Submitted:
25 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Inhibitor and Solution Preparation
2.3. Gravimetric (Weight Loss) Measurements
2.4. Studies on Electrochemistry
2.5. Surface Characterization
2.5.1. X-Ray Photoelectron Spectroscopy (XPS):
2.5.2. SEM and AFM
2.5.3. Surface Wettability (Contact Angle) Measurements:
2.6. Quantum Chemical Studies
2.6.1. Theoretical Computations
2.6.2. Molecular Simulation (MD) Dynamics
2.6.3. NBO Analysis
3. Results and Discussion
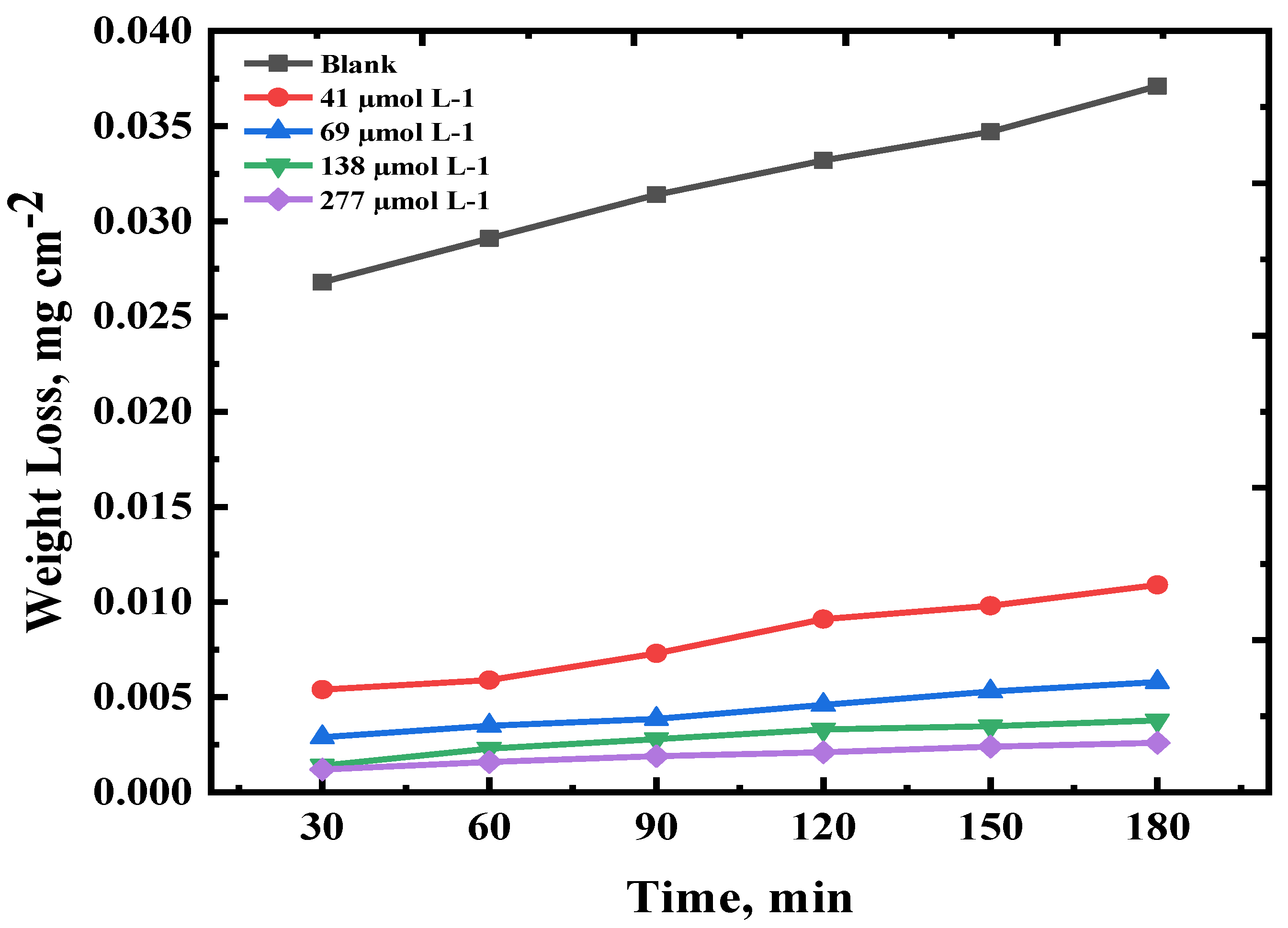
3.1. Gravimetric (Weight Loss) Measurements
3.2. Electrochemical Measurements
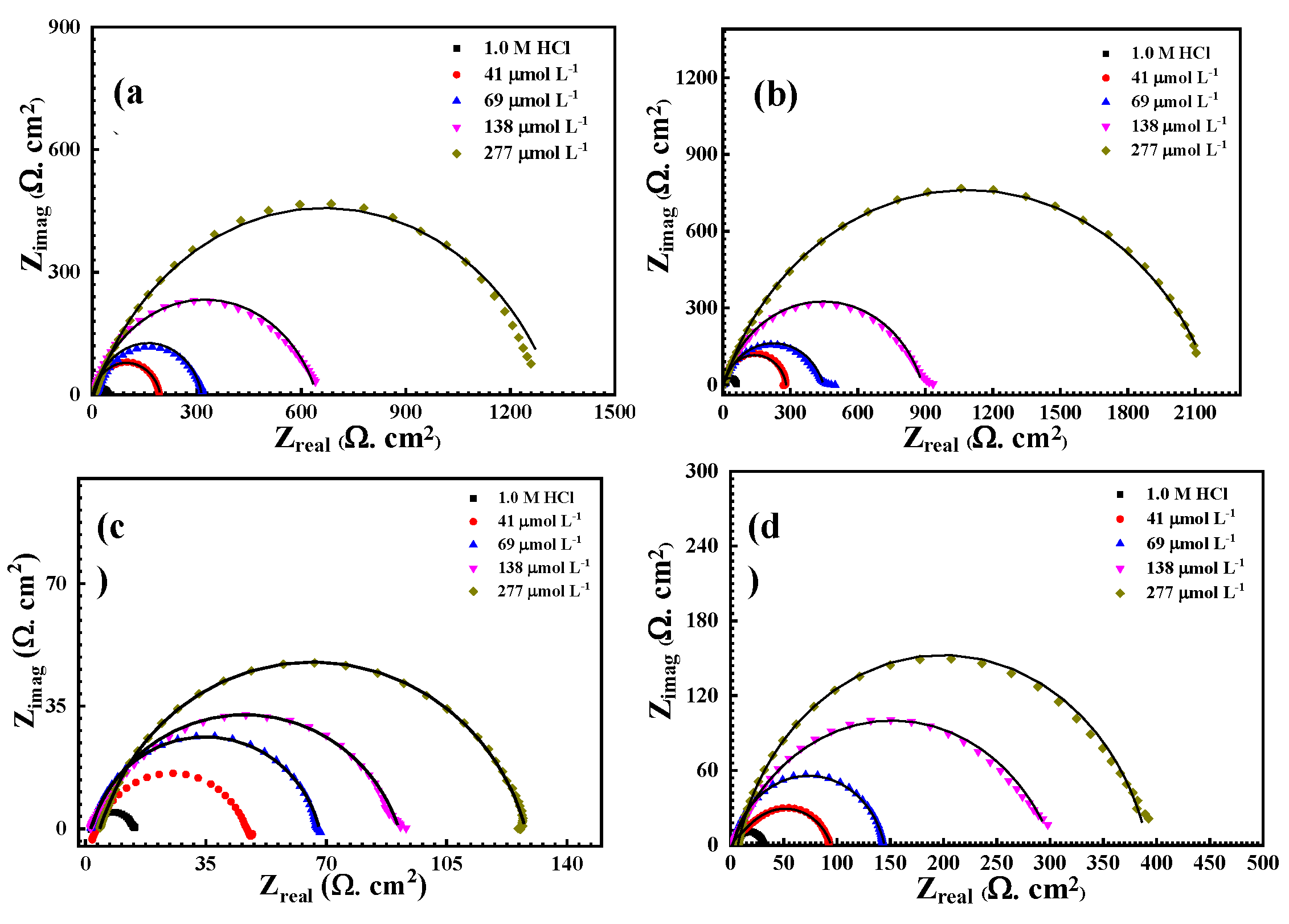
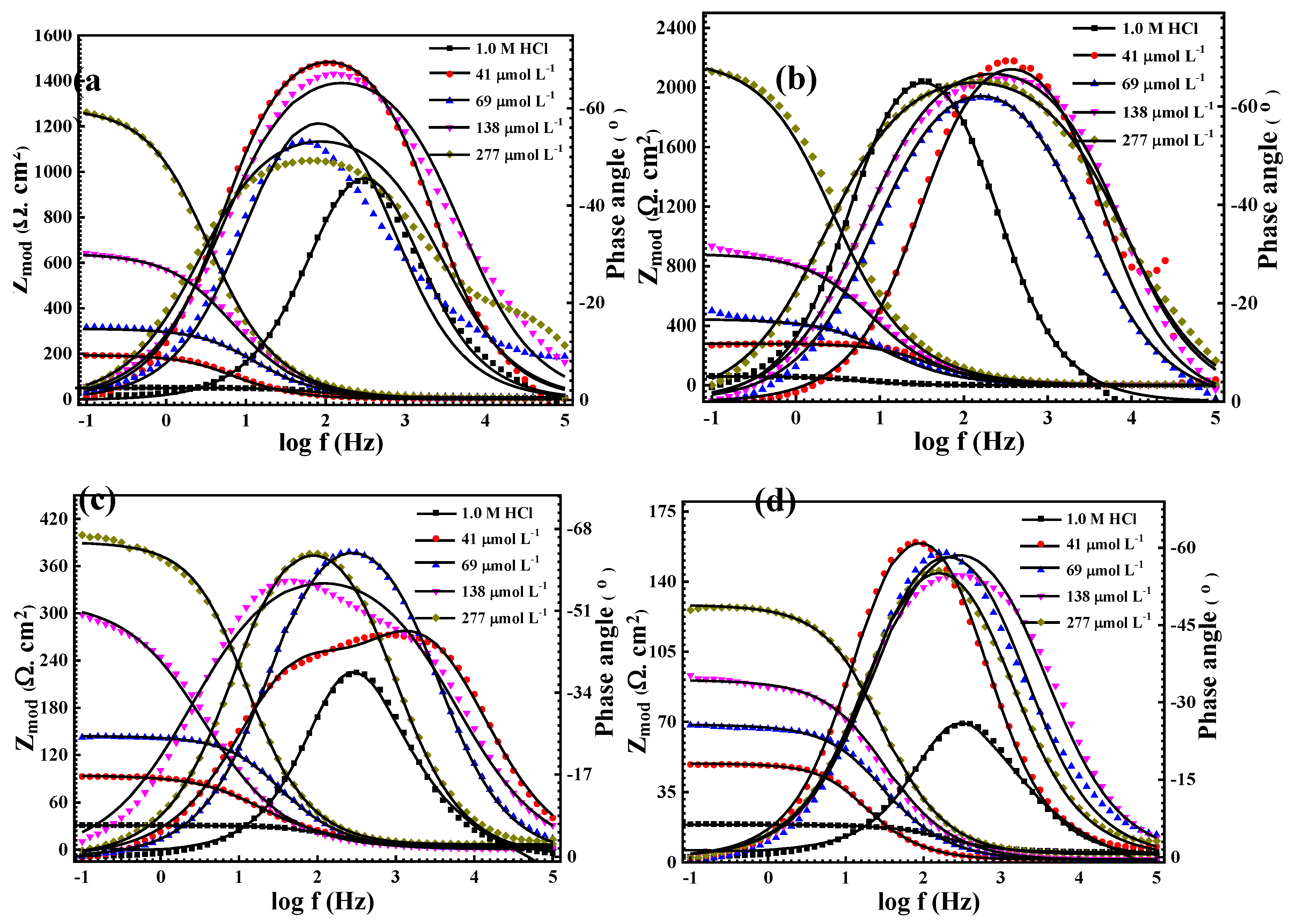
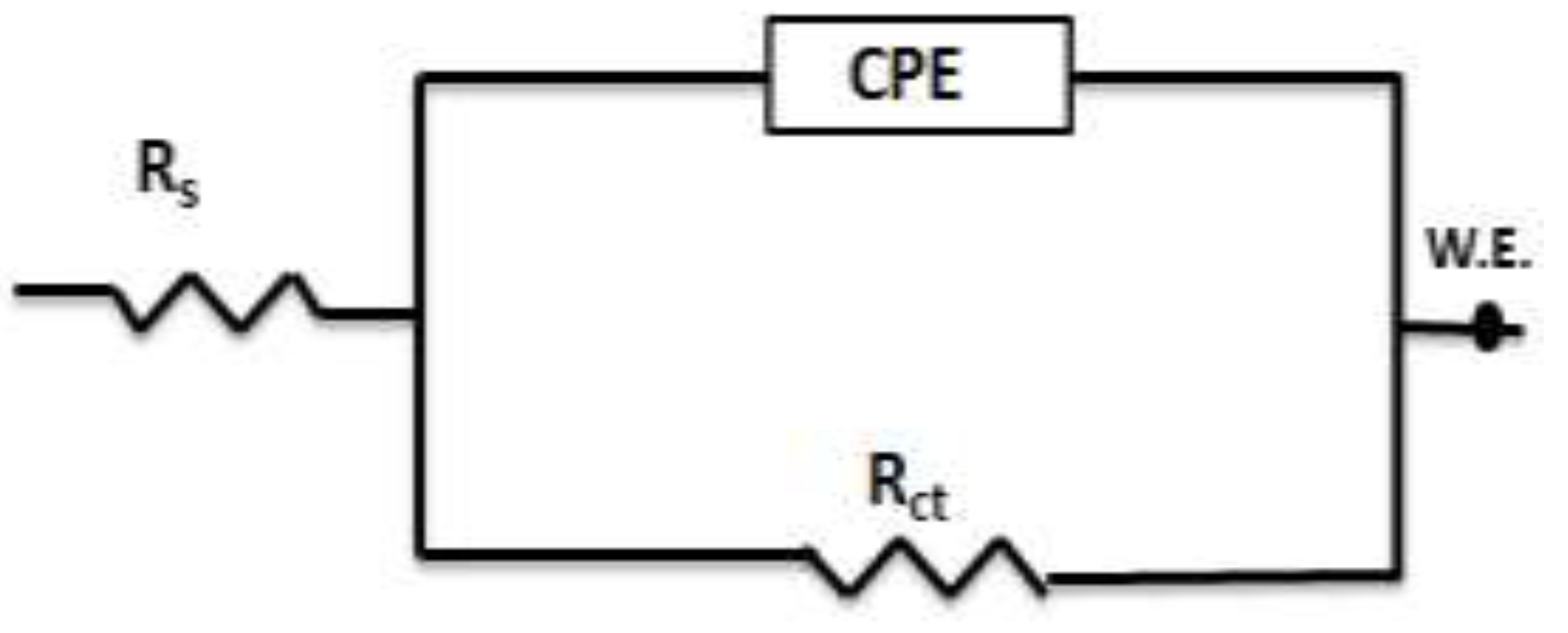
3.2.1. Electrochemical Impedance Spectroscopy (EIS) Measurements
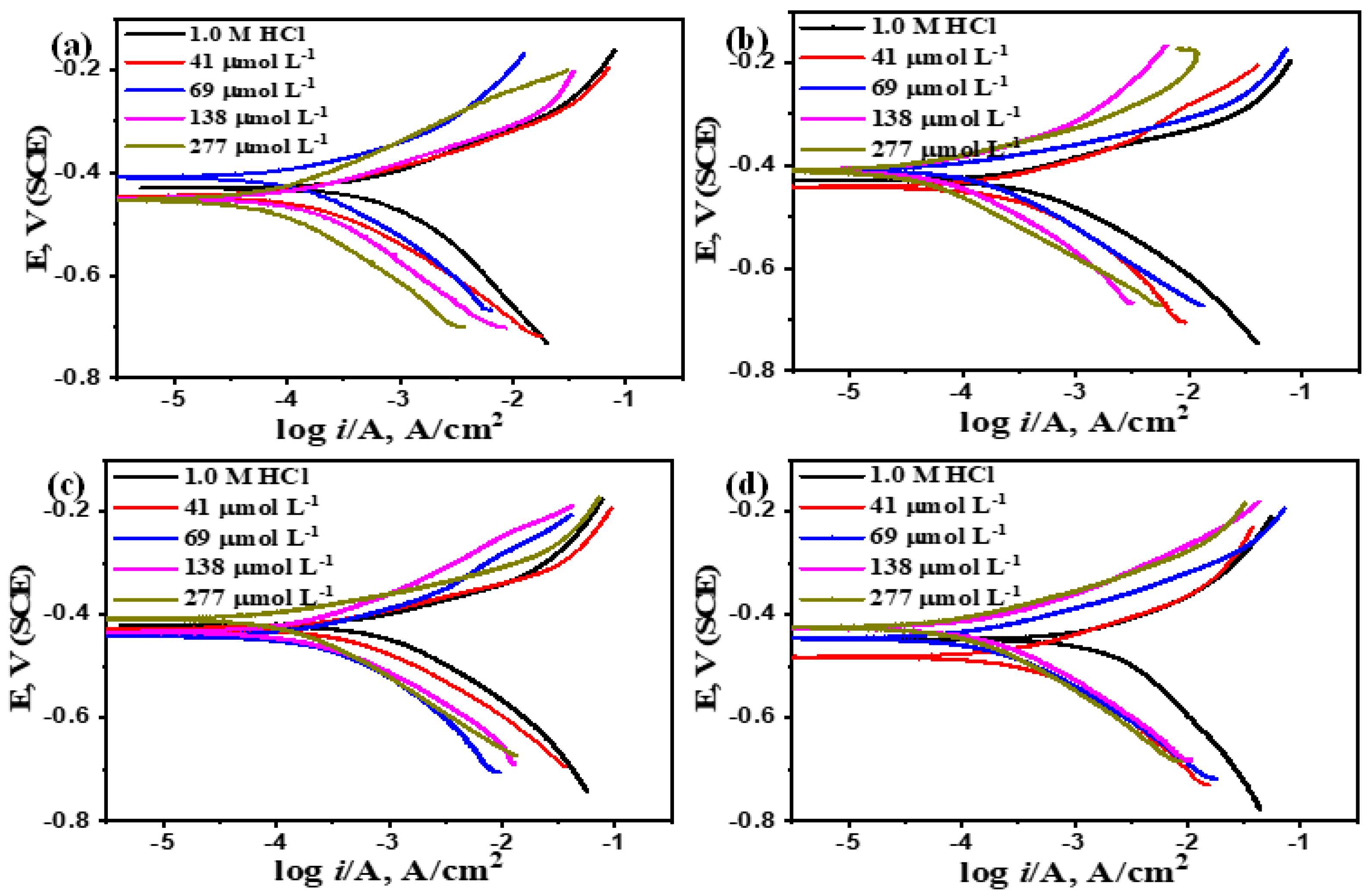
3.2.2. Potentiodynamic Polarization
- I.
- The obtained cathodic and anodic polarization curves exhibit Tafel-type behavior. Addition of the investigated inhibitors to the corrosive acid medium decreases the anodic polarization current more than the cathodic one at all temperatures (mixed control) with