Submitted:
25 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
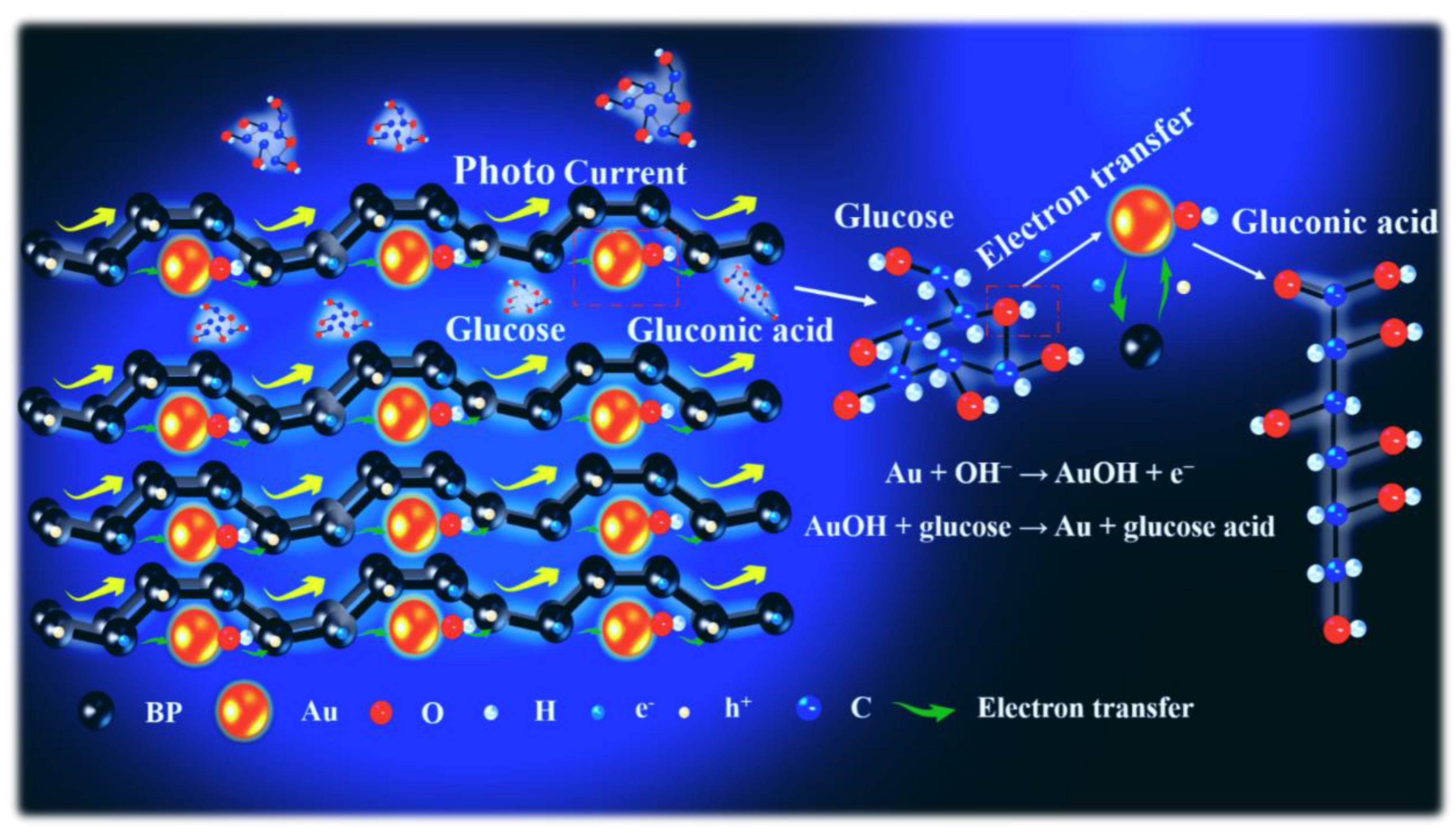
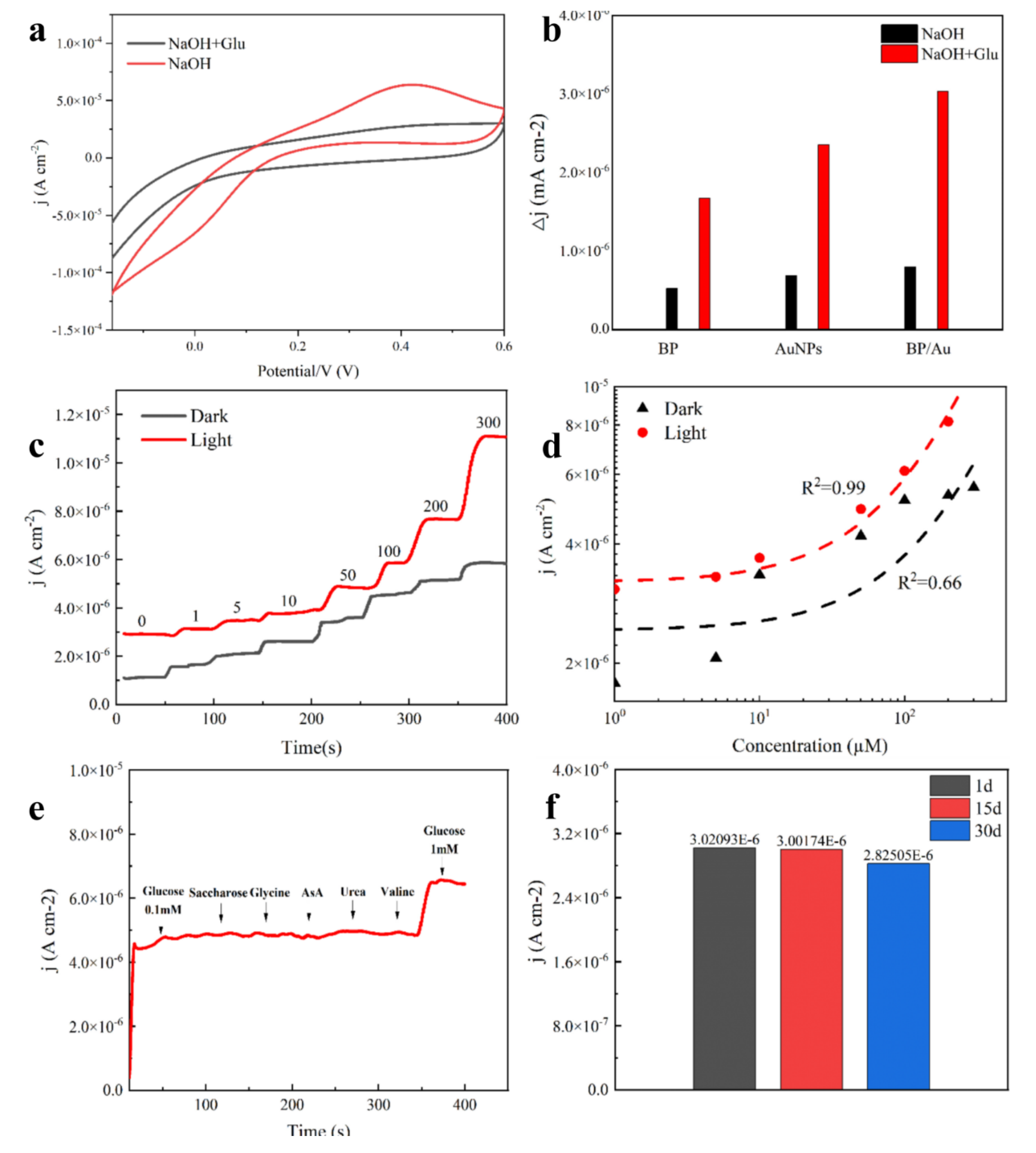
Although noninvasive glucose monitoring in sweat is a promising, pain-free method for diabetes management, it requires highly sensitive and stable sensors to overcome practical limitations. To overcome this challenge, a photoelectrochemical sensor based on a plasmon-enhanced black phosphorus (BP)/gold (Au) heterojunction was developed in this study. BP nanosheets possess a unique layered structure and intrinsic catalytic activity, but their instability and limited efficiency hinder direct use. Therefore, BP/Au was synthesized