidence and Implications
3.2.1. Resource Feedback, Survival Capital, and the Value of Living Longer
3.2.2. Physiological Synergy and Survival Efficacy
3.2.3. Ecological Shifts and Threshold Dynamics
3.3. Limitations and Future Directions
4. Summary and Conclusions
Conflicts of Interest and Funding Declaration
Appendix A: Implicit Differentiation of Early Survival Investment
Appendix B: Linearization and Matrix Derivation of the Stage-Extension Criterion
- Aging as endogenous survival capital, not fixed resource depletion
- Maintenance and reproduction evolve as intertemporal complements
- The > 1 threshold triggers a switch from fast to slow life histories
- Feedback synergies drive the runaway evolution of extreme healthspans
- Provides a theoretical basis for multi-target geroscience
| 1 | While we formalize the evolution of healthspan through the lens of marginal resource allocation, this optimization approach remains logically rooted in population genetics. In this framework, the marginal fitness gains represent the selection coefficient acting on an allele that shifts allocation toward maintenance. We follow the principle that natural selection produces phenotypic designs that maximize fitness subject to trade-offs and constraints (Parker & Maynard Smith, 1990). Under this view, an 'optimum' where marginal benefit equals marginal cost corresponds to a genetic equilibrium where no mutant allele for a different allocation strategy can invade the population (Maynard Smith, 1982). See Grafen (2002, 2006) for the formal correspondence between optimization programs and gene-frequency dynamics via the Price Equation. |
| 2 | Note that L1 is exactly the probability of surviving to age 1 which was p1 in the previous section, and L2 is p1p2. |
| 3 | The use of a script-M () here is intended to signal to the reader that this is a special, model-specific multiplier that encapsulates the product of three interacting forces. It separates the conceptual object “runaway survival capital multiplier” from ordinary variables or constants, reducing ambiguity. |
| 4 | The use of a script-M () here is intended to signal to the reader that this is a special, model-specific multiplier that encapsulates the product of three interacting forces. It separates the conceptual object “runaway survival capital multiplier” from ordinary variables or constants, reducing ambiguity. |
| 5 | This is a concept echoing the "unmaintainability" thresholds explored in recent systems-biology views of aging (Wensink & Cohen, 2022; McAuley, 2025). In those frameworks, senescence is viewed not merely as a consequence of resource scarcity, but as a systemic transition into a state where repair is no longer physically or informationally viable—a constraint that, in our terms, would be represented by a collapse in the survival efficacy parameter as systemic complexity or damage accumulation exceeds a critical limit. orse": an organism may evolve an inherently fragile or "unmaintainable" architecture precisely because its ecological niche disincentivizes long-term survival capital. In this sense, fragility is a designed strategy rather than an unavoidable constraint. A classic example is the Pacific salmon (Oncorhynchus spp.), which undergoes a programmed, systemic collapse—characterized by massive cortisol surges and immune failure—immediately following spawning (Dickhoff, 1989). The salmon does not simply "wear out"; it crosses a programmed unmaintainability threshold because the marginal fitness value of post-reproductive survival has been selected to zero. In short, our view is as consistent with the notion that selection “disincentivises” maintenance beyond a certain age and that complexity is then allows to become fragile or brittle than the reverse direction of causation. |