a concomitant shift of Ecorr towards positive values. The pronounced positive shift in Ecorr values and the greater suppression of the anodic branch classify Q-22 as a mixed-type inhibitor with a predominant anodic effect [31,32], which preferentially restrains the anodic reaction of CS corrosion in HCl solution [33]. A shift in Ecorr greater than 85 mV is commonly used as a benchmark to categorize inhibitors as either anodic or cathodic [34]. Therefore, despite the anodic predominance, both anodic and cathodic reactions are inhibited.
- II.
- The corrosion current density (icorr.) for CS in 1.0 M HCl solution decreases with increasing inhibitor concentration (Q-22), which indicates that the presence of these compounds retards the dissolution of CS in 1.0 M HCl solution, and the degree of inhibition depends on both concentration and temperature of the corrosive medium. A maximum inhibition efficiency of 97.21% was achieved at the optimal concentration of 277 µmol L⁻¹. This concentration-dependent behavior is characteristic of adsorption-based inhibitors forming a protective layer on the metal surface [35]. However, a temperature-dependent decrease in efficiency at fixed concentrations suggests thermal desorption of the inhibitor, compromising the protective layer [36]. The inhibition efficiencies calculated from potentiodynamic polarization align closely with gravimetric measurements.
- III.
- The slopes of the cathodic and anodic Tafel curves (βc and βa) are slightly changed on increasing the concentration of the tested compound. This indicates that the fundamental corrosion reaction mechanism is unaltered. The fact that the values of βc are slightly higher than βa suggests that the inhibitive action of this inhibitor is by blocking the active sites on the CS surface, forming a barrier film without changing the reaction kinetics and consequently decreasing the surface area available for the electrochemical reactions [37,38].
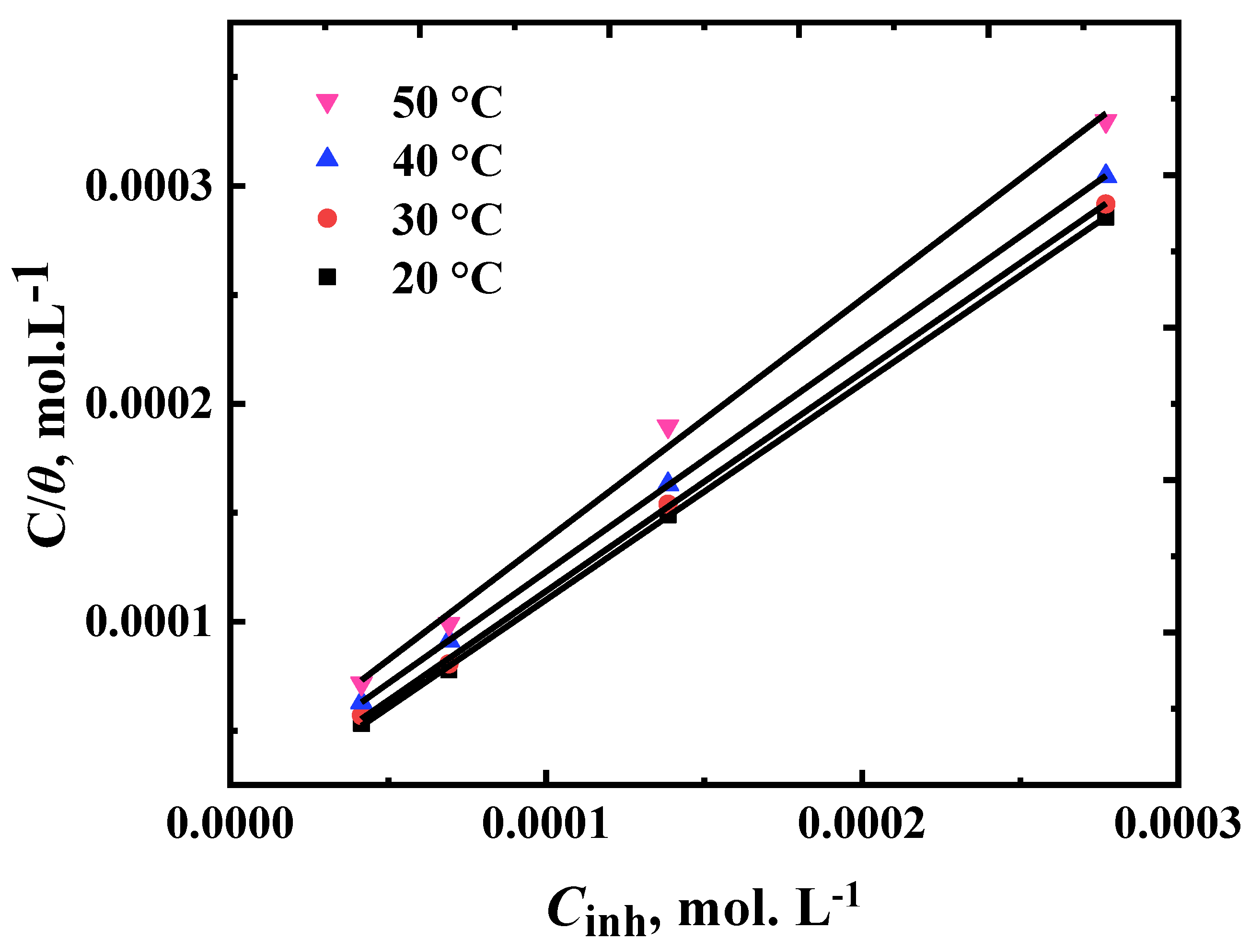
3.3. Adsorption Studies and Thermodynamic Isotherm
| f(θ,x)exp(-a,θ)=KCinh | (10) |
| Kads = 1/55.5 exp (-ΔG°ads. / RT) | (11) |
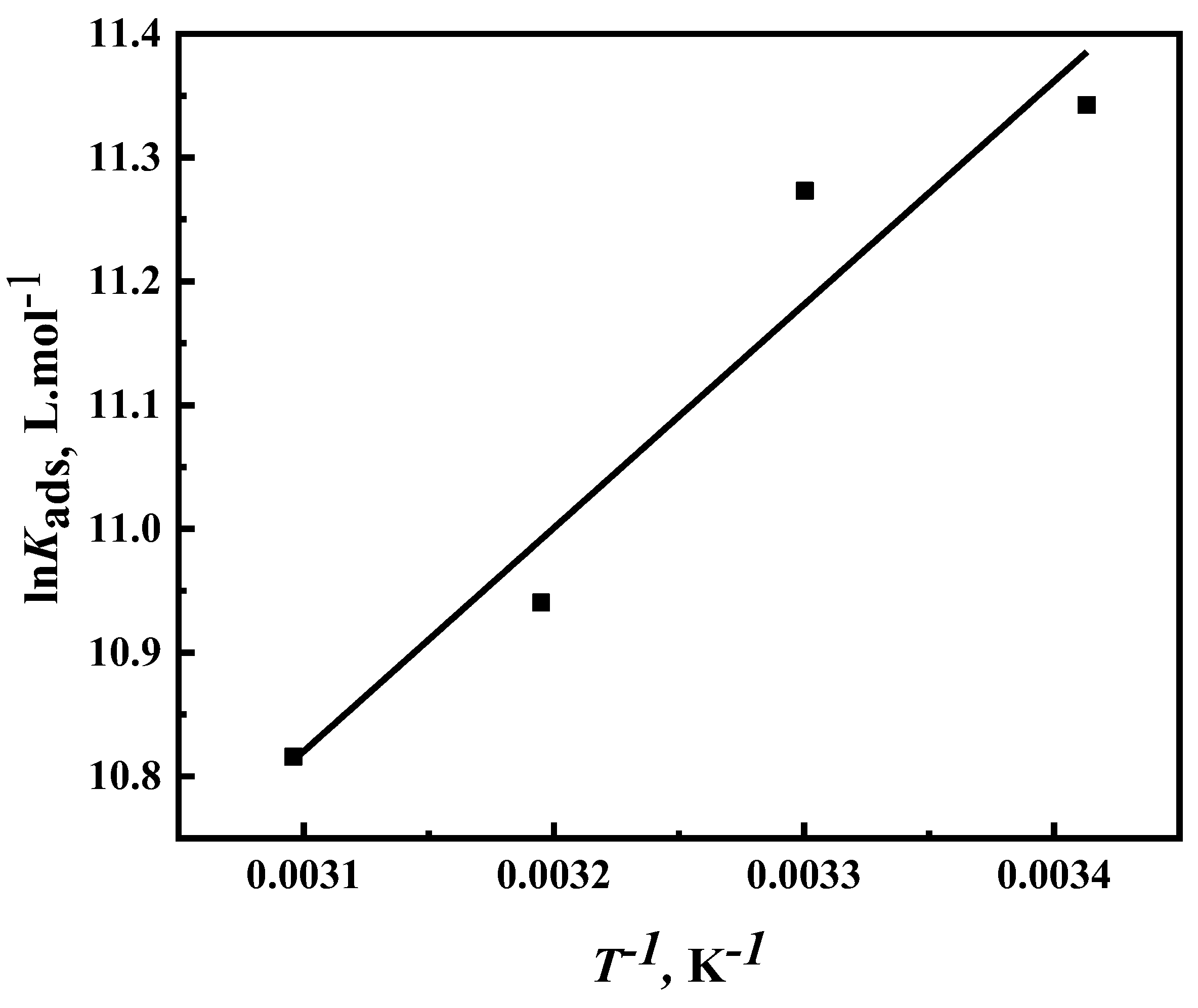
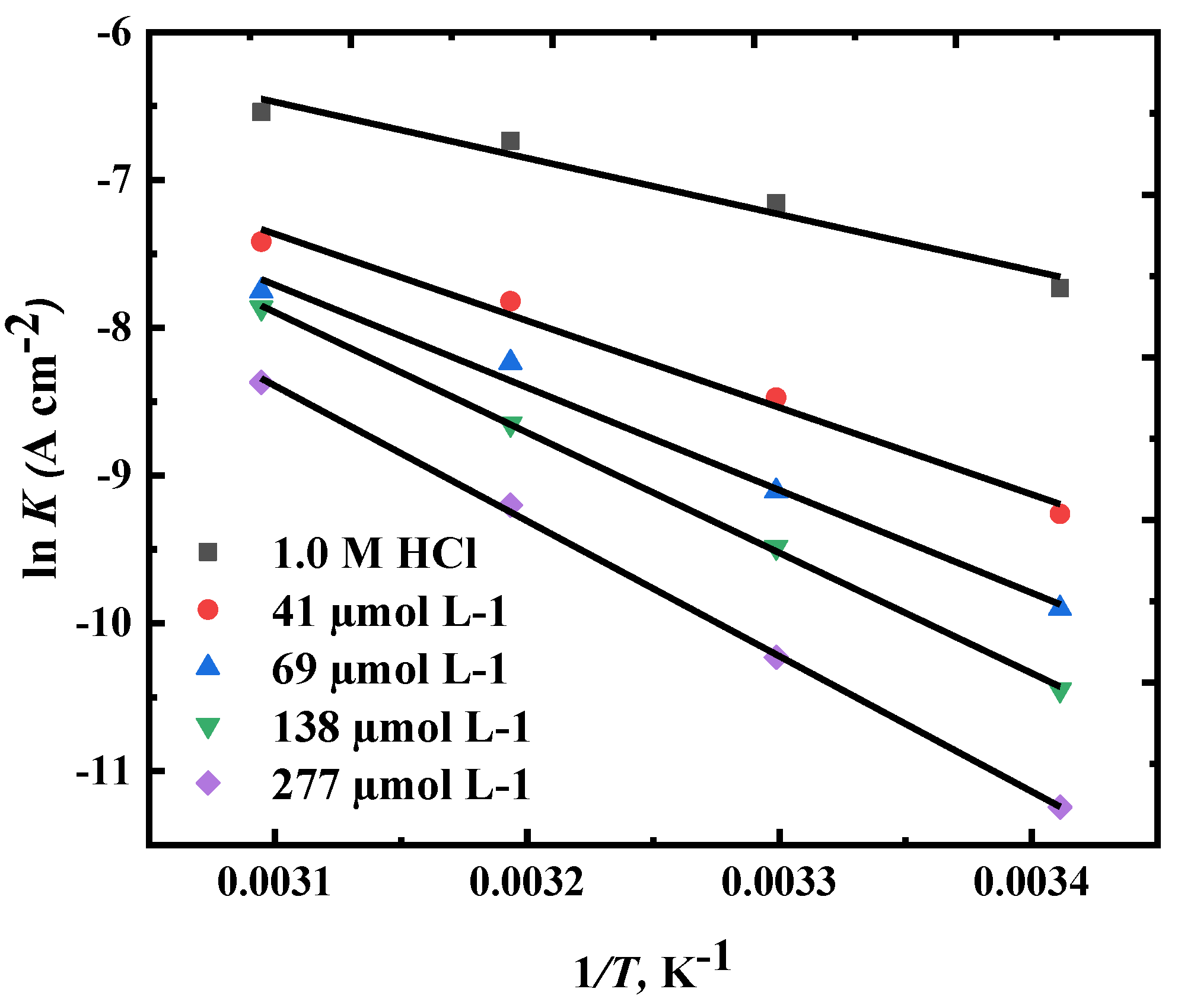
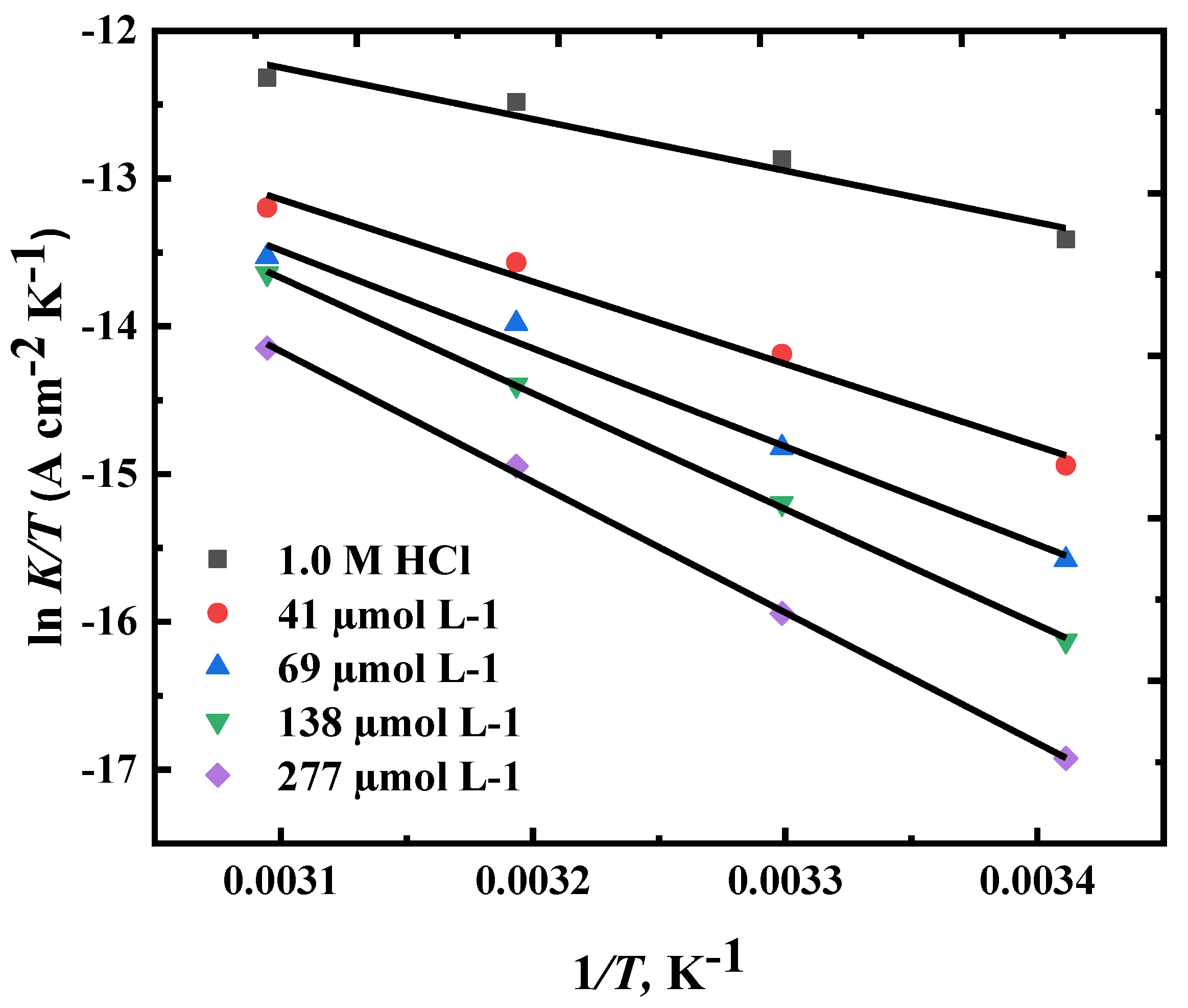