using the one-pot method. First-principles calculations revealed that single-layer BP behaved as a quasi-direct bandgap semiconductor. In comparison, the BP/Au heterojunction exhibited metallic characteristics, with anisotropic electron mobility reaching 1.62 cm2·V−1·s−1 along one direction. Charge density analysis confirmed directional charge transfer. Au donated electrons to adjacent P atoms, whereas P atoms forming shorter bonds lost charge. This process was associated with plasmon-assisted photoexcitation at the Au/BP interface, which modulated interfacial charge distribution and enhanced photoelectrochemical activity. By leveraging the Au component’s surface plasmon resonance, the heterojunction considerably augmented light absorption, accelerated interfacial electron transfer, and utilized the wrinkled BP layers to provide abundant active sites. This synergistic effect substantially lowered the oxidation activation energy of glucose. The resulting sensor achieved exceptional performance, with a sensitivity of 266.9 μA·μM−1·cm−2, a low detection limit, and a wide linear range well-suited for detecting glucose in sweat. The findings emphasized the potential of plasmon–semiconductor coupling for advancing noninvasive glucose monitoring and provided valuable design principles for sweat sensors based on metal–semiconductor heterojunctions.
Keywords:
1. Introduction
2. Experimental
2.1. Materials
2.2. Fabrication of the BP/Au Nanosheet Modified Electrode
2.3. Physicochemical Characterization
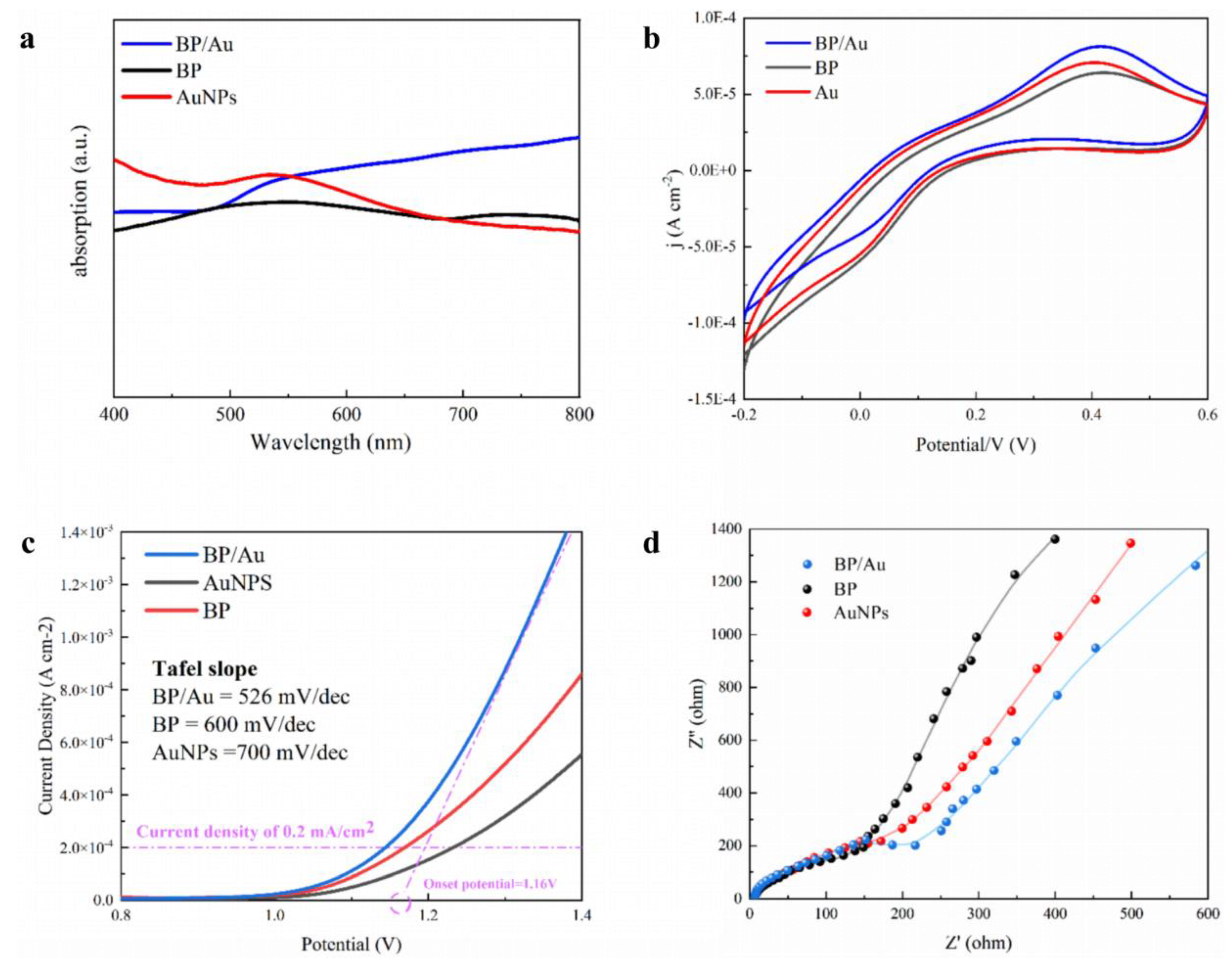
3. Results and Discussion
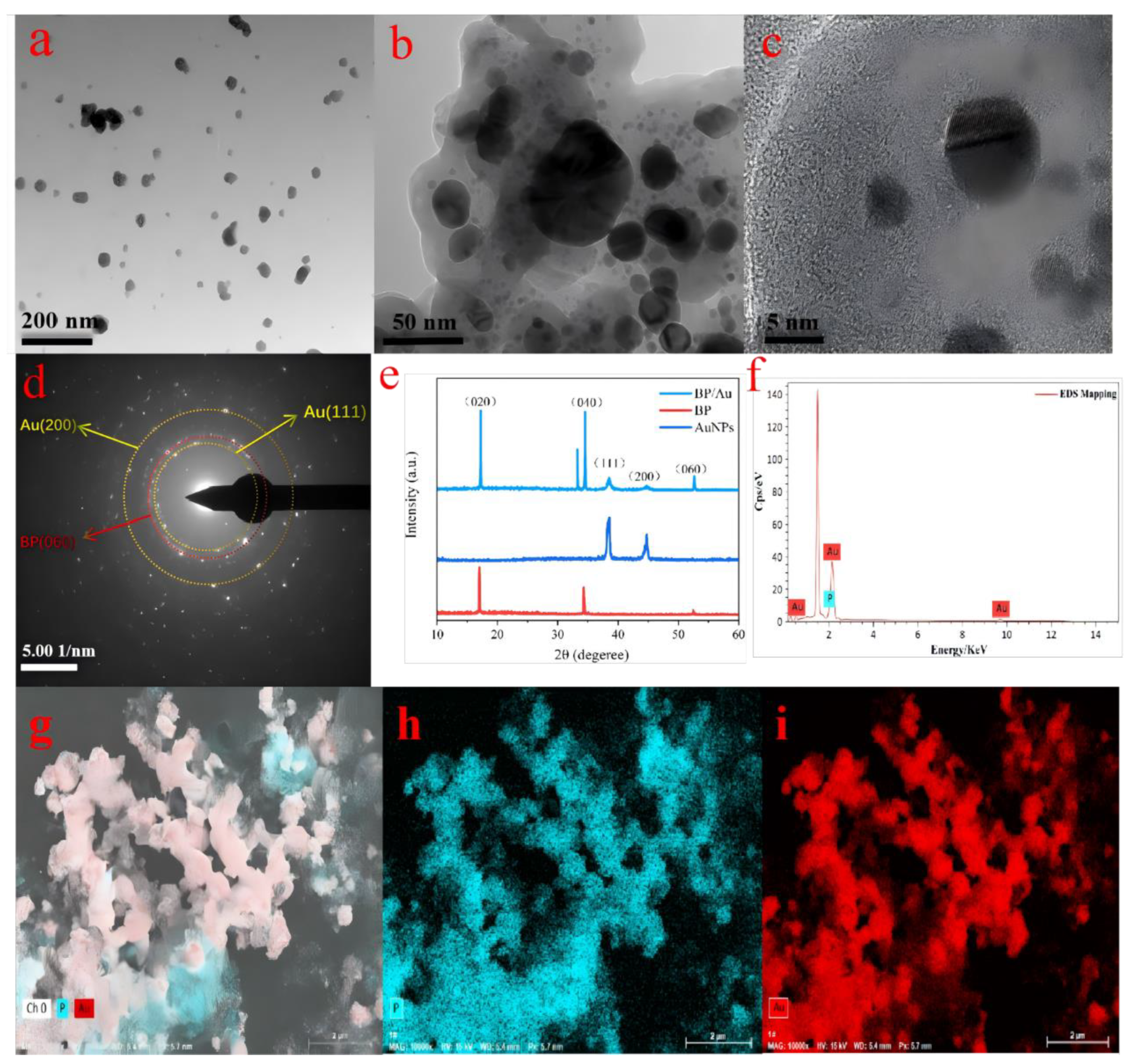
3.1. Morphological and Structural Characterization
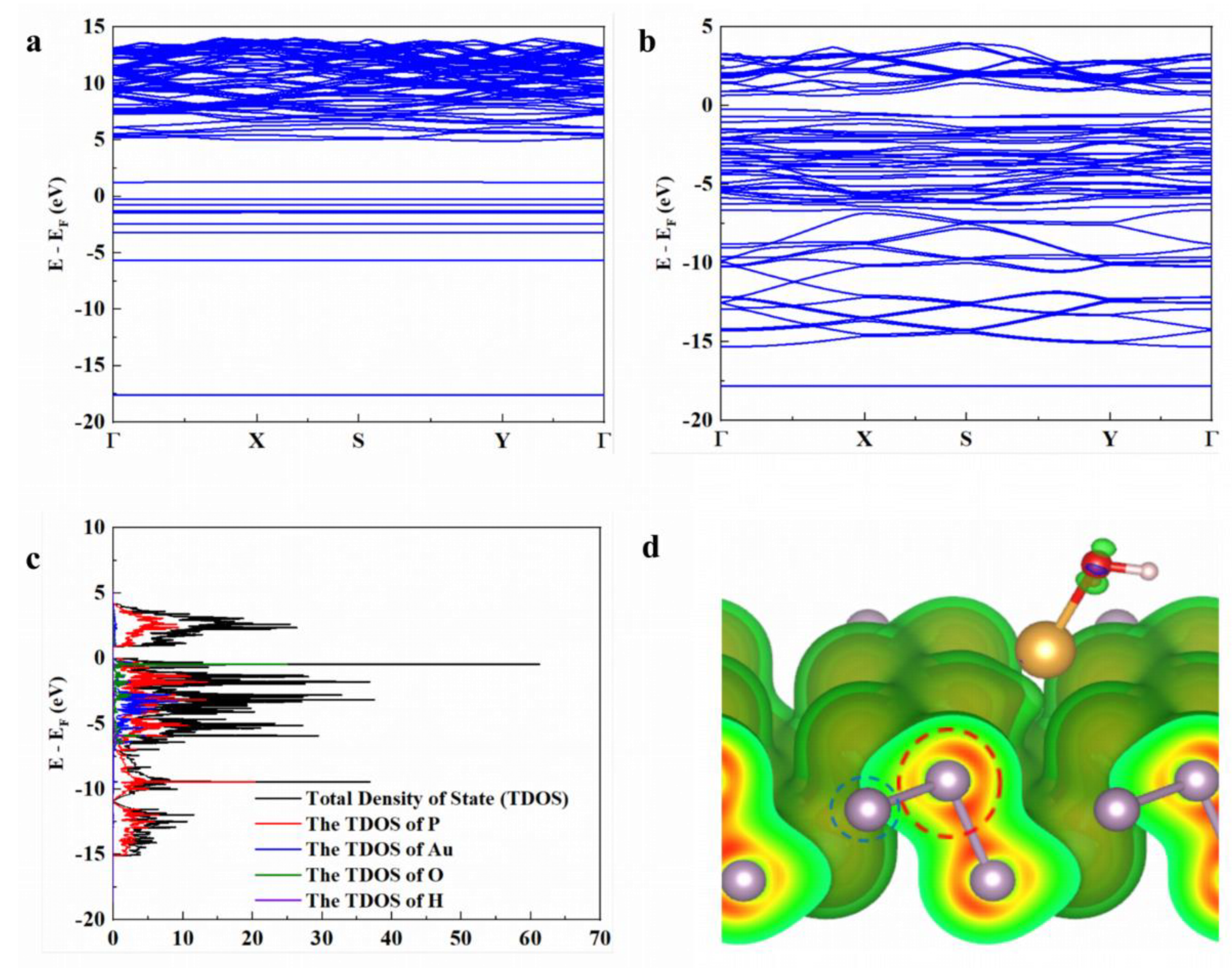
3.2. First-Principles Calculations
3.2.1. Computational Details
3.2.2. Atomic Model Construction
3.2.3. Band Structure
3.2.4. DOS
3.2.5. Charge Density Difference
4. Conclusions
Acknowledgments
References
- Ankit, B., et al., Sensitive electrochemical detection of glucose via a hybrid self-powered biosensing system. 2018. 20: p. 41-46.
