Submitted:
26 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
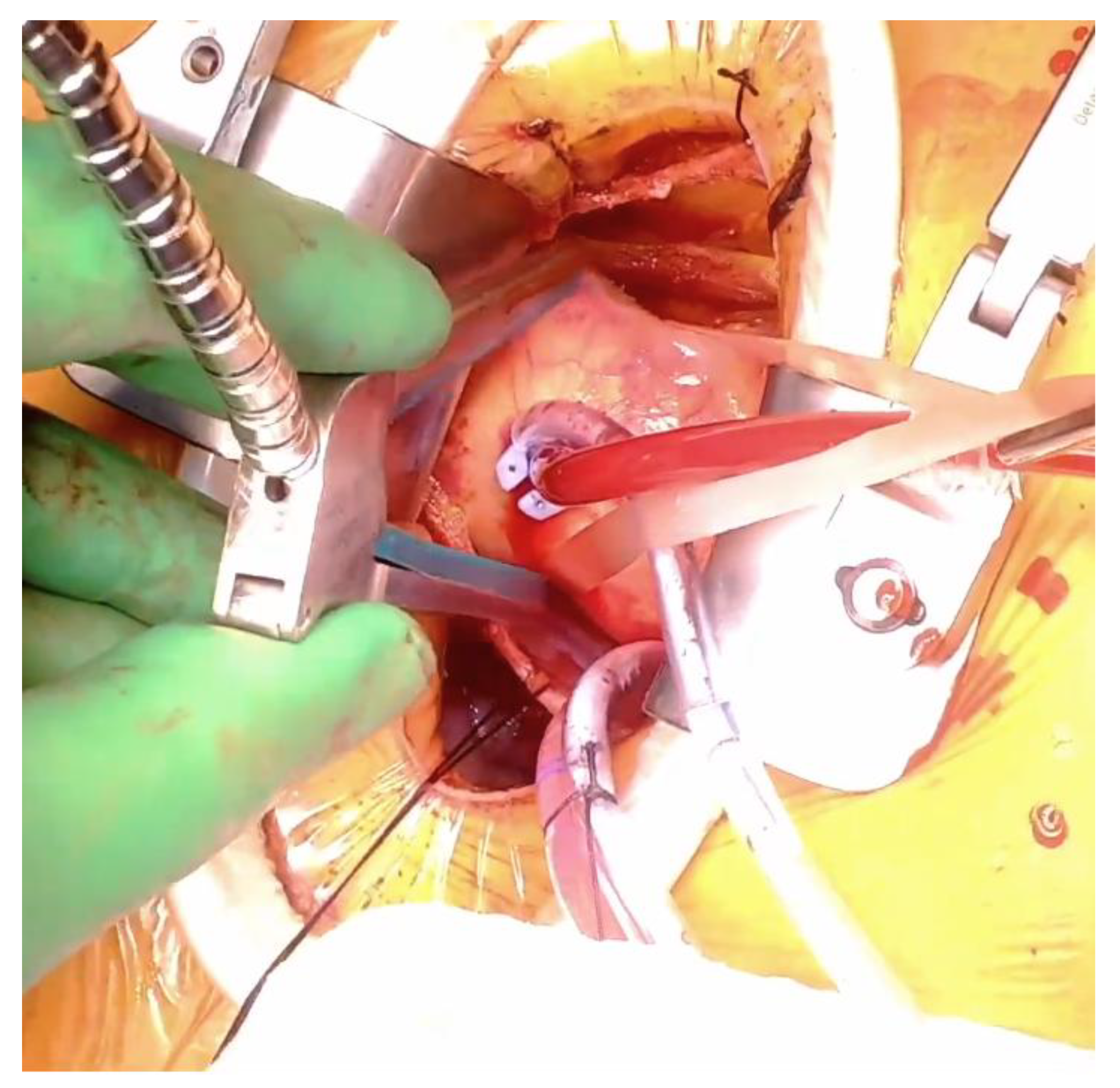
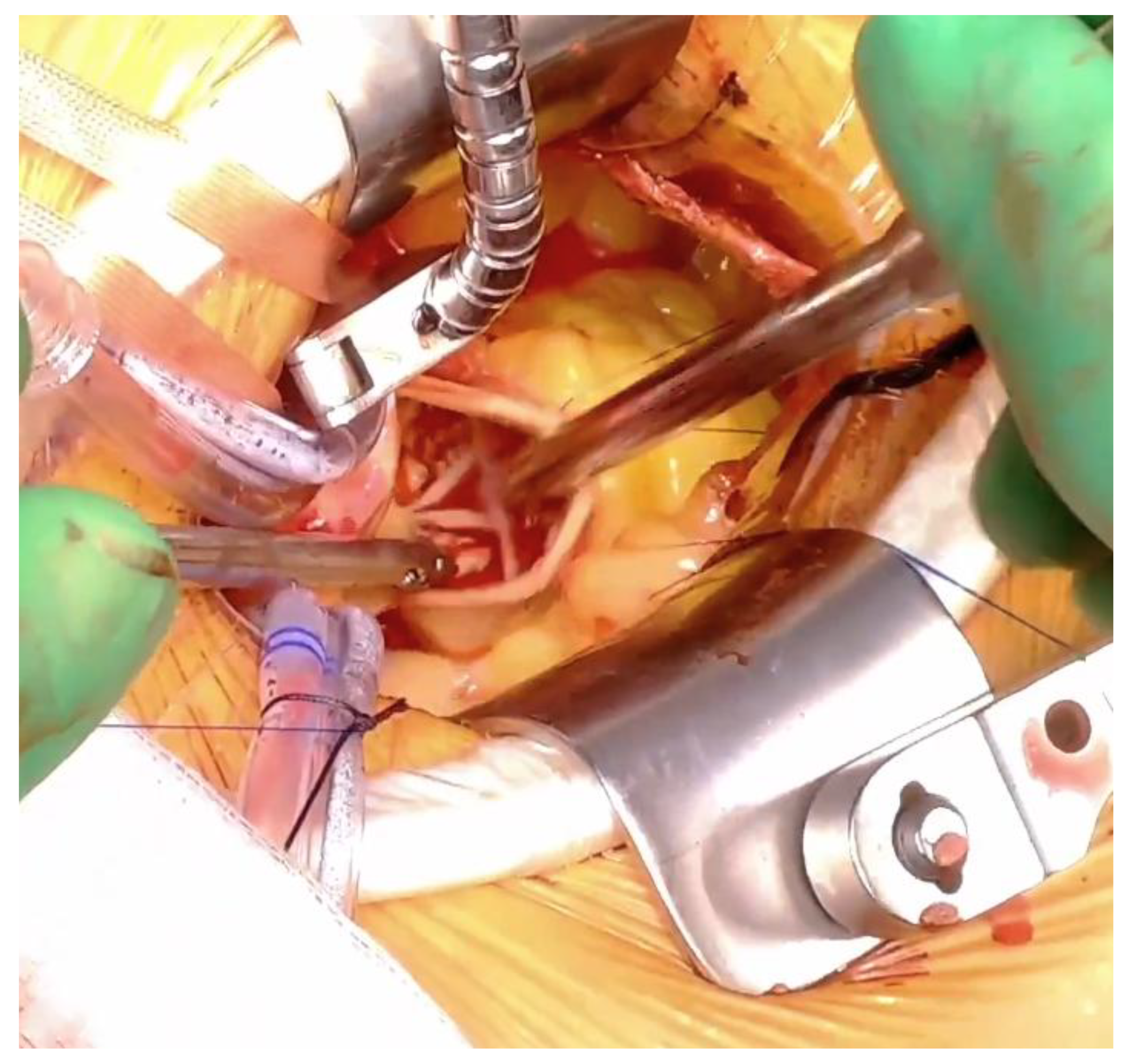
Operative Technique
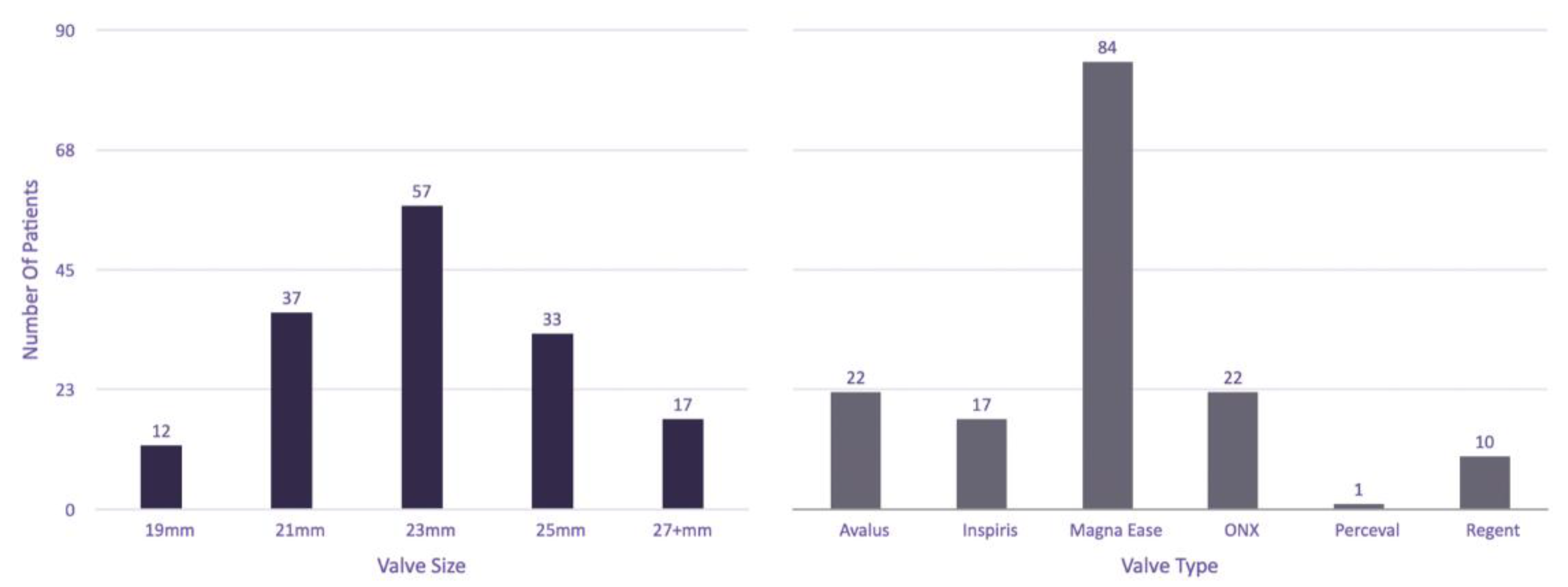
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AVR | Aortic valve replacement |
| RAT-AVR | Right anterior thoracotomy aortic valve replacement |
| CT | Computed tomography |
References
- Shehada, S.-E.; Elhmidi, Y.; Mourad, F.; Wendt, D.; El Gabry, M.; Benedik, J.; Thielmann, M.; Jakob, H. Minimal access versus conventional aortic valve replacement: a meta-analysis of propensity-matched studies. Interact Cardiovasc Thorac Surg 2017, 25, 624–632. [Google Scholar] [CrossRef] [PubMed]
- Shen, H.; Li, D.; Cheng, N.; Li, L.; Dong, S.; Shen, H.; Zhang, L.; Jiang, S. Comparative efficacy of totally thoracoscopic, mini-thoracotomy, and mini-sternotomy approaches in aortic valve replacement. Sci Rep 2024, 14, 17625. [Google Scholar] [CrossRef] [PubMed]
- Jahangiri, M.; Bilkhu, R.; Embleton-Thirsk, A.; Dehbi, H.-M.; Mani, K.; Anderson, J.; Avlonitis, V.; Baghai, M.; Birdi, I.; Booth, K.; et al. Surgical aortic valve replacement in the era of transcatheter aortic valve implantation: a review of the UK national database. BMJ Open 2021, 11, e046491. [Google Scholar] [CrossRef] [PubMed]
- Praz, F.; Borger, M.A.; Lanz, J.; Marin-Cuartas, M.; Abreu, A.; Adamo, M.; Ajmone Marsan, N.; Barili, F.; Bonaros, N.; Cosyns, B.; et al. 2025 ESC/EACTS guidelines for the management of valvular heart disease. Eur Heart J 2025, 46, 4635–4736. [Google Scholar] [CrossRef] [PubMed]