3.4. Corrosion Kinetic and Thermodynamic Studies
3.5. Surface Characterization
3.5.1. Scanning Electron Microscopy (SEM) Analysis
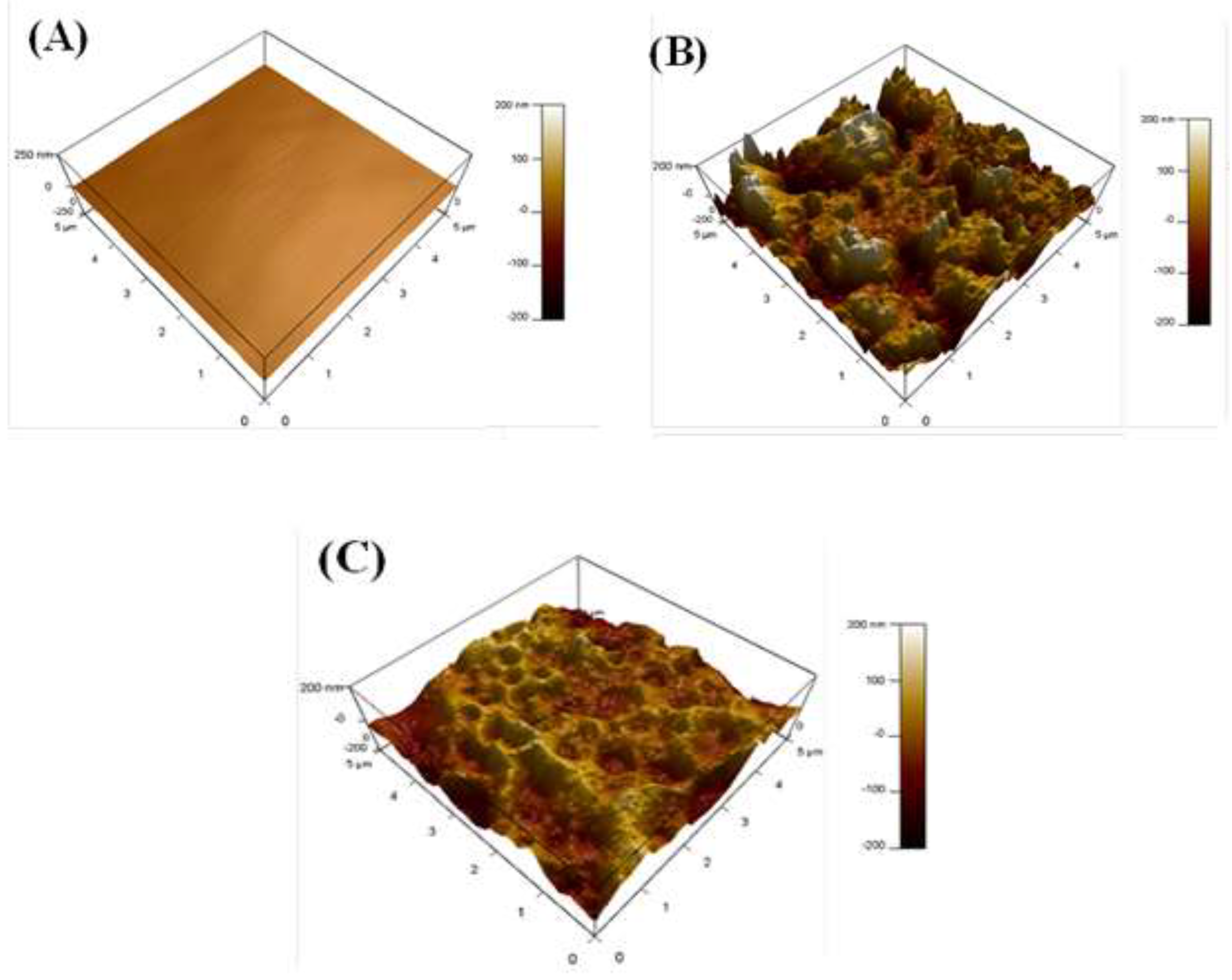
3.5.2. AFM Analysis
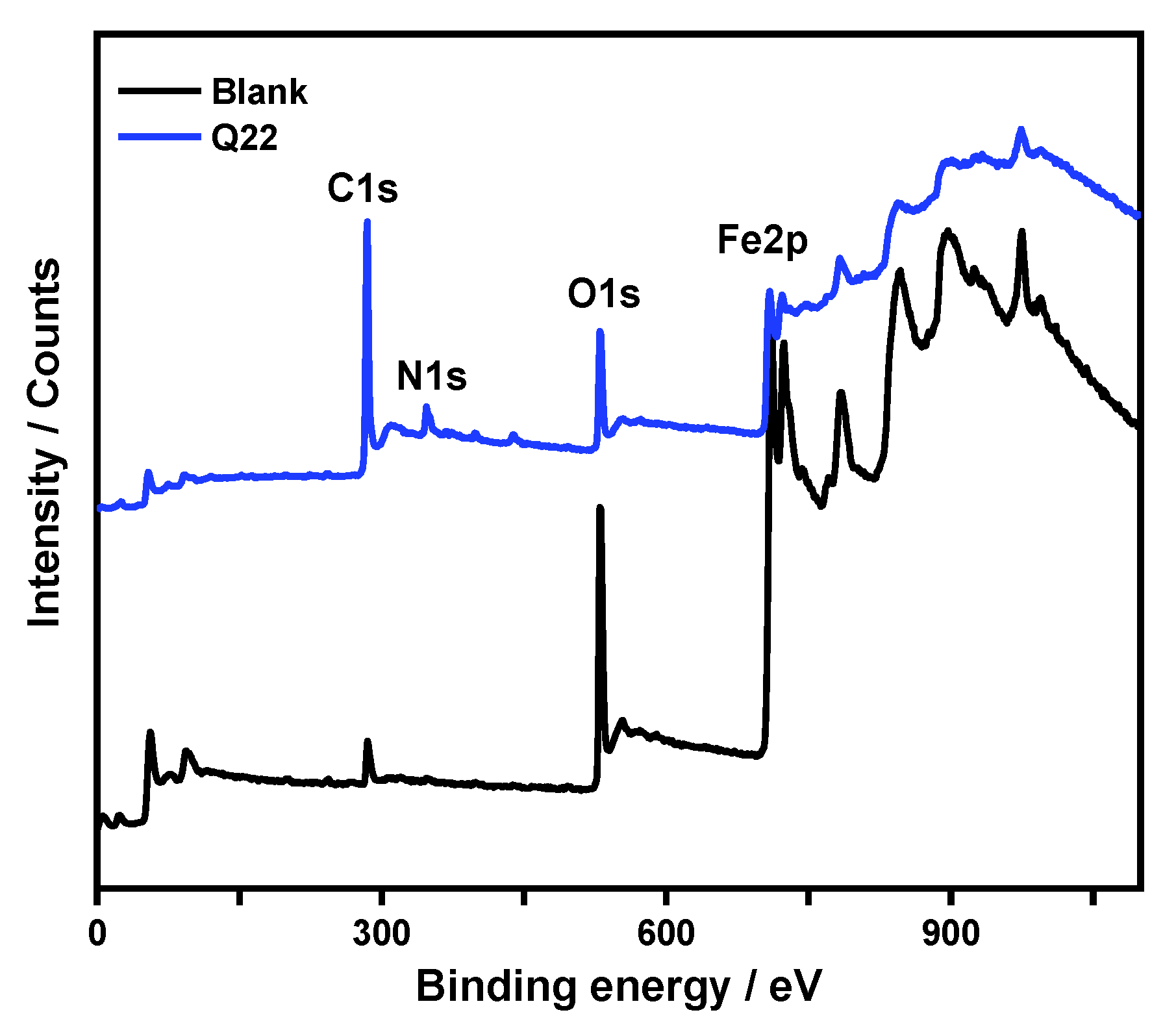
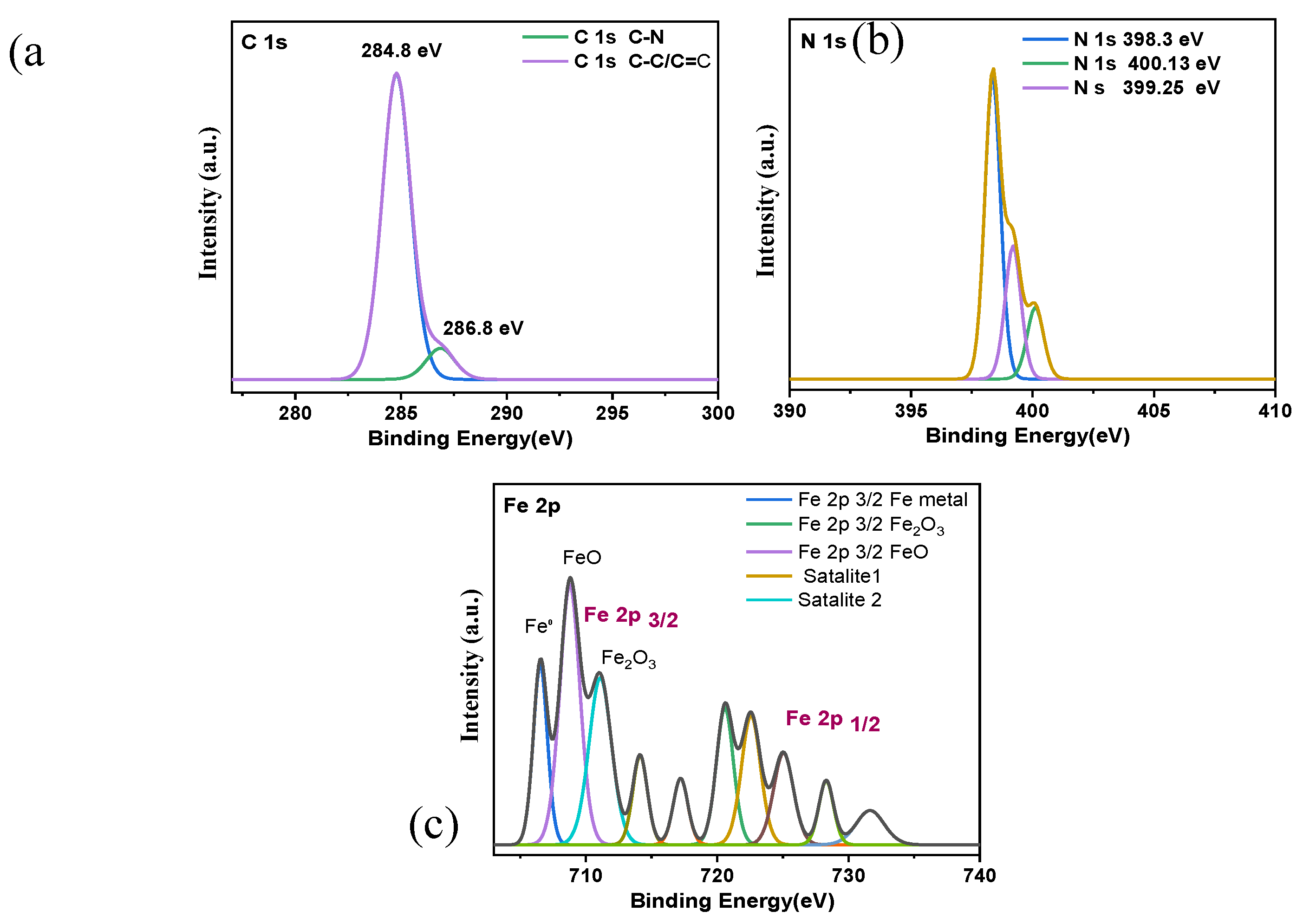
3.5.3. XPS Analysis
3.5.4. Surface Wettability Analysis (Contact Angle)
3.6. DFT studies
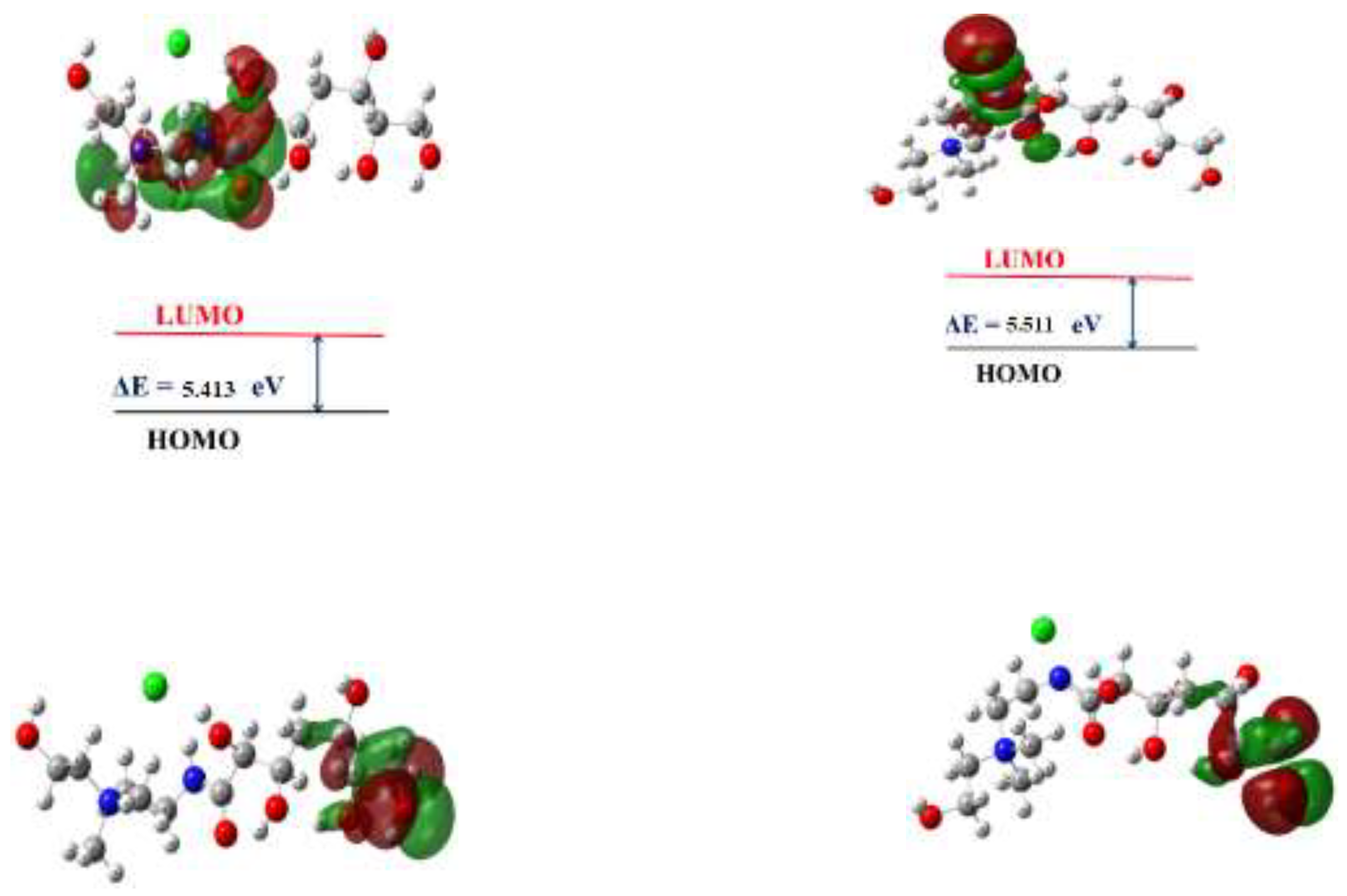
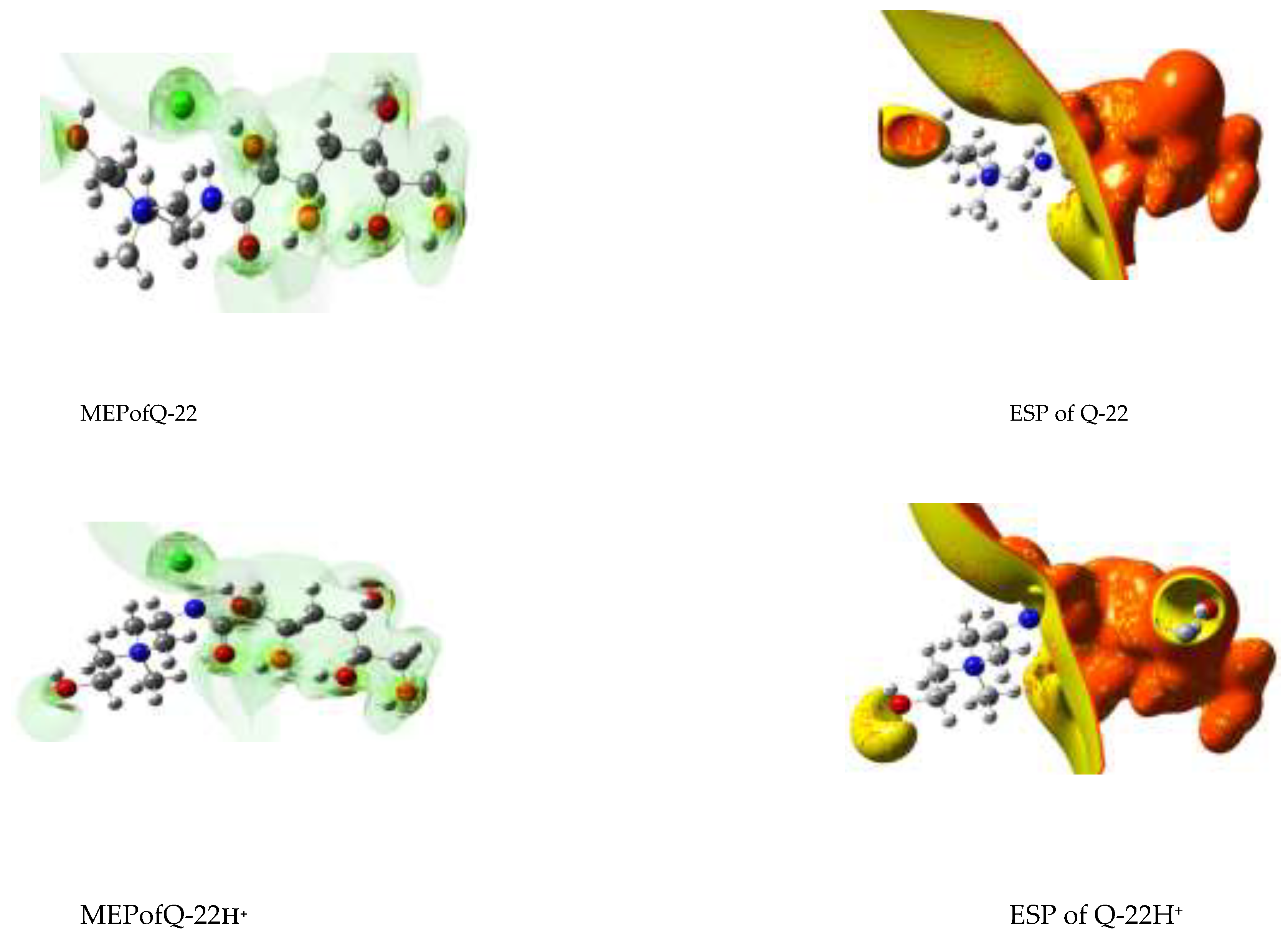
3.6.1. Natural Bond Orbital (NBO) Analysis
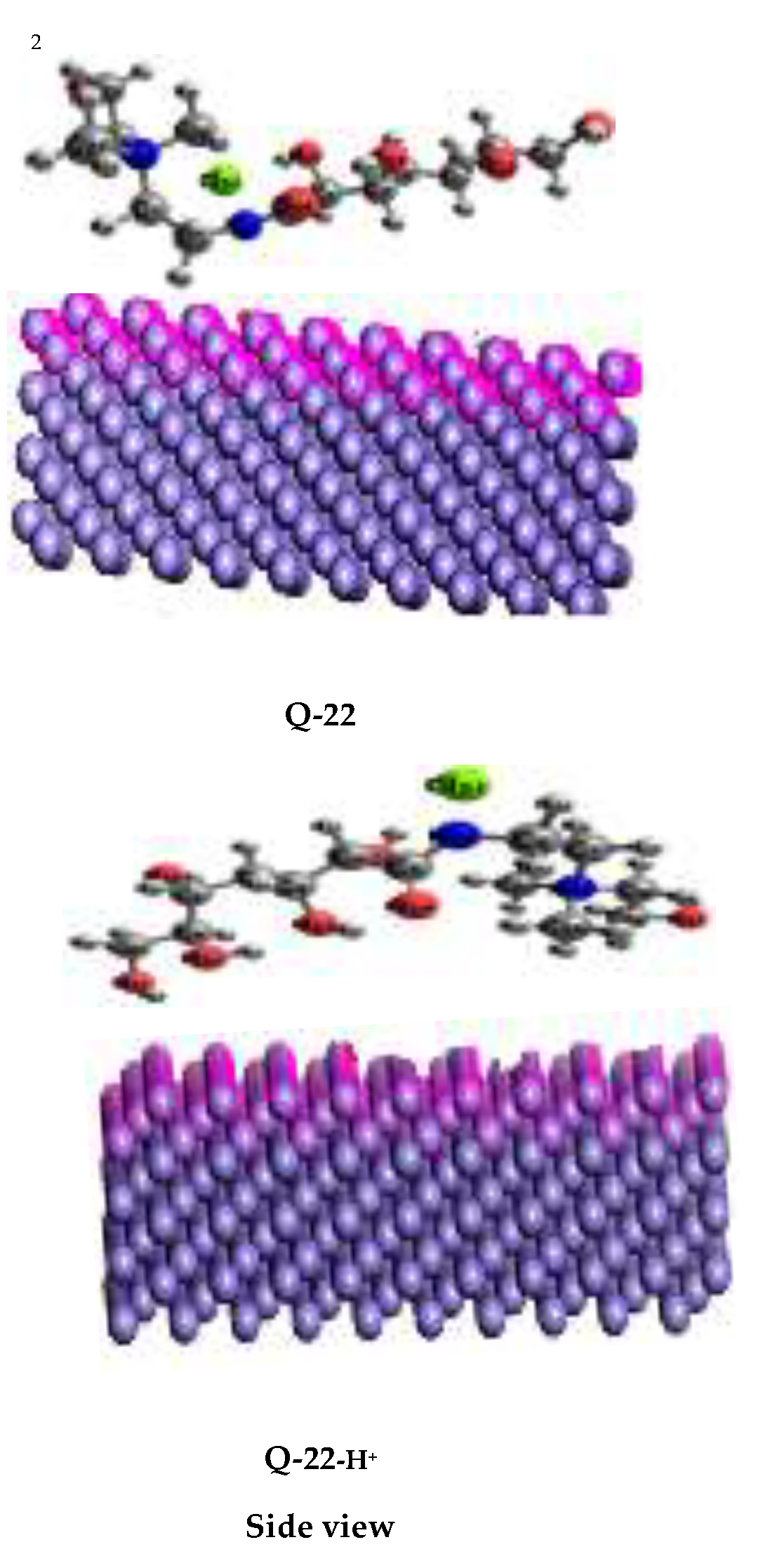
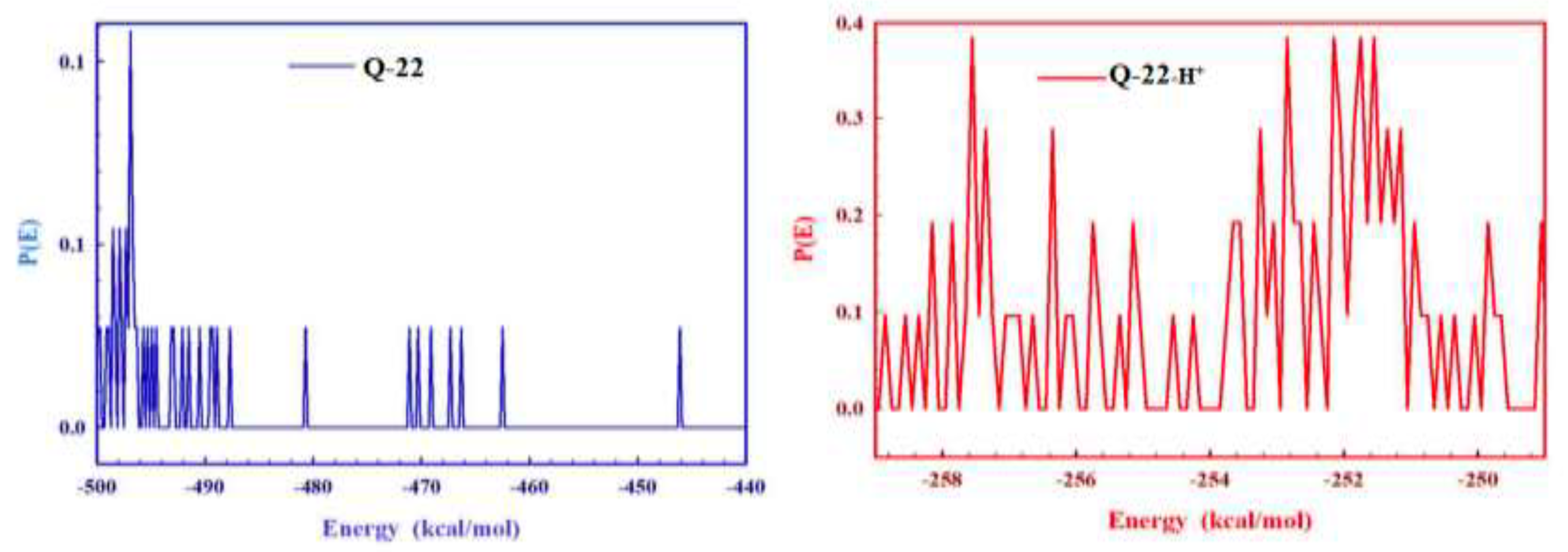
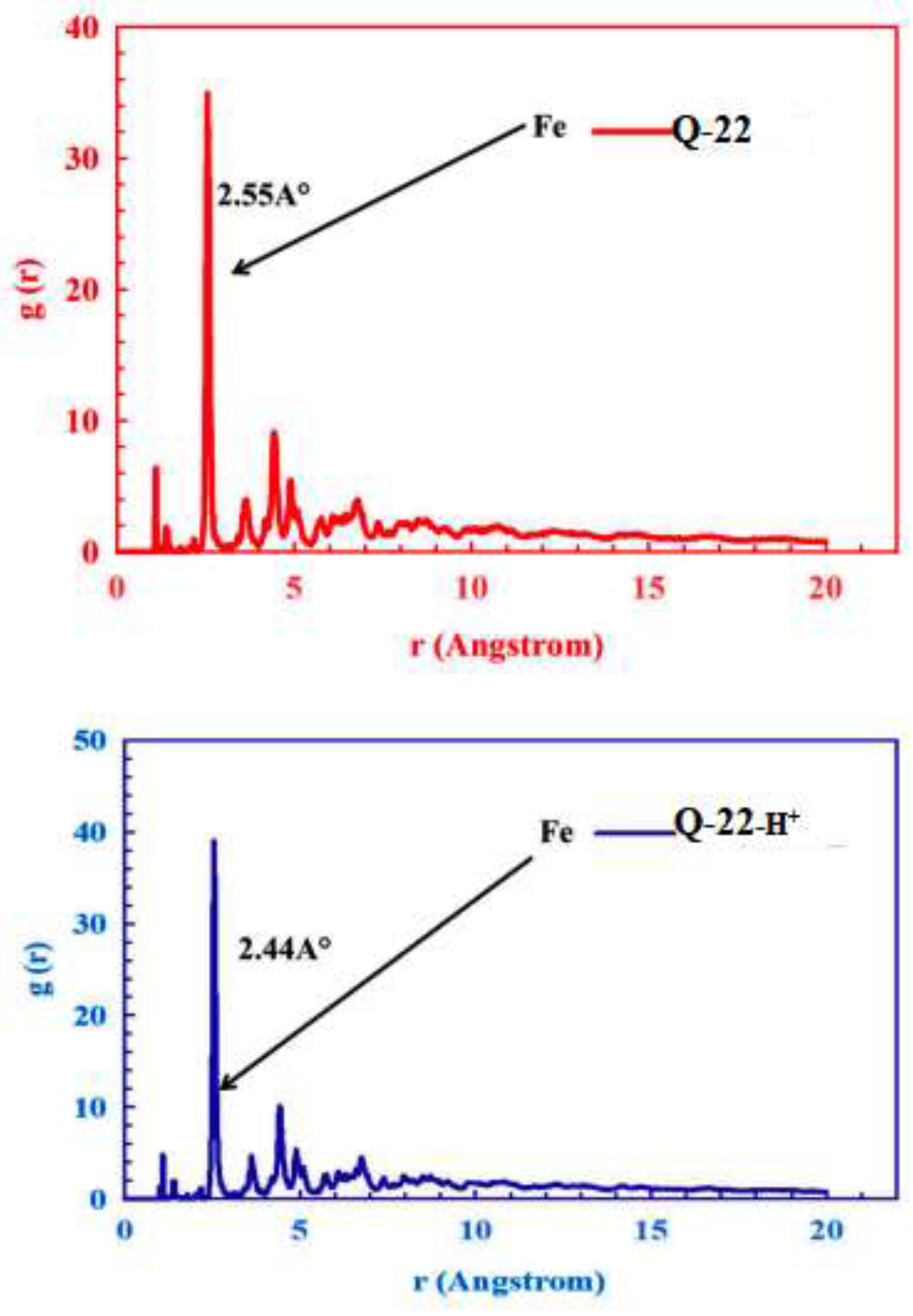
3.6.2. Molecular Simulation Dynamics
5. Conclusions
- Q-22 functions as an outstanding mixed-type corrosion inhibitor, achieving a maximum inhibition efficiency of 97.2% at an optimal concentration of 277 µmol L⁻¹. Its performance is concentration-dependent, with efficiency increasing as the inhibitor dosage rises.
- Electrochemical studies confirm that Q-22 adsorbs strongly onto the CS surface, leading to a substantial increase in charge transfer resistance (Rct) and a marked reduction in double-layer capacitance (Cdl). These findings indicate the formation of a dense, adherent, and resistive protective film that impedes ionic and molecular transport to the metal surface.
- Adsorption studies reveal that the inhibitor’s adsorption on the steel surface is spontaneous and monolayer-forming, following the Langmuir adsorption isotherm. The calculated standard free energy of adsorption (ΔG° ≈ -37 kJ mol⁻¹) suggests that both physisorption and chemisorption contribute to the inhibition mechanism.
- Thermodynamic analysis reveals that the presence of Q-22 substantially increases the apparent activation energy (Ea) of the corrosion process, indicating that the inhibitor raises the energy barrier for metal dissolution, primarily through a physisorption–chemisorption adsorption mechanism.
- Direct visual evidence from SEM, AFM, and XPS analyses corroborates the electrochemical findings. The inhibited surface remains smooth and uniform, with a drastically reduced roughness (RMS = 30 nm) compared to the severely corroded, pitted surface in the blank acid (RMS = 200 nm), signifying the formation of a continuous and stable protective layer.