- Research, A.J.D. and C. Practice, Over 250 million people worldwide unaware they have diabetes, according to new research from the International Diabetes Federation (IDF). 2025. 223(000).
- Global, regional, and national burden of diabetes from 1990 to 2021, with projections of prevalence to 2050: a systematic analysis for the Global Burden of Disease Study 2021. Lancet, 2023. 402(10397): p. 203-234.
- Witkowska Nery, E., et al., Electrochemical Glucose Sensing: Is There Still Room for Improvement? Anal Chem, 2016. 88(23): p. 11271-11282.
- Li, J., Z. Lou, and B.J.J.o.M.C.A. Li. Engineering plasmonic semiconductors for enhanced photocatalysis.
- Chen, L.-C., et al., Improving the reproducibility, accuracy, and stability of an electrochemical biosensor platform for point-of-care use. 2020. 155: p. 112111.
- Adeel, M., et al., Recent advances of electrochemical and optical enzyme-free glucose sensors operating at physiological conditions. Biosens Bioelectron, 2020. 165: p. 112331.
- Park, S., H. Boo, and T.D.J.A.c.a. Chung, Electrochemical non-enzymatic glucose sensors. 2006. 556(1): p. 46-57.
- van Enter, B.J. and E. von Hauff, Challenges and perspectives in continuous glucose monitoring. Chem Commun (Camb), 2018. 54(40): p. 5032-5045.
- Bariya, M., H.Y.Y. Nyein, and A.J.N.E. Javey, Wearable sweat sensors. 2018. 1(3): p. 160-171.