- Glauber, M.; Gilmanov, D.; Farneti, P.A.; Kallushi, E.; Miceli, A.; Chiaramonti, F.; Murzi, M.; Solinas, M. Right anterior minithoracotomy for aortic valve replacement: 10-year experience of a single center. J Thorac Cardiovasc Surg 2015, 150, 548–556.e2. [Google Scholar] [CrossRef] [PubMed]
- Gilmanov, D.; Miceli, A.; Ferrarini, M.; Farneti, P.; Murzi, M.; Solinas, M.; Glauber, M. Aortic valve replacement through right anterior minithoracotomy: can sutureless technology improve clinical outcomes? Ann Thorac Surg 2014, 98, 1585–1592. [Google Scholar] [CrossRef] [PubMed]
- Fatehi Hassanabad, A.; King, M.A.; Karolak, W.; Dokollari, A.; Castejon, A.; de Waard, D.; Smith, H.N.; Holloway, D.D.; Adams, C.; Kent, W.D.T. Right anterior mini thoracotomy approach for aortic valve replacement. Innovations (Phila) 2024, 19, 494–508. [Google Scholar] [CrossRef] [PubMed]
- Martella, A.; Kernis, S.; Curiale, S.; Ndubizu, U.; Cichelli, J. Right thoracotomy minimally invasive AVR: use of preoperative CT scan to plan incision. J Cardiothorac Surg 2015, 10, A28. [Google Scholar] [CrossRef]
- Masuda, T.; Nakamura, Y.; Ito, Y.; Kuroda, M.; Nishijima, S.; Okuzono, Y.; Hirano, T.; Hori, T. The learning curve of minimally invasive aortic valve replacement for aortic valve stenosis. Gen Thorac Cardiovasc Surg 2020, 68, 565–570. [Google Scholar] [CrossRef] [PubMed]