- The experimental results and quantum chemical (DFT) and molecular modeling analyses, such as NBO and MD simulations, show that Q-22 has strong electron-donating and accepting capabilities that facilitate efficient adsorption on the metal surface and support the suggested inhibition mechanism.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Koch, G.; Brongers, M.; Thompson, N.; Virmani, Y.; Payer, J. International measures of prevention, application, and economics of corrosion technologies; NACE International, 2016. [Google Scholar]
- Revie, R. W.; Uhlig, H. H. Corrosion and corrosion control: An introduction to corrosion science and engineering, 4th ed.; John Wiley & Sons, 2008. [Google Scholar]
- Roberge, P. R. Corrosion engineering: Principles and practice; McGraw-Hill, 2008. [Google Scholar]
- Goyal, M.; Kumar, S.; Bahadur, I.; Verma, C.; Ebenso, E. E. Organic corrosion inhibitors for industrial cleaning of ferrous and non-ferrous metals in acidic solutions: A review. Journal of Molecular Liquids 2018, 256, 565–573. [Google Scholar] [CrossRef]
- Popoola, L. T.; Grema, A. S.; Latinwo, G. K.; Gutti, B.; Balogun, A. S. Corrosion problems during oil and gas production and its mitigation. Corrosion Reviews 2013, 31(3–6), 137–147. [Google Scholar] [CrossRef]
- Papavinasam, S. Corrosion control in the oil and gas industry; Gulf Professional Publishing, 2013. [Google Scholar] [CrossRef]
- Belhadi, M.; Oubahou, M.; Hammoudan, I.; et al. A comprehensive assessment of carbon steel corrosion inhibition by 1,10-phenanthroline in the acidic environment: Insights from experimental and computational studies. Environmental Science and Pollution Research 2024, 31(53), 62038–62055. [Google Scholar] [CrossRef] [PubMed]
- Rafique, M.; Rafique, M. S.; Akbar, R.; Quraishi, M. A. Integrated experimental and theoretical assessment of new pyrazole derivatives as corrosion inhibitors for mild steel in acidic medium. Journal of Molecular Liquids 2019, 276, 101–112. [Google Scholar] [CrossRef]
- Afifi, M.; Sliem, M. H.; Elbasiony, N.; Berdimurodov, E.; Guo, L.; El-Tabei, A. S.; Ibrahim, M. A. M.; Abdullah, A. M. Adsorption mechanism and inhibition efficiency of Quaternium-15 (Q15) surfactant on carbon steel in 1 M HCl: A combined theoretical and experimental approach. Results in Engineering 2025, 28, 106887. [Google Scholar] [CrossRef]
- Khedr, B. M. A.; Ramadan, S. K.; Abdelkader, S. A.; Abdelhamed, S.; El-Etre, M. A.; Elaraby, A.; Ibrahim, M. A. M. Multifaceted insights of experimental, surface, and computational investigations for synthesized carbon steel corrosion inhibitor based on pyrazolyl derivative in acidic environment. Scientific Reports 2025, 15, 43246. [Google Scholar] [CrossRef] [PubMed]
- Omar, I. M. A.; Elshamy, I. H.; Abd El Halim, S.; Ibrahim, M. A. M. Electrochemical and computational analysis of thiocolchicoside as a new corrosion inhibitor for biomedical Ti6Al4V alloy in saline solution: DFT, NBO, and MD approaches. Surfaces 2025, 8(4), 77. [Google Scholar] [CrossRef]
- Abd El Rehim, S. S.; Ibrahim, M. A. M.; Khaled, K. F. Corrosion inhibition and adsorption behaviour of 4-aminoantipyrine on mild steel in H₂SO₄. Corrosion Prevention and Control 1999, 46(1), 30–38. [Google Scholar]
- Ibrahim, M. A. M.; Hamza, M. M.; Ryl, J.; Amin, M. A.; Abd El Rehim, S. S. The inhibition effect of eco-friendly sodium dodecyl sulfate on the corrosion behavior of SUS304L, SUS304H, and SUS316H stainless steels in sulfuric acid solution. International Journal of Electrochemical Science 2021, 16(7), 1–15. [Google Scholar] [CrossRef]
- Singh, A.; Ebenso, E. E.; Quraishi, M. A. Adsorption and corrosion inhibition behavior of polyethylene glycol and surfactants on mild steel in acidic media. Journal of Molecular Liquids 2021, 334, 116495. [Google Scholar] [CrossRef]
- Aslam, R.; Mobin, M.; Shoeb, M.; Zehra, S. Corrosion inhibition of steel using different families of chemical compounds: A review. Journal of Molecular Liquids 2021, 321, 114888. [Google Scholar] [CrossRef]
- Dehghani, A.; Bahlakeh, G.; Ramezanzadeh, B. Green corrosion inhibitors for carbon steel in acidic environments: A review. Journal of the Taiwan Institute of Chemical Engineers 2020, 109, 111–123. [Google Scholar] [CrossRef]
- Chigondo, M.; Chigondo, F. Recent natural corrosion inhibitors for mild steel: An overview. Journal of Chemistry 2016, 2016, 6208937. [Google Scholar] [CrossRef]
- Tezel, Ü.; Guven, K. C. Biodegradation of quaternary ammonium compounds in environmental systems. Environmental Toxicology and Pharmacology 2009, 28(3), 484–489. [Google Scholar] [CrossRef]
- Abd El-Lateef, H. M. Experimental and computational investigation of novel cationic surfactants as corrosion inhibitors for carbon steel in HCl solution. Journal of Industrial and Engineering Chemistry 2021, 93, 417–429. [Google Scholar] [CrossRef]
- Fouda, A. S.; El-Desouky, T. M.; El-Badry, B. A.; El-Azzam, E. M. The role of some organic compounds as corrosion inhibitors for carbon steel in acidic medium: A comprehensive study. Scientific Reports 2020, 10, 10476. [Google Scholar] [CrossRef]
- Hefnawy, A.; Khaled, K. F.; Abdelshafi, N. S.; Ibrahim, M. A. M. Eco-friendly corrosion inhibitors based on carbohydrate derivatives for carbon steel in acidic environments: A review. Journal of Bio- and Tribo-Corrosion 2022, 8, 32. [Google Scholar] [CrossRef]
- El-Mokadem, T. H.; Hashem, A. I.; Abd El-Sattar, N. E. A.; Dawood, E. A.; Abdelshafi, N. S. Green synthesis, electrochemical, DFT studies, and MD simulation of novel synthesized thiourea derivatives on carbon steel corrosion inhibition in 1.0 M HCl. Journal of Molecular Structure 2023, 1274, 134567. [Google Scholar] [CrossRef]
- Abdelshafi, N. S.; Farag, A. A.; Heakal, F. E.-T.; Badran, A.-S.; Abdel-Azim, K. M.; Ibrahim, M. A. In-depth experimental assessment of two new aminocoumarin derivatives as corrosion inhibitors for carbon steel in HCl media combined with AFM, SEM/EDX, contact angle, and DFT/MD simulations. Journal of Molecular Structure 2024, 1304, 137638. [Google Scholar] [CrossRef]
- Hadisaputra, S.; Purwoko, A. A.; Savalas, L. R. T.; Prasetyo, N.; Yuanita, E. Quantum chemical and Monte Carlo simulation studies on inhibition performance of caffeine and its derivatives against corrosion of copper. Coatings 2020, 10(11), 1086. [Google Scholar] [CrossRef]
- Arrousse, N.; Salim, R.; Kaddouri, Y.; Zarrouk, A.; Zahri, D.; Hajjaji, F. E.; Touzani, R.; Taleb, M.; Jodeh, S. The inhibition behavior of two pyrimidine–pyrazole derivatives against corrosion in hydrochloric solution: Experimental, surface analysis, and in silico approach studies. Arabian Journal of Chemistry 2020, 13(11), 5949–5967. [Google Scholar] [CrossRef]
- Masroor, S.; Mobin, M. Non-ionic surfactant as corrosion inhibitor for aluminium in 1 M HCl and synergistic influence of gemini surfactant. Chemical Science Review and Letters 2014, 3(10), 33–48. [Google Scholar]
- Khaled, K. F. The inhibition of benzimidazole derivatives on corrosion of iron in 1 M HCl solutions. Electrochimica Acta 2003, 48(17), 2493–2503. [Google Scholar] [CrossRef]
- Pajkossy, T. Impedance of rough capacitive electrodes. Journal of Electroanalytical Chemistry 1994, 364(1–2), 111–125. [Google Scholar] [CrossRef]
- Caprani, A.; Epelboin, I.; Morel, P.; Takenouti, H. Influence of anodic dissolution on the electrochemical impedance of iron in acidic sulfate medium. In Proceedings of the 4th European Symposium on Corrosion Inhibitors, University of Ferrara, Italy, 1975; pp. 571–583. [Google Scholar]
- Bentiss, F.; Traisnel, M.; Lagrenée, M. The substituted 1,3,4-oxadiazoles: A new class of corrosion inhibitors of mild steel in acidic media. Corrosion Science 2000, 42(1), 127–146. [Google Scholar] [CrossRef]
- Obot, I. B.; Macdonald, D. D.; Gasem, Z. M. Density functional theory (DFT) as a powerful tool for the design of new organic corrosion inhibitors. Part 1: An overview. Corrosion Science 2015, 99, 1–30. [Google Scholar] [CrossRef]
- Fouda, A. S.; El-Askalany, A. H.; El-Habab, A. T. The role of some thiosemicarbazide derivatives as corrosion inhibitors for carbon steel in acidic media. Journal of Molecular Liquids 2017, 240, 372–388. [Google Scholar] [CrossRef]
- Zhao, J.; Duan, H.; Jiang, R. Synergistic corrosion inhibition effect of quinoline quaternary ammonium salt and gemini surfactant in H₂S and CO₂ saturated brine solution. Corrosion Science 2015, 91, 108–119. [Google Scholar] [CrossRef]
- ASTM International. ASTM G3-14: Standard practice for conventions applicable to electrochemical measurements in corrosion testing; ASTM International, 2014. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, W.; Liu, F.; Han, E.-H. Insight into the corrosion inhibition of novel macromolecular surfactants for carbon steel in acidic media. Corrosion Science 2022, 194, 109941. [Google Scholar] [CrossRef]
- El-Etre, A. Y. Inhibition of aluminum corrosion using Opuntia extract. Corrosion Science 2003, 45(11), 2485–2495. [Google Scholar] [CrossRef]
- McCafferty, E. Introduction to corrosion science.; Springer, 2010. [Google Scholar] [CrossRef]
- Popova, A.; Sokolova, E.; Raicheva, S.; Christov, M. AC and DC study of the temperature effect on mild steel corrosion in acid media in the presence of benzimidazole derivatives. Corrosion Science 2003, 45(1), 33–58. [Google Scholar] [CrossRef]
- Lu, B. T.; Chen, Z. K.; Luo, J. L.; Patchett, B. M.; Xu, Z. H. Pitting and stress corrosion cracking behavior in welded austenitic stainless steel. Electrochimica Acta 2005, 50(6), 1391–1403. [Google Scholar] [CrossRef]
- Khamis, E.; Ameen, M. A.; Al-Andi, N. M.; Al-Senani, G. Effect of nonionic surfactant on the corrosion inhibition of aluminum in hydrochloric acid. Corrosion 2000, 56(12), 127–135. [Google Scholar] [CrossRef]
- Voudrias, E.; Fytianos, F.; Bozani, E. Sorption–desorption isotherms of dyes from aqueous solutions and wastewaters with different sorbent materials. Global Nest: The International Journal 2002, 4(1), 75–83. [Google Scholar]
- Mohan, S.; Karthikeyan, J. Removal of lignin and tannin color from aqueous solution by adsorption onto activated carbon. Environmental Pollution 1997, 97(1–2), 183–187. [Google Scholar] [CrossRef]
- Djeddi, N.; Benahmed, M.; Akkal, S.; Laouer, H.; Makhloufi, E.; Gherraf, N. Chemical composition and antimicrobial activity of the essential oil of Ruta montana L. Research on Chemical Intermediates 2014, 41(7), 4595–4605. [Google Scholar] [CrossRef]
- Deyab, M. A.; Abd El-Rehim, S. S. Effect of nonionic surfactant as an electrolyte additive on the performance of aluminum-air battery. Journal of the Taiwan Institute of Chemical Engineers 2013, 45(4), 1065–1070. [Google Scholar] [CrossRef]
- Ali, S. A.; El-Shareef, A. M.; Al-Ghamdi, R. F.; Saeed, M. T. The isoxazolidines: The effects of steric factor and hydrophobic chain length on the corrosion inhibition of mild steel in acidic medium. Corrosion Science 2005, 47(11), 2659–2678. [Google Scholar] [CrossRef]