| 6 | To take a dramatic real-world example, some species of octopus are semelparous, meaning that adults die after a single bout of reproduction. It seems that the act of spawning causes female Octopus hummelincki to undergo a hormonal shift that leads to a rapid deterioration in physical health and then death (Wodinsky, 1977). This involves a sudden drop in neuropeptides and a surge in steroid signalling that causes metabolic collapse (Wang & Ragsdale, 2018). Experimentally preventing this endocrinological change by removing the optic gland can artificially extend their lifespan by several months, effectively switching the organism from a "suicide" program to something of a maintenance regime. |
| 7 | The capacity to maintain genomic stability is a central determinant of aging. Deficient DNA repair systems are tightly linked to the pathology of premature aging syndromes in both human and mouse models, as unrepaired DNA damage triggers cellular senescence and systemic dysfunction (Hoeijmakers, 2009). |
| 8 | This analytical result provides a formal mechanism for the simulation-based findings of Pang (2020), who demonstrated that late-life investment strategies in descendants incur the adaptive evolution of prolonged post-reproductive lifespans. |
| 9 | This logic of "gated" survival may provide a novel explanation for terrestrial gigantism, such as that seen in Mesozoic dinosaurs. While environmental productivity is often cited, the SSC framework suggests a "Size-Refuge" feedback loop: if predators (e.g., theropods) are gape-limited and lack the cooperative hunting strategies of modern mammalian carnivores (e.g., wolves, lions, humans), the survival efficacy of increasing body size becomes extremely high. Once an organism crosses a size threshold that effectively nullifies extrinsic predation, the marginal value of future resources spikes, justifying further massive investment in somatic capital (size and armour) over early reproduction. This creates a runaway selection for gigantism: a strategy that, in our terms, persists until the "multiplier" is disrupted by ecological collapse or the evolution of more efficient, cooperative predatory strategies that lower the survival efficacy of size. (For similar views in the explanation of large sizes among Dinosauria see Codron et al., 2012; Hone & Benton, 2005; Sander et al., 2011). |
References
- Admasu, T. D.; et al. Drug synergy slows aging and improves healthspan through IGF and SREBP lipid signaling. Developmental Cell (2018), 47(1), 67–79. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, A. A.; Laforsch, C.; Tollrian, R. Transgenerational induction of defences in animals and plants. Nature (1999), 401(6748), 60–63. [Google Scholar] [CrossRef]
- Barcena, M. L.; et al. Role of AMPK and sirtuins in aging heart: basic and translational aspects. Aging and Disease (2024), 16(6), 3335. [Google Scholar] [PubMed]
- Becker, G. S. A Theory of the Allocation of Time. The Economic Journal (1965), 75(299), 493–517. [Google Scholar] [CrossRef]
- Becker, G. S. (1975); Human capital: A theoretical and empirical analysis; National Bureau of Economic Research. [Google Scholar]
- Becker, G. S. Human capital. The Concise Encyclopedia of Economics (2002), 2, 1–12. [Google Scholar]
- Bryant, M. J.; Reznick, D. Comparative studies of senescence in natural populations of guppies. The American Naturalist (2004), 163(1), 55–68. [Google Scholar] [CrossRef]
- Bürkle, A.; Beneke, S.; Brabeck, C.; Leake, A.; Meyer, R.; Muiras, M. L.; Pfeiffer, R. Poly (ADP-ribose) polymerase-1, DNA repair and mammalian longevity. Experimental gerontology (2002), 37(10-11), 1203–1205. [Google Scholar] [CrossRef]
- Byars, S. G.; Voskarides, K. Antagonistic pleiotropy in human disease. Journal of Molecular Evolution (2020), 88(1), 12–25. [Google Scholar] [CrossRef]
- Clutton-Brock, T. H.; O'riain, M. J.; Brotherton, P. N.; Gaynor, D.; Kansky, R.; Griffin, A. S.; Manser, M. Selfish sentinels in cooperative mammals. science (1999), 284(5420), 1640–1644. [Google Scholar] [CrossRef]
- Codron, D.; Carbone, C.; Müller, D. W.; Clauss, M. Ontogenetic niche shifts in dinosaurs influenced size, diversity and extinction in terrestrial vertebrates. Biology Letters (2012), 8(4), 620–623. [Google Scholar] [CrossRef]