- Tran, L.; Williams-Spence, J.; Shardey, G.C.; Smith, J.A.; Reid, C.M. The Australian and New Zealand Society of Cardiac and Thoracic Surgeons Database Program – two decades of quality assurance data. Heart Lung Circ 2019, 28, 1459–1462. [Google Scholar] [CrossRef] [PubMed]
- Ramchandani, M.; Al Jabbari, O.; Abu Saleh, W.K.; Ramlawi, B. Cannulation strategies and pitfalls in minimally invasive cardiac surgery. Methodist Debakey Cardiovasc J 2016, 12, 10–13. [Google Scholar] [CrossRef] [PubMed]
- Lamelas, J.; Williams, R.F.; Mawad, M.; LaPietra, A. Complications associated with femoral cannulation during minimally invasive cardiac surgery. Ann Thorac Surg 2017, 103, 1927–1932. [Google Scholar] [CrossRef] [PubMed]
- Mundy, J; Williams-Spence, J; Tran, L; Solman, N; McLaren, J; Marrow, N; Ong, J; Reid, C. The Australian and New Zealand Society of Cardiac and Thoracic Surgeons’ Cardiac Surgery Database Program Annual Report 2023. Report No. 17. September 2024. [Google Scholar]

| Demographics | |
|---|---|
| Age | 70.0 [62.0 - 75.0] |
| Male Gender | 112/156 (72%) |
| BMI | 28.9 [25.4 - 31.8] |
| Smoking History | 68/156 (44%) |
| Significant Comorbidities | |
| Lung Disease (Asthma or COPD) | 21/156 (13%) |
| Diabetes | 30/156 (19%) |
| Previous Stroke or TIA | 10/156 (6%) |
| Peripheral Vascular Disease | 5/156 (3%) |
| Current Infective Endocarditis | 1/156 (1%) |
| Immunosuppressive therapy | 6/156 (4%) |
| Permanent Pacemaker | 5/156 (3%) |
| Haemodynamics | N = 156 |