- Seifzadeh, D.; Bezaatpour, A. A Schiff base compound as effective corrosion inhibitor for magnesium in acidic media. Materials Chemistry and Physics 2013, 138(2–3), 794–802. [Google Scholar] [CrossRef]
- Mobin, M.; Rizvi, M. Inhibitory effect of xanthan gum and synergistic surfactant additives for mild steel corrosion in 1 M HCl. Carbohydrate Polymers 2016, 136, 384–393. [Google Scholar] [CrossRef]
- Eddy, N. O.; Ekop, A. S. Inhibition of the corrosion of mild steel in HCl by penicillin V potassium. Materials Science Applications International Journal 2007, 4(1), 2008–2016. [Google Scholar]
- Saratha, R.; Devi, S. S.; Meenakshi, H. N.; Shyamala, R. Investigation of Solanum nigrum leaves extract as corrosion inhibitor for mild steel in acid medium *. International Journal of Current Research 2011, 2(1), 92–98. [Google Scholar]
- Hebrodike, M. M.; Uroh, A. A.; Okeoma, K. B.; Alozie, G. A. Inhibition of the corrosion of mild steel in HCl by Vernonia amygdalina *. African Journal of Pure and Applied Chemistry 2010, 4(8), 158–164. [Google Scholar]
- Hamdy, A.; El-Gendy, N. S. Thermodynamic, adsorption, and electrochemical studies for corrosion inhibition of carbon steel by henna extract in acid medium. Egyptian Journal of Petroleum 2013, 22(1), 17–25. [Google Scholar] [CrossRef]
- Ostovari, A.; Hoseinieh, S. M.; Peikari, M.; Shadizadeh, S. R.; Hashemi, S. J. Corrosion inhibition of mild steel in 1 M HCl solution by henna extract: A comparative study of the inhibition by henna and its constituents (lawsone, gallic acid, α-D-glucose, and tannic acid). Corrosion Science 2009, 51(9), 1935–1949. [Google Scholar] [CrossRef]
- Yadav, D. K.; Maiti, B.; Quraishi, M. A. Electrochemical and quantum chemical studies of 3,4-dihydropyrimidin-2(1H)-ones as corrosion inhibitors for mild steel in hydrochloric acid solution. Corrosion Science 2010, 52(11), 3586–3598. [Google Scholar] [CrossRef]
- Putilova, I. N.; Balezin, S. A.; Barannik, V. P. Metallic corrosion inhibitors.; Pergamon Press, 1960. [Google Scholar]
- Tebbji, K.; Faska, N.; Tounsi, A.; Oudda, H.; Benkaddour, M.; Hammouti, B. The effect of some lactones as inhibitors for the corrosion of mild steel in 1 M hydrochloric acid. Materials Chemistry and Physics 2007, 106(2–3), 260–267. [Google Scholar] [CrossRef]
- Shukla, S. K.; Quraishi, M. A.; Prakash, R. A self-doped conducting polymer “polyanthranilic acid”: An efficient corrosion inhibitor for mild steel in acidic solution. Corrosion Science 2008, 50(10), 2867–2872. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, J.; Yu, W.; Yan, Y.; You, L.; Liu, L. Investigation of inhibition properties of sophorolipids for X65 steel corrosion in simulated oilfield produced water saturated with carbon dioxide. Industrial & Engineering Chemistry Research 2015, 54(19), 5197–5203. [Google Scholar] [CrossRef]
- Abd El-Rehim, S. S.; Refaey, S. A. M.; Taha, F.; Saleh, M. B.; Ahmed, R. A. Corrosion inhibition of mild steel in acidic medium using 2-amino-thiophenol and 2-cyanomethyl-benzothiazole. Journal of Applied Electrochemistry 2001, 31(4), 429–435. [Google Scholar] [CrossRef]
- Finšgar, M.; Jackson, J. Application of corrosion inhibitors for steels in acidic media for the oil and gas industry: A review. Corrosion Science 2014, 86, 17–41. [Google Scholar] [CrossRef]
- Verma, C.; Ebenso, E. E.; Bahadur, I.; Quraishi, M. A. An overview on plant extracts as environmentally sustainable and green corrosion inhibitors for metals and alloys in aggressive corrosive media. Journal of Molecular Liquids 2018, 266, 577–590. [Google Scholar] [CrossRef]
- Quraishi, M. A.; Singh, A.; Chauhan, D. S. Recent progress in surfactant-type corrosion inhibitors for metals in acidic media. Journal of Molecular Liquids 2022, 367, 120505. [Google Scholar] [CrossRef]
- Zheng, T.; Zhang, Z.; Zhao, X.; Liu, Y.; Xu, X. Synergistic corrosion inhibition effects of quaternary ammonium salt cationic surfactants and thiourea on Q235 steel in sulfuric acid: Experimental and theoretical research. Corrosion Science 2022, 199, 110199. [Google Scholar] [CrossRef]
- Li, X.; Deng, S.; Du, G. Nonionic surfactant of coconut diethanolamide as a novel corrosion inhibitor for cold-rolled steel in both HCl and H₂SO₄ solutions. Journal of the Taiwan Institute of Chemical Engineers 2022, 131, 104171. [Google Scholar] [CrossRef]
- Mobin, M.; Aslam, J.; Alreface, S. H. An investigation on the synthesis, characterization and anti-corrosion properties of choline-based ionic liquids as novel and environmentally friendly inhibitors for mild steel corrosion in 5% HCl. Journal of Colloid and Interface Science 2022, 620, 293–312. [Google Scholar] [CrossRef]
- Paiva, V. M.; Silva, T. F.; Falcão, S. G.; Rocha, C.; Villamil, R. F. Novel eco-friendly green inhibitor of corrosion based on acerola (Malpighia glabra) waste aqueous extract for mild steel in 1 mol L⁻¹ HCl solution. Surfaces and Interfaces 2024, 47, 104187. [Google Scholar] [CrossRef]
- El-Etre, A. Y.; Abdallah, M.; El-Tantawy, Z. E. The adsorption behavior of some thiosemicarbazide derivatives on mild steel in acidic medium. Corrosion Science 2008, 50(5), 1465–1471. [Google Scholar] [CrossRef]
- Oguzie, E. E.; Li, Y.; Wang, F.; Wang, S. G. Understanding corrosion inhibition mechanisms through surface analysis and quantum chemical calculations: A review. The Journal of Physical Chemistry C 2009, 113(19), 8420–8429. [Google Scholar] [CrossRef]
- Zhang, D.; Wang, Y.; Liu, L.; Wei, Y. Insight into the anti-corrosion performance of a novel hydrophobic film constructed on copper surface. Corrosion Science 2022, 194, 109941. [Google Scholar] [CrossRef]
- He, T.; Ma, H.; Wang, Y.; Zhang, Y. Construction of a superhydrophobic surface on AZ31B magnesium alloy for corrosion protection. Surface and Coatings Technology 2020, 385, 125429. [Google Scholar] [CrossRef]
- Ramezanzadeh, B.; Bahlakeh, G.; Ramezanzadeh, M. Study of the effect of a new eco-friendly surface treatment on the corrosion resistance of mild steel in HCl solution. Journal of the Taiwan Institute of Chemical Engineers 2018, 91, 439–455. [Google Scholar] [CrossRef]
- Ye, Y.; Zhang, D.; Li, J.; Liu, T.; Wang, J.; Wang, Y. One-step fabrication of a superhydrophobic surface for corrosion protection of aluminum alloy. Colloids and Surfaces A: Physicochemical and Engineering Aspects 2021, 612, 125991. [Google Scholar] [CrossRef]
- Radwan, A. B.; Ibrahim, M. A.; Ismail, E. H.; Santo, L. Beyond wetting: Unveiling the theoretical and practical power of superhydrophobic coatings in multifunctional applications. In Industrial & Engineering Chemistry Research.; Advance online publication, 2025. [Google Scholar] [CrossRef]
- Khaled, K. F.; Hamed, M. N. H.; Abdel-Azim, K. M.; Abdelshafi, N. S. Inhibition of copper corrosion in 3.5% NaCl solutions by a new pyrimidine derivative: Electrochemical and computer simulation techniques. Journal of Solid State Electrochemistry 2011, 15(4), 663–675. [Google Scholar] [CrossRef]
- Zuo, X.; Li, W.; Luo, W.; Zhang, X.; Qiang, Y.; Zhang, J.; Li, H.; Tan, B. Research of Lilium brownii leaves extract as a commendable and green inhibitor for X70 steel corrosion in hydrochloric acid. Journal of Molecular Liquids 2021, 321, 114914. [Google Scholar] [CrossRef]
- Zarrouk, A.; El Ouali, I.; Bouachrine, M.; Hammouti, B.; Ramli, Y.; Essassi, E. M.; Warad, I.; Aouniti, A.; Salghi, R. Theoretical approach to the corrosion inhibition efficiency of some quinoxaline derivatives of steel in acid media using the DFT method. Research on Chemical Intermediates 2013, 39(3), 1125–1136. [Google Scholar] [CrossRef]
- Saady, A.; Rais, Z.; Benhiba, F.; Salim, R.; Ismaily Alaoui, K.; Arrousse, N.; Elhajjaji, F.; Taleb, M.; Jarmoni, K.; Kandri Rodi, Y.; Warad, I.; Zarrouk, A. Chemical, electrochemical, quantum, and surface analysis evaluation on the inhibition performance of novel imidazo[4,5-b]pyridine derivatives against mild steel corrosion. Corrosion Science 2021, 189, 109621. [Google Scholar] [CrossRef]
- Laabaissi, T.; Benhiba, F.; Rouifi, Z.; Missioui, M.; Ourrak, K.; Oudda, H.; Ramli, Y.; Warad, I.; Allali, M.; Zarrouk, A. New quinoxaline derivative as a green corrosion inhibitor for mild steel in mild acidic medium: Electrochemical and theoretical studies. International Journal of Corrosion and Scale Inhibition 2019, 8(2), 241–258. [Google Scholar] [CrossRef]
- Gece, G.; Bilgiç, S. A theoretical study on the inhibition efficiencies of some amino acids as corrosion inhibitors of nickel. Corrosion Science 2010, 52(10), 3435–3443. [Google Scholar] [CrossRef]
- Wang, H.; Wang, X.; Wang, H.; Wang, L.; Liu, A. DFT study of new bipyrazole derivatives and their potential activity as corrosion inhibitors. Journal of Molecular Modeling 2007, 13(2), 147–153. [Google Scholar] [CrossRef]
- Rbaa, M.; Lakhrissi, B. Novel oxazole and imidazole based on 8-hydroxyquinoline as corrosion inhibitors of mild steel in HCl solution: Insights from experimental and computational studies. Surfaces and Interfaces 2019, 15, 43–51. [Google Scholar] [CrossRef]
- El-Deeb, M. M.; Abdel-Shafi, N. S.; Shamroukh, A. H. Electrochemical, DFT and Monte Carlo simulations studies to evaluate the inhibition effect of novel pyridazine derivatives on iron pitting corrosion in 3.5% NaCl. International Journal of Electrochemical Science 2018, 13, 5352–5371. [Google Scholar] [CrossRef]
- Abdelsalam, M. M.; Bedair, M. A.; Hassan, A. M.; Heakal, B. H.; Younis, A.; Elbialy, Z. I.; Badawy, M. A.; Fawzy, H. E.-D.; Fareed, S. A. Green synthesis, electrochemical, and DFT studies on the corrosion inhibition of steel by some novel triazole Schiff base derivatives in hydrochloric acid solution. Arabian Journal of Chemistry 2022, 15, 103491. [Google Scholar] [CrossRef]
- Abdelshafi, N. S.; Ibrahim, M. A.; Badran, A.-S.; Halim, S. A. Experimental and theoretical evaluation of a newly synthesized quinoline derivative as corrosion inhibitor for iron in 1.0 M hydrochloric acid solution. Journal of Molecular Structure 2022, 1250, 131750. [Google Scholar] [CrossRef]
- Belghiti, M. E.; Echihi, S.; Dafali, A.; Karzazi, Y.; Bakasse, M.; Elalaoui-Elabdallaoui, H.; Olasunkanmi, L. O.; Ebenso, E. E.; Tabyaoui, M. Computational simulation and statistical analysis on the relationship between corrosion inhibition efficiency and molecular structure of some hydrazine derivatives in phosphoric acid on mild steel surface. Applied Surface Science 2019, 491, 707–722. [Google Scholar] [CrossRef]
- Cao, Z.; Tang, Y.; Cang, H.; Xu, J.; Lu, G.; Jing, W. Novel benzimidazole derivatives as corrosion inhibitors of mild steel in the acidic media. Part II: Theoretical studies. Corrosion Science 2014, 83, 292–298. [Google Scholar] [CrossRef]
- Politzer, P.; Murray, J. S. The fundamental nature and role of the electrostatic potential in atoms and molecules. Theoretical Chemistry Accounts 2002, 108(3), 134–142. [Google Scholar] [CrossRef]
- Abd El-Lateef, H. M.; Sayed, A. R.; Gomha, S. M.; Bakir, E. M.; Shalabi, K. Synthesis and study of poly[(hydrazinylazo)]thiazoles as potent corrosion inhibitors for cast iron–carbon alloy in molar HCl: A collective computational and experiential methods approach. Journal of Molecular Liquids 2021, 337, 116555. [Google Scholar] [CrossRef]