- Mohan, A.V., et al., Recent advances and perspectives in sweat based wearable electrochemical sensors. 2020. 131: p. 116024.
- Moyer, J., et al., Correlation between sweat glucose and blood glucose in subjects with diabetes. Diabetes Technol Ther, 2012. 14(5): p. 398-402.
- Saeloo, B., et al., Role of transition metal dichalcogenides as a catalyst support for decorating gold nanoparticles for enhanced hydrogen evolution reaction. 2024. 63(40): p. 18750-18762.
- Mu, X., J. Wang, and M.J.M.T. Sun, Two-Dimensional Black Phosphorus: Physical Properties and Applications. 2019.
- Peng, J., et al., Sensitive Detection of Carcinoembryonic Antigen Using Stability-Limited Few-Layer Black Phosphorus as an Electron Donor and a Reservoir. 2017. 13(15): p. 1-11.
- Black Phosphorus Nanosheets Modified with Au Nanoparticles as High Conductivity and High Activity Electrocatalyst for Oxygen Evolution Reaction %J Advanced Energy Materials. 2020. 10(44).
- He, C., et al., Noble metal construction for electrochemical nonenzymatic glucose detection. 2023. 8(1): p. 2200272.
- Lee, S.-H., et al., Direct observation of plasmon-induced interfacial charge separation in metal/semiconductor hybrid nanostructures by measuring surface potentials. 2018. 18(1): p. 109-116.
- Huang, Y., et al., Degradation of Black Phosphorus (BP): The Role of Oxygen and Water. 2015.
- Nicolosi, V., et al., Liquid Exfoliation of Layered Materials. 2013. 340(6139): p. 1420.
- Rohiman, A., et al., Study of Colloidal Gold Synthesis Using Turkevich Method. 2011.
- Chen, Z., et al., Development of Screen-Printable Nafion Dispersion for Electrochemical Sensor. 2022. 12(13): p. 6533.
- Yi, Y., et al., Two-dimensional black phosphorus: Synthesis, modification, properties, and applications. 2017. 120: p. 1-33.
- Ghobadi, T.G.U., et al., Strategies for Plasmonic Hot-Electron-Driven Photoelectrochemical Water Splitting. 2018. 2.
- Ding, Y., et al., A novel NiO–Au hybrid nanobelts based sensor for sensitive and selective glucose detection. 2011. 28(1): p. 393-398.
- Kresse, G. and J.J.C.m.s. Furthmüller, Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. 1996. 6(1): p. 15-50.
- Kresse, G. and D.J.P.R.B. Joubert, From ultrasoft pseudopotentials to the projector augmented-wave method. 1999. 59(3): p. 1758-1775.
- Wang, V., et al., VASPKIT: A user-friendly interface facilitating high-throughput computing and analysis using VASP code. Computer Physics Communications, 2021. 267: p. 108033.
- Blöchl, P.E., Projector augmented-wave method. Physical Review B, 1994. 50(24): p. 17953-17979.
- Zhang, R., B. Li, and J.J.T.J.o.P.C.C. Yang, A first-principles study on electron donor and acceptor molecules adsorbed on phosphorene. 2015. 119(5): p. 2871-2878.
- Liu, Y., et al., Van der Waals heterostructures and devices. 2016. 1(9): p. 1-17.
- Kumar, S., K. Ojha, and A.K.J.A.M.I. Ganguli, Interfacial charge transfer in photoelectrochemical processes. 2017. 4(7): p. 1600981.
- Wei, C. and Z.J.J.S.M. Xu, The comprehensive understanding of as an evaluation parameter for electrochemical water splitting. 2018, Wiley Online Library. p. 1800168.
- Retter, U. and H. Lohse, Electrochemical impedance spectroscopy, in Electroanalytical Methods: Guide to experiments and applications. 2009, Springer. p. 159-177.
- Lavín, Á., et al., On the determination of uncertainty and limit of detection in label-free biosensors. 2018. 18(7): p. 2038.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