|---|---|
| Aortic Valve Primary Pathology | |
| Severe AS | 127/156 (81%) |
| Severe AR | 29/156 (19%) |
| Pathogenesis | |
| Idiopathic Calcific | 108/156 (69%) |
| Bicuspid | 45/156 (28%) |
| Failed TAVR | 1/156 (1%) |
| Active IE | 1/156 (1%) |
| Rheumatic | 1/156 (1%) |
| Pre-operative Ejection Fraction | |
| Normal | 118/156 (76%) |
| Mild Reduction | 28/156 (18%) |
| Moderate Reduction | 7/156 (4%) |
| Severe Reduction | 3/156 (2%) |
| Intra-operative Characteristic | N = 156 |
|---|---|
| Cross-Clamp Time | 79.0 [69.0 – 91.5] |
| CPB Time | 109 [98.0 – 129.5] |
| Arterial Cannulation Site | |
| Central | 7/156 (4%) |
| Femoral | 149/156 (96%) |
| Venous Cannulation Site | |
| Central | 2/156 (1%) |
| Femoral | 154/156 (99%) |
| Characteristic | N = 156 |
|---|---|
| Major Outcomes | |
| 30-day mortality | 2/156 (1%) |
| Return to Theatre | 6/156 (4%) |
| Conversion to Sternotomy | 2/156 (1%) |
| Length of Stay | |
| Ventilation Time (Hours) | 7.9 [5.4 - 16.5] |
| ICU Length of Stay (Hours) | 47.8 [28.4 - 72.2] |
| Hospital Length of Stay (Days) | 6.0 [5.0 - 8.0] |
| Bleeding and Transfusion | |
| ICC output first 4 hours (ml) | 130.0 [90.0 - 190.0] |
| Any Blood Transfusion | 27/156 (17%) |
| Complications | |
| New AKI | 4/156 (3%) |
| TIA | 2/156 (1%) |
| Stroke (Permanent) | 0/156 |
| New AF | 38/156 (24%) |
| PPM Insertion | 10/156 (6%) |
| Groin Seroma or Infection | 3/156 (2%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