- Cohen, A. A.; Ferrucci, L.; Fülöp, T.; Gravel, D.; Hao, N.; Kriete, A.; Varadhan, R. A complex systems approach to aging biology. Nature aging (2022), 2(7), 580–591. [Google Scholar] [CrossRef]
- Covas, R. Evolution of reproductive life histories in island birds worldwide. Proceedings of the Royal Society B: Biological Sciences (2012), 279(1733), 1531–1537. [Google Scholar] [CrossRef]
- Dickhoff, W. W. (1989); Salmonids and annual fishes: Death after sex. In ‘‘Development, Maturation, and Senescence of Neuroendocrine Systems: A Comparative Approach’’; Schreibman, MP, Scanes, CG, Eds.; [Google Scholar]
- Fitzpatrick, S. W.; Torres-Dowdall, J.; Reznick, D. N.; Ghalambor, C. K.; Chris Funk, W. Parallelism isn’t perfect: could disease and flooding drive a life-history anomaly in Trinidadian guppies? The American Naturalist (2014), 183(2), 290–300. [Google Scholar] [CrossRef] [PubMed]
- Flatt, T.; Partridge, L. Horizons in the evolution of aging. BMC Biology (2018), 16(1), 93. [Google Scholar] [CrossRef]
- Foster, E. A.; Franks, D. W.; Mazzi, S.; Darden, S. K.; Balcomb, K. C.; Ford, J. K.; Croft, D. P. Adaptive prolonged postreproductive life span in killer whales. Science (2012), 337(6100), 1313–1313. [Google Scholar] [CrossRef]
- French, J. A.; Carp, S. B. Early-life social adversity and developmental processes in nonhuman primates. Current opinion in behavioral sciences (2016), 7, 40–46. [Google Scholar] [CrossRef]
- Froldi, G. View on metformin: antidiabetic and pleiotropic effects. Pharmaceuticals (2024), 17(4), 478. [Google Scholar] [CrossRef]
- Garratt, M.; Lagisz, M.; Staerk, J.; Neyt, C.; Stout, M. B.; Isola, J. V.; Nakagawa, S. Sterilization and contraception increase lifespan across vertebrates. Nature (2025), 1–9. [Google Scholar] [CrossRef]
- Grafen, A. A first formal link between the Price equation and an optimization program. Journal of theoretical biology (2002), 217(1), 75–91. [Google Scholar] [CrossRef] [PubMed]
- Grafen, A. Optimization of inclusive fitness. Journal of theoretical Biology (2006), 238(3), 541–563. [Google Scholar] [CrossRef] [PubMed]
- Griesser, M.; Drobniak, S. M.; Nakagawa, S.; Botero, C. A. Family living sets the stage for cooperative breeding and ecological resilience in birds. PLoS biology (2017), 15(6), e2000483. [Google Scholar] [CrossRef] [PubMed]
- Grossman, M. On the concept of health capital and the demand for health. Journal of Political economy (1972), 80(2), 223–255. [Google Scholar] [CrossRef]
- Hamilton, W. D. The moulding of senescence by natural selection. Journal of theoretical biology (1966), 12(1), 12–45. [Google Scholar] [CrossRef]
- Hamilton, W. D. (1996); Narrow Roads of Gene Land: The Collected Papers of W.D. Hamilton Volume 1: Evolution of Social Behaviour; Oxford University Press: Oxford; Volume 1. [Google Scholar]
- Healy, K.; Guillerme, T.; Finlay, S.; Kane, A.; Kelly, S.; McClean, D.; Cooper, N. Ecology and mode-of-life explain lifespan variation in birds and mammals. Proceedings of the Royal Society B: Biological Sciences (2014), 281, 1784. [Google Scholar] [CrossRef]
- Herzig, S.; Shaw, R. J. AMPK: guardian of metabolism and mitochondrial homeostasis. Nature Reviews Molecular Cell Biology (2018), 19(2), 121–135. [Google Scholar] [CrossRef]
- Hoeijmakers, J. H. DNA damage, aging, and cancer. New England journal of medicine (2009), 361(15), 1475–1485. [Google Scholar] [CrossRef]
- Holekamp, K. E.; Smith, J. E.; Strelioff, C. C.; Van Horn, R. C.; Watts, H. E. Society, demography and genetic structure in the spotted hyena. Molecular Ecology (2012), 21(3), 613–632. [Google Scholar] [CrossRef]
- Holmes, D. J.; Flückiger, R.; Austad, S. N. Comparative biology of aging in birds: an update. Experimental gerontology (2001), 36(4-6), 869–883. [Google Scholar] [CrossRef]
- Hone, D. W.; Benton, M. J. The evolution of large size: how does Cope's Rule work? Trends in ecology & evolution (2005), 20(1), 4–6. [Google Scholar]