- Fouda, A.; Ismail, M.; Abousalem, A. S.; Elewady, G. Experimental and theoretical studies on corrosion inhibition of 4-amidinophenyl-2,2′-bifuran and its analogues in acidic media. RSC Advances 2017, 7(73), 46414–46430. [Google Scholar] [CrossRef]
- Mert, B. D.; Yüce, A. O.; Kardaş, G.; Yazıcı, B. Inhibition effect of 2-amino-4-methylpyridine on mild steel corrosion: Experimental and theoretical investigation. Corrosion Science 2014, 85, 287–295. [Google Scholar] [CrossRef]
- Guerrab, W.; Chung, I.-M.; Kansiz, S.; Mague, J. T.; Dege, N.; Taoufik, J.; Salghi, R.; Ali, I. H.; Khan, M. I.; Lgaz, H. Synthesis, structural and molecular characterization of 2,2-diphenyl-2H,3H,5H,6H,7H-imidazo[2,1-b][1,3]thiazin-3-one. Journal of Molecular Structure 2019, 1197, 369–376. [Google Scholar] [CrossRef]
- Guerrab, W.; Lgaz, H.; Kansiz, S.; Mague, J. T.; Dege, N.; Ansar, M.; Marzouki, R.; Taoufik, J.; Ali, I. H.; Chung, I.-M. Synthesis of a novel phenytoin derivative: Crystal structure, Hirshfeld surface analysis, and DFT calculations. Journal of Molecular Structure 2020, 1205, 127630. [Google Scholar] [CrossRef]
- Abdallah, M.; Al Bahir, A.; Altass, H. M.; Fawzy, A.; El Guesmi, N.; Al-Gorair, A. S.; Benhiba, F.; Warad, I.; Zarrouk, A. Anticorrosion and adsorption performance of expired antibacterial drugs on Sabic iron corrosion in HCl solution: Chemical, electrochemical, and theoretical approach. Journal of Molecular Liquids 2021, 330, 115702. [Google Scholar] [CrossRef]


| Elements | Carbon (C) | Silicon (Si) | Manganese (Mn) | Sulfur (S) | Copper (Cu) | Iron (Fe) |
| Content (wt.%) | 0.26 | 0.28 | 1.05 | 0.003 | 0.2 | Balance |
| Organic Compound | |
| Quaternium-22 (Q-22) | |
| Structure | 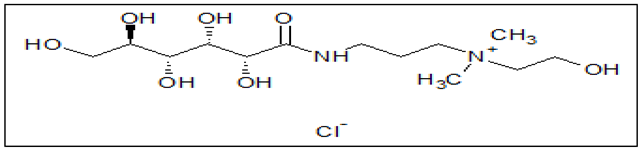 |
| IUPAC Name &Chemical Formula |
N-(2-hydroxyethyl)-N, N-dimethyl-3-(gluconoylamino)propanaminium chloride C13H29N2ClO7 |
| Molecular Weight | 360.83 g/mol |
|
Conc. (μmol L-1) t (min) |
Surface Coverage (ϴ) | |||||
| 30 | 60 | 90 | 120 | 150 | 180 | |
| Blank | - | - | - | - | - | - |
| 41 | 0.7985 | 0.7972 | 0.7675 | 0.7259 | 0.7175 | 0.7062 |
| 69 | 0.8917 | 0.8797 | 0.8770 | 0.8614 | 0.8472 | 0.8436 |
| 138 | 0.9477 | 0.9209 | 0.9108 | 0.9006 | 0.8997 | 0.8981 |
| 277 | 0.9552 | 0.9450 | 0.9394 | 0.9367 | 0.9308 | 0.9299 |
| Inhibition efficiency (%IE) | ||||||
| Blank | - | - | - | - | - | - |
| 41 | 79.85 | 79.72 | 76.75 | 72.59 | 71.75 | 70.61 |
| 69 | 89.17 | 87.97 | 87.70 | 86.14 | 84.72 | 84.36 |
| 138 | 94.77 | 92.09 | 91.08 | 90.06 | 89.97 | 89.81 |
| 277 | 95.52 | 94.50 | 93.94 | 93.67 | 93.08 | 92.99 |
| T (°C) |
Cinh (µmol L-1) |
Rct(Ω cm2) |
Yοx 10-6 µΩ−1 sncm−2 |
Cdl, µF | n | ϴ | %IE |
| 20° | Blank | 59.63 | 570 | 267.04 | 0.918 | ---- | ---- |
| 41 | 271.5 | 34.8 | 58.65 | 0.884 | 0.781 | 78.14 | |
| 69 | 459.2 | 86.76 | 34.68 | 0.806 | 0.870 | 87.01 | |
| 138 | 934.5 | 56.82 | 17.04 | 0.808 | 0.936 | 93.60 | |
| 277 | 2104 | 58.41 | 7.57 | 0.770 | 0.971 | 97.00 | |
| 30° | Blank | 45.7 | 649.6 | 348.44 | 0.897 | ---- | ---- |
| 41 | 192.8 | 257.2 | 82.59 | 0.861 | 0.762 | 76.29 | |
| 69 | 316.6 | 106.7 | 50.30 | 0.832 | 0.855 | 85.55 | |
| 138 | 640.6 | 98.22 | 24.86 | 0.799 | 0.928 | 92.28 | |
| 277 | 1259 | 65.01 | 12.65 | 0.808 | 0.963 | 96.63 | |
| 40° | Blank | 30.7 | 709.3 | 518.68 | 0.893 | ---- | ---- |
| 41 | 92.26 | 146 | 172.59 | 0.767 | 0.667 | 66.72 | |
| 69 | 142.5 | 102.1 | 111.74 | 0.847 | 0.784 | 78.45 | |
| 138 | 297.9 | 459.7 | 53.45 | 0.854 | 0.896 | 89.67 | |
| 277 | 399.6 | 87.64 | 39.85 | 0.702 | 0.923 | 92.23 | |
| 50° | Blank | 18.9 | 886.4 | 842.52 | 0.871 | ---- | ---- |
| 41 | 48.6 | 181.2 | 327.65 | 0.843 | 0.611 | 61.11 | |
| 69 | 66.88 | 262.4 | 238.09 | 0.826 | 0.717 | 71.74 | |
| 138 | 89.61 | 214.5 | 177.70 | 0.800 | 0.789 | 78.90 | |
| 277 | 127.1 | 154.3 | 125.28 | 0.821 | 0.851 | 85.12 |
| T (°C) |
Cinh µmol L-1 |
-Ecorr (mV) SCE |
icorr (µA cm-2) |
βa, mV decade-1 |
βc, mV decade-1 |
Rp, Ω cm2 |
CR, mpy |
θ | IE% |
| 20 | Blank | 407 | 439 | 170.9 | 224.8 | 96.15 | 401.4 | ---- | ---- |
| 41 | 409 | 95.1 | 107.6 | 164.1 | 296.73 | 43.47 | 0.781 | 78.14 | |
| 69 | 453 | 50.2 | 80.5 | 110.3 | 402.53 | 22.93 | 0.890 | 89.01 | |
| 138 | 449 | 29 | 45.6 | 79.3 | 433.49 | 13.24 | 0.936 | 93.62 | |
| 277 | 487 | 13.1 | 34.8 | 60.8 | 733.61 | 7.381 | 0.972 | 97.20 | |
| 30 | Blank | 405 | 780 | 229.7 | 283.7 | 70.75 | 713.2 | ---- | ---- |
| 41 | 441 | 209 | 77.6 | 110 | 94.53 | 95.41 | 0.732 | 73.29 | |
| 69 | 408 | 111 | 50.8 | 120.7 | 139.86 | 50.5 | 0.865 | 86.55 | |
| 138 | 409 | 76.1 | 87.8 | 146.1 | 312.92 | 34.78 | 0.902 | 90.28 | |
| 277 | 411 | 36.1 | 59.4 | 111.5 | 466.14 | 16.51 | 0.956 | 95.63 | |
| 40 | Blank | 396 | 1187 | 362.8 | 332.5 | 63.54 | 1085 | ---- | ---- |
| 41 | 427 | 401.0 | 65.3 | 126.7 | 46.66 | 183.4 | 0.667 | 66.72 | |
| 69 | 441 | 265 | 86.7 | 142.8 | 88.39 | 121.1 | 0.764 | 76.45 | |
| 138 | 433 | 175 | 99.3 | 97.1 | 121.81 | 80.07 | 0.856 | 85.67 | |
| 277 | 408 | 101 | 50 | 109.0 | 147.36 | 46.11 | 0.912 | 91.23 | |
| 50 | Blank | 389 | 1443 | 371.5 | 361.1 | 55.17 | 1319 | ---- | ---- |
| 41 | 482 | 600 | 128.9 | 237.6 | 60.48 | 274.3 | 0.581 | 58.11 | |
| 69 | 425 | 430 | 119.6 | 238.6 | 80.45 | 196.3 | 0.707 | 70.74 | |
| 138 | 426 | 384 | 124.2 | 220.6 | 89.85 | 175.6 | 0.739 | 73.90 | |
| 277 | 425 | 232 | 71.9 | 109.1 | 81.11 | 105.9 | 0.841 | 84.12 |
|
Cinh (µmol L-1) |
Ea(kJ mol-1) | ΔH* (kJ Mol-1) | ΔS* (J mol-1 K-1) |
| 0 | 31.5 | 29.01 | -209.354 |
| 41 | 48.8 | 46.23 | -163.387 |
| 69 | 57.6 | 55.12 | -138.662 |
| 138 | 67.6 | 65.05 | -109.429 |
| 277 | 76.01 | 73.46 | -87.493 |
| No. | CS sample | Roughness (Ra)/nm |
| A | Blank (Polished CS) | 10 |
| B | 1.0 M HCl (Corroded CS) | 200 |
| C | 1.0 M HCl + 277 µmol L-1 Q-22 (Inhibited CS) | 30 |
| Descriptors | Equations | Q-22 | Q-22H+ |
| Energy of highest occupied molecular orbital (EHOMO), (eV) | -6.465 | -6.370 | |
| Energy of lowest unoccupied molecular orbital (ELUMO), (eV) | -1.052 | -0.8587 | |
| Energy Gap ΔE | (LUMO-HOMO) | 5.413 | 5.511 |
| Dipole moment, (µ), (Debye) | 16.401 | 17.146 | |
| Ionization energy (I) (ev) | 6.465 | 6.370 | |
| Electron affinity ( ) (ev) | 1.052 | 0.8587 | |
| Electronegativity (φ ) | φ= | 3.758 | 3.614 |
| Global hardness ψ | ψ = | 2.706 | 2.756 |
| Global softness (S) | s = | 0.370 | 0.363 |
| Global electrophilicity (ω) | ω = φ2/2ψ | 2.609 | 2.370 |
| Global nucleophilicity (ε) | 0.383 | 0.422 | |
| Electroaccepting (ω+) power | 1.068 | 0.9076 | |
| Electrodonating (ω−) power | 4.827 | 4.522 | |
| Net electrophilicity (Δω± = ω+ +ω−) | (Δω± = ω+ +ω−) | 5.895 | 5.430 |
| Fraction of transferred electrons (ΔN) | -0.1009 | -0.1197 | |
| Back-donation energy ΔE back-donation (ev) | -0.6765 |
-0.6890 | |
| Metal/inhibitor interaction energy ΔE Metal /inhibitor (ev) |
0.0357 |
0.0509 |
| Q-22 | Q-22H+ | |||||||
| Atoms | atoms | |||||||
| Cl (1) | 0.022 | 0.003 | 0.019 | Cl (1) | 0.01 | 0.002 | 0.008 | |
| O (2) | 0.028 | 0.001 | 0.027 | O (2) | 0.024 | 0.016 | 0.008 | |
| N (4) | 0.003 | 0.008 | -0.005 | N (4) | 0 | 0.001 | -0.001 | |
| C (5) | 0.014 | 0.005 | 0.009 | C (5) | 0.016 | 0.01 | 0.006 | |
| C (6) | 0.015 | 0.001 | 0.014 | C (6) | 0.014 | -0.001 | 0.015 | |
| C (7) | 0.01 | -0.01 | 0.02 | C (7) | 0.004 | 0.007 | -0.003 | |
| N (9) | 0.015 | -0.002 | 0.017 | N (9) | 0.004 | 0 | 0.004 | |
| C (10) | 0.024 | -0.001 | 0.025 | C (10) | 0.023 | 0.008 | 0.015 | |
| C (11) | 0.012 | 0.004 | 0.008 | C (11) | 0.004 | 0.006 | -0.002 | |
| C (13) | 0.021 | 0.003 | 0.018 | C (13) | 0.019 | 0 | 0.019 | |
| O (14) | 0.01 | 0.015 | -0.005 | O (14) | 0.014 | 0.004 | 0.01 | |
| O (15) | 0.032 | -0.002 | 0.034 | O (15) | 0.033 | 0.003 | 0.03 | |
| C (16) | -0.002 | 0.011 | -0.013 | C (16) | -0.004 | -0.004 | 0 | |
| O (17) | -0.002 | 0.089 | -0.091 | O (17) | -0.001 | -0.002 | 0.001 | |
| O (18) | -0.003 | 0.065 | -0.068 | O (18) | 0.007 | -0.009 | 0.016 | |
| C (19) | 0.005 | 0.041 | -0.036 | C (19) | 0.014 | 0 | 0.014 | |
| O (22) | 0.001 | 0.061 | -0.06 | O (22) | 0.01 | 0.005 | 0.005 | |
| O (23) | 0.005 | 0.038 | -0.033 | O (23) | 0.009 | 0.005 | 0.004 | |
| C (27) | 0.001 | 0.004 | -0.003 | C (27) | -0.007 | -0.004 | -0.003 | |
| C (28) | 0.001 | 0.005 | -0.004 | C (28) | 0 | 0 | 0 | |
| C (34) | -0.001 | 0.01 | -0.011 | C (34) | -0.007 | -0.002 | -0.005 | |
| C (35) | 0.002 | 0.028 | -0.026 | C (35) | 0 | -0.002 | 0.002 | |
| C (36) | 0.001 | 0.022 | -0.021 | C (36) | 0.004 | -0.001 | 0.005 | |
| C (38) | 0.019 | 0.009 | 0.01 | C (38) | 0.013 | 0.001 | 0.012 | |
| C (39) | 0.01 | -0.001 | 0.011 | C (39) | -0.003 | 0.023 | -0.026 | |
| C (40) | 0.019 | 0.001 | 0.018 | C (40) | 0.004 | 0.014 | -0.01 | |
| C (41) | 0.022 | 0.002 | 0.02 | C (41) | 0.005 | 0.012 | -0.007 | |
| C (42) | 0.019 | 0 | 0.019 | C (42) | 0.003 | 0.013 | -0.01 | |
| C (43) | 0.022 | 0.003 | 0.019 | C (43) | 0.003 | 0.013 | -0.01 | |
| C (45) | 0.037 | 0.005 | 0.032 | C (45) | 0.012 | 0.027 | -0.015 | |
| C (46) | 0.003 | -0.003 | 0.006 | C (46) | -0.004 | 0.029 | -0.033 | |
| C (47) | 0.006 | 0.004 | 0.002 | C (47) | 0.004 | 0.014 | -0.01 | |
| C (48) | 0.01 | -0.001 | 0.011 | C (48) | 0.003 | 0.016 | -0.013 | |
| C (49) | 0.009 | 0.008 | 0.001 | C (49) | 0.002 | 0.015 | -0.013 | |
| C (50) | 0.012 | 0.004 | 0.008 | C (50) | 0.003 | 0.015 | -0.012 | |
| C (51) | 0.018 | 0.01 | 0.008 | C (51) | 0.008 | 0.032 | -0.024 | |
| Compound | Donor | Acceptor | E(2)a(kcal/mol) | Occupancy |
|
Q-22 |
πC3–O13 | π*C25–O11 | 23.52 | 1.77 |
| πC4–O15 | π*C26–O12 | 25.13 | 1.65 | |
| πC5–O14 | π*C5–O14 | 33.52 | 1.67 | |
| πC10–O11 | π*C4–O15 | 35.13 | 1.66 | |
| πC9–O12 | π*C9–O20 | 44.52 | 1.68 | |
| πC1–O20 | π*C1–O28 | 36.13 | 1.75 | |
| πC25–O28 | π*C27–O13 | 55.05 | 1.55 | |
| LP (1) N22 | π*Cl51–N24 | 26.74 | 1.87 | |
| LP (1) N24 | π*C16–N22 | 65.21 | 1.45 | |
| πC6–N22 | π*C24–N24 | 36.58 | 1.82 | |
| πC26–N24 | π*O14–C6 | 16.47 | 1.94 | |
| LP (1) O11 | π*O23–O15 | 31.04 | 1.84 | |
| LP (1) O12 | π*C13–O26 | 59.83 | 1.75 | |
| LP (1) O13 | π*C32–O15 | 18.81 | 1.88 | |
| LP (1) O14 | π*C10–O24 | 60.05 | 1.70 | |
| LP (1) O15 | π*O14–N22 | 26.74 | 1.57 | |
| LP (3) Cl51 | π*Cl51–H50 | 45.21 | 1.35 | |
| LP (2) O20 | π*C10–C7 | 36.58 | 1.82 | |
| LP (2) O28 | π*O15–C6 | 26.47 | 1.94 |
| Structures | Q-22 | Q-22-H+ |
| Total energy | 157.26 | 360.54 |
| Adsorption energy | -543.38 | -269.28 |
| Rigid adsorption energy | -452.06 | -8.14 |
| Deformation energy | -121.32 | -253.14 |
| dEad/dNi | -543.38 | -269.28 |
| Temperature (K) | Kads (L mol-1) | (1/KadsL mol-1) (intercept) | R2 |
| 293 | 84.3 | 1.88 | 0.999 |
| 303 | 74.1 | 1.93 | 0.999 |
| 313 | 50.4 | 1.95 | 0.999 |
| 323 | 41.4 | 2.02 | 0.996 |
| Temp, (K) | Langmuir isotherm | ||
|
ΔGºads. (kJ mol-1) |
ΔHºads. (kJ mol-1) |
ΔSºads. (J mol-1 K-1) |
|
| 293 | -37.4 | - 8.32 | 99.3 |
| 303 | -38.5 | 99.2 | |
| 313 | -38.9 | 97.5 | |
| 323 | -39.8 | 98.4 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