- Holt-Lunstad, J.; Smith, T. B.; Layton, J. B. Social relationships and mortality risk: a meta-analytic review. PLoS Medicine (2010), 7(7), e1000316. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, H.; Hill, K.; Lancaster, J.; Hurtado, A. M. A theory of human life history evolution: Diet, intelligence, and longevity. Evolutionary Anthropology (2000), 9(4), 156–185. [Google Scholar] [CrossRef]
- Kirkwood, T. B. Evolution of longevity. Nature (1977), 270, 301–304. [Google Scholar] [CrossRef]
- Kirkwood, T. B.; Holliday, R. The evolution of ageing and longevity. Proceedings of the Royal Society of London. Series B. Biological Sciences (1979), 205(1161), 531–546. [Google Scholar] [CrossRef]
- Lewis, M. A.; Shuai, Z.; van den Driessche, P. A general theory for target reproduction numbers with applications to ecology and epidemiology. Journal of mathematical biology (2019), 78(7), 2317–2339. [Google Scholar] [CrossRef]
- Lieberman, D. E.; Kistner, T. M.; Richard, D.; Lee, I. M.; Baggish, A. L. The active grandparent hypothesis: Physical activity and the evolution of extended human healthspans and lifespans. Proceedings of the National Academy of Sciences (2021), 118(50), e2107621118. [Google Scholar] [CrossRef] [PubMed]
- López-Otín, C.; Blasco, M. A.; Partridge, L.; Serrano, M.; Kroemer, G. The hallmarks of aging. Cell (2013), 153(6), 1194–1217. [Google Scholar] [CrossRef] [PubMed]
- Ma, W.; Zhou, S. Metabolic rewiring in the face of genomic assault: integrating DNA damage response and cellular metabolism. Biomolecules (2025), 15(2), 168. [Google Scholar] [CrossRef]
- Mannick, J. B.; Lamming, D. W. Targeting the biology of aging with mTOR inhibitors. Nature aging (2023), 3(6), 642–660. [Google Scholar] [CrossRef]
- Manzoli, L.; et al. Marital status and mortality in the elderly: a systematic review and meta-analysis. Social Science & Medicine (2007), 64(1), 77–94. [Google Scholar] [CrossRef]
- Maynard Smith, J. (1982); Evolution and the Theory of Games.; Cambridge University Press. [Google Scholar]
- Mc Auley, M. T. The evolution of ageing: classic theories and emerging ideas. Biogerontology (2025), 26(1), 6. [Google Scholar] [CrossRef] [PubMed]
- McComb, K.; Shannon, G.; Durant, S. M.; Sayialel, K.; Slotow, R.; Poole, J.; Moss, C. Leadership in elephants: the adaptive value of age. Proceedings of the Royal Society B: Biological Sciences (2011), 278(1722), 3270–3276. [Google Scholar] [CrossRef]
- Medawar, P. B. An Unsolved Problem of Biology: An inaugural lecture delivered at University College, London, 6 December 1951; H. K. Lewis & Co: London, (1952). [Google Scholar]
- Meng, F.; et al. Synergy between SIRT1 and SIRT6 helps recognize DNA breaks. eLife (2020), 9, e55828. [Google Scholar] [CrossRef]
- Menk, A. V.; et al. 4-1BB costimulation induces T cell mitochondrial function and biogenesis enabling cancer immunotherapeutic responses. Journal of Experimental Medicine (2018), 215(4), 1091–1100. [Google Scholar] [CrossRef]
- Murphy, D.; Mumby, H. S.; Henley, M. D. Age differences in the temporal stability of a male African elephant (Loxodonta africana) social network. Behavioral Ecology (2020), 31(1), 21–31. [Google Scholar] [CrossRef]
- Nattrass, S.; Croft, D. P.; Ellis, S.; Cant, M. A.; Weiss, M. N.; Wright, B. M.; Franks, D. W. Postreproductive killer whale grandmothers improve the survival of their grandoffspring. Proceedings of the National Academy of Sciences (2019), 116(52), 26669–26673. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, J.; Hedeholm, R. B.; Heinemeier, J.; Bushnell, P. G.; Christiansen, J. S.; Olsen, J.; Steffensen, J. F. Eye lens radiocarbon reveals centuries of longevity in the Greenland shark (Somniosus microcephalus). Science (2016), 353(6300), 702–704. [Google Scholar] [CrossRef]
- Pang, T. Y. On age-specific selection and extensive lifespan beyond menopause. Royal Society Open Science (2020), 7(5). [Google Scholar] [CrossRef]
- Parker, G. A.; Maynard Smith, J. Optimality theory in evolutionary biology. Nature (1990), 348(6296), 27–33. [Google Scholar] [CrossRef]
- Parker, J. M.; Wittemyer, G. Orphaning stunts growth in wild African elephants. Conservation Physiology (2022), 10(1), coac053. [Google Scholar] [CrossRef]
- Reznick, D. Costs of reproduction: an evaluation of the empirical evidence. Oikos (1985), 257–267. [Google Scholar] [CrossRef]
- Reznick, D. A.; Bryga, H.; Endler, J. A. Experimentally induced life-history evolution in a natural population. Nature (1990), 346(6282), 357–359. [Google Scholar] [CrossRef]
- Reznick, D.; Butler, M. J., IV; Rodd, H. Life-history evolution in guppies. VII. The comparative ecology of high-and low-predation environments. The American Naturalist (2001), 157(2), 126–140. [Google Scholar] [CrossRef]
- Roy, D. (2026); Investment Cascades and Fitness Capital; Preprints. [Google Scholar] [CrossRef]
- Riessen, H. P. Predator-induced life history shifts in Daphnia: a synthesis of studies using meta-analysis. Canadian Journal of Fisheries and Aquatic Sciences (1999), 56(12), 2487–2494. [Google Scholar] [CrossRef]
- Sander, P. M.; Christian, A.; Clauss, M.; Fechner, R.; Gee, C. T.; Griebeler, E. M.; Witzel, U. Biology of the sauropod dinosaurs: the evolution of gigantism. Biological Reviews (2011), 86(1), 117–155. [Google Scholar] [CrossRef]
- Simpson, S. J.; Raubenheimer, D. (2012); The nature of nutrition: a unifying framework from animal adaptation to human obesity; Princeton university press. [Google Scholar]
- Silk, J. B. The adaptive value of sociality in mammalian groups. Philosophical Transactions of the Royal Society B: Biological Sciences (2007), 362(1480), 539–559. [Google Scholar] [CrossRef]
- Smeele, S. Q.; Conde, D. A.; Baudisch, A.; Bruslund, S.; Iwaniuk, A.; Staerk, J.; Aplin, L. Coevolution of relative brain size and life expectancy in parrots. Proceedings of the Royal Society B (2022), 289(1971), 20212397. [Google Scholar] [CrossRef]
- Stearns, S. C. Trade-offs in life-history evolution. Functional ecology (1989), 3(3), 259–268. [Google Scholar] [CrossRef]
- Stott, I.; Salguero-Gómez, R.; Jones, O. R.; Ezard, T. H.; Gamelon, M.; Lachish, S.; Hodgson, D. J. Life histories are not just fast or slow. Trends in Ecology & Evolution (2024), 39(9), 830–840. [Google Scholar] [CrossRef] [PubMed]
- Vijg, J.; Campisi, J. Puzzles, promises and a cure for ageing. Nature (2008), 454(7208), 1065–1071. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z. Y.; Ragsdale, C. W. Multiple optic gland signaling pathways implicated in octopus maternal behaviors and death. Journal of Experimental Biology (2018), 221(19), jeb185751. [Google Scholar] [CrossRef] [PubMed]
- Wensink, M. J.; Cohen, A. A. The Danaid theory of aging. Frontiers in cell and developmental biology (2022), 9, 671208. [Google Scholar] [CrossRef] [PubMed]
- Werner, C.; et al. Differential effects of endurance, interval, and resistance training on telomerase activity and telomere length among 266 healthy adults. European Heart Journal (2019), 40(1), 34–46. [Google Scholar] [CrossRef] [PubMed]
- Wensink, M. J.; et al. The maintenance gap: a new theoretical perspective on the evolution of aging. Biogerontology (2012), 13(2), 197–201. [Google Scholar] [CrossRef]
- Wilkinson, G. S.; Adams, D. M. Recurrent evolution of extreme longevity in bats. Biology letters (2019), 15(4), 20180860. [Google Scholar] [CrossRef]
- Williams, G. C. Pleiotropy, Natural Selection, and the Evolution of Senescence. Evolution. (1957), 11(4), 398–411. [Google Scholar] [CrossRef]
- Williams, G. C. (1966); Adaptation and Natural Selection; Princeton University Press: Princeton, NJ. [Google Scholar]
- Wodinsky, J. Hormonal inhibition of feeding and death in octopus: control by optic gland secretion. Science (1977), 198(4320), 948–951. [Google Scholar] [CrossRef]
- Zera, A. J.; Harshman, L. G. The physiology of life history trade-offs in animals. Annual review of Ecology and Systematics (2001), 32(1), 95–126. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



